The Influence of Ultrafiltration of Citrus limon L. Burm. cv Femminello Comune Juice on Its Chemical Composition and Antioxidant and Hypoglycemic Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Material and Juice Preparation
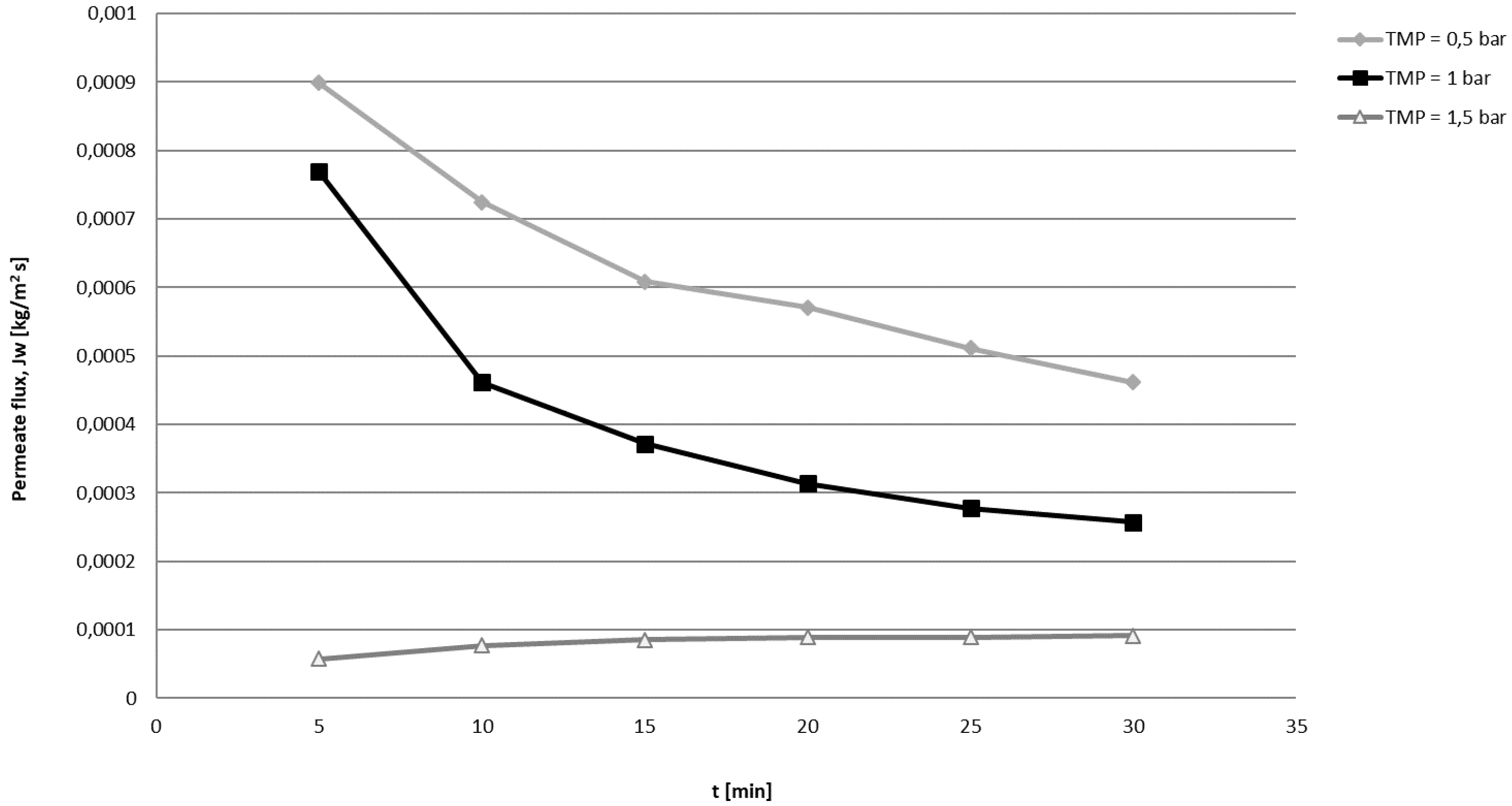
2.3. Ultrafiltration Process
2.4. Physiochemical Characteristics of C. Limon Juice
2.5. Ascorbic Acid Content
2.6. Total Phenols and Flavonoids Content
2.7. HPLC-DAD Analyses
2.8. Antioxidant Effects
2.8.1. DPPH (2,2-diphenyl-1-picrylhydrazyl) Assay
2.8.2. ABTS, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) Assay
2.8.3. Ferric Reducing Ability Power (FRAP) Assay
2.9. RACI Calculation
2.10. In Vitro Hypoglycaemic Activity
2.11. Statistical Analysis
3. Results and Discussion
3.1. Juice Content and Physicochemical Parameters
3.2. Bioactive Compounds
3.3. Antioxidant Activity
3.4. Carbohydrate Hydrolysing Enzymes Inhibitory Activities
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mulder, M. Basic Principles of Membrane Technology; Kluwer Academic Publishers: Norwell, MA, USA, 1991. [Google Scholar]
- Tundis, R.; Loizzo, M.R.; Bonesi, M.; Sicari, V.; Ursino, C.; Manfredi, I.; Conidi, C.; Figoli, A.; Cassano, A. Concentration of bioactive compounds from elderberry (Sambucus nigra L.) juice by nanofiltration membranes. Plant Foods Hum. Nutr. 2018, 73, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Girard, B.; Fukumoto, L.R. Apple juice clarification using microfiltration and ultrafiltration polymeric membranes. LWT 1999, 32, 290–298. [Google Scholar] [CrossRef]
- Wang, B.J.; Wei, T.C.; Yu, Z.R. Effect of operating temperature on component distribution of West Indian cherry juice in a microfiltration system. LWT 2005, 38, 683–689. [Google Scholar] [CrossRef]
- Razi, B.; Aroujalian, A.; Raisi, A.; Fathizadeh, M. Clarification of tomato juice by cross-flow microfiltration. Int. J. Food Sci. Technol. 2011, 46, 138–145. [Google Scholar] [CrossRef]
- Cassano, A.; Drioli, E.; Galaverna, G.; Marchelli, R.; Disilvestro, G.; Cagnasso, P. Clarification and concentration of citrus and carrot juices by integrated membrane processes. J. Food Eng. 2003, 57, 153–163. [Google Scholar] [CrossRef]
- Tundis, R.; Loizzo, M.R.; Bonesi, M.; Menichini, F.; Mastellone, V.; Colica, C.; Menichini, F. Comparative study on the antioxidant capacity and cholinesterase inhibitory activity of Citrus aurantifolia Swingle, C. aurantium L., and C. bergamia Risso and Poit. peel essential oils. J. Food Sci. 2012, 77, H40–H46. [Google Scholar] [CrossRef]
- Tundis, R.; Loizzo, M.R.; Menichini, F. An overview on chemical aspects and potential health benefits of limonoids and their derivatives. Crit. Rev. Food Sci. Nutr. 2014, 54, 225–250. [Google Scholar] [CrossRef] [PubMed]
- Loizzo, M.R.; Tundis, R.; Bonesi, M.; Menichini, F.; De Luca, D.; Colica, C.; Menichini, F. Evaluation of Citrus aurantifolia peel and leaves extracts for their chemical composition, antioxidant and anti-cholinesterase activities. J. Sci. Food Agric. 2012, 92, 2960–2967. [Google Scholar] [CrossRef]
- Okwu, D.E. Citrus fruits: A rich source of phytochemicals and their roles in human health. Int. J. Chem. Sci. 2008, 6, 451–471. [Google Scholar]
- Del Caro, A.; Piga, A.; Vacca, V.; Agabbio, M. Changes of flavonoids, vitamin C and antioxidant capacity in minimally processed citrus segments and juices during storage. Food Chem. 2004, 84, 99–105. [Google Scholar] [CrossRef]
- Wright, E.; Scism-Bacon, J.L.; Glass, L.C. Oxidative stress in type 2 diabetes: The role of fasting and postprandial glycaemia. Int. J. Clin. Pract. 2006, 60, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Selim, F.; Wael, A.; Keith, E.J. Diabetes-induced reactive oxygen species: Mechanism of their generation and role in renal injury. J. Diabetes Res. 2017, 2017, 8379327. [Google Scholar]
- Pareek, S.; Paliwal, R.; Mukherjee, S. Effect of juice extraction methods and processing temperature-time on juice quality of Nagpur mandarin (Citrus reticulata Blanco) during storage. J. Food Sci. Technol. 2010, 48, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis; Association of Official Analytical Chemists: Washington, DC, USA, 2000. [Google Scholar]
- Gao, X.; Ohlander, M.; Jeppsson, N.; Björk, L.; Trajkovski, V. Changes in antioxidant effects and their relationship to phytonutrients in fruits of Sea buckthorn (Hippophae rhamnoides L.) during maturation. J. Agr. Food Chem. 2000, 48, 1485–1490. [Google Scholar] [CrossRef]
- Yoo, K.M.; Lee, C.H.; Lee, H.; Moon, B.K.; Lee, C.Y. Relative antioxidant and cytoprotective activities of common herbs. Food Chem. 2008, 106, 929–936. [Google Scholar] [CrossRef]
- Loizzo, M.R.; Leporini, M.; Sicari, V.; Falco, T.; Pellicanò, T.; Tundis, R. Investigating the in vitro hypoglycaemic and antioxidant properties of Citrus × clementina Hort. juice. Eur. Food Res. Technol. 2017, 244, 523–534. [Google Scholar] [CrossRef]
- Loizzo, M.R.; Tundis, R.; Bonesi, M.; Menichini, F.; Mastellone, V.; Avallone, L.; Menichini, F. Radical scavenging, antioxidant and metal chelating activities of Annona cherimola Mill. (cherimoya) peel and pulp in relation to their total phenolic and total flavonoid contents. J. Food Comp. Anal. 2012, 25, 179–184. [Google Scholar] [CrossRef]
- Al-Mouei, R.; Wafaa, C. Physiochemical juice characteristics of various Citrus species in Syria. J. Plant Nutr. Soil Sci. 2014, 3, 1083–1095. [Google Scholar] [CrossRef]
- Espamer, L.; Pagliero, C.; Ochoab, A.; Marchese, J. Clarification of lemon juice using membrane process. Desalination 2006, 200, 565–567. [Google Scholar] [CrossRef]
- Chornomaz, P.M.; Pagliero, C.; Marchese, J.; Ochoa, N.A. Impact of structural and textural membrane properties on lemon juice clarification. Food Bioprod. Process. 2013, 91, 67–73. [Google Scholar] [CrossRef]
- Cassano, A.; Marchio, M.; Drioli, E. Clarification of blood orange juice by ultrafiltration: Analyses of operating parameters, membrane fouling and juice quality. Desalination 2007, 212, 15–27. [Google Scholar] [CrossRef]
- Pagliero, C.; Ochoa, N.A.; Marchese, J. Orange juice clarification by microfiltration: Effect of operational variables on membrane fouling. Latin Am. Appl. Res. 2011, 41, 279–284. [Google Scholar]
- Gattuso, G.; Barreca, D.; Gargiulli, C.; Leuzzi, U.; Caristi, C. Flavonoid composition of Citrus juices. Molecules 2007, 12, 1641. [Google Scholar] [CrossRef]
- Hajimahmoodi, M.; Moghaddam, G.; Mohsen, M.; Sadeghi, S.; Oveisi, N.; Reza, M.; Behrooz, J. Total antioxidant activity, and hesperidin, diosmin, eriocitrin and quercetin contents of various lemon juices. Trop. J. Pharm. Res. 2014, 13, 951–956. [Google Scholar] [CrossRef]
- Peterson, J.J.; Beecher, G.R.; Bhagwat, S.A.; Dwyer, J.T.; Gebhardt, S.E.; Haytowitz, D.B.; Holden, J.M. Flavanones in grapefruit, lemons, and limes: A compilation and review of the data from the analytical literature. J. Food Compost. Anal. 2006, 19, 74–80. [Google Scholar] [CrossRef]
- Fejzić, A.; Ćavar, S. Phenolic compounds and antioxidant activity of some citruses. Bull. Chem. Technol. Bosn. Herzeg. 2014, 42, 1–4. [Google Scholar]
- Antolovich, M.; Prenzler, P.D.; Patsalides, E.S.; Mc Donald, S.; Robards, K. Methods for testing antioxidant activity. Analyst 2002, 127, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Oikeh, E.I.; Omoregie, E.S.; Oviasogie, F.E.; Oriakhi, K. Phytochemical, antimicrobial, and antioxidant activities of different citrus juice concentrates. Food Sci. Nutr. 2015, 4, 103–109. [Google Scholar] [CrossRef]
- Xi, W.; Lu, J.; Qun, J.; Jiao, B. Characterization of phenolic profile and antioxidant capacity of different fruit part from lemon (Citrus limon Burm.) cultivars. J. Food Sci. Technol. 2017, 54, 1108–1118. [Google Scholar] [CrossRef]
- Matsui, T.; Ogunwande, I.A.; Abesundara, K.J.M.; Matsumoto, K. Anti-hyperglycemic potential of natural products. Med. Chem. 2006, 6, 349–356. [Google Scholar] [CrossRef]
- Tundis, R.; Bonesi, M.; Sicari, V.; Pellicanò, T.M.; Tenuta, M.C.; Leporini, M.; Menichini, F.; Loizzo, M.R. Poncirus trifoliata (L.) Raf.: Chemical composition, antioxidant properties and hypoglycaemic activity via the inhibition of α-amylase and α-glucosidase enzymes. J. Funct. Foods 2016, 25, 477–485. [Google Scholar] [CrossRef]
- Riaz, A.; Alam Khan, R.; Ahmed, M. Glycemic response of Citrus limon, pomegranate and their combinations in alloxan-induced diabetic rats. Aust. J. Basic Appl. Sci. 2013, 7, 215–219. [Google Scholar]
- Mollace, V.; Sacco, I.; Janda, E.; Malara, C.; Ventrice, D.; Colica, C.; Visalli, V.; Muscoli, S.; Ragusa, S.; Muscoli, C.; et al. Hypolipemic and hypoglycaemic activity of bergamot polyphenols: From animal models to human studies. Fitoterapia 2011, 82, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Hamed, W.M.A.; Abid, K.Y.; Al-Amin, S.A.U. Hypoglycemic and hypolipidemic effects of grapefruit juice in diabetic rats. Tikrit J. Pure Sci. 2008, 13, 129–131. [Google Scholar]
- Mallick, N.; Khan, R. Effect of Citrus paradisi and Citrus sinensis on glycemic control in rats. Afr. J. Pharm. Pharmacol. 2015, 9, 60–64. [Google Scholar]
- Tadera, K.; Minami, Y.; Takamatsu, K.; Matsuoka, T. Inhibition of alpha-glucosidase and alpha-amylase by flavonoids. J. Nutr. Sci. Vitaminol. 2010, 52, 149–153. [Google Scholar] [CrossRef]
- Kim, Y.; Keogh, J.B.; Clifton, P.M. Polyphenols and glycemic control. Nutrients 2016, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Sahnoun, M.; Trabelsi, S.; Bejar, S. Citrus flavonoids collectively dominate the α-amylase and α-glucosidase inhibitions. Biologia 2017, 72, 764–773. [Google Scholar] [CrossRef]
- Jia, S.; Hu, Y.; Zhang, W.; Zhao, X.; Chen, Y.; Sun, C.; Li, X.; Chen, K. Hypoglycemic and hypolipidemic effects of neohesperidin derived from Citrus aurantium L. in diabetic KKA(y) mice. Food Funct. 2015, 6, 878–888. [Google Scholar] [CrossRef]
- Oboh, G.; Ademosun, A.O.; Ayeni, P.O.; Omojokun, O.S.; Bello, F. Comparative effect of quercetin and rutin on α-amylase, α-glucosidase, and some pro-oxidant-induced lipid peroxidation in rat pancreas. Comp. Clin. Pathol. 2015, 24, 1103–1110. [Google Scholar] [CrossRef]
| Juice | P (bar) | Juice (%) | pH % Inhibition | TSS (°Brix) µM Fe(II) | % Citric Acid | Ascorbic Acid Content (mg/100 mL) |
|---|---|---|---|---|---|---|
| Untreated | 0 | 45.2 ± 2.6 | 2.5 ± 0.01 c | 8.5 ± 2.1 a | 6.3 ± 1.8 a | 30.8 ± 2.7 a |
| J1 | 0.5 | - | 2.6 ± 0.02 b | 7.8 ± 2.4 b | 5.6 ± 2.0 c | 27.7 ± 3.1 d |
| J2 | 1 | - | 2.7 ± 0.03 a | 3.5 ± 2.2 c | 5.7 ± 2.1 b | 28.6 ± 2.5 c |
| J3 | 1.5 | - | 2.6 ± 0.01 b | 2.7 ± 2.4 d | 5.7 ± 2.2 b | 29.4 ± 3.0 b |
| Juice | TPC a | TFC b | Rutin c | Eriocitrin c | Hesperidin c | Neohesperidin c |
|---|---|---|---|---|---|---|
| Untreated | 151.7 ± 3.4 a | 30.8 ± 2.5 a | 2.9 ± 0.9 a | 16.7 ± 1.9 a | 14.1 ± 1.8 a | 6.0 ± 0.3 a |
| J1 | 102.0 ± 1.3 d | 19.7 ± 3.5 c | 2.2 ± 0.9 b | 14.8 ± 1.4 b | 12.6 ± 1.3 b | 5.0 ± 0.7 b |
| J2 | 126.4 ± 4.9 c | 22.4 ± 3.0 b | 2.3 ± 0.8 b | 15.2 ± 1.3 b | 12.1 ± 1.5 b | 5.6 ± 1.2 b |
| J3 | 148.4 ± 3.1 b | 31.9 ± 4.1 a | 2.8 ± 0.7 a | 15.8 ± 1.5 ba | 13.5 ± 1.3 ba | 5.7 ± 1.0 ba |
| Juice | DPPH Test (IC50 g/mL) | ABTS Test (IC50 g/mL) | FRAP Test a (M Fe(II)/g) | RACI Values |
|---|---|---|---|---|
| (IC50 μg/mL) | (IC50 μg/mL) | (μM Fe(II)/g) | ||
| Untreated | 40.3 ± 1.0 * | 46.5 ± 1.2 * | 49.7 ± 2.8 * | 0.06 |
| J1 | 42.1 ± 1.3 * | 51.3 ± 1.6 * | 52.7 ± 2.0 * | 0.68 |
| J2 | 41.2 ± 4.0 * | 39.7 ± 1.1 * | 49.8 ± 1.6 * | −0.09 |
| J3 | 35.2 ± 1.0 * | 37.3 ± 1.4 * | 48.9 ± 1.5 * | −0.66 |
| Ascorbic acid | 5.0 ± 0.8 | 1.7 ± 0.9 | ||
| BHT | 63.2 ± 4.3 |
| Juice | α-Amylase a (IC50 µL/mL) | α-Glucosidase a % Inhibition |
|---|---|---|
| (IC50 μg/mL) | (IC50 μg/mL) | |
| Untreated | 40.3 ± 1.0 * | 46.5 ± 1.2 * |
| J1 | 42.1 ± 1.3 * | 51.3 ± 1.6 * |
| J2 | 41.2 ± 4.0 * | 49.7 ± 1.1 * |
| J3 | 31.1 ± 1.0 * | 35.3 ± 1.4 * |
| Acarbose | 50.0 ± 1.4 | 35.5 ± 1.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loizzo, M.R.; Sicari, V.; Tundis, R.; Leporini, M.; Falco, T.; Calabrò, V. The Influence of Ultrafiltration of Citrus limon L. Burm. cv Femminello Comune Juice on Its Chemical Composition and Antioxidant and Hypoglycemic Properties. Antioxidants 2019, 8, 23. https://doi.org/10.3390/antiox8010023
Loizzo MR, Sicari V, Tundis R, Leporini M, Falco T, Calabrò V. The Influence of Ultrafiltration of Citrus limon L. Burm. cv Femminello Comune Juice on Its Chemical Composition and Antioxidant and Hypoglycemic Properties. Antioxidants. 2019; 8(1):23. https://doi.org/10.3390/antiox8010023
Chicago/Turabian StyleLoizzo, Monica Rosa, Vincenzo Sicari, Rosa Tundis, Mariarosaria Leporini, Tiziana Falco, and Vincenza Calabrò. 2019. "The Influence of Ultrafiltration of Citrus limon L. Burm. cv Femminello Comune Juice on Its Chemical Composition and Antioxidant and Hypoglycemic Properties" Antioxidants 8, no. 1: 23. https://doi.org/10.3390/antiox8010023
APA StyleLoizzo, M. R., Sicari, V., Tundis, R., Leporini, M., Falco, T., & Calabrò, V. (2019). The Influence of Ultrafiltration of Citrus limon L. Burm. cv Femminello Comune Juice on Its Chemical Composition and Antioxidant and Hypoglycemic Properties. Antioxidants, 8(1), 23. https://doi.org/10.3390/antiox8010023








