Use of Olive Oil Industrial By-Product for Pasta Enrichment
Abstract
:1. Introduction
2. Material and Methods
2.1. Raw Materials
2.2. Chemicals
2.3. Olive Paste Flour Preparation
2.4. Spaghetti Preparation
2.5. Sensory Analysis
2.6. Extraction and Analysis of Triterpenic Acids
2.7. Tocochromanols and Carotenoids Extraction and Analysis
2.8. Extraction and Fatty Acids Analysis
2.9. Extraction and Analysis of Free and Total Phenolic Compounds
2.10. Pasta Characteristics Determination
2.11. Statistical Analysis
3. Results and Discussion
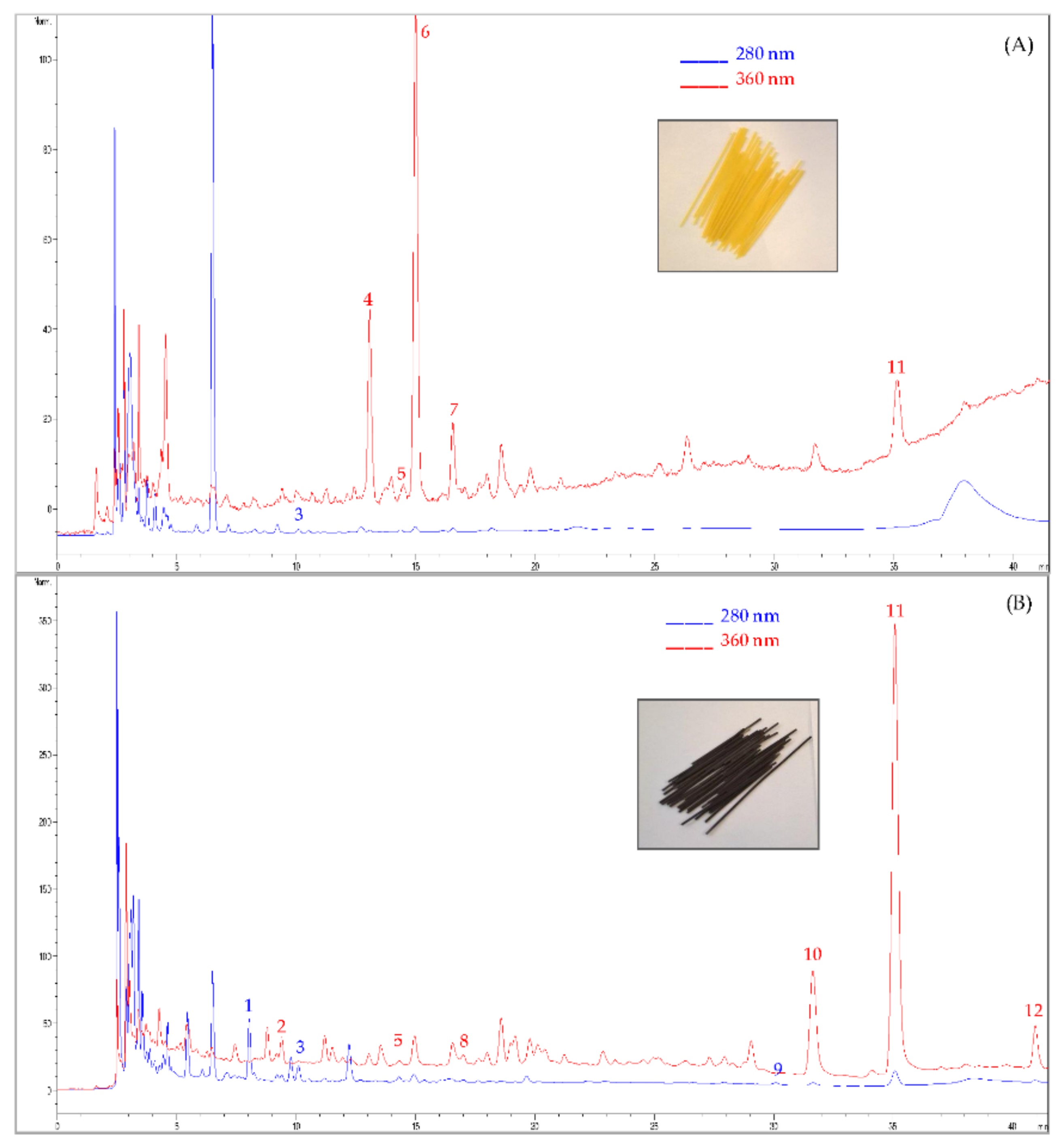
3.1. Step 1: Biochemical Composition of Semolina and OPF
3.2. Step 2: Sensory Quality of Spaghetti Enriched with OPF
3.3. Step 3: Effect of Transglutaminase Addition on Spaghetti Enriched with OPF
3.3.1. Sensory Quality
3.3.2. Biochemical Composition
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Laufenberg, G.; Kunz, B.; Nystroem, M. Transformation of vegetable waste into value added products: (A) the upgrading concept; (B) practical implementations. Bioresour. Technol. 2003, 87, 167–198. [Google Scholar] [CrossRef]
- D’Antuono, I.; Kontogianni, V.G.; Kotsiou, K.; Linsalata, V.; Logrieco, A.F.; Tasioula-Margari, M.; Cardinali, A. Polyphenolic characterization of olive mill wastewaters, coming from Italian and Greek olive cultivars, after membrane technology. Food Res. Int. 2014, 65, 301–310. [Google Scholar] [CrossRef]
- Cardinali, A.; Pati, S.; Minervini, F.; D’Antuono, I.; Linsalata, V.; Lattanzio, V. Verbascoside, isoverbascoside, and their derivatives recovered from olive mill waste water as possible food antioxidants. J. Agric. Food Chem. 2012, 60, 1822–1829. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, G. Lipids and phenols in table olives. Eur. J. Lipid Sci. Technol. 2003, 105, 229–242. [Google Scholar] [CrossRef]
- Potter, R.; Stojceska, V.; Plunkett, A. The use of fruit powders in extruded snacks suitable for Children’s diets. LWT Food Sci. Technol. 2013, 51, 537–544. [Google Scholar] [CrossRef]
- Duranti, M. Grain legume proteins and nutraceutical properties. Fitoterapia 2006, 77, 67–82. [Google Scholar] [CrossRef] [PubMed]
- Dexter, J.E.; Matson, R.R. Changes in spaghetti proteins solubility during cooking. Cereal Chem. 1979, 56, 394–397. [Google Scholar]
- Brennan, C.S.; Tudorica, C.M. Evaluation of potential mechanisms by which dietary fibre additions reduce the predicted glycaemic index of fresh pastas. Int. J. Food Sci. Technol. 2008, 43, 2151–2162. [Google Scholar] [CrossRef]
- Lodi, A.; Tiziani, S.; Vodovatz, Y. Molecular changes in soy and wheat breads during storage as probed by nuclear magnetic resonance (NMR). J. Agric. Food Chem. 2007, 55, 5850–5857. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, K.; Nio, N.; Kikuchi, Y. Properties and applications of microbial transglutaminase. Appl. Microbiol. Biot. 2004, 64, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Kuraishi, C.; Yamazaki, K.; Susa, Y. Transglutaminase: Its utilization in the food industry. Food Rev. Int. 2001, 17, 221–246. [Google Scholar] [CrossRef]
- Padalino, L.; Mastromatteo, M.; Lecce, L.; Spinelli, S.; Contò, F.; Del Nobile, M.A. Chemical composition, sensory and cooking quality evaluation of durum wheat spaghetti enriched with pea flour. Int. J. Food Sci. Technol. 2013, 49, 1544–1556. [Google Scholar] [CrossRef]
- ISO 11036/7304-2. Alimentary Pasta Produced from Durum Wheat Semolina-Estimation of Cooking Quality by Sensory Analysis—Part 2: Routine Method; International Organization for Standardization: Geneva, Switzerland, 2008. [Google Scholar]
- Romero, C.; García, A.; Medina, E.; Ruíz-Méndez, M.V.; de Castro, A.; Brenes, M. Triterpenic acids in table olives. Food Chem. 2010, 118, 670–674. [Google Scholar] [CrossRef]
- Lozano-Mena, G.; Juan, M.E.; García-Granados, A.; Planas, J.M. Determination of Maslinic Acid, a Pentacyclic Triterpene from Olives, in Rat Plasma by High-Performance Liquid Chromatography. J. Agric. Food Chem. 2012, 60, 10220–10225. [Google Scholar] [CrossRef] [PubMed]
- Panfili, G.; Fratianni, A.; Irano, M. Normal phase high performance liquid chromatography method for the determination of tocopherols and tocotrienols in cereals. J. Agric. Food Chem. 2003, 51, 3940–3944. [Google Scholar] [CrossRef] [PubMed]
- Fraser, P.D.; Pinto, M.E.S.; Holloway, D.E.; Bramley, P.M. Application of high-performance liquid chromatography with photodiode array detection to the metabolic profiling of plant isoprenoids. Plant J. 2000, 24, 551–558. [Google Scholar] [CrossRef] [PubMed]
- American Association for Clinical Chemistry (AACC). Approved Methods of the American Association of Cereal Chemists, 10th ed.; Methods 30–10 and 66–41; AACC: St. Paul, MN, USA, 2003. [Google Scholar]
- Durante, M.; Lenucci, M.S.; Marrese, P.P.; Rizzi, V.; De Caroli, M.; Piro, G.; Fini, P.; Russo, G.L.; Mita, G. α-Cyclodextrin encapsulation of supercritical CO2 extracted oleoresins from different plant matrices: A stability study. Food Chem. 2016, 199, 684–693. [Google Scholar] [CrossRef] [PubMed]
- Mattila, P.; Pihlava, J.M.; Hellström, J. Contents of phenolic acids, alkyl- and alkenylresorcinols, and avenanthramides in commercial grain products. J. Agric. Food Chem. 2005, 53, 8290–8295. [Google Scholar] [CrossRef] [PubMed]
- American Association for Clinical Chemistry (AACC). Approved Methods of the American Association of Cereal Chemistry; Method 66–50; AACC: St. Paul, MN, USA, 2000. [Google Scholar]
- Laddomada, B.; Durante, M.; Minervini, F.; Garbetta, A.; Cardinali, A.; D’Antuono, I.; Caretto, S.; Blanco, A.; Mita, G. Phytochemical Composition and Anti-Inflammatory Activity of Extracts from the Whole-Meal Flour of Italian Durum Wheat Cultivars. Int. J. Mol. Sci. 2015, 16, 3512–3527. [Google Scholar] [CrossRef] [PubMed]
- Beleggia, R.; Platani, C.; Spano, G.; Monteleone, M.; Cattivelli, L. Metabolic profiling and analysis of volatile composition of durum wheat semolina and pasta. J. Cereal Sci. 2009, 49, 301–309. [Google Scholar] [CrossRef]
- Breinholt, V.; Kumpulainen, J.T.; Salonen, J.T. Desirable versus harmful levels of intake of flavonoids and phenolic acids. Natural antioxidants and anti-carcinogens in nutrition, health and disease. In Proceedings of the Second International Conference on Natural Antioxidants and Anticarcionogens in Nutrition, Health and Disease, Helsinki, Finland, 24–27 June 1998; Royal Society of Chemistry: London, UK, 1999. [Google Scholar]
- Brenes, M.; De Castro, A. Transformation of oleuropein and its hydrolysis products during Spanish-style green olive processing. J. Sci. Food Agric. 1998, 77, 353–358. [Google Scholar] [CrossRef]
- Arranz, S.; Silván, J.M.; Saura-Calixto, F. Nonextractable polyphenols, usually ignored, are the major part of dietary polyphenols: A study on the Spanish diet. Mol. Nutr. Food Res. 2010, 54, 1646–1658. [Google Scholar] [CrossRef] [PubMed]
- Debbouz, A.; Pitz, W.J.; Moore, W.R.; Dappolonia, B.L. Effect of bleaching on durum-wheat and spaghetti quality. Cereal Chem. 1995, 72, 128–131. [Google Scholar]
- Lončarić, A.; Kosović, I.; Jukić, M.; Ugarčić, Ž.; Piližota, V. Effect of apple by-product as a supplement on antioxidant activity and quality parameters of pasta Croat. J. Food Sci. Technol. 2014, 6, 97–103. [Google Scholar]
- Kang, C.S.; Kim, K.H.; Choi, I.D.; Son, J.H.; Park, C.S. Effects of transglutminase on the quality of white salted noodles made from Korean wheat cultivars. Int. Food Res. J. 2014, 21, 195–202. [Google Scholar]
- Wu, J.P.; Corke, H. Quality of dried white salted noodles affected by microbial transglutaminase. J. Sci. Food Agric. 2005, 85, 2587–2594. [Google Scholar] [CrossRef]
- Gerrard, J.A.; Fayle, S.E.; Wilson, A.J.; Newberry, M.P.; Ross, M.; Kavale, S. Dough properties and crumb strength of white pan bread as affected by microbial transglutaminase. J. Food Sci. 1998, 63, 472–475. [Google Scholar] [CrossRef]
- Sözer, N.; Kaya, A. Changes in cooking and textural properties of spaghetti cooked with different levels of salt in the cooking water. J. Texture Stud. 2003, 34, 381–390. [Google Scholar] [CrossRef]
- Kovács, M.I.P.; Fu, B.X.; Woods, S.M.; Khan, K. Thermal stability of wheat gluten protein: Its effect on dough properties and noodle texture. J. Cereal Sci. 2004, 39, 9–19. [Google Scholar] [CrossRef]
- Food and Agriculture Organization. FAO Production Yearbook of 1997; FAO: Rome, Italy, 1997; Volume 51. [Google Scholar]
- Verardo, V.; Gómez-Caravaca, A.M.; Messia, M.C.; Marconi, E.; Caboni, M.F. Development of Functional Spaghetti Enriched in Bioactive Compounds Using Barley Coarse Fraction Obtained by Air Classification. J. Agric. Food Chem. 2011, 59, 9127–9134. [Google Scholar] [CrossRef] [PubMed]
- Sun-Waterhouse, D.; Jin, D.; Waterhouse, G.I.N. Effect of adding elderberry juice concentrate on the quality attributes, polyphenol contents and antioxidant activity of three fibre-enriched pastas. Food Res. Int. 2013, 54, 781–789. [Google Scholar] [CrossRef]
| Olive Paste Flour | Semolina | |||||
|---|---|---|---|---|---|---|
| Tocochromanols (µg/g DW) | ||||||
| β T3 | ND | 11.95 ± 0.71 | ||||
| α T | 107.17 ± 1.78 | ND | ||||
| Carotenoids (µg/g DW) | ||||||
| Lutein | 4.03 ± 0.27 | 5.59 ± 0.26 | ||||
| Zeaxanthin | 0.19 ± 0.001 | 0.25 ± 0.004 | ||||
| α-carotene | 0.45 ± 0.03 | ND | ||||
| β-carotene | 0.69 ± 0.06 | 0.037 ± 0.001 | ||||
| Triterpenic acids (mg/g DW) | ||||||
| Maslinic | 6.76 ± 0.45 | ND | ||||
| Eonolic | 3.65 ± 0.32 | ND | ||||
| Fatty acids (%) | ||||||
| Myristic C14:0 | ND | 0.24 ± 0.01 | ||||
| Palmitic C16:0 | 21.40 ± 2.70 | 43.93 ± 0.63 | ||||
| Palmitoleic C16:1 | 1.81 ± 0.21 | 5.96 ± 0.24 | ||||
| Stearic C18:0 | 3.25 ± 0.47 | 5.04 ± 0.15 | ||||
| Cis Oleic C18:1 | 57.20 ± 0.24 | 7.01 ± 0.16 | ||||
| Trans oleic C18:1 | 3.08 ± 0.37 | 0.82 ± 0.02 | ||||
| Linoleic C18:2 | 12.50 ± 1.36 | 35.20 ± 0.50 | ||||
| Linolenic C18:3 | 0.79 ± 0.06 | 1.80 ± 0.04 | ||||
| Phenols (µg/g DW) | ||||||
| Free | Non-Extractable | Total | Free | Conjugated + Bound | Total | |
| Caffeic acid | 52.01 ± 2.54 | 576.70 ± 2.83 | 628.71 ± 5.37 | ND | ND | ND |
| 4-Hydroxybenzoic acid | ND | ND | ND | ND | 0.94 ± 0.17 | 0.90 ± 0.20 |
| Vanillic acid | 156.13 ± 7.83 | ND | 156.13 ± 7.83 | 0.64 ± 0.07 | 0.60 ± 0.18 | 1.20 ± 0.30 |
| Syringic acid | ND | ND | ND | ND | 0.71 ± 0.24 | 0.70 ± 0.20 |
| Cumaric acid | 117.74 ± 7.09 | 39.57 ± 2.33 | 157.31 ± 9.42 | 0.25 ± 0.01 | 1.80 ± 0.34 | 2.00 ± 0.35 |
| Ferulic acid | ND | 16.46 ± 2.31 | 16.46 ± 2.31 | 0.67 ± 0.04 | 72.50 ± 8.54 | 73.17 ± 8.78 |
| Sinapic acid | ND | ND | ND | 0.21 ± 0.01 | 8.50 ± 1.30 | 8.71 ± 1.31 |
| Caffeic acid derivative | ND | ND | ND | ND | 1.41 ± 0.34 | 1.41 ± 0.30 |
| Apigenin derivate | ND | ND | ND | 0.74 ± 0.38 | ND | 0.74 ± 0.38 |
| Vitexin | ND | ND | ND | 2.36 ± 1.21 | ND | 2.36 ± 1.21 |
| Quercetin 3-O-glucoside | 72.90 ± 3.81 | ND | 72.90 ± 3.81 | ND | ND | ND |
| Glicosylated luteolin derivate | 27.31 ± 0.91 | ND | 27.13 ± 0.91 | ND | ND | ND |
| Quercetin derivate | 308.23 ± 34.36 | ND | 308.23 ± 34.36 | ND | ND | ND |
| Luteolin | 532.59 ± 53.91 | ND | 532.59 ± 53.91 | 1.07 ± 0.08 | ND | 1.07 ± 0.08 |
| Apigenin | 30.06 ± 3.02 | ND | 30.06 ± 3.02 | 0.10 ± 0.02 | ND | 0.10 ± 0.02 |
| Luteolin derivate | 12.24 ± 1.24 | ND | 12.24 ± 1.24 | ND | ND | ND |
| Tyrosol | 932.12 ± 42.13 | ND | 936.12 ± 42.13 | ND | ND | ND |
| Oleuropein | 371.42 ± 25.41 | ND | 371.42 ± 25.41 | ND | ND | ND |
| Total | 2616.57 ± 182.25 | 632.73 ± 7.47 | 3249.30 ± 189.72 | 6.04 ± 1.84 | 86.50 ± 11.30 | 92.54 ± 13.10 |
| Uncooked Spaghetti | Cooked Spaghetti | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Color | Resistance to Break | Overall Quality | Elasticity | Firmness | Fibrous | Bulkiness | Adhesiveness | Color | Odor | Taste | Overall Quality | |
| CTRL | 7.20 ± 0.27 a | 7.27 ± 0.28 a | 7.23 ± 0.26 a | 7.08 ± 0.28 a | 7.21 ± 0.35 a | 7.02 ± 0.31 a | 6.14 ± 0.33 a | 6.21 ± 0.26 a | 7.56 ± 0.38 a | 7.23 ± 0.28 a | 7.01 ± 0.34 a | 7.20 ± 0.26 a |
| 10% OPF | 6.07 ± 0.30 b | 6.20 ± 0.27 b | 6.20 ± 0.27 b | 5.20 ± 0.26 b | 6.24 ± 0.30 b | 5.80 ± 0.25 b | 5.27 ± 0.30 b | 5.31 ± 0.34 b | 6.72 ± 0.32 b | 6.98 ± 0.30 a | 6.71 ± 0.36 a | 5.32 ± 0.32 b |
| 15% OPF | 5.43 ± 0.37 b | 5.20 ± 0.27 c | 5.34 ± 0.38 c | 4.20 ± 0.26 c | 5.80 ± 0.30 b | 4.80 ± 0.25 c | 4.77 ± 0.41 b | 4.95 ± 0.28 b | 5.36 ± 0.50 c | 6.18 ± 0.30 b | 5.84 ± 0.27 b | 4.42 ± 0.36 c |
| Uncooked Spaghetti | Cooked Spaghetti | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Color | Resistance to Break | Overall Quality | Elasticity | Firmness | Fibrous | Bulkiness | Adhesiveness | Color | Odor | Taste | Overall Quality | |
| CTRL | 7.20 ± 0.27 a | 7.27 ± 0.28 a | 7.23 ± 0.26 a | 7.08 ± 0.28 a | 7.21 ± 0.35 a | 7.02 ± 0.31 a | 6.20 ± 0.28 a | 6.05 ± 0.26 a | 7.56 ± 0.38 a | 7.23 ± 0.28 a | 7.01 ± 0.34 a | 7.20 ± 0.26 a |
| 10% OPF | 6.07 ± 0.30 b | 6.20 ± 0.27 c | 6.20 ± 0.27 b | 5.20 ± 0.26 b | 6.24 ± 0.30 b | 5.80 ± 0.25 b | 5.27 ± 0.30 b | 5.31 ± 0.34 b | 6.72 ± 0.32 b | 6.98 ± 0.30 a | 6.71 ± 0.36 a | 5.32 ± 0.32 b |
| 10% OPF-0.3 TG | 6.00 ± 0.30 b | 6.41 ± 0.36 b,c | 6.55 ± 0.36 b | 5.30 ± 0.35 b | 6.26 ± 0.31 b | 5.82 ± 0.20 b | 5.61 ± 0.38 b | 5.41 ± 0.38 b | 6.75 ± 0.28 b | 7.00 ± 0.31 a | 6.73 ± 0.36 a | 5.73 ± 0.36 b |
| 10% OPF-0.6 TG | 6.05 ± 0.26 b | 6.85 ± 0.30 a,b | 6.77 ± 0.31 a,b | 5.65 ± 0.21 b | 6.31 ± 0.30 b | 5.86 ± 0.21 b | 6.55 ± 0.25 a | 6.08 ± 0.28 a | 6.21 ± 0.33 b | 7.03 ± 0.32 a | 6.83 ± 0.25 a | 6.56 ± 0.33 a |
| OCT (min) | Cooking Loss (%) | Swelling Index (g Water Per g Dry Spaghetti) | Water Absorption (%) | Adhesiveness (Nmm) | Hardness (N) | |
|---|---|---|---|---|---|---|
| CTRL | 10.30 | 5.05 ± 0.28 c | 1.86 ± 0.07 a | 141 ± 3.64 a | 0.69 ± 0.06 a | 6.69 ± 0.32 c |
| 10% OPF | 9.00 | 6.20 ± 0.12 a | 1.75 ± 0.05 b | 138 ± 8.78 a,b | 0.78 ± 0.13 a | 7.96 ± 0.23 b |
| 10% OPF-0.3 TG | 9.30 | 5.93 ± 0.23 a,b | 1.62 ± 0.04 c | 128 ± 4.38 b,c | 0.68 ± 0.12 a | 8.15 ± 0.42 b |
| 10% OPF-0.6 TG | 10.00 | 5.65 ± 0.14 b | 1.61 ± 0.02 c | 126 ± 2.36 c | 0.62 ± 0.13 a | 9.55 ± 0.71 a |
| CTRL | Spaghetti 10% OPF | |||||
|---|---|---|---|---|---|---|
| Tocochromanols (µg/g DW) | ||||||
| α T | ND | 8.63 ± 0.04 | ||||
| Carotenoids (µg/g DW) | ||||||
| Lutein | 4.22 ± 0.95 a | 4.72 ± 0.09 a | ||||
| Zeaxanthin | 0.18 ± 0.002 a | 0.16 ± 0.02 a | ||||
| α-carotene | ND | 0.08 ± 0.001 | ||||
| β-carotene | ND | 0.27 ± 0.003 | ||||
| Total | 4.40 ± 0.95 a | 5.23 ± 0.11 b | ||||
| Triterpenic acids (mg/g DW) | ||||||
| Maslinic | ND | 1.32 ± 0.04 | ||||
| Eonolic | ND | 0.54 ± 0.05 | ||||
| Total | ND | 1.86 ± 0.07 | ||||
| Fatty acids (%) | ||||||
| Myristic C14:0 | 0.37 ± 0.01 a | 0.12 ± 0.01 b | ||||
| Palmitic C16:0 | 41.73 ± 0.19 a | 23.20 ± 0.32 b | ||||
| Palmitoleic C16:1 | 1.95 ± 0.11 a | 2.31 ± 0.09 b | ||||
| Stearic C18:0 | 7.50 ± 0.44 a | 2.49 ± 0.29 b | ||||
| Cis Oleic C18:1 | 12.60 ± 2.22 a | 38.90 ± 2.41 b | ||||
| Trans oleico C18:1 | 1.48 ± 0.15 a | 2.96 ± 0.20 b | ||||
| Linoleic C18:2 | 32.61 ± 1.46 a | 28.68 ± 2.75 a | ||||
| Linolenic C18:3 | 1.76 ± 0.18 a | 1.34 ± 0.64 a | ||||
| Phenols (µg/g DW) | ||||||
| Free | Conjugated + bound | Total | Free | Conjugated + bound | Total | |
| Caffeic acid | ND | ND | ND | 1.32 ± 0.14 | 2.25 ± 0.31 | 3.57 ± 0.45 |
| 4-Hydroxybenzoic acid | ND | 1.28 ± 0.07 a | 1.28 ± 0.07 a | ND | 2.81 ± 0.16 b | 2.81 ± 0.16 b |
| Vanillic acid | 0.56 ± 0.04 a | 0.78 ± 0.01 a | 1.34 ± 0.05 a | 7.28 ± 0.46 b | 1.99 ± 0.62 b | 9.27 ± 1.08 b |
| Syringic acid | ND | 0.75 ± 0.08 | 0.75 ± 0.08 | ND | ND | ND |
| Cumaric acid | 0.17 ± 0.01 | 1.75 ± 0.01 a | 1.92 ± 0.02 a | 1.22 ± 0.13 b | 23.47 ± 1.90 b | 24.69 ± 2.03 b |
| Ferulic acid | 0.35 ± 0.02 | 67.70 ± 0.19 a | 68.05 ± 0.21 a | ND | 60.93 ± 4.28 a | 60.93 ± 4.28 b |
| Sinapic acid | ND | 5.78 ± 0.55 a | 5.78 ± 0.55 a | ND | 9.79 ± 0.73 b | 9.79 ± 0.73 b |
| Caffeic acid derivative | ND | 2.05 ± 0.12 | 2.05 ± 0.12 a | 0.84 ± 0.08 | ND | 0.84 ± 0.08 b |
| Apigenin derivate | 0.27 ± 0.02 | ND | 0.27 ± 0.02 | ND | ND | ND |
| Vitexin | 0.78 ± 0.12 | ND | 0.78 ± 0.12 | ND | ND | ND |
| Quercetin 3-O-glucoside | ND | ND | ND | ND | ND | ND |
| Quercetin | ND | ND | ND | 5.49 ± 0.89 | ND | 5.49 ± 0.89 |
| Luteolin | 0.17 ± 0.08 a | ND | 0.17 ± 0.08 a | 11.46 ± 2.16 b | ND | 11.46 ± 2.16 b |
| Apigenin | ND | ND | ND | 1.18 ± 0.19 | ND | 1.18 ± 0.19 |
| Tyrosol | ND | ND | ND | 66.10 ± 2.47 | 36.23 ± 4.52 | 102.33 ± 6.99 |
| Oleuropein | ND | ND | ND | 12.72 ± 1.17 | ND | 12.72 ± 1.17 |
| Total | 2.30 ± 0.29 a | 80.09 ± 1.03 a | 82.39 ± 1.32 a | 107.61 ± 7.69 b | 137.47 ± 12.52 b | 245.08 ± 20.21 b |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Padalino, L.; D’Antuono, I.; Durante, M.; Conte, A.; Cardinali, A.; Linsalata, V.; Mita, G.; Logrieco, A.F.; Del Nobile, M.A. Use of Olive Oil Industrial By-Product for Pasta Enrichment. Antioxidants 2018, 7, 59. https://doi.org/10.3390/antiox7040059
Padalino L, D’Antuono I, Durante M, Conte A, Cardinali A, Linsalata V, Mita G, Logrieco AF, Del Nobile MA. Use of Olive Oil Industrial By-Product for Pasta Enrichment. Antioxidants. 2018; 7(4):59. https://doi.org/10.3390/antiox7040059
Chicago/Turabian StylePadalino, Lucia, Isabella D’Antuono, Miriana Durante, Amalia Conte, Angela Cardinali, Vito Linsalata, Giovanni Mita, Antonio F. Logrieco, and Matteo Alessandro Del Nobile. 2018. "Use of Olive Oil Industrial By-Product for Pasta Enrichment" Antioxidants 7, no. 4: 59. https://doi.org/10.3390/antiox7040059
APA StylePadalino, L., D’Antuono, I., Durante, M., Conte, A., Cardinali, A., Linsalata, V., Mita, G., Logrieco, A. F., & Del Nobile, M. A. (2018). Use of Olive Oil Industrial By-Product for Pasta Enrichment. Antioxidants, 7(4), 59. https://doi.org/10.3390/antiox7040059









