Avocado Seeds: Extraction Optimization and Possible Use as Antioxidant in Food
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Avocado Preparation
2.3. Extraction Procedure
2.4. Total Phenolic Content (TPC)
| Temperature (°C) | Ethanol Concentration (%) | Time (min) | TPC (mg GAE/g dw) | ORAC (mg TE/g dw) |
|---|---|---|---|---|
| 60.00 | 60.00 | 25.00 | 41.00 ± 0.97 | 104.16 ± 2.13 |
| 60.00 | 93.63 | 25.00 | 35.10 ± 0.24 | 116.12 ± 1.03 |
| 80.00 | 80.00 | 5.00 | 46.78 ± 0.59 | 153.17 ± 3.84 |
| 26.36 | 60.00 | 25.00 | 40.78 ± 0.17 | 70.54 ± 0.97 |
| 60.00 | 60.00 | 25.00 | 41.10 ± 0.57 | 106.10 ± 2.40 |
| 40.00 | 40.00 | 45.00 | 43.24 ± 0.76 | 104.01 ± 2.35 |
| 80.00 | 80.00 | 45.00 | 45.43 ± 0.49 | 144.94 ± 2.84 |
| 80.00 | 40.00 | 45.00 | 45.37 ± 1.39 | 130.08 ± 2.65 |
| 80.00 | 40.00 | 5.00 | 43.70 ± 0.66 | 150.03 ± 1.73 |
| 60.00 | 60.00 | 25.00 | 40.90 ± 0.47 | 104.28 ± 1.03 |
| 60.00 | 60.00 | 55.22 | 42.87 ± 0.70 | 158.77 ± 1.33 |
| 40.00 | 40.00 | 5.00 | 41.19 ± 0.55 | 99.17 ± 1.81 |
| 60.00 | 60.00 | 2.77 | 42.92 ± 1.13 | 155.44 ± 2.71 |
| 93.64 | 60.00 | 25.00 | 46.95 ± 0.09 | 126.23 ± 3.35 |
| 60.00 | 26.36 | 25.00 | 42.33 ± 0.10 | 129.78 ± 3.84 |
| 40.00 | 80.00 | 45.00 | 38.98 ± 0.45 | 100.72 ± 3.27 |
| 40.00 | 80.00 | 5.00 | 35.48 ± 0.55 | 91.01 ± 3.51 |
2.5. ORAC Assay
2.6. Statistical Analysis
2.7. Water-Oil Emulsions
2.8. Peroxide Value (PV)
2.9. Meat Preparation
2.10. Thiobarbituric Reactive Substances
3. Results and Discussion
3.1. Extraction Optimization
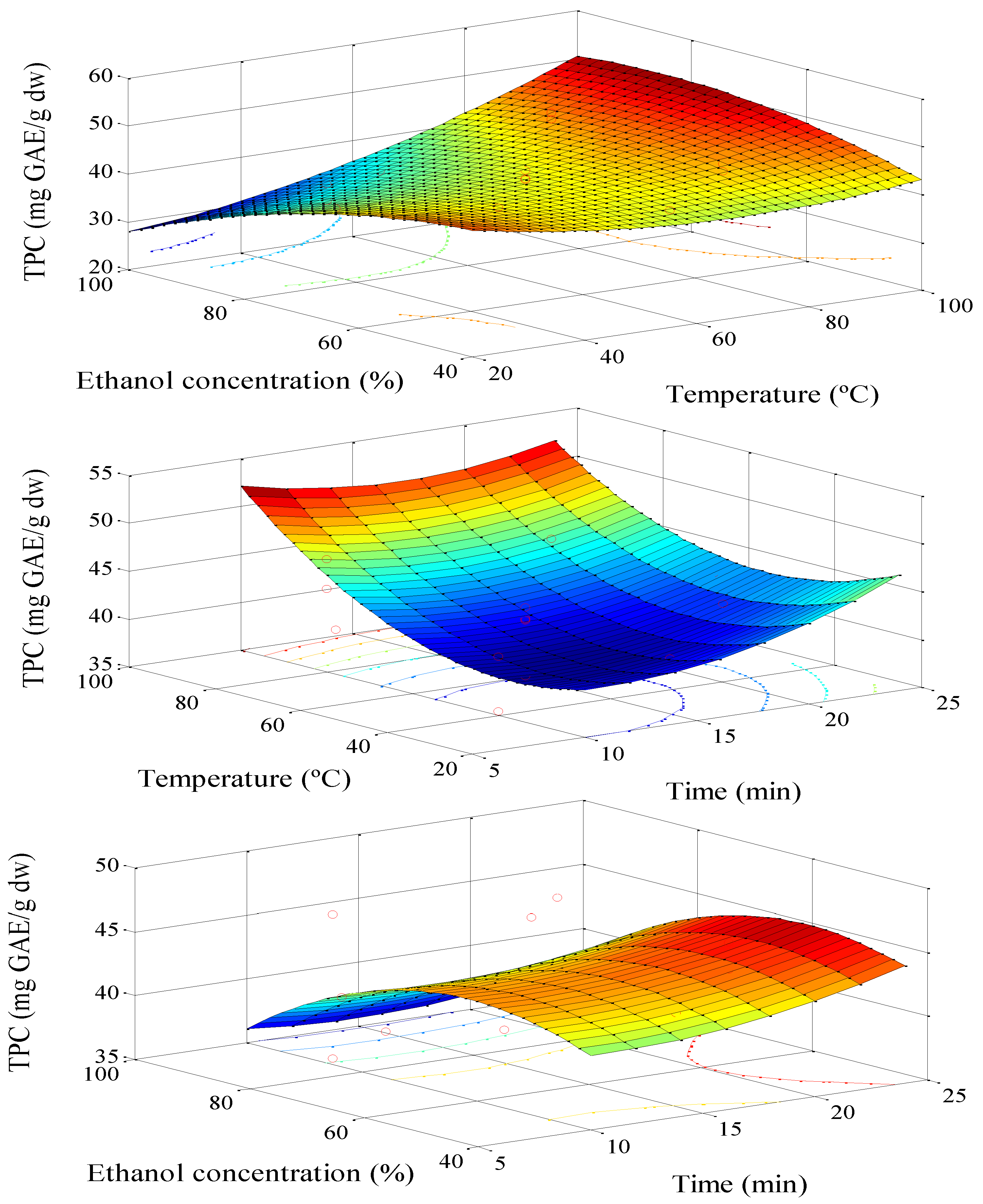
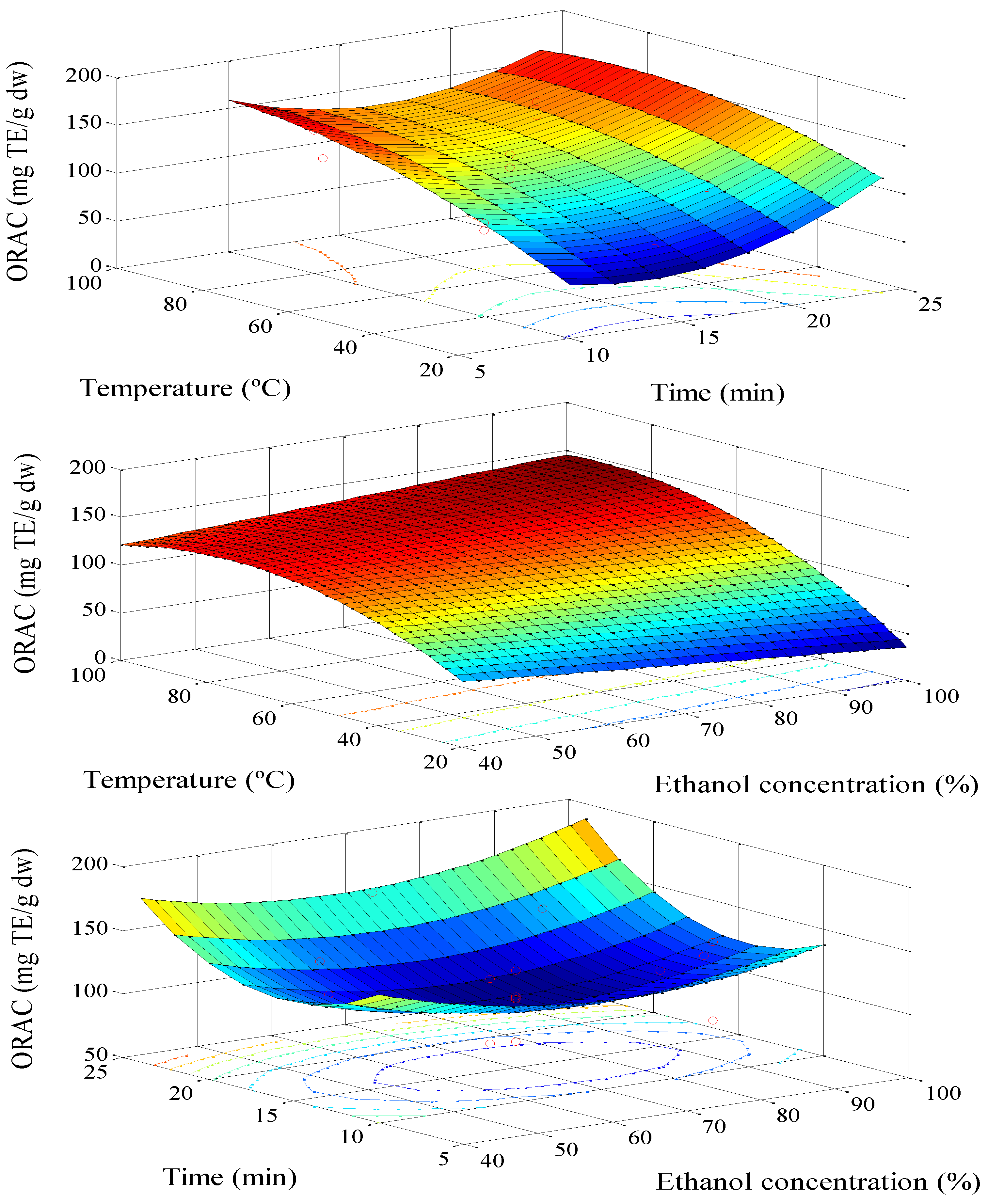
| Term | p-Value | |
|---|---|---|
| Response | ||
| TPC | ORAC | |
| Complete Model | ||
| Constant | 0.001 | 0.006 |
| Temperature (°C) | 0.012 | 0.069 |
| Ethanol (%) | 0.291 | 0.022 |
| Time (min) | 0.804 | 0.001 |
| Temperature (°C) × Temperature (°C) | 0.014 | 0.135 |
| Ethanol (%) × Ethanol (%) | 0.622 | 0.046 |
| Time (min) × Time (min) | 0.068 | 0.000 |
| Temperature (°C) × Ethanol (%) | 0.003 | 0.186 |
| Temperature (°C) × Time (min) | 0.119 | 0.071 |
| Ethanol (%) × Time (min) | 0.610 | 0.435 |
| Reduced Model | ||
| Constant | 0.000 | 0.000 |
| Temperature (°C) | 0.005 | 0.000 |
| Ethanol (%) | 0.001 | 0.031 |
| Time (min) | - | 0.000 |
| Temperature (°C) × Temperature (°C) | 0.029 | - |
| Ethanol (%) × Ethanol (%) | - | 0.033 |
| Time (min) × Time (min) | - | 0.000 |
| Temperature (°C) × Ethanol (%) | 0.004 | - |
| Response | Equation | R2 Value | |
|---|---|---|---|
| R2 | R2 Pred. | ||
| Complete Model | |||
| TPC | 62.87 − 0.47 T − 0.25 [%] − 0.14 t + 0.003 T2 − 0.001 [%]2 + 0.03 t2 + 0.006 T × [%] − 0.007 T × t − 0.003 [%] × t | 94.69 | 57.0 |
| ORAC | 318.2 + 2.03 T − 04.41 [%] − 019.5 t − 00.009 T2 + 0.023 [%]2+ 0.7 t2 + 0.012 T × [%] − 00.053 T × t − 00.03 [%] × t | 96.7 | 75.0 |
| Reduced Model | |||
| TPC | 69.7 − 00.53 T − 00.39 [%] + 0.002 T2 − 00.006 T × [%] | 85.7 | 66.76 |
| ORAC | 345.7 + 1.01 T − 03.92 [%] − 022.01 t + 0.027 [%]2 + 0.73 t2 | 91.88 | 77.88 |
| Model | Conditions | Response | ||||
|---|---|---|---|---|---|---|
| Temperature (°C) | Ethanol (%) | Time (min) | Predicted | Predicted RM | Experimental | |
| TPC | 63 | 56 | 23 | 51.75 | 43.6 | 45.01 |
| ORAC | 93.6 | 44.7 | 7 | 206.82 | 200.66 | 154.3 |
3.2. Extract Optimized Effect in Oil-in-Water Emulsions (O/W)
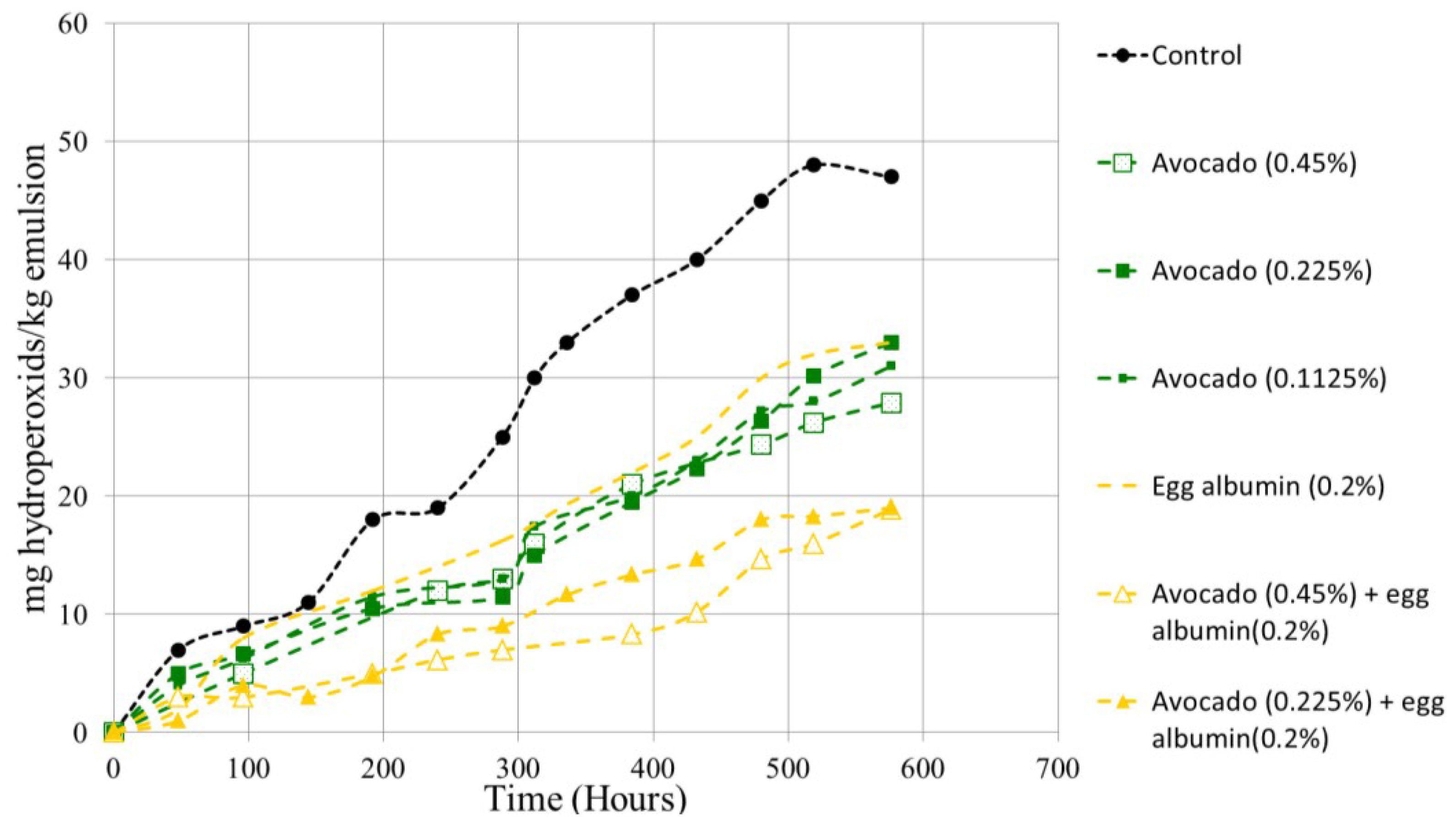
3.3. Effect of the Extract in Burger Meat

4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Martínez, R.; Torres, P.; Meneses, M.A.; Figueroa, J.G.; Pérez-Álvarez, J.A.; Viuda-Martos, M. Chemical, technological and in vitro antioxidant properties of mango, guava, pineapple and passion fruit dietary fibre concentrate. Food Chem. 2012, 135, 1520–1526. [Google Scholar] [CrossRef]
- Ignat, I.; Volf, I.; Popa, V.I. A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chem. 2011, 126, 1821–1835. [Google Scholar] [CrossRef]
- Rubilar, M.; Pinelo, M.; Ihl, M.; Scheuermann, E.; Sineiro, J.; Nuñez, M.J. Murta leaves (Ugni molinae Turcz) as a source of antioxidant polyphenols. J. Agric. Food Chem. 2006, 54, 59–64. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jime, L. Polyphenols: Food sources and bioavailability 1,2. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Perumalla, A.V.S.; Hettiarachchy, N.S. Green tea and grape seed extracts—Potential applications in food safety and quality. Food Res. Int. 2011, 44, 827–839. [Google Scholar] [CrossRef]
- Wettasinghe, M.; Shahidi, F. Phenolic acids in defatted seeds of borage (Borago officinalis L.). Food Chem. 2001, 75, 49–56. [Google Scholar] [CrossRef]
- Jordán, M.; Lax, V.; Rota, M.C.; Lora, S.; Sotomayor, J.A. Relevance of carnosic acid, carnosol, and rosmarinic acid concentrations in the in vitro antioxidant and antimicrobial activities of Rosmarinus officinalis (L.) methanolic extracts. J. Agric. Food Chem. 2012, 60, 9603–9608. [Google Scholar] [CrossRef]
- Ayala-Zavala, J.F.; Vega-Vega, V.; Rosas-Domínguez, C.; Palafox-Carlos, H.; Villa-Rodriguez, J.A.; Siddiqui, M.W.; Dávila-Aviña, J.E.; González-Aguilar, G.A. Agro-industrial potential of exotic fruit byproducts as a source of food additives. Food Res. Int. 2011, 44, 1866–1874. [Google Scholar] [CrossRef]
- Wijngaard, H.H.; Brunton, N. The optimisation of solid–liquid extraction of antioxidants from apple pomace by response surface methodology. J. Food Eng. 2010, 96, 134–140. [Google Scholar] [CrossRef]
- Lagha-Benamrouche, S.; Madani, K. Phenolic contents and antioxidant activity of orange varieties (Citrus sinensis L. and Citrus aurantium L.) cultivated in Algeria: Peels and leaves. Ind. Crops Prod. 2013, 50, 723–730. [Google Scholar] [CrossRef]
- Aguedo, M.; Kohnen, S.; Rabetafika, N.; Vanden Bossche, S.; Sterckx, J.; Blecker, C.; Beauve, C.; Paquot, M. Composition of by-products from cooked fruit processing and potential use in food products. J. Food Compos. Anal. 2012, 27, 61–69. [Google Scholar] [CrossRef]
- Wijngaard, H.; Hossain, M.B.; Rai, D.K.; Brunton, N. Techniques to extract bioactive compounds from food by-products of plant origin. Food Res. Int. 2012, 46, 505–513. [Google Scholar] [CrossRef]
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Guerrero, M.S.; Torres, J.S.; Nuñez, M.J. Extraction of polyphenols from white distilled grape pomace: Optimization and modelling. Bioresour. Technol. 2008, 99, 1311–1318. [Google Scholar] [CrossRef]
- Rodríguez-Carpena, J.G.; Morcuende, D.; Estévez, M. Avocado by-products as inhibitors of color deterioration and lipid and protein oxidation in raw porcine patties subjected to chilled storage. Meat Sci. 2011, 89, 166–173. [Google Scholar] [CrossRef]
- Soong, Y.-Y.; Barlow, P.J. Antioxidant activity and phenolic content of selected fruit seeds. Food Chem. 2004, 88, 411–417. [Google Scholar] [CrossRef]
- Moure, A.; Cruz, J.M.; Franco, D.; Domínguez, J.M.; Sineiro, J.; Domínguez, H.; Jose, M.; Parajo, J.C. Natural antioxidants from residual sources. Food Chem. 2001, 72, 145–171. [Google Scholar] [CrossRef]
- Saha, J.; Debnath, M.; Saha, A.; Ghosh, T.; Sarkar, P.K. Response surface optimisation of extraction of antioxidants from strawberry fruit, and lipid peroxidation inhibitory potential of the fruit extract in cooked chicken patties. J. Sci. Food Agric. 2011, 91, 1759–1765. [Google Scholar] [CrossRef]
- Díaz Reinoso, B.; Couto, D.; Moure, A.; Fernandes, E.; Domínguez, H.; Parajó, J.C. Optimization of antioxidants—Extraction from Castanea sativa leaves. Chem. Eng. J. 2012, 203, 101–109. [Google Scholar] [CrossRef]
- Pompeu, D.R.; Silva, E.M.; Rogez, H. Optimisation of the solvent extraction of phenolic antioxidants from fruits of Euterpe oleracea using Response Surface Methodology. Bioresour. Technol. 2009, 100, 6076–6082. [Google Scholar] [CrossRef]
- Wijngaard, H.; Brunton, N. The optimization of extraction of antioxidants from apple pomace by pressurized liquids. J. Agric. Food Chem. 2009, 57, 10625–10631. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Casettari, L.; Gennari, L.; Angelino, D.; Ninfali, P.; Castagnino, E. ORAC of chitosan and its derivatives. Food Hydrocoll. 2012, 28, 243–247. [Google Scholar] [CrossRef]
- Singh, G.; Maurya, S.; DeLampasona, P.; Catalan, C. A comparison of chemical, antioxidant and antimicrobial studies of cinnamon leaf and bark volatile oils, oleoresins and their constituents. Food Chem. Toxicol. 2007, 45, 1650–1661. [Google Scholar] [CrossRef]
- Doménech-Asensi, G.; García-Alonso, F.J.; Martínez, E.; Santaella, M.; Martín-Pozuelo, G.; Bravo, S.; Periago, M.J. Effect of the addition of tomato paste on the nutritional and sensory properties of mortadella. Meat Sci. 2013, 93, 213–219. [Google Scholar] [CrossRef]
- Spigno, G.; de Faveri, D.M. Antioxidants from grape stalks and marc: Influence of extraction procedure on yield, purity and antioxidant power of the extracts. J. Food Eng. 2007, 78, 793–801. [Google Scholar] [CrossRef]
- Amendola, D.; de Faveri, D.M.; Spigno, G. Grape marc phenolics: Extraction kinetics, quality and stability of extracts. J. Food Eng. 2010, 97, 384–392. [Google Scholar] [CrossRef]
- Boussetta, N.; Vorobiev, E.; Le, L.H.; Cordin-falcimaigne, A.; Lanoisellé, J. Application of electrical treatments in alcoholic solvent for polyphenols extraction from grape seeds. LWT-Food Sci. Technol. 2012, 46, 127–134. [Google Scholar] [CrossRef]
- Rodríguez-Rojo, S.; Visentin, A.; Maestri, D.; Cocero, M.J. Assisted extraction of rosemary antioxidants with green solvents. J. Food Eng. 2012, 109, 98–103. [Google Scholar] [CrossRef]
- Cacace, J.E.; Mazza, G. Mass transfer process during extraction of phenolic compounds from milled berries. J. Food Eng. 2003, 59, 379–389. [Google Scholar] [CrossRef]
- Tubtimdee, C.; Shotipruk, A. Extraction of phenolics from Terminalia chebula Retz with water–ethanol and water–propylene glycol and sugaring-out concentration of extracts. Sep. Purif. Technol. 2011, 77, 339–346. [Google Scholar] [CrossRef]
- Qu, W.; Pan, Z.; Ma, H. Extraction modeling and activities of antioxidants from pomegranate marc. J. Food Eng. 2010, 99, 16–23. [Google Scholar] [CrossRef]
- Thoo, Y.Y.; Ho, S.K.; Liang, J.Y.; Ho, C.W.; Tan, C.P. Effects of binary solvent extraction system, extraction time and extraction temperature on phenolic antioxidants and antioxidant capacity from mengkudu (Morinda citrifolia). Food Chem. 2010, 120, 290–295. [Google Scholar] [CrossRef]
- Pahua-Ramos, M.E.; Ortiz-Moreno, A.; Chamorro-Cevallos, G.; Hernández-Navarro, M.D.; Garduño-Siciliano, L.; Necoechea-Mondragón, H.; Hernández-Ortega, M. Hypolipidemic effect of avocado (Persea americana Mill) seed in a hypercholesterolemic mouse model. Plant Foods Hum. Nutr. 2012, 67, 10–16. [Google Scholar] [CrossRef]
- Sun, Y.; Xu, W.; Zhang, W.; Hu, Q.; Zeng, X. Optimizing the extraction of phenolic antioxidants from kudingcha made frrom Ilex kudingcha C.J. Tseng by using response surface methodology. Sep. Purif. Technol. 2011, 78, 311–320. [Google Scholar] [CrossRef]
- Kahyaoglu, T. Optimization of the pistachio nut roasting process using response surface methodology and gene expression programming. LWT-Food Sci. Technol. 2008, 41, 26–33. [Google Scholar] [CrossRef]
- Kargar, M.; Spyropoulos, F.; Norton, I.T. The effect of interfacial microstructure on the lipid oxidation stability of oil-in-water emulsions. J. Colloid Interface Sci. 2011, 357, 527–533. [Google Scholar] [CrossRef]
- Poyato, C.; Navarro-blasco, I.; Isabel, M.; Yolanda, R.; Astiasarán, I.; Ansorena, D. Oxidative stability of O/W and W/O/W emulsions: Effect of lipid composition and antioxidant polarity. Food Res. Int. 2013, 51, 132–140. [Google Scholar] [CrossRef]
- Sun, C.; Gunasekaran, S. Effects of protein concentration and oil-phase volume fraction on the stability and rheology of menhaden oil-in-water emulsions stabilized by whey protein isolate with xanthan gum. Food Hydrocoll. 2009, 23, 165–174. [Google Scholar] [CrossRef]
- Almajano, M.P.; Delgado, M.E.; Gordon, M.H. Changes in the antioxidant properties of protein solutions in the presence of epigallocatechin gallate. Food Chem. 2007, 101, 126–130. [Google Scholar] [CrossRef]
- Bonoli-Carbognin, M.; Erretani, L.O.C.; Endini, A.L.B.; Goidanich, P.; Cesena, I. Bovine serum albumin produces a synergistic increase in the antioxidant activity of virgin olive oil phenolic compounds in oil-in-water emulsions. J. Agric. Food Chem. 2008, 56, 7076–7081. [Google Scholar] [CrossRef]
- Sasaki, K.; Alamed, J.; Weiss, J.; Villeneuve, P.; López, L.J.; Lecomte, J.; Figueroa-espinoza, M.; Decker, E.A. Relationship between the physical properties of chlorogenic acid esters and their ability to inhibit lipid oxidation in oil-in-water emulsions. Food Chem. 2010, 118, 830–835. [Google Scholar] [CrossRef]
- Mendes, R.; Cardoso, C.; Pestana, C. Measurement of malondialdehyde in fish: A comparison study between HPLC methods and the traditional spectrophotometric test. Food Chem. 2009, 112, 1038–1045. [Google Scholar] [CrossRef]
- Seljeskog, E.; Hervig, T.; Mansoor, M.A. A novel HPLC method for the measurement of thiobarbituric acid reactive substances (TBARS). A comparison with a commercially available kit. Clin. Biochem. 2006, 39, 947–954. [Google Scholar] [CrossRef]
- Wenjiao, F.; Yongkui, Z.; Yunchuan, C.; Junxiu, S.; Yuwen, Y. TBARS predictive models of pork sausages stored at different temperatures. Meat Sci. 2014, 96, 1–4. [Google Scholar] [CrossRef]
- Weiss, J.; Gibis, M.; Schuh, V.; Salminen, H. Advances in ingredient and processing systems for meat and meat products. Meat Sci. 2010, 86, 196–213. [Google Scholar] [CrossRef]
- Rodríguez-Carpena, J.G.; Morcuende, D.; Estévez, M. Avocado, sunflower and olive oils as replacers of pork back-fat in burger patties: Effect on lipid composition, oxidative stability and quality traits. Meat Sci. 2012, 90, 106–115. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gómez, F.S.; Sánchez, S.P.; Iradi, M.G.G.; Azman, N.A.M.; Almajano, M.P. Avocado Seeds: Extraction Optimization and Possible Use as Antioxidant in Food. Antioxidants 2014, 3, 439-454. https://doi.org/10.3390/antiox3020439
Gómez FS, Sánchez SP, Iradi MGG, Azman NAM, Almajano MP. Avocado Seeds: Extraction Optimization and Possible Use as Antioxidant in Food. Antioxidants. 2014; 3(2):439-454. https://doi.org/10.3390/antiox3020439
Chicago/Turabian StyleGómez, Francisco Segovia, Sara Peiró Sánchez, Maria Gabriela Gallego Iradi, Nurul Aini Mohd Azman, and María Pilar Almajano. 2014. "Avocado Seeds: Extraction Optimization and Possible Use as Antioxidant in Food" Antioxidants 3, no. 2: 439-454. https://doi.org/10.3390/antiox3020439
APA StyleGómez, F. S., Sánchez, S. P., Iradi, M. G. G., Azman, N. A. M., & Almajano, M. P. (2014). Avocado Seeds: Extraction Optimization and Possible Use as Antioxidant in Food. Antioxidants, 3(2), 439-454. https://doi.org/10.3390/antiox3020439





