Abstract
Nowadays, sweet potato (Ipomoea batata L.; Lam.) is considered a very interesting nutritive food because it is rich in complex carbohydrates, but as a tubercle, contains high amounts of health-promoting secondary metabolites. The aim of this review is to summarize the most recently published information on this root vegetable, focusing on its bioactive phytochemical constituents, potential effects on health, and the impact of processing technologies. Sweet potato is considered an excellent source of dietary carotenoids, and polysaccharides, whose health benefits include antioxidant, anti-inflammatory and hepatoprotective activity, cardiovascular protection, anticancer properties and improvement in neurological and memory capacity, metabolic disorders, and intestinal barrier function. Moreover, the purple sweet potato, due to its high anthocyanin content, represents a unique food option for consumers, as well as a potential source of functional ingredients for healthy food products. In this context, the effects of commercial processing and domestic cooking techniques on sweet potato bioactive compounds require further study to understand how to minimize their loss.
1. Introduction
Sweet potato (Ipomoea batata L.; Lam.) is a dicotyledonous vegetable belonging to the family Convolvulaceae. It is the seventh most produced crop worldwide after wheat, rice, maize, potato, barley, and cassava, and the fifth in developing countries [1]. Sweet potato tubers, leaves, and shoots are good sources of nutrients for humans and animals, with around 50% of the crop used for animal feed.
Sweet potato tubers contain macronutrients such as starch, dietary fiber, and protein, in addition to an extensive range of micronutrients, including minerals (manganese, copper, potassium, and iron), vitamins (mainly B complex, C, and E), and provitamin A (as carotenoids), anthocyanins (purple sweet potatoes), flavonoids, and coumarins [2]. Compared to other root and tuber crops, the sweet potato contains more carbohydrates and proteins, as well as certain vitamins and minerals [3], and it has higher levels of provitamin A, vitamin C, and minerals than wheat or rice [4].
Due to its content of several bioactive secondary metabolites, the sweet potato is attracting the attention of the food industry, consumers, and scientists [5], not only as a healthy product but also as an ingredient for functional foods [6]. These phytochemicals provide physiological benefits that ultimately, either individually or collectively, promote health and longevity in consumers [7].
The color of this food is linked to its beneficial health effects [6]. Lighter fleshed varieties are reported to have higher levels of phenolic compounds, whereas a more intense yellow color is associated with a higher content of carotenoids, mainly ß-carotene [8]. Additionally, yellow- and orange-fleshed sweet potatoes are rich in phenolic acids, while those varieties that are purple have very high levels of anthocyanins [9,10].
Recent reviews on sweet potato leaves covered current knowledge of their bioactive composition and possible health effects [11] and the protocols developed for plant regeneration as an alternative method to produce disease-free planting material [12]. Beyond the widely studied potential of sweet potato leaves and their cultivars practices, sweet potato roots are more than just foods used for centuries as a major source of carbohydrates. Today they are recognized as a highly nutritious and useful food for the prevention of chronic diseases. To corroborate this, we have integrated for the first time updated information on their content in bioactive compounds, and an extensive revision of the in vitro and in vivo evidence of its benefits for human health. In addition, since phenolic compounds are important constituents of a healthy human diet, we have explored the effect of sweet potato processing on phenolic compounds.
2. Bioactive Compounds in Sweet Potato Tubers
Sweet potatoes are a good source of several bioactive compounds, above all (poly)phenols, terpenoids, tannins, saponins, glycosides, alkaloids, and phytosterols. The diversity of skin and flesh color in this root vegetable arises from the different levels of (poly)phenols and carotenoids [9,13,14,15,16,17]. Thus, the dominant pigments in purple sweet potatoes are anthocyanins and phenolic acids, whereas, in yellow and orange-fleshed sweet potatoes, they are phenolic acids, flavonoids, and carotenoids [13,14]. In addition to genetics, the concentration and bioavailability of bioactive compounds in sweet potatoes and derived products are affected by external factors such as environment and cultivar, storage conditions, and processing; moreover, the available data can be influenced by the extraction and analytical methods [18,19].
2.1. (Poly)phenols
Sweet potatoes are characterized by a high concentration of flavonoids and phenolic acids [14]. Flavonoids are mainly found in purple-fleshed potatoes in the form of anthocyanins and quercetin glycosides [13,14]. The yellow- and orange-fleshed tubers contain a mixture of phenolic acids, above all caffeic acid, chlorogenic acid, and caffeoylquinic acid derivatives [14].
2.1.1. Total (Poly)phenol Content
The total (poly)phenol content (TPC) in sweet potato flesh has been determined by several authors using a simple spectrophotometric method, and the reported values are present in a broad range from 10 to 408 mg of chlorogenic acid equivalents/100 g of fresh weight [9,20,21] or from 1.8 to 136.1 mg of gallic acid equivalents/100 g fresh weight (Table 1, Table 2 and Table 3) [13,22]. The wide variability of (poly)phenol content in sweet potatoes is associated with their genetic diversity, being higher in those with light orange, white, and yellow flesh [9]. This variation can also be influenced by environmental factors such as the type of soil, sun exposure, rainfall, and level of ripeness, as well as the cultivation method [13,23]. (Poly)phenol concentrations in sweet potatoes are lower in locations with more daylight hours and cooler temperatures. Thus, the TPC of commercial sweet potatoes cultivated in the US ranged from 57.1 to 78.6 mg of chlorogenic acid equivalents/100 g of fresh weight [20], whereas orange sweet potatoes grown in Bangladesh contained about 94.3–136.1 mg gallic acid equivalents/100 g fresh weight (Table 1) [13]. Moreover, food processing techniques, including cooking, can also alter the TPC in this root vegetable [1,18,24].

Table 2.
(Poly)phenol content of purple sweet potato flesh.

Table 3.
(Poly)phenol content of white, yellow, and red sweet potato flesh.

Table 1.
(Poly)phenol content of orange sweet potato flesh.
Table 1.
(Poly)phenol content of orange sweet potato flesh.
| Origin | Sample Extraction | Analytical Method | Phytochemical | Amount of Phytochemical | Ref. |
|---|---|---|---|---|---|
| USA | Methanol (80%) | Folin–Ciocalteu | TPC | ~1.4 mg CA/g FW | [9] |
| Methanol (80%) | pH-differential | TAC | <0.1 mg anthocyanins/g FW | ||
| USA | Ethanol (80%) | Folin–Ciocalteu | TPC | ~0.1 mg CA/g FW | [20] |
| Bangladesh | Acetone:water (7:3, v/v) | Folin-Ciocalteu | TPC | 94.3 to 136.1 mg GA/100 g FW | [13] |
| Kenya | Methanol (80%) | Folin–Ciocalteu | TPC | TPC mg GA/100 g DW:
| [18,25] |
| Aluminum chloride | TPC | TPC mg catechin/100 g DW:
| |||
| USA | Ethanol (80%) | Folin–Ciocalteu | TPC | 57.1 to 78.6 mg CA/100 g FW | [20] |
| HPLC-DAD and LC-MS/MS | Phenolic acids: chlorogenic acid, caffeic acid, di-O-caffeoylquinic acids | Phenolic acids mg/100 g FW:
| |||
| Pakistan | Ethyl acetate | Folin–Ciocalteu | TPC | 319.8 μg GA/mg DE | [26] |
| Methanol | 262.6 μg GA/mg DE | ||||
| Ethyl acetate | Aluminum chloride | TFC | 208.8 μg quercetin/mg DE | ||
| Methanol | 177.8 μg quercetin/mg DE | ||||
| Korea | Methanol (50%) withn1.2 M HCl at 80 °C | HPLC system | Quercetin, myricetin, kaempferol, luteolin, ferulic, p-coumaric, p-hydroxybenzoic, sinapic, syringic, and vanillic acids. | Flavonoids: 127.1 µg/g DW (Quercetin: 59.9, myricetin: 39.8, kaempferol: 18.9, luteolin: 8.5) Phenolic acids: 71.1 µg/g DW (p-hydroxybenzoic acid:7.8, vanillic acid 7.9, syringic acid:3.8, p-coumaric:11.9, ferulic acid:24.6, sinapic acid:15.2) | [27] |
CA: chlorogenic acid; DE: dry extract; DW: dry weight; FW: Fresh weight; GA: gallic acid, HPLC: high-performance liquid chromatography; MS: mass spectrometry; TPC: Total phenolic content; TFC: Total flavonoid content; TAC: Total anthocyanin content.
2.1.2. Flavonoids
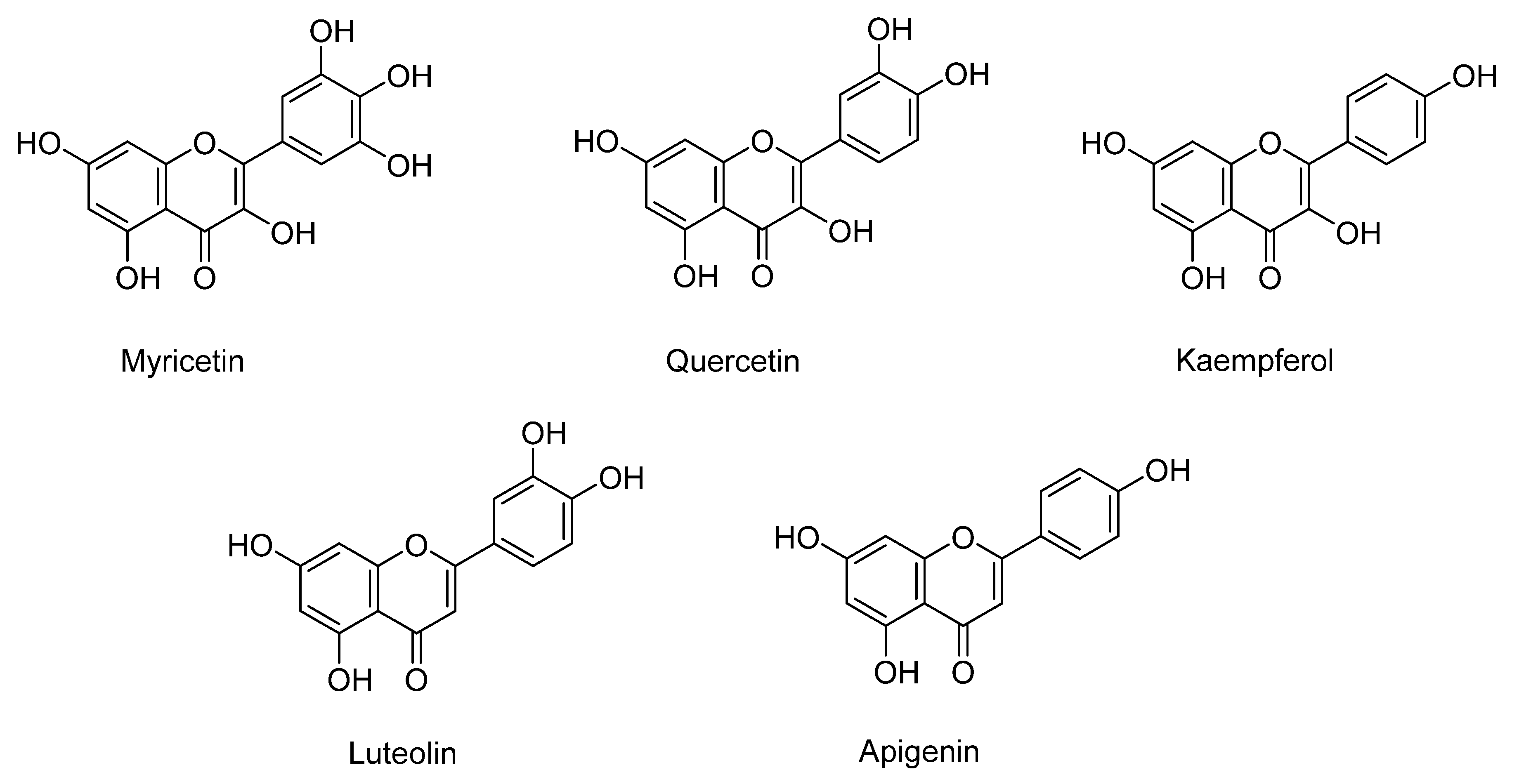
Quercetin, myricetin, luteolin, kaempferol, and apigenin are the flavonoids identified in sweet potatoes, especially in varieties with orange and purple flesh (Figure 1). As mentioned, the flavonoid content depends on the variety, being higher in purple-fleshed (579.5 µg/g dry weight) than orange- (121.1 µg/g dry weight) and white-fleshed (45.4 µg/g dry weight) sweet potatoes [27]. The major flavonoid in purple and orange potatoes is quercetin, followed by myricetin, kaempferol, and luteolin [27].
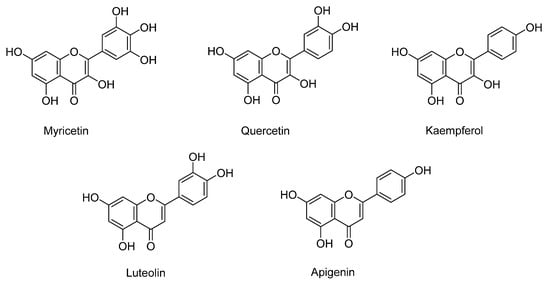
Figure 1.
The main flavonoids identified in sweet potato.
2.1.3. Anthocyanins (In Purple Sweet Potato)
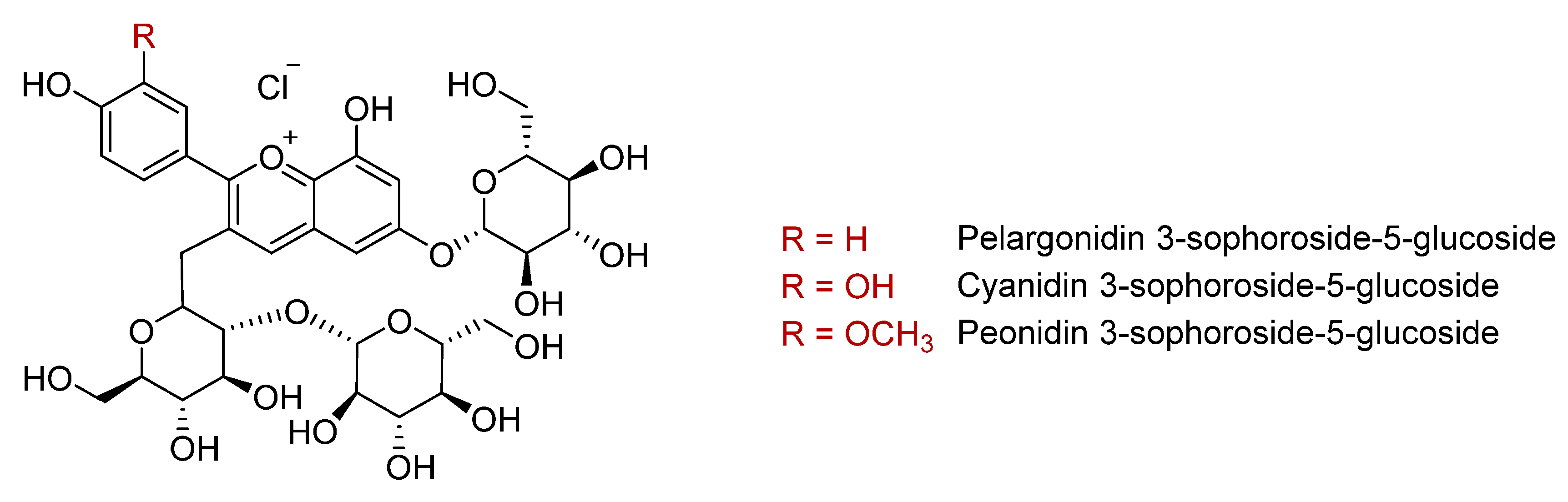
Anthocyanins are the flavonoid family responsible for the purple coloration of sweet potato flesh and skin. Total anthocyanin concentrations are higher in the purple varieties than those with orange flesh, with reported values of 14–182 mg/100 g fresh weight (Table 2) [9,21,31,38,39]. More than 20 anthocyanins have been identified in sweet potatoes [17,33,34,35,40,41,42]. The main ones in purple varieties are 3-sophoroside-5-glucoside derivatives of peonidin, cyanidin, and pelargonidin aglycones (Figure 2), almost all of them mono- or di-acylated with p-hydroxybenzoic acid, ferulic acid, or caffeic acid [16,29,32,33,34,37,40,42]. According to Fossen et al., these acylated forms represent more than 98% of the total anthocyanin content in purple sweet potato [43]. However, the anthocyanin composition differs widely between varieties. Thus, Im et al. reported that ‘Sinjami’, ‘Danjami’, and ‘Yeonjami’ contained 72–77% di-acylated anthocyanins compared to 90–95% in the Korean varieties ‘Jami’ and ‘Borami’ [22]. Conversely, a higher proportion of mono-acylated anthocyanins (21–24%) was found in ‘Sinjami’, ‘Danjami’, and ‘Yeonjami’ compared to ‘Jami’ and ‘Borami’ [22]. Acylation with the phenolic acids p-coumaric, ferulic, or caffeic acids enhances the stability of anthocyanins in conditions of heat, pH, and ultraviolet radiation, which facilitates their application in the food industry as natural colorants [42,44,45]. Compared to non- and di-acylated anthocyanins, mono-acylated forms have a higher resistance to heat, especially those derived from cyanidin 3- -hydroxybenzoylsophoroside-5-glucoside [35]. Therefore, these acylated forms could contribute to the higher antioxidant activity of purple sweet potatoes compared to those of other colours [35].
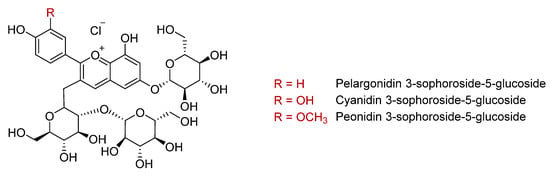
Figure 2.
Main anthocyanins in purple sweet potato.
2.1.4. Phenolic Acids
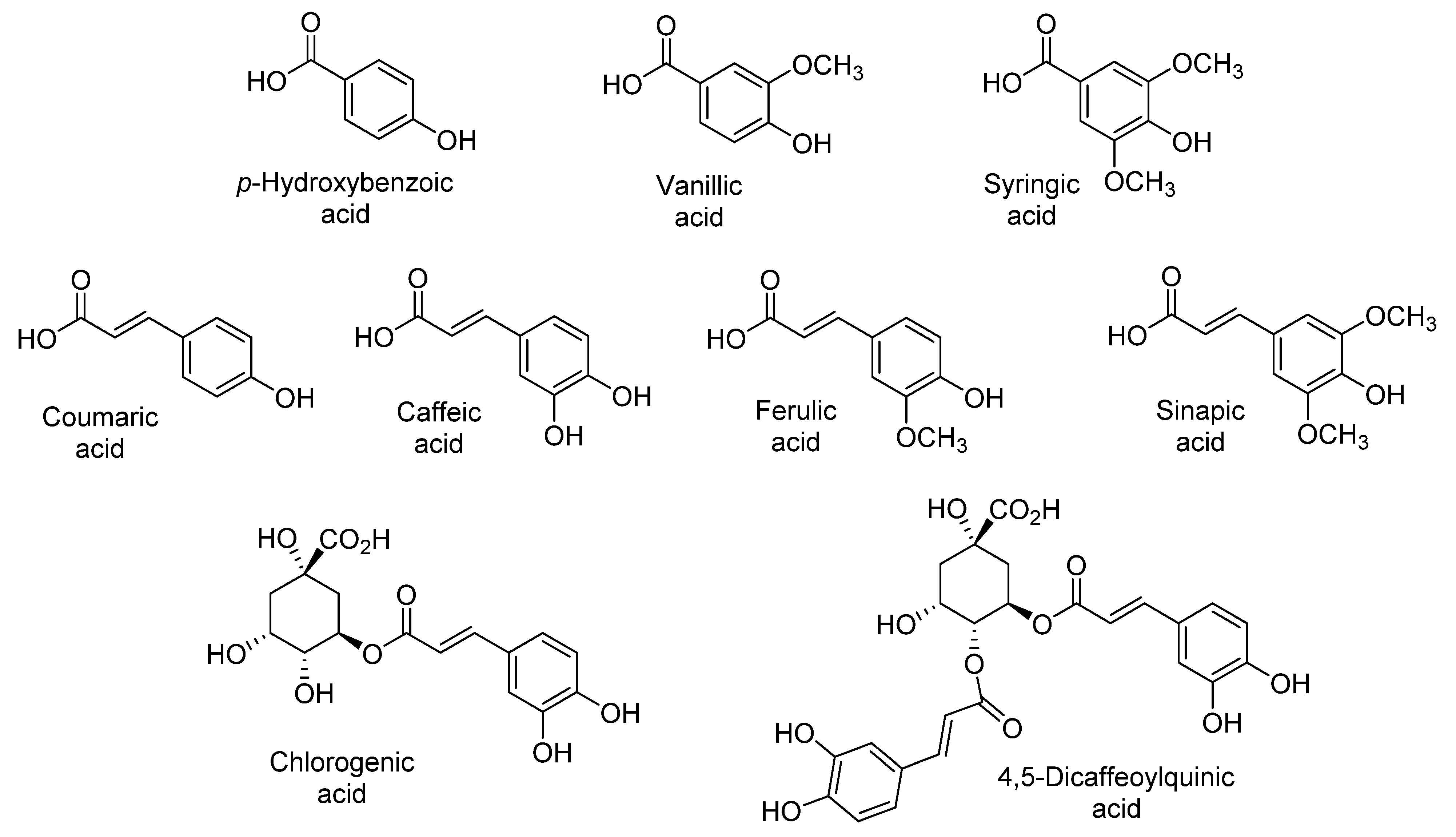
The sensory qualities of sweet potato are associated with their content of phenolic acids, namely chlorogenic, dicaffeoylquinic, caffeic, ferulic, p-hydroxybenzoic, coumaric, sinapic, syringic, and vanillic acids (Figure 3) [9,13,20,21,22,26,27,30]. Park et al. reported that the level of these phenolic acids varies with the flesh color, being almost ten-fold higher in purple (744.3 µg/g dry weight) than in orange and white sweet potatoes (71.1 µg/g dry weight and 52.5 µg/g dry weight, respectively) (Table 1, Table 2 and Table 3) [27]. The predominance of chlorogenic, ferulic, coumaric, and caffeic acids in purple-fleshed sweet potatoes is due to the stability afforded by their interaction with anthocyanins [22,27,44,45,46].
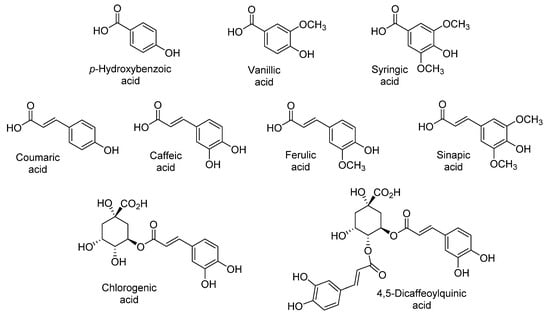
Figure 3.
Main phenolic acids identified in sweet potato.
2.1.5. Isolation, Identification, and Quantification of Phenolic Compounds in Sweet Potatoes
The isolation of total monomeric anthocyanins and other phenolic compounds from sweet potatoes is usually carried out using solvent extraction processes. Methanol and ethanol (80:20, solvent: water) are the most used polar solvents, as they provide higher phenolic extraction efficiency [9,20,30,31,46]. According to the results of Vishnu et al. (2019), the extraction of anthocyanins from purple sweet potato is more efficient when using acidified methanol with 0.5% trifluoroacetic acid compared to ethanol, methanol/trifluoroacetic acid/water, and ethanol/trifluoroacetic acid/water [32].
Spectrophotometric methods are used to quantify total (poly)phenols and anthocyanins. Folin–Ciocalteu reagent has been applied to quantify total (poly)phenols in sweet potatoes, commonly expressed as equivalents of chlorogenic acid or gallic acid [13,20,21,22,24,26,30]. Nevertheless, this reagent could provide overestimated results because of a tendency to react with other compounds in sweet potatoes, such as sugars and ascorbic acid [47,48]. Padda et al. reported that the average TPC in sweet potato tubers measured with Folin–Denis and Folin–Ciocalteu reagents were 60.9 and 74.6 mg/100 g fresh weight, respectively, indicating that the Folin–Denis reagent is an alternative option for (poly)phenol quantification in sweet potatoes [46]. Regarding anthocyanins, the total content is commonly quantified using a pH differential-spectrophotometry method reading at ranges from 420 to 730 nm [9,21,30,37]. The major individual phenolic compounds in sweet potatoes are identified and quantified using phase-reverse phase high-performance liquid chromatography (HPLC), electrospray ionization mass spectrometry, and tandem MS [20,22,27,31,32,33,35,40,41,42].
2.2. Carotenoids
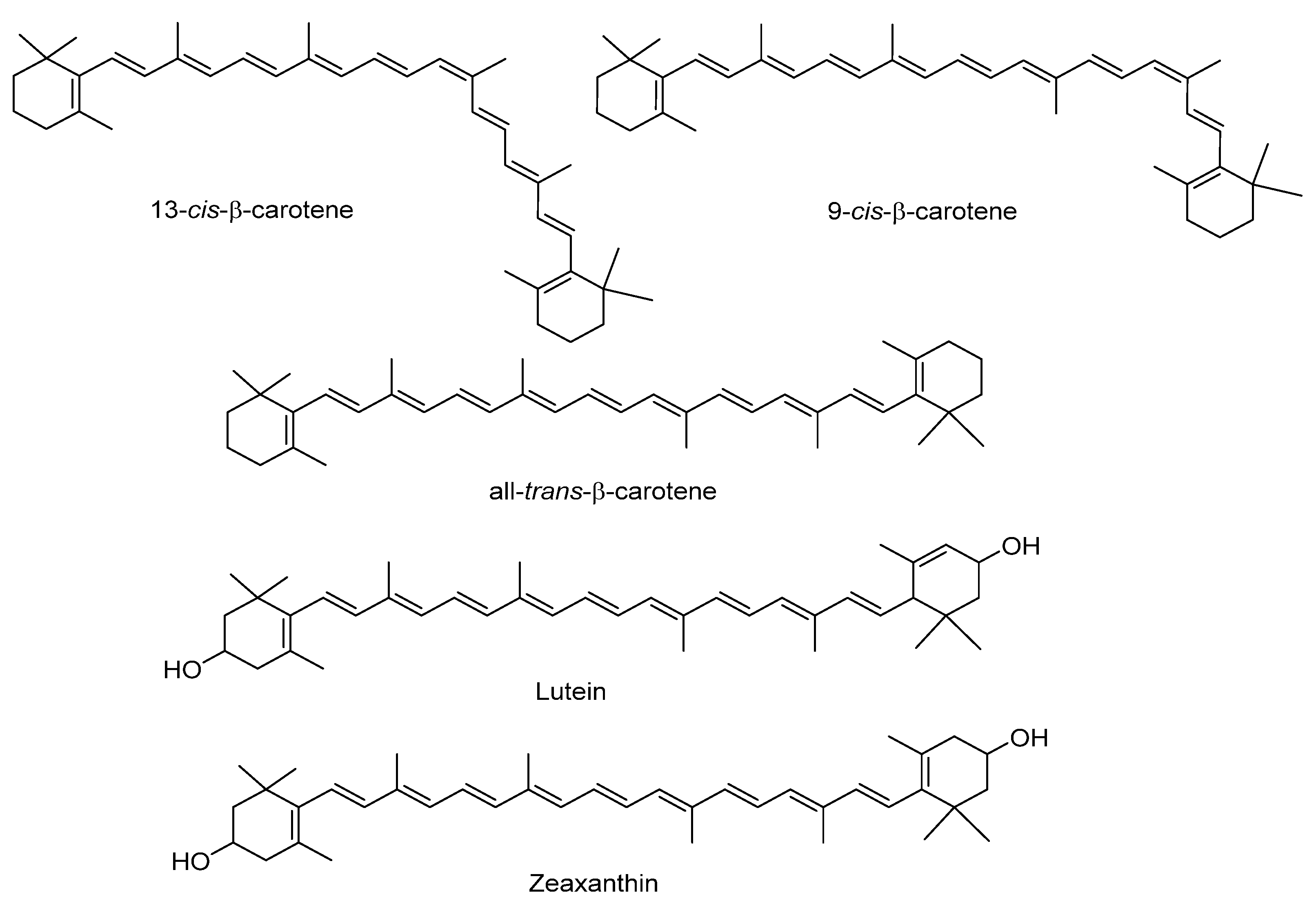
Sweet potato tubers are an excellent source of carotenoids, which contribute to their yellow and orange color (Figure 4). The total carotenoid content differs significantly according to the potato color and variety (Table 4 and Table 5). β-carotene is the most abundant in those with orange flesh (more than 99% of total carotenoid content), whereas trans-β-carotene predominates in those with white and purple flesh. In orange varieties, Vimala et al. (2011) reported 5.9–12.9 mg/100 g of β-carotene [49], and Alam et al. (2016), 0.38–7.38 mg/100 g [13]. Lutein and zeaxanthin have been identified as minor carotenoids in sweet potatoes, with reported ranges of 0.1–0.4 and 0.1–0.2 mg/100 g dry weight, respectively [19,27]. According to the results of Park et al. (2016), lutein and zeaxanthin levels are higher in potatoes with purple versus white flesh [27].
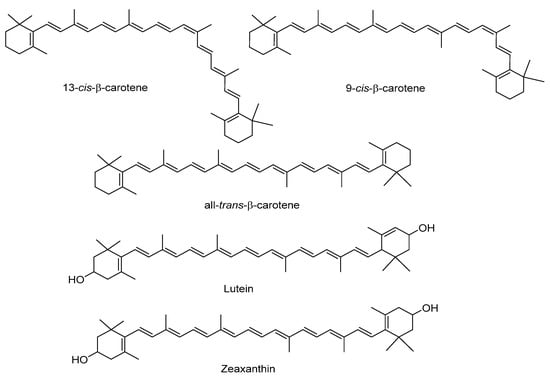
Figure 4.
Main carotenoids identified in sweet potato.
2.3. Other Phytochemicals
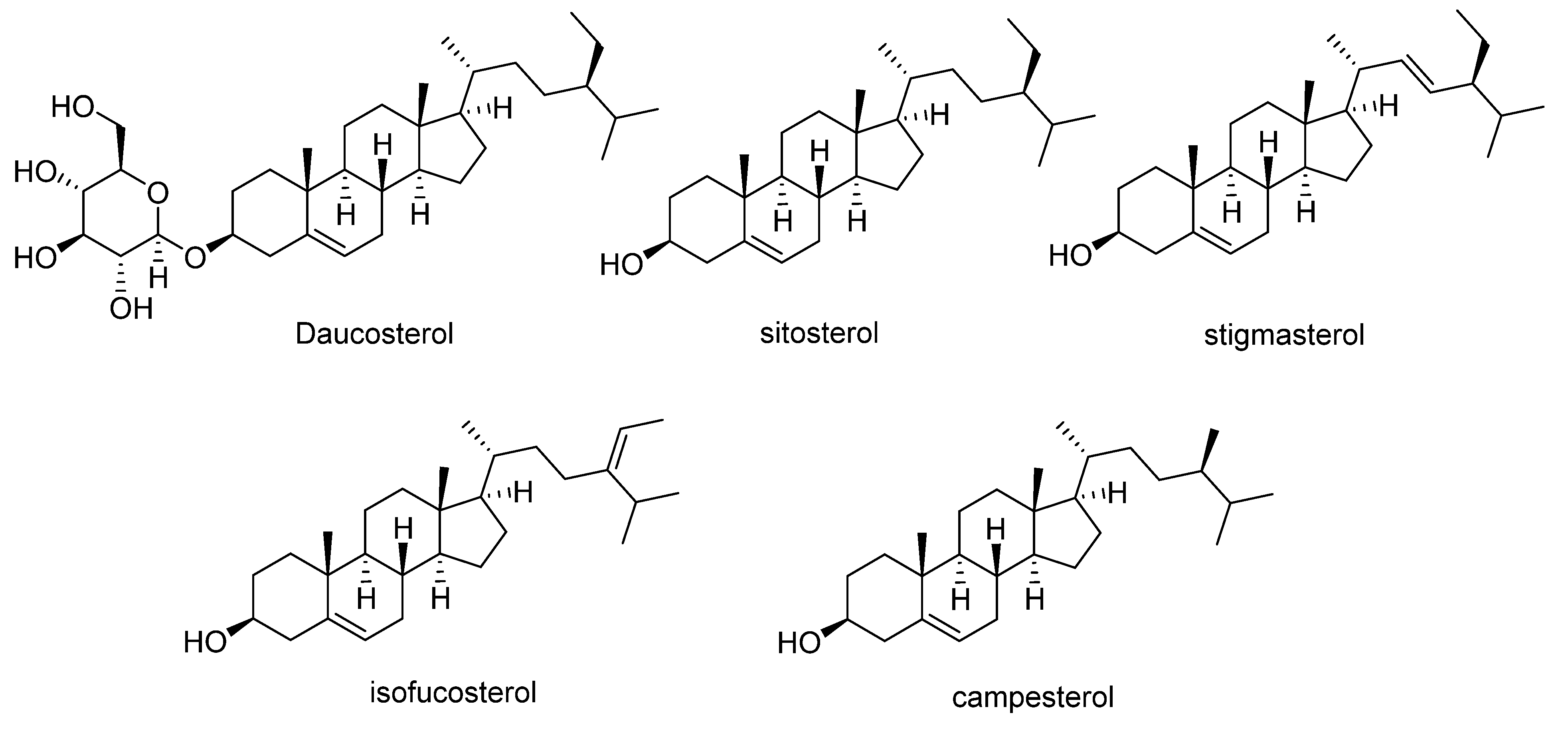
Orange-fleshed sweet potatoes also contain phytosterols (Figure 5). Daucosterol has been reported at concentrations ranging from 0.01 to 0.5 mg/g dry weight [50], whereas β-sitosterol, the most abundant phytosterol in sweet potato (55 to 78% of total phytosterols), is found at ranges of 122–358 mg/kg, followed by campesterol (35–101 mg/kg), stigmasterol (5–58 mg/kg), and isofucosterol (5–23 mg/kg) (Table 4) [51].
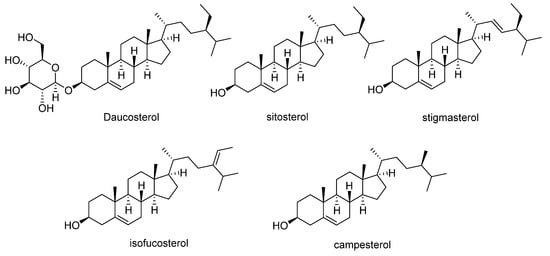
Figure 5.
Main phytosterols identified in sweet potato.

Table 4.
Carotenoid, phytosterol, and other phytochemical contents of orange sweet potato flesh.
Table 4.
Carotenoid, phytosterol, and other phytochemical contents of orange sweet potato flesh.
| Origin | Sample Extraction | Analythical Method | Phytochemical | Amount of Phytochemical | Ref. |
|---|---|---|---|---|---|
| Carotenoids | |||||
| USA | Hexane-acetone (1:1) | Reverse-phase HPLC | β-Carotene | ~122.0 µg/g FW, ~18.2 µg/g FW (light-orange) | [9] |
| Bangladesh | Hexane-acetone (1:1) | Spectrophotometry | TC | 0.38 to 7.24 µ * | [13] |
| Reverse-phase HPLC | TC | 19.3 to 61.9 µ * | |||
| trans-β-Carotene | 76.6 to 96.5 µ * | ||||
| cis-β-Carotene: | 3.5 to 23.4 µ * | [52] | |||
| Brazil | Acetone-petroleum ether. Petroleum ether: diethyl ether (1:1) | Reverse-phase HPLC | trans-β-Carotene | Raw: 79.1 to 128.5 mg *, boiled: 68.9 to 133.3 mg *, roasted: 64.6 to 127.0 mg *, steamed: 69.4 to 131.0 mg *, flour: 45.4 to 79.7 mg * | [19] |
| 13-cis-β-Carotene | Raw: 9.3 to 9.6 mg *, boiled: 4.3 to 8.6 mg *, roasted: 4.3 to 11.1 mg *, steamed: 7.1 to 8.5 mg *, flour: 2.7 to 4.7 mg * | ||||
| 9-cis-β-Carotene | Raw: 4.9 to 6.1 mg *, boiled: 3.9 to 6.0 mg *, roasted: 3.8 to 5.5 mg *, steamed: 5.2 to 7.4 mg *, flour: 1.5 to 2.1 mg * | ||||
| 5,6-Eepoxy-β-carotene | Raw: 7.0 to 11.3 mg *, boiled: 7.8 to 13.1 mg *, roasted: 7.0 to 9.6 mg *, steamed: 7.6 to 15.4 mg *, flour: 3.8 to 6.5 mg * | ||||
| Lutein | Raw: 0.1 to 0.4 mg *, boiled: 0.2 to 0.4 mg *, roasted: 0.1 to 0.6 mg *, steamed: 0.1 to 1.1 mg *, flour: 0.1 to 0.3 mg * | ||||
| Zeaxanthin | Raw: 0.1 to 0.2 mg *, boiled: 0.1 to 0.3 mg *, roasted: 0.1 to 0.2 mg *, steamed: 0.1 to 0.6 mg *, flour: 0.1 to 0.2 mg * | ||||
| Kenya | Methanol and tetrahydrofuran | Reverse-phase HPLC | Lutein | 0.01 to 0.1 mg * | [18,25] |
| Zeaxanthin | 0.02 to 0.5 mg * | ||||
| β-Xanthin | 0.1 to 0.5 mg * | ||||
| 13-cis-β-Carotene | 0.05 to 0.4 mg * | ||||
| All trans β-Carotene | 2.6 to 18.2 mg * | ||||
| β-9-cis-β-Carotene | 0.05 to 0.4 mg * | ||||
| Korea | Ethanol (0.1% ascorbic acid) | HPLC system | TC | 93.4 µg ** | |
| Lutein | 0.15 µg ** | [27] | |||
| α-Carotene | 0.44 µg ** | ||||
| (all E)-β-Carotene | 68.74 µg ** | ||||
| (9Z)-β-Carotene | 1.45 µg ** | ||||
| (13Z)-β-Carotene | 22.64 µg ** | ||||
| India | Hexane-acetone (6:4) | HPLC | TC | 7.47 to 15.47 mg/100 g FW | |
| β-Carotene | 5.85 to 13.63 mg/100 g FW | [46] | |||
| Phytosterols | |||||
| China | Ethanol (70%) | HPLC system | Daucosterol linolenate | 0.05 to 0.2 mg ** | [50] |
| Acetone-petroleum ether, ethyl acetate, and n-butanol (1:1) | Daucosterol linoleate | 0.2 to 0.5 mg ** | |||
| Daucosterol palmitate | 0.3 to 0.6 mg ** | ||||
| Other phytochemicals | |||||
| Kenya | Water | UV spectrophotometry | Tannic acid | 0.04 to 0.13 g * | [25] |
| Water | HPLC | Oxalic acid | 0.003 to 0.132 g * | ||
| NS | ELISA kit | Phytic acid | 0.05 to 0.42 g * | ||
HPLC: high-performance liquid chromatography; DW: dry weight; FW: Fresh weight; TC: total carotenoids; * Results expressed per 100 g DW, ** Results expressed per 1 g DW.

Table 5.
Carotenoid content of purple, yellow, and white sweet potato flesh.
Table 5.
Carotenoid content of purple, yellow, and white sweet potato flesh.
| Color of Sweet Potato Flesh (Origin) | Sample Extraction | Analythical Method | Phytochemical | Amount of Phytochemical | Ref. |
|---|---|---|---|---|---|
| Purple (USA) | Hexane–acetone (1:1) | Reverse-phase HPLC | β-Carotene | ~22.5 µg/g FW, ~50.6 µg/g FW (light purple) | [9] |
| Purple (Korea) | Ethanol (0.1% ascorbic acid) | HPLC system | TC | −2.22 µg ** | [27] |
| Lutein | −0.28 µg ** | ||||
| Zeaxanthin | −0.11 µg ** | ||||
| (all E)-β-Carotene | −1.53 µg ** | ||||
| (9Z)-β-Carotene | −0.02 µg ** | ||||
| (13Z)-β-Carotene | −0.28 µg ** | ||||
| Yellow (USA) | Hexane–acetone (1:1) | Reverse-phase HPLC | β-Carotene | ~−1.9 µg/g FW | [9] |
| Yellowish cream (Bangladesh) | Acetone:petroleum ether | Spectrophotometry Reverse pase HPLC | TC | −3.3 to 5.6 µ * | [52] |
| trans--β-Carotene | −83.6 to 84.3 µ * | ||||
| cis--β-Carotene: | −13.4 to 15.7 µ * | ||||
| White (USA) | Hexane–acetone (1:1) | Reverse-phase HPLC | β-Carotene | −0.2 µg/g FW | [9] |
| White (Bangladesh) | Acetone:petroleum ether | Spectrophotometry Reverse pase HPLC | TC | −1.0 µ * | [52] |
| White (Korea) | Ethanol (0.1% ascorbic acid) | HPLC system | TC | −1.37 µg ** | [27] |
| Lutein | −0.27 µg ** | ||||
| Zeaxanthin | −0.03 µg ** | ||||
| α-Carotene | −0.01 µg ** | ||||
| (all E)-β-Carotene | −0.83 µg ** | ||||
| (9Z)-β-Carotene | −0.09 µg ** | ||||
| (13Z)-β-Carotene | −0.14 µg ** |
HPLC: high-performance liquid chromatography; DW: dry weight; FW: Fresh weight; TC: total carotenoids; * Results expressed per 100 g DW, ** Results expressed per 1 g DW.
3. Beneficial Health Effects of Sweet Potatoes
Traditionally, sweet potatoes have been used as an important source of carbohydrates and energy for both human beings and livestock because of their high content of starch [53]. Nowadays, sweet potatoes are recognized as a highly nutritious and useful food for the prevention of chronic diseases, mainly due to its content of dietary fiber, naturally occurring sugars, protein content, vitamins A and C, potassium, iron, and calcium, and its low amount of fat (mainly saturated fat), sodium, and cholesterol [54].
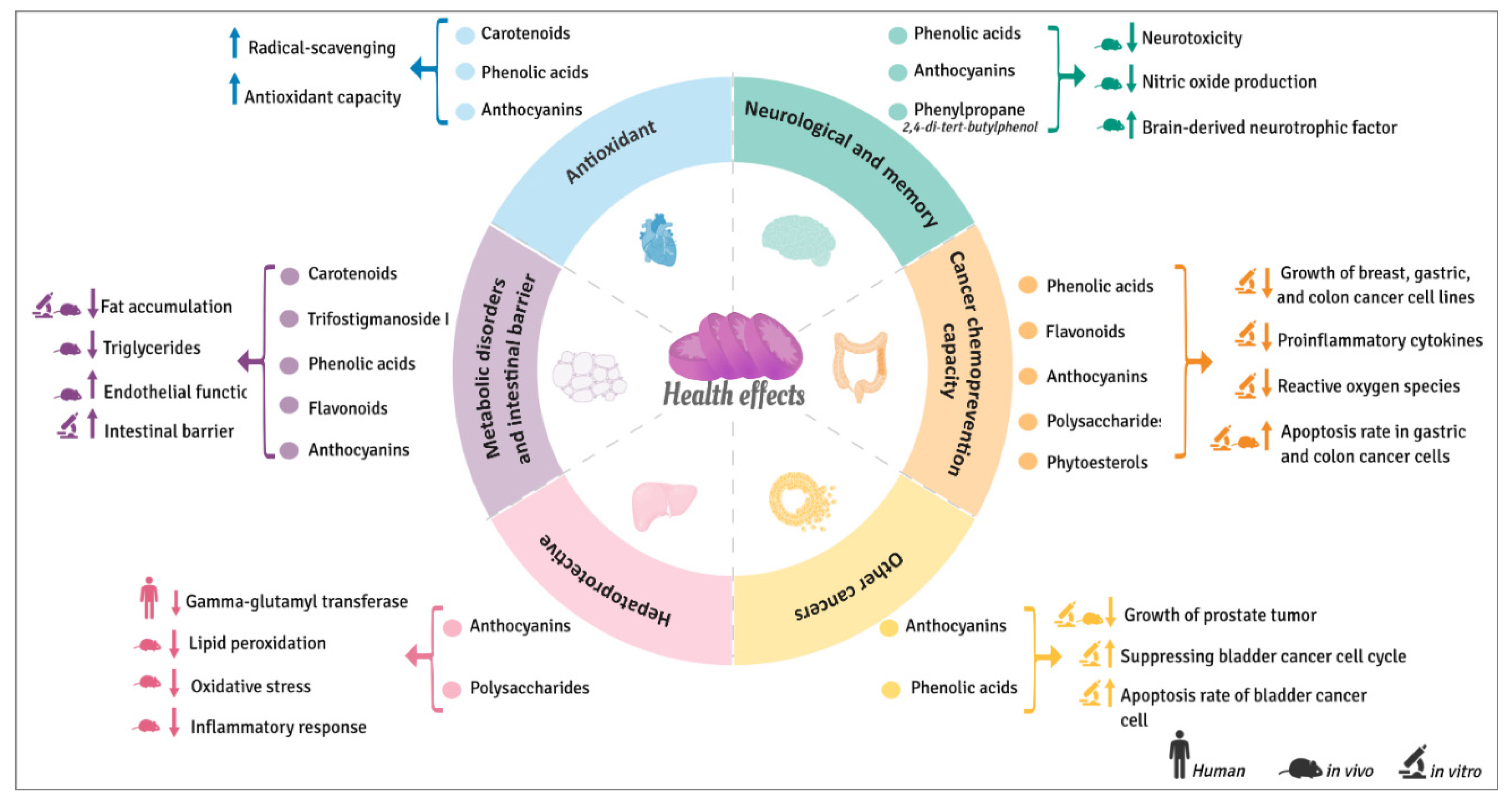
The regular consumption of sweet potatoes or extracts rich in their bioactive phytochemicals seems to also be beneficial for human health as evidenced by The Okinawan Diet [54]. However, most of the positive health effects reported until now (Figure 6), which will be explained in detail in the following sections, are based on in vitro studies and just a few of them in animal models. So far only one human trial has been carried out.
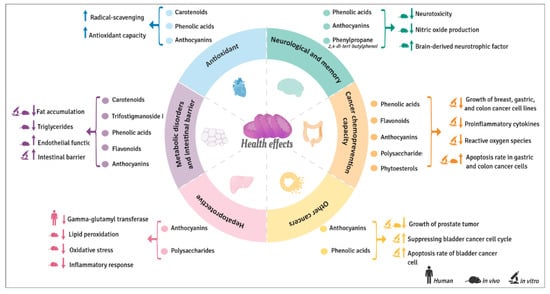
Figure 6.
Beneficial health effects of bioactive compounds from sweet potatoes.
3.1. Antioxidant Properties
The potent antioxidant capacity of purple sweet potatoes is mainly attributed to their anthocyanin and carotenoid content. These compounds exhibit free radical scavenging activity, and consequently, the consumption of anthocyanin-rich products is associated with a lower risk of diabetes cardiovascular disease, cancer, and cognitive performance [55]. Anthocyanins from purple sweet potato showed stronger 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging capacity than those from red cabbage, grape skin, elderberry, or purple corn, and some were also more active than either vitamin C or vitamin E [56,57].
The overall antioxidant capacity of white-fleshed cultivars was attributed to their high content of phenolic acids and carotenoids, and it was concluded that their consumption might protect the human body from oxidative stress [46]. Rather than anthocyanins, chlorogenic acid was the main DPPH radical scavenger in extracts of purple-fleshed varieties “Miyanou-36” and “Bise” [28]. On the other hand, in yellow or orange sweet potatoes, the carotenoid β-carotene, which is the main pigment, has a strong antioxidant capacity due to its conjugated double bonds [58] and acts as a source of provitamin A [8].
3.2. Hepatoprotective Effects
To date, most of the studies on hepatoprotective effects have been carried out using animal models and only one study has been carried out in human studies. In addition, it should be noted that all these studies have focused on evaluating the protective effect of anthocyanins from sweet potatoes.
The anthocyanins offer protection from injury induced by hepatotoxins mainly by inhibition of lipid peroxidation and scavenging free radicals. In healthy men with borderline hepatitis, purple sweet potato beverages significantly decreased the serum levels of some liver enzymes, particularly gamma-glutamyl transferase (GGT) [59].
In animal models, a protective effect against CCl4-induced acute liver damage has been extensively reported after the intake of anthocyanin-rich purple sweet potato extract [57,60,61], as well as sweet potato polysaccharides [62]. Inhibition of lipid peroxidation was also observed in male rats fed a high-cholesterol diet after the administration of anthocyanin-rich sweet potato flakes [63] and in the acetaminophen-induced hepatotoxicity mouse model [64]. Purple sweet potato anthocyanin (PSPA) attenuated the oxidative stress and inflammatory response induced by D-galactose in mouse liver [65], whereas an extract from Shinzami, a variety of purple sweet potato, prevented ischaemia–reperfusion-induced liver damage in rats by improvement of the antioxidant status [66].
The fact that the protective effect has been demonstrated in animal studies indicates that human studies are needed to show whether there is strong evidence for the hepatoprotective effects of anthocyanins in purple sweet potato.
3.3. Cognitive and Memory Improvement
The bioactive compounds from purple sweet potato exhibit memory-enhancing effects. According to Isoda et al. (2010, 2013), caffeoylquinic acid-rich purple sweet potato extracts improved spatial learning and memory in a mouse model of aging [67,68]. In addition, D-galactose-induced impairment of memory and spatial learning was repaired through the regulation of synaptic protein expression in the hippocampus and cerebral cortex of mice by different PSPA treatments [69]. Protection against Aβ-induced neurotoxicity by caffeoylquinic acids has also been reported in a mouse model [70,71,72]. Similarly, in mice injected with Aβ₁₋₄₂, attenuation of cognitive dysfunction and neuronal cell damage was observed after the administration of 2,4-di-tert-butylphenol extracts from sweet potatoes [73].
A neuroprotective effect of purple sweet potato anthocyanins in a Wistar rat model with ischemic stroke was reported by Adnyana et al. (2018, 2019), which was attributed to the inhibition of damaging effects of reactive oxygen species (ROS) [74]. The treated rats showed an enhanced neurological score between day-3 and day-7 post-stroke, an increase in the brain-derived neurotrophic factor level, and a reduced apoptosis rate [75].
Cognitive deterioration is also associated with obesity, a growing public health concern. In this context, purple sweet potato anthocyanins were observed to improve cognitive function in high-fat diet-fed mice via the activation of AMP-activated protein kinase, which protects against hippocampal apoptosis by restoring impaired autophagy [76]. The protective role of this pigment in high-fat diet-associated neuroinflammation in the mouse brain was also examined by Li (2018), who found significant improvement in impaired memory function and behavior, as well as suppression of the increment in body weight, hyperlipemia, fat content, and endotoxin levels [77].
In summary, numerous studies have been carried out attempting to explain the neuroprotective effect of sweet potato. However, all studies have only been conducted in animals, which leaves the need to confirm this protective effect in human studies. In addition, future studies are needed to evaluate the brain-protective effect of other sweet potato varieties (yellow- and orange-fleshed sweet potatoes).
3.4. In Vitro and In-Vivo Cancer Chemoprevention Capacity
Most of the studies on protective cancer effects derived from sweet potato consumption have been carried out using in-vitro and animal models. Further research is needed in this field, especially in randomized clinical trials in humans.
Extracts of purple-fleshed sweet potato Tainung 73 (PFSP TNG 73) are reported to have in vitro anti-inflammatory and anticancer activities, attributed to a high content of antioxidative compounds, including phenolics, flavonoids, and the pigment anthocyanin [78]. Anthocyanin-rich extracts of PFSP TNG 73 suppressed the production of nitric oxide and some proinflammatory cytokines, such as NF𝜅-𝛽, TNF-𝛼, and IL-6, in macrophage cells stimulated by lipopolysaccharides. They also inhibited the growth of some in vitro cancer cell lines, including human breast cancer (MCF-7), gastric cancer (SNU-1), and colon adenocarcinoma (WiDr), in a concentration- and time-dependent manner. Additionally, PFSP TNG 73 extracts induced apoptosis in MFC-7 cells. Thus, this variety of sweet potato is a source of metabolites with potential application in the development of drugs, nutritional foods, and health supplements.
Purple sweet potatoes also contain polysaccharides with promising antitumor properties. Three beta-type polysaccharides, PSPP1-1, PSPP2-1, and PSPP3, with low amounts of proteins and uronic acids, were isolated from crude purple sweet potato polysaccharides. In an in vitro antitumor assay, PSPP1-1 exhibited strong activities against gastric cancer SGC7901 cells and colon cancer SW620 cells, whereas PSPP2-1 and PSPP3-1 had moderate activities. Furthermore, PSPP1-1 was found to induce apoptosis in both types of cancer cells [6].
3.4.1. Breast Cancer
Breast cancer is a prominent cause of mortality in women throughout the world. In a study by Kato et al. [79] using E0771 murine breast cancer cells, lipid-soluble polyphenols (mainly caffeic acid derivatives) from fermented sweet potato were found to accumulate in cell cytoplasm due to their high lipophilicity and reduce ROS through their strong antioxidant activity. These metabolites also arrested the cell cycle at G0/G1 by suppressing Akt activity and enhancing the cytotoxicity of anti-cancer agents. Thus, lipid-soluble polyphenols from sweet potatoes inhibited tumor growth and improved the efficacy of chemotherapy drugs, suggesting they have application as a functional food to support cancer therapy.
Another study revealed that three phytosterols from sweet potato, daucosterol linolenate (DLA), daucosterol linoleate (DL), and daucosterol palmitate (DP), had a stronger inhibitory effect against the MCF-7 than the MDA-MB-231 breast cancer cell line, and had no impact on non-tumorigenic MCF-10A cells [50]. In vivo experiments demonstrated that DLA, DL, and DP suppressed MCF-7 xenografts in nude mice. In another study, sitosterol-d-glucoside (β-SDG), a recently isolated phytosterol from sweet potato, also displayed potent anticancer activity [80] against MCF7 and MDA-MB-231 cell lines and suppressed the growth of MCF7 xenografts in nude mice. This effect of β-SDG was due to the up-regulation of miR-10a expression and inactivation of the PI3K–Akt signaling pathway.
A component of the new sweet potato variety Zhongshu NO. 1, the glycoprotein SPG-56, was reported to inhibit proliferation and promote apoptosis of MCF-7 cells in mice in a dose- and time-dependent manner [81]. The serum tumor markers CEA, CA125, and CA153 were reduced by 54.8%, 91.8%, and 90.3%, respectively, in mice orally administered 240 mg/kg/d of SPG-56, with a significant difference (p < 0.01) compared with the untreated control. The inhibitory effect of SPG-56 against MCF-7 cells was found to be mediated by the altered expression of specific genes. It was concluded that SPG-56 merits further research as a novel anti-tumor agent for breast cancer treatment.
3.4.2. Colon Cancer
Colon cancer is responsible for a high proportion of cancer mortality throughout the world. A new small molecule, glycoprotein SPG-8700, isolated from Zhongshu-1 sweet potatoes [82] was found to promote apoptosis in HCT-116 colon cancer cells by regulating the expression of Bcl-2 and Bax genes, with no effect on normal cell growth. Sporamin, another molecule isolated from sweet potato, has promising effects against colorectal cancer in vitro and in vivo [83]. This proteinase inhibitor was able to modify the gene expression profile of colon cancer cells, up-regulating genes involved in the homeostasis of intracellular metal ions and the activities of essential enzymes and DNA damage repair.
A study by Lim et al. [84] on the anthocyanin-enriched sweet potato (clone P40) found it offered protection against colorectal cancer by inducing cell cycle arrest, inhibiting proliferation, and apoptotic mechanisms. The anticancer activity of this clone was demonstrated in both in vitro cell culture and an in vivo animal model. Treatment of human colon SW480 cancer cells with a P40 anthocyanin extract resulted in a dose–dependent inhibition of cell proliferation due to a cytostatic but not cytotoxic mechanism.
3.4.3. Other Cancers
Phenolic phytochemicals present in fruits and vegetables indisputably confer anticancer benefits upon regular consumption. The protective effect of PSPA and polyphenol-rich sweet potato extract in other types of cancer has been assessed in studies with cell culture and in vivo.
PSPA has been shown to have in vitro antitumor effects in bladder cancer, a common malignant disease. Li et al. [85] reported that PSPA reduced bladder cancer cell viability in a dose-dependent manner, increasing the apoptosis rate and suppressing the cell cycle. The mechanism of action underlying the anticancer effects of PSPA includes upregulation of pro-apoptosis genes and a lower expression of the anti-apoptotic gene Bcl-2. Thus, the results of this study provide new insights into the treatment of bladder cancer and the potential role that PSPA plays in cancer prevention.
The effect of PSPA on the bladder cancer cell line BIU87 was also investigated [85]. Compared with the control, the proliferation of BIU87 cells was significantly inhibited in groups treated with this phenolic compound, which induced cell apoptosis in a dose-dependent manner.
The growth-inhibitory and apoptosis-inducing properties of polyphenol-rich sweet potato extract were recently demonstrated in cell culture and in vivo prostate cancer xenograft models [86]. Thus, the extract is a candidate for use as a dietary supplement for prostate cancer management. Despite the growth and apoptosis inhibitory properties of phenolic compounds, future studies should be carried out in humans to support the findings detected in vitro.
3.5. Metabolic Disorders and Intestinal Barrier Function
Anthocyanin and carotenoid-rich extracts from purple and orange-fleshed sweet potatoes, respectively, may be useful as supplementary ingredients for the treatment of obesity and related diseases. Their anti-obesity effects were studied both in vitro (3T3-L1 cells) and in vivo (high-fat diet-induced obese mice) [87]. Both extracts exhibited the potential to inhibit fat accumulation in adipocytes, reduce weight gain, and restore triglyceride levels to normal with an improvement in the ratio between triglyceride and high-density lipoprotein cholesterol, a cardio-metabolic biomarker that predicts a higher risk of heart disease and arteriosclerosis.
Although full evidence from clinical trials is still lacking, and the long-term effects have not been studied yet, sweet potato has proved effective in treating hyperglycemia, as concluded in a recent systematic review based on in vitro and in vivo studies [88]. Cardiovascular disorders are one of the most important causes of mortality in type 2 diabetes mellitus, and atherosclerosis is the major cardiovascular complication related to diabetes [89]. In fact, the endothelial dysfunction found in diabetes has been established as the initial point in the atherosclerotic process [90]. Dietary flavonoids from sweet potatoes were found to attenuate atherosclerosis in mice by alleviating inflammation, inhibiting platelet aggregation, and reducing LDL cholesterol, which improved endothelial function [91]. More data were provided by another study, which reported that flavonoids isolated from purple sweet potato protected against endothelium dysfunction in type 2 diabetes mellitus by reducing endothelial premature senescence and atherogenesis via suppressing ROS levels and the NLRP3 inflammasome [92].
Recently, the sesquiterpene trifostigmanoside I (TS I) was identified as the main compound responsible for the protective effect of sweet potato extracts on intestinal barrier function [5]. Through PKCα/β-ERK1/2 signaling, TS I induced the production of mucin (MUC2), a polypeptide secreted by specialized epithelial cells that forms a protective hydrated gel over mucosal surfaces, and partially protected tight junctions, both effects helping to maintain the intestinal barrier function. More research is needed on other phytochemicals in sweet potato to determine their effect on the regulation of multiple signaling pathways involved in gastrointestinal disorders.
4. Effect of Sweet-Potato Processing on Phenolic Compounds
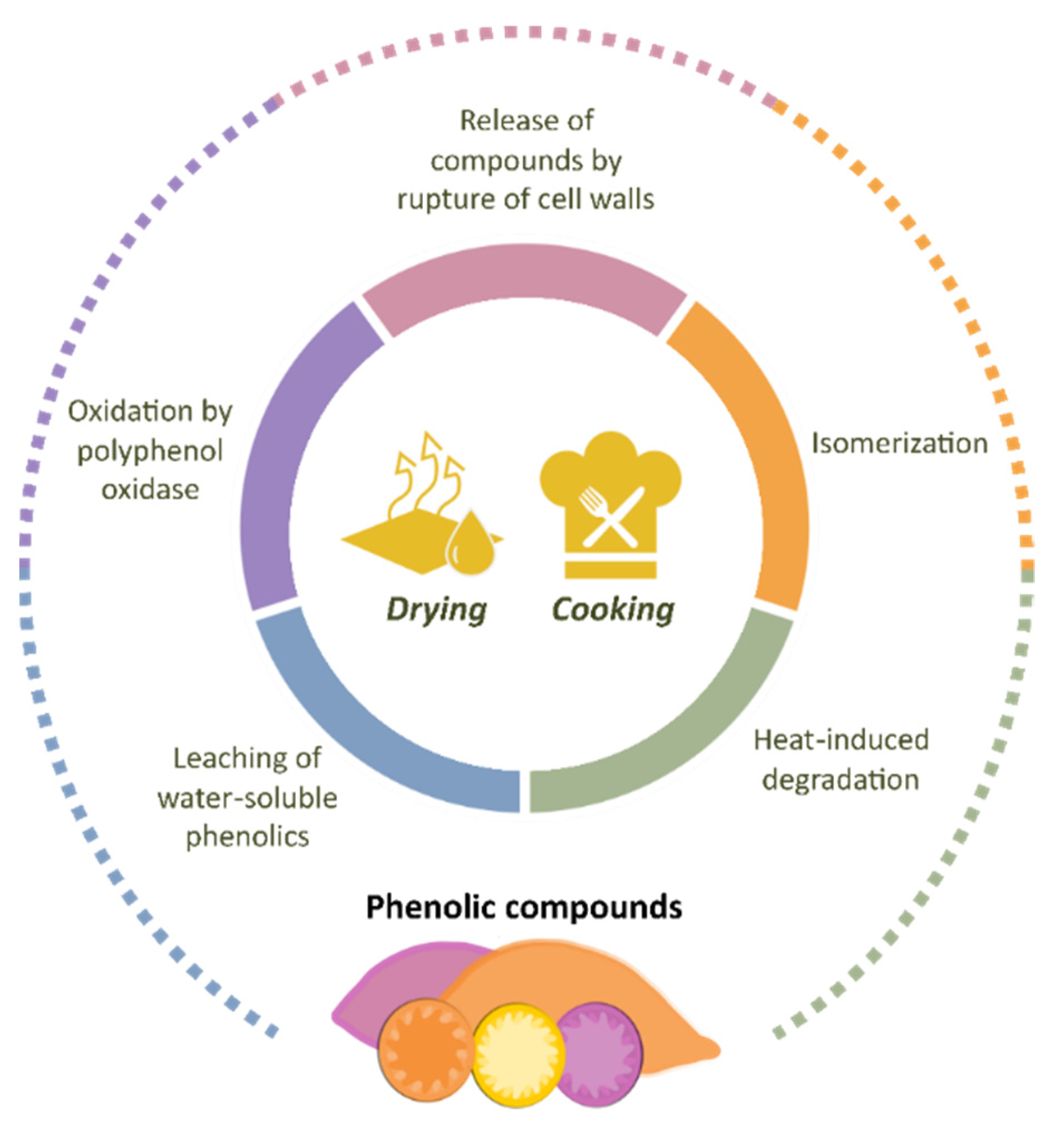
Sweet potato bioactive compounds have attracted the interest of researchers in the field of human nutrition and the agro-food sector. Unfortunately, the fresh tubers are highly perishable and difficult to conserve due to their high moisture content, sustained metabolism, and microbial attack [93]. The resulting losses generate extra costs for farmers, traders, consumers, and other stakeholders in the sweet potato value chain [18]. Aiming to exploit the economic and nutritional value of this crop more effectively, researchers have joined efforts to explore the impact of different drying and cooking methods on the nutritional composition and physicochemical properties of sweet potatoes. The chemical profile of foods can be affected by several factors, ranging from agronomic practices to processing and storage conditions [94,95]. As phenolic compounds are important constituents of a healthy human diet, their retention in food is desirable from the nutritional point of view. The main findings in terms of phenolic composition are presented in the following sections and in Figure 7.
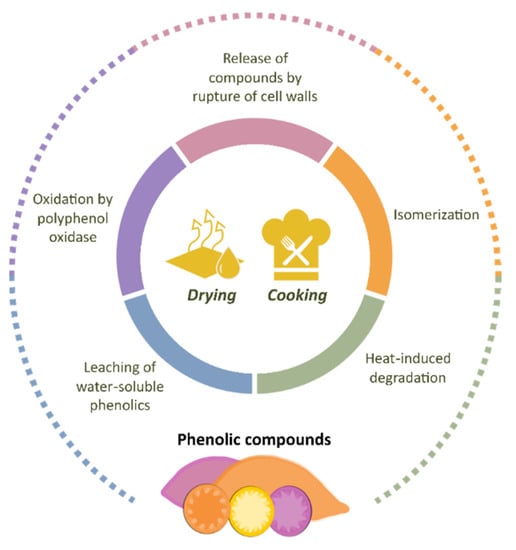
Figure 7.
Main factors affecting the phenolic profile in sweet potato during processing.
4.1. Drying Treatments
The drying of foods is a hugely important technique for the food industry, as it allows the shelf life of fresh food to be extended and the development of new ingredients and products [96,97]. However, improper drying methods can cause a decline in product quality, including color deterioration, malformation, and loss of nutrients [96].
Convective hot-air and sun-drying are among the methods commonly used to preserve starch-containing products, as they inhibit enzyme activity, prevent microbiological spoilage, and delay decay [93]. However, in addition to being energy- and time-consuming, these processes can also induce loss of nutrients and change the physical and chemical characteristics of the final products [93].
4.1.1. Hot-Air Drying, Microwave Drying, and Vacuum-Freeze Drying
Different drying methods (hot-air, microwave, and vacuum-freeze) were tested in sweet potato tubers to determine their effects on the TPC. In dried sweet potatoes, the TPC was found to be higher than in the fresh tuber, increasing by 116–225%. Microwave-dried samples retained the highest TPC [98], which could be due to the enhanced release of bound phenolics by the heat-induced breakdown of cellular constituents and/or changes in the water content. The increase in phenolic content could also be explained, at least partially, by the formation of Maillard reaction products with a phenolic-type structure during the thermal process [98]. A recent study carried out by Savas [99] compared the effect of freeze-drying and convective drying on the TPC, total flavonoids, and total anthocyanins in sliced sweet potatoes. The TPC and total flavonoid content in freeze-dried samples, and total anthocyanin content in convectively dried samples were clearly higher than in fresh tubers. After the optimization of variables in the convective drying process, this method led to an increase in both types of functional compounds.
4.1.2. Spray Drying
One of the strategies aimed at promoting the consumption of sweet potatoes is the conversion of sweet potato puree into dry powder for use as a functional ingredient in food systems [100]. Freeze-drying and spray drying are commonly used to obtain sweet potato powders. Spray drying, one of the most rapid and least expensive procedures, involves spraying the food material into a chamber where hot dry air rapidly causes the small droplets to evaporate, leaving the spray-dried particles [101]. However, the high temperatures used during the process reduces the (poly)phenol content. In a recent study on purple-fleshed sweet potato, spray drying was found to negatively affect different phenolic groups (anthocyanins and cinnamoylquinic acids) with total (poly)phenol losses representing around 90% of the initial concentrations [102]. The least affected compounds were mono-cinnamoylquinic acids with feruloyl moieties and mono-acylated peonidin derivatives with p-hydroxybenzoic acid.
Flour, in addition to being obtained by methods such as hot air-drying, freeze-drying, and microwave-drying, can also be produced by spray-drying. The use of carriers (polysaccharides, proteins, and lipids) was tested as a strategy to improve the properties of spray-dried sweet potato flour [103]. The results showed that apart from increasing flour yields, the use of maltodextrins as a carrier led to a higher retention rate of anthocyanins, flavonoids, and total phenolics compared to flours produced without or with other carriers [103].
4.2. Pretreatments
As well as being more stable during storage, dried processed foods can be healthy and nutritious if prepared with the appropriate technologies [104]. Pretreatments before drying and processing can avoid nutrient losses and change the physical and chemical characteristics of the final products, even improving the nutritional value of the sweet potato. Ultrasound (US) [104] and vacuum impregnation (VI) [105,106] are the most frequently applied pretreatments for sweet potatoes.
4.2.1. Ultrasound
Ultrasound (US) technology is based on acoustic vibration, during which the food undergoes rapid compressions and expansions, the so-called “sponge effect”. This results in alterations in surface tension and viscosity, cell wall disruption, the formation of microscopic channels, and denaturation of enzymes [107,108]. These changes can increase the energy transfer rate in the drying procedure, thereby shortening it, and maintain or improve the product quality [96,109].
Despite the importance of phenolic compounds, to date, little research has focused on evaluating the effect of US pretreatment on the phenolic profile of sweet potatoes. One study concluded that pretreatment with US does not alter antioxidant activity (determined by the DPPH method) [110]. The effect of combining US and other pretreatments with drying methods (hot air, microwave vacuum, and freeze) on the stability of quality attributes of sweet potato slices were studied [104]. Compared with the unprocessed samples, the TPC in processed potatoes was found to be lower after 90 days of storage at room temperature. However, it is notable that the reduction in phenolic content was higher in the samples without pre-treatment (76%) than in pretreated (blanching + US in distilled water + hot air drying + microwave vacuum or blanching + US-assisted osmotic dehydration + hot air drying + microwave vacuum) samples (46 and 33%, respectively).
Finally, another study demonstrated that the TPC and total flavonoid content in US-assisted dry sweet potatoes increased compared (15 and 87%, respectively) to control samples when the lowest drying temperature (70 °C) was used [111]. Thus, although US application can accelerate the drying process and maintain the quality of dried products, its positive effect depends on the drying method applied.
4.2.2. Vacuum Impregnation
Vacuum impregnation (VI) is a non-destructive treatment that takes advantage of the highly porous nature of most foods (i.e., vegetables, fruits, meats, cheese, etc.). Thus, the main purpose of VI is to add substances of interest to porous foods, replacing the native liquids or gases [105,112,113]. Overall, VI may be used to achieve qualitative, technological, or nutritional functionality [105,113]. One of its benefits is that it permits the preparation of a fortified functional food without subjecting the product to a high temperature [113]. VI is used as a pretreatment before drying, freezing, and frying foods to preserve the color, natural flavor, aroma, and heat-sensitive nutrient components [113]. To date, only two studies have evaluated the use of VI in sweet potatoes, with the aim of polyphenol fortification.
Using VI technology (25 min at 20 °C), Abalos et al. (2020) [105] produced sweet potato slices with a 473% increase in phenolic concentration compared to the control. In this study, a commercial solution of polyphenol extract (95% [v/v] proanthocyanidins) was used as an impregnation medium. The influence of cooking (steam, cook vide, and sous vide) on the concentration of polyphenols in VI pre-treated samples was also compared, with sous vide causing lower losses than cooking under normal atmospheric pressure.
In the second study [106], sweet potatoes were impregnated with freshly squeezed onion juice, kale juice, an onion–kale mixture (1:1), or sodium chloride solution. The samples were then dried using vacuum drying and freeze-drying methods. The pretreatment with VI increased the sweet potato content of polyphenols, carotenoids, chlorophyll, and volatile organic compounds, and improved their antioxidant capacity.
According to the studies carried out so far, VI pretreatment can increase the phenolic content of sweet potatoes, although the final concentration depends on the subsequent method of drying or cooking.
4.3. Cooking Techniques
Sweet potatoes can be cooked by various methods including boiling, steaming, roasting, microwave roasting, and baking. During these processes, the phenolic compounds in sweet potato are susceptible to changes [114], attributed mainly to (i) degradation by heat, (ii) oxidation by polyphenol oxidase, (iii) leaching of water-soluble phenolics, and (iv) isomerization and/or release of compounds by rupture of cell walls [1,19,114,115]. The relation between the type of cooking method and the final phenolic concentration in sweet potatoes has been the subject of various studies [1,19,115,116,117,118,119].
The TPC in sweet potato can be increased by cooking. Roasting, for example, resulted in significantly higher levels compared to the raw vegetable (5.42 mg and 3.34 mg chlorogenic acid equivalent/g DW, respectively) [118]. Other studies have found that steaming, microwaving, baking, and boiling also enhance the TPC [119,120]. After boiling and frying, phenolic compounds were retained in orange-fleshed sweet potatoes [18,121], whereas steam-cooking drastically increased the TPC in purple sweet potatoes (about four-fold) compared to raw tubers [115]. This could be due to the softening or disruption of plant cell walls or the breakdown of complex compounds during thermal treatment, which facilitates the release of phenolic compounds [118,120].
Other studies have compared the effect of different cooking methods without including a control (raw sweet potato). In a comparison [1] of domestic processing techniques (baking, boiling, frying, microwaving, sautéing, and steaming), the TPC was least affected by boiling, whereas the biggest reductions were induced by deep frying, followed by microwaving. In purple-fleshed sweet potato, baking and steaming did not influence the TPC, but boiling resulted in a significant decrease [117]. Conversely, in orange-fleshed sweet potato, boiling, and roasting had a less negative effect on the TPC than steaming [19].
A clear trend has not been observed in the effect of cooking on phenolic acids in sweet potato, which include chlorogenic acids, a family of esters formed from certain cinnamic acids and quinic acid. A study by Carrera et al. (2021) demonstrated that orange-fleshed sweet potatoes had higher concentrations of caffeoylquinic acids (4-caffeoylquinic acid and 4,5-dicaffeoylquinic acid) after oven-roasting compared with boiling or steaming in the microwave [116]. The fact that boiled sweet potatoes generally contain a lower concentration of caffeoylquinic acids may be due to the extraction effect of water during cooking or by temperature. The same study demonstrated that phenolic compounds decreased with prolonged cooking in water. Another comparative study reported that total caffeoylquinic acids were reduced by different home processing techniques in the following order: boiling > deep-frying > sautéing or steaming > microwaving or oven baking [1]. Finally, a positive effect of cooking on the chlorogenic acid content was detected in a purple-fleshed variety; the biggest increase was found in sweet potatoes cooked by steaming, followed by baking > microwaving > boiling [120].
Anthocyanins are well known to be sensitive to heat and light and susceptible to variations in pH and the presence of oxygen, but the way they are affected by cooking depends on the technique used [114,122]. The highest loss of anthocyanins in purple-fleshed sweet potato was reported to be induced by baking, followed by steaming and boiling [117]. In contrast, another study found that boiling, steaming, microwaving, and baking increased the content in almost all tested samples (orange and purple) compared to raw sweet potato [119]. An increase in total flavonoids was also found in boiled and fried, orange-fleshed sweet potatoes [18]. Finally, steam-cooking of purple sweet potato had a positive effect on total monomeric anthocyanin and flavonoid contents, which increased by approximately 13-fold and 5-fold, respectively, compared to the control [115].
Regarding the carotenoids, food processing can change their levels in this vegetable. Abong et al. (2021) reported that after frying and boiling sweet potatoes retained more than 90% of their β-carotene content [18]. Additionally, boiling has been found to reduce trans-β-carotene but increase cis-β-carotene [114].
In summary, the effect of cooking on the TPC, individual phenolic compounds, and carotenoids in sweet potato depends on the technique. The temperature and time of cooking and contact with water should be controlled to minimize the loss of these bioactive compounds.
5. Conclusions
Ipomoea batata L.; Lam. is an important food crop with a comprehensive content of macronutrients and micronutrients, and it stands out as a dietary source of several active secondary metabolites, principally carotenoids, phenolic acids, tocopherols, anthocyanins, flavonoids, and coumarins. Overall, it can be concluded that the concentration of phytochemicals in sweet potato depends on the variety, as well as the processing and storage conditions.
Growing evidence for the protective health effects of its bioactive constituents has attracted new interest in this root vegetable from the food industry, consumers, and scientists. The powerful antioxidant capacity of purple sweet potatoes is mainly attributed to their anthocyanin and carotenoid content. These compounds have a high capacity to eliminate free radicals and contribute to the inhibition of lipid peroxidation activity, offering protection against injuries induced by hepatotoxins. In addition, consumption of sweet potatoes rich in anthocyanins is closely associated with a lower risk of diabetes, cardiovascular disease, and cancer, and has also been associated with improved cognitive function. Some minor sesquiterpene compounds of sweet potato have also been described to improve intestinal barrier function. Although the protection of the purple sweet potato anthocyanin from injury induced by hepatotoxins has been studied in healthy men with borderline hepatitis, most of the studies describing the potential of the sweet potato phytochemicals have been performed in vitro and in animal models, so human intervention trials are necessary to demonstrate the possible health effect in humans. Therefore, further research is needed in this field, especially in randomized clinical trials in humans. The challenge today is to evaluate the effect of prolonged consumption of different sweet potatoes, especially purple ones, on indicators of diseases such as oxidative stress. Positive results would open future market prospects and encourage consumers to adopt sweet potatoes as a functional food with the ability to prevent the incidence of chronic diseases in the long term.
Finally, the application of commercial processing (drying) and domestic cooking methods (boiling, frying, steaming, baking, and microwaving) can affect the functional properties of sweet potatoes to different degrees. Although studies have shown that the concentration of phenolic compounds can change during processing (due to oxidation, leaching, isomerization, degradation, and/or release from cell walls), no clear trend has been observed as to what constitutes the optimal strategy for their preservation or enhancement. Therefore, further research is required on the effects of processing and cooking on sweet potatoes to provide consumers with guidelines on how to maximize their health properties.
Funding
This research was funded by the project (PID2020-114022RB-I00), funded by MCIN/AEI/10.13039/501100011033; CIBEROBN from the Instituto de Salud Carlos III, ISCIII from the Ministerio de Ciencia, Innovacion y Universidades, (AEI/FEDER, UE) and Generalitat de Catalunya (2017SGR 196). Emily P. Laveriano-Santos thanks for the FI-SDUR (EMC/3345/2020) fellowship from the Generalitat de Catalunya. Carolina Jaime-Rodriguez thanks the Ministry of Science, Technology, and Innovation (MinCiencias), Scholarship Program of Colombia (Announcement 885,2020). Johana González-Coria the National Scholarship Program of Paraguay “Don Carlos Antonio López” (BECAL). Anna Vallverdu-Queralt thanks the Ministry of Science Innovation and Universities for the Ramon y Cajal contract (RYC-2016-19355).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Jung, J.-K.; Lee, S.-U.; Kozukue, N.; Levin, C.E.; Friedman, M. Distribution of Phenolic Compounds and Antioxidative Activities in Parts of Sweet Potato (Ipomoea batata L.) Plants and in Home Processed Roots. J. Food Compos. Anal. 2011, 24, 29–37. [Google Scholar] [CrossRef]
- Bovell-Benjamin, A.C. Sweet Potato: A Review of Its Past, Present, and Future Role in Human Nutrition. Adv. Food Nutr. Res. 2007, 52, 1–59. [Google Scholar] [CrossRef] [PubMed]
- Shih, P.-H.; Yeh, C.-T.; Yen, G.-C. Anthocyanins Induce the Activation of Phase II Enzymes through the Antioxidant Response Element Pathway against Oxidative Stress-Induced Apoptosis. J. Agric. Food Chem. 2007, 55, 9427–9435. [Google Scholar] [CrossRef]
- Wang, H.; Cao, G.; Prior, R.L. Oxygen Radical Absorbing Capacity of Anthocyanins. J. Agric. Food Chem. 1997, 45, 304–309. [Google Scholar] [CrossRef]
- Parveen, A.; Choi, S.; Kang, J.-H.; Oh, S.H.; Kim, S.Y. Trifostigmanoside I, an Active Compound from Sweet Potato, Restores the Activity of MUC2 and Protects the Tight Junctions through PKCα/β to Maintain Intestinal Barrier Function. Int. J. Mol. Sci. 2020, 22, 291. [Google Scholar] [CrossRef] [PubMed]
- Mohanraj, R.; Sivasankar, S. Sweet Potato (Ipomoea batatas [L.] Lam)-A Valuable Medicinal Food: A Review. J. Med. Food 2014, 17, 733–741. [Google Scholar] [CrossRef]
- Shandilya, U.K.; Sharma, A. Functional Foods and Their Benefits: An Overview. J. Nutr. Health Food Eng. 2017, 7, 2–5. [Google Scholar] [CrossRef]
- Tang, Y.; Cai, W.; Xu, B. Profiles of Phenolics, Carotenoids and Antioxidative Capacities of Thermal Processed White, Yellow, Orange and Purple Sweet Potatoes Grown in Guilin, China. Food Sci. Hum. Wellness 2015, 4, 123–132. [Google Scholar] [CrossRef]
- Teow, C.C.; Truong, V.-D.; McFeeters, R.F.; Thompson, R.L.; Pecota, K.V.; Yencho, G.C. Antioxidant Activities, Phenolic and β-Carotene Contents of Sweet Potato Genotypes with Varying Flesh Colours. Food Chem. 2007, 103, 829–838. [Google Scholar] [CrossRef]
- Li, L.; Aldini, G.; Carini, M.; Chen, C.-Y.O.; Chun, H.-K.; Cho, S.-M.; Park, K.-M.; Correa, C.R.; Russell, R.M.; Blumberg, J.B. Characterisation, Extraction Efficiency, Stability and Antioxidant Activity of Phytonutrients in Angelica Keiskei. Food Chem. 2009, 115, 227–232. [Google Scholar] [CrossRef]
- Nguyen, H.C.; Chen, C.-C.; Lin, K.-H.; Chao, P.-Y.; Lin, H.-H.; Huang, M.-Y. Bioactive Compounds, Antioxidants, and Health Benefits of Sweet Potato Leaves. Molecules 2021, 26, 1820. [Google Scholar] [CrossRef] [PubMed]
- Behera, S.; Chauhan, V.B.S.; Pati, K.; Bansode, V.; Nedunchezhiyan, M.; Verma, A.K.; Monalisa, K.; Naik, P.K.; Naik, S.K. Biology and Biotechnological Aspect of Sweet Potato (Ipomoea batatas L.): A Commercially Important Tuber Crop. Planta 2022, 256, 40. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.; Rana, Z.; Islam, S. Comparison of the Proximate Composition, Total Carotenoids and Total Polyphenol Content of Nine Orange-Fleshed Sweet Potato Varieties Grown in Bangladesh. Foods 2016, 5, 64. [Google Scholar] [CrossRef] [PubMed]
- Kurata, R.; Sun, H.N.; Oki, T.; Okuno, S.; Ishiguro, K.; Sugawara, T. Sweet Potato Polyphenols. In Sweet Potato Chemistry, Processing and Nutrition; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 177–222. [Google Scholar] [CrossRef]
- Luo, D.; Mu, T.; Sun, H. Profiling of Phenolic Acids and Flavonoids in Sweet Potato (Ipomoea batatas L.) Leaves and Evaluation of Their Anti-Oxidant and Hypoglycemic Activities. Food Biosci. 2021, 39, 100801. [Google Scholar] [CrossRef]
- Sun, H.; Mu, B.; Song, Z.; Ma, Z.; Mu, T. The In Vitro Antioxidant Activity and Inhibition of Intracellular Reactive Oxygen Species of Sweet Potato Leaf Polyphenols. Oxid. Med. Cell. Longev. 2018, 2018, 9017828. [Google Scholar] [CrossRef]
- Wang, S.; Nie, S.; Zhu, F. Chemical Constituents and Health Effects of Sweet Potato. Food Res. Int. 2016, 89, 90–116. [Google Scholar] [CrossRef]
- Abong’, G.O.; Muzhingi, T.; Okoth, M.W.; Ng’ang’a, F.; Emelda Ochieng, P.; Mbogo, D.M.; Malavi, D.; Akhwale, M.; Ghimire, S. Processing Methods Affect Phytochemical Contents in Products Prepared from Orange-fleshed Sweetpotato Leaves and Roots. Food Sci. Nutr. 2021, 9, 1070–1078. [Google Scholar] [CrossRef]
- Donado-Pestana, C.M.; Salgado, J.M.; de Oliveira Rios, A.; dos Santos, P.R.; Jablonski, A. Stability of Carotenoids, Total Phenolics and In Vitro Antioxidant Capacity in the Thermal Processing of Orange-Fleshed Sweet Potato (Ipomoea batatas Lam.) Cultivars Grown in Brazil. Plant Foods Hum. Nutr. 2012, 67, 262–270. [Google Scholar] [CrossRef]
- Truong, V.D.; McFeeters, R.F.; Thompson, R.T.; Dean, L.L.; Shofran, B. Phenolic Acid Content and Composition in Leaves and Roots of Common Commercial Sweetpotato (Ipomea batatas L.) Cultivars in the United States. J. Food Sci. 2007, 72, C343–C349. [Google Scholar] [CrossRef]
- Cevallos-Casals, B.A.; Cisneros-Zevallos, L. Stoichiometric and Kinetic Studies of Phenolic Antioxidants from Andean Purple Corn and Red-Fleshed Sweetpotato. J. Agric. Food Chem. 2003, 51, 3313–3319. [Google Scholar] [CrossRef]
- Im, Y.R.; Kim, I.; Lee, J. Phenolic Composition and Antioxidant Activity of Purple Sweet Potato (Ipomoea batatas (L.) Lam.): Varietal Comparisons and Physical Distribution. Antioxidants 2021, 10, 462. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Oki, T.; Kai, Y.; Nishiba, Y.; Okuno, S. Effect of Repeated Harvesting on the Content of Caffeic Acid and Seven Species of Caffeoylquinic Acids in Sweet Potato Leaves. Biosci. Biotechnol. Biochem. 2015, 79, 1308–1314. [Google Scholar] [CrossRef] [PubMed]
- Nicoletto, C.; Vianello, F.; Sambo, P. Effect of Different Home-Cooking Methods on Textural and Nutritional Properties of Sweet Potato Genotypes Grown in Temperate Climate Conditions. J. Sci. Food Agric. 2018, 98, 574–581. [Google Scholar] [CrossRef]
- Ooko Abong’, G.; Muzhingi, T.; Wandayi Okoth, M.; Ng’ang’a, F.; Ochieng’, P.E.; Mahuga Mbogo, D.; Malavi, D.; Akhwale, M.; Ghimire, S. Phytochemicals in Leaves and Roots of Selected Kenyan Orange Fleshed Sweet Potato (OFSP) Varieties. Int. J. Food Sci. 2020, 3567972. [Google Scholar] [CrossRef] [PubMed]
- Majid, M.; Nasir, B.; Zahra, S.S.; Khan, M.R.; Mirza, B.; Haq, I.u. Ipomoeabatatas L. Lam. Ameliorates Acute and Chronic Inflammations by Suppressing Inflammatory Mediators, a Comprehensive Exploration Using In Vitro and In Vivo Models. BMC Complement Altern. Med. 2018, 18, 216. [Google Scholar] [CrossRef]
- Park, S.Y.; Lee, S.Y.; Yang, J.W.; Lee, J.S.; Oh, S.D.; Oh, S.; Lee, S.M.; Lim, M.H.; Park, S.K.; Jang, J.S.; et al. Comparative Analysis of Phytochemicals and Polar Metabolites from Colored Sweet Potato (Ipomoea batatas L.) Tubers. Food Sci. Biotechnol. 2016, 25, 283–291. [Google Scholar] [CrossRef]
- Oki, T.; Masuda, M.; Furuta, S.; Nishiba, Y.; Terahara, N.; Suda, I. Involvement of Anthocyanins and Other Phenolic Compounds in Radical-scavenging Activity of Purple-fleshed Sweet Potato Cultivars. J. Food Sci. 2002, 67, 1752–1756. [Google Scholar] [CrossRef]
- Sun, H.; Zhang, P.; Zhu, Y.; Lou, Q.; He, S. Antioxidant and Prebiotic Activity of Five Peonidin-Based Anthocyanins Extracted from Purple Sweet Potato (Ipomoea batatas (L.) Lam.). Sci. Rep. 2018, 8, 5018. [Google Scholar] [CrossRef]
- Steed, L.E.; Truong, V.D. Anthocyanin Content, Antioxidant Activity, and Selected Physical Properties of Flowable Purple-Fleshed Sweetpotato Purees. J. Food Sci. 2008, 73, S215–S221. [Google Scholar] [CrossRef]
- Zhu, F.; Cai, Y.Z.; Yang, X.; Ke, J.; Corke, H. Anthocyanins, Hydroxycinnamic Acid Derivatives, and Antioxidant Activity in Roots of Different Chinese Purple-Fleshed Sweetpotato Genotypes. J. Agric. Food Chem. 2010, 58, 7588–7596. [Google Scholar] [CrossRef]
- Vishnu, V.R.; Renjith, R.S.; Mukherjee, A.; Anil, S.R.; Sreekumar, J.; Jyothi, A.N. Comparative Study on the Chemical Structure and in Vitro Antiproliferative Activity of Anthocyanins in Purple Root Tubers and Leaves of Sweet Potato (Ipomoea batatas). J. Agric. Food Chem. 2019, 67, 2467–2475. [Google Scholar] [CrossRef] [PubMed]
- Montilla, E.C.; Hillebrand, S.; Butschbach, D.; Baldermann, S.; Watanabe, N.; Winterhalter, P. Preparative Isolation of Anthocyanins from Japanese Purple Sweet Potato (Ipomoea batatas L.) Varieties by High-Speed Countercurrent Chromatography. J. Agric. Food Chem. 2010, 58, 9899–9904. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.W.; Kim, J.B.; Cho, S.M.; Chung, M.N.; Lee, Y.M.; Chu, S.M.; Che, J.H.; Kim, S.N.; Kim, S.Y.; Cho, Y.S.; et al. Anthocyanin Changes in the Korean Purple-Fleshed Sweet Potato, Shinzami, as Affected by Steaming and Baking. Food Chem. 2012, 130, 966–972. [Google Scholar] [CrossRef]
- Xu, J.; Su, X.; Lim, S.; Griffin, J.; Carey, E.; Katz, B.; Tomich, J.; Smith, J.S.; Wang, W. Characterisation and Stability of Anthocyanins in Purple-Fleshed Sweet Potato P40. Food Chem. 2015, 186, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Clifford, M.N. Profiling the Chlorogenic Acids of Sweet Potato (Ipomoea batatas) from China. Food Chem. 2008, 106, 147–152. [Google Scholar] [CrossRef]
- Rodriguez-Saona, L.E.; Giusti, M.M.; Wrolstad, R.E. Anthocyanin Pigment Composition of Red-Fleshed Potatoes. J. Food Sci. 1998, 63, 458–465. [Google Scholar] [CrossRef]
- Ji, H.; Zhang, H.; Li, H.; Li, Y.; Ji, H.; Zhang, H.; Li, H.; Li, Y. Analysis on the Nutrition Composition and Antioxidant Activity of Different Types of Sweet Potato Cultivars. Food Nutr. Sci. 2015, 6, 161–167. [Google Scholar] [CrossRef]
- Brown, C.R.; Culley, D.; Yang, C.P.; Durst, R.; Wrolstad, R. Variation of Anthocyanin and Carotenoid Contents and Associated Antioxidant Values in Potato Breeding Lines. J. Am. Soc. Hortic. Sci. 2005, 130, 174–180. [Google Scholar] [CrossRef]
- He, W.; Zeng, M.; Chen, J.; Jiao, Y.; Niu, F.; Tao, G.; Zhang, S.; Qin, F.; He, Z. Identification and Quantitation of Anthocyanins in Purple-Fleshed Sweet Potatoes Cultivated in China by UPLC-PDA and UPLC-QTOF-MS/MS. J. Agric. Food Chem. 2016, 64, 171–177. [Google Scholar] [CrossRef]
- Lee, M.J.; Park, J.S.; Choi, D.S.; Jung, M.Y. Characterization and Quantitation of Anthocyanins in Purple-Fleshed Sweet Potatoes Cultivated in Korea by HPLC-DAD and HPLC-ESI-QTOF-MS/MS. J. Agric. Food Chem. 2013, 61, 3148–3158. [Google Scholar] [CrossRef]
- Li, J.; Li, X.D.; Zhang, Y.; Zheng, Z.D.; Qu, Z.Y.; Liu, M.; Zhu, S.H.; Liu, S.; Wang, M.; Qu, L. Identification and Thermal Stability of Purple-Fleshed Sweet Potato Anthocyanins in Aqueous Solutions with Various PH Values and Fruit Juices. Food Chem. 2013, 136, 1429–1434. [Google Scholar] [CrossRef] [PubMed]
- Fossen, T.; Andersen, Ø.M. Anthocyanins from Tubers and Shoots of the Purple Potato, Solanum tuberosum. J. Hortic. Sci. Biotechnol. 2000, 75, 360–363. [Google Scholar] [CrossRef]
- Matsufuji, H.; Kido, H.; Misawa, H.; Yaguchi, J.; Otsuki, T.; Chino, M.; Takeda, M.; Yamagata, K. Stability to Light, Heat, and Hydrogen Peroxide at Different PH Values and DPPH Radical Scavenging Activity of Acylated Anthocyanins from Red Radish Extract. J. Agric. Food Chem. 2007, 55, 3692–3701. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.L.; Yu, Y.Q.; Chen, Z.J.; Wen, G.S.; Wei, F.G.; Zheng, Q.; Wang, C.; Xiao, X.L. Stability-Increasing Effects of Anthocyanin Glycosyl Acylation. Food Chem. 2017, 214, 119–128. [Google Scholar] [CrossRef]
- Padda, M.S.; Picha, D.H. Quantification of Phenolic Acids and Antioxidant Activity in Sweetpotato Genotypes. Sci. Hortic. 2008, 119, 17–20. [Google Scholar] [CrossRef]
- Escarpa, A.; González, M.C. Approach to the Content of Total Extractable Phenolic Compounds from Different Food Samples by Comparison of Chromatographic and Spectrophotometric Methods. Anal. Chim. Acta 2001, 427, 119–127. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of Total Phenols and Other Oxidation Substrates and Antioxidants by Means of Folin-Ciocalteu Reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Vimala, B.; Nambisan, B.; Hariprakash, B. Retention of Carotenoids in Orange-Fleshed Sweet Potato during Processing. J. Food Sci. Technol. 2011, 48, 520–524. [Google Scholar] [CrossRef]
- Jiang, P.; Han, B.; Jiang, L.; Li, Y.; Yu, Y.; Xu, H.; Li, Z.; Zhou, D.; Jia, X.; Li, X.; et al. Simultaneous Separation and Quantitation of Three Phytosterols from the Sweet Potato, and Determination of Their Anti-Breast Cancer Activity. J. Pharm. Biomed. Anal. 2019, 174, 718–727. [Google Scholar] [CrossRef]
- Cordeiro, N.; Freitas, N.; Faria, M.; Gouveia, M. Ipomoeabatatas (L.) Lam.: A Rich Source of Lipophilic Phytochemicals. J. Agric. Food Chem. 2013, 61, 12380–12384. [Google Scholar] [CrossRef]
- Islam, S.N.; Nusrat, T.; Begum, P.; Ahsan, M. Carotenoids and β-Carotene in Orange Fleshed Sweet Potato: A Possible Solution to Vitamin A Deficiency. Food Chem. 2016, 199, 628–631. [Google Scholar] [CrossRef]
- Tian, S.J.; Rickard, J.E.; Blanshard, J.M.V. Physicochemical Properties of Sweet Potato Starch. J. Sci. Food Agric. 1991, 57, 459–491. [Google Scholar] [CrossRef]
- Willcox, D.C.; Willcox, B.J.; Todoriki, H.; Suzuki, M. The Okinawan Diet: Health Implications of a Low-Calorie, Nutrient-Dense, Antioxidant-Rich Dietary Pattern Low in Glycemic Load. J. Am. Coll. Nutr. 2009, 28, 500S–516S. [Google Scholar] [CrossRef] [PubMed]
- Philpott, M.; Lim, C.C.; Ferguson, L.R. Dietary Protection against Free Radicals: A Case for Multiple Testing to Establish Structure-Activity Relationships for Antioxidant Potential of Anthocyanic Plant Species. Int. J. Mol. Sci. 2009, 10, 1081–1103. [Google Scholar] [CrossRef] [PubMed]
- Philpott, M.; Gould, K.S.; Lim, C.; Ferguson, L.R. In Situ and In Vitro Antioxidant Activity of Sweetpotato Anthocyanins. J. Agric. Food Chem. 2004, 52, 1511–1513. [Google Scholar] [CrossRef] [PubMed]
- Kano, M.; Takayanagi, T.; Harada, K.; Makino, K.; Ishikawa, F. Antioxidative Activity of Anthocyanins from Purple Sweet Potato, Ipomoera Batatas Cultivar Ayamurasaki. Biosci. Biotechnol. Biochem. 2005, 69, 979–988. [Google Scholar] [CrossRef]
- Fu, H.; Xie, B.; Ma, S.; Zhu, X.; Fan, G.; Pan, S. Evaluation of Antioxidant Activities of Principal Carotenoids Available in Water Spinach (Ipomoea aquatica). J. Food Compos. Anal. 2011, 24, 288–297. [Google Scholar] [CrossRef]
- Suda, I.; Ishikawa, F.; Hatakeyama, M.; Miyawaki, M.; Kudo, T.; Hirano, K.; Ito, A.; Yamakawa, O.; Horiuchi, S. Intake of Purple Sweet Potato Beverage Affects on Serum Hepatic Biomarker Levels of Healthy Adult Men with Borderline Hepatitis. Eur. J. Clin. Nutr. 2008, 62, 60–67. [Google Scholar] [CrossRef]
- Wang, W.; Li, J.; Wang, Z.; Gao, H.; Su, L.; Xie, J.; Chen, X.; Liang, H.; Wang, C.; Han, Y. Oral Hepatoprotective Ability Evaluation of Purple Sweet Potato Anthocyanins on Acute and Chronic Chemical Liver Injuries. Cell Biochem. Biophys 2014, 69, 539–548. [Google Scholar] [CrossRef]
- Wang, L.; Zhao, Y.; Zhou, Q.; Luo, C.-L.; Deng, A.-P.; Zhang, Z.-C.; Zhang, J.-L. Characterization and Hepatoprotective Activity of Anthocyanins from Purple Sweet Potato (Ipomoea batatas L. Cultivar Eshu No. 8). J. Food Drug Anal. 2017, 25, 607–618. [Google Scholar] [CrossRef]
- Sun, J.; Zhou, B.; Tang, C.; Gou, Y.; Chen, H.; Wang, Y.; Jin, C.; Liu, J.; Niu, F.; Kan, J. Characterization, Antioxidant Activity and Hepatoprotective Effect of Purple Sweetpotato Polysaccharides. Int. J. Biol. Macromol. 2018, 115, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Han, K.-H.; Shimada, K.; Sekikawa, M.; Fukushima, M. Anthocyanin-Rich Red Potato Flakes Affect Serum Lipid Peroxidation and Hepatic SOD MRNA Level in Rats. Biosci. Biotechnol. Biochem. 2007, 71, 1356–1359. [Google Scholar] [CrossRef]
- Choi, J.H.; Choi, C.Y.; Lee, K.J.; Hwang, Y.P.; Chung, Y.C.; Jeong, H.G. Hepatoprotective Effects of an Anthocyanin Fraction from Purple-Fleshed Sweet Potato against Acetaminophen-Induced Liver Damage in Mice. J. Med. Food 2009, 12, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-F.; Fan, S.-H.; Zheng, Y.-L.; Lu, J.; Wu, D.-M.; Shan, Q.; Hu, B. Purple Sweet Potato Color Attenuates Oxidative Stress and Inflammatory Response Induced by D-Galactose in Mouse Liver. Food Chem. Toxicol. 2009, 47, 496–501. [Google Scholar] [CrossRef]
- Jung, S.; Shin, J.; Kim, J.Y.; Kwon, O. Shinzami Korean Purple-fleshed Sweet Potato Extract Prevents Ischaemia–Reperfusion-induced Liver Damage in Rats. J. Sci. Food Agric. 2015, 95, 2818–2823. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Miyamae, Y.; Shigemori, H.; Isoda, H. Neuroprotective Effect of 3, 5-Di-O-Caffeoylquinic Acid on SH-SY5Y Cells and Senescence-Accelerated-Prone Mice 8 through the up-Regulation of Phosphoglycerate Kinase-1. Neuroscience 2010, 169, 1039–1045. [Google Scholar] [CrossRef]
- Sasaki, K.; Han, J.; Shimozono, H.; Villareal, M.O.; Isoda, H. Caffeoylquinic Acid-Rich Purple Sweet Potato Extract, with or without Anthocyanin, Imparts Neuroprotection and Contributes to the Improvement of Spatial Learning and Memory of SAMP8 Mouse. J. Agric. Food Chem. 2013, 61, 5037–5045. [Google Scholar] [CrossRef]
- Wu, D.; Lu, J.; Zheng, Y.; Zhou, Z.; Shan, Q.; Ma, D. Purple Sweet Potato Color Repairs D-Galactose-Induced Spatial Learning and Memory Impairment by Regulating the Expression of Synaptic Proteins. Neurobiol. Learn. Mem. 2008, 90, 19–27. [Google Scholar] [CrossRef]
- Matthews, D.G.; Caruso, M.; Alcazar Magana, A.; Wright, K.M.; Maier, C.S.; Stevens, J.F.; Gray, N.E.; Quinn, J.F.; Soumyanath, A. Caffeoylquinic Acids in Centella Asiatica Reverse Cognitive Deficits in Male 5XFAD Alzheimer’s Disease Model Mice. Nutrients 2020, 12, 3488. [Google Scholar] [CrossRef]
- Choi, S.J.; Kim, J.K.; Suh, S.H.; Kim, C.R.; Kim, H.K.; Kim, C.-J.; Park, G.G.; Park, C.-S.; Shin, D.-H. Ligularia Fischeri Extract Protects against Oxidative-Stress-Induced Neurotoxicity in Mice and Pc12 Cells. J. Med. Food 2014, 17, 1222–1231. [Google Scholar] [CrossRef]
- Kwon, S.-H.; Lee, H.-K.; Kim, J.-A.; Hong, S.-I.; Kim, H.-C.; Jo, T.-H.; Park, Y.-I.; Lee, C.-K.; Kim, Y.-B.; Lee, S.-Y. Neuroprotective Effects of Chlorogenic Acid on Scopolamine-Induced Amnesia via Anti-Acetylcholinesterase and Anti-Oxidative Activities in Mice. Eur. J. Pharmacol. 2010, 649, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.J.; Kim, J.K.; Kim, H.K.; Harris, K.; Kim, C.-J.; Park, G.G.; Park, C.-S.; Shin, D.-H. 2, 4-Di-Tert-Butylphenol from Sweet Potato Protects against Oxidative Stress in PC12 Cells and in Mice. J. Med. Food 2013, 16, 977–983. [Google Scholar] [CrossRef]
- Adnyana, I.M.O.; Sudewi, A.A.R.; Samatra, D.P.G.P.; Suprapta, D.N. Neuroprotective Effects of Purple Sweet Potato Balinese Cultivar in Wistar Rats with Ischemic Stroke. Open Access Maced. J. Med. Sci. 2018, 6, 1959. [Google Scholar] [CrossRef]
- Adnyana, I.M.O.; Sudewi, R.; Samatra, P.; Suprapta, S. Balinese Cultivar of Purple Sweet Potato Improved Neurological Score and BDNF and Reduced Caspase-Independent Apoptosis among Wistar Rats with Ischemic Stroke. Open Access Maced. J. Med. Sci. 2019, 7, 38–44. [Google Scholar] [CrossRef]
- Zhuang, J.; Lu, J.; Wang, X.; Wang, X.; Hu, W.; Hong, F.; Zhao, X.; Zheng, Y. Purple Sweet Potato Color Protects against High-Fat Diet-Induced Cognitive Deficits through AMPK-Mediated Autophagy in Mouse Hippocampus. J. Nutr. Biochem. 2019, 65, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Shi, Z.; Mi, Y. Purple Sweet Potato Color Attenuates High Fat-Induced Neuroinflammation in Mouse Brain by Inhibiting MAPK and NF-ΚB Activation. Mol. Med. Rep. 2018, 17, 4823–4831. [Google Scholar] [CrossRef]
- Sugata, M.; Lin, C.-Y.; Shih, Y.-C. Anti-Inflammatory and Anticancer Activities of Taiwanese Purple-Fleshed Sweet Potatoes (Ipomoea batatas L. Lam) Extracts. BioMed Res. Int. 2015, 2015, 768093. [Google Scholar] [CrossRef]
- Kato, K.; Nagane, M.; Aihara, N.; Kamiie, J.; Miyanabe, M.; Hiraki, S.; Luo, X.; Nakanishi, I.; Shoji, Y.; Matsumoto, K. Lipid-Soluble Polyphenols from Sweet Potato Exert Antitumor Activity and Enhance Chemosensitivity in Breast Cancer. J. Clin. Biochem. Nutr. 2021, 68, 193–200. [Google Scholar] [CrossRef]
- Xu, H.; Li, Y.; Han, B.; Li, Z.; Wang, B.; Jiang, P.; Zhang, J.; Ma, W.; Zhou, D.; Li, X. Anti-Breast-Cancer Activity Exerted by β-Sitosterol-d-Glucoside from Sweet Potato via Upregulation of Microrna-10a and via the Pi3k–Akt Signaling Pathway. J. Agric. Food Chem. 2018, 66, 9704–9718. [Google Scholar] [CrossRef]
- Li, Z.; Yu, Y.; Wang, M.; Xu, H.; Han, B.; Jiang, P.; Ma, H.; Li, Y.; Tian, C.; Zhou, D. Anti-Breast Cancer Activity of SPG-56 from Sweet Potato in MCF-7 Bearing Mice in Situ through Promoting Apoptosis and Inhibiting Metastasis. Sci. Rep. 2019, 9, 146. [Google Scholar] [CrossRef]
- Tian, C.; Wang, M.; Liu, S.; Ma, H.; He, K.; Zhou, D.; Li, Y.; Ye, X.; Li, X. A New Glycoprotein SPG-8700 Isolated from Sweet Potato with Potential Anti-Cancer Activity against Colon Cancer. Nat. Prod. Res. 2019, 33, 2322–2328. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Chen, S.-J.; Chen, B.-W.; Zhang, K.-W.; Zhang, J.-J.; Xiao, R.; Li, P.-G. Gene Expression Profile of the Human Colorectal Carcinoma LoVo Cells Treated With Sporamin and Thapsigargin. Front. Oncol. 2021, 11, 2003. [Google Scholar] [CrossRef]
- Lim, S.; Xu, J.; Kim, J.; Chen, T.; Su, X.; Standard, J.; Carey, E.; Griffin, J.; Herndon, B.; Katz, B. Role of Anthocyanin-enriched Purple-fleshed Sweet Potato P40 in Colorectal Cancer Prevention. Mol. Nutr. Food Res. 2013, 57, 1908–1917. [Google Scholar] [CrossRef]
- Li, W.-L.; Yu, H.-Y.; Zhang, X.-J.; Ke, M.; Hong, T. Purple Sweet Potato Anthocyanin Exerts Antitumor Effect in Bladder Cancer. Oncol. Rep. 2018, 40, 73–82. [Google Scholar] [CrossRef]
- Gundala, S.R.; Yang, C.; Lakshminarayana, N.; Asif, G.; Gupta, M.V.; Shamsi, S.; Aneja, R. Polar Biophenolics in Sweet Potato Greens Extract Synergize to Inhibit Prostate Cancer Cell Proliferation and in Vivo Tumor Growth. Carcinogenesis 2013, 34, 2039–2049. [Google Scholar] [CrossRef]
- Kim, H.; Koo, K.A.; Park, W.S.; Kang, D.; Kim, H.S.; Lee, B.Y.; Goo, Y.; Kim, J.; Lee, M.K.; Woo, D.K. Anti-obesity Activity of Anthocyanin and Carotenoid Extracts from Color-fleshed Sweet Potatoes. J. Food Biochem. 2020, 44, e13438. [Google Scholar] [CrossRef]
- Naomi, R.; Bahari, H.; Yazid, M.D.; Othman, F.; Zakaria, Z.A.; Hussain, M.K. Potential Effects of Sweet Potato (Ipomoea batatas) in Hyperglycemia and Dyslipidemia—A Systematic Review in Diabetic Retinopathy Context. Int. J. Mol. Sci. 2021, 22, 10816. [Google Scholar] [CrossRef]
- Massi-Benedetti, M.; Federici, M.O. Cardiovascular Risk Factors in Type 2 Diabetes: The Role of Hyperglycaemia. Exp. Clin. Endocrinol. Diabetes 1999, 107, S120–S123. [Google Scholar] [CrossRef]
- Onat, D.; Brillon, D.; Colombo, P.C.; Schmidt, A.M. Human Vascular Endothelial Cells: A Model System for Studying Vascular Inflammation in Diabetes and Atherosclerosis. Curr. Diab. Rep. 2011, 11, 193–202. [Google Scholar] [CrossRef] [Green Version]
- Loke, W.M.; Proudfoot, J.M.; Hodgson, J.M.; McKinley, A.J.; Hime, N.; Magat, M.; Stocker, R.; Croft, K.D. Specific Dietary Polyphenols Attenuate Atherosclerosis in Apolipoprotein E–Knockout Mice by Alleviating Inflammation and Endothelial Dysfunction. Arter. Thromb. Vasc. Biol. 2010, 30, 749–757. [Google Scholar] [CrossRef]
- Sun, C.; Fan, S.; Wang, X.; Lu, J.; Zhang, Z.; Wu, D.; Shan, Q.; Zheng, Y. Purple Sweet Potato Color Inhibits Endothelial Premature Senescence by Blocking the NLRP3 Inflammasome. J. Nutr. Biochem. 2015, 26, 1029–1040. [Google Scholar] [CrossRef]
- Selokela, L.M.; Laurie, S.M.; Sivakumar, D. Impact of Different Postharvest Thermal Processes on Changes in Antioxidant Constituents, Activity and Nutritional Compounds in Sweet Potato with Varying Flesh Colour. South Afr. J. Bot. 2022, 144, 380–388. [Google Scholar] [CrossRef]
- Perez, M.; Domínguez-López, I.; López-Yerena, A.; Vallverdú Queralt, A. Current Strategies to Guarantee the Authenticity of Coffee. Crit. Rev. Food Sci. Nutr. 2021, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Perez, M.; Lopez-Yerena, A.; Vallverdú-Queralt, A. Traceability, Authenticity and Sustainability of Cocoa and Chocolate Products: A Challenge for the Chocolate Industry. Crit. Rev. Food Sci. Nutr. 2021, 62, 475–489. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Zhang, M.; Ye, Y.; Yu, D. Influence of Ultrasonic Pretreatments on Drying Kinetics and Quality Attributes of Sweet Potato Slices in Infrared Freeze Drying (IRFD). LWT 2020, 131, 109801. [Google Scholar] [CrossRef]
- Moses, J.A.; Norton, T.; Alagusundaram, K.; Tiwari, B.K. Novel Drying Techniques for the Food Industry. Food Eng. Rev. 2014, 6, 43–55. [Google Scholar] [CrossRef]
- Yang, J.; Chen, J.F.; Zhao, Y.Y.; Mao, L.C. Effects of Drying Processes on the Antioxidant Properties in Sweet Potatoes. Agric. Sci. China 2010, 9, 1522–1529. [Google Scholar] [CrossRef]
- Savas, E. The Modelling of Convective Drying Variables’ Effects on the Functional Properties of Sliced Sweet Potatoes. Foods 2022, 11, 741. [Google Scholar] [CrossRef]
- Grabowski, J.A.; Truong, V.-D.; Daubert, C.R. Nutritional and Rheological Characterization of Spray Dried Sweetpotato Powder. LWT-Food Sci. Technol. 2008, 41, 206–216. [Google Scholar] [CrossRef]
- Verma, A.; Singh, S.V. Spray Drying of Fruit and Vegetable Juices—A Review. Crit. Rev. Food Sci. Nutr. 2015, 55, 701–719. [Google Scholar] [CrossRef]
- Chamorro, S.; Cueva-Mestanza, R.; de Pascual-Teresa, S. Effect of Spray Drying on the Polyphenolic Compounds Present in Purple Sweet Potato Roots: Identification of New Cinnamoylquinic Acids. Food Chem. 2021, 345, 128679. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Li, J.; Guan, Y.; Zhao, G. Effect of Carriers on Physicochemical Properties, Antioxidant Activities and Biological Components of Spray-Dried Purple Sweet Potato Flours. LWT-Food Sci. Technol. 2013, 51, 348–355. [Google Scholar] [CrossRef]
- Lagnika, C.; Riaz, A.; Jiang, N.; Song, J.; Li, D.; Liu, C.; Wei, Q.; Zhang, M. Effects of Pretreatment and Drying Methods on the Quality and Stability of Dried Sweet Potato Slices during Storage. J. Food Processing Preserv. 2021, 45, e15807. [Google Scholar] [CrossRef]
- Abalos, R.A.; Naef, E.F.; Aviles, M.V.; Gómez, M.B. Vacuum Impregnation: A Methodology for the Preparation of a Ready-to-Eat Sweet Potato Enriched in Polyphenols. LWT 2020, 131, 109773. [Google Scholar] [CrossRef]
- Kręcisz, M.; Kolniak-Ostek, J.; Stępień, B.; Łyczko, J.; Pasławska, M.; Musiałowska, J. Influence of Drying Methods and Vacuum Impregnation on Selected Quality Factors of Dried Sweet Potato. Agriculture 2021, 11, 858. [Google Scholar] [CrossRef]
- Pérez, M.; López-Yerena, A.; Lozano-Castellón, J.; Olmo-Cunillera, A.; Lamuela-Raventós, R.M.; Martin-Belloso, O.; Vallverdú-Queralt, A. Impact of Emerging Technologies on Virgin Olive Oil Processing, Consumer Acceptance, and the Valorization of Olive Mill Wastes. Antioxidants 2021, 10, 417. [Google Scholar] [CrossRef]
- Cui, R.; Zhu, F. Effect of Ultrasound on Structural and Physicochemical Properties of Sweetpotato and Wheat Flours. Ultrason. Sonochem. 2020, 66, 105118. [Google Scholar] [CrossRef]
- Singla, M.; Sit, N. Application of Ultrasound in Combination with Other Technologies in Food Processing: A Review. Ultrason. Sonochem. 2021, 73, 105506. [Google Scholar] [CrossRef]
- Infante, R.A.; Azoubel, P.M.; de Lima, M.A.B.; Stamford, T.C.M.; Araújo, A.S.; de Mendonça, W.S.; da Silva Vasconcelos, M.A. Ultrasound Pretreatment Application in Dehydration: Its Influence on the Microstructure, Antioxidant Activity and Carotenoid Retention of Biofortified Beauregard Sweet Potato (Ipomoea batatas Lam). J. Food Sci. Technol. 2021, 58, 4542–4549. [Google Scholar] [CrossRef]
- Rashid, M.T.; Ma, H.; Jatoi, M.A.; Hashim, M.M.; Wali, A.; Safdar, B. Influence of Ultrasonic Pretreatment with Hot Air Drying on Nutritional Quality and Structural Related Changes in Dried Sweet Potatoes. Int. J. Food Eng. 2019, 15, 20180409. [Google Scholar] [CrossRef]
- Derossi, A.; Husain, A.; Caporizzi, R.; Severini, C. Manufacturing Personalized Food for People Uniqueness. An Overview from Traditional to Emerging Technologies. Crit. Rev. Food Sci. Nutr. 2020, 60, 1141–1159. [Google Scholar] [CrossRef]
- Betoret, E.; Betoret, N.; Rocculi, P.; Dalla Rosa, M. Strategies to Improve Food Functionality: Structure–Property Relationships on High Pressures Homogenization, Vacuum Impregnation and Drying Technologies. Trends Food Sci. Technol. 2015, 46, 1–12. [Google Scholar] [CrossRef]
- Murador, D.C.; da Cunha, D.T.; de Rosso, V.V. Effects of Cooking Techniques on Vegetable Pigments: A Meta-Analytic Approach to Carotenoid and Anthocyanin Levels. Food Res. Int. 2014, 65, 177–183. [Google Scholar] [CrossRef]
- Cakrawati, D.; Srivichai, S.; Hongsprabhas, P. Effect of Steam-Cooking on (Poly) Phenolic Compounds in Purple Yam and Purple Sweet Potato Tubers. Food Res. 2021, 5, 330–336. [Google Scholar] [CrossRef]
- Carrera, C.; Zelaya-Medina, C.F.; Chinchilla, N.; Ferreiro-González, M.; Barbero, G.F.; Palma, M. How Different Cooking Methods Affect the Phenolic Composition of Sweet Potato for Human Consumption (Ipomea batata (L.) Lam). Agronomy 2021, 11, 1636. [Google Scholar] [CrossRef]
- Hong, K.H.; Koh, E. Effects of Cooking Methods on Anthocyanins and Total Phenolics in Purple-fleshed Sweet Potato. J. Food Processing Preserv. 2016, 40, 1054–1063. [Google Scholar] [CrossRef]
- Hou, F.; Mu, T.; Ma, M.; Blecker, C. Optimization of Processing Technology Using Response Surface Methodology and Physicochemical Properties of Roasted Sweet Potato. Food Chem. 2019, 278, 136–143. [Google Scholar] [CrossRef]
- Musilova, J.; Lidikova, J.; Vollmannova, A.; Frankova, H.; Urminska, D.; Bojnanska, T.; Toth, T. Influence of Heat Treatments on the Content of Bioactive Substances and Antioxidant Properties of Sweet Potato (Ipomoea batatas L.) Tubers. J. Food 2020, 2020, 8856260. [Google Scholar] [CrossRef]
- Franková, H.; Musilová, J.; Árvay, J.; Šnirc, M.; Jančo, I.; Lidiková, J.; Vollmannová, A. Changes in Antioxidant Properties and Phenolics in Sweet Potatoes (Ipomoea batatas L.) Due to Heat Treatments. Molecules 2022, 27, 1884. [Google Scholar] [CrossRef]
- Burgos, G.; Amoros, W.; Muñoa, L.; Sosa, P.; Cayhualla, E.; Sanchez, C.; Díaz, C.; Bonierbale, M. Total Phenolic, Total Anthocyanin and Phenolic Acid Concentrations and Antioxidant Activity of Purple-Fleshed Potatoes as Affected by Boiling. J. Food Compos. Anal. 2013, 30, 6–12. [Google Scholar] [CrossRef]
- Huang, Y.; Zhou, S.; Zhao, G.; Ye, F. Destabilisation and Stabilisation of Anthocyanins in Purple-Fleshed Sweet Potatoes: A Review. Trends Food Sci. Technol. 2021, 116, 1141–1154. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).