The Impact of Fermentation on Bee Pollen Polyphenolic Compounds Composition
Abstract
:1. Introduction
2. Materials and Methods
2.1. Pollen Samples
2.2. Chemicals and Reagents
2.3. Solid State Fermentation Bacteria
2.4. Preparation of Extracts
2.5. Spectrophotometric Evaluation
2.6. UPLC-DAD Evaluation
2.7. Statistical Analysis
3. Results and Discussion
3.1. Variation of Total Phenolic Compounds, Flavonoid Content, and Radical Scavenging Activity in Fermented and Non-Fermented Bee-Collected Pollen
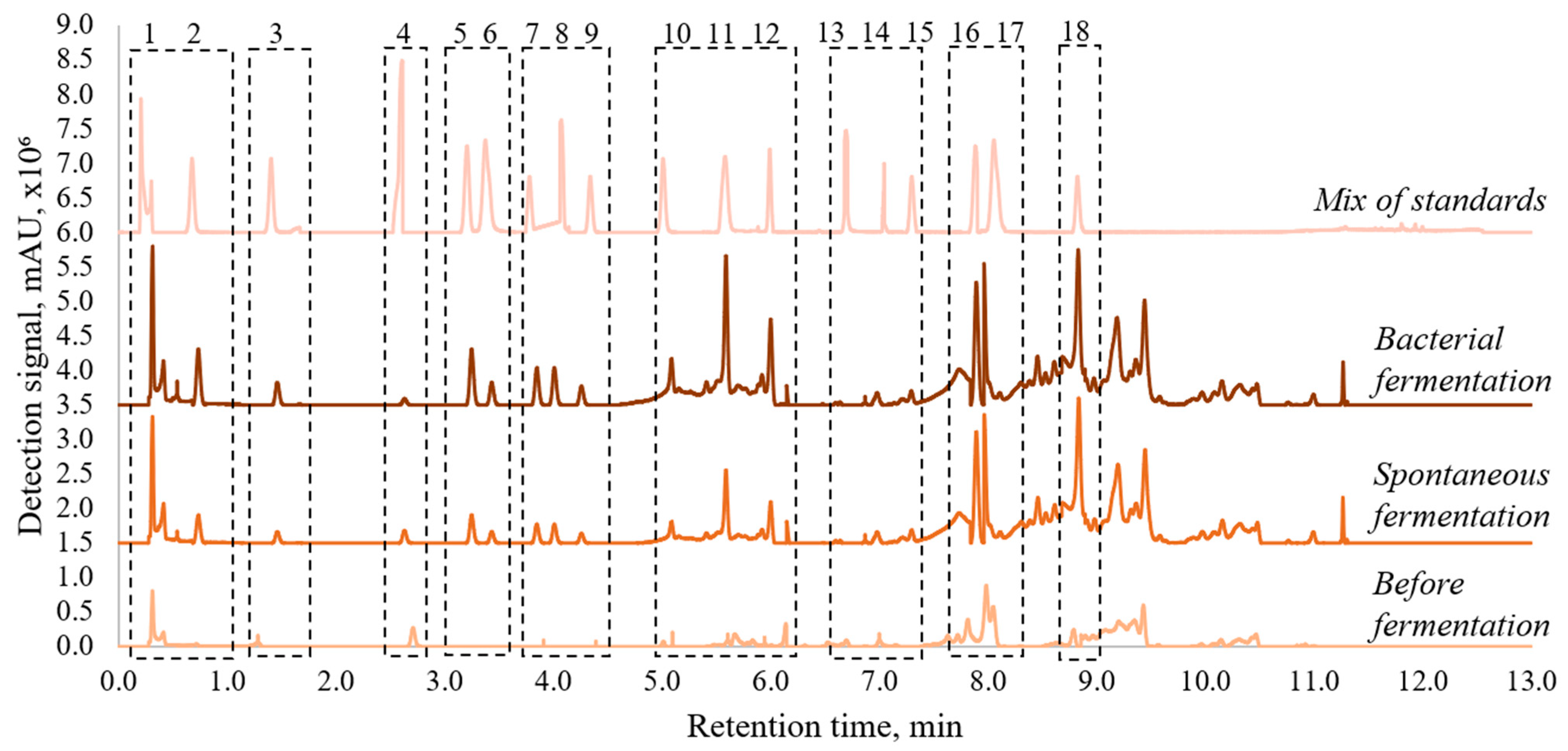
3.2. Qualitative and Qualitative Analysis of Phenolic Compounds and Lactic Acid
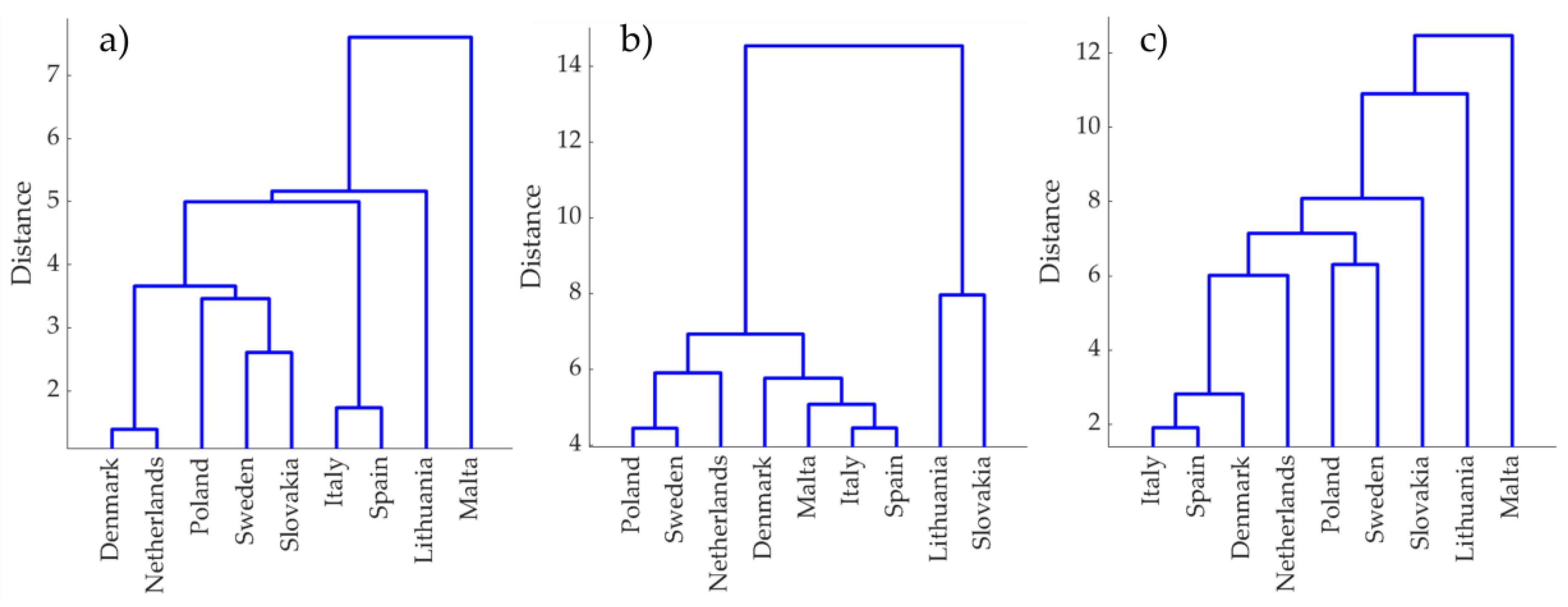
3.3. Chemometric Analysis of the Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kieliszek, M.; Piwowarek, K.; Kot, A.M.; Błażejak, S.; Chlebowska-Śmigiel, A.; Wolska, I. Pollen and Bee Bread as New Health-oriented Products: A review. Trends Food Sci. Technol. 2018, 71, 170–180. [Google Scholar] [CrossRef]
- Denisow, B.; Denisow-Pietrzyk, M. Biological and Therapeutic Properties of Bee Pollen: A review. J. Sci. Food Agric. 2016, 96, 4303–4309. [Google Scholar] [CrossRef] [PubMed]
- Komosinska-Vassev, K.; Olczyk, P.; Kaźmierczak, J.; Mencner, L.; Olczyk, K. Bee Pollen: Chemical Composition and Therapeutic Application. Evid.-Based Complement. Altern. Med. 2015, 2015, 297425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mărgăoan, R.; Strant, M.; Varadi, A.; Topal, E.; Yücel, B.; Cornea-Cipcigan, M.; Campos, M.G.; Vodnar, D.C. Bee Collected Pollen and Bee Bread: Bioactive Constituents and Health Benefits. Antioxidants 2019, 8, 568. [Google Scholar] [CrossRef] [Green Version]
- Kaškonienė, V.; Adaškevičiūtė, V.; Kaškonas, P.; Mickienė, R.; Maruška, A. Antimicrobial and Antioxidant Activities of Natural and Fermented Bee Pollen. Food Biosci. 2020, 34, 100532–100541. [Google Scholar] [CrossRef]
- Kaškonienė, V.; Katilevičiūtė, A.; Kaškonas, P.; Maruška, A. The Impact of Solid State Fermentation on Bee Pollen Phenolic Compounds and Radical Scavenging Capacity. Chem. Pap. 2018, 72, 2115–2120. [Google Scholar] [CrossRef]
- Adaškevičiūtė, V.; Kaškonienė, V.; Maruška, A. Optimization of Conditions for Bee Pollen Lactic Acid Fermentation. In Proceedings of the CYSENI 2021: The 17th International Conference of Young Scientists on Energy Issues, Kaunas, Lithuania, 24–28 May 2021; Volume 17, pp. 1099–1106. [Google Scholar]
- Kaškonienė, V.; Kaškonas, P.; Maruška, A. Volatile Compounds Composition and Antioxidant Activity of Bee Pollen Collected in Lithuania. Chem. Pap. 2015, 69, 291–299. [Google Scholar] [CrossRef]
- Adaškevičiūtė, V.; Kaškonienė, V.; Kaškonas, P.; Barčauskaitė, K.; Maruška, A. Comparison of Physicochemical Properties of Bee Pollen with Other Bee Products. Biomolecules 2019, 9, 819. [Google Scholar] [CrossRef] [Green Version]
- Goh, E.; Gledhill, A. Analysis of Polyphenols in Fruit Juices Using ACQUITY UPLC H-Class with UV and MS Detection. Cardiovasc. Dis. 2011, 1, 3–17. [Google Scholar]
- Gómez-Caravaca, A.M.; Maggio, R.M.; Cerretani, L. Chemometric Applications to Assess Quality and Critical Parameters of Virgin and Extra-Virgin Olive Oil. A Review. Anal. Chim. Acta 2016, 913, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Kaškonienė, V.; Kaškonas, P.; Maruška, A.; Ragažinskienė, O. Essential Oils of Bidens tripartita L. Collected during Period of 3 Years Composition Variation Analysis. Acta Physiol. Plant. 2013, 35, 1171–1178. [Google Scholar] [CrossRef]
- Yan, S.; Li, Q.; Xue, X.; Wang, K.; Zhao, L.; Wu, L. Analysis of Improved Nutritional Composition of Bee Pollen (Brassica campestris L.) After Different Fermentation Treatments. Int. J. Food Sci. 2019, 54, 2169–2181. [Google Scholar] [CrossRef]
- Zuluaga, C.M.; Serratob, J.C.; Quicazan, M.C. Bee-pollen Structure Modification by Physical and Biotechnological Processing: Influence on the Availability of Nutrients and Bioactive Compounds. Chem. Eng. Trans. 2015, 43, 79–84. [Google Scholar]
- Bors, W.; Heller, W.; Michel, C.; Saran, M. Radical Chemistry of Flavonoid Antioxidants. In Antioxidants in Therapy and Preventive Medicine; Springer: Berlin/Heidelberg, Germany, 1990; pp. 165–170. [Google Scholar]
- Platzer, M.; Kiese, S.; Herfellner, T.; Schweiggert-Weisz, U.; Eisner, P. How Does the Phenol Structure Influence the Results of the Folin-Ciocalteu Assay? Antioxidants 2021, 10, 811. [Google Scholar] [CrossRef] [PubMed]
- Zuluaga-Dominguez, C.M.; Quicazan, M. Effect of Fermentation on Structural Characteristics and Bioactive Compounds of Bee-pollen Based Food. J. Apic. Sci. 2019, 63, 209–222. [Google Scholar] [CrossRef] [Green Version]
- Munoz, R.; de las Rivas, B.; Lopez de Felipe, F.; Reveron, I.; Santamaría, R.L.; Esteban-Torres, M.; Curiel, J.A.; Rodríguez, H.; Iranzo, J.M. Biotransformation of Phenolics by Lactobacillus plantarum in Fermented Foods. In Fermented Foods in Health and Disease Prevention; Frias, J., Martinez-Villaluenga, C., Peñas, E., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 63–83. [Google Scholar]
- Shirsat, D.; Kad, S.K.; Wakhle, D.M. Solid State Fermentation of Bee-collected Pollen. Int. J. Curr. Microbiol. 2019, 8, 1557–1563. [Google Scholar] [CrossRef]
- Perveen, S.; Orfali, R.; Al-Taweel, A.; Khan, A.; Alghanem, B.; Shaibah, H. Simultaneous Identification of Phenolic and Flavonoid Contents in Bee Pollen by HPLC-ESI-MS data. Biomed. Res. 2019, 30, 770–774. [Google Scholar] [CrossRef] [Green Version]
- Waś, E.; Szczesna, T.; Rybak-Chmielewska, H.; Teper, D.; Jaśkiewicz, K. Application of HPLC-DAD Technique for Determination of Phenolic Compounds in Bee Pollen Loads. J. Apic. Sci. 2017, 61, 153–162. [Google Scholar] [CrossRef] [Green Version]
- Rzepecka-Stojko, A.; Stojko, J.; Kurek-Górecka, A.; Górecki, M.; Kabała-Dzik, A.; Kubina, R.; Moździerz, A.; Buszman, E. Polyphenols from Bee Pollen: Structure, Absorption, Metabolism and Biological Activity. Molecules 2015, 20, 21732–21749. [Google Scholar] [CrossRef] [Green Version]
- Freire, K.; Lins, A.; Dorea, M.; Santos, F.; Camara, C.; Silva, T. Palynological Origin, Phenolic Content and Antioxidant Properties of Honeybee-collected Pollen from Bahia Brazil. Molecules 2012, 17, 1652–1664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ulusoy, E.; Kolayli, S. Phenolic Composition and Antioxidant Properties of Anzer Bee Pollen. J. Food Biochem. 2014, 38, 73–82. [Google Scholar] [CrossRef]
- Boguta, A.M.; Bringel, F.; Martinussen, J.; Jensen, P.R. Screening of Lactic Acid Bacteria for Their Potential as Microbial Cell Factories for Bioconversion of Lignocellulosic Feedstocks. Microb. Cell Factories 2014, 13, 97–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borghi, A.A.; Palma, M.S.A. Tetracycline: Production, Waste, Treatment and Environmental Impact Assessment. Braz. J. Pharm. Sci. 2014, 50, 25–40. [Google Scholar] [CrossRef]
- Garmienė, G.; Šalomskienė, J.; Jasutienė, I.; Mačionienė, I.; Miliauskienė, I. Production of Benzoic Acid by Lactic Acid Bacteria from Lactobacillus, Lactococcus and Streptococcus Genera in Milk. Milchwissenschaft 2010, 65, 295–298. [Google Scholar]
- Santos, A.; Schieber, A.; Weber, F. Site-specific Hydrolysis of Chlorogenic Acids by Selected Lactobacillus Species. Int. Food Res. J. 2018, 109, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Fritsch, C.; Heinrich, V.; Vogel, R.F.; Toelstede, S. Phenolic Acid Degradation Potential and Growth Behavior of Lactic Acid Bacteria in Sunflower Substrates. Food Microbiol. 2016, 57, 178–186. [Google Scholar] [CrossRef]
- Sawicki, T.; Starowicz, M.; Kłębukowska, L.; Hanus, P. The Profile of Polyphenolic Compounds, Contents of Total Phenolics and Flavonoids, and Antioxidant and Antimicrobial Properties of Bee Products. Molecules 2022, 27, 1301. [Google Scholar] [CrossRef]
- Venkatesan, T.; Choi, Y.W.; Kim, Y.K. Impact of Different Extraction Solvents on Phenolic Content and Antioxidant Potential of Pinus densiflora Bark Extract. Biomed. Res. Int. 2019, 2019, 3520675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Country | Location | GPS Coordinates | Collection Period |
|---|---|---|---|
| Lithuania | Šiauliai region, Kuršėnai | 55°59′ N 22°55′ E | August 2018 |
| Poland | Bialystok | 53°08′ N 23°08′ E | July 2018 |
| Sweden | Hagfors region | 60°02′ N 13°39′ E | August 2018 |
| Denmark | Alsgarde region | 56°04′ N 12°32′ E | August 2018 |
| Slovakia | Trnava region | 48°22′ N 17°35′ E | June 2018 |
| The Netherlands | South Holland, Gouda | 52°0′ N 4°42′ E | August 2018 |
| Republic of Malta | Northern region, Mellieha | 35°57′ N 14°21′ E | August 2018 |
| Italy | Bibbiena region | 43°42′ N 11°49′ E | 2018 |
| Spain | Valencia region | 39°28′ N 0°22′ W | May 2018 |
| Method | Total Phenolic Compounds Content | Total Flavonoid Content | Radical Scavenging Activity |
|---|---|---|---|
| Sample | 8 µL of extract 8 µL Folin-Ciocalteu reagent 240 µL 4% Na2CO3 | 10 µL of extract 240 µL AlCl3 colorimetric stock solution 1 | 5.5 µL of extract 225 µL DPPH reagent |
| Blank | 8 µL of 80% CH3OH 8 µL Folin-Ciocalteu reagent 240 µL 4% Na2CO3 | 10 µL of 80% CH3OH 240 µL AlCl3 colorimetric stock solution 1 | 5.5 µL of 80% CH3OH 225 µL DPPH reagent |
| Incubation duration | 30 min | 30 min | 15 min |
| Incubation conditions | Room temperature | +4 °C | Room temperature, dark |
| Wavelength | 694 nm | 405 nm | 515 nm |
| Compound | Retention Time, min | Linear Regression Equation | R2 |
|---|---|---|---|
| Lactic acid | 0.30 ± 0.01 | y = 1.967 × 103x + 0.561 × 103 | 0.9913 |
| Gallic acid | 0.55 ± 0.05 | y = 1.779 × 105x + 0.511 × 103 | 0.9996 |
| Benzoic acid | 1.23 ± 0.02 | y = 4.066 × 105x − 9.802 × 103 | 0.9992 |
| Chlorogenic acid | 2.55 ± 0.20 | y = 2.586 × 105x + 13.606 × 103 | 0.9991 |
| Vanillic acid | 3.12 ± 0.02 | y = 4.022 × 105x + 7.491 × 103 | 0.9988 |
| Caffeic acid | 3.41 ± 0.05 | y = 2.103 × 105x + 15.115 × 103 | 0.9989 |
| Syringic acid | 3.78 ± 0.03 | y = 2.552 × 105x − 2.430 × 103 | 0.9993 |
| Salicylic acid | 4.06 ± 0.10 | y = 0.644 × 105x + 0.347 × 103 | 0.9996 |
| Coumaric acid | 4.34 ± 0.06 | y = 2.834 × 105x + 1.135 × 103 | 0.9991 |
| Ferulic acid | 4.96 ± 0.04 | y = 2.984 × 105x + 3.121 × 103 | 0.9994 |
| Rutin | 5.49 ± 0.06 | y = 2.733 × 105x + 5.876 × 103 | 0.9974 |
| Ellagic acid | 5.59 ± 0.02 | y = 7.343 × 105x − 39.101 × 103 | 0.9994 |
| Coumarin | 5.84 ± 0.03 | y = 4.232 × 105x + 0.726 × 103 | 0.9975 |
| Myricetin | 6.82 ± 0.04 | y = 2.293 × 105x − 13.132 × 103 | 0.9987 |
| Hesperidin | 7.09 ± 0.02 | y = 8.740 × 103x + 0.431 × 103 | 0.9995 |
| Quercetin | 7.92 ± 0.05 | y = 4.158 × 105x − 15.251 × 103 | 0.9991 |
| Luteolin | 8.07 ± 0.08 | y = 2.753 × 105x − 1.610 × 103 | 0.9996 |
| Naringenin | 9.00 ± 0.04 | y = 1.293 × 105x − 3.407 × 103 | 0.9982 |
| Sample | Total Phenolic Compounds Content, mg/g (RUE) | Total Flavonoid Content, mg/g (RUE) | Radical Scavenging Activity, mg/g (RUE) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Before Fermentation | Fermentation Type | Before Fermentation | Fermentation Type | Before Fermentation | Fermentation Type | ||||
| Bacterial | Spontaneous | Bacterial | Spontaneous | Bacterial | Spontaneous | ||||
| Lithuanian | 11.97 | 22.63 i | 17.99 i | 6.26 | 9.67 i | 8.85 i | 9.23 | 14.69 i | 13.62 i |
| Polish | 10.94 | 16.23 i | 13.75 i | 4.87 | 7.17 i | 6.05 i | 6.67 | 11.20 i | 10.24 i |
| Swedish | 11.67 | 17.57 i | 14.02 i | 5.22 | 8.21 i | 6.26 i | 7.38 | 12.21 i | 11.57 i |
| Danish | 9.81 | 12.15 i | 10.50 i | 4.78 | 6.32 i | 5.50 i | 6.12 | 10.12 i | 9.32 i |
| Slovak | 11.29 | 19.98 i | 18.49 i | 5.36 | 8.24 i | 7.20 i | 8.08 | 13.04 i | 11.82 i |
| Dutch | 9.81 | 14.22 i | 12.62 i | 4.81 | 6.74 i | 5.62 i | 6.53 | 10.27 i | 8.86 i |
| Maltese | 8.08 | 10.68 i | 9.28 i | 3.69 | 5.41 i | 5.00 i | 2.33 | 5.44 i | 4.98 i |
| Italian | 8.67 | 12.46 i | 10.34 i | 4.34 | 6.02 i | 5.16 i | 4.56 | 8.91 i | 6.17 i |
| Spanish | 9.10 | 12.65 i | 10.19 i | 4.58 | 6.11 i | 5.21 i | 4.74 | 9.44 i | 7.56 i |
| Mean | 10.15 | 15.40 | 13.02 | 4.88 | 7.10 | 6.09 | 6.18 | 10.59 | 9.35 |
| SD | 1.39 | 4.02 | 3.39 | 0.71 | 1.36 | 1.24 | 2.07 | 2.66 | 2.8 |
| Min | 8.08 | 10.68 | 9.28 | 3.69 | 5.41 | 5.00 | 2.33 | 5.44 | 4.98 |
| Max | 11.97 | 22.63 | 18.49 | 6.26 | 9.67 | 8.85 | 9.23 | 14.69 | 13.62 |
| Type of Fermentation | Concentration, µg/g | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Denmark | Sweden | Poland | Lithuania | Slovakia | Netherlands | Italy | Spain | Malta | |
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| Gallic acid | |||||||||
| Before fermentation | 3.51 ± 0.13 | nd | 7.01 ± 0.23 | 31.42 ± 0.23 | 8.84 ± 0.50 | 2.31 ± 0.21 | nd | 2.11 ± 0.30 | 11.44 ± 0.22 |
| Bacterial | 4.58 ± 0.24 i | 9.43 ± 0.42 i | 9.93 ± 0.13 i | 52.71 ± 0.41 i | 22.13 ± 0.20 i | 5.52 ± 0.23 i | nd | 4.81 ± 0.13 i | 22.71 ± 0.43 i |
| Spontaneous | 3.33 ± 0.21 | 7.31 ± 0.32 i | 8.14 ± 0.41 i | 42.12 ± 0.42 i | 14.10 ± 0.21 i | 4.30 ± 0.24 i | nd | 3.64 ± 0.11 i | 20.13 ± 0.42 i |
| Benzoic acid | |||||||||
| Before fermentation | nd | nd | nd | 5.51 ± 0.41 | 2.11 ± 0.42 | 1.81 ± 0.21 | 1.12 ± 0.22 | nd | 0.54 ± 0.20 |
| Bacterial | 3.12 ± 0.43 i | 5.50 ± 0.22 i | 3.82 ± 0.11 i | 9.89 ± 0.44 i | 4.93 ± 0.11 i | 4.23 ± 0.23 i | 1.91 ± 0.21 i | 2.11 ± 0.12 i | 1.50 ± 0.11 i |
| Spontaneous | 2.41 ± 0.31 i | 4.12 ± 0.41 i | 2.93 ± 0.44 i | 7.64 ± 0.30 i | 3.34 ± 0.42 i | 3.11 ± 0.21 i | 1.44 ± 0.21 | 1.63 ± 0.21 i | 1.13 ± 0.21 i |
| Chlorogenic acid | |||||||||
| Before fermentation | 3.12 ± 0.43 | 3.82 ± 0.42 | 2.71 ± 0.40 | 4.22 ± 0.40 | 5.51 ± 0.10 | 3.71 ± 0.22 | 1.84 ± 0.11 | 2.50 ± 0.14 | nd |
| Bacterial | 2.01 ± 0.32 d | 2.73 ± 0.34 d | 2.04 ± 0.20 d | 3.19 ± 0.11 d | nd d | nd d | 0.93 ± 0.24 d | 1.64 ± 0.12 d | nd |
| Spontaneous | 2.53 ± 0.31 d | 3.12 ± 0.41 | 2.42 ± 0.41 | 3.62 ± 0.10 d | nd d | nd d | 1.44 ± 0.12 d | 2.03 ± 0.11 d | nd |
| Vanillic acid | |||||||||
| Before fermentation | nd | nd | nd | nd | nd | nd | 1.14 ± 0.31 | nd | nd |
| Bacterial | 2.42 ± 0.41 i | 46.60 ± 0.61 i | 58.72 ± 0.21 i | 68.90 ± 0.51 i | 43.32 ± 0.51 i | 34.10 ± 0.20 i | 22.38 ± 0.54 i | 23.42 ± 0.12 i | 21.64 ± 0.43 i |
| Spontaneous | 1.91 ± 0.42 i | 31.63 ± 0.41 i | 40.22 ± 0.54 i | 45.83 ± 0.12 i | 32.81 ± 0.62 i | 22.24 ± 0.51 i | 15.13 ± 0.40 i | 13.43 ± 0.41 i | 12.61 ± 0.21 i |
| Caffeic acid | |||||||||
| Before fermentation | 2.32 ± 0.23 | nd | 2.14 ± 0.11 | nd | nd | 1.92 ± 0.21 | 2.53 ± 0.10 | nd | 2.12 ± 0.31 |
| Bacterial | 5.49 ± 0.21 i | nd | 5.70 ± 0.41 i | 8.14 ± 0.23 i | nd | 4.91 ± 0.12 i | 4.24 ± 0.21 i | nd | 4.84 ± 0.13 i |
| Spontaneous | 4.34 ± 0.24 i | nd | 4.42 ± 0.30 i | 6.33 ± 0.31 i | nd | 3.83 ± 0.44 i | 3.14 ± 0.23 i | nd | 3.63 ± 0.12 i |
| Syringic acid | |||||||||
| Before fermentation | 3.51 ± 0.11 | nd | 7.04 ± 0.20 | 8.81 ± 0.51 | 6.63 ± 0.33 | 3.31 ± 0.22 | 1.92 ± 0.21 | 2.14 ± 0.33 | 1.54 ± 0.20 |
| Bacterial | 4.62 ± 0.24 i | 9.41 ± 0.41 i | nd d | 22.11 ± 0.22 i | 13.44 ± 0.42 i | 5.49 ± 0.20 i | 3.13 ± 0.12 i | 4.13 ± 0.11 i | 1.91 ± 0.14 i |
| Spontaneous | 3.33 ± 0.21 | 7.32 ± 0.31 i | nd d | 14.13 ± 0.24 i | 9.42 ± 0.31 i | 4.32 ± 0.21 i | 2.24 ± 0.11 | 3.02 ± 0.13 i | 1.70 ± 0.11 |
| Salicylic acid | |||||||||
| Before fermentation | 10.39 ± 0.31 | 14.51 ± 0.61 | 13.67 ± 0.37 | 17.71 ± 0.37 | 16.61 ± 0.40 | nd | nd | 7.11 ± 0.20 | 6.71 ± 0.31 |
| Bacterial | 17.59 ± 0.44 i | 21.30 ± 0.43 i | 18.76 ± 0.39 i | 23.31 ± 0.49 i | 22.10 ± 0.51 i | nd | nd | 14.09 ± 0.21 i | 10.39 ± 0.46 i |
| Spontaneous | 11.91 ± 0.10 i | 19.61 ± 0.12 i | 16.21 ± 0.20 i | 20.16 ± 0.19 i | 19.83 ± 0.55 i | nd | nd | 12.21 ± 0.45 i | 9.45 ± 0.41 i |
| Coumaric acid | |||||||||
| Before fermentation | 1.31 ± 0.22 | nd | nd | 3.61 ± 0.24 | 1.91 ± 0.21 | 1.04 ± 0.30 | 0.31 ± 0.11 | 0.63 ± 0.21 | nd |
| Bacterial | 2.22 ± 0.13 i | nd | nd | 5.63 ± 0.21 i | 3.71 ± 0.21 i | 2.44 ± 0.21 i | 1.54 ± 0.21 i | 2.44 ± 0.64 i | nd |
| Spontaneous | 1.61 ± 0.20 | nd | nd | 4.24 ± 0.20 i | 2.92 ± 0.30 i | 1.64 ± 0.30 | 1.14 ± 0.10 i | 1.30 ± 0.41 i | nd |
| Ferulic acid | |||||||||
| Before fermentation | 13.12 ± 0.23 | 26.61 ± 0.63 | 23.10 ± 0.44 | 38.14 ± 0.54 | 27.71 ± 0.42 | 13.42 ± 0.33 | 9.10 ± 0.34 | 9.42 ± 0.32 | 7.91 ± 0.11 |
| Bacterial | 31.31 ± 0.41 i | 46.64 ± 0.61 i | 43.32 ± 0.52 i | 68.90 ± 0.51 i | 58.73 ± 0.24 i | 34.13 ± 0.21 i | 22.43 ± 0.54 i | 23.43 ± 0.13 i | 21.63 ± 0.44 i |
| Spontaneous | 21.94 ± 0.40 i | 31.62 ± 0.41 i | 32.84 ± 0.64 i | 45.83 ± 0.10 i | 40.22 ± 0.51 i | 22.24 ± 0.50 i | 15.12 ± 0.41 i | 13.44 ± 0.41 i | 12.62 ± 0.20 i |
| Coumarin | |||||||||
| Before fermentation | 7.11 ± 0.22 | 8.92 ± 0.21 | nd | nd | nd | nd | 6.72 ± 0.33 | nd | nd |
| Bacterial | 21.32 ± 0.42 i | 26.63 ± 0.62 i | nd | nd | nd | nd | 10.41 ± 0.54 i | nd | nd |
| Spontaneous | 11.91 ± 0.10 i | 21.61 ± 0.13 i | nd | nd | nd | nd | 9.52 ± 0.41 i | nd | nd |
| Ellagic acid | |||||||||
| Before fermentation | 11.12 ± 0.22 | 11.41 ± 0.63 | 17.72 ± 0.43 | 28.12 ± 0.22 | 19.91 ± 0.10 | 10.43 ± 0.34 | 3.41 ± 0.11 | 6.61 ± 0.33 | 2.71 ± 0.13 |
| Bacterial | 19.31 ± 0.44 i | 23.33 ± 0.51 i | 46.71 ± 0.22 i | 58.91 ± 0.42 i | 36.62 ± 0.62 i | 14.12 ± 0.21 i | 7.52 ± 0.23 i | 9.43 ± 0.14 i | 5.62 ± 0.24 i |
| Spontaneous | 18.43 ± 0.12 i | 12.82 ± 0.62 i | 30.22 ± 0.21 i | 35.82 ± 0.11 i | 21.63 ± 0.11 i | 12.21 ± 0.50 i | 6.64 ± 0.21 i | 8.34 ± 0.42 i | 3.90 ± 0.21 i |
| Rutin | |||||||||
| Before fermentation | 31.12 ± 0.20 | 41.61 ± 0.31 | 41.70 ± 0.43 | 54.12 ± 0.52 | 48.13 ± 0.43 | 33.12 ± 0.30 | 27.61 ± 0.33 | 27.40 ± 0.33 | 21.40 ± 0.21 |
| Bacterial | 52.34 ± 0.42 i | 64.64 ± 0.63 i | 75.74 ± 0.21 i | 89.93 ± 0.53 i | 63.32 ± 0.54 i | 57.73 ± 0.22 i | 44.63 ± 0.54 i | 41.21 ± 0.14 i | 40.04 ± 0.41 i |
| Spontaneous | 42.93 ± 0.41 i | 56.63 ± 0.42 i | 61.23 ± 0.52 i | 64.84 ± 0.12 i | 52.22 ± 0.61 i | 45.10 ± 0.51 i | 37.14 ± 0.42 i | 33.43 ± 0.42 i | 30.63 ± 0.21 i |
| Myricetin | |||||||||
| Before fermentation | 6.52 ± 0.14 | nd | nd | 5.69 ± 0.09 | 6.51 ± 0.10 | 4.86 ± 0.21 | 6.49 ± 0.33 | 1.50 ± 0.41 | 4.44 ± 0.50 |
| Bacterial | 16.29 ± 0.43 i | 11.59 ± 0.33 i | 18.71 ± 0.43 i | 17.10 ± 0.21 i | 18.12 ± 0.43 i | 8.41 ± 0.16 i | 8.91 ± 0.41 i | 6.23 ± 0.09 i | 11.61 ± 0.41 i |
| Spontaneous | 14.89 ± 0.22 i | 8.91 ± 0.40 i | 10.91 ± 0.10 i | 16.01 ± 0.19 i | 7.68 ± 0.21 i | 8.03 ± 0.20 i | 7.43 ± 0.86 i | 4.34 ± 0.20 i | 10.89 ± 0.12 i |
| Hesperidin | |||||||||
| Before fermentation | nd | nd | nd | 11.55 ± 0.21 | nd | 5.41 ± 0.21 | nd | nd | 2.12 ± 0.19 |
| Bacterial | 19.09 ± 0.35 i | nd | nd | 26.51 ± 0.18 i | nd | 10.20 ± 0.35 i | nd | 9.93 ± 0.13 i | 6.64 ± 0.21 i |
| Spontaneous | 9.11 ± 0.19 i | nd | nd | 19.39 ± 0.19 i | nd | 9.12 ± 0.29 i | nd | 6.88 ± 0.11 i | 3.86 ± 0.45 i |
| Quercetin | |||||||||
| Before fermentation | 22.72 ± 0.76 | 40.21 ± 0.77 | 36.31 ± 0.19 | 58.10 ± 0.48 | 47.71 ± 0.38 | 7.11 ± 0.12 | 9.03 ± 0.28 | 4.10 ± 0.41 | 6.62 ± 0.41 |
| Bacterial | 40.29 ± 0.51 i | 61.09 ± 0.45 i | 49.12 ± 0.29 i | 78.86 ± 0.46 i | 68.69 ± 0.19 i | 21.40 ± 0.11 i | 20.41 ± 0.46 i | 8.13 ± 0.24 i | 20.59 ± 0.36 i |
| Spontaneous | 30.81 ± 0.55 i | 45.88 ± 0.10 i | 44.43 ± 0.22 i | 65.81 ± 0.11 i | 60.21 ± 0.12 i | 10.41 ± 0.35 i | 11.10 ± 0.39 i | 7.31 ± 0.29 i | 9.57 ± 0.19 i |
| Luteolin | |||||||||
| Before fermentation | 33.10 ± 0.24 | 43.61 ± 0.26 | 43.12 ± 0.40 | 58.13 ± 0.49 | 47.68 ± 0.37 | 33.41 ± 0.30 | nd | 29.41 ± 0.27 | 29.61 ± 0.22 |
| Bacterial | 51.29 ± 0.40 i | 66.59 ± 0.61 i | 63.33 ± 0.51 i | 88.90 ± 0.46 i | 78.71 ± 0.22 i | 54.12 ± 0.21 i | 42.42 ± 0.51 i | 43.36 ± 0.10 i | 41.56 ± 0.35 i |
| Spontaneous | 41.90 ± 0.35 i | 51.60 ± 0.39 i | 52.79 ± 0.55 i | 65.81 ± 0.09 i | 60.21 ± 0.54 i | 42.23 ± 0.46 i | 35.14 ± 0.42 i | 33.39 ± 0.35 i | 32.62 ± 0.21 i |
| Naringenin | |||||||||
| Before fermentation | 27.61 ± 0.29 | 31.59 ± 0.30 | 41.69 ± 0.39 | 59.21 ± 0.51 | 49.69 ± 0.36 | 21.61 ± 0.29 | 34.41 ± 0.26 | 27.39 ± 0.30 | 21.09 ± 0.21 |
| Bacterial | 44.62 ± 0.51 i | 54.57 ± 0.60 i | 91.81 ± 0.21 i | 135.03 ± 0.49 i | 82.21 ± 0.49 i | 31.71 ± 0.56 i | 79.10 ± 0.19 i | 42.32 ± 0.36 i | 41.22 ± 0.09 i |
| Spontaneous | 37.13 ± 0.35 i | 46.61 ± 0.35 i | 67.82 ± 0.45 i | 124.81 ± 0.09 i | 74.13 ± 0.55 i | 28.71 ± 0.34 i | 45.13 ± 0.46 i | 33.41 ± 0.39 i | 32.91 ± 0.36 i |
| Criteria | Before Fermentation | Bacterial Fermentation | Spontaneous Fermentation | ||||||
|---|---|---|---|---|---|---|---|---|---|
| TPC | TFC | RSA | TPC | TFC | RSA | TPC | TFC | RSA | |
| TPC | 1 | 0.905 | 0.945 | 1 | 0.979 | 0.924 | 1 | 0.906 | 0.892 |
| TFC | 1 | 0.956 | 1 | 0.935 | 1 | 0.872 | |||
| RSA | 1 | 1 | 1 | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adaškevičiūtė, V.; Kaškonienė, V.; Barčauskaitė, K.; Kaškonas, P.; Maruška, A. The Impact of Fermentation on Bee Pollen Polyphenolic Compounds Composition. Antioxidants 2022, 11, 645. https://doi.org/10.3390/antiox11040645
Adaškevičiūtė V, Kaškonienė V, Barčauskaitė K, Kaškonas P, Maruška A. The Impact of Fermentation on Bee Pollen Polyphenolic Compounds Composition. Antioxidants. 2022; 11(4):645. https://doi.org/10.3390/antiox11040645
Chicago/Turabian StyleAdaškevičiūtė, Vaida, Vilma Kaškonienė, Karolina Barčauskaitė, Paulius Kaškonas, and Audrius Maruška. 2022. "The Impact of Fermentation on Bee Pollen Polyphenolic Compounds Composition" Antioxidants 11, no. 4: 645. https://doi.org/10.3390/antiox11040645
APA StyleAdaškevičiūtė, V., Kaškonienė, V., Barčauskaitė, K., Kaškonas, P., & Maruška, A. (2022). The Impact of Fermentation on Bee Pollen Polyphenolic Compounds Composition. Antioxidants, 11(4), 645. https://doi.org/10.3390/antiox11040645









