Analysis of Antioxidant Phytochemicals and Anti-Inflammatory Effect from Vitex rotundifolia L.f
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Extraction Method
2.3. DPPH and ABTS Assays of Total Extracts and Fractions
2.4. Fractionation and Separation of Marker Compounds 1–7
2.5. Chromatographic and Separation Conditions
2.6. Method Validation
2.6.1. Detection of Wavelength
2.6.2. Preparation of Calibration Standard Solution
2.7. Mass Analysis
2.8. Screened Antioxidants by DPPH-HPLC Analysis and Further Verified by ELISA Assay
2.9. Anti-Inflammatory Assay
2.9.1. Cell Culture
2.9.2. Measurement of Cell Viability
2.9.3. Measurement of Nitric Oxide (NO) Production
2.10. Statistical Analysis
3. Results
3.1. Screen DPPH and ABTS Activities Guided Extraction Solvent Selection
3.2. Antioxidant Properties of Fractions
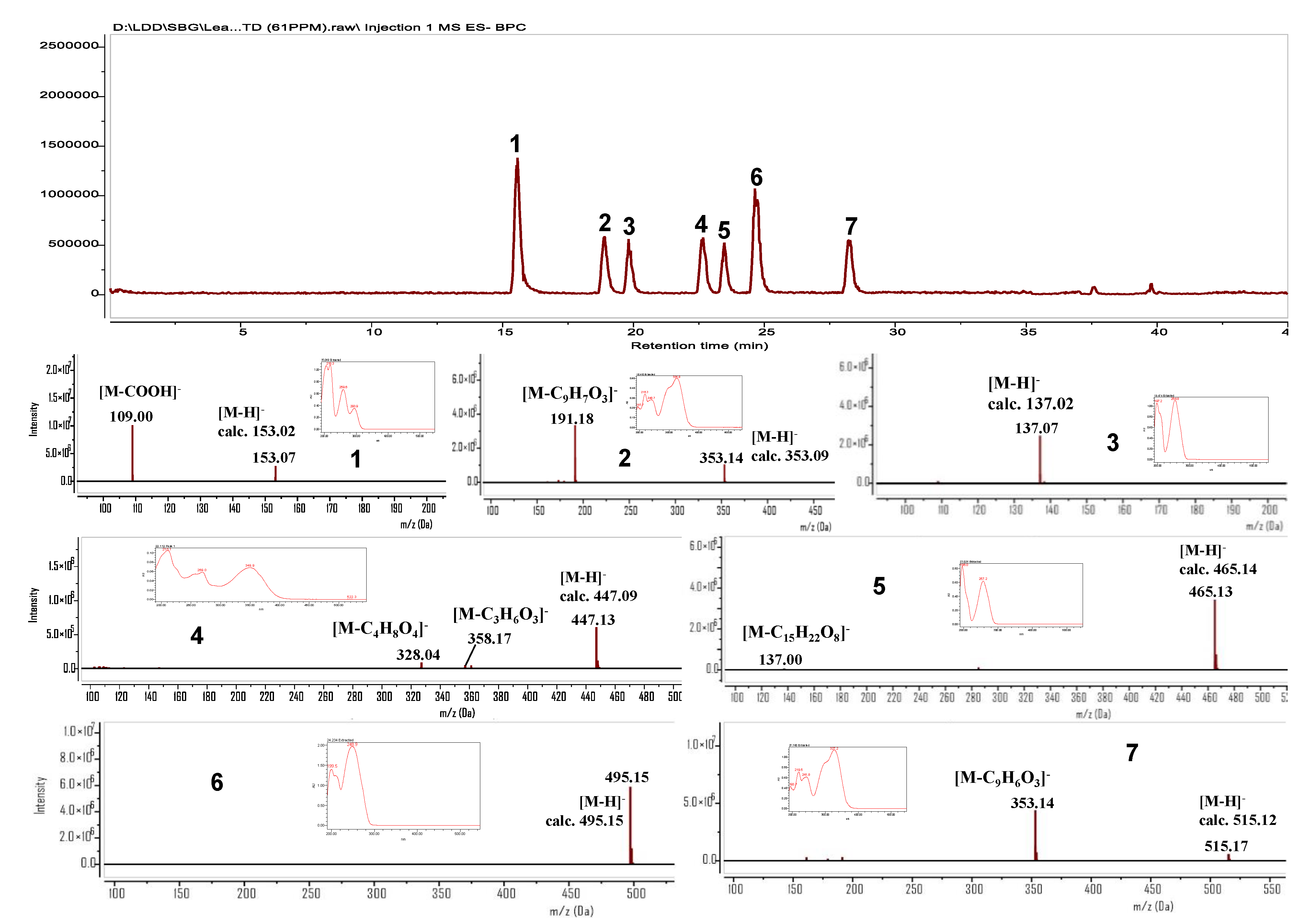
3.3. Isolation and Identification of Marker Compounds 1–7
Spectroscopic Data of Compounds 1–7
3.4. Establishment, Chemometric Profile, and Validation of HPLC/MS Analytical Method
3.4.1. Establishment of Analytical Method
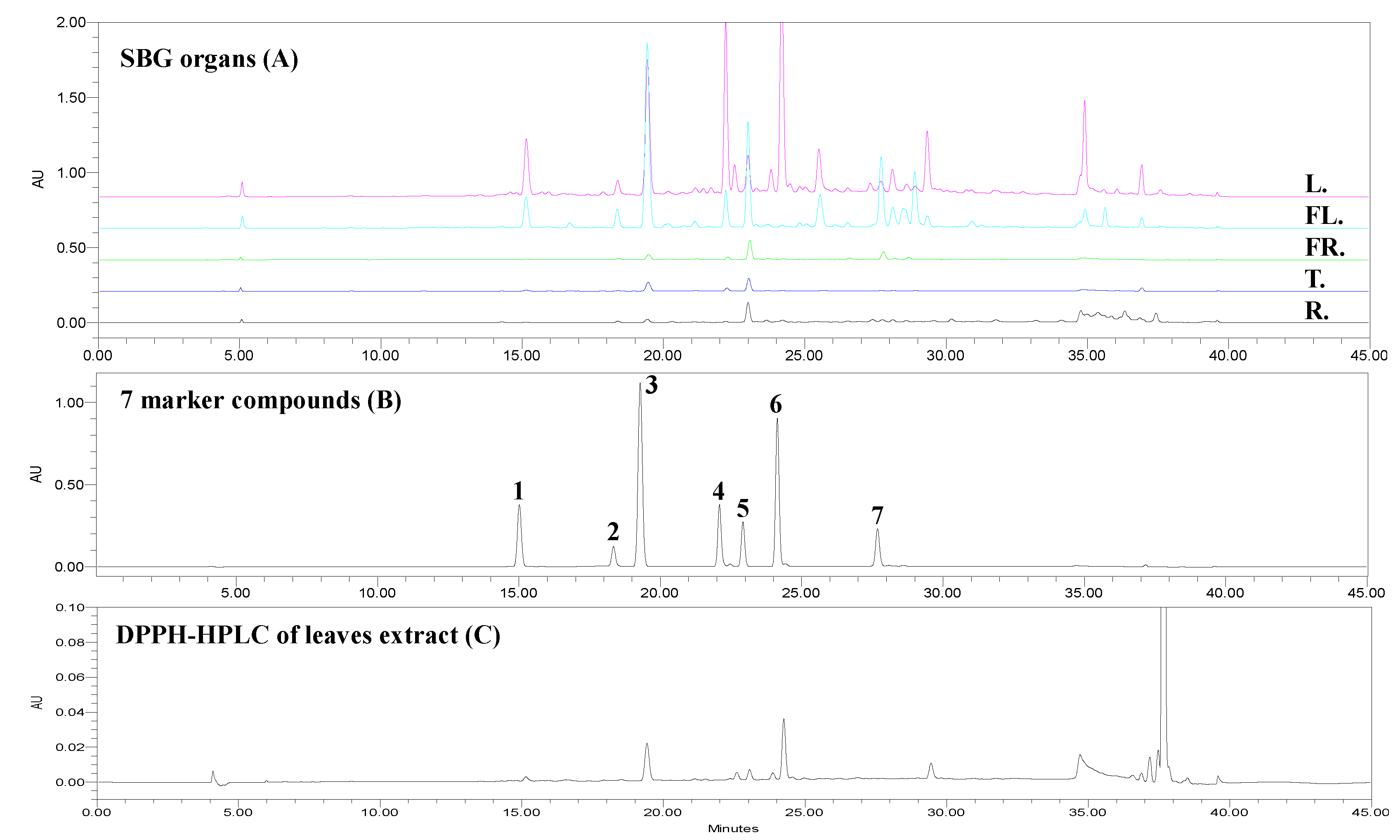
3.4.2. Chemical Profiles of Five Organs of V. rotundifolia
3.4.3. Validation and Quantification of Marker Compounds from Organs of V. rotundifolia
Linearity, LODs, and LOQs
3.5. Screen Antioxidants by DPPH-HPLC Method and ELISA Assay
3.6. Anti-Inflammatory Effects of Extracts and Fractions Derived from Five Organs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Semchyshyn, H.M.; Lozinska, L.M. Fructose protects baker’s yeast against peroxide stress: Potential role of catalase and superoxide dismutase. FEMS Yeast Res. 2012, 12, 761–773. [Google Scholar] [CrossRef] [Green Version]
- Bild, W.; Ciobica, A.; Padurariu, M.; Bild, V. The interdependence of the reactive species of oxygen, nitrogen, and carbon. J. Physiol. Biochem. 2013, 69, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Jacob, C. A scent of therapy: Pharmacological implications of natural products containing redox-active sulfur atoms. Nat. Prod. Rep. 2006, 23, 851–863. [Google Scholar] [CrossRef] [PubMed]
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free radicals: Properties, sources, targets, and their implication in various diseases. Indian J. Clin. Biochem. 2015, 30, 11–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paz-Elizur, T.; Sevilya, Z.; Leitner-Dagan, Y.; Elinger, D.; Roisman, L.C.; Livneh, Z. DNA repair of oxidative DNA damage in human carcinogenesis: Potential application for cancer risk assessment and prevention. Cancer Lett. 2008, 266, 60–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kowluru, R.A.; Kowluru, V.; Xiong, Y.; Ho, Y.-S. Overexpression of mitochondrial superoxide dismutase in mice protects the retina from diabetes-induced oxidative stress. Free. Radic. Biol. Med. 2006, 41, 1191–1196. [Google Scholar] [CrossRef]
- Shim, S.-Y.; Lee, Y.E.; Lee, M. Antioxidant Compounds, Kirenol and Methyl ent-16α, 17-dihydroxy-kauran-19-oate Bioactivity-Guided Isolated from Siegesbeckia glabrescens Attenuates MITF-Mediated Melanogenesis via Inhibition of Intracellular ROS Production. Molecules 2021, 26, 1940. [Google Scholar] [CrossRef]
- Yanfei, B.; Tianshuang, X.; Rui, J.; Yaoli, G.; Yiya, G.; Qi, Y.; Chengjian, Z. Vitex Diterpenoids: Structural Diversity and Pharmacological Activity. Curr. Pharm. Des. 2020, 26, 138–159. [Google Scholar]
- Yao, J.-L.; Fang, S.-M.; Liu, R.; Oppong, M.B.; Liu, E.-W.; Fan, G.-W.; Zhang, H. A Review on the Terpenes from Genus Vitex. Molecules 2016, 21, 1179. [Google Scholar] [CrossRef] [Green Version]
- But, P.P.H.; Kimura, T.; Guo, J.-X.; Sung, C.K. Northeast Asia, Part I. In International Collation of Traditional and Folk Medicine; But, P.P.H., Ed.; World Scientific: Singapore, 1996; pp. 141–142. [Google Scholar]
- Sohn, S.-H.; Ko, E.; Oh, B.-G.; Kim, S.-H.; Kim, Y.; Shin, M.; Hong, M.; Bae, H. Inhibition effects of Vitex rotundifolia on inflammatory gene expression in A549 human epithelial cells. Ann. Allergy Asthma Immunol. 2009, 103, 152–159. [Google Scholar] [CrossRef]
- Ramezani, M.; Amin, G.; Jalili, E. Antinociceptive and anti-inflammatory effects of hydroalcohol extract of Vitex agnus-castus fruit. J. Shahrekord Univ. Med. Sci. 2010, 11, 46–51. [Google Scholar]
- Kim, J.-Y.; Shim, S.H. Anti-Atherosclerotic Effects of Fruits of Vitex rotundifolia and Their Isolated Compounds via Inhibition of Human LDL and HDL Oxidation. Biomolecules 2019, 9, 727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, K.S.; Chang, I.-M. Anti-Inflammatory Activity of Aucubin by Inhibition of Tumor Necrosis Factor-α Production in RAW 264.7 Cells. Planta Med. 2004, 70, 778–779. [Google Scholar] [CrossRef] [PubMed]
- Liou, C.-J.; Huang, W.-C. Casticin inhibits interleukin-1β-induced ICAM-1 and MUC5AC expression by blocking NF-κB, PI3K-Akt, and MAPK signaling in human lung epithelial cells. Oncotarget 2017, 8, 101175–101188. [Google Scholar] [CrossRef] [Green Version]
- Saklani, S.; Mishra, A.P.; Chandra, H.; Atanassova, M.S.; Stankovic, M.; Sati, B.; Shariati, M.A.; Nigam, M.; Khan, M.U.; Plygun, S.; et al. Comparative Evaluation of Polyphenol Contents and Antioxidant Activities between Ethanol Extracts of Vitex negundo and Vitex trifolia L. Leaves by Different Methods. Plants 2017, 6, 45. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.-Y.; Kim, S.; Shim, S.H. Anti-Atherosclerotic Activity of (3R)-5-Hydroxymellein from an Endophytic Fungus Neofusicoccum parvum JS-0968 Derived from Vitex rotundifolia through the Inhibition of Lipoproteins Oxidation and Foam Cell Formation. Biomolecules 2020, 10, 715. [Google Scholar] [CrossRef]
- Antolovich, M.; Prenzler, P.D.; Patsalides, E.; McDonald, S.; Robards, K. Methods for testing antioxidant activity. Analyst 2002, 127, 183–198. [Google Scholar] [CrossRef]
- Shim, S.-Y.; Lee, Y.E.; Song, H.Y.; Lee, M. p-Hydroxybenzoic Acid β-d-Glucosyl Ester and Cimidahurinine with Antimelanogenesis and Antioxidant Effects from Pyracantha angustifolia via Bioactivity-Guided Fractionation. Antioxidants 2020, 9, 258. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.; Du, N.; Wen, L.; Zhu, H.; Liu, F.; Wang, X.; Du, J.; Li, S. An Efficient Method for the Preparative Isolation and Purification of Flavonoid Glycosides and Caffeoylquinic Acid Derivatives from Leaves of Lonicera japonica Thunb. Using High Speed Counter-Current Chromatography (HSCCC) and Prep-HPLC Guided by DPPH-HPLC Experiments. Molecules 2017, 22, 229. [Google Scholar] [CrossRef] [Green Version]
- Jeong, D.E.; Shim, S.-Y.; Lee, M. Anti-inflammatory activity of phenylpropyl triterpenoids from Osmanthus fragrans var. aurantiacus leaves. Int. Immunopharmacol. 2020, 86, 106576. [Google Scholar] [CrossRef]
- Benahmed, M.; Akkal, S.; Elomri, A.; Laouer, H.; Vérité, P.; Seguin, E. Constituents from Bupleurum montanum (Coss. & Dur.) (Apiaceae). Arab. J. Chem. 2014, 7, 1065–1069. [Google Scholar]
- Lee, E.J.; Kim, J.S.; Kim, H.P.; Lee, J.-H.; Kang, S.S. Phenolic constituents from the flower buds of Lonicera japonica and their 5-lipoxygenase inhibitory activities. Food Chem. 2010, 120, 134–139. [Google Scholar] [CrossRef]
- Yoshioka, T.; Inokuchi, T.; Fujioka, S.; Kimura, Y. Phenolic Compounds and Flavonoids as Plant Growth Regulators from Fruit and Leaf of Vitex rotundifolia. Z. Für Nat. C 2004, 59, 509–514. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.K. Antioxidative Constituents from the Twigs of Vitex rotundifolia. Biomol. Ther. 2009, 17, 412–417. [Google Scholar] [CrossRef] [Green Version]
- Dutta, P.K.; Chowdhury, U.S.; Chakravarty, A.K.; Achari, B.; Pakrashi, S.C. Studies on indian medicinal plants-part LXXV: Nishindaside, a novel iridoid glycoside from vitex negundo. Tetrahedron 1983, 39, 3067–3072. [Google Scholar] [CrossRef]
- Sehgal, C.K.; Taneja, S.C.; Dhar, K.L.; Atal, C.K. 6′-p-hydroxybenzoylmussaenosidic acid-an iridoid glucoside from Vitex negundo. Phytochemistry 1983, 22, 1036–1038. [Google Scholar] [CrossRef]
- Hu, Y.; Zhang, Q.; Xin, H.; Qin, L.-P.; Lu, B.-R.; Rahman, K.; Zheng, H. Association between chemical and genetic variation of Vitex rotundifolia populations from different locations in China: Its implication for quality control of medicinal plants. Biomed. Chromatogr. 2007, 21, 967–975. [Google Scholar] [CrossRef]
- Ko, W.G.; Kang, T.H.; Lee, S.J.; Kim, N.Y.; Kim, Y.C.; Sohn, D.H.; Lee, B.H. Polymethoxyflavonoids from Vitex rotundifolia inhibit proliferation by inducing apoptosis in human myeloid leukemia cells. Food Chem. Toxicol. 2000, 38, 861–865. [Google Scholar] [CrossRef]
- Ko, W.G.; Kang, T.H.; Lee, S.J.; Kim, Y.C.; Lee, B.H. Rotundifuran, a labdane type diterpene from Vitex rotundifolia, induces apoptosis in human myeloid leukaemia cells. Phytother. Res. 2001, 15, 535–537. [Google Scholar] [CrossRef]
- Kakkar, S.; Bais, S. A Review on Protocatechuic Acid and Its Pharmacological Potential. ISRN Pharmacol. 2014, 2014, 952943. [Google Scholar] [CrossRef] [Green Version]
- Wei, M.; Chu, X.; Jiang, L.; Yang, X.; Cai, Q.; Zheng, C.; Ci, X.; Guan, M.; Liu, J.; Deng, X. Protocatechuic Acid Attenuates Lipolysaccharide-Induced Acute Lung Injury. Inflammation 2012, 35, 1169–1178. [Google Scholar] [CrossRef] [PubMed]
- Liang, N.; Kitts, D.D. Role of Chlorogenic Acids in Controlling Oxidative and Inflammatory Stress Conditions. Nutrients 2015, 8, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, Y.; Gong, X.; Zhang, L.; Jiang, R.; Yang, J.; Wang, B.; Wan, J. Chlorogenic acid ameliorated concanavalin A-induced hepatitis by suppression of Toll-like receptor 4 signaling in mice. Int. Immunopharmacol. 2017, 44, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Li, Y. Chondroprotective and anti-nociceptive effects of caffeoylquinic acid in osteoarthritis by downregulating catabolic activity and oxidative damage in chondrocytes. Biomed. Pharmacother. 2017, 93, 985–994. [Google Scholar] [CrossRef]
- Ono, M.; Yanaka, T.; Yamamoto, M.; Ito, Y.; Nohara, T. New Diterpenes and Norditerpenes from the Fruits of Vitex rotundifolia. J. Nat. Prod. 2002, 65, 537–541. [Google Scholar] [CrossRef]
- Lee, W.; Ku, S.-K.; Bae, J.-S. Vascular barrier protective effects of orientin and isoorientin in LPS-induced inflammation in vitro and in vivo. Vasc. Pharmacol. 2014, 62, 3–14. [Google Scholar] [CrossRef]
- Rios, J.L.; Mañez, S.; Paya, M.; Alcaraz, M.J. Antioxidant activity of flavonoids from Sideritis javalambrensis. Phytochemistry 1992, 31, 1947–1950. [Google Scholar] [CrossRef]
- Cotelle, N.; Bernier, J.L.; Catteau, J.P.; Henichart, J.P.; Pesando, D. Inhibition of Macrophage Superoxide Production by Two Prenylated Flavanones from Erythrina sigmoidea Hua. Nat. Prod. Lett. 1993, 3, 79–86. [Google Scholar] [CrossRef]
- Li, X.; Li, K.; Xie, H.; Xie, Y.; Li, Y.; Zhao, X.; Jiang, X.; Chen, D. Antioxidant and Cytoprotective Effects of the Di-O-Caffeoylquinic Acid Family: The Mechanism, Structure-Activity Relationship, and Conformational Effect. Molecules 2018, 23, 222. [Google Scholar] [CrossRef] [Green Version]
- Sikora, E.; Scapagnini, G.; Barbagallo, M. Curcumin, inflammation, ageing and age-related diseases. Immun. Ageing 2010, 7, 1. [Google Scholar] [CrossRef] [Green Version]

| Total Extracts | Organs | ||||||
|---|---|---|---|---|---|---|---|
| Leaf | Flower | Fruit | Twig | Root | |||
| 80% EtOH (mg/g dried sample) | 27.2 | 16.0 | 40.0 | 24.2 | 2.4 | ||
| 100% MeOH (mg/g dried sample) | 14.8 | 14.0 | 25.6 | 24.0 | 1.6 | ||
| Radical activity | Total extract | Con. (μg/mL) | |||||
| DPPH (%) | 80% EtOH | 10 | 90.28 ± 9.55 * | 101.43 ± 2.60 ** | 94.37 ± 10.73 | 94.00 ± 15.47 | 98.09 ± 3.09 * |
| 100 | 64.16 ± 2.91 | 53.46 ± 3.57 | 78.43 ± 6.59 | 68.25 ± 9.42 | 68.25 ± 6.12 | ||
| 100% MeOH | 10 | 95.12 ± 0.85 * | 101.97 ± 0.93 | 95.03 ± 5.72 * | 101.49 ± 2.04 ** | 100.60 ± 1.49 | |
| 100 | 70.20 ± 2.46 | 77.09 ± 6.39 | 83.73 ± 0.49 | 78.15 ± 3.96 | 76.57 ± 1.48 | ||
| Ascorbic acid | 10 | 49.15 ± 0.21 ** | 49.15 ± 0.21 ** | 49.15 ± 0.21 ** | 49.15 ± 0.21 ** | 49.15 ± 0.21 ** | |
| 100 | 3.91 ± 0.00 ** | 3.91 ± 0.00 ** | 3.91 ± 0.00 ** | 3.91 ± 0.00 ** | 3.91 ± 0.00 ** | ||
| ABTS•+ (%) | 80% EtOH | 10 | 95.57 ± 0.34 * | 98.76 ± 1.34 * | 95.76 ± 1.98 * | 94.39 ± 0.70 * | 93.84 ± 0.62 ** |
| 100 | 63.09 ± 1.17 * | 77.18 ± 0.23 * | 65.97 ± 0.47 * | 58.53 ± 1.03 * | 47.13 ± 0.42 ** | ||
| 100% MeOH | 10 | 96.76 ± 0.96 * | 98.39 ± 0.72 | 96.03 ± 0.48 * | 95.85 ± 0.28 ** | 93.25 ± 2.33 ** | |
| 100 | 65.74 ± 0.28 * | 83.25 ± 1.06 | 67.20 ± 0.36 * | 61.18 ± 0.63 ** | 43.52 ± 0.72 ** | ||
| Ascorbic acid | 10 | 33.92 ± 0.49 ** | 33.92 ± 0.49 ** | 33.92 ± 0.49 ** | 33.92 ± 0.49 ** | 33.92 ± 0.49 ** | |
| 100 | 2.85 ± 0.62 ** | 2.85 ± 0.62 ** | 2.85 ± 0.62 ** | 2.85 ± 0.62 ** | 2.85 ± 0.62 ** | ||
| Organs | Fractions | ABTS•+ EC50 Values (µg/mL) | DPPH• EC50 Values (µg/mL) |
|---|---|---|---|
| Leaf | MC | 185.48 ± 1.74 | >250 |
| E | 70.77 ± 0.40 | 53.05 ± 3.37 | |
| B | 224.12 ± 2.44 | 124.88 ± 7.30 | |
| Flower | E | 32.85 ± 1.23 | 19.10 ± 2.94 |
| B | 104.99 ± 1.64 | 70.00 ± 1.79 | |
| Fruit | MC | 119.43 ± 1.13 | >250 |
| E | 70.98 ± 1.16 | 35.61 ± 7.64 | |
| B | 207.56 ± 4.76 | 189.17 ± 5.06 | |
| Twig | E | 123.33 ± 0.95 | 87.12 ± 6.90 |
| B | 160.75 ± 0.92 | 146.21 ± 16.68 | |
| Root | MC | 35.11 ± 0.19 | 72.86 ± 2.41 |
| E | 55.30 ± 0.75 | 68.20 ± 1.30 | |
| B | 124.90 ± 2.60 | 202.30 ± 15.26 | |
| Ascorbic acid * | 6.35 ± 3.30 | 8.38 ± 0.40 |
| Locations | Samples | Marker Compounds | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | ||
| Goheung | Leaf | 0.4546 ± 0.0036 | 0.2871 ± 0.0008 | 0.3552 ± 0.0010 | 4.2427 ± 0.0012 | 0.3944 ± 0.0030 | 1.0858 ± 0.0001 | 0.0943 ± 0.0013 |
| Flower | 0.2446 ± 0.0022 | 0.4216 ± 0.0031 | 0.4963 ± 0.0020 | 0.8751 ± 0.0023 | 1.1459 ± 0.0043 | 0.0062 ± 0.0001 | 0.6787 ± 0.0040 | |
| Fruit | 0.0040 ± 0.0001 | 0.0056 ± 0.0000 | 0.0184 ± 0.0003 | 0.0425 ± 0.0009 | 1.4030 ± 0.0010 | 0.0039 ± 0.0001 | 0.0072 ± 0.0001 | |
| Twig | 0.0012 ± 0.0001 | 0.0271 ± 0.0003 | 0.0077 ± 0.0002 | 0.0270 ± 0.0001 | 0.2129 ± 0.0027 | 0.0037 ± 0.0001 | 0.0079 ± 0.0003 | |
| Root | N.D. | 0.0287 ± 0.0001 | 0.0027 ±0.0001 | N.D. | 0.2112 ± 0.0009 | 0.0095 ± 0.0001 | 0.0237 ± 0.0003 | |
| Jeju | Leaf | 0.1983 ± 0.0007 | 2.2644 ± 0.0001 | 0.1433 ± 0.0011 | 1.2529 ± 0.0075 | 0.5012 ± 0.0045 | 0.1109 ± 0.0001 | 0.7863 ± 0.0003 |
| Flower | 0.1141 ± 0.0002 | 0.5471 ± 0.0010 | 0.2475 ± 0.0010 | 0.5781 ± 0.0034 | 0.7146 ± 0.0017 | 0.0043 ± 0.0001 | 1.1429 ± 0.0024 | |
| Fruit | 0.0039 ± 0.0001 | 0.1330 ± 0.0013 | 0.0168 ± 0.0002 | 0.2427 ± 0.0006 | 0.0984 ± 0.0007 | 0.0018 ± 0.0001 | 0.0809 ± 0.0003 | |
| Twig | 0.0514 ± 0.0001 | 0.1226 ± 0.0010 | 0.0396 ± 0.0002 | 0.0742 ± 0.0007 | 0.2011 ± 0.0002 | 0.0021 ± 0.0001 | 0.4464 ± 0.0008 | |
| Sinan | Leaf | 0.0239 ± 0.0003 | 1.4998 ± 0.0067 | 0.0042 ± 0.0001 | 1.0546 ± 0.0024 | 1.8050 ± 0.0033 | 0.4516 ± 0.0014 | 0.4922 ± 0.0034 |
| Fruit | 0.0080 ± 0.0004 | 0.9908 ± 0.0014 | 0.0182 ± 0.0002 | 0.6788 ± 0.0015 | 0.7085 ± 0.0014 | 0.0134 ± 0.0003 | 0.1416 ± 0.0002 | |
| Twig | N.D. | 0.1949 ± 0.0005 | N.D. | N.D. | 0.4229 ± 0.0008 | 0.0032 ± 0.0001 | 0.2466 ± 0.0006 | |
| Root | N.D. | 0.3941 ± 0.0003 | 0.0023 ± 0.0001 | N.D. | 0.5594 ± 0.0003 | 0.0006 ± 0.0001 | 0.2451 ± 0.0003 | |
| Busan | Leaf | N.D. | 0.5883 ± 0.0005 | 0.0022 ± 0.0001 | 1.2095 ± 0.0038 | 0.7462 ± 0.0008 | 0.0519 ± 0.0003 | 0.3789 ± 0.0004 |
| Fruit | 0.0146 ± 0.0004 | 0.2424 ± 0.0002 | 0.0282 ± 0.0007 | 0.6423 ± 0.0003 | 0.0067 ± 0.0001 | N.D. | 0.1255 ± 0.0006 | |
| Twig | N.D. | 0.8450 ± 0.0057 | N.D. | 0.1631 ± 0.0005 | N.D. | N.D. | 0.7905 ± 0.0042 | |
| Root | N.D. | 0.3527 ± 0.0011 | N.D. | N.D. | 0.4607 ± 0.0026 | N.D. | N.D. | |
| Compounds | a Reduction of the Peak Area (%) | EC50 Values (µM) |
|---|---|---|
| Protocatechuic acid (1) | 69.54 ± 2.33 | 52.15 ± 0.49 |
| Chlorogenic acid (2) | 77.39 ± 1.14 | 68.25 ± 0.78 |
| 4-Hydroxybenzoic acid (3) | 3.22 ± 0.52 | - |
| Orientin (4) | 84.85 ± 1.07 | 56.45 ± 0.07 |
| Agnuside (5) | 21.82 ± 1.33 | - |
| 6′-p-Hydroxybenzoylmussaenosidic acid (6) | 4.90 ± 0.21 | - |
| 3,5-Di-CQA (7) | 89.06 ± 1.17 | 25.05 ± 0.49 |
| Ascobic acid * | - | 46.04 ± 0.67 |
| Organs | IC50 Values (µg/mL) | Fractions | IC50 Values (µg/mL) |
|---|---|---|---|
| Leaf | 169.14 | H | 2.21 ± 0.97 |
| MC | 6.32 ± 0.83 | ||
| E | 188.64 ± 5.91 | ||
| B | >300 | ||
| DW | >300 | ||
| Flower | 75.06 | E | 90.10 ± 3.95 |
| B | 84.42 ± 4.29 | ||
| Fruit | 100.75 | E | 36.24 ± 6.48 |
| B | 229.95 ± 34.76 | ||
| Twig | 101.97 | E | 92.31 ± 3.49 |
| B | 42.89 ± 1.48 | ||
| Root | 138.50 | E | 38.85 ± 5.56 |
| B | 285.98 ± 17.38 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le, D.; Han, S.; Ahn, J.; Yu, J.; Kim, C.-K.; Lee, M. Analysis of Antioxidant Phytochemicals and Anti-Inflammatory Effect from Vitex rotundifolia L.f. Antioxidants 2022, 11, 454. https://doi.org/10.3390/antiox11030454
Le D, Han S, Ahn J, Yu J, Kim C-K, Lee M. Analysis of Antioxidant Phytochemicals and Anti-Inflammatory Effect from Vitex rotundifolia L.f. Antioxidants. 2022; 11(3):454. https://doi.org/10.3390/antiox11030454
Chicago/Turabian StyleLe, DucDat, Sanghee Han, Jeongjun Ahn, Jayeon Yu, Chang-Kwon Kim, and Mina Lee. 2022. "Analysis of Antioxidant Phytochemicals and Anti-Inflammatory Effect from Vitex rotundifolia L.f" Antioxidants 11, no. 3: 454. https://doi.org/10.3390/antiox11030454
APA StyleLe, D., Han, S., Ahn, J., Yu, J., Kim, C.-K., & Lee, M. (2022). Analysis of Antioxidant Phytochemicals and Anti-Inflammatory Effect from Vitex rotundifolia L.f. Antioxidants, 11(3), 454. https://doi.org/10.3390/antiox11030454





