Radiation Type- and Dose-Specific Transcriptional Responses across Healthy and Diseased Mammalian Tissues
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Hybrid Collection and Transcriptomic Analyses
2.2. Functional Enrichment Analysis
2.3. Database Construction
2.4. Machine Learning Approach
2.5. Functional Network
3. Results and Discussion
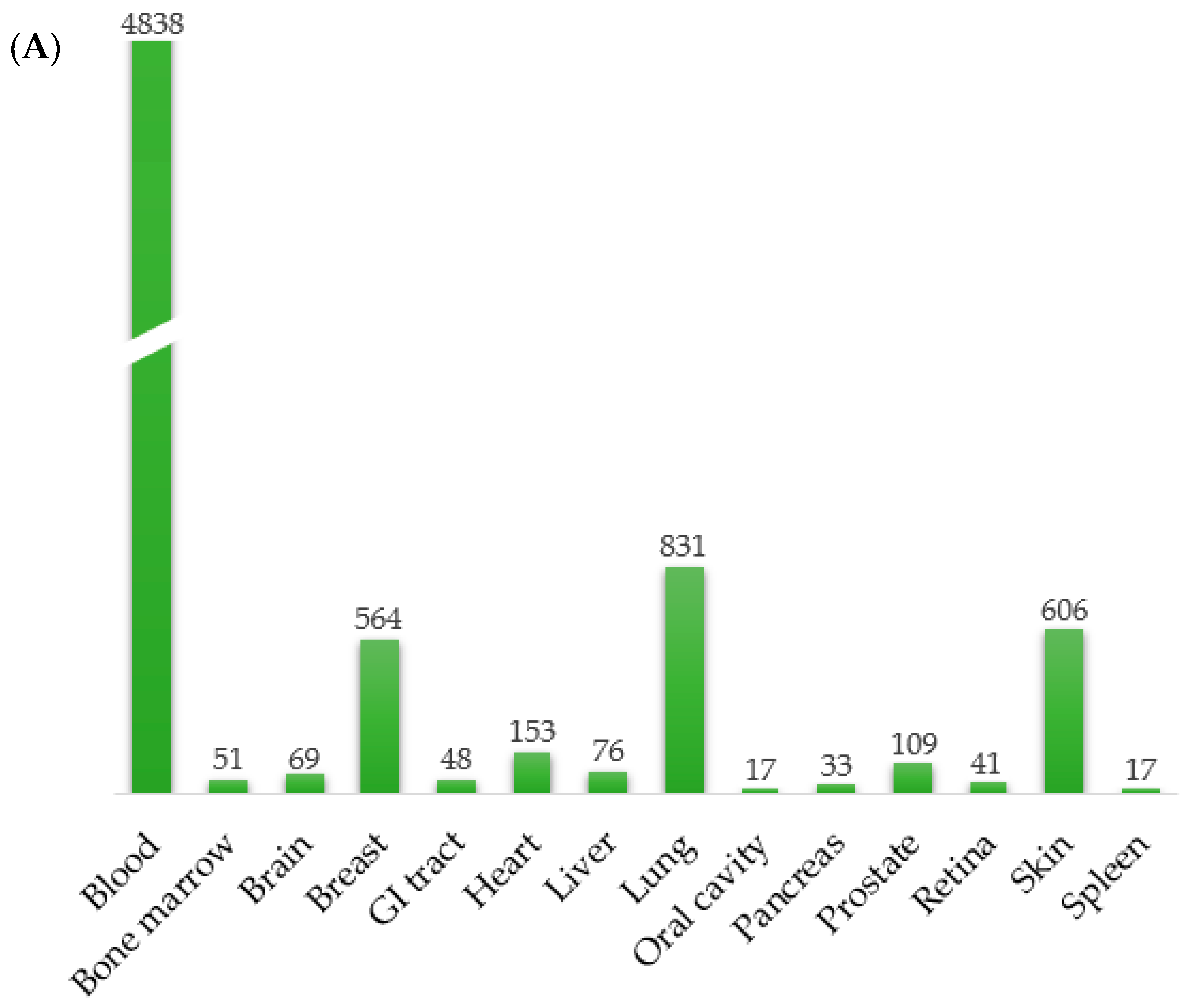
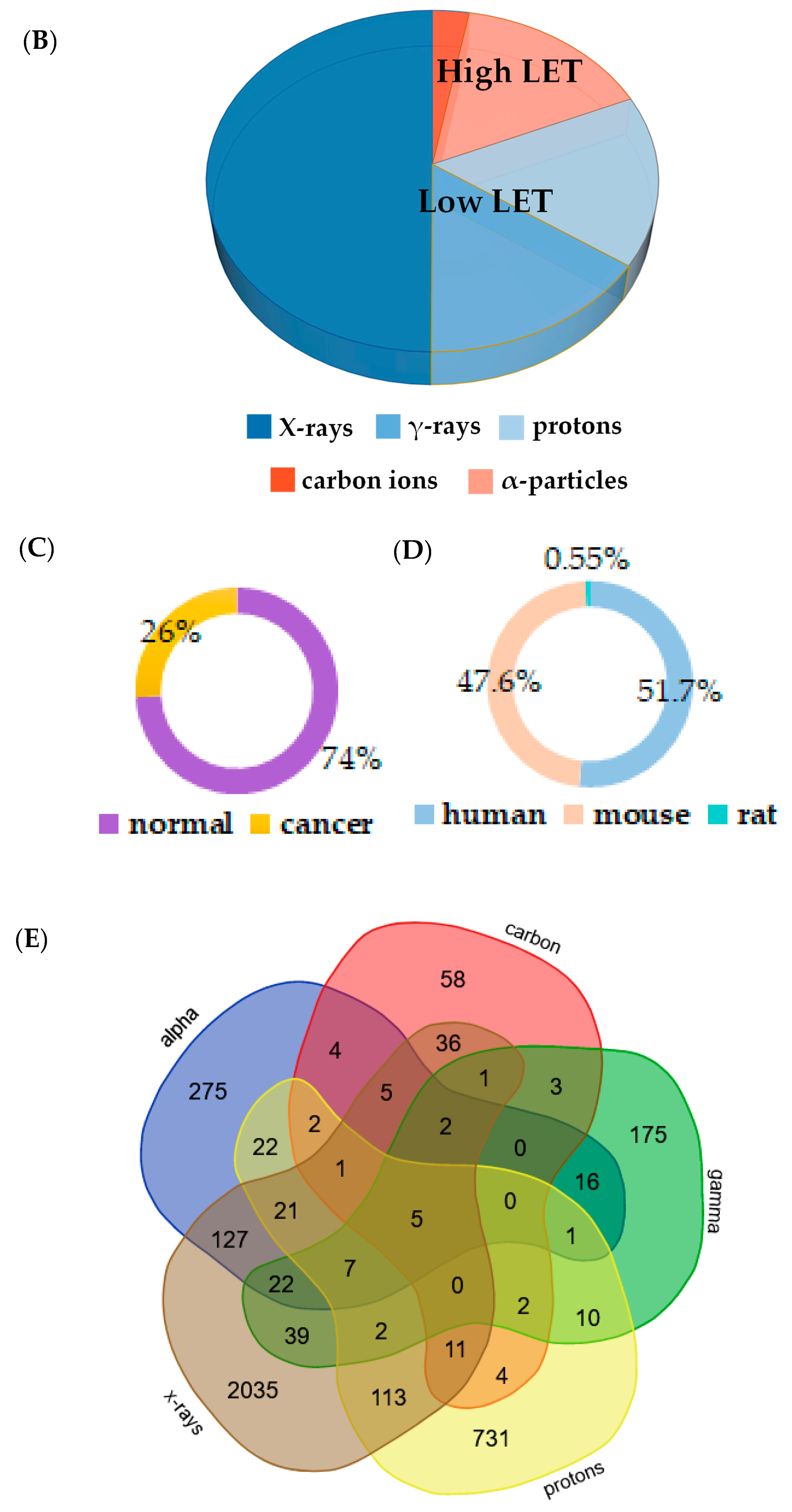
3.1. Development of RadBioBase
- -
- Differential expression of genes: The expression status of the corresponding genes (i.e., up or downregulated in irradiated compared to non-irradiated tissue/cell control groups). In this version of the database, the canonical, full-length transcripts for each gene were used.
- -
- Biological characteristics: Cell type (cancer or normal), organism and tissue/cell line.
- -
- Type of irradiation: X-rays, γ-rays, protons, carbon ions or α-particles.
- -
- Post-irradiation time when provided in the original study.
- -
- Physical characteristics: LET (keV/μm), beam energy (MeV or kV for X-rays), dose (Gy) and average dose rate (Gy/min or Gy/h). In the cases where the LET of particles was not included in the original paper, it was calculated with the Stopping and Range of Ions in Matter (SRIM/TRIM) software, using as entrance parameters the type of ion, the target density and the energy of the irradiation beam when provided. For tissue targets not included in the compound dictionary of SRIM, the elemental compositions and mass densities were obtained from the bibliography [66,67,68]. Notably, the SRIM-calculated LET values were calculated only when provided in the relative studies, for the entrance point (highest energy values) of the beam instead of the Bragg peak, and thus were much smaller than the expected LET values for the Bragg peak region. According to the different energies in the various studies, LET values for protons were calculated as such: energies 100 MeV----> 0.76 keV/μm, 250 MeV----> 0.34 keV/μm, 190.6 MeV----> 0.5 keV/μm, 230 MeV----> 0.38 keV/μm, 4.5 MeV----> 9.54 keV/μm (Table S1). Moreover, those α-particle energies not provided in the original paper were calculated empirically with the help of LET-energy curves [69].
- -
- Comparison with low-LET irradiation: X-rays, γ-rays or electrons, depending on the information given in the original paper.
- -
- DNA damage (in clusters per Gy per Gbp): DSBs and total clusters of DNA damage were calculated using the Monte Carlo Damage Simulation (MCDS) software [70,71] for each radiation type (Table S1). For each MCDS input file, the parameters were set as CELL: DNA = 1 ndia = 5 cdia = 10, SIMCON: nocs = 10,000 seed = 987,654,321, and the oxygen concentration was set to 20%, while X-ray and γ-ray radiation was simulated by a 10 keV electron beam. The inclusion of the “complex damages” is based on the well-documented importance of clustered DNA damages in defying biological responses and can provide the first hints for possible connections of the quality and quantity of DNA damage with specific gene expression [72]. PubMed ID of the corresponding article.
- -
- Type of validation: depending on the method used in the original studies for data validation, we defined values as (a) microarrays, (b) RNA-Seq, (c) qPCR, (d) microarrays and qPCR, and (e) RNA-Seq and qPCR.
3.2. Commonalities among Radiation Types across a Number of Mammalian Tissues
3.3. Radiation Type-Specific Disease Pathways Inferred from Transcriptomes of Irradiated Cells
3.4. Machine Learning-Generated Gene Signatures of Cell Sensitivity to High- Versus Low-LET Radiation Types
3.5. Low-Dose Irradiation Is Associated with Cytokine Cascades, While High with ROS Metabolism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schaue, D.; McBride, W.H. Opportunities and challenges of radiotherapy for treating cancer. Nat. Rev. Clin. Oncol. 2015, 12, 527–540. [Google Scholar] [CrossRef] [PubMed]
- Baskar, R.; Lee, K.A.; Yeo, R.; Yeoh, K.W. Cancer and radiation therapy: Current advances and future directions. Int. J. Med. Sci. 2012, 9, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Delaney, G.; Jacob, S.; Featherstone, C.; Barton, M. The role of radiotherapy in cancer treatment: Estimating optimal utilization from a review of evidence-based clinical guidelines. Cancer 2005, 104, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Hong, A.M.; Stretch, J.R.; Thompson, J.F. Treatment of primary merkel cell carcinoma: Radiotherapy can be an effective, less morbid alternative to surgery. Eur. J. Surg. Oncol. 2021, 47, 483–485. [Google Scholar] [CrossRef] [PubMed]
- Kroemer, G.; Galassi, C.; Zitvogel, L.; Galluzzi, L. Immunogenic cell stress and death. Nat. Immunol. 2022, 23, 487–500. [Google Scholar] [CrossRef] [PubMed]
- Kroemer, G.; Galluzzi, L.; Kepp, O.; Zitvogel, L. Immunogenic cell death in cancer therapy. Annu. Rev. Immunol. 2013, 31, 51–72. [Google Scholar] [CrossRef]
- Golden, E.B.; Apetoh, L. Radiotherapy and immunogenic cell death. Semin. Radiat. Oncol. 2015, 25, 11–17. [Google Scholar] [CrossRef]
- Muroyama, Y.; Nirschl, T.R.; Kochel, C.M.; Lopez-Bujanda, Z.; Theodros, D.; Mao, W.; Carrera-Haro, M.A.; Ghasemzadeh, A.; Marciscano, A.E.; Velarde, E.; et al. Stereotactic radiotherapy increases functionally suppressive regulatory t cells in the tumor microenvironment. Cancer Immunol. Res. 2017, 5, 992–1004. [Google Scholar] [CrossRef]
- Nakad, R.; Schumacher, B. DNA damage response and immune defense: Links and mechanisms. Front. Genet. 2016, 7, 147. [Google Scholar] [CrossRef]
- Pateras, I.S.; Havaki, S.; Nikitopoulou, X.; Vougas, K.; Townsend, P.A.; Panayiotidis, M.I.; Georgakilas, A.G.; Gorgoulis, V.G. The DNA damage response and immune signaling alliance: Is it good or bad? Nature decides when and where. Pharmacol. Ther. 2015, 154, 36–56. [Google Scholar] [CrossRef]
- Mohamad, O.; Sishc, B.J.; Saha, J.; Pompos, A.; Rahimi, A.; Story, M.D.; Davis, A.J.; Kim, D.W.N. Carbon ion radiotherapy: A review of clinical experiences and preclinical research, with an emphasis on DNA damage/repair. Cancers 2017, 9, 66. [Google Scholar] [CrossRef] [PubMed]
- Nikitaki, Z.; Velalopoulou, A.; Zanni, V.; Tremi, I.; Havaki, S.; Kokkoris, M.; Gorgoulis, V.G.; Koumenis, C.; Georgakilas, A.G. Key biological mechanisms involved in high-let radiation therapies with a focus on DNA damage and repair. Expert. Rev. Mol. Med. 2022, 24, e15. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.H.; Park, S.; Peyton, M.; Girard, L.; Xie, Y.; Minna, J.D.; Story, M.D. Distinct transcriptome profiles identified in normal human bronchial epithelial cells after exposure to gamma-rays and different elemental particles of high z and energy. BMC Genom. 2013, 14, 372. [Google Scholar] [CrossRef] [PubMed]
- Macaeva, E.; Tabury, K.; Michaux, A.; Janssen, A.; Averbeck, N.; Moreels, M.; De Vos, W.H.; Baatout, S.; Quintens, R. High-let carbon and iron ions elicit a prolonged and amplified p53 signaling and inflammatory response compared to low-let X-rays in human peripheral blood mononuclear cells. Front. Oncol. 2021, 11, 768493. [Google Scholar] [CrossRef]
- Michalettou, T.D.; Michalopoulos, I.; Costes, S.V.; Hellweg, C.E.; Hada, M.; Georgakilas, A.G. A meta-analysis of the effects of high-let ionizing radiations in human gene expression. Life 2021, 11, 115. [Google Scholar] [CrossRef]
- Nielsen, S.; Bassler, N.; Grzanka, L.; Laursen, L.; Swakon, J.; Olko, P.; Andreassen, C.N.; Alsner, J.; Singers Sorensen, B. Comparison of coding transcriptomes in fibroblasts irradiated with low and high let proton beams and cobalt-60 photons. Int. J. Radiat. Oncol. Biol. Phys. 2019, 103, 1203–1211. [Google Scholar] [CrossRef]
- Niemantsverdriet, M.; van Goethem, M.J.; Bron, R.; Hogewerf, W.; Brandenburg, S.; Langendijk, J.A.; van Luijk, P.; Coppes, R.P. High and low let radiation differentially induce normal tissue damage signals. Int. J. Radiat. Oncol. Biol. Phys. 2012, 83, 1291–1297. [Google Scholar] [CrossRef][Green Version]
- Sertorio, M.; Nowrouzi, A.; Akbarpour, M.; Chetal, K.; Salomonis, N.; Brons, S.; Mascia, A.; Ionascu, D.; McCauley, S.; Kupneski, T.; et al. Differential transcriptome response to proton versus X-ray radiation reveals novel candidate targets for combinatorial pt therapy in lymphoma. Radiother. Oncol. 2021, 155, 293–303. [Google Scholar] [CrossRef]
- Barrett, T.; Wilhite, S.E.; Ledoux, P.; Evangelista, C.; Kim, I.F.; Tomashevsky, M.; Marshall, K.A.; Phillippy, K.H.; Sherman, P.M.; Holko, M.; et al. Ncbi geo: Archive for functional genomics data sets--update. Nucleic Acids Res. 2013, 41, D991–D995. [Google Scholar] [CrossRef]
- Becker, B.V.; Majewski, M.; Abend, M.; Palnek, A.; Nestler, K.; Port, M.; Ullmann, R. Gene expression changes in human ipsc-derived cardiomyocytes after X-ray irradiation. Int. J. Radiat. Biol. 2018, 94, 1095–1103. [Google Scholar] [CrossRef]
- Mukherjee, S.; Grilj, V.; Broustas, C.G.; Ghandhi, S.A.; Harken, A.D.; Garty, G.; Amundson, S.A. Human transcriptomic response to mixed neutron-photon exposures relevant to an improvised nuclear device. Radiat. Res. 2019, 192, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Fujinaga, H.; Sakai, Y.; Yamashita, T.; Arai, K.; Terashima, T.; Komura, T.; Seki, A.; Kawaguchi, K.; Nasti, A.; Yoshida, K.; et al. Biological characteristics of gene expression features in pancreatic cancer cells induced by proton and X-ray irradiation. Int. J. Radiat. Biol. 2019, 95, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Broustas, C.G.; Xu, Y.; Harken, A.D.; Chowdhury, M.; Garty, G.; Amundson, S.A. Impact of neutron exposure on global gene expression in a human peripheral blood model. Radiat. Res. 2017, 187, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Broustas, C.G.; Xu, Y.; Harken, A.D.; Garty, G.; Amundson, S.A. Comparison of gene expression response to neutron and X-ray irradiation using mouse blood. BMC Genom. 2017, 18, 2. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Wang, F.; Kong, P.; Yu, P.K.N.; Wang, H.; Han, W. Gene profiling characteristics of radioadaptive response in ag01522 normal human fibroblasts. PLoS ONE 2015, 10, e0123316. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Smilenov, L.B.; Elliston, C.D.; Amundson, S.A. Radiation dose-rate effects on gene expression in a mouse biodosimetry model. Radiat. Res. 2015, 184, 24–32. [Google Scholar] [CrossRef]
- Ghandhi, S.A.; Yaghoubian, B.; Amundson, S.A. Global gene expression analyses of bystander and alpha particle irradiated normal human lung fibroblasts: Synchronous and differential responses. BMC Med. Genom. 2008, 1, 63. [Google Scholar] [CrossRef]
- Ghandhi, S.A.; Sinha, A.; Markatou, M.; Amundson, S.A. Time-series clustering of gene expression in irradiated and bystander fibroblasts: An application of fbpa clustering. BMC Genom. 2011, 12, 2. [Google Scholar] [CrossRef]
- Ghandhi, S.A.; Ming, L.; Ivanov, V.N.; Hei, T.K.; Amundson, S.A. Regulation of early signaling and gene expression in the alpha-particle and bystander response of imr-90 human fibroblasts. BMC Med. Genom. 2010, 3, 31. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Iwakawa, M.; Furusawa, Y.; Ishikawa, K.; Aoki, M.; Imadome, K.; Matsumoto, I.; Tsujii, H.; Ando, K.; Imai, T. Gene expression analysis in human malignant melanoma cell lines exposed to carbon beams. Int. J. Radiat. Biol. 2008, 84, 299–314. [Google Scholar] [CrossRef]
- Mezentsev, A.; Amundson, S.A. Global gene expression responses to low- or high-dose radiation in a human three-dimensional tissue model. Radiat. Res. 2011, 175, 677–688. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Wang, J.; Jaehnig, E.J.; Shi, Z.; Zhang, B. Webgestalt 2019: Gene set analysis toolkit with revamped uis and apis. Nucleic Acids Res. 2019, 47, W199–W205. [Google Scholar] [CrossRef] [PubMed]
- Alanni, R.; Hou, J.; Azzawi, H.; Xiang, Y. Deep gene selection method to select genes from microarray datasets for cancer classification. BMC Bioinform. 2019, 20, 608. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Meng, Z.; Su, R. Werfe: A gene selection algorithm based on recursive feature elimination and ensemble strategy. Front. Bioeng. Biotechnol. 2020, 8, 496. [Google Scholar] [CrossRef]
- Zhang, J.G.; Deng, H.W. Gene selection for classification of microarray data based on the bayes error. BMC Bioinform. 2007, 8, 370. [Google Scholar] [CrossRef]
- Su, R.; Liu, X.; Wei, L. Mine-rfe: Determine the optimal subset from rfe by minimizing the subset-accuracy-defined energy. Brief. Bioinform. 2020, 21, 687–698. [Google Scholar] [CrossRef]
- Kolde, R.; Laur, S.; Adler, P.; Vilo, J. Robust rank aggregation for gene list integration and meta-analysis. Bioinformatics 2012, 28, 573–580. [Google Scholar] [CrossRef]
- Szklarczyk, D.; Gable, A.L.; Nastou, K.C.; Lyon, D.; Kirsch, R.; Pyysalo, S.; Doncheva, N.T.; Legeay, M.; Fang, T.; Bork, P.; et al. The string database in 2021: Customizable protein-protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res. 2021, 49, D605–D612. [Google Scholar] [CrossRef]
- Baluchamy, S.; Zhang, Y.; Ravichandran, P.; Ramesh, V.; Sodipe, A.; Hall, J.C.; Jejelowo, O.; Gridley, D.S.; Wu, H.; Ramesh, G.T. Expression profile of DNA damage signaling genes in 2 gy proton exposed mouse brain. Mol. Cell. Biochem. 2010, 341, 207–215. [Google Scholar] [CrossRef]
- Baluchamy, S.; Zhang, Y.; Ravichandran, P.; Ramesh, V.; Sodipe, A.; Hall, J.C.; Jejelowo, O.; Gridley, D.S.; Wu, H.; Ramesh, G.T. Differential oxidative stress gene expression profile in mouse brain after proton exposure. Vitr. Cell. Dev. Biology. Anim. 2010, 46, 718–725. [Google Scholar] [CrossRef]
- Bravatà, V.; Cammarata, F.P.; Minafra, L.; Pisciotta, P.; Scazzone, C.; Manti, L.; Savoca, G.; Petringa, G.; Cirrone, G.A.P.; Cuttone, G.; et al. Proton-irradiated breast cells: Molecular points of view. J. Radiat. Res. 2019, 60, 451–465. [Google Scholar] [CrossRef] [PubMed]
- Bravatà, V.; Minafra, L.; Cammarata, F.P.; Pisciotta, P.; Lamia, D.B.; Marchese, V.; Petringa, G.; Manti, L.; Cirrone, G.A.P.; Gilardi, M.C.; et al. Gene expression profiling of breast cancer cell lines treated with proton and electron radiations. Br. J. Radiol. 2018, 91, 20170934. [Google Scholar] [CrossRef] [PubMed]
- Cammarata, F.P.; Forte, G.I.; Broggi, G.; Bravata, V.; Minafra, L.; Pisciotta, P.; Calvaruso, M.; Tringali, R.; Tomasello, B.; Torrisi, F.; et al. Molecular investigation on a triple negative breast cancer xenograft model exposed to proton beams. Int. J. Mol. Sci. 2020, 21, 6337. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, V.; Howland, M. Genomic profiling of a human leukemic monocytic cell-line (thp-1) exposed to alpha particle radiation. Sci. World J. 2012, 2012, 205038. [Google Scholar] [CrossRef]
- Chauhan, V.; Howland, M.; Greene, H.B.; Wilkins, R.C. Transcriptional and secretomic profiling of epidermal cells exposed to alpha particle radiation. Open Biochem. J. 2012, 6, 103–115. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chauhan, V.; Howland, M. Gene expression responses in human lung fibroblasts exposed to alpha particle radiation. Toxicol. Vitr. 2014, 28, 1222–1229. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, V.; Howland, M.; Mendenhall, A.; O’Hara, S.; Stocki, T.J.; McNamee, J.P.; Wilkins, R.C. Effects of alpha particle radiation on gene expression in human pulmonary epithelial cells. Int. J. Hyg. Environ. Health 2012, 215, 522–535. [Google Scholar] [CrossRef]
- Chauhan, V.; Howland, M.; Wilkins, R. Identification of gene-based responses in human blood cells exposed to alpha particle radiation. BMC Med. Genom. 2014, 7, 43. [Google Scholar] [CrossRef]
- Fushimi, K.; Uzawa, K.; Ishigami, T.; Yamamoto, N.; Kawata, T.; Shibahara, T.; Ito, H.; Mizoe, J.e.; Tsujii, H.; Tanzawa, H. Susceptible genes and molecular pathways related to heavy ion irradiation in oral squamous cell carcinoma cells. Radiother. Oncol. 2008, 89, 237–244. [Google Scholar] [CrossRef]
- Gridley, D.S.; Freeman, T.L.; Makinde, A.Y.; Wroe, A.J.; Luo-Owen, X.; Tian, J.; Mao, X.W.; Rightnar, S.; Kennedy, A.R.; Slater, J.M.; et al. Comparison of proton and electron radiation effects on biological responses in liver, spleen and blood. Int. J. Radiat. Biol. 2011, 87, 1173–1181. [Google Scholar] [CrossRef]
- Gridley, D.S.; Pecaut, M.J.; Rizvi, A.; Coutrakon, G.B.; Luo-Owen, X.; Makinde, A.Y.; Slater, J.M. Low-dose, low-dose-rate proton radiation modulates cd4(+) t cell gene expression. Int. J. Radiat. Biol. 2009, 85, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Konings, K.; Belmans, N.; Vermeesen, R.; Baselet, B.; Lamers, G.; Janssen, A.; Isebaert, S.; Baatout, S.; Haustermans, K.; Moreels, M. Targeting the Hedgehog pathway in combination with X-ray or carbon ion radiation decreases migration of MCF-7 breast cancer cells. Int. J. Oncol. 2019, 55, 1339–1348. [Google Scholar] [CrossRef] [PubMed]
- Konings, K.; Vandevoorde, C.; Belmans, N.; Vermeesen, R.; Baselet, B.; Walleghem, M.V.; Janssen, A.; Isebaert, S.; Baatout, S.; Haustermans, K.; et al. The combination of particle irradiation with the hedgehog inhibitor gant61 differently modulates the radiosensitivity and migration of cancer cells compared to X-ray irradiation. Front. Oncol. 2019, 9, 391. [Google Scholar] [CrossRef]
- Leduc, A.; Chaouni, S.; Pouzoulet, F.; De Marzi, L.; Megnin-Chanet, F.; Corre, E.; Stefan, D.; Habrand, J.L.; Sichel, F.; Laurent, C. Differential normal skin transcriptomic response in total body irradiated mice exposed to scattered versus scanned proton beams. Sci. Rep. 2021, 11, 5876. [Google Scholar] [CrossRef]
- Lindgren, T.; Stigbrand, T.; Riklund, K.; Johansson, L.; Eriksson, D. Gene expression profiling in molt-4 cells during gamma-radiation-induced apoptosis. Tumour Biol. 2012, 33, 689–700. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Wang, Z.; Li, W.; Wei, Y. Transcriptional response of murine bone marrow cells to total-body carbon-ion irradiation. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2019, 839, 49–58. [Google Scholar] [CrossRef]
- Liu, Q.J.; Zhang, D.Q.; Zhang, Q.Z.; Feng, J.B.; Lu, X.; Wang, X.R.; Li, K.P.; Chen, D.Q.; Mu, X.F.; Li, S.; et al. Dose-effect of ionizing radiation-induced pig3 gene expression alteration in human lymphoblastoid ahh-1 cells and human peripheral blood lymphocytes. Int. J. Radiat. Biol. 2015, 91, 71–80. [Google Scholar] [CrossRef]
- Mao, X.W.; Green, L.M.; Mekonnen, T.; Lindsey, N.; Gridley, D.S. Gene expression analysis of oxidative stress and apoptosis in proton-irradiated rat retina. Vivo. 2021, 24, 425–430. [Google Scholar]
- Purgason, A.; Zhang, Y.; Hamilton, S.R.; Gridley, D.S.; Sodipe, A.; Jejelowo, O.; Ramesh, G.T.; Moreno-Villanueva, M.; Wu, H. Apoptosis and expression of apoptosis-related genes in mouse intestinal tissue after whole-body proton exposure. Mol. Cell. Biochem. 2018, 442, 155–168. [Google Scholar] [CrossRef]
- Rizvi, A.; Pecaut, M.J.; Gridley, D.S. Low-dose gamma-rays and simulated solar particle event protons modify splenocyte gene and cytokine expression patterns. J. Radiat. Res. 2011, 52, 701–711. [Google Scholar] [CrossRef][Green Version]
- Song, L.H.; Yan, H.L.; Cai, D.L. Gene expression profiles in the liver of mice irradiated with (60)co gamma rays and treated with soybean isoflavone. Eur. J. Nutr. 2006, 45, 406–417. [Google Scholar] [CrossRef] [PubMed]
- Suetens, A.; Moreels, M.; Quintens, R.; Chiriotti, S.; Tabury, K.; Michaux, A.; Grégoire, V.; Baatout, S. Carbon ion irradiation of the human prostate cancer cell line pc3: A whole genome microarray study. Int. J. Oncol. 2014, 44, 1056–1072. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Tian, S.; Gridley, D.S. Comparison of acute proton, photon, and low-dose priming effects on genes associated with extracellular matrix and adhesion molecules in the lungs. Fibrogenesis Tissue Repair 2013, 6, 4. [Google Scholar] [CrossRef]
- Uehara, Y.; Ito, Y.; Taki, K.; Nenoi, M.; Ichinohe, K.; Nakamura, S.; Tanaka, S.; Oghiso, Y.; Tanaka, K.; Matsumoto, T.; et al. Gene expression profiles in mouse liver after long-term low-dose-rate irradiation with gamma rays. Radiat. Res. 2010, 174, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.; Bassler, N.; Grzanka, L.; Swakon, J.; Olko, P.; Andreassen, C.N.; Overgaard, J.; Alsner, J.; Sorensen, B.S. Differential gene expression in primary fibroblasts induced by proton and cobalt-60 beam irradiation. Acta Oncol. 2017, 56, 1406–1412. [Google Scholar] [CrossRef] [PubMed]
- Nsarsam, D.; Mohammed Saied, D.; Nasir Aklo, K. Theoretical study of energy loss of proton in human tissues. Al-Qadisiyah J. Pure Sci. 2013, 18, 23–31. [Google Scholar]
- Raheem, Z.J.; Saied, B.M. Energy loss of carbon ion in lung tissue. AIP Conf. Proc. 2019, 2190, 020021. [Google Scholar]
- Usta, M.; Tufan, M.Ç. Stopping power and range calculations in human tissues by using the hartree-fock-roothaan wave functions. RaPC 2017, 140, 43–50. [Google Scholar] [CrossRef]
- Nikitaki, Z.; Choulilitsa, E.; Kalospyros, S.A.; Kaisaridi, S.; Terzoudi, G.I.; Kokkoris, M.; Georgakilas, A.G. Construction and evaluation of an α-particle-irradiation exposure apparatus. Int. J. Radiat. Biol. 2021, 97, 1404–1416. [Google Scholar] [CrossRef]
- Semenenko, V.A.; Stewart, R.D. Fast monte carlo simulation of DNA damage formed by electrons and light ions. Phys. Med. Biol. 2006, 51, 1693–1706. [Google Scholar] [CrossRef]
- Stewart, R.D.; Yu, V.K.; Georgakilas, A.G.; Koumenis, C.; Park, J.H.; Carlson, D.J. Effects of radiation quality and oxygen on clustered DNA lesions and cell death. Radiat. Res. 2011, 176, 587–602. [Google Scholar] [CrossRef] [PubMed]
- Pavlopoulou, A.; Asfa, S.; Gioukakis, E.; Mavragani, I.V.; Nikitaki, Z.; Takan, I.; Pouget, J.P.; Harrison, L.; Georgakilas, A.G. In silico investigation of the biological implications of complex DNA damage with emphasis in cancer radiotherapy through a systems biology approach. Molecules 2021, 26, 7602. [Google Scholar] [CrossRef]
- Prasanna, P.G.; Citrin, D.E.; Hildesheim, J.; Ahmed, M.M.; Venkatachalam, S.; Riscuta, G.; Xi, D.; Zheng, G.; Deursen, J.V.; Goronzy, J.; et al. Therapy-induced senescence: Opportunities to improve anticancer therapy. J. Natl. Cancer Inst. 2021, 113, 1285–1298. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.J. Targeting the p53 protein for cancer therapies: The translational impact of p53 research. Cancer Res. 2022, 82, 362–364. [Google Scholar] [CrossRef]
- Azzam, E.I.; Jay-Gerin, J.P.; Pain, D. Ionizing radiation-induced metabolic oxidative stress and prolonged cell injury. Cancer Lett. 2012, 327, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Maier, P.; Hartmann, L.; Wenz, F.; Herskind, C. Cellular pathways in response to ionizing radiation and their targetability for tumor radiosensitization. Int. J. Mol. Sci. 2016, 17, 102. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, T. Roles of sulfur metabolism and rhodanese in detoxification and anti-oxidative stress functions in the liver: Responses to radiation exposure. Med. Sci. Monit. 2015, 21, 1721. [Google Scholar] [CrossRef]
- Yuan, Z.H.; Liu, T.; Wang, H.; Xue, L.X.; Wang, J.J. Fatty acids metabolism: The bridge between ferroptosis and ionizing radiation. Front. Cell Dev. Biol. 2021, 9, 675617. [Google Scholar] [CrossRef]
- Shibata, Y.; Yasui, H.; Higashikawa, K.; Miyamoto, N.; Kuge, Y. Erastin, a ferroptosis-inducing agent, sensitized cancer cells to X-ray irradiation via glutathione starvation in vitro and in vivo. PLoS ONE 2019, 14, e0225931. [Google Scholar] [CrossRef]
- Liao, P.; Wang, W.; Wang, W.; Kryczek, I.; Li, X.; Bian, Y.; Sell, A.; Wei, S.; Grove, S.; Johnson, J.K.; et al. Cd8(+) t cells and fatty acids orchestrate tumor ferroptosis and immunity via acsl4. Cancer Cell 2022, 40, 365–378 e366. [Google Scholar] [CrossRef]
- Olechnowicz, J.; Tinkov, A.; Skalny, A.; Suliburska, J. Zinc status is associated with inflammation, oxidative stress, lipid, and glucose metabolism. J. Physiol. Sci. 2018, 68, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Stork, C.J.; Martorano, L.M.; Li, Y.V. Uvb radiation induces an increase in intracellular zinc in human epidermal keratinocytes. Int. J. Mol. Med. 2010, 26, 463–469. [Google Scholar] [PubMed]
- Wiseman, D.A.; Wells, S.M.; Hubbard, M.; Welker, J.E.; Black, S.M. Alterations in zinc homeostasis underlie endothelial cell death induced by oxidative stress from acute exposure to hydrogen peroxide. Am. J. Physiol.—Lung Cell. Mol. Physiol. 2007, 292, 165–177. [Google Scholar] [CrossRef]
- Matsubara, J.; Shida, T.; Ishioka, K.; Egawa, S.; Inada, T.; Machida, K. Protective effect of zinc against lethality in irradiated mice. Environ. Res. 1986, 41, 558–567. [Google Scholar] [CrossRef]
- Fukada, T.; Yamasaki, S.; Nishida, K.; Murakami, M.; Hirano, T. Zinc homeostasis and signaling in health and diseases. J. Biol. Inorg. Chem. 2011, 16, 1123–1134. [Google Scholar] [CrossRef] [PubMed]
- Chan, R.K.; Ibrahim, S.I.; Verna, N.; Carroll, M.; Moore, F.D.; Hechtman, H.B. Ischaemia–reperfusion is an event triggered by immune complexes and complement. Br. J. Surg. 2003, 90, 1470–1478. [Google Scholar] [CrossRef]
- Brewer, G.J. Anticopper therapy against cancer and diseases of inflammation and fibrosis. Drug Discov. Today 2005, 10, 1103–1109. [Google Scholar] [CrossRef]
- Goodman, V.L.; Brewer, G.J.; Merajver, S.D. Copper deficiency as an anti-cancer strategy. Endocr.-Relat. Cancer 2004, 11, 255–263. [Google Scholar] [CrossRef]
- Lowndes, S.A.; Harris, A.L. The role of copper in tumour angiogenesis. J. Mammary Gland. Biol. Neoplasia 2005, 10, 299–310. [Google Scholar] [CrossRef]
- Wang, F.; Jiao, P.; Qi, M.; Frezza, M.; Dou, Q.P.; Yan, B. Turning tumor-promoting copper into an anti-cancer weapon via high-throughput chemistry. Curr. Med. Chem. 2010, 17, 2685. [Google Scholar] [CrossRef]
- Rex, D.A.B.; Agarwal, N.; Prasad, T.S.K.; Kandasamy, R.K.; Subbannayya, Y.; Pinto, S.M. A comprehensive pathway map of il-18-mediated signalling. J. Cell Commun. Signal. 2020, 14, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Piette, J. Signalling pathway activation by photodynamic therapy: Nf-κb at the crossroad between oncology and immunology. Photochem. Photobiol. Sci. 2015, 14, 1510–1517. [Google Scholar] [CrossRef] [PubMed]
- Hellweg, C.E. The nuclear factor kappab pathway: A link to the immune system in the radiation response. Cancer Lett. 2015, 368, 275–289. [Google Scholar] [CrossRef] [PubMed]
- Buonanno, M.; de Toledo, S.M.; Pain, D.; Azzam, E.I. Long-term consequences of radiation-induced bystander effects depend on radiation quality and dose and correlate with oxidative stress. Radiat. Res. 2011, 175, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Bednarski, J.J.; Sleckman, B.P. At the intersection of DNA damage and immune responses. Nat. Rev. Immunol. 2019, 19, 231–242. [Google Scholar] [CrossRef]
- Cancer Stat Facts: Leukemia—Acute Myeloid Leukemia (aml). Available online: https://seer.cancer.gov/statfacts/html/amyl.html (accessed on 7 June 2021).
- Chedotal, A.; Kerjan, G.; Moreau-Fauvarque, C. The brain within the tumor: New roles for axon guidance molecules in cancers. Cell Death Differ. 2005, 12, 1044–1056. [Google Scholar] [CrossRef]
- Logotheti, S.; Marquardt, S.; Richter, C.; Sophie Hain, R.; Murr, N.; Takan, I.; Pavlopoulou, A.; Putzer, B.M. Neural networks recapitulation by cancer cells promotes disease progression: A novel role of p73 isoforms in cancer-neuronal crosstalk. Cancers 2020, 12, 3789. [Google Scholar] [CrossRef]
- Yilmaz, H.; Toy, H.I.; Marquardt, S.; Karakulah, G.; Kucuk, C.; Kontou, P.I.; Logotheti, S.; Pavlopoulou, A. In silico methods for the identification of diagnostic and favorable prognostic markers in acute myeloid leukemia. Int. J. Mol. Sci. 2021, 22, 9601. [Google Scholar] [CrossRef]
- Rola, R.; Sarkissian, V.; Obenaus, A.; Nelson, G.A.; Otsuka, S.; Limoli, C.L.; Fike, J.R. High-let radiation induces inflammation and persistent changes in markers of hippocampal neurogenesis. Radiat. Res. 2005, 164, 556–560. [Google Scholar] [CrossRef]
- Alboni, S.; Cervia, D.; Sugama, S.; Conti, B. Interleukin 18 in the cns. J. Neuroinflammation 2010, 7, 1–12. [Google Scholar] [CrossRef]
- Felderhoff-Mueser, U.; Schmidt, O.I.; Oberholzer, A.; Bührer, C.; Stahel, P.F. Il-18: A key player in neuroinflammation and neurodegeneration? Trends Neurosci. 2005, 28, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Min, S.S.; Wierzbicki, A.S. Radiotherapy, chemotherapy and atherosclerosis. Curr. Opin. Cardiol. 2017, 32, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Sun, C.; Gerdes, N.; Liu, C.; Liao, M.; Liu, J.; Shi, M.A.; He, A.; Zhou, Y.; Sukhova, G.K.; et al. Interleukin 18 function in atherosclerosis is mediated by the interleukin 18 receptor and the na-cl co-transporter. Nat. Med. 2015, 21, 820–826. [Google Scholar] [CrossRef] [PubMed]
- Bodo, S.; Campagne, C.; Thin, T.H.; Higginson, D.S.; Vargas, H.A.; Hua, G.; Fuller, J.D.; Ackerstaff, E.; Russell, J.; Zhang, Z.; et al. Single-dose radiotherapy disables tumor cell homologous recombination via ischemia/reperfusion injury. J. Clin. Investig. 2019, 129, 786–801. [Google Scholar] [CrossRef]
- Wambersie, A.; Hendry, J.; Gueulette, J.; Gahbauer, R.; Potter, R.; Gregoire, V. Radiobiological rationale and patient selection for high-let radiation in cancer therapy. Radiother. Oncol. 2004, 73 (Suppl. S2), S1–S14. [Google Scholar] [CrossRef]
- Dressman, H.K.; Muramoto, G.G.; Chao, N.J.; Meadows, S.; Marshall, D.; Ginsburg, G.S.; Nevins, J.R.; Chute, J.P. Gene expression signatures that predict radiation exposure in mice and humans. PLoS Med. 2007, 4, e106. [Google Scholar] [CrossRef]
- Tran, K.A.; Kondrashova, O.; Bradley, A.; Williams, E.D.; Pearson, J.V.; Waddell, N. Deep learning in cancer diagnosis, prognosis and treatment selection. Genome Med. 2021, 13, 152. [Google Scholar] [CrossRef]
- Hiraga, T.; Ito, S.; Nakamura, H. Cancer stem-like cell marker cd44 promotes bone metastases by enhancing tumorigenicity, cell motility, and hyaluronan production. Cancer Res. 2013, 73, 4112–4122. [Google Scholar] [CrossRef]
- Owyong, M.; Chou, J.; van den Bijgaart, R.J.; Kong, N.; Efe, G.; Maynard, C.; Talmi-Frank, D.; Solomonov, I.; Koopman, C.; Hadler-Olsen, E.; et al. Mmp9 modulates the metastatic cascade and immune landscape for breast cancer anti-metastatic therapy. Life Sci. Alliance 2019, 2, e201800226. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, J.; Wan, L.; Zhou, X.; Wang, Z.; Wei, W. Targeting cdc20 as a novel cancer therapeutic strategy. Pharmacol. Ther. 2015, 151, 141–151. [Google Scholar] [CrossRef]
- Mahner, S.; Baasch, C.; Schwarz, J.; Hein, S.; Wolber, L.; Janicke, F.; Milde-Langosch, K. C-fos expression is a molecular predictor of progression and survival in epithelial ovarian carcinoma. Br. J. Cancer 2008, 99, 1269–1275. [Google Scholar] [CrossRef] [PubMed]
- Asem, M.S.; Buechler, S.; Wates, R.B.; Miller, D.L.; Stack, M.S. Wnt5a signaling in cancer. Cancers 2016, 8, 79. [Google Scholar] [CrossRef] [PubMed]
- Toy, H.I.; Karakulah, G.; Kontou, P.I.; Alotaibi, H.; Georgakilas, A.G.; Pavlopoulou, A. Investigating molecular determinants of cancer cell resistance to ionizing radiation through an integrative bioinformatics approach. Front. Cell Dev. Biol. 2021, 9, 620248. [Google Scholar] [CrossRef] [PubMed]
- Bridges, A.E.; Ramachandran, S.; Pathania, R.; Parwal, U.; Lester, A.; Rajpurohit, P.; Morera, D.S.; Patel, N.; Singh, N.; Korkaya, H.; et al. Rad51ap1 deficiency reduces tumor growth by targeting stem cell self-renewal. Cancer Res. 2020, 80, 3855–3866. [Google Scholar] [CrossRef] [PubMed]
- Chandler, B.C.; Moubadder, L.; Ritter, C.L.; Liu, M.; Cameron, M.; Wilder-Romans, K.; Zhang, A.; Pesch, A.M.; Michmerhuizen, A.R.; Hirsh, N.; et al. Ttk inhibition radiosensitizes basal-like breast cancer through impaired homologous recombination. J. Clin. Investig. 2020, 130, 958–973. [Google Scholar] [CrossRef]
- Shih, H.T.; Chen, W.Y.; Wang, H.Y.; Chao, T.; Huang, H.D.; Chou, C.H.; Chang, Z.F. Dnmt3b protects centromere integrity by restricting r-loop-mediated DNA damage. Cell Death Dis. 2022, 13, 546. [Google Scholar] [CrossRef]
- Wu, C.; Guo, E.; Ming, J.; Sun, W.; Nie, X.; Sun, L.; Peng, S.; Luo, M.; Liu, D.; Zhang, L.; et al. Radiation-induced dnmt3b promotes radioresistance in nasopharyngeal carcinoma through methylation of p53 and p21. Mol. Ther. Oncolytics 2020, 17, 306–319. [Google Scholar] [CrossRef]
- Soo Lee, N.; Jin Chung, H.; Kim, H.J.; Yun Lee, S.; Ji, J.H.; Seo, Y.; Hun Han, S.; Choi, M.; Yun, M.; Lee, S.G.; et al. Traip/rnf206 is required for recruitment of rap80 to sites of DNA damage. Nat. Commun. 2016, 7, 10463. [Google Scholar] [CrossRef]
- Herrera, F.G.; Ronet, C.; Ochoa de Olza, M.; Barras, D.; Crespo, I.; Andreatta, M.; Corria-Osorio, J.; Spill, A.; Benedetti, F.; Genolet, R.; et al. Low-dose radiotherapy reverses tumor immune desertification and resistance to immunotherapy. Cancer Discov. 2022, 12, 108–133. [Google Scholar] [CrossRef]
- Algara, M.; Arenas, M.; Marin, J.; Vallverdu, I.; Fernandez-Leton, P.; Villar, J.; Fabrer, G.; Rubio, C.; Montero, A. Low dose anti-inflammatory radiotherapy for the treatment of pneumonia by covid-19: A proposal for a multi-centric prospective trial. Clin. Transl. Radiat. Oncol. 2020, 24, 29–33. [Google Scholar] [CrossRef]
- Zhang, Q.; Matzke, M.; Schepmoes, A.A.; Moore, R.J.; Webb-Robertson, B.J.; Hu, Z.; Monroe, M.E.; Qian, W.J.; Smith, R.D.; Morgan, W.F. High and low doses of ionizing radiation induce different secretome profiles in a human skin model. PLoS ONE 2014, 9, e92332. [Google Scholar] [CrossRef] [PubMed]
- Multhoff, G.; Radons, J. Radiation, inflammation, and immune responses in cancer. Front. Oncol. 2012, 2, 58. [Google Scholar] [CrossRef] [PubMed]
- Casey, D.L.; Pitter, K.L.; Wexler, L.H.; Slotkin, E.K.; Gupta, G.P.; Wolden, S.L. Tp53 mutations increase radioresistance in rhabdomyosarcoma and ewing sarcoma. Br. J. Cancer 2021, 125, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Logotheti, S.; Pavlopoulou, A.; Galtsidis, S.; Vojtesek, B.; Zoumpourlis, V. Functions, divergence and clinical value of tap73 isoforms in cancer. Cancer Metastasis Rev. 2013, 32, 511–534. [Google Scholar] [CrossRef] [PubMed]
- Logotheti, S.; Pavlopoulou, A.; Marquardt, S.; Takan, I.; Georgakilas, A.G.; Stiewe, T. P73 isoforms meet evolution of metastasis. Cancer Metastasis Rev. 2022. [Google Scholar] [CrossRef]
- Aznar, M.C.; Bacchus, C.; Coppes, R.P.; Deutsch, E.; Georg, D.; Haustermans, K.; Hoskin, P.; Krause, M.; Lartigau, E.F.; Lock, S.; et al. Radiation oncology in the new virtual and digital era. Radiother. Oncol. 2021, 154, A1–A4. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sagkrioti, E.; Biz, G.M.; Takan, I.; Asfa, S.; Nikitaki, Z.; Zanni, V.; Kars, R.H.; Hellweg, C.E.; Azzam, E.I.; Logotheti, S.; et al. Radiation Type- and Dose-Specific Transcriptional Responses across Healthy and Diseased Mammalian Tissues. Antioxidants 2022, 11, 2286. https://doi.org/10.3390/antiox11112286
Sagkrioti E, Biz GM, Takan I, Asfa S, Nikitaki Z, Zanni V, Kars RH, Hellweg CE, Azzam EI, Logotheti S, et al. Radiation Type- and Dose-Specific Transcriptional Responses across Healthy and Diseased Mammalian Tissues. Antioxidants. 2022; 11(11):2286. https://doi.org/10.3390/antiox11112286
Chicago/Turabian StyleSagkrioti, Eftychia, Gökay Mehmet Biz, Işıl Takan, Seyedehsadaf Asfa, Zacharenia Nikitaki, Vassiliki Zanni, Rumeysa Hanife Kars, Christine E. Hellweg, Edouard I. Azzam, Stella Logotheti, and et al. 2022. "Radiation Type- and Dose-Specific Transcriptional Responses across Healthy and Diseased Mammalian Tissues" Antioxidants 11, no. 11: 2286. https://doi.org/10.3390/antiox11112286
APA StyleSagkrioti, E., Biz, G. M., Takan, I., Asfa, S., Nikitaki, Z., Zanni, V., Kars, R. H., Hellweg, C. E., Azzam, E. I., Logotheti, S., Pavlopoulou, A., & Georgakilas, A. G. (2022). Radiation Type- and Dose-Specific Transcriptional Responses across Healthy and Diseased Mammalian Tissues. Antioxidants, 11(11), 2286. https://doi.org/10.3390/antiox11112286









