On the Potential Role of the Antioxidant Couple Vitamin E/Selenium Taken by the Oral Route in Skin and Hair Health
Abstract
:1. Introduction
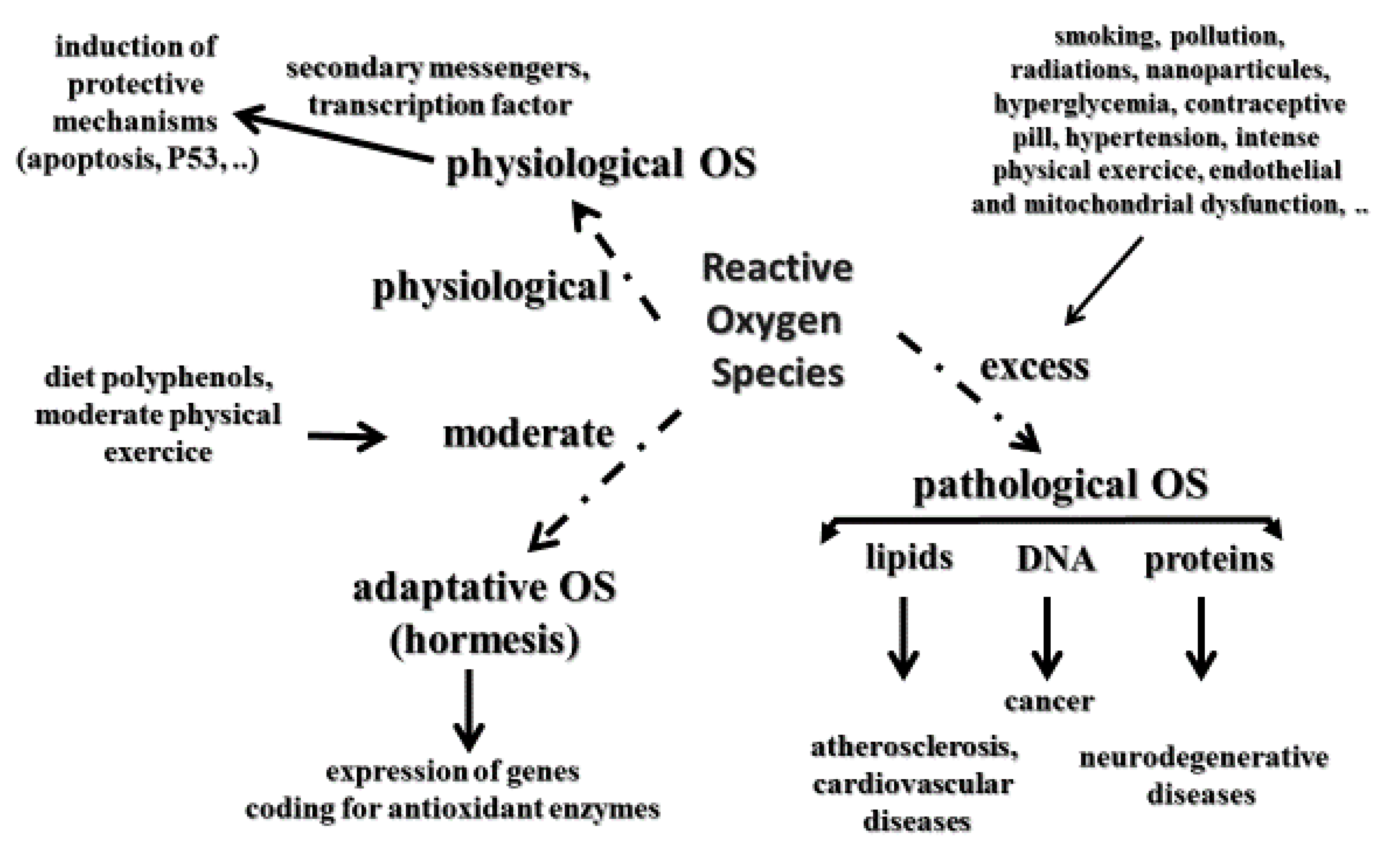
2. Oxidative Stress Definitions
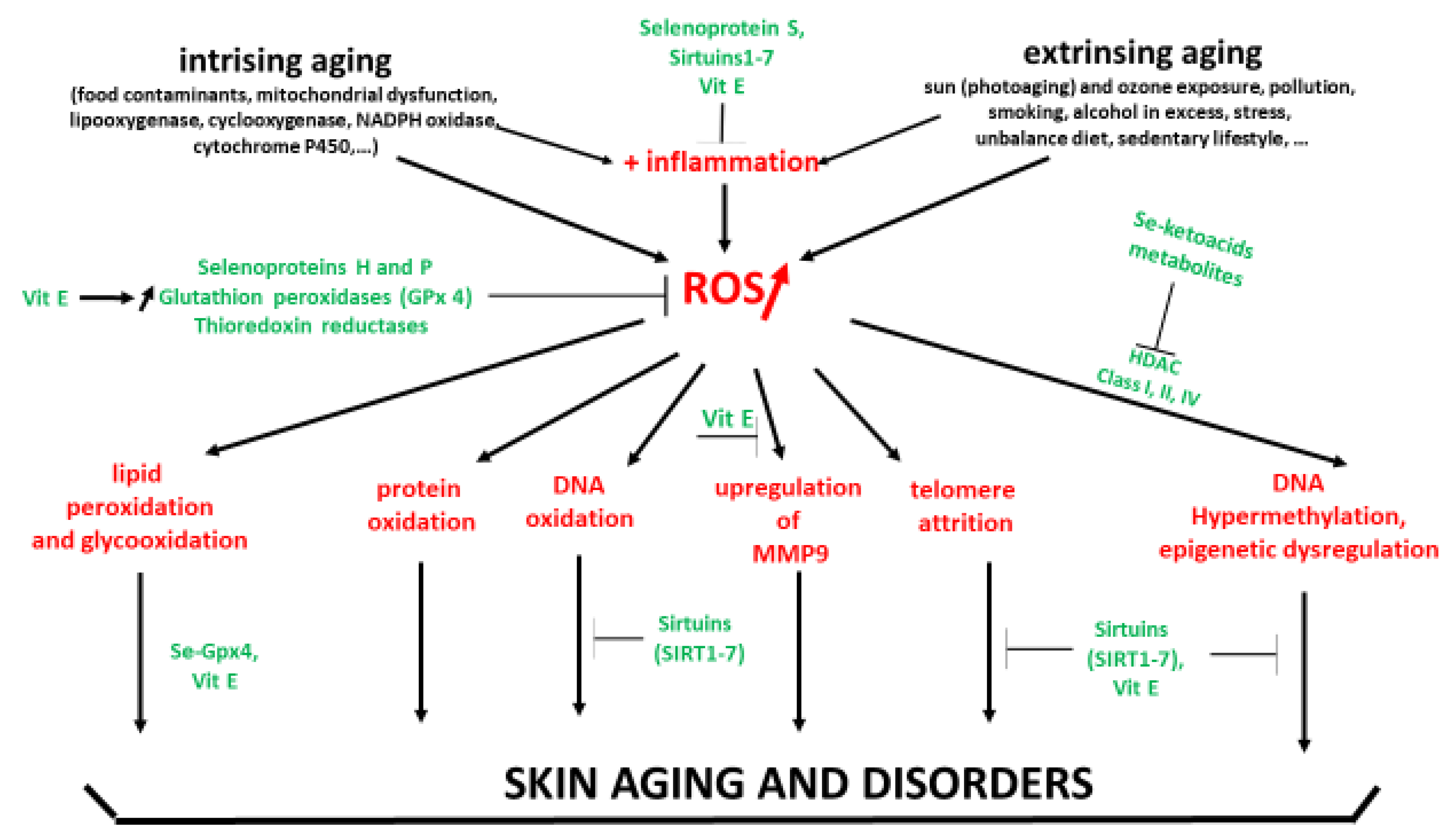
3. Oxidative Stress and Skin
4. The Skin Antioxidant Network
5. Selenium
5.1. Physiological Functions
5.2. Selenium, Skin Aging and Disorders
5.3. Se Plasma Concentration, Skin Disorders and Supplementation
6. Vitamin E
6.1. Physiological Function
6.2. Vitamin E, Skin Aging and Disorders
6.3. Vitamin E Plasma Concentration, Skin Disorders and Supplementation
7. Oxidative Stress, Antioxidants, and Aging Hair
8. Synergic Antioxidant Action of Vitamin E/Se
9. Discussion
10. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AA | alopecia aerata |
| AE | antioxidant efficacy |
| AGA | androgenetic alopecia |
| AGEs | advanced glycation end products |
| AMK | N1-acetyl-5-methoxykynuramine |
| AFMK | N1-acetyl-N2-formyl-5-methoxykynuramine |
| AP-1 | activator protein |
| ARE | Antioxidant Response Element |
| CV | cyclic voltametry |
| Dis | 3-deiodinases |
| DNA | deoxyribonucleic acid |
| DNAm | deoxyribonucleic acid methylation |
| DNDMT | DNA demethyltransferase |
| DNMT | DNA methyltransferase |
| GPx | glutathione peroxidase |
| GSH | glutathione |
| GSSG | oxidized glutathione |
| HAT | histone acetyltransferase |
| HDAC | Histone deacetylase |
| HDMT | histone demethyltransferase |
| HMT | histone methyltransferase |
| H2O2 | hydrogen peroxide |
| hTERT | telomerase reverse transcriptase |
| Keap 1 | Kelch-like ECH-associated protein 1 |
| MDA | malonaldehyde |
| MED | minimal erythrema dose |
| MFRTA | Mitochondrial Free Radical Theory of Aging |
| MMP | matrix metalloproteinase |
| NF-κB | nuclear factor kappa B |
| Nrf2 | Nuclear factor (erythroid-derived 2)-like 2 |
| O2 | oxygen |
| 8-OHdG | 8-hydroxy-2′-deoxyguanosine |
| 8-oxoGua | 8-oxo-7,8-dihydroguanine |
| OS | oxidative stress |
| OSWV | osteryoung square wave voltammetry |
| PI3-K | phosphatidylinositol 3-kinase |
| PKC | protein kinace C |
| R• | lipidic radical |
| RH | polyunsaturated free fatty acid |
| ROO• | peroxyl radical |
| ROOH | lipid peroxide |
| ROS | reactive oxygen species |
| SC | striatum corneum |
| -SH | thiol group |
| SIRT1-7 | silencing information regulator 1-7 |
| SM | sodium selenomethionine |
| Se | selenium |
| SeCyS | selenocysteine |
| SELECT | Selenium and Vitamin E Cancer Prevention Trial |
| SePP1 or SELENOP | selenoprotein P |
| SOD | superoxide dismutase |
| SS | sodium selenite |
| TE | telogen effluvium |
| TRxR1, TRxR2, TRxR3 | Thioredoxin reductases 1, 2, 3 |
| UVA and UVB | ultraviolet A and B |
| Vit C | vitamin C |
| Vit C• | ascorbyl radical |
| Vit E | vitamin E |
| Vit E• | tocopheryl radical |
| WHO | World Health Organization |
References
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harman, D. Aging: A theory based on free radical and radiation chemistry. J. Geront. 1956, 11, 298–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sohal, R.S.; Orr, W.C. The redox stress hypothesis of aging. Free. Radic. Biol. Med. 2012, 52, 539–555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viña, J.; Borras, C.; Abdelaziz, K.M.; Garcia-Valles, R.; Gomez-Cabrera, M.C. The free radical theory of aging revisited: The cell signaling disruption theory of aging. Antiox Redox Signal. 2013, 19, 779–787. [Google Scholar] [CrossRef] [Green Version]
- Ludovico, P.; Burhans, W.C. Reactive oxygen species, aging and the hormesis police. FEMS Yeast Res. 2014, 14, 33–39. [Google Scholar] [CrossRef] [Green Version]
- Farage, M.A.; Miller, K.W.; Elsner, P.; Maibach, H.I. Intrinsic and extrinsic factors in skin aging: A review. Int. J. Cosmet. Sci. 2008, 30, 87–95. [Google Scholar] [CrossRef]
- Poljšak, B.; Dahmane, R.; Godic, A. Intrinsic skin aging: The role of oxidative stress. Acta Derm. Alp Pannonica Adriat. 2012, 21, 33–36. [Google Scholar]
- Poljšak, B.; Dahmane, R. Free radicals and extrinsic skin aging. Dermatol. Res. Pract. 2012, 2012, 135206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trüeb, R.M. Oxidative stress and its impact on skin, scalp and hair. Int. J. Cosm. Sci. 2021, 43, 9–13. [Google Scholar] [CrossRef]
- Schagen, S.K.; Zampeli, V.A.; Makrantonaki, E.; Zouboulis, C.C. Discovering the link between nutrition and skin aging. Dermato-endocrinology 2012, 4, 298–307. [Google Scholar] [CrossRef] [Green Version]
- Cao, C.; Xiao, Z.; Wu, Y.; Ge, C. Diet and skin aging- from the perspective food nutrition. Nutrients 2020, 12, 870. [Google Scholar] [CrossRef] [PubMed]
- Diotallevi, F.; Campanati, A.; Martina, E.; Radi, G.; Paolinelli, M.; Marani, A.; Molinelli, E.; Candelora, M.; Taux, M.; Galeazzi, T.; et al. The role of nutrition in immnune-mediated, infammatory skin disease: A narrative review. Nutrients 2020, 14, 591. [Google Scholar] [CrossRef] [PubMed]
- Boelsma, E.; Hendriks, H.F.; Roza, L. Nutritional skin care: Health effects of micronutrients and fatty acids. Clin. Nutr. 2001, 73, 853–864. [Google Scholar] [CrossRef] [Green Version]
- Oresajo, C.; Pillai, S.; Yatskayer, M.; Puccetti, G.; McDaniel, D.H. Antioxidants and skin aging: A Review. Cosmet. Dermatol. 2009, 22, 563–570. [Google Scholar]
- Sant’Anna Addor, F.A.; Ponzio, H.; Ravelli, F.N. Oral Photoprotection. In Daily Routine in Cosmetic Dermatology; Clinical Approaches and Procedures in Cosmetic Dermatology; Issa, M., Tamura, B., Eds.; Springer: Cham, Switzerland, 2016. [Google Scholar] [CrossRef]
- Pérez-Sánchez, A.; Barrajón-Catalán, E.; Herranz-López, M.; Micol, V. Nutraceuticals for skin care: A comprehensive review of human clinical studies. Nutrients 2018, 10, 403. [Google Scholar] [CrossRef] [Green Version]
- Vollmer, D.L.; West, V.A.; Lephart, E.D. Enhancing skin heatlh: By oral administration of natural compounds and minerals with implications to the dermal microbiome. Int. J. Mol. Sci. 2018, 19, 359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, Q.; Zhang, Y.; Lou, H.; Ou, Z.; Liu, J.; Duan, W.; Wang, H.; Ge, Y.; Min, J.; Wang, F.; et al. GPX4 and vitamin E cooperatively protect hematopoietic stem and progenitor cells from lipid peroxidation and ferroptosis. Cell Death Dis. 2021, 12, 706. [Google Scholar] [CrossRef]
- Briganti, S.; Picardo, M. Antioxidant activity, lipid peroxidation and skin diseases. What’s new? J. Eur. Acad. Dermatol. Venereol. 2003, 17, 663–669. [Google Scholar] [CrossRef]
- Packer, L.; Slater, T.F.; Willson, R.L. Direct observation of a free radical interaction between vitamin E and vitamin C. Nature 1979, 278, 737–738. [Google Scholar] [CrossRef]
- Cozza, G.; Rossetto, M.; Bosello-Travain, V.; Maiorino, M.; Roveri, A.; Toppo, S.; Zaccarin, M.; Zennaro, L.; Ursini, F. Glutathione peroxidase 4-catalyzed reduction of lipid hydroperoxides in membranes: The polar head of membrane phospholipids binds the enzyme and addresses the fatty acid hydroperoxide group toward the redox center. Free Radic. Biol. Med. 2017, 112, 1–11. [Google Scholar] [CrossRef]
- Juan, C.A.; Pérez de la Lastra, J.M.; Plou, F.J.; Pérez-Lebeña, E. The chemistry of reactive oxygen species (ROS) revisited: Outlining their role in biological macromolecules (DNA, lipids and proteins) and induced pathologies. Int. J. Mol. Sci. 2021, 22, 4642. [Google Scholar] [CrossRef] [PubMed]
- Birben, E.; Sahiner, M.; Sackesen, S.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense. WAO J. 2012, 5, 9–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- D’Oria, R.; Schipani, R.; Leonardini, A.; Natalicchio, A.; Perrini, S.; Cignarelli, A.; Laviola, L.; Giorgino, F. The role of oxidative stress in cardiac disease: From physiological response to injury factor. Oxidative Med. Cell. Longev. 2020, 2020, 5732956. [Google Scholar] [CrossRef] [PubMed]
- Loft, S.; Poulsen, H.E. Cancer risk and oxidative DNA damage in man. J. Mol. Med. 1996, 74, 297–312. [Google Scholar] [CrossRef] [PubMed]
- Grimm, S.; Hoehn, A.; Davies, K.J.; Grune, T. Protein oxidative modifications in the aging brain: Consequence for the onset of neurodegenerative disease. Free Radic. Res. 2011, 45, 73–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, R.; Rinker, L.; Peng, J.; Chilian, W.M. Reactive oxygen species: The good and the bad. Reactive oxygen species in living cells. In Reactive Oxygen Species in Living Cells; Cristiana, F., Elena, A., Eds.; Chapter 2; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef] [Green Version]
- Taufenberger, A.; Magistretti, P.J. Reactive oxygen species: Beyond their reactive behavior. Neurochem. Res. 2021, 46, 77–87. [Google Scholar] [CrossRef]
- Checa, J.; Aran, J.M. Reactive oxygen species: Drivers of physiological and pathological processes. J. Inflamm. Res. 2020, 13, 1057–1073. [Google Scholar] [CrossRef]
- Sies, H.; Jones, D.P. Oxidative stress. In Encyclopedia of Stress, 2nd ed.; Fink, G., Ed.; Elsevier: Amsterdam, The Netherlands, 2007; pp. 45–48. [Google Scholar]
- Birringer, M. Hormetics: Dietary triggers of an adaptative stress response. Pharm. Res. 2011, 28, 2680–2694. [Google Scholar] [CrossRef]
- Nitti, M.; Marengo, B.; Furfaro, A.L.; Pronzato, M.A.; Marinari, U.M.; Domenicotti, C.; Traverso, N. Hormesis and oxidative distress: Pathophysiology of reactive oxygen species and the open question of antioxidant modulation and supplementation. Antioxidants 2022, 11, 1613. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Kozumbo, W.J. The hormetic dose-response mechanism: Nrf2 activation. Pharmacol. Res. 2021, 167, 105526. [Google Scholar] [CrossRef]
- Omenn, G.S.; Goodman, G.E.; Thornquist, M.D.; Balmes, J.; Cullen, M.R.; Glass, A.; Keogh, J.P.; Meyskens, F.L., Jr.; Valanis, B.; Williams, J.H., Jr.; et al. Risk factors for lung cancer and for intervention effects in CARET, the Beta-Carotene and Retinol Efficacy Trial. J. Natl. Cancer Inst. 1996, 88, 1550–1559. [Google Scholar]
- Hercberg, S.; Ezzedine, K.; Guinot, C.; Preziosi, P.; Galan, P.; Bertrais, S.; Estaquio, C.; Briancxon, S.; Favier, A.; Latreille, J.; et al. Antioxidant supplementation increases the risk of skin cancers in women but not in men. J. Nutr. 2007, 137, 2098–2105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trouba, J.; Hamade, H.K.; Amin, R.O.; Germolec, D.R.J. Oxidative stress and its role in skin disease. Antioxid. Redox Signal. 2002, 4, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Nishigori, C.; Hattori, Y.; Toyokuni, S. Role of reactive oxygen species in skin carcinogenesis. Antioxid. Redox Signal. 2004, 6, 561–570. [Google Scholar] [CrossRef]
- Bickers, D.R.; Athar, M. Oxidative stress in the pathogenesis of skin disease. J. Investig. Dermatol. 2006, 126, 2565–2575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rinnerthaler, M.; Bischof, J.; Streubel, M.K.; Trost, A.; Richter, K. Oxidative stress in aging human skin. Biomolecules 2015, 5, 545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xian, D.; Lai, R.; Song, J.; Xiong, X.; Zhong, J. Emerging perspective: Role of increased ROS and redox imbalance in skin carcinogenesis. Oxidative Med. Cell. Longev. 2019, 2019, 8127362. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.; Park, H.J. Molecular mechanisms of skin aging and rejuvenation. In Molecular Mechanisms of the Aging Process and Rejuvenation; Shiomi, N., Ed.; IntechOpen: London, UK, 2016. [Google Scholar] [CrossRef] [Green Version]
- Parrado, C.; Mercado-Saenz, S.; Perez-Davo, A.; Gilaberte, Y.; Gonzalez, S.; Juarranz, A. Environmental stressors on skin aging. Mechanistic Insights. Front. Pharmacol. 2019, 10, 759. [Google Scholar] [CrossRef]
- Uitto, J. The role of elastin and collagen in cutaneous aging: Intrinsic aging versus photoexposure. J. Drugs Dermatol. 2008, 7, s12. [Google Scholar]
- Vierkötter, A.; Krutmann, J. Environmental influences on skin aging and ethnic-specific manifestations. Dermato-Endocrinology 2012, 4, 227–231. [Google Scholar] [CrossRef] [Green Version]
- El-Domyati, M.; Attia, S.; Saleh, F.; Brown, D.; Birk, D.E.; Gasparro, F.; Ahmad, H.; Uitto, J. Intrinsic aging vs. photoaging: A comparative histopathological, immunohistochemical, and ultrastructural study of skin. Exp. Dermatol. 2002, 11, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Zheng, Y.W.; Liu, Q.; Liu, L.P.; Luo, F.-L.; Zhou, H.-C.; Isoda, H.; Ohkohchi, N.; Li, Y.-M. Reactive oxygen species in skin repair, regeneration, aging and inflammation. In Reactive Oxygen Species (ROS) in Living Cells; Chapter 5; Filip, C., Albu, E., Eds.; IntechOpen: London, UK, 2018. [Google Scholar]
- Zastrow, L.; Groth, N.; Klein, F.; Kockott, D.; Lademann, J.; Renneberg, R.; Ferrero, L. The missing link—Light-induced (280–1600 nm) free radical formation in human skin. Ski. Pharmacol. Physiol. 2009, 22, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Jurkiewicz, B.A.; Buettner, G.R. EPR detection of free radicals in UV-irradiated skin: Mouse versus human. Photochem. Photobiol. 1996, 64, 918–922. [Google Scholar] [CrossRef] [PubMed]
- Ou-Yang, H.; Stamatas, G.; Saliou, C.; Kollias, N. A chemiluminescence study of UVA-induced oxidative stress in human skin in vivo. J. Investig. Dermatol. 2004, 122, 1020–1029. [Google Scholar] [CrossRef] [Green Version]
- Sakurai, H.; Yasui, H.; Yamada, Y.; Nishimuraa, H.; Shigemotoa, M. Detection of reactive oxygen species in the skin of live mice and rats exposed to UVA light: A research review on chemiluminescence and trials for UVA protection. Photochem. Photobiol. Sci. 2005, 4, 715–720. [Google Scholar] [CrossRef]
- Hakozaki, T.; Date, A.; Yoshii, T.; Toyokuni, S.; Yasui, H.; Sakurai, H. Visualization and characterization of UVB-induced reactive oxygen species in a human skin equivalent model. Arch Dermatol. Res. 2008, 300, 51–56. [Google Scholar] [CrossRef]
- Meinke, M.C.; Lauer, A.C.; Haaga, S.F.; Darvina, M.E.; Groth, N.; Lademann, J. Cutaneous radical scavenging effects of orally administered antioxidants measured by electron paramagnetic resonance spectroscopy. E-SPEN J. 2012, 7, e160–e166. [Google Scholar] [CrossRef]
- Wenk, P.; Brenneisen, P.; Meewes, C.; Wlaschek, M.; Peters, T.; Blaudschun, R.; Ma, W.; Kuhr, L.; Schneider, L.; Scharffetter-Kochanek, K. UV-induced oxidative stress and photoaging. In Oxidants and Antioxidants in Cutaneous Biology; Thiele, J., Elsner, P., Eds.; Karger: Basel, Switzerland, 2001; pp. 83–94. [Google Scholar]
- Niki, E. Lipid oxidation in skin. Free. Radic. Res. 2015, 49, 827–834. [Google Scholar] [CrossRef]
- Sanders, S.; Chang, H.; Salzmann, S.; Müller, C.S.L.; Ekanayake-Mudiyanselage, S.; Elsner, P.; Thiele, J.J. Photoaging is associated with protein oxidation in human skin in vivo. J. Investig. Dermatol. 2002, 118, 618–625. [Google Scholar] [CrossRef] [Green Version]
- Ahsanuddin, S.; Lam, M.; Baron, E.D. Skin aging and oxidative stress. Mol. Sci. 2016, 3, 187–195. [Google Scholar] [CrossRef]
- Kwam, E.; Tyrell, R.M. Induction of oxidative DNA base damage in human skin cells by UV and near visible radiation. Carcinogenesis 1997, 18, 2379–2384. [Google Scholar]
- Pourzand, C.; Albieri-Borges, A.; Raczek, N.N. Shedding a new light on skin aging, Iron- and redox-homeostasis and emerging natural antioxidants. Antioxidants 2022, 11, 471. [Google Scholar] [CrossRef] [PubMed]
- Fuks, K.B.; Woodby, B.; Valacchi, G. Skin damage by tropospheric Ozone. Der Hautarzt 2019. [Google Scholar] [CrossRef] [PubMed]
- e Silva, S.A.M.; Michniak-Kohn, B.; Leonardy, G.R. An overview about oxidation in clinical practice of aging. Ann. Bras. Dermatol. 2017, 92, 367–374. [Google Scholar] [CrossRef] [Green Version]
- Blazer, S.; Khankin, E.; Segev, Y.; Ofir, R.; Yalon-Hacohen, M.; Kra-Oz, Z.; Gottfried, Y.; Larisch, S.; Skorecki, K.L. High glucose-induced replicative senescence: Point of no return and effect of telomerase. Biochem. Biophys. Res. Commun. 2002, 296, 93–101. [Google Scholar] [CrossRef]
- Noordam, R.; Gunn, D.A.; Tomlin, C.C.; Maier, A.B.; Mooijaart, S.P.; Slagboom, P.E.; Westendorp, R.G.J.; de Craen, A.J.M.; van Heemst, D. High serum glucose levels are associated with a higherperceived age. Age 2013, 35, 189–195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gkogkolou, P.; Böhm, M. Advanced glycation end products. Key players in skin aging? Dermato-endocrinology 2012, 4, 259–270. [Google Scholar] [CrossRef] [Green Version]
- Sajithal, G.B.; Chandrakasan, G. Role of lipid peroxidation products in the formation of advanced glycation end products: An in vitro study on collagen. Proc. Indian Acad. Sci. 1999, 111, 215–229. [Google Scholar] [CrossRef]
- Sergi, D.; Boulestin, H.; Campbell, F.M.; Williams, L.M. The role of dietary advanced glycation end products in metabolic dysfunction. Mol. Nutr. Food Res. 2021, 65, e1900934. [Google Scholar] [CrossRef]
- Wang, X.; Liu, J.; Yang, Y.; Zhang, X. An update on the potential role of advanced glycation end products in glycolipid metabolism. Life Sci. 2020, 245, 117344. [Google Scholar] [CrossRef]
- Song, Q.; Liu, J.; Dong, L.; Wang, X.; Zhang, X. Novel advances in inhibiting advanced glycation end product formation using natural compounds. Biomed. Pharmacother. 2021, 140, 111750. [Google Scholar] [CrossRef] [PubMed]
- Nowotny, K.; Jung, T.; Höhn, A.; Weber, D.; Grune, T. Advanced glycation end products and oxidative stress in type 2 diabetes mellitus. Biomolecules 2015, 5, 194–222. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Cao, X.; Song, F.; Xie, T.; Ji, S.; Miao, M.; Dong, J.; Tian, M.; Lin, Y.; Lu, S. Reduced dermis thickness and AGE accumulation in diabetic abdominal skin. Int. J. Low. Extrem. Wounds 2012, 11, 224–230. [Google Scholar] [CrossRef]
- Masaki, H.; Okano, Y.; Sakurai, H. Generation of active oxygen species from advanced glycation end-products (AGE) under ultraviolet light A (UVA) irradiation. Biochem. Biophys. Res. Commun. 1997, 235, 306–310. [Google Scholar] [CrossRef]
- Jeanmaire, C.; Danoux, L.; Pauly, G. Glycation during human dermal intrinsic and actinic aging: An in vivo and in vitro model study. Br. J. Dermatol. 2001, 145, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Danby, F.W. Nutrition and aging skin: Sugar and glycation. Clin. Dermatol. 2010, 28, 409–411. [Google Scholar] [CrossRef]
- Beisswenger, P.J.; Howell, S.; Mackenzie, T.; Corstjens, H.; Muizzuddin, N.; Matsui, M.S. Two fluorescent wavelengths, 440(ex)/520(em) nm and 370(ex)/440(em) nm, reflect advanced glycation and oxidation end products in human skin without diabetes. Diabetes Technol. Ther. 2012, 14, 285–292. [Google Scholar] [CrossRef]
- Daffu, G.; Hurtado del Pozo, C.; O’Shea, M.; Ananthakrishnan, R.; Ramasamy, R.; Schmidt, A.-M. Radical roles for RAGE in the pathogenesis of oxidative stress in cardiovascular diseases and beyond. Int. J. Mol. Sci. 2013, 14, 19891–19910. [Google Scholar] [CrossRef] [Green Version]
- Guarneri, F.; Custurone, P.; Papaianni, V.; Gangemi, S. Involvement of RAGE and oxidative stress in inflammatory and infectious skin diseases. Antioxidants 2021, 10, 82. [Google Scholar] [CrossRef]
- Louzona, M.; Coeurdassiera, M.; Gimbert, F.; Pauget, B.; de Vaufleurya, A. Telomere dynamic in humans and animals: Review and perspectives in environmental toxicology. Environ. Int. 2019, 131, 105025. [Google Scholar] [CrossRef]
- Vaiserman, A.; Krasnienkov, D. Telomere length as a marker of biological age: State-of-the-art, open Iissues, and future perspectives. Front. Genet. 2021, 11, 630186. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Hog, Y.; Kim, M. Structural and functional changes and possible molecular mechanisms in aged skin. Int. J. Mol. Sci. 2021, 22, 12489. [Google Scholar] [CrossRef]
- Oikawa, S.; Kawanishi, S. Site-specific DNA damage at GGG sequence by oxidative stress may accelerate telomere shortening. FEBS Lett. 1999, 453, 365–368. [Google Scholar] [CrossRef] [Green Version]
- von Zglinicki, T. Oxidative stress shortens telomeres. Trends Biochem. Sci. 2002, 27, 339–344. [Google Scholar] [CrossRef]
- Reichert, S.; Stier, A. Does oxidative stress shorten telomeres in vivo? A review. Biol. Lett. 2017, 13, 20170463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barnes, R.P.; Fouquerel, E.; Opresko, P.L. The impact of DNA damage and stress on telomere homeostasis. Mech. Aging Dev. 2019, 177, 37–45. [Google Scholar] [CrossRef]
- Buckingham, E.M.; Klingelhutz, A.J. The role of telomeres in the aging of human skin. Exp. Dermatol. 2011, 20, 297–302. [Google Scholar] [CrossRef]
- Sugimoto, M.; Yamashita, R.; Ueda, M. Telomere length of the skin in association with chronological aging and photoaging. J. Dermatol. Sci. 2006, 43, 43–47. [Google Scholar] [CrossRef]
- Kosmadaki, M.G.; Gilchrest, B.A. The role of telomeres in skin aging/photoaging. Micron 2004, 35, 155–159. [Google Scholar] [CrossRef]
- Jacczak, B.; Rubis, B.; Totoni, Z. Potential of naturally derived compounds in telomerase and telomere modulation in skin senescence and aging. Int. J. Mol. Sci. 2021, 22, 6381. [Google Scholar] [CrossRef]
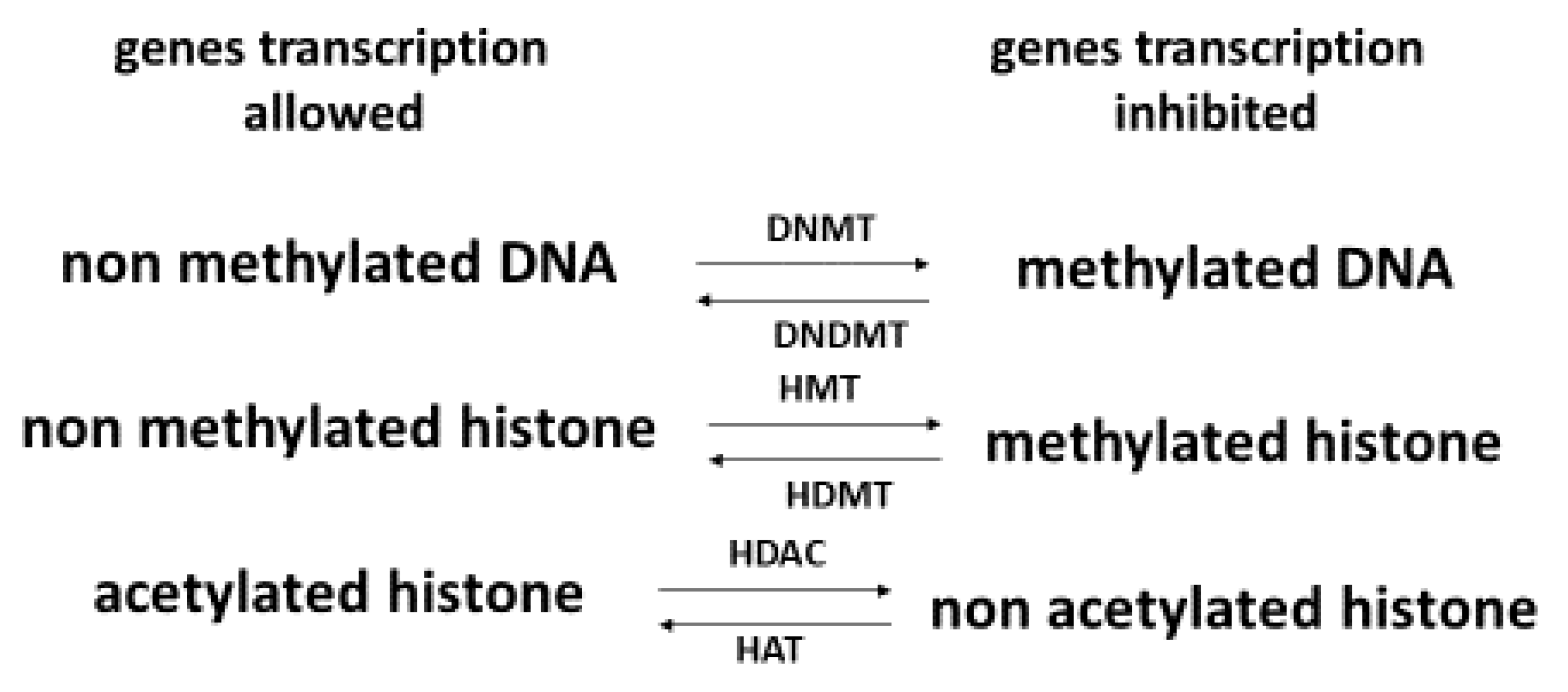
- Zhang, L.; Lu, Q.; Chang, C. Epigenetics in health and disease. Adv. Exp. Med. Biol. 2020, 1253, 3–55. [Google Scholar] [CrossRef] [PubMed]
- Perera, B.P.U.; Faulk, C.; Svoboda, L.K.; Goodrich, J.M.; Dolinoy, D.C. The role of environmental exposures and the epigenome in health and disease. Environ. Mol. Mutagen. 2020, 61, 176–192. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-T.; Oh, S.; Ro, D.H.; Yoo, H.; Kwon, Y.-W. The key role of DNA methylation and histone acetylation in epigenetics of atherosclerosis. J. Lipid Atheroscler. 2020, 9, 419–434. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.A.; Akman, K.; Calimport, S.R.G.; Wuttke, D.; Stolzing, A.; de Magalhaes, J.P. The Role of DNA Methylation in aging, rejuvenation, and age-related disease. Rejuvenation Res. 2012, 15, 483–494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Russell-Goldman, E.; Murphy, G.F. The Pathobiology of skin aging: New insights into an old dilemma. Am. J. Pathol. 2020, 190, 1356–1369. [Google Scholar] [CrossRef] [PubMed]
- Köhler, F.; Rodrıguez-Paredes, M. DNA Methylation in epidermal differentiation, aging, and cancer. J. Investig. Dermatol. 2020, 140, 38–47. [Google Scholar] [CrossRef] [Green Version]
- de Oliveira, N.F.P.; de Souza, B.F.; de Castro Coêlho, M. Radiation and its relation to DNA methylation in epidermal cells: A review. Epigenomes 2020, 4, 23. [Google Scholar] [CrossRef]
- Boroni, M.; Zonari, A.; de Oliveira, C.R.; Alkatib, K.; Cruz, E.A.O.; Brace, L.E.; de Carvalho, J.L. Highly accurate skin-specific methylome analysis algorithm as a platform to screen and validate therapeutics for healthy aging. Clin. Epigenetics 2020, 12, 1–16. [Google Scholar] [CrossRef]
- Orioli, D.; Dellambra, E. Epigenetic regulation of skin cells in natural aging and premature aging diseases. Cells 2018, 7, 268. [Google Scholar] [CrossRef] [Green Version]
- Thiele, J.J.; Schroeter, C.; Hsieh, S.N.; Podda, M.; Packer, L. The antioxidant network of the stratum corneum. In Oxidants and Antioxidants in Cutaneous Biology; Thiele, J., Elsner, P., Eds.; Karger: Basel, Switzerland, 2001; Volume 29, pp. 26–42. [Google Scholar]
- Slominski, A.; Tobin, D.J.; Zmijewski, M.A.; Wortsman, J.; Paus, R. Melatonin in the skin: Synthesis, metabolism and functions. Trends Endocrinol. Metab. 2007, 19, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Rusanova, Y.; Martínez-Ruiz, L.; Florido, J.; Rodríguez-Santana, C.; Guerra-Librero, A.; Acuña-Castroviejo, D.; Escames, G. Protective effects of melatonin on the skin: Future perspectives. Int. J. Mol. Sci. 2019, 20, 4948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bocheva, G.; Slominski, R.M.; Janjetovic, Z.; Kim, T.-K.; Böhm, M.; Steinbrink, K.; Reiter, R.J.; Kleszczynski, K.; Slominski, A.T. Protective role of melatonin and its metabolites in skin aging. Int. J. Mol. Sci. 2022, 23, 1238. [Google Scholar] [CrossRef]
- Ressmeyer, A.-R.; Mayo, J.C.; Zelosko, V.; Sáinz, R.M.; Tan, D.-X.; Poeggeler, B.; Antolín, I.; Zsizsiki, B.K.; Reiter, R.J.; Hardeland, R. Antioxidant properties of the melatonin metabolite N1-acetyl-5-methoxykynuramine (AMK): Scavenging of free radicals and prevention of protein destruction. Redox Rep. 2003, 8, 205–213. [Google Scholar] [CrossRef] [Green Version]
- Shindo, Y.; Witt, E.; Han, D.; Epstein, W.; Packer, L. Enzymic and non-enzymic antioxidants in epidermis and dermis. J. Investig. Dermatol. 1994, 102, 122–124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thiele, J.J.; Weber, S.U.; Packer, L. Sebaceous gland secretion is a major physiological route of vitamin E delivery to skin. J. Investig. Dermatol. 1999, 113, 1006–1010. [Google Scholar] [PubMed] [Green Version]
- Shapiro, S.S.; Saliou, C. Role of vitamins in skin care. Nutrition 2001, 1, 839–844. [Google Scholar] [CrossRef]
- Jahan, I.; Islam, M.R.; Islam, M.R.; Ali, R.; Rahman, S.M.; Nahar, Z.; Hasnat, A.; Islam, M.S. Altered serum elements, antioxidants, MDA, and immunoglobulins are associated with an increased risk of seborrheic dermatitis. Heliyon 2021, 7, e06621. [Google Scholar] [CrossRef]
- Boyera, N.; Galey, I.; Bernard, B.A. Effect of vitamin C and its derivatives on collagen synthesis and crosslinking by normal human fibroblasts. Int. J. Cosmet. Sci. 1998, 20, 151–158. [Google Scholar] [CrossRef]
- Dunaway, S.; Odin, R.; Zhou, L.; Ji, L.; Yuhang, Z.; Kadekaro, A.L. Natural antioxidants: Multiple 95. Mechanisms to protect skin from solar radiation. Front. Pharmacol. 2018, 9, 392. [Google Scholar] [CrossRef] [Green Version]
- Ganguly, B.; Hota, M.; Pradhan, J. Skin aging: Implications of UV radiation, reactive oxygen species and natural antioxidants. In Reactive Oxygen Species; Rizwan, A., Ed.; IntechOpen: London, UK, 2021; pp. 1–21. [Google Scholar]
- Primavesi, L.; Piantanida, M.; Pravettoni, V. Mediterranean diet and skin health. In Bioactive Dietary Factors and Plant Extracts in Dermatology; Chapter I; Watson, R.R., Zibadi, S., Eds.; Humana: Louisville, KY, USA, 2013; pp. 3–14. [Google Scholar]
- Williams, M.C.B.; Williams, G.M.; Pageon, H.; Fourtanier, A.; Green, A.C. Dietary antioxidant capacity and skin photo aging: A 15-year longitudinal study. J. Investig. Dermatol. 2021, 141, 1111–1118. [Google Scholar]
- Packer, L. Ultraviolet radiation (UVA, UVB) and skin antioxidants. In Free Radical Damage and Its Control; Chapter 9; Rice-Evans, C.A., Burdon, R.H., Eds.; Elsevier: Amsterdam, The Netherlands, 1994. [Google Scholar]
- Pandel, R.; Poljšak, B.; Godic, A.; Dahmane, R. Skin photoaging and the role of antioxidants in its prevention. ISRN Dermatol. 2013, 2013, 930164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.; Liu, Y.; Zhao, Z.; Qiu, J. Oxidative stress in the skin: Impact and related protection. Int. J. Cosmet. Sci. 2021, 43, 495–509. [Google Scholar] [CrossRef] [PubMed]
- Rhie, G.; Shin, M.H.; Seo, J.Y.; Choi, W.W.; Cho, K.H.; Kim, K.H.; Park, K.C.; Eun, H.C.; Chung, J.H. Aging-and photoaging-dependent changes of enzymic and nonenzymic antioxidants in the epidermis and dermis of human skin. J. Investig. Dermatol. 2001, 117, 1212–1217. [Google Scholar] [PubMed]
- White, W.S.; Kim, C.; Kalkwarf, H.J.; Bustos, P.; Roe, D.A. Ultraviolet light-induced reductions in plasma carotenoids levels. Am. J. Clin. Nutr. 1988, 47, 879–883. [Google Scholar] [CrossRef]
- Biesalski, H.K.; Hemmes, C.; Hopfenmuller, W.; Schmid, C.; Gollnick, H.P.M. Effects of controlled exposure of sunlight on plasma and skin levels of β-carotene. Free Radic. Res. 1996, 24, 215–224. [Google Scholar] [CrossRef]
- Keegan, G.T.; Keegan, L. Foods high in antioxidants, selenium, and vitamin E. Altern Med. Alert. 2005, 8, S1–S2. [Google Scholar]
- Hariharan, S.; Dharmaraj, S. Selenium and selenoproteins: Its role in regulation of infammation. Infammopharmacology 2020, 28, 667–695. [Google Scholar] [CrossRef]
- Tinggi, U. Selenium, its role as antioxidant in human health. Environ. Health Prev. Med. 2008, 13, 102–108. [Google Scholar] [CrossRef] [Green Version]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Cai, Z.; Zhang, J.; Li, H. Selenium, aging and aging-related diseases. Aging Clin. Exp. Res. 2019, 31, 1035–1047. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, P.B.; Fain, H.D.; Cassidy, J.P., Jr.; Tran, S.M.; Moos, P.J.; Boucher, K.M.; Gerads, R.; Florell, S.R.; Grossman, D.; Leachman, S.A. Selenium for the prevention of cutaneaous melanoma. Nutrients 2013, 5, 725–749. [Google Scholar] [CrossRef] [Green Version]
- Kieliszek, A.; Bano, I.; Zare, H. A comprehensive review on selenium and its effects on human health and distribution in middle eastern countries. Biol. Trace Elem. Res. 2022, 200, 971–987. [Google Scholar] [CrossRef] [PubMed]
- Barichelli, G.; Capperucci, A.; Tanini, D. The role of selenium in pathologies: An updated review. Antioxidants 2022, 11, 251. [Google Scholar] [CrossRef] [PubMed]
- Moghadaszadeh, B.; Beggs, A.H. Selenoproteins and their impact on human Health through diverse physiological pathways. Physiology 2006, 21, 307–315. [Google Scholar] [CrossRef]
- Steinbrenner, H.; Speckmann, B.; Klotz, L.-O. Selenoproteins: Antioxidant selenoenzymes and beyond. Arch. Biochem. Biophys. 2016, 595, 113–119. [Google Scholar] [CrossRef]
- Ye, R.; Huang, J.; Wang, Z.; Chen, Y.; Dong, Y. The Role and Mechanism of Essential Selenoproteins for Homeostasis. Antioxidants 2022, 11, 973. [Google Scholar] [CrossRef]
- Brown, K.M.; Arthur, J.R. Selenium, selenoproteins and human health: A review. Public Health Nutr. 2001, 4, 593–599. [Google Scholar] [CrossRef] [Green Version]
- Saito, Y. Selenoprotein P as an in vivo redox regulator: Disorders related to its deficiency and excess. J. Clin. Biochem. Nutr. 2020, 66, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Tinkov, A.A.; Bjørklund, G.; Skalny, A.V.; Holmgren, A.; Skalnaya, M.G.; Chirumbolo, C.A.; Aaseth, J. The role of the thioredoxin/thioredoxin reductase system in the metabolic syndrome: Towards a possible prognostic marker? Cell Mol. Life Sci. 2018, 75, 1567–1586. [Google Scholar] [CrossRef]
- Sarikaya, E.; Dogan, S. Glutathione peroxidase in health and diseases. In Glutathione System and Oxidative Stress in Health and Disease; Bagatini, M.D., Blumenberg, M., Eds.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef] [Green Version]
- Xia, Y.; Hill, K.E.; Byrne, D.W.; Xu, J.; Burk, R.F. Effectiveness of selenium supplements in a low-selenium area of China. Am. J. Clin. Nutr. 2005, 81, 829–834. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hurst, R.; Armah, C.N.; Dainty, J.R.; Hart, D.J.; Teucher, B.; Goldson, A.J.; Broadley, M.R.; Motley, A.K.; Fairweather-Tait, S.J. Establishing optimal selenium status: Results of a randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2010, 1, 923–931. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Müller, S.M.; Dawczynski, C.; Wiest, J.; Lorkowski, S.; Kipp, A.P.; Schwerdtle, T. Functional biomarkers for the selenium status in a human nutritional intervention study. Nutrients 2020, 12, 676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steinbrenner, H.; Duntas, L.H.; Rayman, M.P. The role of selenium in type-2 diabetes mellitus and its metabolic comorbidities. Redox Biol. 2022, 50, 102236. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.D. Selenium. In Encyclopedia of Human Nutrition, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 186–192. [Google Scholar]
- McKenzie, R.C. Selenium, ultraviolet radiation and the skin. Exp. Dermatol. 2000, 25, 631–636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jobeili, L.; Rousselle, P.; Béal, D.; Blouin, E.; Roussel, A.-M.; Damour, O.; Rachidi, W. Selenium preserves keratinocyte stemness and delays senescence by maintaining epidermal adhesion. Aging 2017, 9, 2302–2315. [Google Scholar] [CrossRef] [Green Version]
- Rafferty, T.D.; McKenzie, R.C.; Hunter, J.A.; Howie, A.F.; Arthur, J.R.; Nicol, F.; Beckett, G.J. Differential expression of selenoproteins by human skin cells and protection by selenium from UVB-radiation-induced cell death. Biochem. J. 1998, 332, 231–236. [Google Scholar] [CrossRef] [Green Version]
- Favrot, C.; Beal, D.; Blouin, E.; Leccia, M.Y.; Roussel, A.-M.; Rachidi, W. Age-dependent protective effect of selenium against UVA irradiation in primary human keratinocytes and the associated DNA repair signature. Oxidative Med. Cell. Longev. 2018, 2018, 5895439. [Google Scholar] [CrossRef]
- Burke, K.E.; Burford, R.G.; Combs, G.F., Jr.; French, I.W.; Skeffington, D.R. The effect of topical L-selenomethionine on minimal erythema dose of ultraviolet irradiation in humans. Photodermatol. Photoimmunol. Photomed. 1992, 9, 52–57. [Google Scholar]
- Alehagen, U.; Opstad, T.N.; Alexander, J.; Larsson, A.; Aaseth, J. Impact of selenium on biomarkers and clinical aspects related to aging. A Review. Biomolecules 2021, 11, 1478. [Google Scholar] [CrossRef]
- Shu, Y.; Wu, M.; Yang, S.; Wang, H.; Li, H. Association of dietary selenium intake with telomere length in middle-aged and older adults. Clin. Nutr. 2020, 39, 3086–3091. [Google Scholar] [CrossRef] [PubMed]
- Tarwadi, K.V.; Agte, V.V. Effect of micronutrients on methylglyoxal-mediated in vitro glycation of albumin. Biol. Trace. Elem. Res. 2011, 143, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Vinson, J.A.; Howard, T.B., III. Inhibition of protein glycation and advanced glycation end products by ascorbic acid and other vitamins and nutrients. Nut. Biochem. 1996, 7, 659–663. [Google Scholar] [CrossRef]
- Du, P.-C.; Tu, Z.-C.; Wang, H. Mechanism of selenium nanoparticles inhibiting advanced glycation end products. J. Agric. Food Chem. 2020, 68, 10586–10595. [Google Scholar] [CrossRef]
- Speckmann, B.; Tilman, G. Epigenetic effects of selenium and their implications for health. Epigenetics 2015, 10, 179–190. [Google Scholar] [CrossRef]
- Pilsner, J.R.; Hall, M.N.; Liu, X.; Ahsan, H.; Ilievski, V.; Slavkovich, V.; Levy, D.; Factor-Litvak, P.; Graziano, J.H.; Gamble, M.V. Associations of plasma selenium with arsenic and genomic methylation of leukocyte DNA in Bangladesh. Environ. Health Perspect. 2011, 119, 113–118. [Google Scholar] [CrossRef]
- Bradshaw, P.C. Acetyl-CoA Metabolism and histone acetylation in the regulation of aging and lifespan. Antioxidants 2021, 10, 572. [Google Scholar] [CrossRef]
- Lee, J.-I.; Nian, H.; Cooper, A.J.L.; Sinha, R.; Dai, J.; Bisson, W.J.; Dashwood, R.H.; Pinto, J.T. α-keto acid metabolites of naturally-occuring organoselenium compounds as inhibitors of histone deacetylase in human prostate cancer cells. Cancer Prev. Res. 2009, 2, 683–693. [Google Scholar] [CrossRef] [Green Version]
- Jing, H.; Lin, H. Sirtuins and epigenetic regulation. Chem. Rev. 2015, 115, 2350–2375. [Google Scholar] [CrossRef] [Green Version]
- Houtkooper, R.H.; Pirinen, E.; Auwerx, J. Sirtuins as regulators of metabolism and health span. Nat. Rev. Mol. Cell Biol. 2012, 13, 225–238. [Google Scholar] [CrossRef] [Green Version]
- Zhao, L.; Cao, J.; Hu, K.; He, X.; Yun, D.; Tong, T.; Han, L. Sirtuins and their biological relevance in aging and age-related diseases. Aging Dis. 2020, 4, 927–945. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Peterson, L.M.; Willking-Busch, M.J.; Ndiaye, M.A.; Philippe, G.A.P.; Setaluri, V.; Ahmad, N. Sirtuins in skin and skin cancers. Ski. Pharmacol. Physiol. 2017, 30, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Grabowska, W.; Sikora, E.; Bielak-Zmijewska, A. Sirtuins, a promosing target in slowing down the aging process. Biogerontology 2017, 18, 447–476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bielach-Bazyluk, A.; Zbroch, E.; Mysliwiec, H.; Rydzewska-Rosolowska, A.; Kakareko, K.; Flisiak, I.; Hryszko, T. Sirtuin 1 and skin: Implications in intrinsic and extrinsic aging-a systematic review. Cells 2021, 10, 813. [Google Scholar] [CrossRef]
- Giacconi, R.; Chiodi, L.; Boccoli, G.; Costarelli, L.; Piacenza, F.; Provinciali, M.; Malavolta, M. Reduced levels of plasma selenium are associated with increased inflammation and cardiovascular disease in an Italian elderly population. Exp. Gerontol. 2021, 145, 111219. [Google Scholar] [CrossRef]
- Reilly, R. (Ed.) Selenium in diets. In Selenium in Food and Heath; Chapter 10; Springer: Berlin/Heidelberg, Germany, 2006; pp. 173–198. [Google Scholar]
- Agostoni, C.; Canani, R.B.; Fairweather-Tait, S.; Heinonen, M.; Korhonen, H.; La Vieille, S.; Marchelli, R.; Martin, A.; Naska, A.; Neuhäuser-Berthold, M.; et al. Scientific Opinion on Dietary Reference Values for selenium. EFSA J. 2014, 12, 3846. [Google Scholar]
- Burk, R.F.; Norsworthy, B.K.; Hill, K.E.; Motley, A.K.; Byrneshowed, D.W. Effects of chemical form of selenium on plasma biomarkers in a high-dose human supplementation trial. Cancer Epidemiol. Biomark. Prev. 2006, 15, 804–810. [Google Scholar] [CrossRef] [Green Version]
- Lv, J.; Aib, P.; Leia, S.; Zhoua, F.; Chena, S.; Zhanga, Y. Selenium levels and skin diseases: Systematic review and meta-analysis. J. Trace Elem. Med. Biol. 2020, 62, 126548. [Google Scholar] [CrossRef]
- Juhlin, L.; Edqvist, L.E.; Ekman, L.G.; Ljunghall, K.; Olsson, M. Blood glutathione-peroxidase levels in skin diseases: Effect of selenium and vitamin E treatment. Acta Derm. Venereol. 1982, 62, 211–214. [Google Scholar]
- Vaughn, A.R.; Foolad, N.; Maarouf, M.; Tran, K.A.; Shi, V.Y. Micronutrients in atopic dermatis: A systematic review. J. Altern. Complement. Med. 2019, 25, 567–577. [Google Scholar] [CrossRef]
- Yamada, T.; Saunders, T.; Kuroda, S.; Sera, K.; Nakamura, T.; Takatsuji, T. Cohort study for prevention of atopic dermatitis using hair mineral contents. J. Trace Elem. Med. Biol. 2013, 27, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Michaëlsson, G.; Edqvist, L.E. Erythrocyte glutathione peroxidase activity in acne vulgaris and the effect of selenium and vitamin E treatment. Acta Derm. Venereol. 1984, 649, 9–14. [Google Scholar] [CrossRef]
- Ljunghall, K.; Juhlin, L.; Edqvist, L.E.; Plantin, L.O. Selenium, glutathione-peroxidase and dermatitis herpetiformis. Acta Derm. Venereol. 1984, 64, 546–547. [Google Scholar]
- Fairris, G.M.; Lloydt, B.; Hinst, L.; Perkins, P.J. The effect of supplementation with selenium and vitamin E in psoriasis. Ann. Clin. Biochem. 1989, 26, 83–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Food Safety Authority. Tolerable Upper Intake Levels for Vitamins and Minerals; February Report; European Food Safety Authority: Parma, Italy, 2006. [Google Scholar]
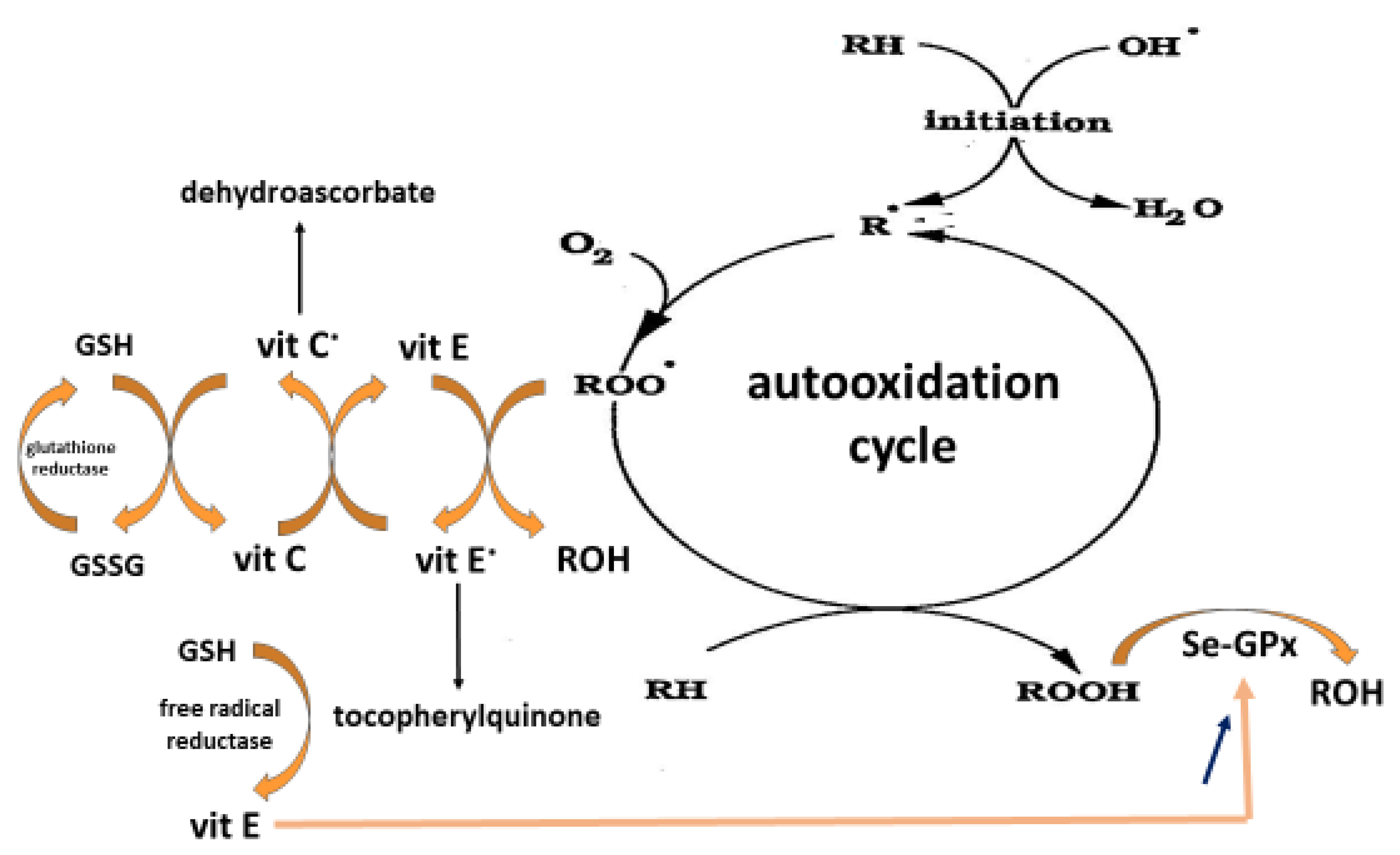
- Bowry, V.W.; Ingold, K.U.; Stocker, R. Vitamin E in human low-density lipoprotein. When and how this antioxidant becomes a pro-oxidant. Biochem J. 1992, 288, 341–344. [Google Scholar] [CrossRef] [Green Version]
- Niki, E.; Tsuchiya, J.; Tanimura, R.; Kamiya, Y. Regeneration of vitamin E from alpha-chromanoxyl radical by glutathione and vitamin C. Chem. Lett. 1982, 6, 789–792. [Google Scholar] [CrossRef]
- Buettner, G.R. The pecking order of free radicals and antioxidants: Lipid peroxidation, alpha-tocopherol and ascorbate. Arch. Biochem. Biophys. 1993, 300, 535–543. [Google Scholar] [CrossRef]
- Yang, N.Y.; MacDonald, I.B.; Indrajit, A.N.D.; Desai, D. Vitamin E supplementation and glutathione peroxidase activity. Proc. Soc. Exp. Biol. Med. 1976, 151, 770–774. [Google Scholar] [CrossRef]
- Li, R.K.; Cowan, D.B.; Mickle, D.A.; Weisel, R.D.; Burton, G.W. Effect of vitamin E on human glutathione peroxidase (GSH-PX1) expression in cardiomyocytes. Free Radic. Biol. Med. 1996, 21, 419–426. [Google Scholar] [CrossRef]
- Van Haaften, R.I.M.; Haenen, G.R.M.M.; Evelo, C.T.A.; Bast, A. Effect of vitamin E on glutathione-dependent enzymes. Drug Metab. Rev. 2003, 35, 215–253. [Google Scholar] [CrossRef]
- Park, K. Role of micronutrients in skin health and function. Biomol. Ther. 2015, 23, 207–217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Machlin, L.J.; Filipski, R.; Nelson, J.; Horn, L.R.; Brin, M. Effects of a prolonged vitamin E deficiency in the rat. J. Nutr. 1977, 107, 1200–1208. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Yang, G.; Luo, M.; Lan, Q.; Shi, X.; Deng, H.; Wang, N.; Xu, X.; Zhang, C. Serum vitamin E levels and chronic inflammatory skin diseases: A systematic review and meta-analysis. PLoS ONE 2021, 16, e0261259. [Google Scholar] [CrossRef] [PubMed]
- Passi, S.; Morrone, A.; De Luca, C.; Picardo, M.; Ippolito, F. Blood levels of vitamin E, polyunsaturated fatty acids of phospholipids, lipoperoxides and glutathione peroxidase in patients affected with seborrheic dermatitis. J. Dermatol. Sci. 1991, 2, 171–178. [Google Scholar] [CrossRef]
- Packer, L.; Valacchi, G. Antioxidants and the response of skin to oxidative stress: Vitamin E as a key indicator. J. Nutr. 2002, 15, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Fryer, M.J. Evidence for the photoprotective effects of vitamin E. Photochem. Ad Photobiol. 1993, 2, 304–312. [Google Scholar] [CrossRef]
- Nachbar, F.; Korting, H. The role of vitamin E in normal and damaged skin. J. Mol. Med. 1995, 73, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Thiele, J.J.; Traber, M.G.; Packer, L. Depletion of human stratum corneum vitamin E: An early and sensitive in vivo maker of UV induced photo-oxidation. J. Investig. Dermatol. 1998, 110, 756–761. [Google Scholar] [CrossRef]
- Jurkiewicz, B.A.; Bissett, D.L.; Buettner, G.R. Effect of topically applied tocopherol on ultraviolet radiation-mediated free radical damage in skin. J. Investig. Dermatol. 1995, 104, 484–488. [Google Scholar] [CrossRef] [Green Version]
- Lopez-Torres, M.; Thiele, J.; Shindo, Y.; Han, D.; Packer, L. Topical application of α–tocopherol modules the antioxidant network and diminishes ultraviolet oxidative damage in murine skin. Br. J. Dermatol. 1998, 138, 207–215. [Google Scholar] [CrossRef]
- Pelle, E.; Miranda, E.P.; Fthenakis, C.; Mammone, T.; Marenus, K.; Maes, D. Cigarette smoke-induced lipid peroxidation in human skin and its inhibition by topically applied antioxidants. Ski. Pharmacol. App. Ski. Physiol. 2002, 15, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Thiele, J.J.; Hsieh, S.N.; Ekanayake-Mudiyanselage, S. Vitamin E: Critical review of its current use in cosmetic and clinical dermatology. Dermatol. Surg. 2005, 31, 805–813. [Google Scholar] [CrossRef]
- Wu, S.; Gao, J.; Dinh, Q.T.; Chen, C.; Fimmel, S. IL-8 production and AP-1 transactivation induced by UVA in human keratinocytes.: Roles of alpha-tocopherol. Mol. Immunol. 2008, 45, 2288–2296. [Google Scholar] [CrossRef] [PubMed]
- Michels, A.J. Vitamin E and Skin Health. Available online: https://lpi.oregonstate.edu/mic/vitamins/vitamin-E2012 (accessed on 6 June 2022).
- Hantke, B.; Lahmann, C.; Venzke, K.; Fisher, T.; Kocourek, A.; Windsor, L.J.; Bergemann, J.; Stäb, F.; Tschesche, T. Influence of flavonoids and vitamins on the MMP-and TIMP-expression of human dermal fibroblasts after UVA irradiation. Photochem. Photobiol. Sci. 2002, 1, 826–833. [Google Scholar] [CrossRef]
- Zappe, K.; Pointner, A.; Switzeny, O.J.; Magnet, U.; Tomeva, E.; Heller, J.; Mare, G.; Wagner, K.-H.; Knasmueller, S.; Haslberger, A.G. Counteraction of oxidative stress by vitamin E affects epigenetic regulation by increasing global methylation and gene expression of MLH1 and DNMT1 Dose Dependently in Caco-2 Cells. Oxidative Med. Cell. Longev. 2018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makpol, M.; Durani, L.W.; Chua, K.H.; Anum Mohd Yusof, Y.; Wan Ngah, W.Z. Tocotrienol-Rich fraction prevents cell cycle arrest and elongates telomere length in senescent human diploid fibroblasts. J. Biomed. Biotechnol. 2011, 2011, 506171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Oliveira Pinto, C.A.S.; Martins, T.E.A.; Martinez, R.M.; Freire, T.B.; Robles Velasco, M.V.; Baby, A.R. Vitamin E in human skin: Functionality and topical products. In Vitamin E in Health and Disease; Pınar, E., Júlia, S.S., Eds.; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef]
- Delinasios, G.J.; Karbaschi, M.; Cooke, M.S.; Young, A.R. Vitamin E inhibits the UVAI induction of “light” and dark” cyclobutane pyrimidine dimers, and oxidatively generated DNA damage, in keratinocytes. Sci. Rep. 2018, 8, 423. [Google Scholar] [CrossRef] [PubMed]
- La Ruche, G.; Cesarini, J.P. Protective effect of oral selenium plus copper associated with vitamin complex on sunburn cell formation in human skin. Photodermatol. Photoimmunol. Photomed. 1991, 8, 232–235. [Google Scholar]
- Fuchs, J.; Kern, H. Modulation of UV-light-induced skin inflammation by D-alpha-tocopherol and L-ascorbic acid: A clinical study using solar simulated radiation. Free Radic. Biol. Med. 1998, 25, 1006–1012. [Google Scholar] [CrossRef]
- Mireles-Rocha, H.; Galindo, I.; Huerta, M.; Trujillo-Hernandez, B.; Elizalde, T.; Cortes-Franco, R. UVB phototoprotection with antioxidants: Effects of oral therapy with d-α-tocopherol an ascorbic acid on the minimal erythrema dose. Acta Derm. Venereol. 2002, 82, 21–24. [Google Scholar] [CrossRef]
- McArdle, F.; Rhodes, L.E.; Parslew, R.A.G.; Close, G.L.; Jack, C.I.A.; Friedmann, P.S.; Jackson, M.J. Effects of oral vitamin E and 𝛽-carotene supplementation on ultraviolet radiation-induced oxidative stress in human skin. Am. J. Clin. Nutr. 2004, 80, 1270–1275. [Google Scholar] [CrossRef] [Green Version]
- Werninghaus, L.; Meydani, M.; Bhawan, J.; Randall Margolis, R.; Blumberg, J.B.; Gilchres, B.A. Evaluation of the photoprotective effect of oral vitamin E supplementation. Arch. Dermatol. 1994, 130, 1257–1261. [Google Scholar] [CrossRef] [PubMed]
- Meydani, S.N.; Meydani, M.; Blumberg, J.B.; Leka, L.S.; Siber, G.; Loszewski, R.; Thompson, C.; Pedrosa, M.C.; Diamond, R.D.; Stollar, B.D. Vitamin E supplementation and in vivo immune response in healthy elderly subjects. A randomized controlled trial. JAMA 1997, 277, 1380–1386. [Google Scholar] [CrossRef] [PubMed]
- Pallast, E.G.; Schouten, E.G.; de Waart, F.G.; Fonk, H.C.; Doekes, G.; von Blomberg, B.M.; Kok, F.J. Effect of 50- and 100-mg vitamin E supplements on cellular immune function in noninstitutionalized elderly persons. Am. J. Clin. Nutr. 1999, 69, 273–281. [Google Scholar] [CrossRef] [Green Version]
- Tsoureli-Nikita, E.; Hercogova, J.; Lott, T.; Menchini, G. Evaluation of dietary intake of vitamin E in the treatment of atopic dermatitis: A study of the clinical course and evaluation of the immunoglobulin E serum levels. Int. J. Dermatol. 2002, 41, 146–150. [Google Scholar] [CrossRef] [PubMed]
- Elgoweini, M.; Nour El Din, N. Response of vitiligo to narrowband ultraviolet B and oral antioxidants. J. Clin. Pharmacol. 2009, 49, 852–855. [Google Scholar] [CrossRef] [PubMed]
- Kharaeva, Z.; Gostova, E.; De Luca, C.; Raskovic, D.; Korkina, L. Clinical and biochemical effects of coenzyme Q(10), vitamin E, and selenium supplementation to psoriasis patients. Nutrition 2009, 25, 295–302. [Google Scholar] [CrossRef]
- Oh, S.; Chung, J.; Kim, M.-K.; Kwon, S.O.; Cho, B.-H. Antioxidant nutrient intakes and corresponding biomarkers associated with the risk of atopic dermatitis in young children. Eur. J. Clin. Nutr. 2010, 64, 245–252. [Google Scholar] [CrossRef]
- Javanbakht, M.H.; Keshavarz, S.A.; Djalali, M.; Siassi, F.; Eshraghian, M.R.; Firooz, A.; Seirafi, H.; Ehsani, A.H.; Chamari, M.; Mirshafiey, A. Randomized controlled trial using vitamins E and D supplementation in atopic dermatitis. J. Dermatol. Treat. 2011, 22, 144–150. [Google Scholar] [CrossRef]
- Goforoushan, F.; Azimi, H.; Goldus, M. Efficacy of vitamin E to prevent dermal complications of isotretinoin. Pak. J. Biol. Sci. 2013, 16, 548–550. [Google Scholar] [CrossRef] [Green Version]
- Jaffary, F.; Faghihi, G.; Mokhtarian, A.; Hosseini, S.M. Effects of oral vitamin E on treatment of atopic dermatitis: A randomized controlled trial. J. Res. Med. Sci. 2015, 20, 1053–1057. [Google Scholar] [PubMed]
- Berardesca, E.; Cameli, N. Vitamin E supplementation in inflammatory skin diseases. Dermatol. Ther. 2021, 34, e15160. [Google Scholar] [CrossRef] [PubMed]
- Eberlein-Köning, B.; Placzek, B.; Przybilla, B. Protective effect against sunburn of combined systemic ascorbic acid (vitamin C) and d-α-tocopherol (vitamin E). J. Am. Acad. Dermatol. 1998, 38, 45–48. [Google Scholar] [CrossRef]
- Eberlein-König, B.; Ring, J. Relevance of vitamins C and E in cutaneous photoprotection. J. Cosmet. Dermatol. 2005, 4, 4–9. [Google Scholar] [CrossRef]
- Greul, A.K.; Grundmann, J.U.; Heinrich, F.; Pfitzner, I.; Bernhardt, J.; Ambach, A.; Biesalski, H.-K.; Gollnick, H. Photoprotection of UV-irradiated human skin: An antioxidative combination of vitamins E and C, carotenoids, selenium and proanthocyanidins. Ski. Pharmacol. Appl. Ski. Physiol. 2002, 15, 307–315. [Google Scholar] [CrossRef]
- Césarini, J.P.; Michel, L.; Maurette, J.M.; Adhoute, H.; Béjot, M. Immediate effects of UV radiation on the skin: Modification by an antioxidant complex containing carotenoids. Photodermatol. Photoimmunol. Photomed. 2003, 19, 182–189. [Google Scholar] [CrossRef] [Green Version]
- Placzek, M.; Gaube, S.; Kerkmann, U.; Gilbertz, K.P.; Herzinger, T.; Haen, E.; Przybilla, B. Ultraviolet B-induced DNA damage in human epidermis is modified by the antioxidants ascorbic acid and D-alpha-tocopherol. J. Investig. Dermatol. 2005, 124, 304–307. [Google Scholar] [CrossRef] [Green Version]
- Heinrich, U.; Tronnier, H.; Stahl, W.; Béjot, M. Maurette, J.-M. Antioxidant supplements improve parameters related to skin structure in humans. Ski. Pharmacol. Physiol. 2006, 19, 224–231. [Google Scholar] [CrossRef]
- Udompataikul, M.; Sripiroj, P.; Palungwachira, P. An oral nutraceutical containing antioxidants, minerals and glycosaminoglycans improves skinroughness and fine wrinkles. Int. J. Cosmet. Sci. 2009, 31, 427–435. [Google Scholar] [CrossRef]
- Granger, C.; Aladren, S.; Delgado, J.; Garre, A.; Trullas, C.; Gilaberte, Y. Prospective evaluation of the efficacy of a food supplement in increasing photoprotection and improving selective markers related to skin photo-aging. Dermatol. Ther. 2020, 10, 163–178. [Google Scholar] [CrossRef] [Green Version]
- Burke, K.E. Photoprotection of the skin with vitamins C and E: Antioxidants and synergies. In Nutrition and Skin; Pappas, A., Ed.; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar] [CrossRef]
- Pincemail, J.; Le Goff, C.; Charlier, C.; Gillion, P.; Cheramy-Bien, J.-P.; Van Honacker, E.; Chapelle, J.-P.; Defraigne, J.-O. Evaluation biologique du stress oxydant. Application en routine clinique. Nutr. Endocrinol. 2009, 16–30. [Google Scholar]
- Roberts, L.J., 2nd; Oates, J.A.; Linton, M.R.F.; Fazio, S.; Meador, B.P.; Gross, M.D.; Shyr, Y.; Morrow, J.D. The relationship between dose of vitamin E and suppression of oxidative stress in humans. Free Radic. Biol. Med. 2007, 43, 1388–1393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ozuguz, P.; Kacar, S.D.; Ekiz, O.; Takci, Z.; Balta, I.; Kalkan, G. Evaluation of serum vitamins A and E and zinc levels according to the severity of acne vulgaris. Cutan. Ocul. Toxicol. 2014, 33, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Trüeb, R.M. Aging of hair. J. Cosmet. Dermatol. 2005, 4, 60–72. [Google Scholar] [CrossRef]
- Trüeb, R.M. Pharmacologic interventions in aging hair. Clin. Interv. Aging 2006, 1, 121–129. [Google Scholar] [CrossRef]
- Trüeb, R.M. Oxidative stress in ageing of hair. Int. J. Trichology 2009, 1, 6–14. [Google Scholar] [CrossRef] [Green Version]
- Ho, C.H.; Sood, T.; Zito, P.M. Androgenetic Alopecia; StatPearls Publishing: Tampa, FL, USA, 2022. [Google Scholar]
- Upton, J.H.; Hannen, R.F.; Bahta, A.W.; Farjo, N.; Farjo, B.; Philpott, M.P. Oxidative stress associated senescence in dermal papilla cells of men with androgenetic alopecia. J. Investig. Dermatol. 2015, 135, 1244–1252. [Google Scholar] [CrossRef] [Green Version]
- Prie, B.E.; Iosif, L.; Tivig, I.; Stoian, I.; Giurcaneanu, C. Oxidative stress in androgenetic alopecia. J. Med. Life 2016, 9, 79–83. [Google Scholar]
- Prie, B.E.; Voiculescu, V.M.; Ionescu-Bozdog, O.B.; Petrutescu, B.; Iosif, L.; Gaman, L.E.; Clatici, V.G.; Stoian, I.; Giurcaneanu, C. Oxidative stress and alopecia areata. J. Med. Life 2015, 8, 43–46. [Google Scholar]
- Acharya, P.; Mathur, M.C. Oxidative stress in alopecia areata: A systematic review and meta-analysis. Int. J. Dermatol. 2020, 59, 434–440. [Google Scholar] [CrossRef]
- Almohanna, H.M.; Ahmed, A.A.; Tsatalis, J.P.; Tosti, A. The role of vitamins and minerals in hair loss: A review. Dermatol. Ther. 2019, 9, 51–70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramadan, R.; Tawdy, A.; Hay, R.A.; Rashed, L.; Tawfik, D. The antioxidant role of paraoxonase 1 and vitamin E in three autoimmune diseases. Ski. Pharmacol. Physiol. 2013, 26, 2–7. [Google Scholar] [CrossRef]
- Naziroglu, M.; Kokcam, I. Antioxidants and lipid peroxidation status in the blood of patients with alopecia. Cell Biochem. Funct. 2000, 18, 169–173. [Google Scholar] [CrossRef]
- Thompson, J.M.; Mirza, M.A.; Park, M.K.; Qureshi, A.A.; Cho, E. The role of micronutrients in alopecia areata: A review. Am. J. Clin. Dermatol. 2017, 18, 663–679. [Google Scholar] [CrossRef] [PubMed]
- El-Tahlavi, S.M.; El-Ramly, A.Z.; El-Nabarawy, E.; El-Ghaffar, N. Serum selenium levels in alopecia areata. J. Egypt Women’s Dermatol. Soc. 2012, 9, 174–177. [Google Scholar] [CrossRef]
- Nguyen, B.; Tosti, A. Alopecia in patients with COVID-19: A systematic review and meta-analysis. JAAD Int. 2022, 7, 67–77. [Google Scholar] [CrossRef]
- Sharquie, K.E.; Jabbar, R.I. COVID-19 infection is a major cause of acute telogen effluvium. Ir. J. Med. Sci. 2021, 191, 1677–1681. [Google Scholar] [CrossRef]
- Czech, T.; Sugihara, S.; Nishimura, Y. Characteristics of hair loss after COVID-19: A systematic scoping review. J. Cosmet. Dermatol. 2022, 21, 3655–3662. [Google Scholar] [CrossRef]
- Pincemail, J.; Cavalier, E.; Charlier, C.; Cheramy–Bien, J.-P.; Brevers, E.; Courtois, A.; Fadeur, M.; Meziane, S.; Le Goff, C.; Misset, B.; et al. Oxidative stress status in COVID-19 patients hospitalized in intensive care unit for severe pneumonia. A pilot sudy. Antioxidants 2021, 10, 257. [Google Scholar] [CrossRef]
- Pincemail, J.; Rousseau, A.-F.; Kaux, J.-F.; Cheramy-Bien, J.-P.; Bruyère, C.; Prick, J.; Stern, D.; Kaci, M.M.; Maertens de Noordhout, B.; Albert, A.; et al. Oxidative stress status and its correlation with redox status as measured by an electrochemical (PAOT®) methodology: A Pilot Study in Critical COVID-19 Pneumonia Survivors. Free Radic. Biol. Med. 2021, 177, S120. [Google Scholar] [CrossRef]
- Schomburg, L. Selenium deficiency in COVID-19-a possible long-lasting toxic relationship. Nutrients 2022, 14, 283. [Google Scholar] [CrossRef] [PubMed]
- Du Laing, G.; Petrovic, M.; Lachat, C.; De Boevre, M.; Klingenberg, G.J.; Sun, Q.; De Saeger, S.; De Clercq, J.; Ide, L.; Vanderkerckhove, L. Course and survival of COVID-19 patients with comorbidities in relation to the trace element status at hospital admission. Nutrients 2021, 13, 3304. [Google Scholar] [CrossRef]
- Guo, E.L.; Katta, R. Diet and hair loss: Effects of nutrient deficiency and supplement use. Dermatol. Pract. Concept. 2017, 7, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Majeed, M.; Nagabhushanam, K.; Mundkur, L. Can selenium reduce the susceptibility and severity of SARS-CoV-2? A comprehensive review. Int. J. Mol. Sci. 2022, 23, 4809. [Google Scholar] [CrossRef] [PubMed]
- Siegl-Cachedenier, E.; Flores, I.; Klatt, P.; Blasco, M.A. Telomerase reverses epidermal hair follicle stem cell defects and loss of long-term survival associated with critically short telomeres. J. Cell Biol. 2007, 22, 277–290. [Google Scholar] [CrossRef] [Green Version]
- Stone, R.C.; Aviv, A.; Paus, R. Telomere dynamics and telomerase in the biology of hair follicles and their stem cells as a model for aging research. J. Investig. Dermatol. 2021, 141, 1031–1040. [Google Scholar] [CrossRef]
- Patrnogic, A. Is Vitamin E Oil Good for Hair? Available online: https://wowskinscience.com (accessed on 12 September 2022).
- Beoy, L.A.; Woei, W.J.; Hay, Y.K. Effects of tocotrienol supplementation on hair growth in human volunteers. Trop. Life Sci. Res. 2010, 21, 91–99. [Google Scholar]
- Rushton, D.H. Nutritional factors and hair loss. Clin. Dermatol. 2002, 27, 396–404. [Google Scholar] [CrossRef] [Green Version]
- Bertolino, F.A.; Stege, P.W.; Salinas, E.; Messina, G.A.; Rabhave, J. Electrochemical study of the antioxidant activity and the synergic eEffect of selenium with natural and synthetic antioxidants. Anal. Lett. 2010, 43, 2078–2090. [Google Scholar] [CrossRef]
- Ebhohimen, I.E.; Okanlawon, T.S.; Osagie, A.O.; Izevbigie, O.N. Vitamin E in human health and oxidative stress related Diseases. In Vitamin E in Health and Disease–Interactions, Diseases and Health Aspects; Pınar, E., Júlia, S.S., Eds.; IntechOpen: London, UK, 2021. [Google Scholar] [CrossRef]
- Brigelius-Flohé, R. Selenium in human health and disease: An Overview. In Molecular and Integrative Toxicology; Bernhard, M., Ed.; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar] [CrossRef]
- Gruber, F.; Marchetti-Deschmann, M.; Kremslehner, C.; Schosserer, M. The skin epilipidome in stress, aging and inflammation. Front. Endocrinol. 2021, 11, 607076. [Google Scholar] [CrossRef] [PubMed]
- Hogan, J.S.; Weiss, W.P.; Smith, K.L. Role of vitamin E and selenium in host defense against mastitis. J. Dairy Sci. 1993, 76, 2795–2803. [Google Scholar] [CrossRef]
- Ghassemi, F.; Yuosefinasab, A. The protective effect of vitamin E and selenium on liver tissue in fructose- induced diabetic rat. Pars. J. Med. Sci. 2014, 12, 47–55. [Google Scholar] [CrossRef] [Green Version]
- Celebi, S. Effect of dietary Vitamin E, selenium and their combination on concentration of selenium, MDA, and antioxidant enzyme activities in some tissues of laying hens. Pak. J. Zool. 2019, 51, 1155–1161. [Google Scholar] [CrossRef]
- Dawood, M.A.O.; Zommora, M.; Eweedah, N.M.; Helal, A.I. Synergistic effects of selenium nanoparticles and vitamin E on growth, immune-gene expression and regulation of antioxidant status of Nile Tilapia (Oreochromis nilocitus). Biol. Trace Elem. Res. 2020, 195, 624–635. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Milhail, S.S.; Ding, Y.W.; Yang, G.-Y.; Bondoc, F.; Yang, C.S. Effects of vitamin E and selenium supplementation on esophageal adenocarcinogenesis in surgical model with rats. Carcinogenesis 2000, 21, 1531–1536. [Google Scholar] [CrossRef] [PubMed]
- Sokmen, B.B.; Basaraner, H.; Yanardag, R. Combined effects of treatment with vitamin C, vitamin E and selenium on the skin of diabetic rats. Hum. Exp. Toxicol. 2013, 32, 379–384. [Google Scholar] [CrossRef]
- Lippman, S.M.; Klein, E.A.; Goodman, P.J.; Lucia, M.S.; Thompson, I.M.; Ford, L.G.; Parnes, H.L.; Minasian, L.M.; Gaziano, J.M.; Hartline, J.A.; et al. Effect of selenium and vitamin E on risk of prostate cancer and cancers: The Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 2009, 301, 39–51. [Google Scholar] [CrossRef] [Green Version]
- Fam, V.W.; Charoenwoodhipong, P.; Sivamani, R.K.; Holt, R.R.; Keen, C.L. Plant-based foods for skin heath: A narrative review. J. Acad. Nutr. Diet. 2022, 122, 614–629. [Google Scholar] [CrossRef]
- World Health Organization. Global Action Plan for the Prevention and Control of Noncommunicable Diseases 2013–2020; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Daily consumption of fruit and vegetables by sex, age and educational attainment level. Eurostat report. 2002. Available online: http://tc.ontologycentral.com/page/hlth_ehis_fv3e (accessed on 10 October 2022).
- Candido, T.M.; Ariede, M.B.; Lima, F.V.; de Souza Guedes, L.; Robles Velasco, M.V.; Baby, A.R.; Rosado, C. Dietary supplements and the skin: Focus on photoprotection and antioxidant activity-a review. Nutrients 2022, 14, 1248. [Google Scholar] [CrossRef]
- Gonzalez, S.; Gilaberte, Y.; Philips, N.; Juarranz, A. Current trends in photoprotection-a new generation of oral photoprotectors. Open Dermatol. J. 2011, 5, 6–14. [Google Scholar] [CrossRef] [Green Version]
- Woodby, B.; Penta, K.; Pecorelli, A.; Lila, M.-A.; Valacchi, G. Skin health from inside out. Ann. Rev. Food Sci. Technol. 2020, 11, 235–254. [Google Scholar] [CrossRef] [Green Version]
- Santos, J.S.; Tavares, G.D.; Barradas, T.N. Vitamin E and derivatives in skin health promotion. In Vitamin E in Health and Disease; Pınar, E., Júlia, S.S., Eds.; IntechOpen: London, UK, 2021. [Google Scholar] [CrossRef]
- Miller, E.R., III; Pastor-Barriuso, R.; Dalal, D.; Riemersma, R.A.; Appel, L.J.; Guallar, E. Meta-Analysis: High-Dosage vitamin E supplementation may increase all-cause mortality. Ann. Intern. Med. 2005, 142, 37–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rayman, M.P.; Winther, K.H.; Pastor-Barriuso, R.; Cold, F.; Thvilum, M.; Stranges, S.; Guallar, E.; Cold, S. Effect of long-term selenium supplementation on mortality: Results from a multiple-dose, randomised controlled trial. Free. Radic. Biol. Med. 2018, 127, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Dumoulin, M.; Gaudout, D.; Lemaire, B. Clinical effects of an oral supplementation rich in antioxidants on skin radiance in women. Clin. Coesmetic Investig. Dermatol. 2016, 9, 315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petruk, G.; Del Giuduce, R.; Rigano, M.M.; Monti, D.M. Antioxidants from plants protect again skin photoaging. Oxidative Med. Cell. Longev. 2018, 2018, 1454936. [Google Scholar] [CrossRef] [Green Version]
- Luo, J.; Si, H.; Jia, Z.; Liu, D. Dietary anti-aging polyphenols and potential mechanisms. Antioxidants 2021, 10, 283. [Google Scholar] [CrossRef]
- Martel, J.; Ojcius, D.M.; Ko, Y.-F.; Ke, P.-Y.; Wu, C.-Y.; Peng, H.-H.; Young, J.D. Effects of phytochemicals on health and longevity. Trends Endocrinol. Metab. 2019, 30, 335–346. [Google Scholar] [CrossRef]
- Stefanson, A.L.; Bakovic, M. Dietary Regulation of Keap1/Nrf2/ARE Pathway: Focus on plant-derived compounds and trace minerals. Nutrients 2014, 6, 3777–3801. [Google Scholar] [CrossRef]
- Nobile, V.; Schiano, I.; Peral, A.; Giardina, S.; Sparta, E.; Caturla, N. Antioxidant and reduced skin aging effects of a polyphenol enriched dietary supplement in response to air pollution: A randomized, double-blind, placebo-controlled study. Food Nutr. Res. 2021. [Google Scholar] [CrossRef]
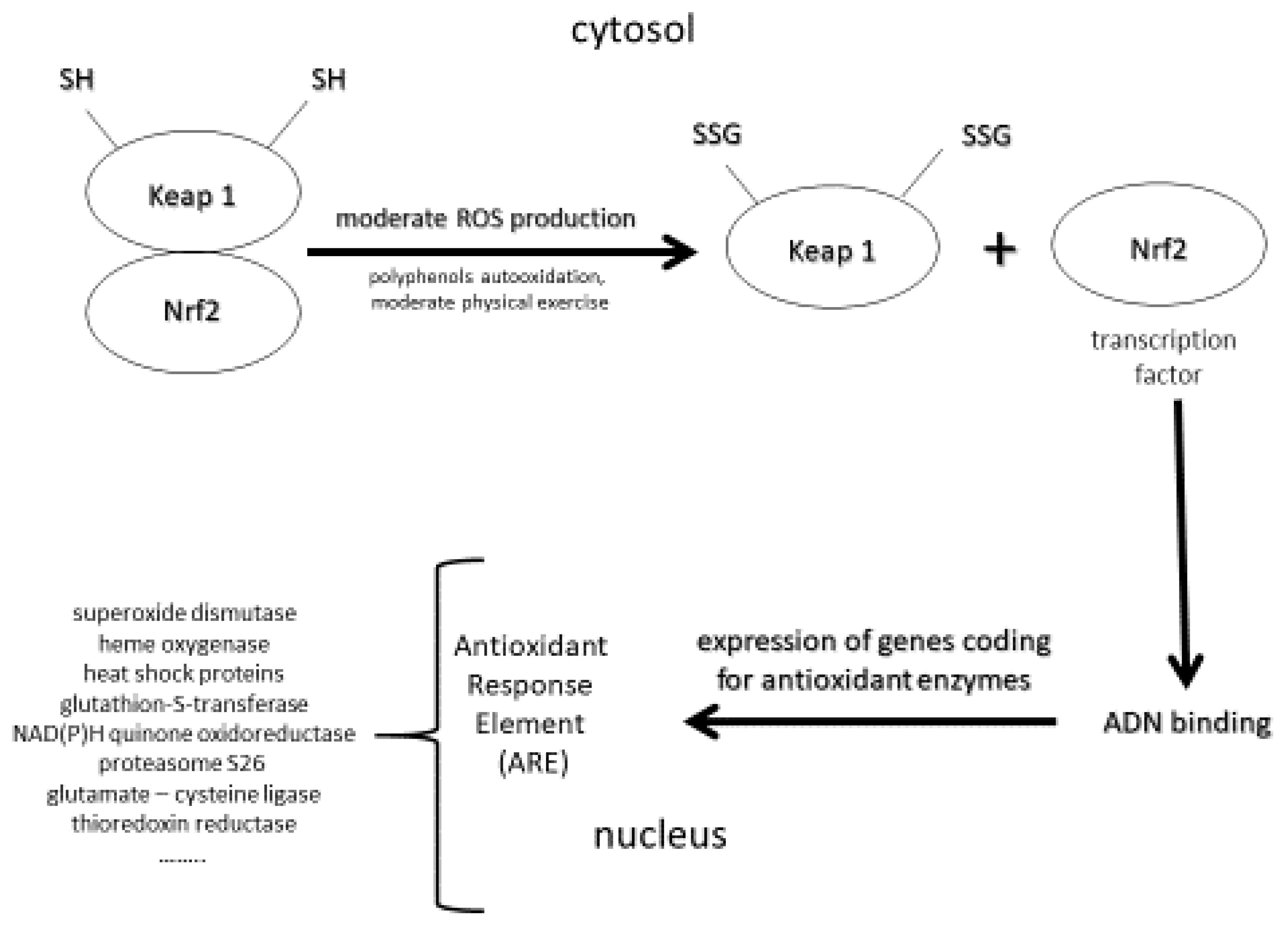
- Boo, Y.C. Natural NRF2 modulators for skin protection. Antioxidants 2020, 9, 812. [Google Scholar] [CrossRef] [PubMed]
- Greenwald, M.B.-Y.; Ben-Sasson, S.; Bianco-Peled, H.; Kohen, R. Skin redox balance maintenance: The need for an NrF2-activator delivery system. Cometics 2016, 3, 1. [Google Scholar] [CrossRef] [Green Version]
- Ogawa, T.; Ishitsuka, Y. The role of KEAP1-NRF2 system in atopic dermatitis and psoriasis. Antioxidants 2022, 11, 1397. [Google Scholar] [CrossRef] [PubMed]
- Bocheva, G.; Slominski, R.M.; Slominski, A.T. The impact of vitamin D on skin aging. Int. J. Mol. Sci. 2021, 22, 9097. [Google Scholar] [CrossRef] [PubMed]
- Tagliaferri, S.; Porri, D.; De Giuseppe, R.; Manuelli, M.; Alessio, F.; Cena, H. The controversial role of vitamin D as an antioxidant: Results from randomised controlled trials. Nutr. Res. Rev. 2019, 32, 99–105. [Google Scholar] [CrossRef]
- Kechichian, E.; Ezzedine, K. Vitamin D and the skin: An update for dermatologists. Am. J. Clin. Dermatol. 2018, 19, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Triviñoa, F.J.; Arias-Santiago, S.; Gilaberte-Calzadac, Y. Vitamin D and the Skin: A Review for Dermatologists. Actas Dermo-Sifiliográficas (Engl. Ed.) 2019, 110, 262–272. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pincemail, J.; Meziane, S. On the Potential Role of the Antioxidant Couple Vitamin E/Selenium Taken by the Oral Route in Skin and Hair Health. Antioxidants 2022, 11, 2270. https://doi.org/10.3390/antiox11112270
Pincemail J, Meziane S. On the Potential Role of the Antioxidant Couple Vitamin E/Selenium Taken by the Oral Route in Skin and Hair Health. Antioxidants. 2022; 11(11):2270. https://doi.org/10.3390/antiox11112270
Chicago/Turabian StylePincemail, Joël, and Smail Meziane. 2022. "On the Potential Role of the Antioxidant Couple Vitamin E/Selenium Taken by the Oral Route in Skin and Hair Health" Antioxidants 11, no. 11: 2270. https://doi.org/10.3390/antiox11112270
APA StylePincemail, J., & Meziane, S. (2022). On the Potential Role of the Antioxidant Couple Vitamin E/Selenium Taken by the Oral Route in Skin and Hair Health. Antioxidants, 11(11), 2270. https://doi.org/10.3390/antiox11112270





