Microcurrent and Gold Nanoparticles Combined with Hyaluronic Acid Accelerates Wound Healing
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Excisional Wound Model
2.3. Treatment
2.4. Euthanasia
2.5. Synthesis and Characterization of the GNPs
2.6. Wound Size Analysis
2.7. Histomorphometry
2.8. Determination of Cytokine Content Using ELISA
2.9. Biochemical Analysis
2.10. Statistical Analysis
3. Results
3.1. Physicochemical Properties of the GNP Solution
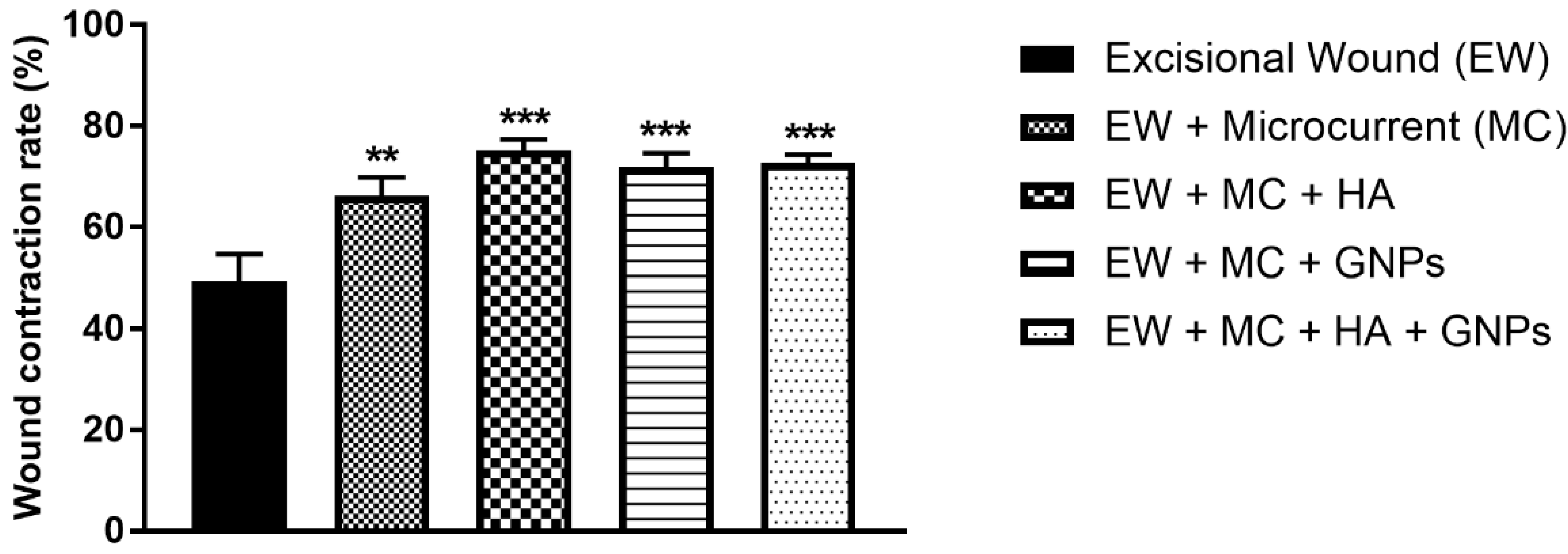
3.2. Analysis of the Wound Contraction Rate
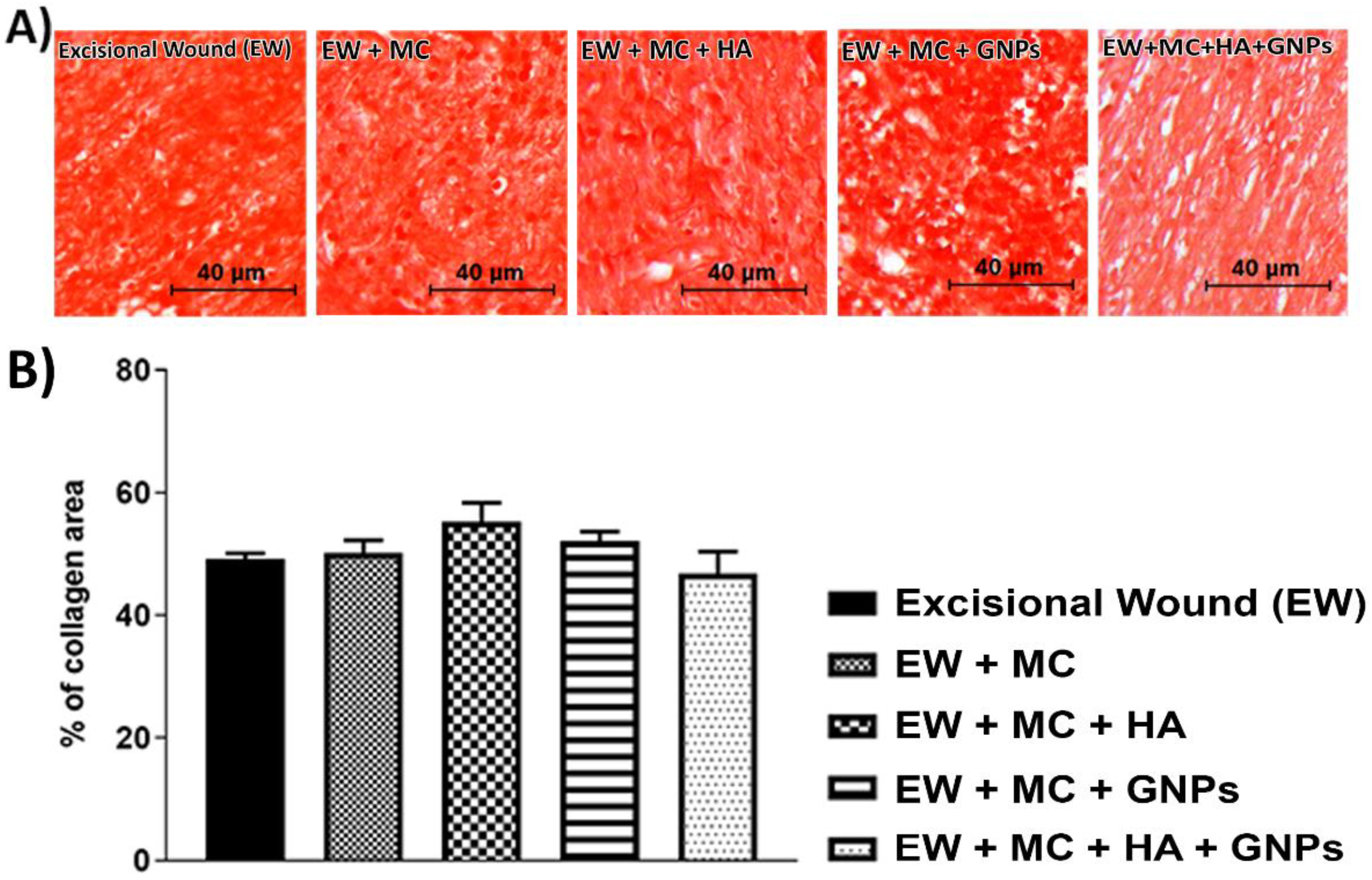
3.3. Histological Analysis
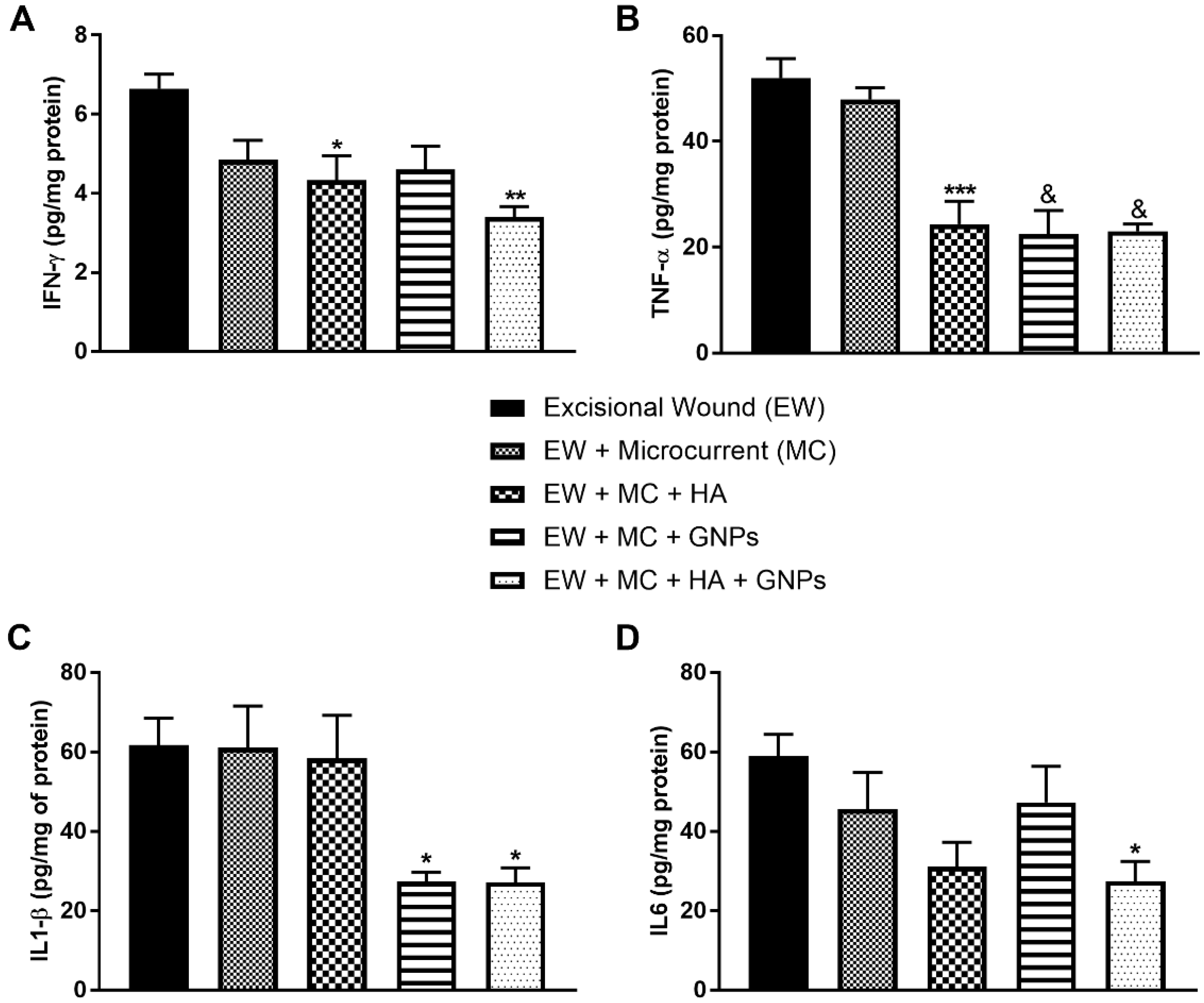
3.4. Evaluation of Pro-Inflammatory Cytokines
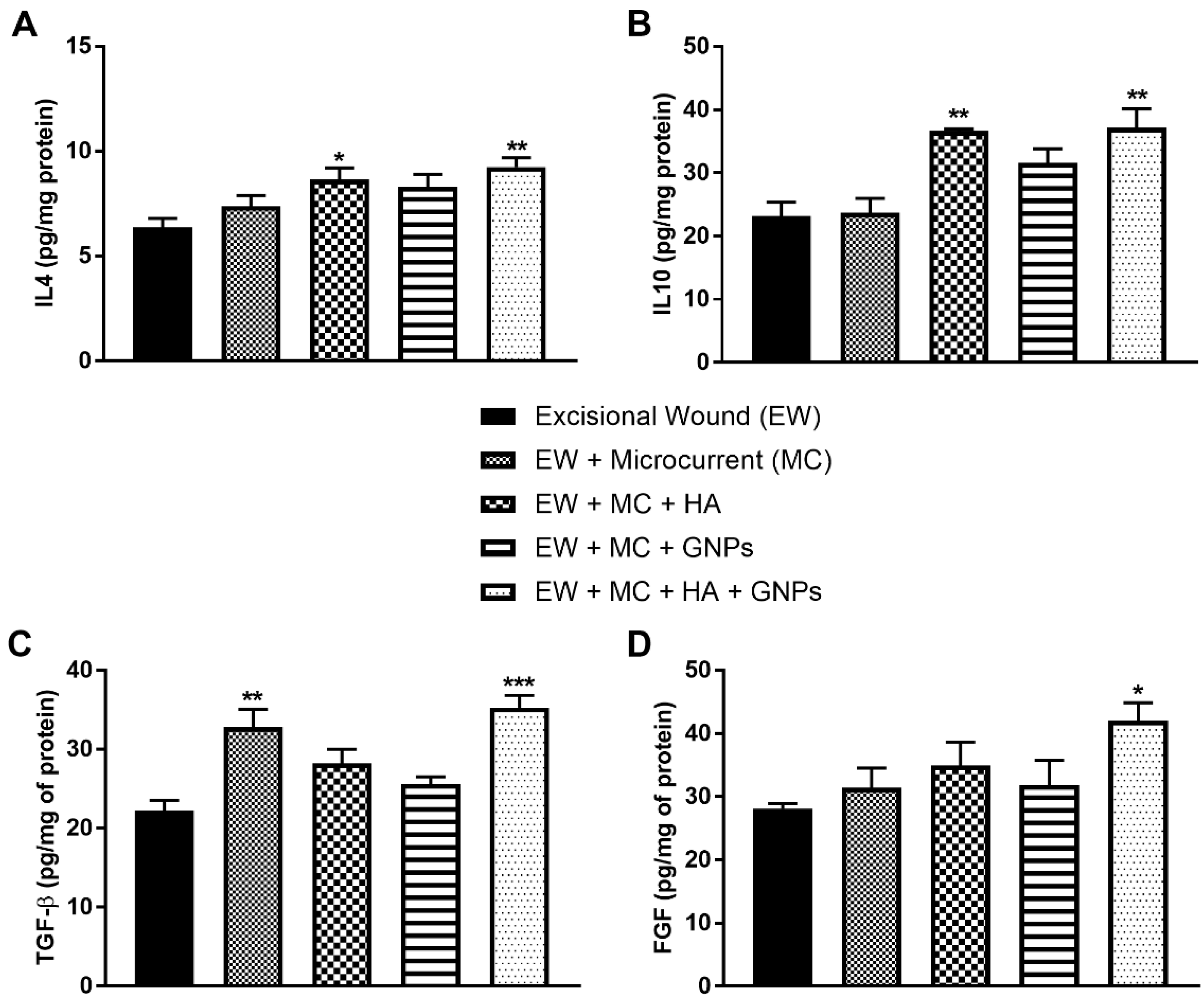
3.5. Evaluation of Anti-Inflammatory Cytokines and Growth Factors
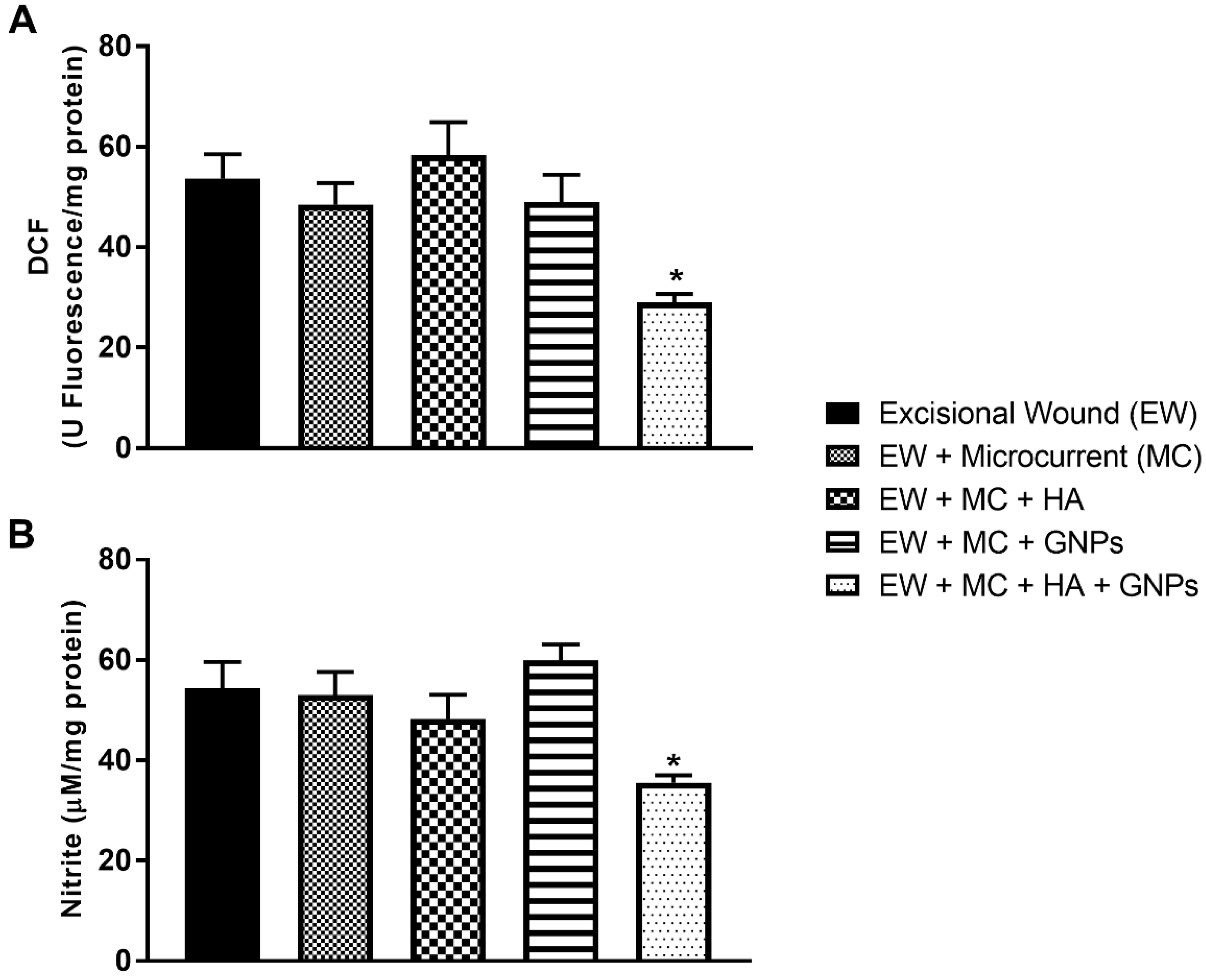
3.6. Intracellular Determination of Oxidants
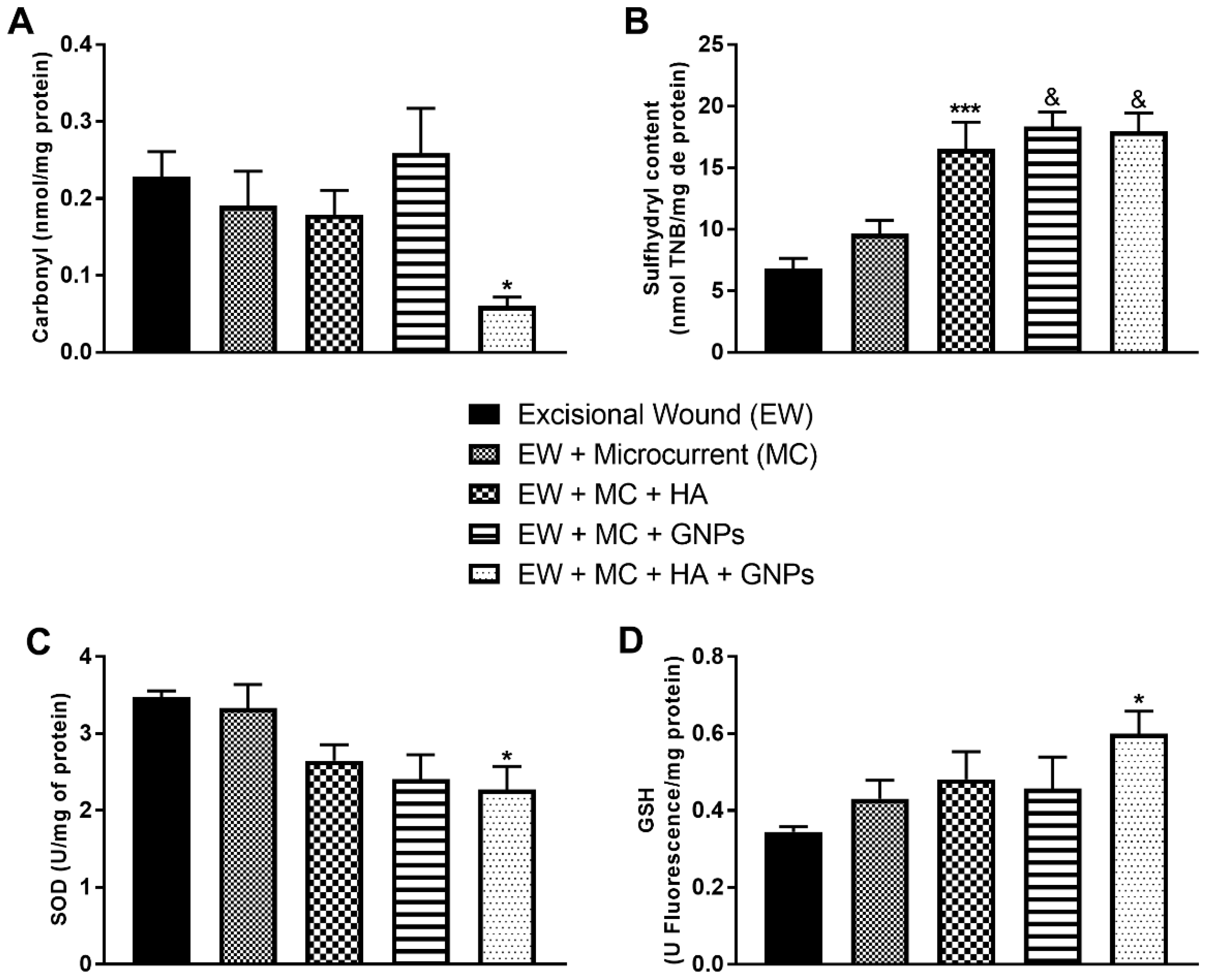
3.7. Markers of Oxidative Damage and Antioxidants
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Martinengo, L.; Olsson, M.; Bajpai, R.; Soljak, M.; Upton, Z.; Schmidtchen, A.; Car, J.; Järbrink, K. Prevalence of chronic wounds in the general population: Systematic review and meta-analysis of observational studies. Ann. Epidemiol. 2019, 29, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Nussbaum, S.R.; Carter, M.J.; Fife, C.E.; DaVanzo, J.; Haught, R.; Nusgart, M.; Cartwright, D. An Economic Evaluation of the Impact, Cost, and Medicare Policy Implications of Chronic Nonhealing Wounds. Value Health J. Int. Soc. Pharm. Outcomes Res. 2018, 21, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Holzer-Geissler, J.C.J.; Schwingenschuh, S.; Zacharias, M.; Einsiedler, J.; Kainz, S.; Reisenegger, P.; Holecek, C.; Hofmann, E.; Wolff-Winiski, B.; Fahrngruber, H.; et al. The Impact of Prolonged Inflammation on Wound Healing. Biomedicines 2022, 10, 856. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, S.B.; Challen, K.; Wright, K.L.; Hardy, J.G. Electrical Stimulation to Enhance Wound Healing. J. Funct. Biomater. 2021, 12, 40. [Google Scholar] [CrossRef]
- Sahana, T.G.; Rekha, P.D. Biopolymers: Applications in wound healing and skin tissue engineering. Mol. Biol. Rep. 2018, 45, 2857–2867. [Google Scholar] [CrossRef]
- Veith, A.P.; Henderson, K.; Spencer, A.; Sligar, A.D.; Baker, A.B. Therapeutic strategies for enhancing angiogenesis in wound healing. Adv. Drug Deliv. Rev. 2019, 146, 97–125. [Google Scholar] [CrossRef]
- Zaccaron, R.P.; Barbieri, R.T.; Mendes, C.; Venturini, L.M.; Alves, N.; Mariano, S.S.; de Andrade, T.A.M.; Hermes de Araújo, P.H.; Feuser, P.E.; Thirupathi, A.; et al. Photobiomodulation associated with lipid nanoparticles and hyaluronic acid accelerate the healing of excisional wounds. J. Biomater. Appl. 2022, 37, 8853282221109344. [Google Scholar] [CrossRef]
- Foulds, I.S.; Barker, A.T. Human skin battery potentials and their possible role in wound healing. Br. J. Dermatol. 1983, 109, 515–522. [Google Scholar] [CrossRef]
- Nair, H.K.R. Microcurrent as an adjunct therapy to accelerate chronic wound healing and reduce patient pain. J. Wound Care 2018, 27, 296–306. [Google Scholar] [CrossRef]
- Poltawski, L.; Watson, T. Bioelectricity and microcurrent therapy for tissue healing—A narrative review. Phys. Ther. Rev. 2009, 14, 104–114. [Google Scholar] [CrossRef]
- Vieira, A.C.; Reid, B.; Cao, L.; Mannis, M.J.; Schwab, I.R.; Zhao, M. Ionic components of electric current at rat corneal wounds. PLoS ONE 2011, 6, e17411. [Google Scholar] [CrossRef] [PubMed]
- Jennings, J.; Chen, D.; Feldman, D. Transcriptional response of dermal fibroblasts in direct current electric fields. Bioelectromagnetics 2008, 29, 394–405. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, O.M.; Mertz, P.M.; Smerbeck, R.V.; Eaglstein, W.H. The healing of superficial skin wounds is stimulated by external electrical current. J. Investig. Dermatol. 1983, 81, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Bai, H.; Wang, E.; Forrester, J.V.; McCaig, C.D. Electrical stimulation directly induces pre-angiogenic responses in vascular endothelial cells by signaling through VEGF receptors. J. Cell Sci. 2004, 117, 397–405. [Google Scholar] [CrossRef]
- Park, R.J.; Son, H.; Kim, K.; Kim, S.; Oh, T. The Effect of Microcurrent Electrical Stimulation on the Foot Blood Circulation and Pain of Diabetic Neuropathy. J. Phys. Ther. Sci. 2011, 23, 515–518. [Google Scholar] [CrossRef][Green Version]
- Clarke Moloney, M.; Lyons, G.M.; Breen, P.; Burke, P.E.; Grace, P.A. Haemodynamic study examining the response of venous blood flow to electrical stimulation of the gastrocnemius muscle in patients with chronic venous disease. Eur. J. Vasc. Endovasc. Surg. Off. J. Eur. Soc. Vasc. Surg. 2006, 31, 300–305. [Google Scholar] [CrossRef]
- Sharifi, S.; Hajipour, M.J.; Gould, L.; Mahmoudi, M. Nanomedicine in Healing Chronic Wounds: Opportunities and Challenges. Mol. Pharm. 2021, 18, 550–575. [Google Scholar] [CrossRef]
- Mahmoudi, A.; Kesharwani, P.; Majeed, M.; Teng, Y.; Sahebkar, A. Recent advances in nanogold as a promising nanocarrier for curcumin delivery. Colloids Surfaces. B Biointerfaces 2022, 215, 112481. [Google Scholar] [CrossRef]
- da Rocha, F.R.; Haupenthal, D.P.d.S.; Zaccaron, R.P.; Corrêa, M.E.A.B.; Tramontin, N.d.S.; Fonseca, J.P.; Nesi, R.T.; Muller, A.P.; Pinho, R.A.; Paula, M.M.d.S.; et al. Therapeutic effects of iontophoresis with gold nanoparticles in the repair of traumatic muscle injury. J. Drug Target. 2020, 28, 307–319. [Google Scholar] [CrossRef]
- Akturk, O.; Kismet, K.; Yasti, A.C.; Kuru, S.; Duymus, M.E.; Kaya, F.; Caydere, M.; Hucumenoglu, S.; Keskin, D. Collagen/gold nanoparticle nanocomposites: A potential skin wound healing biomaterial. J. Biomater. Appl. 2016, 31, 283–301. [Google Scholar] [CrossRef]
- Mendes, C.; dos Santos Haupenthal, D.P.; Zaccaron, R.P.; de Bem Silveira, G.; Corrêa, M.E.A.B.; de Roch Casagrande, L.; de Sousa Mariano, S.; de Souza Silva, J.I.; de Andrade, T.A.M.; Feuser, P.E.; et al. Effects of the association between photobiomodulation and hyaluronic acid linked gold nanoparticles in wound healing. ACS Biomater. Sci. Eng. 2020, 6, 5132–5144. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Lee, Y.-S.; Choe, J.; Lee, H.; Kim, Y.-M.; Jeoung, D. CD44-epidermal growth factor receptor interaction mediates hyaluronic acid-promoted cell motility by activating protein kinase C signaling involving Akt, Rac1, Phox, reactive oxygen species, focal adhesion kinase, and MMP-2. J. Biol. Chem. 2008, 283, 22513–22528. [Google Scholar] [CrossRef]
- Xu, Z.; Liu, G.; Liu, P.; Hu, Y.; Chen, Y.; Fang, Y.; Sun, G.; Huang, H.; Wu, J. Hyaluronic acid-based glucose-responsive antioxidant hydrogel platform for enhanced diabetic wound repair. Acta Biomater. 2022, 147, 147–157. [Google Scholar] [CrossRef]
- Corrêa, M.; Mendes, C.; Bittencourt, J.V.S.; Takejima, A.; de Souza, I.C.; de Carvalho, S.C.D.; Orlandini, I.G.; de Andrade, T.A.M.; Guarita-Souza, L.C.; Silveira, P.C.L. Effects of the Application of Decellularized Amniotic Membrane Solubilized with Hyaluronic Acid on Wound Healing. Ann. Biomed. Eng. 2022. [Google Scholar] [CrossRef]
- Wu, S.; Zhao, W.; Sun, M.; He, P.; Lv, H.; Wang, Q.; Ma, J. Novel bi-layered dressing patches constructed with radially-oriented nanofibrous pattern and herbal compound-loaded hydrogel for accelerated diabetic wound healing. Applied Materials Today. 2022, 28, 101542. [Google Scholar] [CrossRef]
- Wu, S.; Dong, T.; Li, Y.; Sun, M.; Qi, Y.; Liu, J.; Duan, B. State-of-the-art review of advanced electrospun nanofiber yarn-based textiles for biomedical applications. Appl. Mater. Today 2022, 27, 101473. [Google Scholar] [CrossRef]
- Yu, R.; Zhang, H.; Guo, B. Conductive biomaterials as bioactive wound dressing for wound healing and skin tissue engineering. Nano-Micro Lett. 2022, 14, 1–46. [Google Scholar] [CrossRef]
- Li, T.; Sun, M.; Wu, S. State-of-the-art review of electrospun gelatin-based nanofiber dressings for wound healing applications. Nanomaterials 2022, 12, 784. [Google Scholar] [CrossRef] [PubMed]
- Della Vechia, I.C.; Steiner, B.T.; Freitas, M.L.; Fidelis, G.d.S.P.; Galvani, N.C.; Ronchi, J.M.; Possato, J.C.; Fagundes, M.Í.; Rigo, F.K.; Feuser, P.E.; et al. Comparative cytotoxic effect of citrate-capped gold nanoparticles with different sizes on noncancerous and cancerous cell lines. J. Nanoparticle Res. 2020, 22, 133. [Google Scholar] [CrossRef]
- Andrade, T.A.M.; Masson-Meyers, D.S.; Caetano, G.F.; Terra, V.A.; Ovidio, P.P.; Jordão-Júnior, A.A.; Frade, M.A.C. Skin changes in streptozotocin-induced diabetic rats. Biochem. Biophys. Res. Commun. 2017, 490, 1154–1161. [Google Scholar] [CrossRef]
- Jiang, X.; Ge, H.; Zhou, C.; Chai, X.; Deng, H. The role of transforming growth factor β1 in fractional laser resurfacing with a carbon dioxide laser. Lasers Med. Sci. 2014, 29, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Vidal, B.C.; Mello, M.L.S. Supramolecular order following binding of the dichroic birefringent sulfonic dye Ponceau SS to collagen fibers. Biopolymers 2005, 78, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Curtolo, G.; de Paula Araújo, J.; Lima, J.A.; Brandt, J.V.; Bittencourt, J.V.S.; Venturini, L.M.; Silveira, P.C.L.; Rogers, S.; Franzini, C.M.; de Goes, V.F. Silver nanoparticles formulations for healing traumatic injuries in oral mucosa of rats. Arch. Oral Biol. 2021, 129, 105202. [Google Scholar] [CrossRef] [PubMed]
- Corrêa, M.E.A.B.; dos Santos Haupenthal, D.P.; Mendes, C.; Zaccaron, R.P.; de Roch Casagrande, L.; Venturini, L.M.; Porto, G.D.; Bittencourt, J.V.S.; de Souza Silva, J.I.; de Sousa Mariano, S.; et al. Effects of Percutaneous Collagen Induction Therapy Associated with Hyaluronic Acid on Inflammatory Response, Oxidative Stress, and Collagen Production. Inflammation 2020, 43, 2232–2244. [Google Scholar] [CrossRef] [PubMed]
- Bannister, J.V.; Calabrese, L. Assays for superoxide dismutase. Methods Biochem. Anal. 1987, 32, 279–312. [Google Scholar]
- Dos Santos Haupenthal, D.P.; Mendes, C.; de Bem Silveira, G.; Zaccaron, R.P.; Correa, M.; Nesi, R.T.; Pinho, R.A.; da Silva Paula, M.M.; Silveira, P.C.L. Effects of treatment with gold nanoparticles in a model of acute pulmonary inflammation induced by lipopolysaccharide. J. Biomed. Mater. Research. Part A 2020, 108, 103–115. [Google Scholar] [CrossRef]
- Franek, A.; Kostur, R.; Polak, A.; Taradaj, J.; Szlachta, Z.; Blaszczak, E.; Dolibog, P.; Dolibog, P.; Koczy, B.; Kucio, C. Using high-voltage electrical stimulation in the treatment of recalcitrant pressure ulcers: Results of a randomized, controlled clinical study. Ostomy Wound Manag. 2012, 58, 30–44. [Google Scholar]
- Polak, A.; Franek, A.; Taradaj, J. High-Voltage Pulsed Current Electrical Stimulation in Wound Treatment. Adv. Wound Care 2014, 3, 104–117. [Google Scholar] [CrossRef]
- Ahmed, A.F.; Elgayed, S.S.A.; Ibrahim, I.M. Polarity effect of microcurrent electrical stimulation on tendon healing: Biomechanical and histopathological studies. J. Adv. Res. 2012, 3, 109–117. [Google Scholar] [CrossRef]
- Kaur, U.; Banerjee, P.; Bir, A.; Sinha, M.; Biswas, A.; Chakrabarti, S. Reactive oxygen species, redox signaling and neuroinflammation in Alzheimer’s disease: The NF-kappaB connection. Curr. Top. Med. Chem. 2015, 15, 446–457. [Google Scholar] [CrossRef]
- Yu, C.; Hu, Z.-Q.; Peng, R.-Y.J.M.M.R. Effects and mechanisms of a microcurrent dressing on skin wound healing: A review. Mil. Med. Res. 2014, 1, 24. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, M.; Maeshige, N.; Honda, H.; Yoshikawa, Y.; Uemura, M.; Yamamoto, M.; Terashi, H. Optimum microcurrent stimulation intensity for galvanotaxis in human fibroblasts. J. Wound Care 2012, 21, 5–6. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.Y.; Al-Waili, N.; Stubbs, D.; Wendell, K.; Butler, G.; Al-Waili, T.; Al-Waili, A. Ultra-low microcurrent in the management of diabetes mellitus, hypertension and chronic wounds: Report of twelve cases and discussion of mechanism of action. Int. J. Med. Sci. 2009, 7, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zeng, L.; Song, W.; Liu, J. Influencing factors and drug application of iontophoresis in transdermal drug delivery: An overview of recent progress. Drug Deliv. Transl. Res. 2021, 12, 15–26. [Google Scholar] [CrossRef]
- Petrilli, R.; Lopez, R.F.V. Physical methods for topical skin drug delivery: Concepts and applications. Braz. J. Pharm. Sci. 2018, 54, e01008. [Google Scholar] [CrossRef]
- Manabe, E.; Numajiri, S.; Sugibayashi, K.; Morimoto, Y. Analysis of skin permeation-enhancing mechanism of iontophoresis using hydrodynamic pore theory. J. Control. Release 2000, 66, 149–158. [Google Scholar] [CrossRef]
- Arunkumar, S.; Ashok, P.; Desai, B.; Shivakumar, H.N. Technology. Effect of chemical penetration enhancer on transdermal iontophoretic delivery of diclofenac sodium under constant voltage. J. Drug Deliv. Sci. Technol. 2015, 30, 171–179. [Google Scholar] [CrossRef]
- Kim, J.W.; Shim, J.S.; Maeng, C.; Kim, Y.S.; Ahn, J.; Kwak, M.G.; Hong, S.J.; Cho, H.M. Fabrication of SiC nanoparticles by physical milling for ink-jet printing. J. Nanosci. Nanotechnol. 2013, 13, 5586–5589. [Google Scholar] [CrossRef]
- Dohnert, M.B.; Venancio, M.; Possato, J.C.; Zeferino, R.C.; Dohnert, L.H.; Zugno, A.I.; De Souza, C.T.; Paula, M.M.; Luciano, T.F. Gold nanoparticles and diclofenac diethylammonium administered by iontophoresis reduce inflammatory cytokines expression in Achilles tendinitis. Int. J. Nanomed. 2012, 7, 1651–1657. [Google Scholar] [CrossRef]
- Jeon, K.I.; Byun, M.S.; Jue, D.M. Gold compound auranofin inhibits IkappaB kinase (IKK) by modifying Cys-179 of IKKbeta subunit. Exp. Mol. Med. 2003, 35, 61–66. [Google Scholar] [CrossRef]
- Sumbayev, V.V.; Yasinska, I.M.; Garcia, C.P.; Gilliland, D.; Lall, G.S.; Gibbs, B.F.; Bonsall, D.R.; Varani, L.; Rossi, F.; Calzolai, L. Gold nanoparticles downregulate interleukin-1beta-induced pro-inflammatory responses. Small 2013, 9, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Boomi, P.; Ganesan, R.; Poorani, G.P.; Jegatheeswaran, S.; Balakumar, C.; Prabu, H.G.; Anand, K.; Prabhu, N.M.; Jeyakanthan, J.; Saravanan, M. Phyto-Engineered Gold Nanoparticles (AuNPs) with Potential Antibacterial, Antioxidant, and Wound Healing Activities Under in vitro and in vivo Conditions. Int. J. Nanomed. 2020, 15, 7553. [Google Scholar] [CrossRef] [PubMed]
- Ni, C.; Zhou, J.; Kong, N.; Bian, T.; Zhang, Y.; Huang, X.; Xiao, Y.; Yang, W.; Yan, F. Gold nanoparticles modulate the crosstalk between macrophages and periodontal ligament cells for periodontitis treatment. Biomaterials 2019, 206, 115–132. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.-L.; Truong, C.-T.; Nguyen, B.C.Q.; Vo, T.-N.V.; Dao, T.-T.; Nguyen, V.-D.; Trinh, D.-T.T.; Huynh, H.K.; Bui, C.-B. Anti-inflammatory and wound healing activities of calophyllolide isolated from Calophyllum inophyllum Linn. PLoS ONE 2017, 12, e0185674. [Google Scholar] [CrossRef] [PubMed]
- Singer, A.J.; Clark, R.A. Cutaneous wound healing. N. Engl. J. Med. 1999, 341, 738–746. [Google Scholar] [CrossRef]
- Trengove, N.J.; Stacey, M.C.; MacAuley, S.; Bennett, N.; Gibson, J.; Burslem, F.; Murphy, G.; Schultz, G. Analysis of the acute and chronic wound environments: The role of proteases and their inhibitors. Wound Repair. Regen. 1999, 7, 442–452. [Google Scholar] [CrossRef]
- Altman, R.; Bedi, A.; Manjoo, A.; Niazi, F.; Shaw, P.; Mease, P. Anti-inflammatory effects of intra-articular hyaluronic acid: A systematic review. Cartilage 2019, 10, 43–52. [Google Scholar] [CrossRef]
- Anderson, I. The properties of hyaluronan and its role in wound healing. Prof. Nurse. 2001, 17, 232–235. [Google Scholar]
- Zhao, J.Y.; Chai, J.K.; Song, H.F.; Zhang, J.; Xu, M.H.; Liang, Y.-D. Influence of hyaluronic acid on wound healing using composite porcine acellular dermal matrix grafts and autologous skin in rabbits. Int. Wound J. 2013, 10, 562–572. [Google Scholar] [CrossRef]
- Senel, O.; Cetinkale, O.; Ozbay, G.; Ahçioğlu, F.; Bulan, R. Oxygen free radicals impair wound healing in ischemic rat skin. Ann. Plast. Surg. 1997, 39, 516–523. [Google Scholar] [CrossRef]
- Litwiniuk, M.; Krejner, A.; Speyrer, M.S.; Gauto, A.R.; Grzela, T. Hyaluronic acid in inflammation and tissue regeneration. Wounds 2016, 28, 78–88. [Google Scholar] [PubMed]
- Trabucchi, E.; Pallotta, S.; Morini, M.; Corsi, F.; Franceschini, R.; Casiraghi, A.; Pravettoni, A.; Foschi, D.; Minghetti, P. Low molecular weight hyaluronic acid prevents oxygen free radical damage to granulation tissue during wound healing. Int. J. Tissue React. 2002, 24, 65. [Google Scholar] [PubMed]
- Cheng, H.; Lai, G.; Fu, L.; Zhang, H.; Yu, A. Enzymatically catalytic deposition of gold nanoparticles by glucose oxidase-functionalized gold nanoprobe for ultrasensitive electrochemical immunoassay. Biosens. Bioelectron. 2015, 71, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.; Singh, P.; Castro-Aceituno, V.; Yesmin Simu, S.; Kim, Y.-J.; Mathiyalagan, R.; Yang, D.-C. Gold nanoparticles synthesized using Panax ginseng leaves suppress inflammatory-mediators production via blockade of NF-κB activation in macrophages. Artif. Cells Nanomed. Biotechnol. 2017, 45, 270–276. [Google Scholar] [CrossRef]
- Pinho, R.A.; Haupenthal, D.P.; Fauser, P.E.; Thirupathi, A.; Silveira, P.C.L. Gold Nanoparticle-Based Therapy for Muscle Inflammation and Oxidative Stress. J. Inflamm. Res. 2022, 15, 3219–3234. [Google Scholar] [CrossRef]
- Harding, K.G.; Morris, H.L.; Patel, G.K. Science, medicine and the future: Healing chronic wounds. BMJ 2002, 324, 160–163. [Google Scholar] [CrossRef]
- Tai, G.; Tai, M.; Zhao, M. Electrically stimulated cell migration and its contribution to wound healing. Burn. Trauma 2018, 6. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mendes, C.; Thirupathi, A.; Zaccaron, R.P.; Corrêa, M.E.A.B.; Bittencourt, J.V.S.; Casagrande, L.d.R.; de Lima, A.C.S.; de Oliveira, L.L.; de Andrade, T.A.M.; Gu, Y.; et al. Microcurrent and Gold Nanoparticles Combined with Hyaluronic Acid Accelerates Wound Healing. Antioxidants 2022, 11, 2257. https://doi.org/10.3390/antiox11112257
Mendes C, Thirupathi A, Zaccaron RP, Corrêa MEAB, Bittencourt JVS, Casagrande LdR, de Lima ACS, de Oliveira LL, de Andrade TAM, Gu Y, et al. Microcurrent and Gold Nanoparticles Combined with Hyaluronic Acid Accelerates Wound Healing. Antioxidants. 2022; 11(11):2257. https://doi.org/10.3390/antiox11112257
Chicago/Turabian StyleMendes, Carolini, Anand Thirupathi, Rubya Pereira Zaccaron, Maria Eduarda Anastácio Borges Corrêa, João V. S. Bittencourt, Laura de Roch Casagrande, Anadhelly C. S. de Lima, Lara L. de Oliveira, Thiago A. M. de Andrade, Yaodong Gu, and et al. 2022. "Microcurrent and Gold Nanoparticles Combined with Hyaluronic Acid Accelerates Wound Healing" Antioxidants 11, no. 11: 2257. https://doi.org/10.3390/antiox11112257
APA StyleMendes, C., Thirupathi, A., Zaccaron, R. P., Corrêa, M. E. A. B., Bittencourt, J. V. S., Casagrande, L. d. R., de Lima, A. C. S., de Oliveira, L. L., de Andrade, T. A. M., Gu, Y., Feuser, P. E., Machado-de-Ávila, R. A., & Silveira, P. C. L. (2022). Microcurrent and Gold Nanoparticles Combined with Hyaluronic Acid Accelerates Wound Healing. Antioxidants, 11(11), 2257. https://doi.org/10.3390/antiox11112257






