Physical and Oxidative Water-in-Oil Emulsion Stability by the Addition of Liposomes from Shrimp Waste Oil with Antioxidant and Anti-Inflammatory Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Extraction of Low Purified Phospholipids
2.3. Liposome Preparation and Particle Properties
2.4. Cryo-Transmission Electron Microscopy (Cryo-TEM)
2.5. Differential Scanning Calorimetry
2.6. Antioxidant Activity
2.7. Astaxanthin Determination
2.8. EPA and DHA Determination
2.9. Encapsulation Efficiency
2.10. Cellular Viability and Anti-Inflammatory Properties
2.10.1. Cell Culture
2.10.2. Cell Viability
2.10.3. Immuno-Stimulation
2.11. Emulsion Preparation
2.12. Optical Microscopy
2.13. Rheological Properties
2.14. Evaluation of Lipid Oxidation
2.15. Statistical Analyses
3. Results and Discussion
3.1. Particle Properties and Morphology
3.2. Thermal Properties
3.3. Encapsulation Efficiency (EE) and Antioxidant Activity
3.4. Cellular Viability and Anti-Inflammatory Properties
3.5. Water-in-Oil Emulsion Stability
3.5.1. Optical Microscopy
3.5.2. Rheological Properties
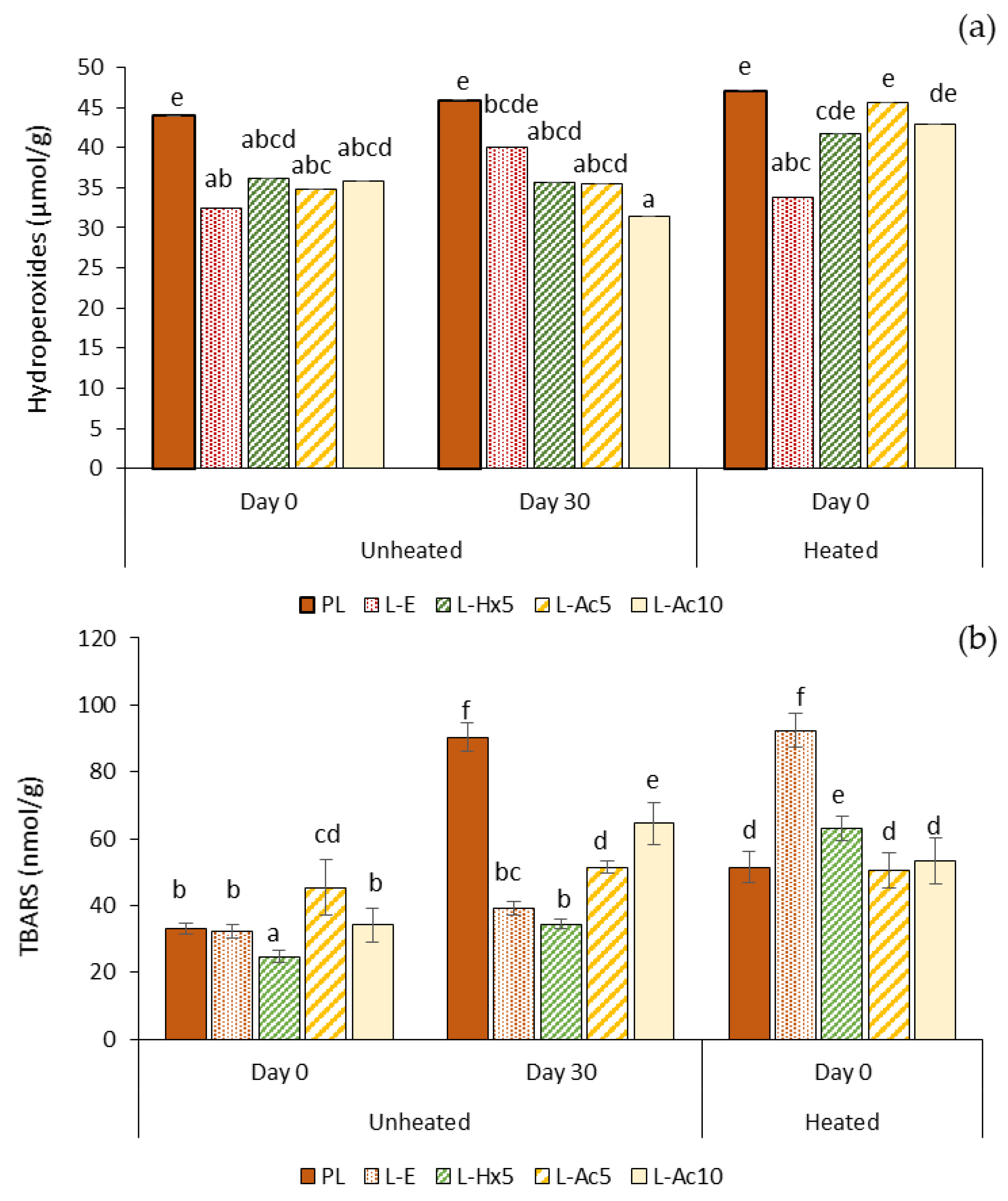
3.5.3. Oxidative Stability
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Gómez-Estaca, J.; Calvo, M.M.; Álvarez-Acero, I.; Montero, P.; Gómez-Guillén, M.C. Characterization and storage stability of astaxanthin esters, fatty acid profile and α-tocopherol of lipid extract from shrimp (L. vannamei) waste with potential applications as food ingredient. Food Chem. 2017, 216, 37–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cretton, M.; Malanga, G.; Mazzuca Sobczuk, T.; Mazzuca, M. Lipid Fraction from Industrial Crustacean Waste and Its Potential as a Supplement for the Feed Industry: A Case Study in Argentine Patagonia. Waste Biomass Valorization 2021, 12, 2311–2319. [Google Scholar] [CrossRef]
- Saini, R.K.; Song, M.-H.; Rengasamy, K.R.R.; Ko, E.-Y.; Keum, Y.-S. Red Shrimp Are a Rich Source of Nutritionally Vital Lipophilic Compounds: A Comparative Study among Edible Flesh and Processing Waste. Foods 2020, 9, 1179. [Google Scholar] [CrossRef] [PubMed]
- Pascual-Silva, C.; Alemán, A.; Montero, M.P.; Gómez-Guillén, M.C. Extraction and characterization of Argentine red shrimp (Pleoticus muelleri) phospholipids as raw material for liposome production. Food Chem. 2022, 374, 131766. [Google Scholar] [CrossRef]
- Marín, D.; Alemán, A.; Sánchez-Faure, A.; Montero, P.; Gómez-Guillén, M.C. Freeze-dried phosphatidylcholine liposomes encapsulating various antioxidant extracts from natural waste as functional ingredients in surimi gels. Food Chem. 2018, 245, 525–535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Darachai, P.; Limpawattana, M.; Hawangjoo, M.; Klaypradit, W. Effects of Shrimp Waste Types and Their Cooking on Properties of Extracted Astaxanthin and Its Characteristics in Liposomes. J. Food Nutr. Res. 2019, 7, 530–536. [Google Scholar] [CrossRef]
- Gulzar, S.; Benjakul, S. Characteristics and storage stability of nanoliposomes loaded with shrimp oil as affected by ultrasonication and microfluidization. Food Chem. 2020, 310, 125916. [Google Scholar] [CrossRef]
- Hassane Hamadou, A.; Huang, W.-C.; Xue, C.; Mao, X. Formulation of vitamin C encapsulation in marine phospholipids nanoliposomes: Characterization and stability evaluation during long term storage. LWT—Food Sci. Technol. 2020, 127, 109439. [Google Scholar] [CrossRef]
- Burri, L.; Hoem, N.; Banni, S.; Berge, K. Marine omega-3 phospholipids: Metabolism and biological activities. Int. J. Mol. Sci. 2012, 13, 15401–15419. [Google Scholar] [CrossRef] [Green Version]
- Murru, E.; Banni, S.; Carta, G. Nutritional properties of dietary omega-3-enriched phospholipids. BioMed Res. Int. 2013, 2013, 965417. [Google Scholar] [CrossRef]
- Lordan, R.; Tsoupras, A.; Zabetakis, I. Phospholipids of animal and marine origin: Structure, function, and anti-inflammatory properties. Molecules 2017, 22, 1964. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, S.; Zhang, T.-T.; Cong, P.-X.; Xu, J.; Xue, C.-H.; Jiang, X.-M.; Wang, Y.-M. Comparative study on the digestion and absorption characteristics of n-3 LCPUFA-enriched phospholipids in the form of liposomes and emulsions. Food Res. Int. 2020, 137, 109428. [Google Scholar] [CrossRef]
- Li, J.; He, Y.; Anankanbil, S.; Guo, Z. Chapter 7—Phospholipid-Based Surfactants. In Biobased Surfactants, 2nd ed.; Hayes, D.G., Solaiman, D.K.Y., Ashby, R.D., Eds.; AOCS Press: Urbana, IL, USA, 2019; pp. 243–286. [Google Scholar] [CrossRef]
- Huang, L.; Lu, X.; Zhang, L.; Liang, P. Insight into the emulsifying properties of DHA-enriched phospholipids from large yellow croaker (Larimichthys Crocea) roe. LWT—Food Sci. Technol. 2021, 150, 111984. [Google Scholar] [CrossRef]
- Sommerling, J.H.; de Matos, M.B.C.; Hildebrandt, E.; Dessy, A.; Kok, R.J.; Nirschl, H.; Leneweit, G. Instability Mechanisms of Water-in-Oil Nanoemulsions with Phospholipids: Temporal and Morphological Structures. Langmuir 2018, 34, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Gao, H.; McClements, D.J.; Zeng, H.; Ma, L.; Zou, L.; Miao, J.; Wu, X.; Tan, J.; Liang, R.; et al. Impact of polysaccharide mixtures on the formation, stability and EGCG loading of water-in-oil high internal phase emulsions. Food Chem. 2022, 372, 131225. [Google Scholar] [CrossRef]
- Wang, M.; Yan, W.; Zhou, Y.; Fan, L.; Liu, Y.; Li, J. Progress in the application of lecithins in water-in-oil emulsions. Trends Food Sci. Technol. 2021, 118, 388–398. [Google Scholar] [CrossRef]
- Waraho, T.; McClements, D.J.; Decker, E.A. Mechanisms of lipid oxidation in food dispersions. Trends Food Sci. Technol. 2011, 22, 3–13. [Google Scholar] [CrossRef]
- Chen, B.; Rao, J.; Ding, Y.; McClements, D.J.; Decker, E.A. Lipid oxidation in base algae oil and water-in-algae oil emulsion: Impact of natural antioxidants and emulsifiers. Food Res. Int. 2016, 85, 62–169. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Liu, J.; Salt, L.J.; Ridout, M.J.; Han, J.; Wilde, P.J. Structural stability of liposome-stabilized oil-in-water pickering emulsions and their fate during: In vitro digestion. Food Funct. 2019, 10, 7262–7274. [Google Scholar] [CrossRef]
- Cengiz, A.; Schroën, K.; Berton-Carabin, C. Towards Oxidatively Stable Emulsions Containing Iron-Loaded Liposomes: The Key Role of Phospholipid-to-Iron Ratio. Foods 2021, 10, 1293. [Google Scholar] [CrossRef]
- Taladrid, D.; Marín, D.; Alemán, A.; Álvarez-Acero, I.; Montero, P.; Gómez-Guillén, M.C. Effect of chemical composition and sonication procedure on properties of food-grade soy lecithin liposomes with added glycerol. Food Res. Int. 2017, 100, 541–550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Popov, I.N.; Lewin, G. Photochemiluminescent detection of antiradical activity; IV: Testing of lipid-soluble antioxidants. J. Biochem. Biophys. Methods 1996, 31, 1–8. [Google Scholar] [CrossRef]
- Alemán, A.; Mastrogiacomo, I.; López-Caballero, M.E.; Ferrari, M.; Montero, P.; Gómez-Guillén, M.C. A novel functional wrapping design by complexation of ε-polylysine with liposomes entrapping bioactive peptides. Food Bioprocess Technol. 2016, 9, 1113–1124. [Google Scholar] [CrossRef] [Green Version]
- Gómez-Guillén, M.C.; Montero, P.; López-Caballero, M.E.; Baccan, G.C.; Gómez-Estaca, J. Bioactive and technological functionality of a lipid extract from shrimp (L. vannamei) cephalothorax. LWT-Food Sci. Technol. 2018, 89, 704–711. [Google Scholar] [CrossRef] [Green Version]
- Tong, L.; Sasaki, S.; McClements, D.; Decker, E. Antioxidant Activity of Whey in a Salmon Oil Emulsion. J. Food Sci. 2000, 65, 1325–1329. [Google Scholar] [CrossRef]
- Hama, S.; Uenishi, S.; Yamada, A.; Ohgita, T.; Tsuchiya, H.; Yamashita, E.; Kogure, K. Scavenging of hydroxyl radicals in aqueous solution by astaxanthin encapsulated in liposomes. Biol. Pharm. Bull. 2012, 35, 2238–2242. [Google Scholar] [CrossRef] [Green Version]
- Marín, D.; Alemán, A.; Montero, P.; Gómez-Guillén, M.C. Encapsulation of food waste compounds in soy phosphatidylcholine liposomes: Effect of freeze-drying, storage stability and functional aptitude. J. Food Eng. 2018, 223, 132–143. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.; Briggs, J.L. Rheological and thermal properties of soybean oils with modified FA compositions. J. Am. Oil Chem. Soc. 2002, 79, 831–836. [Google Scholar] [CrossRef]
- Peng, C.-H.; Chang, C.-H.; Peng, R.Y.; Chyau, C.-C. Improved membrane transport of astaxanthine by liposomal encapsulation. Eur. J. Pharm. Biopharm. 2010, 75, 154–161. [Google Scholar] [CrossRef]
- Saito, H.; Ishihara, K. Antioxidant activity and active sites of phospholipids as antioxidants. J. Am. Oil Chem. Soc. 1997, 74, 1531–1536. [Google Scholar] [CrossRef]
- Bakır, T.; Beker, B.Y.; Sönmezoğlu, İ.; İmer, F.; Apak, R. Antioxidant and prooxidant effects of α- tocopherol in a linoleic acid- copper (II)-ascorbate system. Eur. J. Lipid Sci. Technol. 2013, 115, 372–376. [Google Scholar] [CrossRef]
- Alemán, A.; Marín-Peñalver, D.; Fernández de Palencia, P.; Gómez-Guillén, M.C.; Montero, P. Anti-inflammatory properties, bioaccessibility and intestinal absorption of sea fennel (Crithmum maritimum) extract encapsulated in soy phosphatidylcholine liposomes. Nutrients 2022, 14, 210. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jiao, G.; Song, C.; Gu, S.; Brown, R.E.; Zhang, J.; Zhang, P.; Gagnon, J.; Locke, S.; Stefanova, R.; et al. An Extract from Shrimp Processing By-Products Protects SH-SY5Y Cells from Neurotoxicity Induced by Aβ25–35. Mar. Drugs 2017, 15, 83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos, D.D.; Cahú, T.B.; Firmino, G.O.; de Castro, C.C.M.M.B.; Carvalho, L.B., Jr.; Bezerra, R.S.; Lima Filho, J.L. Shrimp Waste Extract and Astaxanthin:Rat Alveolar Macrophage, OxidativeStress and Inflammation. J. Food Sci. 2012, 77, H141–H146. [Google Scholar] [CrossRef] [PubMed]
- Kao, Y.-F.; Wu, Y.-H.S.; Chou, C.-H.; Fu, S.-G.; Liu, C.-W.; Chai, H.-J.; Chen, Y.-C. Manufacture and characterization of anti-inflammatory liposomes from jumbo flying squid (Dosidicus gigas) skin phospholipid extraction. Food Funct. 2018, 9, 3986–3996. [Google Scholar] [CrossRef]
- McClements, D.J.; Gumus, C.E. Natural emulsifiers—Biosurfactants, phospholipids, biopolymers, and colloidal particles: Molecular and physicochemical basis of functional performance. Adv. Colloid Interface Sci. 2016, 234, 3–26. [Google Scholar] [CrossRef] [Green Version]
- Xie, L.; Shi, C.; Cui, X.; Zeng, H. Surface Forces and Interaction Mechanisms of Emulsion Drops and Gas Bubbles in Complex Fluids. Langmuir 2017, 33, 3911–3925. [Google Scholar] [CrossRef] [Green Version]
- Xiong, W.; Ren, C.; Tian, M.; Yang, X.; Li, J.; Li, B. Emulsion stability and dilatational viscoelasticity of ovalbumin/chitosan complexes at the oil-in-water interface. Food Chem. 2018, 252, 181–188. [Google Scholar] [CrossRef]
- Zhu, Y.; Gao, H.; Liu, W.; Zou, L.; McClements, D.J. A review of the rheological properties of dilute and concentrated food emulsions. J. Texture Stud. 2020, 51, 45–55. [Google Scholar] [CrossRef]
- Gumus, C.E.; Decker, E.A.; McClements, D.J. Formation and stability of omega-3 oil emulsion-based delivery systems using plant proteins as emulsifiers: Lentil, pea, and Faba bean proteins. Food Biophys. 2017, 12, 186–197. [Google Scholar] [CrossRef]
- Zaccone, A.; Winter, H.H.; Siebenburger, M.; Ballauff, M. Linking self-assembly, rheology, and gel transition in attractive colloids. J. Rheol. 2014, 58, 1219–1244. [Google Scholar] [CrossRef]
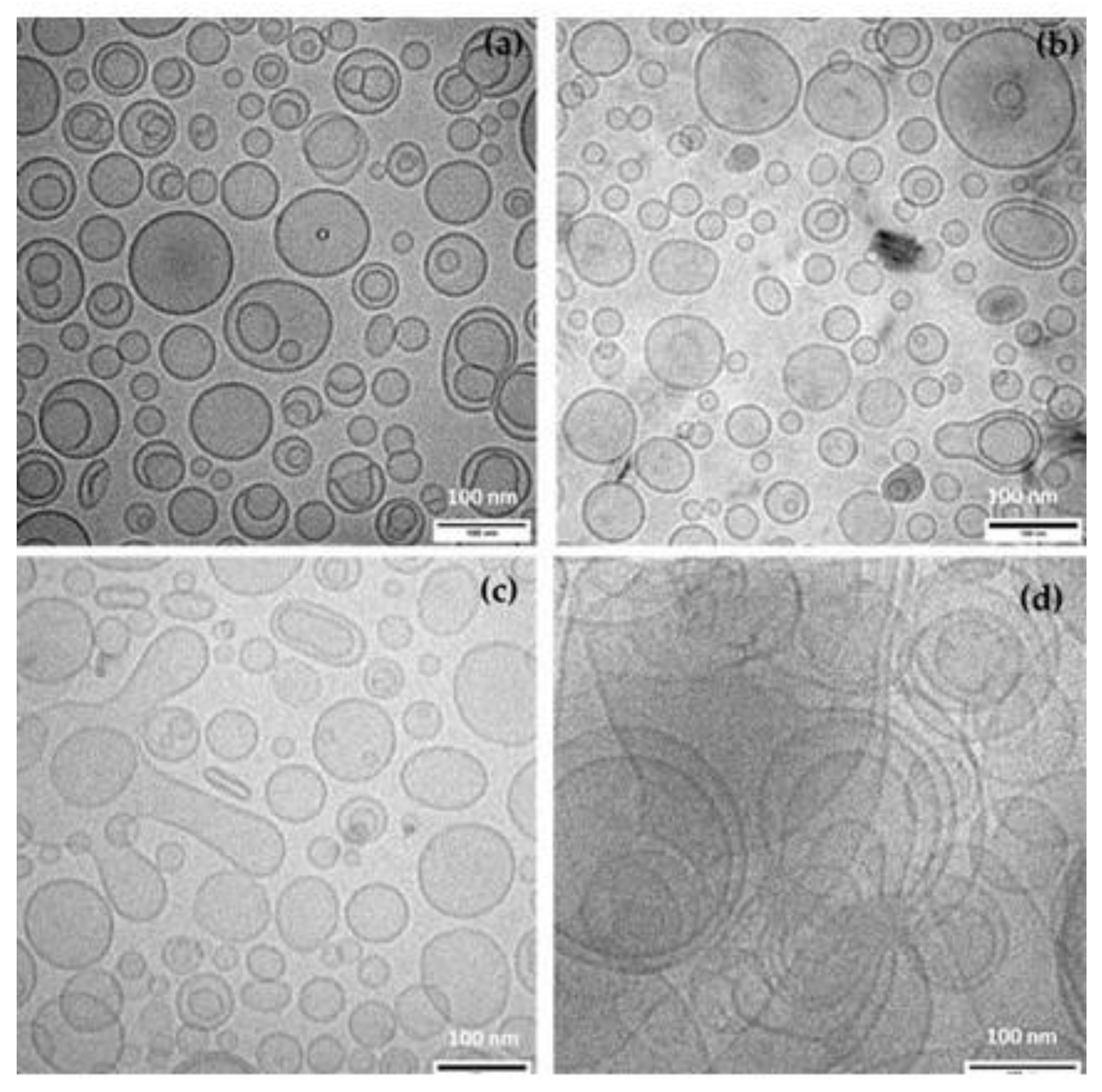




| Sample | 0 Days | 14 Days | 28 Days | |
|---|---|---|---|---|
| z-average (nm) | L-Hx5 | 143.1 ± 2.1 A/a | 145.7 ± 1.4 A/a | 144.9 ± 0.3 A/a |
| L-Ac5 | 145.3 ± 0.9 A/a | 145.9 ± 1.8 A/ab | 150.5 ± 2.4 B/b | |
| L-Ac10 | 147.1 ± 2.2 A/b | 149.2 ± 0.5 A/b | 148.3 ± 1.4 A/ab | |
| L-PLlow | 190.5 ± 1.7 A/c | 198.7 ± 2.3 B/c | 208.6 ± 2.4 C/c | |
| PDI | L-Hx5 | 0.17 ± 0.01 A/a | 0.18 ± 0.03 A/a | 0.17 ± 0.02 A/a |
| L-Ac5 | 0.24 ± 0.02 A/b | 0.25 ± 0.01 A/b | 0.28 ± 0.02 B/b | |
| L-Ac10 | 0.24 ± 0.01 A/b | 0.27 ± 0.01 AB/b | 0.27 ± 0.02 B/b | |
| L-PLlow | 0.22 ± 0.01 A/b | 0.21 ± 0.02 A/a | 0.22 ± 0.02 A/c | |
| ζ Potential (mV) | L-Hx5 | −67.1 ± 0.9 A/a | −64.1 ± 1.6 B/a | −64.1 ± 1.6 B/a |
| L-Ac5 | −66.9 ± 0.6 A/a | −63.1 ± 1.5 B/a | −56.3 ± 0.7 C/b | |
| L-Ac10 | −68.4 ± 0.8 A/ab | −64.5 ± 1.6 B/a | −63.4 ± 1.2 B/a | |
| L-PLlow | −69.4 ± 1.4 A/b | −75.2 ± 0.9 B/b | −66.8 ± 2.1 C/c |
| EEAsx | EEEPA+DHA | PCL | ABTS | |
|---|---|---|---|---|
| PL | - | - | 189 ± 2.83 a | 3.26 ± 0.07 a |
| Hx | - | - | 596 ± 96.8 b | 2.53 ± 0.05 b |
| Ac | - | - | 58.2 ± 2.90 c | 2.24 ± 0.43 b |
| L-E | - | - | 6.68 ± 1.58 a | 209 ± 14.5 a |
| L-Hx5 | 94.0 ± 0.10 a | 69.0 ± 0.81 a | 25.9 ± 1.53 b | 207 ± 7.66 a |
| L-Ac5 | 98.4 ± 0.30 b | 95.8 ± 5.99 b | 12.2 ± 2.81 c | 245 ± 5.62 b |
| L-Ac10 | 98.0 ± 0.13 b | 91.1 ± 12.6 b | 11.9 ± 3.22 c | 228 ± 1.02 c |
| κup (Pa·s−n) | nup | κdown (Pa·s−n) | ndown | G0′ (Pa) | G0′ (Pa) | n′ | n′′ | tanδ | |
|---|---|---|---|---|---|---|---|---|---|
| PL | 0.65 ± 0.03 | 0.51 ± 0.02 | 1.04 ± 0.01 | 0.37 ± 0.02 | - | - | - | - | 0.43 ± 0.05 |
| L-E | 8.43 ± 0.24 | 0.26 ± 0.02 | 5.88 ± 0.03 | 0.33 ± 0.01 | 55.1 ± 2.37 | 5.24 ± 0.32 | 0.07 ± 0.01 | 0.26 ± 0.01 | 0.13 ± 0.01 |
| L-Hx5 | 6.19 ± 0.40 | 0.28 ± 0.05 | 2.59 ± 0.94 | 0.45 ± 0.01 | 64.9 ± 1.92 | 4.90 ± 0.32 | 0.04 ± 0.01 | 0.27 ± 0.03 | 0.10 ± 0.01 |
| L-Ac5 | 3.56 ± 0.11 | 0.34 ± 0.00 | 2.16 ± 0.01 | 0.46 ± 0.01 | 16.8 ± 0.66 | 3.34 ± 0.39 | 0.14 ± 0.01 | 0.28 ± 0.02 | 0.25 ± 0.03 |
| L-Ac10 | 1.10 ± 0.22 | 0.59 ± 0.03 | 1.78 ± 0.02 | 0.40 ± 0.01 | 1.87 ± 0.32 | 0.76 ± 0.18 | 0.17 ± 0.03 | 0.42 ± 0.01 | 0.58 ± 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pascual-Silva, C.; Alemán, A.; López-Caballero, M.E.; Montero, M.P.; Gómez-Guillén, M.d.C. Physical and Oxidative Water-in-Oil Emulsion Stability by the Addition of Liposomes from Shrimp Waste Oil with Antioxidant and Anti-Inflammatory Properties. Antioxidants 2022, 11, 2236. https://doi.org/10.3390/antiox11112236
Pascual-Silva C, Alemán A, López-Caballero ME, Montero MP, Gómez-Guillén MdC. Physical and Oxidative Water-in-Oil Emulsion Stability by the Addition of Liposomes from Shrimp Waste Oil with Antioxidant and Anti-Inflammatory Properties. Antioxidants. 2022; 11(11):2236. https://doi.org/10.3390/antiox11112236
Chicago/Turabian StylePascual-Silva, Carolina, Ailén Alemán, María Elvira López-Caballero, María Pilar Montero, and María del Carmen Gómez-Guillén. 2022. "Physical and Oxidative Water-in-Oil Emulsion Stability by the Addition of Liposomes from Shrimp Waste Oil with Antioxidant and Anti-Inflammatory Properties" Antioxidants 11, no. 11: 2236. https://doi.org/10.3390/antiox11112236
APA StylePascual-Silva, C., Alemán, A., López-Caballero, M. E., Montero, M. P., & Gómez-Guillén, M. d. C. (2022). Physical and Oxidative Water-in-Oil Emulsion Stability by the Addition of Liposomes from Shrimp Waste Oil with Antioxidant and Anti-Inflammatory Properties. Antioxidants, 11(11), 2236. https://doi.org/10.3390/antiox11112236





