Targeting Ferroptosis as a Promising Therapeutic Strategy for Ischemia-Reperfusion Injury
Abstract
1. Introduction
2. Pathological Mechanism of Ferroptosis in I/R Injury
3. Therapeutic Strategies Targeting Ferroptosis in I/R Injury
3.1. Ferroptosis in Cerebral I/R Injury
3.1.1. Therapeutic Targets of Ferroptosis in Cerebral I/R Injury
3.1.2. Pharmacological Therapies Targeting Ferroptosis in Cerebral I/R Injury
3.2. Ferroptosis in Myocardial I/R Injury
3.2.1. Therapeutic Targets of Ferroptosis in Myocardial I/R Injury
3.2.2. Pharmacological Therapies Targeting Ferroptosis in Myocardial I/R Injury
3.3. Ferroptosis in Lung I/R Injury
3.4. Ferroptosis in Hepatic I/R Injury
3.4.1. Therapeutic Targets of Ferroptosis in Hepatic I/R Injury
3.4.2. Pharmacological Therapies Targeting Ferroptosis in Hepatic I/R Injury
3.5. Ferroptosis in Renal I/R Injury
3.5.1. Therapeutic Targets of Ferroptosis in Renal I/R Injury
3.5.2. Pharmacological Therapies Targeting Ferroptosis in Renal I/R Injury
3.6. Ferroptosis in Intestinal I/R Injury
4. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Eltzschig, H.K.; Eckle, T. Ischemia and reperfusion—From mechanism to translation. Nat. Med. 2011, 17, 1391–1401. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.Q.; Guo, Y.; Li, X.; Zhang, G.Q.; Li, P. Small molecules as modulators of regulated cell death against ischemia/reperfusion injury. Med. Res. Rev. 2022, 42, 2067–2101. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Zandkarimi, F.; Zhang, Y.; Meena, J.K.; Kim, J.; Zhuang, L.; Tyagi, S.; Ma, L.; Westbrook, T.F.; Steinberg, G.R.; et al. Energy-stress-mediated ampk activation inhibits ferroptosis. Nat. Cell. Biol. 2020, 22, 225–234. [Google Scholar] [CrossRef]
- Heusch, G. Myocardial ischaemia-reperfusion injury and cardioprotection in perspective. Nat. Rev. Cardiol. 2020, 17, 773–789. [Google Scholar] [CrossRef] [PubMed]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An iron-dependent form of nonapoptotic cell death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.F.; Tuo, Q.Z.; Yin, Q.Z.; Lei, P. The pathological role of ferroptosis in ischemia/reperfusion-related injury. Zool Res. 2020, 41, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.J.; Luo, X.J.; Tu, H.; Chen, H.; Xiong, X.M.; Li, N.S.; Peng, J. Ferroptosis occurs in phase of reperfusion but not ischemia in rat heart following ischemia or ischemia/reperfusion. Naunyn Schmiedebergs Arch. Pharmacol. 2021, 394, 401–410. [Google Scholar] [CrossRef]
- Wang, H.; Liu, C.; Zhao, Y.; Gao, G. Mitochondria regulation in ferroptosis. Eur. J. Cell. Biol. 2020, 99, 151058. [Google Scholar] [CrossRef]
- Gao, M.; Yi, J.; Zhu, J.; Minikes, A.M.; Monian, P.; Thompson, C.B.; Jiang, X. Role of mitochondria in ferroptosis. Mol. Cell. 2019, 73, 354–363.e3. [Google Scholar] [CrossRef]
- Loor, G.; Kondapalli, J.; Iwase, H.; Chandel, N.S.; Waypa, G.B.; Guzy, R.D.; Hoek, T.L.V.; Schumacker, P.T. Mitochondrial oxidant stress triggers cell death in simulated ischemia-reperfusion. Biochim. Biophys. Acta 2011, 1813, 1382–1394. [Google Scholar] [CrossRef]
- Li, X.; Ma, N.; Xu, J.; Zhang, Y.; Yang, P.; Su, X.; Xing, Y.; An, N.; Yang, F.; Zhang, G.; et al. Targeting ferroptosis: Pathological mechanism and treatment of ischemia-reperfusion injury. Oxid. Med. Cell. Longev. 2021, 2021, 1587922. [Google Scholar] [CrossRef] [PubMed]
- Tuo, Q.Z.; Lei, P.; Jackman, K.A.; Li, X.L.; Xiong, H.; Li, X.L.; Liuyang, Z.Y.; Roisman, L.; Zhang, S.T.; Ayton, S.; et al. Tau-mediated iron export prevents ferroptotic damage after ischemic stroke. Mol. Psychiatry 2017, 22, 1520–1530. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Jiang, L.; Hu, Y.; Tang, N.; Liang, N.; Li, X.F.; Chen, Y.W.; Qin, H.; Wu, L. Ferritin reduction is essential for cerebral ischemia-induced hippocampal neuronal death through p53/slc7a11-mediated ferroptosis. Brain Res. 2021, 1752, 147216. [Google Scholar] [CrossRef]
- Li, C.; Sun, G.; Chen, B.; Xu, L.; Ye, Y.; He, J.; Bao, Z.; Zhao, P.; Miao, Z.; Zhao, L.; et al. Nuclear receptor coactivator 4-mediated ferritinophagy contributes to cerebral ischemia-induced ferroptosis in ischemic stroke. Pharmacol. Res. 2021, 174, 105933. [Google Scholar] [CrossRef]
- Wang, P.; Cui, Y.; Ren, Q.; Yan, B.; Zhao, Y.; Yu, P.; Gao, G.; Shi, H.; Chang, S.; Chang, Y.Z. Mitochondrial ferritin attenuates cerebral ischaemia/reperfusion injury by inhibiting ferroptosis. Cell. Death Dis. 2021, 12, 447. [Google Scholar] [CrossRef]
- Huang, Y.; Liu, J.; He, J.; Hu, Z.; Tan, F.; Zhu, X.; Yuan, F.; Jiang, Z. Ubiad1 alleviates ferroptotic neuronal death by enhancing antioxidative capacity by cooperatively restoring impaired mitochondria and golgi apparatus upon cerebral ischemic/reperfusion insult. Cell. Biosci. 2022, 12, 42. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Liu, Y.; Li, K.; Yuan, D.; Yang, S.; Zhou, L.; Zhao, Y.; Miao, S.; Lv, C.; Zhao, J. Cox-2/pge2 pathway inhibits the ferroptosis induced by cerebral ischemia reperfusion. Mol. Neurobiol. 2022, 59, 1619–1631. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Wu, Y.; Liang, S.; Piao, X. Activation of ssat1/alox15 axis aggravates cerebral ischemia/reperfusion injury via triggering neuronal ferroptosis. Neuroscience 2022, 485, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Tuo, Q.Z.; Liu, Y.; Xiang, Z.; Yan, H.F.; Zou, T.; Shu, Y.; Ding, X.L.; Zou, J.J.; Xu, S.; Tang, F.; et al. Thrombin induces acsl4-dependent ferroptosis during cerebral ischemia/reperfusion. Signal Transduct. Target Ther. 2022, 7, 59. [Google Scholar] [CrossRef]
- Lu, J.; Xu, F.; Lu, H. Lncrna pvt1 regulates ferroptosis through mir-214-mediated tfr1 and p53. Life Sci. 2020, 260, 118305. [Google Scholar] [CrossRef]
- Gao, M.; Monian, P.; Quadri, N.; Ramasamy, R.; Jiang, X. Glutaminolysis and transferrin regulate ferroptosis. Mol. Cell. 2015, 59, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Sun, L.; Wu, W.; Wu, J.; Sun, Z.; Ren, J. Usp22 protects against myocardial ischemia-reperfusion injury via the sirt1-p53/slc7a11-dependent inhibition of ferroptosis-induced cardiomyocyte death. Front. Physiol. 2020, 11, 551318. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.J.; Zhou, Y.J.; Xiong, X.M.; Li, N.S.; Zhang, J.J.; Luo, X.J.; Peng, J. Ubiquitin-specific protease 7 promotes ferroptosis via activation of the p53/tfr1 pathway in the rat hearts after ischemia/reperfusion. Free Radic. Biol. Med. 2021, 162, 339–352. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Xiong, Y.; Zhang, Y.; Leng, Y.; Tao, J.; Li, L.; Qiu, Z.; Xia, Z. Activation of nrf2/fpn1 pathway attenuates myocardial ischemia-reperfusion injury in diabetic rats by regulating iron homeostasis and ferroptosis. Cell. Stress Chaperones 2021, 27, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Li, W.; Wang, Y.; Leng, Y.; Xia, Z. Inhibition of dnmt-1 alleviates ferroptosis through ncoa4 mediated ferritinophagy during diabetes myocardial ischemia/reperfusion injury. Cell. Death Discov. 2021, 7, 267. [Google Scholar] [CrossRef]
- Stamenkovic, A.; O’Hara, K.A.; Nelson, D.C.; Maddaford, T.G.; Edel, A.L.; Maddaford, G.; Dibrov, E.; Aghanoori, M.; Kirshenbaum, L.A.; Fernyhough, P.; et al. Oxidized phosphatidylcholines trigger ferroptosis in cardiomyocytes during ischemia-reperfusion injury. Am. J. Physiol. Heart Circ. Physiol. 2021, 320, H1170–H1184. [Google Scholar] [CrossRef]
- Chen, H.Y.; Xiao, Z.Z.; Ling, X.; Xu, R.N.; Zhu, P.; Zheng, S.Y. Elavl1 is transcriptionally activated by foxc1 and promotes ferroptosis in myocardial ischemia/reperfusion injury by regulating autophagy. Mol. Med. 2021, 27, 14. [Google Scholar] [CrossRef]
- Sun, W.; Shi, R.; Guo, J.; Wang, H.; Shen, L.; Shi, H.; Yu, P.; Chen, X. Mir-135b-3p promotes cardiomyocyte ferroptosis by targeting gpx4 and aggravates myocardial ischemia/reperfusion injury. Front. Cardiovasc. Med. 2021, 8, 663832. [Google Scholar] [CrossRef]
- Sun, W.; Wu, X.; Yu, P.; Zhang, Q.; Shen, L.; Chen, J.; Tong, H.; Fan, M.; Shi, H.; Chen, X. Lncaabr07025387.1 enhances myocardial ischemia/reperfusion injury via mir-205/acsl4-mediated ferroptosis. Front. Cell. Dev. Biol. 2022, 10, 672391. [Google Scholar] [CrossRef]
- Zhang, J.K.; Zhang, Z.; Guo, Z.A.; Fu, Y.; Chen, X.J.; Chen, W.J.; Wu, H.F.; Cui, X.J. The bmsc-derived exosomal lncrna mir9-3hg suppresses cardiomyocyte ferroptosis in ischemia-reperfusion mice via the pum2/prdx6 axis. Nutr. Metab. Cardiovasc. Dis. 2022, 32, 515–527. [Google Scholar] [CrossRef]
- Wu, S.; Yang, J.; Sun, G.; Hu, J.; Zhang, Q.; Cai, J.; Yuan, D.; Li, H.; Hei, Z.; Yao, W. Macrophage extracellular traps aggravate iron overload-related liver ischaemia/reperfusion injury. Br. J. Pharmacol. 2021, 178, 3783–3796. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Jiao, H.; Yue, Y.; He, K.; Jin, Y.; Zhang, J.; Zhang, J.; Wei, Y.; Luo, H.; Hao, Z.; et al. Ubiquitin ligase e3 huwe1/mule targets transferrin receptor for degradation and suppresses ferroptosis in acute liver injury. Cell. Death Differ. 2022, 29, 1705–1718. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wu, L.; Tian, X.; Zheng, W.; Yuan, M.; Tian, X.; Zuo, H.; Song, H.; Shen, Z. Mir-29a-3p in exosomes from heme oxygenase-1 modified bone marrow mesenchymal stem cells alleviates steatotic liver ischemia-reperfusion injury in rats by suppressing ferroptosis via iron responsive element binding protein 2. Oxid. Med. Cell. Longev. 2022, 2022, 6520789. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.L.; Liao, X.H.; Sun, H.; Jiang, X.; Liu, Q.; Zhang, L. Augmenter of liver regeneration protects the kidney from ischaemia-reperfusion injury in ferroptosis. J. Cell. Mol. Med. 2019, 23, 4153–4164. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Jiang, X.; Yang, C.; Zhang, J.; Chen, B.; Li, Y.; Yao, S.; Xie, Q.; Gomez, H.; Murugan, R.; et al. Pannexin 1 mediates ferroptosis that contributes to renal ischemia/reperfusion injury. J. Biol. Chem. 2019, 294, 19395–19404. [Google Scholar] [CrossRef]
- Sui, M.; Xu, D.; Zhao, W.; Lu, H.; Chen, R.; Duan, Y.; Li, Y.; Zhu, Y.; Zhang, L.; Zeng, L. Cirbp promotes ferroptosis by interacting with elavl1 and activating ferritinophagy during renal ischaemia-reperfusion injury. J. Cell. Mol. Med. 2021, 25, 6203–6216. [Google Scholar] [CrossRef]
- Chen, C.; Wang, D.; Yu, Y.; Zhao, T.; Min, N.; Wu, Y.; Kang, L.; Zhao, Y.; Du, L.; Zhang, M.; et al. Legumain promotes tubular ferroptosis by facilitating chaperone-mediated autophagy of gpx4 in aki. Cell. Death Dis. 2021, 12, 65. [Google Scholar] [CrossRef]
- Eleftheriadis, T.; Pissas, G.; Golfinopoulos, S.; Liakopoulos, V.; Stefanidis, I. Role of indoleamine 2,3-dioxygenase in ischemia-reperfusion injury of renal tubular epithelial cells. Mol. Med. Rep. 2021, 23, 472. [Google Scholar] [CrossRef]
- Feng, R.; Xiong, Y.; Lei, Y.; Huang, Q.; Liu, H.; Zhao, X.; Chen, Z.; Chen, H.; Liu, X.; Wang, L.; et al. Lysine-specific demethylase 1 aggravated oxidative stress and ferroptosis induced by renal ischemia and reperfusion injury through activation of tlr4/nox4 pathway in mice. J. Cell. Mol. Med. 2022, 26, 4254–4267. [Google Scholar] [CrossRef]
- Ding, C.; Ding, X.; Zheng, J.; Wang, B.; Li, Y.; Xiang, H.; Dou, M.; Qiao, Y.; Tian, P.; Xue, W. Mir-182-5p and mir-378a-3p regulate ferroptosis in i/r-induced renal injury. Cell. Death Dis. 2020, 11, 929. [Google Scholar] [CrossRef]
- Tao, W.; Liu, F.; Zhang, J.; Fu, S.; Zhan, H.; Qian, K. Mir-3587 inhibitor attenuates ferroptosis following renal ischemia-reperfusion through ho-1. Front. Mol. Biosci. 2021, 8, 789927. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Feng, D.; Wang, Z.; Zhao, Y.; Sun, R.; Tian, D.; Liu, D.; Zhang, F.; Ning, S.; Yao, J.; et al. Ischemia-induced acsl4 activation contributes to ferroptosis-mediated tissue injury in intestinal ischemia/reperfusion. Cell. Death Differ. 2019, 26, 2284–2299. [Google Scholar] [CrossRef] [PubMed]
- Deng, F.; Zhao, B.C.; Yang, X.; Lin, Z.B.; Sun, Q.S.; Wang, Y.F.; Yan, Z.Z.; Liu, W.F.; Li, C.; Hu, J.J.; et al. The gut microbiota metabolite capsiate promotes gpx4 expression by activating trpv1 to inhibit intestinal ischemia reperfusion-induced ferroptosis. Gut Microbes 2021, 13, 1902719. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Qiang, Z.; Chai, D.; Peng, J.; Xia, Y.; Hu, R.; Jiang, H. Nrf2 inhibits ferroptosis and protects against acute lung injury due to intestinal ischemia reperfusion via regulating slc7a11 and ho-1. Aging 2020, 12, 12943–12959. [Google Scholar] [CrossRef] [PubMed]
- Qiang, Z.; Dong, H.; Xia, Y.; Chai, D.; Hu, R.; Jiang, H. Nrf2 and stat3 alleviates ferroptosis-mediated iir-ali by regulating slc7a11. Oxid. Med. Cell. Longev. 2020, 2020, 5146982. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Xia, Y.; Jin, S.; Xue, C.; Wang, Y.; Hu, R.; Jiang, H. Nrf2 attenuates ferroptosis-mediated iir-ali by modulating tert and slc7a11. Cell. Death Dis. 2021, 12, 1027. [Google Scholar] [CrossRef]
- Li, Y.; Cao, Y.; Xiao, J.; Shang, J.; Tan, Q.; Ping, F.; Huang, W.; Wu, F.; Zhang, H.; Zhang, X. Inhibitor of apoptosis-stimulating protein of p53 inhibits ferroptosis and alleviates intestinal ischemia/reperfusion-induced acute lung injury. Cell. Death Differ. 2020, 27, 2635–2650. [Google Scholar] [CrossRef]
- Alim, I.; Caulfield, J.T.; Chen, Y.; Swarup, V.; Geschwind, D.H.; Ivanova, E.; Seravalli, J.; Ai, Y.; Sansing, L.H.; Marie, E.J.S.; et al. Selenium drives a transcriptional adaptive program to block ferroptosis and treat stroke. Cell 2019, 177, 1262–1279.e25. [Google Scholar] [CrossRef]
- Tuo, Q.Z.; Masaldan, S.; Southon, A.; Mawal, C.; Ayton, S.; Bush, A.I.; Lei, P.; Belaidi, A.A. Characterization of selenium compounds for anti-ferroptotic activity in neuronal cells and after cerebral ischemia-reperfusion injury. Neurotherapeutics 2021, 18, 2682–2691. [Google Scholar] [CrossRef]
- Shi, Y.; Han, L.; Zhang, X.; Xie, L.; Pan, P.; Chen, F. Selenium alleviates cerebral ischemia/reperfusion injury by regulating oxidative stress, mitochondrial fusion and ferroptosis. Neurochem. Res. 2022, 47, 2992–3002. [Google Scholar] [CrossRef]
- Guan, X.; Li, X.; Yang, X.; Yan, J.; Shi, P.; Ba, L.; Cao, Y.; Wang, P. The neuroprotective effects of carvacrol on ischemia/reperfusion-induced hippocampal neuronal impairment by ferroptosis mitigation. Life Sci. 2019, 235, 116795. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.; Wu, Y.; Liu, S.; Luo, C.; Lu, Y.; Liu, M.; Wang, L.; Zhang, Y.; Liu, X. Rehmannioside a improves cognitive impairment and alleviates ferroptosis via activating pi3k/akt/nrf2 and slc7a11/gpx4 signaling pathway after ischemia. J. Ethnopharmacol. 2022, 289, 115021. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.; Li, Z.; Zhu, S.; Cheng, M.; Ju, Y.; Ren, L.; Yang, G.; Min, D. Galangin attenuated cerebral ischemia-reperfusion injury by inhibition of ferroptosis through activating the slc7a11/gpx4 axis in gerbils. Life Sci. 2021, 264, 118660. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Zhu, L.; Tang, P.; Chen, D.; Li, Y.; Li, J.; Bao, C. Carthamin yellow improves cerebral ischemia-reperfusion injury by attenuating inflammation and ferroptosis in rats. Int. J. Mol. Med. 2021, 47, 52. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhai, Y.; Chen, J.; Xu, X.; Wang, H. Kaempferol ameliorates oxygen-glucose deprivation/reoxygenation-induced neuronal ferroptosis by activating nrf2/slc7a11/gpx4 axis. Biomolecules 2021, 11, 923. [Google Scholar] [CrossRef]
- Feng, Y.; Madungwe, N.B.; Aliagan, A.D.I.; Tombo, N.; Bopassa, J.C. Liproxstatin-1 protects the mouse myocardium against ischemia/reperfusion injury by decreasing vdac1 levels and restoring gpx4 levels. Biochem. Biophys. Res. Commun. 2019, 520, 606–611. [Google Scholar] [CrossRef]
- Xu, Y.; Li, X.; Cheng, Y.; Yang, M.; Wang, R. Inhibition of acsl4 attenuates ferroptotic damage after pulmonary ischemia-reperfusion. FASEB J. 2020, 34, 16262–16275. [Google Scholar] [CrossRef]
- Friedmann Angeli, J.P.; Schneider, M.; Proneth, B.; Tyurina, Y.Y.; Tyurin, V.A.; Hammond, V.J.; Herbach, N.; Aichler, M.; Walch, A.; Eggenhofer, E.; et al. Inactivation of the ferroptosis regulator gpx4 triggers acute renal failure in mice. Nat. Cell. Biol. 2014, 16, 1180–1191. [Google Scholar] [CrossRef]
- Luo, Y.; Apaijai, N.; Liao, S.; Maneechote, C.; Chunchai, T.; Arunsak, B.; Benjanuwattra, J.; Yanpiset, P.; Chattipakorn, S.C.; Chattipakorn, N. Therapeutic potentials of cell death inhibitors in rats with cardiac ischaemia/reperfusion injury. J. Cell. Mol. Med. 2022, 26, 2462–2476. [Google Scholar] [CrossRef]
- Chen, K.; Xu, Z.; Liu, Y.; Wang, Z.; Li, Y.; Xu, X.; Chen, C.; Xia, T.; Liao, Q.; Yao, Y.; et al. Irisin protects mitochondria function during pulmonary ischemia/reperfusion injury. Sci. Transl. Med. 2017, 9, eaao6298. [Google Scholar] [CrossRef]
- Yamada, N.; Karasawa, T.; Wakiya, T.; Sadatomo, A.; Ito, H.; Kamata, R.; Watanabe, S.; Komada, T.; Kimura, H.; Sanada, Y.; et al. Iron overload as a risk factor for hepatic ischemia-reperfusion injury in liver transplantation: Potential role of ferroptosis. Am. J. Transplant. 2020, 20, 1606–1618. [Google Scholar] [CrossRef]
- Zhou, L.; Xue, X.; Hou, Q.; Dai, C. Targeting ferroptosis attenuates interstitial inflammation and kidney fibrosis. Kidney Dis. 2022, 8, 57–71. [Google Scholar] [CrossRef] [PubMed]
- Tu, H.; Zhou, Y.J.; Tang, L.J.; Xiong, X.M.; Zhang, X.J.; Sheikh, M.S.A.; Zhang, J.J.; Luo, X.J.; Yuan, C.; Peng, J. Combination of ponatinib with deferoxamine synergistically mitigates ischemic heart injury via simultaneous prevention of necroptosis and ferroptosis. Eur. J. Pharmacol. 2021, 898, 173999. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Wang, H.; Han, D.; Xie, E.; Yang, X.; Wei, J.; Gu, S.; Gao, F.; Zhu, N.; Yin, X.; et al. Ferroptosis as a target for protection against cardiomyopathy. Proc. Natl. Acad. Sci. USA 2019, 116, 2672–2680. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.H.; Yang, K.T.; Ting, P.C.; Luo, Y.P.; Lin, D.J.; Wang, Y.S.; Chang, J.C. Gossypol acetic acid attenuates cardiac ischemia/reperfusion injury in rats via an antiferroptotic mechanism. Biomolecules 2021, 11, 1667. [Google Scholar] [CrossRef]
- Hwang, J.W.; Park, J.H.; Park, B.W.; Kim, H.; Kim, J.J.; Sim, W.S.; Mishchenko, N.P.; Fedoreyev, S.A.; Vasileva, E.A.; Ban, K.; et al. Histochrome attenuates myocardial ischemia-reperfusion injury by inhibiting ferroptosis-induced cardiomyocyte death. Antioxidants 2021, 10, 1624. [Google Scholar] [CrossRef]
- Shan, X.; Lv, Z.Y.; Yin, M.J.; Chen, J.; Wang, J.; Wu, Q.N. The protective effect of cyanidin-3-glucoside on myocardial ischemia-reperfusion injury through ferroptosis. Oxid. Med. Cell. Longev. 2021, 2021, 8880141. [Google Scholar] [CrossRef]
- Lin, J.H.; Yang, K.T.; Lee, W.S.; Ting, P.C.; Luo, Y.P.; Lin, D.J.; Wang, Y.S.; Chang, J.C. Xanthohumol protects the rat myocardium against ischemia/reperfusion injury-induced ferroptosis. Oxid. Med. Cell. Longev. 2022, 2022, 9523491. [Google Scholar] [CrossRef]
- Lv, Z.; Wang, F.; Zhang, X.; Zhang, X.; Zhang, J.; Liu, R. Etomidate attenuates the ferroptosis in myocardial ischemia/reperfusion rat model via nrf2/ho-1 pathway. Shock 2021, 56, 440–449. [Google Scholar] [CrossRef]
- Xu, S.; Wu, B.; Zhong, B.; Lin, L.; Ding, Y.; Jin, X.; Huang, Z.; Lin, M.; Wu, H.; Xu, D. Naringenin alleviates myocardial ischemia/reperfusion injury by regulating the nuclear factor-erythroid factor 2-related factor 2 (nrf2) /system xc-/ glutathione peroxidase 4 (gpx4) axis to inhibit ferroptosis. Bioengineered 2021, 12, 10924–10934. [Google Scholar] [CrossRef]
- Lu, H.; Xiao, H.; Dai, M.; Xue, Y.; Zhao, R. Britanin relieves ferroptosis-mediated myocardial ischaemia/reperfusion damage by upregulating gpx4 through activation of ampk/gsk3β/nrf2 signalling. Pharm. Biol. 2022, 60, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Lei, Z.; Yang, X.; Zhao, M.; Hou, Y.; Wang, D.; Tang, S.; Li, J.; Yu, J. Propofol protects myocardium from ischemia/reperfusion injury by inhibiting ferroptosis through the akt/p53 signaling pathway. Front. Pharmacol. 2022, 13, 841410. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Qi, K.; Gong, Y.; Long, X.; Zhu, S.; Lu, F.; Lin, K.; Xu, J. Ferulic acid alleviates myocardial ischemia reperfusion injury via upregulating ampkα2 expression-mediated ferroptosis depression. J. Cardiovasc. Pharmacol. 2021, 79, 489–500. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ren, X.; Wang, Y.; Chen, D.; Jiang, L.; Li, X.; Li, T.; Huo, M.; Li, Q. Targeting ferroptosis by polydopamine nanoparticles protects heart against ischemia/reperfusion injury. ACS Appl. Mater. Interfaces 2021, 13, 53671–53682. [Google Scholar] [CrossRef]
- Jin, T.; He, Q.; Cheng, C.; Li, H.; Liang, L.; Zhang, G.; Su, C.; Xiao, Y.; Bradley, J.; Peberdy, M.A.; et al. Uamc-3203 or/and deferoxamine improve post-resuscitation myocardial dysfunction through suppressing ferroptosis in a rat model of cardiac arrest. Shock 2022, 57, 344–350. [Google Scholar] [CrossRef]
- Li, T.; Tan, Y.; Ouyang, S.; He, J.; Liu, L. Resveratrol protects against myocardial ischemia-reperfusion injury via attenuating ferroptosis. Gene 2022, 808, 145968. [Google Scholar] [CrossRef]
- Yu, P.; Zhang, J.; Ding, Y.; Chen, D.; Sun, H.; Yuan, F.; Li, S.; Li, X.; Yang, P.; Fu, L.; et al. Dexmedetomidine post-conditioning alleviates myocardial ischemia-reperfusion injury in rats by ferroptosis inhibition via slc7a11/gpx4 axis activation. Hum. Cell. 2022, 35, 836–848. [Google Scholar] [CrossRef]
- Zhang, J.; Bi, J.; Ren, Y.; Du, Z.; Li, T.; Wang, T.; Zhang, L.; Wang, M.; Wei, S.; Lv, Y.; et al. Involvement of gpx4 in irisin’s protection against ischemia reperfusion-induced acute kidney injury. J. Cell. Physiol. 2021, 236, 931–945. [Google Scholar] [CrossRef]
- Jiang, G.P.; Liao, Y.J.; Huang, L.L.; Zeng, X.J.; Liao, X.H. Effects and molecular mechanism of pachymic acid on ferroptosis in renal ischemia reperfusion injury. Mol. Med. Rep. 2021, 23, 63. [Google Scholar] [CrossRef]
- Linkermann, A.; Skouta, R.; Himmerkus, N.; Mulay, S.R.; Dewitz, C.; de Zen, F.; Prokai, A.; Zuchtriegel, G.; Krombach, F.; Welz, P.S.; et al. Synchronized renal tubular cell death involves ferroptosis. Proc. Natl. Acad. Sci. USA 2014, 111, 16836–16841. [Google Scholar] [CrossRef]
- Zhao, Z.; Wu, J.; Xu, H.; Zhou, C.; Han, B.; Zhu, H.; Hu, Z.; Ma, Z.; Ming, Z.; Yao, Y.; et al. Xjb-5-131 inhibited ferroptosis in tubular epithelial cells after ischemia-reperfusion injury. Cell. Death Dis. 2020, 11, 629. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Quan, F.; Cao, Q.; Lin, Y.; Yue, C.; Bi, R.; Cui, X.; Yang, H.; Yang, Y.; Birnbaumer, L.; et al. Quercetin alleviates acute kidney injury by inhibiting ferroptosis. J. Adv. Res. 2021, 28, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Tonnus, W.; Meyer, C.; Steinebach, C.; Belavgeni, A.; von Mässenhausen, A.; Gonzalez, N.Z.; Maremonti, F.; Gembardt, F.; Himmerkus, N.; Latk, M.; et al. Dysfunction of the key ferroptosis-surveilling systems hypersensitizes mice to tubular necrosis during acute kidney injury. Nat. Commun. 2021, 12, 4402. [Google Scholar] [CrossRef]
- Yang, J.; Sun, X.; Huang, N.; Li, P.; He, J.; Jiang, L.; Zhang, X.; Han, S.; Xin, H. Entacapone alleviates acute kidney injury by inhibiting ferroptosis. FASEB J. 2022, 36, e22399. [Google Scholar] [CrossRef]
- Feng, Y.D.; Ye, W.; Tian, W.; Meng, J.R.; Zhang, M.; Sun, Y.; Zhang, H.N.; Wang, S.J.; Wu, K.H.; Liu, C.X.; et al. Old targets, new strategy: Apigenin-7-o-β-d-(-6″-p-coumaroyl)-glucopyranoside prevents endothelial ferroptosis and alleviates intestinal ischemia-reperfusion injury through ho-1 and mao-b inhibition. Free Radic. Biol. Med. 2022, 184, 74–88. [Google Scholar] [CrossRef] [PubMed]
- Zhongyin, Z.; Wei, W.; Juan, X.; Guohua, F. Isoliquiritin apioside relieves intestinal ischemia/reperfusion-induced acute lung injury by blocking hif-1α-mediated ferroptosis. Int. Immunopharmacol. 2022, 108, 108852. [Google Scholar] [CrossRef] [PubMed]
- Farmer, E.E.; Mueller, M.J. Ros-mediated lipid peroxidation and res-activated signaling. Annu. Rev. Plant Biol. 2013, 64, 429–450. [Google Scholar] [CrossRef]
- Riegman, M.; Sagie, L.; Galed, C.; Levin, T.; Steinberg, N.; Dixon, S.J.; Wiesner, U.; Bradbury, M.S.; Niethammer, P.; Zaritsky, A.; et al. Ferroptosis occurs through an osmotic mechanism and propagates independently of cell rupture. Nat. Cell. Biol. 2020, 22, 1042–1048. [Google Scholar] [CrossRef]
- Berenshtein, E.; Vaisman, B.; Goldberg-Langerman, C.; Kitrossky, N.; Konijn, A.M.; Chevion, M. Roles of ferritin and iron in ischemic preconditioning of the heart. Mol. Cell. Biochem. 2002, 234–235, 283–292. [Google Scholar] [CrossRef]
- Nakamura, T.; Naguro, I.; Ichijo, H. Iron homeostasis and iron-regulated ros in cell death, senescence and human diseases. Biochim. Biophys. Acta Gen. Subj. 2019, 1863, 1398–1409. [Google Scholar] [CrossRef]
- Li, J.; Cao, F.; Yin, H.L.; Huang, Z.J.; Lin, Z.T.; Mao, N.; Sun, B.; Wang, G. Ferroptosis: Past, present and future. Cell. Death Dis. 2020, 11, 88. [Google Scholar] [CrossRef]
- Yang, W.S.; Kim, K.J.; Gaschler, M.M.; Patel, M.; Shchepinov, M.S.; Stockwell, B.R. Peroxidation of polyunsaturated fatty acids by lipoxygenases drives ferroptosis. Proc. Natl. Acad. Sci. USA 2016, 113, E4966–E4975. [Google Scholar] [CrossRef] [PubMed]
- Müller, T.; Dewitz, C.; Schmitz, J.; Schröder, A.S.; Bräsen, J.H.; Stockwell, B.R.; Murphy, J.M.; Kunzendorf, U.; Krautwald, S. Necroptosis and ferroptosis are alternative cell death pathways that operate in acute kidney failure. Cell. Mol. Life Sci. 2017, 74, 3631–3645. [Google Scholar] [CrossRef] [PubMed]
- Haeggström, J.Z.; Funk, C.D. Lipoxygenase and leukotriene pathways: Biochemistry, biology, and roles in disease. Chem. Rev. 2011, 111, 5866–5898. [Google Scholar] [CrossRef] [PubMed]
- Braughler, J.M.; Duncan, L.A.; Chase, R.L. The involvement of iron in lipid peroxidation. Importance of ferric to ferrous ratios in initiation. J. Biol. Chem. 1986, 261, 10282–10289. [Google Scholar] [CrossRef]
- Doll, S.; Conrad, M. Iron and ferroptosis: A still ill-defined liaison. IUBMB Life 2017, 69, 423–434. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.S.; SriRamaratnam, R.; Welsch, M.E.; Shimada, K.; Skouta, R.; Viswanathan, V.S.; Cheah, J.H.; Clemons, P.A.; Shamji, A.F.; Clish, C.B.; et al. Regulation of ferroptotic cancer cell death by gpx4. Cell 2014, 156, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Bersuker, K.; Hendricks, J.M.; Li, Z.; Magtanong, L.; Ford, B.; Tang, P.H.; Roberts, M.A.; Tong, B.; Maimone, T.J.; Zoncu, R.; et al. The coq oxidoreductase fsp1 acts parallel to gpx4 to inhibit ferroptosis. Nature 2019, 575, 688–692. [Google Scholar] [CrossRef]
- Mao, C.; Liu, X.; Zhang, Y.; Lei, G.; Yan, Y.; Lee, H.; Koppula, P.; Wu, S.; Zhuang, L.; Fang, B.; et al. Dhodh-mediated ferroptosis defence is a targetable vulnerability in cancer. Nature 2021, 593, 586–590. [Google Scholar] [CrossRef]
- Kraft, V.A.N.; Bezjian, C.T.; Pfeiffer, S.; Ringelstetter, L.; Müller, C.; Zandkarimi, F.; Merl-Pham, J.; Bao, X.; Anastasov, N.; Kössl, J.; et al. Gtp cyclohydrolase 1/tetrahydrobiopterin counteract ferroptosis through lipid remodeling. ACS Cent. Sci. 2020, 6, 41–53. [Google Scholar] [CrossRef]
- Dietrich, R.B.; Bradley, W.G., Jr. Iron accumulation in the basal ganglia following severe ischemic-anoxic insults in children. Radiology 1988, 168, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Scindia, Y.; Dey, P.; Thirunagari, A.; Liping, H.; Rosin, D.L.; Floris, M.; Okusa, M.D.; Swaminathan, S. Hepcidin mitigates renal ischemia-reperfusion injury by modulating systemic iron homeostasis. J. Am. Soc. Nephrol. 2015, 26, 2800–2814. [Google Scholar] [CrossRef] [PubMed]
- Kakhlon, O.; Cabantchik, Z.I. The labile iron pool: Characterization, measurement, and participation in cellular processes. Free Radic. Biol. Med. 2002, 33, 1037–1046. [Google Scholar] [CrossRef]
- Valko, M.; Jomova, K.; Rhodes, C.J.; Kuča, K.; Musílek, K. Redox- and non-redox-metal-induced formation of free radicals and their role in human disease. Arch. Toxicol. 2016, 90, 1–37. [Google Scholar] [CrossRef]
- Hess, M.L.; Manson, N.H. Molecular oxygen: Friend and foe. The role of the oxygen free radical system in the calcium paradox, the oxygen paradox and ischemia/reperfusion injury. J. Mol. Cell. Cardiol. 1984, 16, 969–985. [Google Scholar] [CrossRef]
- Dare, A.J.; Bolton, E.A.; Pettigrew, G.J.; Bradley, J.A.; Saeb-Parsy, K.; Murphy, M.P. Protection against renal ischemia-reperfusion injury in vivo by the mitochondria targeted antioxidant mitoq. Redox Biol. 2015, 5, 163–168. [Google Scholar] [CrossRef]
- Stockwell, B.R.; Angeli, J.P.F.; Bayir, H.; Bush, A.I.; Conrad, M.; Dixon, S.J.; Fulda, S.; Gascón, S.; Hatzios, S.K.; Kagan, V.E.; et al. Ferroptosis: A regulated cell death nexus linking metabolism, redox biology, and disease. Cell 2017, 171, 273–285. [Google Scholar] [CrossRef]
- Chen, Y.; Fan, H.; Wang, S.; Tang, G.; Zhai, C.; Shen, L. Ferroptosis: A novel therapeutic target for ischemia-reperfusion injury. Front. Cell. Dev. Biol. 2021, 9, 688605. [Google Scholar] [CrossRef]
- Hankey, G.J. Stroke. Lancet 2017, 389, 641–654. [Google Scholar] [CrossRef]
- Park, U.J.; Lee, Y.A.; Won, S.M.; Lee, J.H.; Kang, S.H.; Springer, J.E.; Lee, Y.B.; Gwag, B.J. Blood-derived iron mediates free radical production and neuronal death in the hippocampal ca1 area following transient forebrain ischemia in rat. Acta Neuropathol. 2011, 121, 459–473. [Google Scholar] [CrossRef]
- Won, S.M.; Lee, J.H.; Park, U.J.; Gwag, J.; Gwag, B.J.; Lee, Y.B. Iron mediates endothelial cell damage and blood-brain barrier opening in the hippocampus after transient forebrain ischemia in rats. Exp. Mol. Med. 2011, 43, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Dávalos, A.; Fernandez-Real, J.M.; Ricart, W.; Soler, S.; Molins, A.; Planas, E.; Genís, D. Iron-related damage in acute ischemic stroke. Stroke 1994, 25, 1543–1546. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, M.; Puig, N.; Carbonell, T.; Castillo, J.; Martinez, J.; Rama, R.; Dávalos, A. Iron intake increases infarct volume after permanent middle cerebral artery occlusion in rats. Brain Res. 2002, 952, 1–6. [Google Scholar] [CrossRef]
- Prass, K.; Ruscher, K.; Karsch, M.; Isaev, N.; Megow, D.; Priller, J.; Scharff, A.; Dirnagl, U.; Meisel, A. Desferrioxamine induces delayed tolerance against cerebral ischemia in vivo and in vitro. J. Cereb. Blood Flow Metab. 2002, 22, 520–525. [Google Scholar] [CrossRef]
- Lei, P.; Ayton, S.; Appukuttan, A.T.; Moon, S.; Duce, J.A.; Volitakis, I.; Cherny, R.; Wood, S.J.; Greenough, M.; Berger, G.; et al. Lithium suppression of tau induces brain iron accumulation and neurodegeneration. Mol. Psychiatry 2017, 22, 396–406. [Google Scholar] [CrossRef]
- Pham, C.G.; Bubici, C.; Zazzeroni, F.; Papa, S.; Jones, J.; Alvarez, K.; Jayawardena, S.; de Smaele, E.; Cong, R.; Beaumont, C.; et al. Ferritin heavy chain upregulation by nf-kappab inhibits tnfalpha-induced apoptosis by suppressing reactive oxygen species. Cell 2004, 119, 529–542. [Google Scholar] [CrossRef]
- Hou, W.; Xie, Y.; Song, X.; Sun, X.; Lotze, M.T.; Zeh, H.J., 3rd; Kang, R.; Tang, D. Autophagy promotes ferroptosis by degradation of ferritin. Autophagy 2016, 12, 1425–1428. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Chang, S.Y.; Wu, Q.; Gou, Y.J.; Jia, L.; Cui, Y.M.; Yu, P.; Shi, Z.H.; Wu, W.S.; Gao, G.; et al. The protective role of mitochondrial ferritin on erastin-induced ferroptosis. Front. Aging Neurosci. 2016, 8, 308. [Google Scholar] [CrossRef]
- Mancias, J.D.; Vaites, L.P.; Nissim, S.; Biancur, D.E.; Kim, A.J.; Wang, X.; Liu, Y.; Goessling, W.; Kimmelman, A.C.; Harper, J.W. Ferritinophagy via ncoa4 is required for erythropoiesis and is regulated by iron dependent herc2-mediated proteolysis. Elife 2015, 4, e10308. [Google Scholar] [CrossRef]
- Mancias, J.D.; Wang, X.; Gygi, S.P.; Harper, J.W.; Kimmelman, A.C. Quantitative proteomics identifies ncoa4 as the cargo receptor mediating ferritinophagy. Nature 2014, 509, 105–109. [Google Scholar] [CrossRef]
- Gao, M.; Monian, P.; Pan, Q.; Zhang, W.; Xiang, J.; Jiang, X. Ferroptosis is an autophagic cell death process. Cell. Res. 2016, 26, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Mugoni, V.; Postel, R.; Catanzaro, V.; de Luca, E.; Turco, E.; Digilio, G.; Silengo, L.; Murphy, M.P.; Medana, C.; Stainier, D.Y.; et al. Ubiad1 is an antioxidant enzyme that regulates enos activity by coq10 synthesis. Cell 2013, 152, 504–518. [Google Scholar] [CrossRef] [PubMed]
- Karuppagounder, S.S.; Alin, L.; Chen, Y.; Brand, D.; Bourassa, M.W.; Dietrich, K.; Wilkinson, C.M.; Nadeau, C.A.; Kumar, A.; Perry, S.; et al. N-acetylcysteine targets 5 lipoxygenase-derived, toxic lipids and can synergize with prostaglandin e(2) to inhibit ferroptosis and improve outcomes following hemorrhagic stroke in mice. Ann. Neurol. 2018, 84, 854–872. [Google Scholar] [CrossRef] [PubMed]
- Shirhan, M.D.; Moochhala, S.M.; Ng, P.Y.; Lu, J.; Ng, K.C.; Teo, A.L.; Yap, E.; Ng, I.; Hwang, P.; Lim, T.; et al. Spermine reduces infarction and neurological deficit following a rat model of middle cerebral artery occlusion: A magnetic resonance imaging study. Neuroscience 2004, 124, 299–304. [Google Scholar] [CrossRef]
- Ou, Y.; Wang, S.J.; Li, D.; Chu, B.; Gu, W. Activation of sat1 engages polyamine metabolism with p53-mediated ferroptotic responses. Proc. Natl. Acad. Sci. USA 2016, 113, E6806–E6812. [Google Scholar] [CrossRef]
- Xiao, X.; Lu, Z.; Lin, V.; May, A.; Shaw, D.H.; Wang, Z.; Che, B.; Tran, K.; Du, H.; Shaw, P.X. Microrna mir-24-3p reduces apoptosis and regulates keap1-nrf2 pathway in mouse cardiomyocytes responding to ischemia/reperfusion injury. Oxid. Med. Cell. Longev. 2018, 2018, 7042105. [Google Scholar] [CrossRef]
- Huang, Z.P.; Ding, Y.; Chen, J.; Wu, G.; Kataoka, M.; Hu, Y.; Yang, J.H.; Liu, J.; Drakos, S.G.; Selzman, C.H.; et al. Long non-coding rnas link extracellular matrix gene expression to ischemic cardiomyopathy. Cardiovasc. Res. 2016, 112, 543–554. [Google Scholar] [CrossRef]
- Dharap, A.; Nakka, V.P.; Vemuganti, R. Effect of focal ischemia on long noncoding rnas. Stroke 2012, 43, 2800–2802. [Google Scholar] [CrossRef]
- Bao, J.; Zhou, S.; Pan, S.; Zhang, Y. Molecular mechanism exploration of ischemic stroke by integrating mrna and mirna expression profiles. Clin. Lab 2018, 64, 559–568. [Google Scholar] [CrossRef]
- Li, W.; Ning, J.Z.; Cheng, F.; Yu, W.M.; Rao, T.; Ruan, Y.; Yuan, R.; Zhang, X.B.; Du, Y.; Xiao, C.C. Malat1 promotes cell apoptosis and suppresses cell proliferation in testicular ischemia-reperfusion injury by sponging mir-214 to modulate trpv4 expression. Cell. Physiol. Biochem. 2018, 46, 802–814. [Google Scholar] [CrossRef]
- Ingold, I.; Berndt, C.; Schmitt, S.; Doll, S.; Poschmann, G.; Buday, K.; Roveri, A.; Peng, X.; Freitas, F.P.; Seibt, T.; et al. Selenium utilization by gpx4 is required to prevent hydroperoxide-induced ferroptosis. Cell 2018, 172, 409–422.e21. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Xu, B.; Xiao, A.; Liu, L.; Fang, X.; Liu, R.; Turlova, E.; Barszczyk, A.; Zhong, X.; Sun, C.L.; et al. Trpm7 inhibitor carvacrol protects brain from neonatal hypoxic-ischemic injury. Mol Brain 2015, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Arrigo, M.; Price, S.; Baran, D.A.; Pöss, J.; Aissaoui, N.; Bayes-Genis, A.; Bonello, L.; François, B.; Gayat, E.; Gilard, M.; et al. Optimising clinical trials in acute myocardial infarction complicated by cardiogenic shock: A statement from the 2020 critical care clinical trialists workshop. Lancet Respir. Med. 2021, 9, 1192–1202. [Google Scholar] [CrossRef]
- Ibáñez, B.; Heusch, G.; Ovize, M.; van de Werf, F. Evolving therapies for myocardial ischemia/reperfusion injury. J. Am. Coll. Cardiol. 2015, 65, 1454–1471. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Y.; Liu, S.Q.; Yao, R.Q.; Tian, Y.P.; Yao, Y.M. A novel insight into the fate of cardiomyocytes in ischemia-reperfusion injury: From iron metabolism to ferroptosis. Front. Cell. Dev. Biol. 2021, 9, 799499. [Google Scholar] [CrossRef] [PubMed]
- Lillo-Moya, J.; Rojas-Solé, C.; Muñoz-Salamanca, D.; Panieri, E.; Saso, L.; Rodrigo, R. Targeting ferroptosis against ischemia/reperfusion cardiac injury. Antioxidants 2021, 10, 667. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Yang, H.; Kong, Q.; Li, J.; Lee, S.M.; Gao, B.; Dong, H.; Wei, J.; Song, J.; Zhang, D.D.; et al. Usp22 antagonizes p53 transcriptional activation by deubiquitinating sirt1 to suppress cell apoptosis and is required for mouse embryonic development. Mol. Cell. 2012, 46, 484–494. [Google Scholar] [CrossRef]
- Lakhal-Littleton, S.; Wolna, M.; Carr, C.A.; Miller, J.J.; Christian, H.C.; Ball, V.; Santos, A.; Diaz, R.; Biggs, D.; Stillion, R.; et al. Cardiac ferroportin regulates cellular iron homeostasis and is important for cardiac function. Proc. Natl. Acad. Sci. USA 2015, 112, 3164–3169. [Google Scholar] [CrossRef]
- Harada, N.; Kanayama, M.; Maruyama, A.; Yoshida, A.; Tazumi, K.; Hosoya, T.; Mimura, J.; Toki, T.; Maher, J.M.; Yamamoto, M.; et al. Nrf2 regulates ferroportin 1-mediated iron efflux and counteracts lipopolysaccharide-induced ferroportin 1 mrna suppression in macrophages. Arch. Biochem. Biophys. 2011, 508, 101–109. [Google Scholar] [CrossRef]
- Zhang, B.; Zhai, M.; Li, B.; Liu, Z.; Li, K.; Jiang, L.; Zhang, M.; Yi, W.; Yang, J.; Yi, D.; et al. Honokiol ameliorates myocardial ischemia/reperfusion injury in type 1 diabetic rats by reducing oxidative stress and apoptosis through activating the sirt1-nrf2 signaling pathway. Oxid. Med. Cell. Longev. 2018, 2018, 3159801. [Google Scholar] [CrossRef]
- Yeang, C.; Hasanally, D.; Que, X.; Hung, M.Y.; Stamenkovic, A.; Chan, D.; Chaudhary, R.; Margulets, V.; Edel, A.L.; Hoshijima, M.; et al. Reduction of myocardial ischaemia-reperfusion injury by inactivating oxidized phospholipids. Cardiovasc. Res. 2019, 115, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Simone, L.E.; Keene, J.D. Mechanisms coordinating elav/hu mrna regulons. Curr. Opin. Genet. Dev. 2013, 23, 35–43. [Google Scholar] [CrossRef] [PubMed]
- de Bruin, R.G.; Rabelink, T.J.; van Zonneveld, A.J.; van der Veer, E.P. Emerging roles for rna-binding proteins as effectors and regulators of cardiovascular disease. Eur. Heart J. 2017, 38, 1380–1388. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yao, Z.; Wang, L.; Ding, H.; Shao, J.; Chen, A.; Zhang, F.; Zheng, S. Activation of ferritinophagy is required for the rna-binding protein elavl1/hur to regulate ferroptosis in hepatic stellate cells. Autophagy 2018, 14, 2083–2103. [Google Scholar] [CrossRef]
- Bao, C.; Lu, Y.; Chen, J.; Chen, D.; Lou, W.; Ding, B.; Xu, L.; Fan, W. Exploring specific prognostic biomarkers in triple-negative breast cancer. Cell. Death Dis. 2019, 10, 807. [Google Scholar] [CrossRef]
- Thomson, D.W.; Dinger, M.E. Endogenous microrna sponges: Evidence and controversy. Nat. Rev. Genet. 2016, 17, 272–283. [Google Scholar] [CrossRef]
- Cai, H.; Yang, X.; Gao, Y.; Xu, Z.; Yu, B.; Xu, T.; Li, X.; Xu, W.; Wang, X.; Hua, L. Exosomal microrna-9-3p secreted from bmscs downregulates esm1 to suppress the development of bladder cancer. Mol. Ther. Nucleic Acids 2019, 18, 787–800. [Google Scholar] [CrossRef]
- Kamat, P.; Vandenberghe, S.; Christen, S.; Bongoni, A.K.; Meier, B.; Rieben, R.; Khattab, A.A. Dexrazoxane shows no protective effect in the acute phase of reperfusion during myocardial infarction in pigs. PLoS ONE 2016, 11, e0168541. [Google Scholar] [CrossRef]
- El-Sharaky, A.S.; Wahby, M.M.; El-Dein, M.M.B.; Fawzy, R.A.; El-Shahawy, I.N. Mutual anti-oxidative effect of gossypol acetic acid and gossypol-iron complex on hepatic lipid peroxidation in male rats. Food Chem. Toxicol. 2009, 47, 2735–2741. [Google Scholar] [CrossRef]
- Reynolds, J.M.; Tone, J.N. Subchronic oral administration of gossypol-acetic acid (gaa) alters the distribution and utilization of radioiron in male rats. Drug Chem. Toxicol. 1988, 11, 135–150. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, N.K.; Lim, H.J.; Mazumder, S.; Rethineswaran, V.K.; Kim, Y.J.; Jang, W.B.; Ji, S.T.; Kang, S.; Kim, D.Y.; et al. Therapeutic cell protective role of histochrome under oxidative stress in human cardiac progenitor cells. Mar. Drugs 2019, 17, 368. [Google Scholar] [CrossRef] [PubMed]
- Anufriev, V.; Novikov, V.L.; Maximov, O.B.; Elyakov, G.B.; Levitsky, D.O.; Lebedev, A.V.; Sadretdinov, S.M.; Shvilkin, A.V.; Afonskaya, N.I.; Ruda, M.; et al. Synthesis of some hydroxynaphthazarins and their cardioprotective effects under ischemia-reperfusion in vivo. Bioorg. Med. Chem. Lett. 1998, 8, 587–592. [Google Scholar] [CrossRef]
- Olivas-Aguirre, F.J.; Rodrigo-García, J.; Martínez-Ruiz, N.D.; Cárdenas-Robles, A.I.; Mendoza-Díaz, S.O.; Álvarez-Parrilla, E.; González-Aguilar, G.A.; de la Rosa, L.A.; Ramos-Jiménez, A.; Wall-Medrano, A. Cyanidin-3-o-glucoside: Physical-chemistry, foodomics and health effects. Molecules 2016, 21, 1264. [Google Scholar] [CrossRef] [PubMed]
- Petroni, K.; Trinei, M.; Fornari, M.; Calvenzani, V.; Marinelli, A.; Micheli, L.A.; Pilu, R.; Matros, A.; Mock, H.P.; Tonelli, C.; et al. Dietary cyanidin 3-glucoside from purple corn ameliorates doxorubicin-induced cardiotoxicity in mice. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 462–469. [Google Scholar] [CrossRef]
- Aloud, B.M.; Raj, P.; McCallum, J.; Kirby, C.; Louis, X.L.; Jahan, F.; Yu, L.; Hiebert, B.; Duhamel, T.A.; Wigle, J.T.; et al. Cyanidin 3-o-glucoside prevents the development of maladaptive cardiac hypertrophy and diastolic heart dysfunction in 20-week-old spontaneously hypertensive rats. Food Funct. 2018, 9, 3466–3480. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhao, Y.; Song, J.; Wang, R.; Gao, L.; Zhang, L.; Fang, L.; Lu, Y.; Du, G. Total flavonoids from anchusa italica retz. Improve cardiac function and attenuate cardiac remodeling post myocardial infarction in mice. J. Ethnopharmacol. 2020, 257, 112887. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Song, X.; Sun, X.; Huang, J.; Zhong, M.; Lotze, M.T.; Zeh, H.J.R.; Kang, R.; Tang, D. Identification of baicalein as a ferroptosis inhibitor by natural product library screening. Biochem. Biophys. Res. Commun. 2016, 473, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, P.J.; Carvalho, D.O.; Cruz, J.M.; Guido, L.F.; Barros, A.A. Fundamentals and health benefits of xanthohumol, a natural product derived from hops and beer. Nat. Prod. Commun. 2009, 4, 591–610. [Google Scholar] [CrossRef] [PubMed]
- Drummond, J.C.; McKay, L.D.; Cole, D.J.; Patel, P.M. The role of nitric oxide synthase inhibition in the adverse effects of etomidate in the setting of focal cerebral ischemia in rats. Anesth. Analg. 2005, 100, 841–846. [Google Scholar] [CrossRef]
- Bakar, E.; Ulucam, E.; Cerkezkayabekir, A.; Sanal, F.; Inan, M. Investigation of the effects of naringin on intestinal ischemia reperfusion model at the ultrastructural and biochemical level. Biomed. Pharmacother. 2019, 109, 345–350. [Google Scholar] [CrossRef]
- Kim, S.G.; Lee, E.; Park, N.Y.; Park, H.H.; Jeong, K.T.; Kim, K.J.; Lee, Y.J.; Jin, M.; Lee, E. Britanin attenuates ovalbumin-induced airway inflammation in a murine asthma model. Arch. Pharm. Res. 2016, 39, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Zhu, L.; Yuan, X.; Chen, H.; Xiong, R.; Zhang, S.; Cheng, H.; Shen, Y.; An, H.; Li, T.; et al. Britanin ameliorates cerebral ischemia-reperfusion injury by inducing the nrf2 protective pathway. Antioxid. Redox. Signal. 2017, 27, 754–768. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Bi, J.; Yang, L.; Zhang, J.; Wan, Y.; Chen, X.; Wang, Y.; Wu, Z.; Lv, Y.; Wu, R. Serum irisin levels are decreased in patients with sepsis, and exogenous irisin suppresses ferroptosis in the liver of septic mice. Clin. Transl. Med. 2020, 10, e173. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Zhao, T.; Song, Y.; Lin, L.; Fan, X.; Cui, B.; Feng, H.; Wang, X.; Yu, Q.; Zhang, J.; et al. The emerging role of ferroptosis in non-cancer liver diseases: Hype or increasing hope? Cell. Death Dis. 2020, 11, 518. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Q.; Gao, W.; Du, F.; Wang, X. Mule/arf-bp1, a bh3-only e3 ubiquitin ligase, catalyzes the polyubiquitination of mcl-1 and regulates apoptosis. Cell 2005, 121, 1085–1095. [Google Scholar] [CrossRef]
- Ghosh, M.C.; Tong, W.H.; Zhang, D.; Ollivierre-Wilson, H.; Singh, A.; Krishna, M.C.; Mitchell, J.B.; Rouault, T.A. Tempol-mediated activation of latent iron regulatory protein activity prevents symptoms of neurodegenerative disease in irp2 knockout mice. Proc. Natl. Acad. Sci. USA 2008, 105, 12028–12033. [Google Scholar] [CrossRef]
- Polimeno, L.; Pesetti, B.; de Santis, F.; Resta, L.; Rossi, R.; de Palma, A.; Girardi, B.; Amoruso, A.; Francavilla, A. Decreased expression of the augmenter of liver regeneration results in increased apoptosis and oxidative damage in human-derived glioma cells. Cell. Death Dis. 2012, 3, e289. [Google Scholar] [CrossRef]
- Jankowski, J.; Perry, H.M.; Medina, C.B.; Huang, L.; Yao, J.; Bajwa, A.; Lorenz, U.M.; Rosin, D.L.; Ravichandran, K.S.; Isakson, B.E.; et al. Epithelial and endothelial pannexin1 channels mediate aki. J. Am. Soc. Nephrol. 2018, 29, 1887–1899. [Google Scholar] [CrossRef]
- Miller, G.; Matthews, S.P.; Reinheckel, T.; Fleming, S.; Watts, C. Asparagine endopeptidase is required for normal kidney physiology and homeostasis. FASEB J. 2011, 25, 1606–1617. [Google Scholar] [CrossRef]
- Mohib, K.; Wang, S.; Guan, Q.; Mellor, A.L.; Sun, H.; Du, C.; Jevnikar, A.M. Indoleamine 2,3-dioxygenase expression promotes renal ischemia-reperfusion injury. Am. J. Physiol.-Renal Physiol. 2008, 295, F226–F234. [Google Scholar] [CrossRef]
- Zou, Y.F.; Wen, D.; Zhao, Q.; Shen, P.Y.; Shi, H.; Zhao, Q.; Chen, Y.X.; Zhang, W. Urinary microrna-30c-5p and microrna-192-5p as potential biomarkers of ischemia-reperfusion-induced kidney injury. Exp. Biol. Med. 2017, 242, 657–667. [Google Scholar] [CrossRef] [PubMed]
- Wilflingseder, J.; Sunzenauer, J.; Toronyi, E.; Heinzel, A.; Kainz, A.; Mayer, B.; Perco, P.; Telkes, G.; Langer, R.M.; Oberbauer, R. Molecular pathogenesis of post-transplant acute kidney injury: Assessment of whole-genome mrna and mirna profiles. PLoS ONE 2014, 9, e104164. [Google Scholar] [CrossRef] [PubMed]
- Wilflingseder, J.; Jelencsics, K.; Bergmeister, H.; Sunzenauer, J.; Regele, H.; Eskandary, F.; Reindl-Schwaighofer, R.; Kainz, A.; Oberbauer, R. Mir-182-5p inhibition ameliorates ischemic acute kidney injury. Am. J. Pathol. 2017, 187, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, S.; Wang, H.; Cao, J.; Huang, X.; Chen, Z.; Xu, P.; Sun, G.; Xu, J.; Lv, J.; et al. Circular rna circnrip1 acts as a microrna-149-5p sponge to promote gastric cancer progression via the akt1/mtor pathway. Mol. Cancer 2019, 18, 20. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Ju, Y.; Dai, X.; Ni, N.; Liu, Y.; Zhang, D.; Gao, H.; Sun, H.; Zhang, J.; Gu, P. Ho-1-mediated ferroptosis as a target for protection against retinal pigment epithelium degeneration. Redox. Biol. 2021, 43, 101971. [Google Scholar] [CrossRef]
- Cai, M.; Ma, Y.; Zhang, W.; Wang, S.; Wang, Y.; Tian, L.; Peng, Z.; Wang, H.; Qingrong, T. Apigenin-7-o-β-d-(-6’’-p-coumaroyl)-glucopyranoside treatment elicits neuroprotective effect against experimental ischemic stroke. Int. J. Biol. Sci. 2016, 12, 42–52. [Google Scholar] [CrossRef][Green Version]
- Efimova, I.; Catanzaro, E.; van der Meeren, L.; Turubanova, V.D.; Hammad, H.; Mishchenko, T.A.; Vedunova, M.V.; Fimognari, C.; Bachert, C.; Coppieters, F.; et al. Vaccination with early ferroptotic cancer cells induces efficient antitumor immunity. J. Immunother. Cancer 2020, 8, e001369. [Google Scholar] [CrossRef]
- Hambright, W.S.; Fonseca, R.S.; Chen, L.; Na, R.; Ran, Q. Ablation of ferroptosis regulator glutathione peroxidase 4 in forebrain neurons promotes cognitive impairment and neurodegeneration. Redox. Biol. 2017, 12, 8–17. [Google Scholar] [CrossRef]
- Kang, R.; Zeng, L.; Zhu, S.; Xie, Y.; Liu, J.; Wen, Q.; Cao, L.; Xie, M.; Ran, Q.; Kroemer, G.; et al. Lipid peroxidation drives gasdermin d-mediated pyroptosis in lethal polymicrobial sepsis. Cell. Host Microbe 2018, 24, 97–108.e4. [Google Scholar] [CrossRef]
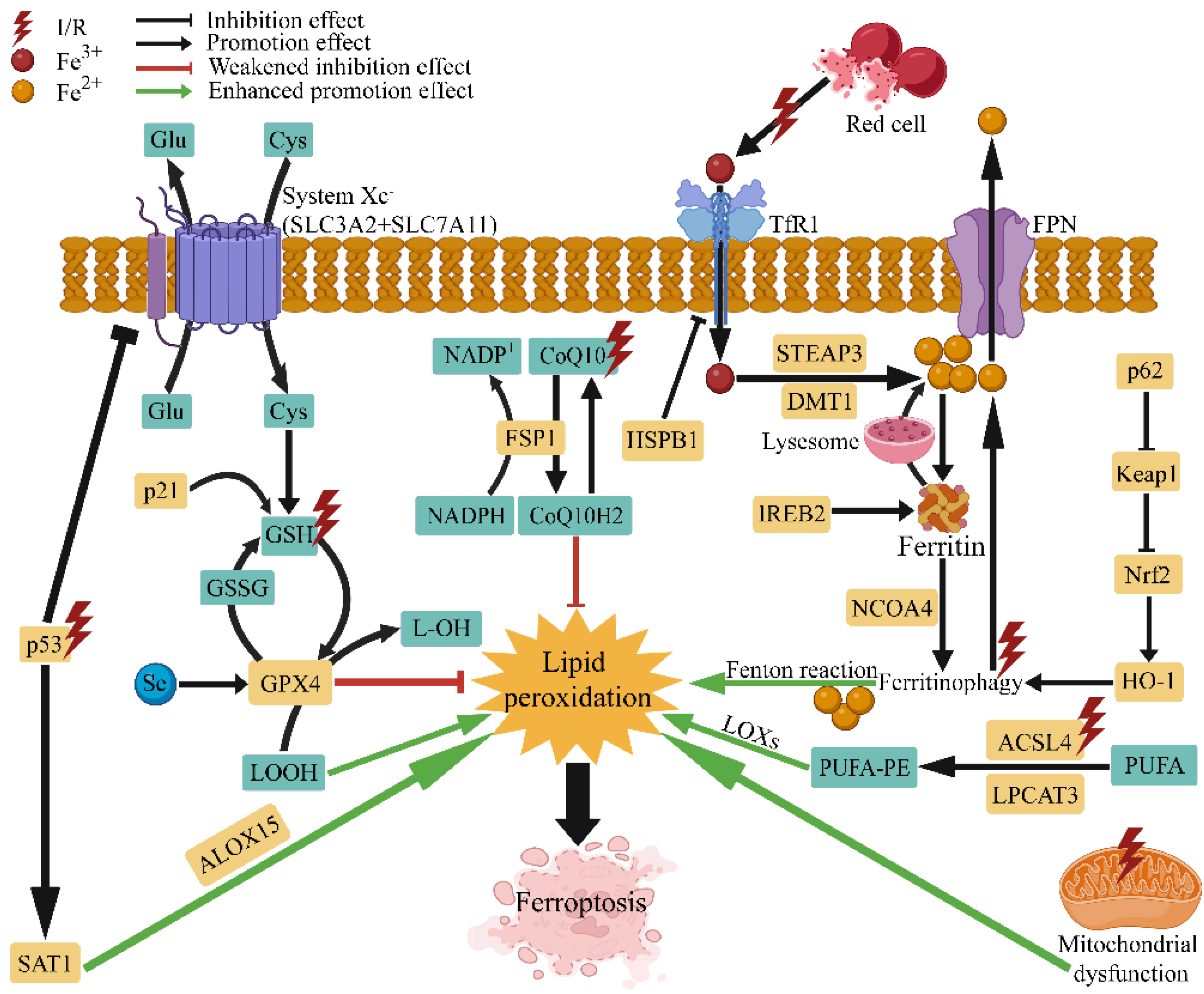
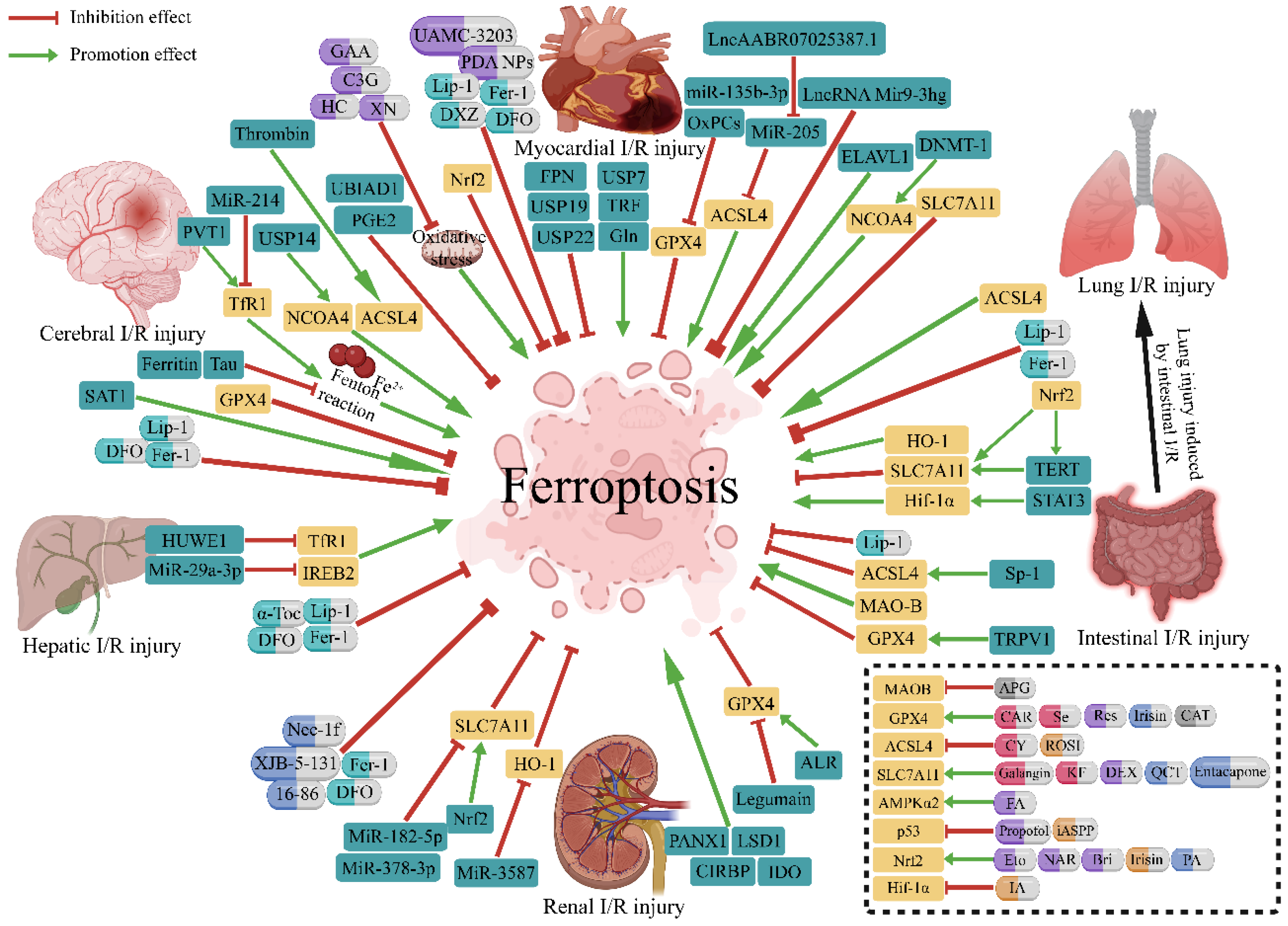
| Therapeutic Targets | Diseases | Model | Key Mechanism | References |
|---|---|---|---|---|
| Tau | Cerebral IRI | In vivo | Tau-iron interaction, inhibit iron overload | [12] |
| Ferritin | Cerebral IRI | In vivo/In vitro | Regulate p53 and SLC7A11 | [13,14] |
| Mitochondrial ferritin | Cerebral IRI | In vivo | Inhibit iron overload, inhibit lipid peroxidation | [15] |
| NCOA4 and USP14 | Cerebral IRI | In vivo/In vitro | Promote ferritinophagy | [14] |
| UBIAD1 | Cerebral IRI | In vivo/In vitro | Inhibit lipid peroxidation | [16] |
| PGE2 | Cerebral IRI | In vivo/Human samples | Inhibit iron accumulation and lipid peroxidation | [17] |
| SAT1 | Cerebral IRI | In vivo/In vitro | Transcriptional target of p53, induce lipid peroxidation | [18] |
| Thrombin | Cerebral IRI | In vivo/In vitro | Instigate esterification of ACSL4 | [19] |
| LncRNA PVT1/miR-214 | Cerebral IRI | In vivo/In vitro | Inhibit TfR1 and p53 | [20] |
| Transferrin and glutamine | Myocardial IRI | In vivo/In vitro | Ferroptosis inducer | [21] |
| USP22 | Myocardial IRI | In vivo/In vitro | Regulate SIRT1/p53/SLC7A11 axis | [22] |
| USP7 | Myocardial IRI | In vivo/In vitro | Upregulate p53/TfR1 pathway | [23] |
| FPN | Myocardial IRI | In vivo/In vitro | Regulate iron homeostasis | [24] |
| DNMT-1 | Myocardial IRI | In vivo/In vitro | Promote NCOA4-mediated ferritinophagy | [25] |
| OxPCs | Myocardial IRI | In vivo/In vitro | Suppress GPX4 activity | [26] |
| ELAVL1 | Myocardial IRI | In vivo/In vitro | Promote autophagic ferroptosis | [27] |
| MiR-135b-3p | Myocardial IRI | In vivo/In vitro | Downregulate GPX4 expression | [28] |
| LncAABR07025387.1 | Myocardial IRI | In vivo/In vitro | Sponge miR-205 to enhance ACSL4 expression | [29] |
| LncRNA Mir9-3hg | Myocardial IRI | In vivo/In vitro | Regulate Pum2/PRDX6 axis | [30] |
| MET | Liver IRI | In vivo/In vitro | Disrupt iron metabolism | [31] |
| HUWE1 | Liver IRI | In vivo/In vitro/Human samples | Target TfR1 for proteasomal degradation | [32] |
| MiR-29a-3p | Liver IRI | In vivo/In vitro | Downregulate IREB2 expression | [33] |
| ALR | Renal IRI | In vitro | Anti-oxidant, upregulate GPX4 expression | [34] |
| Panx1 | Renal IRI | In vivo/In vitro | Regulate HO-1, NCOA4 and FTH1 | [35] |
| CIRBP | Renal IRI | In vivo/In vitro | Regulate ELAVL1 to promote ferritinophagy | [36] |
| Legumain | Renal IRI | In vivo/In vitro | Promote degradation of GPX4 | [37] |
| IDO | Renal IRI | In vitro | Induce AhR-mediated ferroptosis | [38] |
| LSD1 | Renal IRI | In vivo/In vitro | Upregulate TLR4/NOX4 pathway | [39] |
| MiR-182-5p and miR-378-3p | Renal IRI | In vivo/In vitro | Downregulate GPX4 and SLC7A11 expression | [40] |
| MiR-3587 | Renal IRI | In vitro | Downregulate HO-1 expression | [41] |
| Sp1 | Intestinal IRI | In vivo/In vitro/Human samples | Increase ACSL4 transcription | [42] |
| TRPV1 | Intestinal IRI | In vivo/In vitro/Human samples | Upregulate GPX4 expression | [43] |
| Nrf2 | IIR-induced lung injury | In vivo/In vitro | Upregulate SLC7A11-related axis | [44,45,46] |
| p53 | IIR-induced lung injury | In vivo/In vitro | Regulate Nrf2 signaling pathway | [47] |
| Reagents | Diseases | Model | Function | References |
|---|---|---|---|---|
| Selenium compounds | Cerebral IRI | In vivo/In vitro | Drive GPX4 expression | [48,49,50] |
| Carvacrol | Cerebral IRI | In vitro | Upregulate GPX4 expression | [51] |
| Rehmannioside A | Cerebral IRI | In vivo/In vitro/Human samples | Activate SLC7A11/GPX4 axis | [52] |
| Galangin | Cerebral IRI | In vivo/In vitro | Activate SLC7A11/GPX4 axis | [53] |
| Carthamin yellow | Cerebral IRI | In vivo | Inhibit ACSL4 expression | [54] |
| Kaempferol | Cerebral IRI | In vitro | Activate Nrf2/SLC7A11/GPX4 axis | [55] |
| Liproxstatin-1 | Cerebral, myocardial, lung, liver, intestinal IRI | In vivo/In vitro/Human samples | Inhibit lipid peroxidation | [12,42,56,57,58] |
| Ferrostatin-1 | Cerebral, myocardial, lung, liver, renal IRI | In vivo/In vitro/Human samples | Inhibit lipid peroxidation | [12,20,26,31,59,60,61,62] |
| Deferoxamine | Myocardial, liver, renal IRI | In vivo/In vitro | Iron chelator | [21,31,61,62,63] |
| Dexrazoxane | Myocardial IRI | In vivo/In vitro | Iron chelator | [64] |
| Gossypol acetic acid | Myocardial IRI | In vivo/In vitro | Anti-oxidant/iron-chelating | [65] |
| Histochrome | Myocardial IRI | In vivo/In vitro | Anti-oxidant/iron-chelating | [66] |
| Cyanidin-3-glucoside | Myocardial IRI | In vivo/In vitro | Anti-oxidant | [67] |
| Xanthohumol | Myocardial IRI | In vivo/In vitro | Anti-oxidant/upregulate GPX4 expression | [68] |
| Etomidate | Myocardial IRI | In vivo | Upregulate Nrf2/HO-1 pathway | [69] |
| Naringenin | Myocardial IRI | In vivo/In vitro | Upregulate Nrf2/SLC7A11/GPX4 axis | [70] |
| Britanin | Myocardial IRI | In vivo/In vitro | Upregulate Nrf2/GPX4 axis | [71] |
| Propofol | Myocardial IRI | In vivo/In vitro | Regulate AKT/p53 pathway | [72] |
| Ferulic acid | Myocardial IRI | In vivo | Upregulate AMPKα2 expression | [73] |
| PDA NPs | Myocardial IRI | In vivo/In vitro | Inhibit iron deposition and lipid peroxidation | [74] |
| UAMC-3203 | Myocardial IRI | In vivo | Inhibit lipid peroxidation | [75] |
| Resveratrol | Myocardial IRI | In vivo/In vitro | Regulate USP19-Beclin 1 autophagy/upregulate GPX4 | [76] |
| Dexmedetomidine | Myocardial IRI | In vivo | Upregulate SLC7A11/GPX4 axis | [77] |
| Irisin | Lung, renal IRI | In vivo/In vitro | Upregulate Nrf2/HO-1 axis/upregulate GPX4 | [60,78] |
| Rosiglitazone | Lung, intestinal IRI | In vivo/In vitro | Inhibit ACSL4 expression | [42,57] |
| α-tocopherol | Liver IRI | In vivo | Inhibit lipid peroxidation | [61] |
| Pachymic acid | Renal IRI | In vivo | Upregulate Nrf2 signaling pathway | [79] |
| 16–86 | Renal IRI | In vivo/In vitro | Inhibit lipid peroxidation | [80] |
| XJB-5-131 | Renal IRI | In vivo | Inhibit lipid peroxidation/anti-oxidant | [81] |
| Quercetin | Renal IRI | In vivo/In vitro | Inhibit ATF3/SLC7A11/GPX4 axis | [82] |
| Nec-1f | Renal IRI | In vivo/In vitro | Inhibit RIPK1 kinase activity and ferroptosis | [83] |
| Entacapone | Renal IRI | In vivo/In vitro | Upregulate SLC7A11 repression | [84] |
| APG | Intestinal IRI | In vivo/In vitro | Inhibit MAO-B/anti-oxidant | [85] |
| Capsiate | Intestinal IRI | In vivo/In vitro | Enhance GPX4 expression/activate TRPV1 | [43] |
| iASPP | IIR-induced lung injury | In vivo/In vitro | Upregulate Nrf2 signaling/p53 inhibitor | [47] |
| Isoliquiritin apioside | IIR-induced lung injury | In vivo/In vitro | Downregulate Hif-1α expression | [86] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, Y.; Wang, X.; Liu, X.; Shen, L.; Chen, Q.; Shu, Q. Targeting Ferroptosis as a Promising Therapeutic Strategy for Ischemia-Reperfusion Injury. Antioxidants 2022, 11, 2196. https://doi.org/10.3390/antiox11112196
Pan Y, Wang X, Liu X, Shen L, Chen Q, Shu Q. Targeting Ferroptosis as a Promising Therapeutic Strategy for Ischemia-Reperfusion Injury. Antioxidants. 2022; 11(11):2196. https://doi.org/10.3390/antiox11112196
Chicago/Turabian StylePan, Yihang, Xueke Wang, Xiwang Liu, Lihua Shen, Qixing Chen, and Qiang Shu. 2022. "Targeting Ferroptosis as a Promising Therapeutic Strategy for Ischemia-Reperfusion Injury" Antioxidants 11, no. 11: 2196. https://doi.org/10.3390/antiox11112196
APA StylePan, Y., Wang, X., Liu, X., Shen, L., Chen, Q., & Shu, Q. (2022). Targeting Ferroptosis as a Promising Therapeutic Strategy for Ischemia-Reperfusion Injury. Antioxidants, 11(11), 2196. https://doi.org/10.3390/antiox11112196







