Hepatic Homeostasis of Metal Ions Following Acute Repeated Stress Exposure in Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Animal
2.2. Experimental Protocol
2.3. Haptoglobin Enzyme Activity Assay
2.4. Ceruloplasmin Enzyme Activity Assay
2.5. Liver Tissue Preparation
2.6. Circulating and Liver Metal Concentrations
2.6.1. Iron
2.6.2. Copper
2.6.3. Zinc
2.7. mRNA Expression
2.8. miRNA Expression
2.9. Protein Determination
2.10. Data Analysis
3. Results
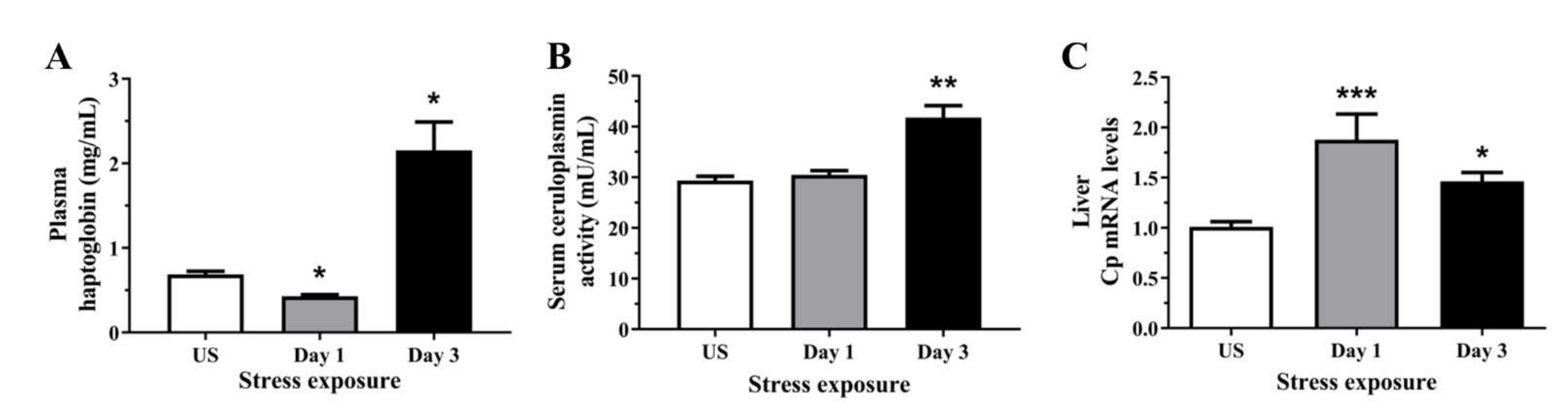
3.1. Stress Increased Both Positive Acute Phase Proteins Including Haptoglobin and Ceruloplasmin Indicative of Liver Inflammation
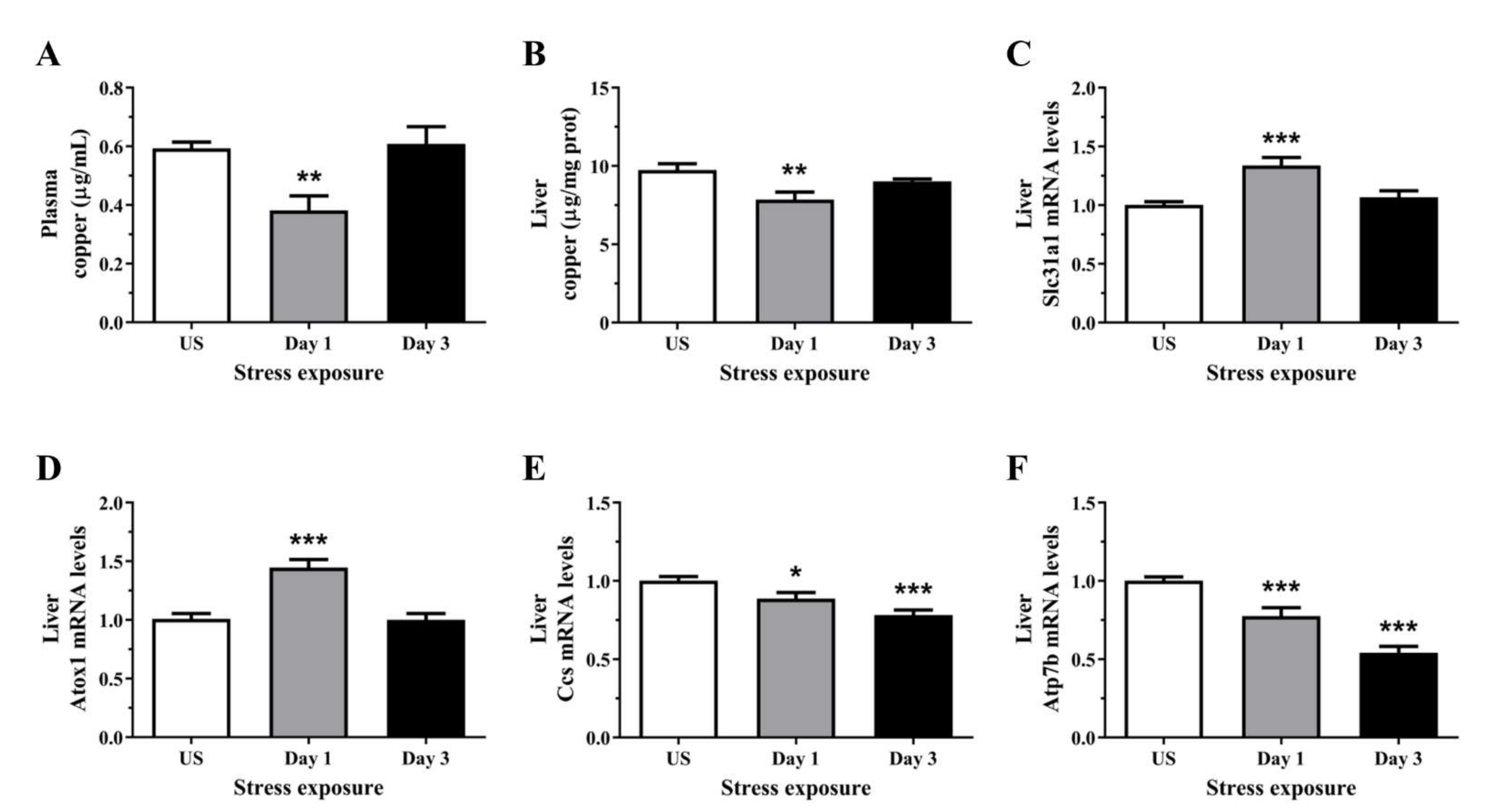
3.2. Acute Stress Decreases Plasma and Hepatic Copper Levels Demonstrating an Alteration in Copper Homeostasis
3.3. Zinc Content Was Reduced in the Plasma but Increase in the Liver Following Repeated Acute Stress
3.4. Acute Stress Leads to an Overall Reduction in Serum and Hepatic Liver Iron Content
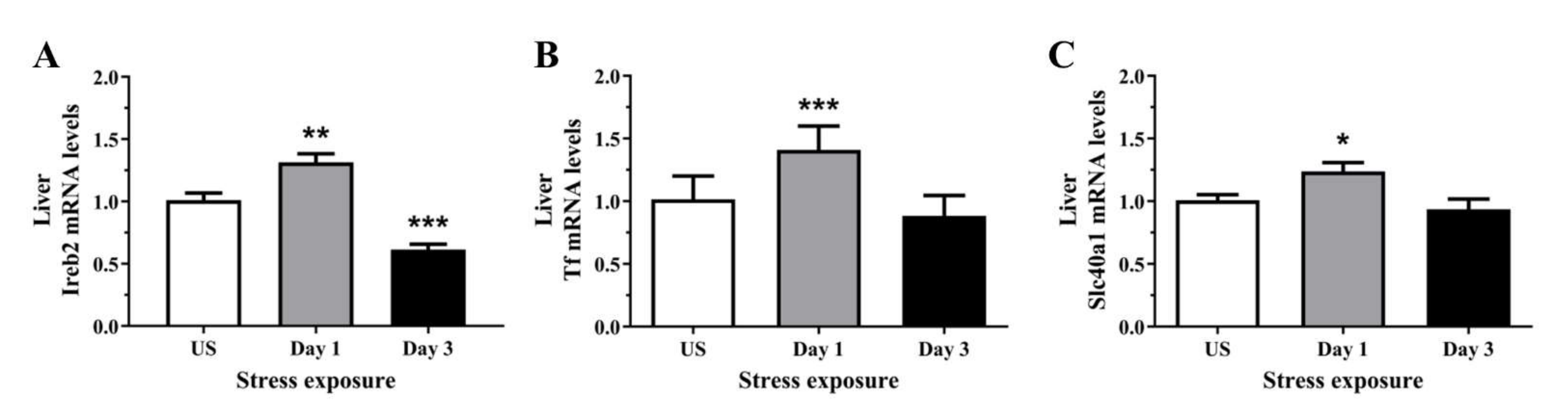
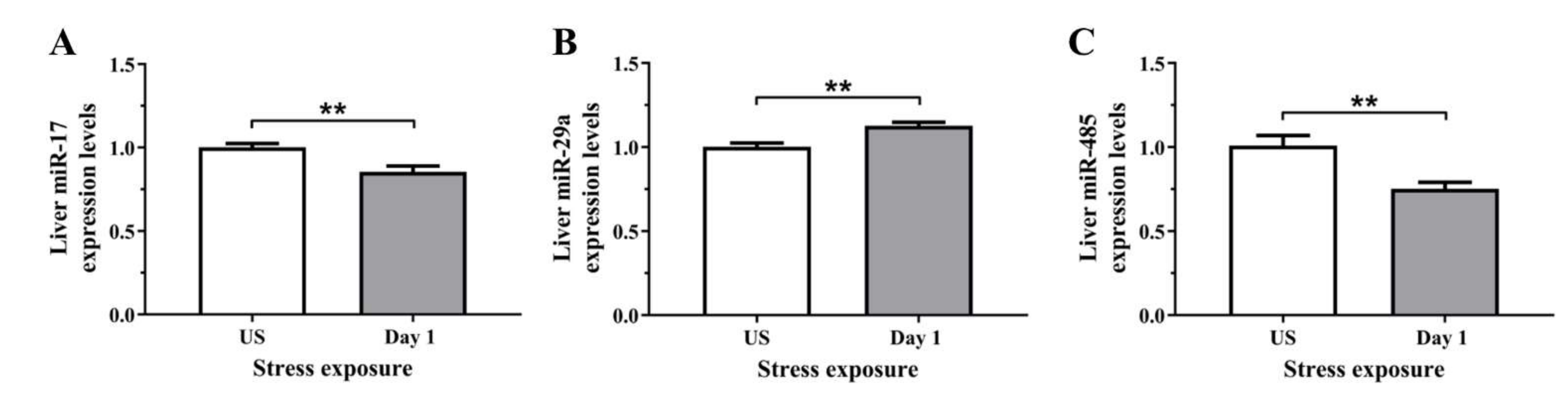
3.5. The Decrease in Serum and Hepatic Iron Content and Upregulation of Iron Transporters Suggests There Is a Redistribution of Iron in Response to Stress
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Acevedo, K.; Masaldan, S.; Opazo, C.M.; Bush, A.I. Redox active metals in neurodegenerative diseases. JBIC J. Biol. Inorg. Chem. 2019, 24, 1141–1157. [Google Scholar] [CrossRef] [PubMed]
- Spasojević, I.; Mojović, M.; Stetvić, Z.; Spasić, S.D.; Jones, D.R.; Morina, A.; Spasić, M.B. Bioavailability and catalytic properties of copper and iron for Fenton chemistry in human cerebrospinal fluid. Redox Rep. 2010, 15, 29–35. [Google Scholar] [CrossRef] [Green Version]
- McCall, K.A.; Huang, C.-C.; Fierke, C.A. Function and mechanism of zinc metalloenzymes. J. Nutr. 2000, 130 (Suppl. S5), 1437s–1446s. [Google Scholar] [CrossRef] [Green Version]
- Maret, W. The redox biology of redox-inert zinc ions. Free Radic. Biol. Med. 2019, 134, 311–326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Linder, M.C. Copper homeostasis in mammals, with emphasis on secretion and excretion. A review. Int. J. Mol. Sci. 2020, 21, 4932. [Google Scholar] [CrossRef]
- Skopp, A.; Boyd, S.D.; Ullrich, M.S.; Liu, L.; Winkler, D.D. Copper-zinc superoxide dismutase (Sod1) activation terminates interaction between its copper chaperone (Ccs) and the cytosolic metal-binding domain of the copper importer Ctr1. Biometals 2019, 32, 695–705. [Google Scholar] [CrossRef] [Green Version]
- Hatori, Y.; Lutsenko, S. The role of copper chaperone Atox1 in coupling redox homeostasis to intracellular copper distribution. Antioxidants 2016, 5, 25. [Google Scholar] [CrossRef] [Green Version]
- Lutsenko, S.; Barnes, N.L.; Bartee, M.Y.; Dmitriev, O.Y. Function and regulation of human copper-transporting ATPases. Physiol. Rev. 2007, 87, 1011–1046. [Google Scholar] [CrossRef] [PubMed]
- Bonaccorsi di Patti, M.C.; Cutone, A.; Polticelli, F.; Rosa, L.; Lepanto, M.S.; Valenti, P.; Musci, G. The ferroportin-ceruloplasmin system and the mammalian iron homeostasis machine: Regulatory pathways and the role of lactoferrin. Biometals 2018, 31, 399–414. [Google Scholar] [CrossRef]
- Hentze, M.W.; Muckenthaler, M.U.; Galy, B.; Camaschella, C. Two to tango: Regulation of Mammalian iron metabolism. Cell 2010, 142, 24–38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilkinson, N.; Pantopoulos, K. The IRP/IRE system in vivo: Insights from mouse models. Front. Pharmacol. 2014, 5, 176. [Google Scholar] [CrossRef] [Green Version]
- Rouault, T.A. The role of iron regulatory proteins in mammalian iron homeostasis and disease. Nat. Chem. Biol. 2006, 2, 406–414. [Google Scholar] [CrossRef]
- Nemeth, E.; Ganz, T. Hepcidin-ferroportin interaction controls systemic iron homeostasis. Int. J. Mol. Sci. 2021, 22, 6493. [Google Scholar] [CrossRef] [PubMed]
- Sangokoya, C.; Doss, J.F.; Chi, J.T. Iron-responsive miR-485-3p regulates cellular iron homeostasis by targeting ferroportin. PLoS Genet. 2013, 9, e1003408. [Google Scholar] [CrossRef]
- Kong, Y.; Hu, L.; Lu, K.; Wang, Y.; Xie, Y.; Gao, L.; Yang, G.; Xie, B.; He, W.; Chen, G.; et al. Ferroportin downregulation promotes cell proliferation by modulating the Nrf2-miR-17-5p axis in multiple myeloma. Cell Death Dis. 2019, 10, 624. [Google Scholar] [CrossRef]
- Lim, D.; Kim, K.S.; Jeong, J.-H.; Marques, O.; Kim, H.-J.; Song, M.; Lee, T.-H.; Kim, J.I.; Choi, H.-S.; Min, J.-J.; et al. The hepcidin-ferroportin axis controls the iron content of Salmonella-containing vacuoles in macrophages. Nat. Commun. 2018, 9, 2091. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.-J.C.; Yip, T.; Lee, J.K.; Juliani, J.; Sernia, C.; Hill, A.F.; Lavidis, N.A.; Spiers, J.G. Restraint stress alters expression of glucocorticoid bioavailability mediators, suppresses Nrf2, and promotes oxidative stress in liver tissue. Antioxidants 2020, 9, 853. [Google Scholar] [CrossRef] [PubMed]
- Spiers, J.G.; Steiger, N.; Khadka, A.; Juliani, J.; Hill, A.F.; Lavidis, N.A.; Anderson, S.T.; Chen, H.-J.C. Repeated acute stress modulates hepatic inflammation and markers of macrophage polarisation in the rat. Biochimie 2021, 180, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Spahl, D.U.; Berendji-Grun, D.; Suschek, C.V.; Kolb-Bachofen, V.; Kroncke, K.-D. Regulation of zinc homeostasis by inducible NO synthase-derived NO: Nuclear metallothionein translocation and intranuclear Zn2+ release. Proc. Natl. Acad. Sci. USA 2003, 100, 13952–13957. [Google Scholar] [CrossRef] [Green Version]
- Cortese-Krott, M.M.; Kulakov, L.; Opländer, C.; Kolb-Bachofen, V.; Kröncke, K.D.; Suschek, C.V. Zinc regulates iNOS-derived nitric oxide formation in endothelial cells. Redox Biol. 2014, 2, 945–954. [Google Scholar] [CrossRef] [Green Version]
- Krężel, A.; Maret, W. The bioinorganic chemistry of mammalian metallothioneins. Chem. Rev. 2021, 121, 14594–14648. [Google Scholar] [CrossRef]
- Chasapis, C.T.; Ntoupa, P.-S.A.; Spiliopoulou, C.A.; Stefanidou, M.E. Recent aspects of the effects of zinc on human health. Arch. Toxicol. 2020, 94, 1443–1460. [Google Scholar] [CrossRef]
- Troadec, M.B.; Ward, D.M.; Lo, E.; Kaplan, J.; De Domenico, I. Induction of FPN1 transcription by MTF-1 reveals a role for ferroportin in transition metal efflux. Blood 2010, 116, 4657–4664. [Google Scholar] [CrossRef] [Green Version]
- Günther, V.; Lindert, U.; Schaffner, W. The taste of heavy metals: Gene regulation by MTF-1. Biochim. Et. Biophys. Acta (BBA)-Mol. Cell Res. 2012, 1823, 1416–1425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qu, W.; Waalkes, M.P. Metallothionein blocks oxidative DNA damage induced by acute inorganic arsenic exposure. Toxicol. Appl. Pharmacol. 2015, 282, 267–274. [Google Scholar] [CrossRef] [Green Version]
- Dubé, A.; Harrisson, J.-F.; Saint-Gelais, G.; Séguin, C. Hypoxia acts through multiple signaling pathways to induce metallothionein transactivation by the metal-responsive transcription factor-1 (MTF-1). Biochem. Cell Biol. 2011, 89, 562–577. [Google Scholar] [CrossRef] [Green Version]
- Ohashi, T.; Matsui, T.; Chujo, M.; Nagao, M. Restraint stress up-regulates expression of zinc transporter Zip14 mRNA in mouse liver. Cytotechnology 2008, 57, 181–185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sahin, E.; Gümüşlü, S. Stress-dependent induction of protein oxidation, lipid peroxidation and anti-oxidants in peripheral tissues of rats: Comparison of three stress models (immobilization, cold and immobilization-cold). Clin. Exp. Pharmacol. Physiol. 2007, 34, 425–431. [Google Scholar] [CrossRef]
- Spiers, J.G.; Chen, H.-J.C.; Bradley, A.; Anderson, S.; Sernia, C.; Lavidis, N.A. Acute restraint stress induces rapid and prolonged changes in erythrocyte and hippocampal redox status. Psychoneuroendocrinology 2013, 38, 2511–2519. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-J.C.; Lee, J.K.; Yip, T.; Sernia, C.; Lavidis, N.A.; Spiers, J.G. Sub-acute restraint stress progressively increases oxidative/nitrosative stress and inflammatory markers while transiently upregulating antioxidant gene expression in the rat hippocampus. Free Radic. Biol. Med. 2019, 130, 446–457. [Google Scholar] [CrossRef] [Green Version]
- Black, P.H. Stress and the inflammatory response: A review of neurogenic inflammation. Brain Behav. Immun. 2002, 16, 622–653. [Google Scholar] [CrossRef]
- Arambasić, J.; Poznanović, G.; Ivanović-Matić, S.; Bogojević, D.; Mihailović, M.; Uskoković, A.; Grigorov, I. Association of the glucocorticoid receptor with STAT3, C/EBPbeta, and the hormone-responsive element within the rat haptoglobin gene promoter during the acute phase response. IUBMB Life 2010, 62, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Baumann, H.; Prowse, K.R.; Marinković, S.; Won, K.-A.; Jahreis, G.P. Stimulation of hepatic acute phase response by cytokines and glucocorticoids. Ann. N. Y. Acad. Sci. 1989, 557, 280–295, discussion 295–296. [Google Scholar] [CrossRef] [PubMed]
- Schaer, D.J.; Vinchi, F.; Ingoglia, G.; Tolosano, E.; Buehler, P.W. Haptoglobin, hemopexin, and related defense pathways-basic science, clinical perspectives, and drug development. Front. Physiol. 2014, 5, 415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heo, J.; Kattesh, H.G.; Roberts, M.P.; Morrow, J.L.; Dailey, J.W.; Saxton, A.M. Hepatic corticosteroid-binding globulin (CBG) messenger RNA expression and plasma CBG concentrations in young pigs in response to heat and social stress. J. Anim. Sci. 2005, 83, 208–215. [Google Scholar] [CrossRef]
- Sauerwein, H.; Schmitz, S.; Hiss, S. The acute phase protein haptoglobin and its relation to oxidative status in piglets undergoing weaning-induced stress. Redox Rep. 2005, 10, 295–302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jelena, A.; Mirjana, M.; Desanka, B.; Svetlana, I.-M.; Aleksandra, U.; Goran, P.; Ilijana, G. Haptoglobin and the inflammatory and oxidative status in experimental diabetic rats: Antioxidant role of haptoglobin. J. Physiol. Biochem. 2013, 69, 45–58. [Google Scholar] [CrossRef]
- Yalamanoglu, A.; Deuel, J.; Hunt, R.C.; Baek, J.H.; Hassell, K.; Redinius, K.; Irwin, D.C.; Schaer, D.J.; Buehler, P.W. Depletion of haptoglobin and hemopexin promote hemoglobin-mediated lipoprotein oxidation in sickle cell disease. Am. J. Physiol. Lung Cell Mol. Physiol. 2018, 315, L765–L774. [Google Scholar] [CrossRef] [Green Version]
- Levy, A.P.; Asleh, R.; Blum, S.; Levy, N.S.; Miller-Lotan, R.; Kalet-Litman, S.; Anbinder, Y.; Lache, O.; Nakhoul, F.M.; Asaf, R.; et al. Haptoglobin: Basic and clinical aspects. Antioxid. Redox Signal. 2010, 12, 293–304. [Google Scholar] [CrossRef]
- Conley, L.; Geurs, T.L.; Levin, L.A. Transcriptional regulation of ceruloplasmin by an IL-6 response element pathway. Mol. Brain Res. 2005, 139, 235–241. [Google Scholar] [CrossRef]
- Ramadori, G.; Van Damme, J.; Rieder, H.; Zum Büschenfelde, K.H.M. Interleukin 6, the third mediator of acute-phase reaction, modulates hepatic protein synthesis in human and mouse. Comparison with interleukin 1 beta and tumor necrosis factor-alpha. Eur. J. Immunol. 1988, 18, 1259–1264. [Google Scholar] [CrossRef]
- Shiva, S.; Wang, X.; A Ringwood, L.; Xu, X.; Yuditskaya, S.; Annavajjhala, V.; Miyajima, H.; Hogg, N.; Harris, Z.L.; Gladwin, M.T. Ceruloplasmin is a NO oxidase and nitrite synthase that determines endocrine NO homeostasis. Nat. Chem. Biol. 2006, 2, 486–493. [Google Scholar] [CrossRef]
- Vasilyev, V.B. Looking for a partner: Ceruloplasmin in protein-protein interactions. Biometals 2019, 32, 195–210. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Pin, S.; Gathinji, M.; Fuchs, R.; Harris, Z.L. Aceruloplasminemia: An inherited neurodegenerative disease with impairment of iron homeostasis. Ann. N. Y. Acad. Sci. 2004, 1012, 299–305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gitlin, J.D.; Schroeder, J.J.; Lee-Ambrose, L.M.; Cousins, R.J. Mechanisms of caeruloplasmin biosynthesis in normal and copper-deficient rats. Biochem. J. 1992, 282 Pt 3, 835–839. [Google Scholar] [CrossRef] [Green Version]
- Linder, M.C. Apoceruloplasmin: Abundance, detection, formation, and metabolism. Biomedicines 2021, 9, 233. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Agui, T.; Suzuki, Y.; Sato, M.; Matsumoto, K. Inhibition of the copper incorporation into ceruloplasmin leads to the deficiency in serum ceruloplasmin activity in Long-Evans cinnamon mutant rat. J. Biol. Chem. 1993, 268, 8965–8971. [Google Scholar] [CrossRef]
- Karakoç, Y.; Yurdakos, E.; Gulyasar, T.; Mengi, M.; Barutcu, U.B. Experimental stress-induced changes in trace element levels of various tissues in rats. J. Trace Elem. Exp. Med. 2003, 16, 55–60. [Google Scholar] [CrossRef]
- Scheiber, I.F.; Mercer, J.F.; Dringen, R. Metabolism and functions of copper in brain. Prog. Neurobiol. 2014, 116, 33–57. [Google Scholar] [CrossRef]
- Polishchuk, R.S.; Polishchuk, E.V. From and to the Golgi—Defining the Wilson disease protein road map. FEBS Lett. 2019, 593, 2341–2350. [Google Scholar] [CrossRef] [Green Version]
- Stewart, D.J.; Short, K.K.; Maniaci, B.; Burkhead, J.L. COMMD1 and PtdIns(4,5)P(2) interaction maintain ATP7B copper transporter trafficking fidelity in HepG2 cells. J. Cell Sci. 2019, 132, jcs231753. [Google Scholar] [CrossRef]
- Harada, M.; Sakisaka, S.; Terada, K.; Kimura, R.; Kawaguchi, T.; Koga, H.; Taniguchi, E.; Sasatomi, K.; Miura, N.; Suganuma, T.; et al. Role of ATP7B in biliary copper excretion in a human hepatoma cell line and normal rat hepatocytes. Gastroenterology 2000, 118, 921–928. [Google Scholar] [CrossRef]
- Kim, H.; Son, H.-Y.; Bailey, S.M.; Lee, J. Deletion of hepatic Ctr1 reveals its function in copper acquisition and compensatory mechanisms for copper homeostasis. Am. J. Physiol.-Gastrointest. Liver Physiol. 2009, 296, G356–G364. [Google Scholar] [CrossRef] [Green Version]
- Carroll, M.C.; Girouard, J.B.; Ulloa, J.L.; Subramaniam, J.; Wong, P.C.; Valentine, J.S.; Culotta, V.C. Mechanisms for activating Cu- and Zn-containing superoxide dismutase in the absence of the CCS Cu chaperone. Proc. Natl. Acad. Sci. USA 2004, 101, 5964–5969. [Google Scholar] [CrossRef] [Green Version]
- Tao, L.; Zheng, Y.; Shen, Z.; Li, Y.; Tian, X.; Dou, X.; Qian, J.; Shen, H. Psychological stress-induced lower serum zinc and zinc redistribution in rats. Biol. Trace Elem. Res. 2013, 155, 65–71. [Google Scholar] [CrossRef]
- Tian, X.; Zheng, Y.; Li, Y.; Shen, Z.; Tao, L.; Dou, X.; Qian, J.; Shen, H. Psychological stress induced zinc accumulation and up-regulation of ZIP14 and metallothionein in rat liver. BMC Gastroenterol. 2014, 14, 32. [Google Scholar] [CrossRef] [Green Version]
- Taylor, J.A.; Simons, T.J. Effect of dexamethasone on zinc transport in rat hepatocytes in primary culture. Biochim. Et. Biophys. Acta (BBA)-Biomembr. 1994, 1193, 240–246. [Google Scholar] [CrossRef]
- Russo, A.J. Decreased zinc and increased copper in individuals with anxiety. Nutr. Metab. Insights 2011, 4, 1–5. [Google Scholar] [CrossRef]
- Maserejian, N.N.; Hall, S.A.; McKinlay, J.B. Low dietary or supplemental zinc is associated with depression symptoms among women, but not men, in a population-based epidemiological survey. J. Affect. Disord. 2012, 136, 781–788. [Google Scholar] [CrossRef] [Green Version]
- Anbari-Nogyni, Z.; Bidaki, R.; Madadizadeh, F.; Sangsefidi, Z.S.; Fallahzadeh, H.; Karimi-Nazari, E.; Nadjarzadeh, A. Relationship of zinc status with depression and anxiety among elderly population. Clin. Nutr. ESPEN 2020, 37, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Andrews, G.K. Cellular zinc sensors: MTF-1 regulation of gene expression. Biometals 2001, 14, 223–237. [Google Scholar] [CrossRef]
- Ghoshal, K.; Wang, Y.; Sheridan, J.; Jacob, S.T. Metallothionein induction in response to restraint stress. Transcriptional control, adaptation to stress, and role of glucocorticoid. J. Biol. Chem. 1998, 273, 27904–27910. [Google Scholar] [CrossRef] [Green Version]
- Hidalgo, J.; Campmany, L.; Borras, M.; Garvey, J.S.; Armario, A. Metallothionein response to stress in rats: Role in free radical scavenging. Am. J. Physiol.-Endocrinol. Metab. 1988, 255 Pt 1, E518–E524. [Google Scholar] [CrossRef] [PubMed]
- Chasapis, C.T.; Loutsidou, A.C.; Spiliopoulou, C.A.; Stefanidou, M.E. Zinc and human health: An update. Arch. Toxicol. 2012, 86, 521–534. [Google Scholar] [CrossRef]
- Prasad, A.S. Zinc is an antioxidant and anti-inflammatory agent: Its role in human health. Front. Nutr. 2014, 1, 14. [Google Scholar] [CrossRef] [Green Version]
- Liuzzi, J.P.; Lichten, L.A.; Rivera, S.; Blanchard, R.K.; Aydemir, T.B.; Knutson, M.D.; Ganz, T.; Cousins, R.J. Interleukin-6 regulates the zinc transporter Zip14 in liver and contributes to the hypozincemia of the acute-phase response. Proc. Natl. Acad. Sci. USA 2005, 102, 6843–6848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, M.H.; Aydemir, T.B.; Kim, J.; Cousins, R.J. Hepatic ZIP14-mediated zinc transport is required for adaptation to endoplasmic reticulum stress. Proc. Natl. Acad. Sci. USA 2017, 114, E5805–E5814. [Google Scholar] [CrossRef] [Green Version]
- Zhao, M.; Chen, J.; Wang, W.; Wang, L.; Ma, L.; Shen, H.; Li, M. Psychological stress induces hypoferremia through the IL-6-hepcidin axis in rats. Biochem. Biophys. Res. Commun. 2008, 373, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Yang, C.; Jiang, S.; Ni, Y.; Zhao, R.; Ma, W. Repeated Restraint Stress Enhances Hepatic TFR2 Expression and Induces Hepatic Iron Accumulation in Rats. Biol. Trace Elem. Res. 2020, 196, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Ripa, R.; Dolfi, L.; Terrigno, M.; Pandolfini, L.; Savino, A.; Arcucci, V.; Groth, M.; Tozzini, E.T.; Baumgart, M.; Cellerino, A. MicroRNA miR-29 controls a compensatory response to limit neuronal iron accumulation during adult life and aging. BMC Biol. 2017, 15, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Gene Name | Gene Symbol | TaqMan Assay ID | |
|---|---|---|---|
| Copper | Ceruloplasmin | Cp | Rn00561049_m1 |
| Copper transporter 1 (Ctr1) | Slc31a1 | Rn00683634_m1 | |
| Antioxidant 1 copper chaperone | Atox1 | Rn00584459_m1 | |
| Copper transporting ATPase 7 beta-polypeptide | Atp7b | Rn00560862_m1 | |
| Copper chaperone for SOD | Ccs | Rn00584772_m1 | |
| Zinc | Metallothionein 1a | Mt1a | Rn00821759_g1 |
| Zrt- and Irt-like protein 14 (Zip14) | Slc39a14 | Rn01468336_m1 | |
| Zinc transporter 1 (Znt1) | Slc30a1 | Rn00575737_m1 | |
| Metal-regulatory transcription factor 1 | Mtf1 | Rn01749440_m1 | |
| Iron | Iron responsive element binding protein 2 (Irp2) | Ireb2 | Rn00575852_m1 |
| Transferrin | Tf | Rn01445482_m1 | |
| Ferroportin (Fpn1) | Slc40a1 | Rn00591187_m1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spiers, J.G.; Tan, L.S.; Anderson, S.T.; Hill, A.F.; Lavidis, N.A.; Chen, H.-J.C. Hepatic Homeostasis of Metal Ions Following Acute Repeated Stress Exposure in Rats. Antioxidants 2022, 11, 85. https://doi.org/10.3390/antiox11010085
Spiers JG, Tan LS, Anderson ST, Hill AF, Lavidis NA, Chen H-JC. Hepatic Homeostasis of Metal Ions Following Acute Repeated Stress Exposure in Rats. Antioxidants. 2022; 11(1):85. https://doi.org/10.3390/antiox11010085
Chicago/Turabian StyleSpiers, Jereme G., Li Si Tan, Stephen T. Anderson, Andrew F. Hill, Nickolas A. Lavidis, and Hsiao-Jou Cortina Chen. 2022. "Hepatic Homeostasis of Metal Ions Following Acute Repeated Stress Exposure in Rats" Antioxidants 11, no. 1: 85. https://doi.org/10.3390/antiox11010085
APA StyleSpiers, J. G., Tan, L. S., Anderson, S. T., Hill, A. F., Lavidis, N. A., & Chen, H.-J. C. (2022). Hepatic Homeostasis of Metal Ions Following Acute Repeated Stress Exposure in Rats. Antioxidants, 11(1), 85. https://doi.org/10.3390/antiox11010085








