Therapeutic Potential of Alpha-Lipoic Acid in Viral Infections, including COVID-19
Abstract
1. Introduction: Oxidative Stress in Physiology and Pathophysiology
2. The Redox System in Infectious Diseases and Viral Infections
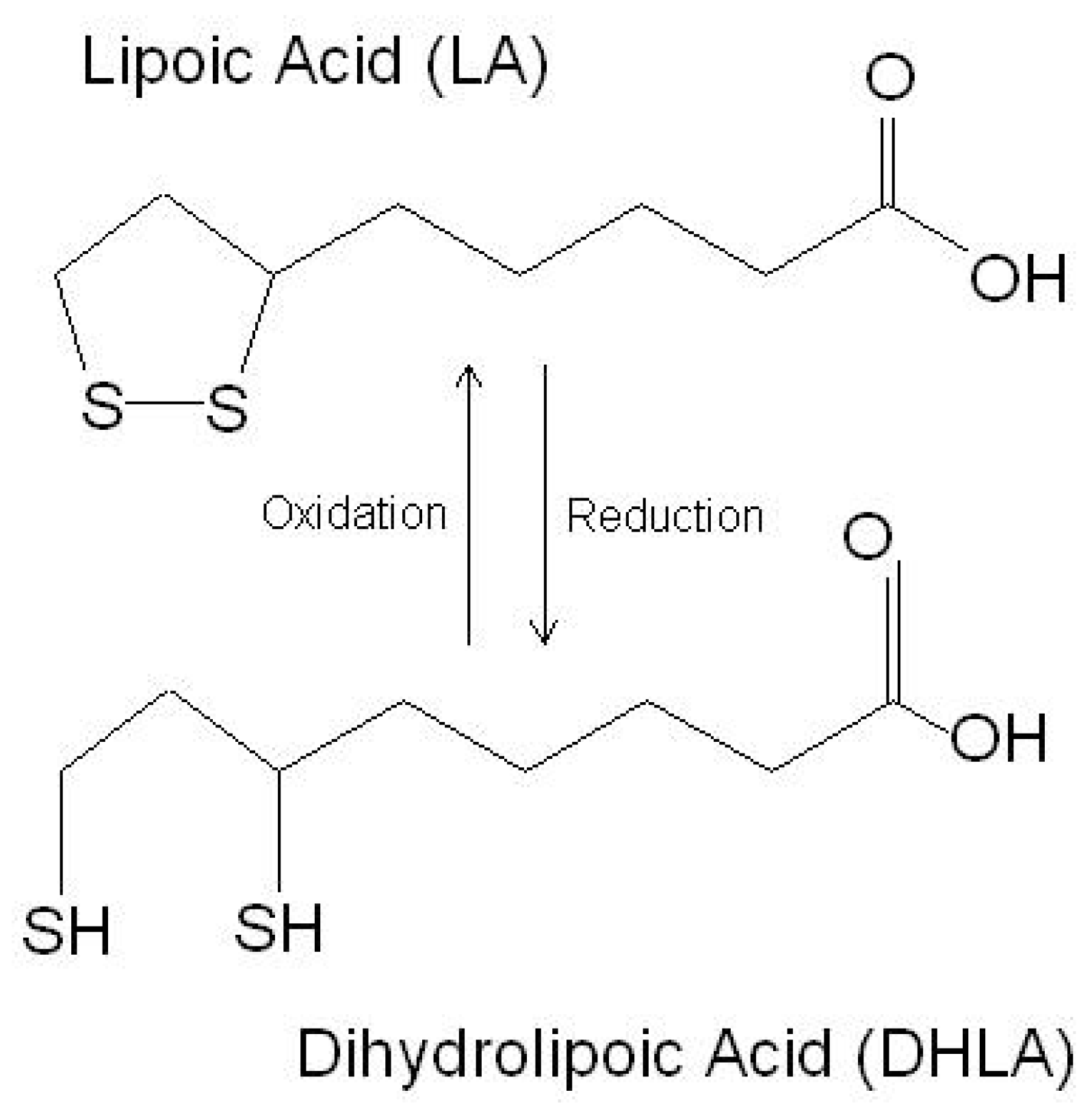
3. Beneficial Effects of LA
4. Antioxidant Supplementation for the Treatment of Viral Infections
5. Effects of LA in Viral Infection
5.1. Effects of LA on Influenza Virus Infection
5.2. Effects of LA in Herpes Infections
5.3. Effects of LA in Smallpox Infection
5.4. Effects of LA in Viral Hepatitis
5.5. Effects of LA in HIV Infection
5.6. Effects of LA in COVID-19
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxid. Med. Cell. Longev. 2016, 2016, 1245049. [Google Scholar] [CrossRef] [PubMed]
- Zarkovic, N. Roles and Functions of ROS and RNS in Cellular Physiology and Pathology. Cells 2020, 9, 767. [Google Scholar] [CrossRef] [PubMed]
- Khomich, O.A.; Kochetkov, S.N.; Bartosch, B.; Ivanov, A.V. Redox biology of respiratory viral infections. Viruses 2018, 10, 392. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.; Kumar, S.; Simon, S.D.; Singh, D.P.; Kumar, A. HIV gp120- and methamphetamine-mediated oxidative stress induces astrocyte apoptosis via cytochrome P450 2E1. Cell Death Dis. 2013, 4, e850. [Google Scholar] [CrossRef]
- Checconi, P.; De Angelis, M.; Marcocci, M.E.; Fraternale, A.; Magnani, M.; Palamara, A.T.; Nencioni, L. Redox-modulating agents in the treatment of viral infections. Int. J. Mol. Sci. 2020, 21, 4084. [Google Scholar] [CrossRef]
- Forman, H.J. Glutathione—From antioxidant to post-translational modifier. Arch. Biochem. Biophys. 2016, 595, 64–67. [Google Scholar] [CrossRef]
- Diotallevi, M.; Checconi, P.; Palamara, A.T.; Celestino, I.; Coppo, L.; Holmgren, A.; Abbas, K.; Peyrot, F.; Mengozzi, M.; Ghezzi, P. Glutathione Fine-Tunes the innate immune response toward antiviral pathways in a macrophage cell line independently of its antioxidant properties. Front. Immunol. 2017, 8, 1239. [Google Scholar] [CrossRef]
- Checconi, P.; Limongi, D.; Baldelli, S.; Ciriolo, M.R.; Nencioni, L.; Palamara, A.T. Role of glutathionylation in infection and inflammation. Nutrients 2019, 11, 1952. [Google Scholar] [CrossRef]
- Kosmider, B.; Messier, E.M.; Janssen, W.J.; Nahreini, P.; Wang, J.; Hartshorn, K.L.; Mason, R.J. Nrf2 protects human alveolar epithelial cells against injury induced by influenza A virus. Respir. Res. 2012, 13, 43. [Google Scholar] [CrossRef]
- Ivanov, A.V.; Smirnova, O.A.; Ivanova, O.N.; Masalova, O.V.; Kochetkov, S.N.; Isaguliants, M.G. Hepatitis C virus proteins activate NRF2/ARE pathway by distinct ROS-dependent and independent mechanisms in HUH7 cells. PLoS ONE 2011, 6, e24957. [Google Scholar] [CrossRef]
- Ivanov, A.V.; Smirnova, O.A.; Petrushanko, I.Y.; Ivanova, O.N.; Karpenko, I.L.; Alekseeva, E.; Sominskaya, I.; Makarov, A.A.; Bartosch, B.; Kochetkov, S.N.; et al. HCV core protein uses multiple mechanisms to induce oxidative stress in human hepatoma huh7 cells. Viruses 2015, 7, 2745–2770. [Google Scholar] [CrossRef]
- Medvedev, R.; Ploen, D.; Spengler, C.; Elgner, F.; Ren, H.; Bunten, S.; Hildt, E. HCV-induced oxidative stress by inhibition of Nrf2 triggers autophagy and favors release of viral particles. Free Radic. Biol. Med. 2017, 110, 300–315. [Google Scholar] [CrossRef]
- Anticoli, S.; Amatore, D.; Matarrese, P.; De Angelis, M.; Palamara, A.T.; Nencioni, L.; Ruggieri, A. Counteraction of HCV-Induced Oxidative Stress Concurs to Establish Chronic Infection in Liver Cell Cultures. Oxid. Med. Cell. Longev. 2019, 2019, 6452390. [Google Scholar] [PubMed]
- Polonikov, A. Endogenous Deficiency of Glutathione as the Most Likely Cause of Serious Manifestations and Death in COVID-19 Patients. ACS Infect. Dis. 2020, 6, 1558–1562. [Google Scholar] [CrossRef]
- Laforge, M.; Elbim, C.; Frère, C.; Hémadi, M.; Massaad, C.; Nuss, P.; Benoliel, J.J.; Becker, C. Tissue damage from neutrophil-induced oxidative stress in COVID-19. Nat. Rev. Immunol. 2020, 20, 515–516. [Google Scholar] [CrossRef] [PubMed]
- De Flora, S.; Balansky, R.; La Maestra, S. Rationale for the use of N-acetylcysteine in both prevention and adjuvant therapy of COVID-19. FASEB J. 2020, 34, 13185–13193. [Google Scholar] [CrossRef]
- Fratta Pasini, A.M.; Stranieri, C.; Cominacini, L.; Mozzini, C. Potential role of antioxidant and anti-inflammatory therapies to prevent severe sars-cov-2 complications. Antioxidants 2021, 10, 272. [Google Scholar] [CrossRef] [PubMed]
- Packer, L.; Cadenas, E. Lipoic acid: Energy metabolism and redox regulation of transcription and cell signaling. J. Clin. Biochem. Nutr. 2011, 48, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, J.; Lodge, J.K.; Marcocci, L.; Tritschler, H.J.; Packer, L.; Rihn, B.H. α-lipoic acid in liver metabolism and disease. Free Radic. Biol. Med. 1998, 24, 1023–1039. [Google Scholar] [CrossRef]
- Clavreul, N.; Bachschmid, M.M.; Hou, X.; Shi, C.; Idrizovic, A.; Ido, Y.; Pimentel, D.; Cohen, R.A. S-glutathiolation of p21ras by peroxynitrite mediates endothelial insulin resistance caused by oxidized low-density lipoprotein. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 2454–2461. [Google Scholar] [CrossRef]
- Ibrahimpasic, K. Alpha lipoic acid and glycaemic control in diabetic neuropathies at type 2 diabetes treatment. Med. Arch. 2013, 67, 7–9. [Google Scholar] [CrossRef]
- Hofmann, M.A.; Schiekofer, S.; Kanitz, M.; Klevesath, M.S.; Joswig, M.; Lee, V.; Morcos, M.; Tritschler, H.; Ziegler, R.; Wahl, P.; et al. Insufficient glycemic control increases nuclear factor-κB binding activity in peripheral blood mononuclear cells isolated from patients with type 1 diabetes. Diabetes Care 1998, 21, 1310–1316. [Google Scholar] [CrossRef]
- Ziegler, D.; Reljanovic, M.; Mehnert, H.; Gries, F.A. α-lipoic acid in the treatment of diabetic polyneuropathy in Germany: Current evidence from clinical trials. Exp. Clin. Endocrinol. Diabetes 1999, 107, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Nagamatsu, M.; Nickander, K.K.; Schmelzer, J.D.; Raya, A.; Wittrock, D.A.; Tritschler, H.; Low, P.A. Lipoic acid improves nerve blood flow, reduces oxidative stress, and improves distal nerve conduction in experimental diabetic neuropathy. Diabetes Care 1995, 18, 1160–1167. [Google Scholar] [CrossRef] [PubMed]
- Androne, L.; Gavan, N.A.; Veresiu, I.A.; Orasan, R. In vivo effect of lipoic acid on lipid peroxidation in patients with diabetic neuropathy. In Vivo 2000, 14, 327–330. [Google Scholar]
- Packer, L.; Kraemer, K.; Rimbach, G. Molecular aspects of lipoic acid in the prevention of diabetes complications. Nutrition 2001, 17, 888–895. [Google Scholar] [CrossRef]
- Jones, W.; Li, X.; Qu, Z.; Perriott, L.; Whitesell, R.R.; May, J.M. Uptake, recycling, and antioxidant actions of α-lipoic acid in endothelial cells. Free Radic. Biol. Med. 2002, 33, 83–93. [Google Scholar] [CrossRef]
- Ziegler, D.; Gries, F.A. α-lipoic acid in the treatment of diabetic peripheral and cardiac autonomic neuropathy. Diabetes 1997, 46, S62–S66. [Google Scholar] [CrossRef] [PubMed]
- Lynch, M.A. Lipoic acid confers protection against oxidative injury in non-neuronal and neuronal tissue. Nutr. Neurosci. 2001, 4, 419–438. [Google Scholar] [CrossRef] [PubMed]
- Tancheva, L.P.; Lazarova, M.I.; Alexandrova, A.V.; Dragomanova, S.T.; Nicoletti, F.; Tzvetanova, E.R.; Hodzhev, Y.K.; Kalfin, R.E.; Miteva, S.A.; Mazzon, E.; et al. Neuroprotective mechanisms of three natural antioxidants on a rat model of parkinson’s disease: A comparative study. Antioxidants 2020, 9, 49. [Google Scholar] [CrossRef]
- Tzvetanova, E.R.; Georgieva, A.P.; Alexandrova, A.V.; Tancheva, L.P.; Lazarova, M.I.; Dragomanova, S.T.; Alova, L.G.; Stefanova, M.O.; Kalfin, R.E. Antioxidant mechanisms in neuroprotective action of lipoic acid on learning and memory of rats with experimental dementia. Bulg. Chem. Commun. 2018, 50, 52–57. [Google Scholar]
- Hager, K.; Marahrens, A.; Kenklies, M.; Riederer, P.; Münch, G. Alpha-lipoic acid as a new treatment option for Azheimer type dementia. Arch. Gerontol. Geriatr. 2001, 32, 275–282. [Google Scholar] [CrossRef]
- Holmquist, L.; Stuchbury, G.; Berbaum, K.; Muscat, S.; Young, S.; Hager, K.; Engel, J.; Münch, G. Lipoic acid as a novel treatment for Alzheimer’s disease and related dementias. Pharmacol. Ther. 2007, 113, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Lovell, M.A.; Xie, C.; Xiong, S.; Markesbery, W.R. Protection against amyloid beta peptide and iron/hydrogen peroxide toxicity by alpha lipoic acid. J. Alzheimer’s Dis. 2003, 5, 229–239. [Google Scholar] [CrossRef]
- Kim, S.M.; Ha, J.S.; Han, A.R.; Cho, S.W.; Yang, S.J. Effects of α-lipoic acid on LPS-induced neuroinflammation and NLRP3 inflammasome activation through the regulation of BV-2 microglial cells activation. BMB Rep. 2019, 52, 613–618. [Google Scholar] [CrossRef]
- Maldonado-Rojas, W.; Olivero-Verbel, J.; Ortega-Zuñiga, C. Searching of protein targets for alpha lipoic acid. J. Braz. Chem. Soc. 2011, 22, 2250–2259. [Google Scholar] [CrossRef]
- Suzuki, Y.J.; Tsuchiya, M.; Packer, L. Thioctic acid and dihydrolipoic acid are novel antioxidants which interact with reactive oxygen species. Free Radic. Res. 1991, 15, 255–263. [Google Scholar] [CrossRef]
- Scott, B.C.; Aruoma, O.I.; Evans, P.J.; O’Neill, C.; Van Der Vliet, A.; Cross, C.E.; Tritschler, H.; Halliwell, B. Lipoic and dihydrolipoic acids as antioxidants. A critical evaluation. Free Radic. Res. 1994, 20, 119–133. [Google Scholar] [CrossRef]
- Trujillo, M.; Radi, R. Peroxynitrite reaction with the reduced and the oxidized forms of lipoic acid: New insights into the reaction of peroxynitrite with thiols. Arch. Biochem. Biophys. 2002, 397, 91–98. [Google Scholar] [CrossRef]
- Biewenga, G.P.; Haenen, G.R.M.M.; Bast, A. The pharmacology of the antioxidant: Lipoic acid. Gen. Pharmacol. 1997, 29, 315–331. [Google Scholar] [CrossRef]
- Ou, P.; Tritschler, H.J.; Wolff, S.P. Thioctic (lipoic) acid: A therapeutic metal-chelating antioxidant? Biochem. Pharmacol. 1995, 50, 123–126. [Google Scholar] [CrossRef]
- Lodge, J.K.; Traber, M.G.; Packer, L. Thiol chelation of Cu2+ by dihydrolipoic acid prevents human low density lipoprotein peroxidation. Free Radic. Biol. Med. 1998, 25, 287–297. [Google Scholar] [CrossRef]
- Goralska, M.; Dackor, R.; Holley, B.; McGahan, M.C. Alpha lipoic acid changes iron uptake and storage in lens epithelial cells. Exp. Eye Res. 2003, 76, 241–248. [Google Scholar] [CrossRef]
- Suh, J.H.; Moreau, R.; Heath, S.H.D.; Hagen, T.M. Dietary supplementation with (R)-α-lipoic acid reverses the age-related accumulation of iron and depletion of antioxidants in the rat cerebral cortex. Redox Rep. 2005, 10, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Lykkesfeldt, J.; Hagen, T.M.; Vinarsky, V.; Ames, B.N. Age-associated decline in ascorbic acid concentration, recycling, and biosynthesis in rat hepatocytes—Reversal with (R)-α-lipoic acid supplementation. FASEB J. 1998, 12, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Xu DP, W.W. Alpha-Lipoic acid dependent regeneration of ascorbic acid from dehydroascorbic acid in rat liver mitochondria. J Bioenerg. Biomembr. 1996, 28, 77–85. [Google Scholar]
- Bast, A.; Haenen, G.R.M.M. Interplay between lipoic acid and glutathione in the protection against microsomal lipid peroxidation. Biochim. Biophys. Acta BBA Lipids Lipid Metab. 1988, 963, 558–561. [Google Scholar] [CrossRef]
- Busse E Zimmer G Schopohl B Kornhuber B Influence of alpha-lipoic acid on intracellular glutathione in vitro and in vivo. Arzneim. Forsch. 1992, 42, 829–831.
- Suh, J.H.; Wang, H.; Liu, R.M.; Liu, J.; Hagen, T.M. (R)-α-Lipoic acid reverses the age-related loss in GSH redox status in post-mitotic tissues: Evidence for increased cysteine requirement for zGSH synthesis. Arch. Biochem. Biophys. 2004, 423, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Suh, J.H.; Shenvi, S.V.; Dixon, B.M.; Liu, H.; Jaiswal, A.K.; Liu, R.M.; Hagen, T.M. Decline in transcriptional activity of Nrf2 causes age-related loss of glutathione synthesis, which is reversible with lipoic acid. Proc. Natl. Acad. Sci. USA 2004, 101, 3381–3386. [Google Scholar] [CrossRef] [PubMed]
- Lii, C.-K.; Liu, K.-L.; Cheng, Y.-P.; Lin, A.-H.; Chen, H.-W.; Tsai, C.-W. Sulforaphane and α-Lipoic Acid Upregulate the Expression of the π Class of Glutathione S-Transferase through c-Jun and Nrf2 Activation. J. Nutr. 2010, 140, 885–892. [Google Scholar] [CrossRef]
- Lee, J.; Jung, S.-Y.; Yang, K.-J.; Kim, Y.; Lee, D.; Lee, M.H.; Kim, D.-K. α-Lipoic acid prevents against cisplatin cytotoxicity via activation of the NRF2/HO-1 antioxidant pathway. PLoS ONE 2019, 14, e0226769. [Google Scholar] [CrossRef]
- Koriyama, Y.; Nakayama, Y.; Matsugo, S.; Kato, S. Protective effect of lipoic acid against oxidative stress is mediated by Keap1/Nrf2-dependent heme oxygenase-1 induction in the RGC-5 cellline. Brain Res. 2013, 1499, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Pilar Valdecantos, M.; Prieto-Hontoria, P.L.; Pardo, V.; Módol, T.; Santamaría, B.; Weber, M.; Herrero, L.; Serra, D.; Muntané, J.; Cuadrado, A.; et al. Essential role of Nrf2 in the protective effect of lipoic acid against lipoapoptosis in hepatocytes. Free Radic. Biol. Med. 2015, 84, 263–278. [Google Scholar] [CrossRef] [PubMed]
- Ying, Z.; Kampfrath, T.; Sun, Q.; Parthasarathy, S.; Rajagopalan, S. Evidence that α-lipoic acid inhibits NF-κB activation independent of its antioxidant function. Inflamm. Res. 2011, 60, 219–225. [Google Scholar] [CrossRef]
- Sen, C.K.; Roy, S.; Packer, L. Fas mediated apoptosis of human Jurkat T-cells: Intracellular events and potentiation by redox-active α-lipoic acid. Cell Death Differ. 1999, 6, 481–491. [Google Scholar] [CrossRef]
- Suzuki, Y.J.; Shi, S.S.; Day, R.M.; Blumberg, J.B. Differential regulation of MAP kinase signaling by pro- and antioxidant biothiols. Ann. N. Y. Acad. Sci. 2000, 899, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.S.; Day, R.M.; Halpner, A.D.; Blumberg, J.B.; Suzuki, Y.J. Homocysteine and α-Lipoic Acid Regulate p44/42 MAP Kinase Phosphorylation in NIH/3T3 Cells. Antioxid. Redox Signal. 1999, 1, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Konrad, D.; Somwar, R.; Sweeney, G.; Yaworsky, K.; Hayashi, M.; Ramlal, T.; Klip, A. The antihyperglycemic drug α-lipoic acid stimulates glucose uptake via both GLUT4 translocation and GLUT4 activation: Potential role of p38 mitogen-activated protein kinase in GLUT4 activation. Diabetes 2001, 50, 1464–1471. [Google Scholar] [CrossRef]
- Petersen Shay, K.; Hagen, T.M. Age-associated impairment of Akt phosphorylation in primary rat hepatocytes is remediated by alpha-lipoic acid through PI3 kinase, PTEN, and PP2A. Biogerontology 2009, 10, 443–456. [Google Scholar] [CrossRef][Green Version]
- Zhang, W.J.; Wei, H.; Hagen, T.; Frei, B. α-Lipoic acid attenuates LPS-induced inflammatory responses by activating the phosphoinositide 3-kinase/Akt signaling pathway. Proc. Natl. Acad. Sci. USA 2007, 104, 4077–4082. [Google Scholar] [CrossRef]
- Smith, A.R.; Hagen, T.M. Vascular endothelial dysfunction in aging: Loss of Akt-dependent endothelial nitric oxide synthase phosphorylation and partial restoration by (R)-α-lipoic acid. Biochem. Soc. Trans. 2003, 31, 1447–1449. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.J.; Moini, H.; Shon, H.K.; Chung, A.S.; Packer, L. α-Lipoic acid decreases thiol reactivity of the insulin receptor and protein tyrosine phosphatase 1B in 3T3-L1 adipocytes. Biochem. Pharmacol. 2003, 66, 849–858. [Google Scholar] [CrossRef]
- Diesel, B.; Kulhanek-Heinze, S.; Höltje, M.; Brandt, B.; Höltje, H.D.; Vollmar, A.M.; Kiemer, A.K. α-lipoic acid as a directly binding activator of the insulin receptor: Protection from hepatocyte apoptosis. Biochemistry 2007, 46, 2146–2155. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Park, J.Y.; Namkoong, C.; Jang, P.G.; Ryu, J.W.; Song, H.S.; Yun, J.Y.; Namgoong, I.S.; Ha, J.; Park, I.S.; et al. Anti-obesity effects of α-lipoic acid mediated by suppression of hypothalamic AMP-activated protein kinase. Nat. Med. 2004, 10, 727–733. [Google Scholar] [CrossRef]
- Woo, J.L.; Song, K.H.; Eun, H.K.; Jong, C.W.; Hyoun, S.K.; Park, H.S.; Kim, M.S.; Kim, S.W.; Lee, K.U.; Park, J.Y. α-Lipoic acid increases insulin sensitivity by activating AMPK in skeletal muscle. Biochem. Biophys. Res. Commun. 2005, 332, 885–891. [Google Scholar]
- Lee, W.J.; In, K.L.; Hyoun, S.K.; Yun, M.K.; Eun, H.K.; Jong, C.W.; Sung, M.H.; Kim, M.S.; Jo, I.; Goo, T.O.; et al. α-lipoic acid prevents endothelial dysfunction in obese rats via activation of AMP-activated protein kinase. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 2488–2494. [Google Scholar] [CrossRef]
- Beck, M.A.; Handy, J.; Levander, O.A. The role of oxidative stress in viral infections. Ann. N. Y. Acad. Sci. 2000, 917, 906–912. [Google Scholar] [CrossRef]
- Camini, F.C.; da Silva Caetano, C.C.; Almeida, L.T.; de Brito Magalhães, C.L. Implications of oxidative stress on viral pathogenesis. Arch. Virol. 2017, 162, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Serkedjieva, J.; Krumova, E.; Stefanova, T.; Tancheva, L. Pulmonary protection of a plant polyphenol extract in influenza virus-infected mice. J. Infect. 2009, 59, S426. [Google Scholar] [CrossRef]
- Abarova, S.; Tancheva, L.; Nikolov, R.; Serkedjieva, J.; Pavlova, E.; Bramanti, A.; Nicoletti, F.; Tzvetkov, N.T. Preventive Effect of a Polyphenol-Rich Extract from Geranium sanguineum L. on Hepatic Drug Metabolism in Influenza Infected Mice. Sci. Pharm. 2020, 88, 45. [Google Scholar] [CrossRef]
- Mileva, M.; Tancheva, L.; Bakalova, R.; Galabov, A.; Savov, V.; Ribarov, S. Effect of vitamin E on lipid peroxidation and liver monooxigenase activity in experimental influenza virus infection. Toxicol. Lett. 2000, 114, 39–45. [Google Scholar] [CrossRef]
- Mileva, M.; Bakalova, R.; Tancheva, L.; Galabov, A.; Ribarov, S. Effect of vitamin E supplementation on lipid peroxidation in blood and lung of influenza virus infected mice. Comp. Immunol. Microbiol. Infect. Dis. 2002, 25, 1–11. [Google Scholar] [CrossRef]
- Murzakhmetova, M.; Moldakarimov, S.; Tancheva, L.; Abarova, S.; Serkedjieva, J. Antioxidant and prooxidant properties of a polyphenol-rich extract from Geranium sanguineum L. in vitro and in vivo. Phyther. Res. 2008, 22, 746–751. [Google Scholar] [CrossRef]
- Savov, V.M.; Galabov, A.S.; Tantcheva, L.P.; Mileva, M.M.; Pavlova, E.L.; Stoeva, E.S.; Braykova, A.A. Effects of rutin and quercetin on monooxygenase activities in experimental influenza virus infection. Exp. Toxicol. Pathol. 2006, 58, 59–64. [Google Scholar] [CrossRef]
- Johari, J.; Kianmehr, A.; Mustafa, M.; Abubakar, S.; Zandi, K. Antiviral Activity of Baicalein and Quercetin against the Japanese Encephalitis Virus. Int. J. Mol. Sci. 2012, 13, 16785–16795. [Google Scholar] [CrossRef]
- Bachmetov, L.; Gal-Tanamy, M.; Shapira, A.; Vorobeychik, M.; Giterman-Galam, T.; Sathiyamoorthy, P.; Golan-Goldhirsh, A.; Benhar, I.; Tur-Kaspa, R.; Zemel, R. Suppression of hepatitis C virus by the flavonoid quercetin is mediated by inhibition of NS3 protease activity. J. Viral Hepat. 2012, 19, e81–e88. [Google Scholar] [CrossRef]
- Fan, D.; Zhou, X.; Zhao, C.; Chen, H.; Zhao, Y.; Gong, X. Anti-inflammatory, antiviral and quantitative study of quercetin-3-O- β-D-glucuronide in Polygonum perfoliatum L. Fitoterapia 2011, 82, 805–810. [Google Scholar] [CrossRef]
- Quiles, J.L.; Rivas-García, L.; Varela-López, A.; Llopis, J.; Battino, M.; Sánchez-González, C. Do nutrients and other bioactive molecules from foods have anything to say in the treatment against COVID-19? Environ. Res. 2020, 191, 110053. [Google Scholar] [CrossRef]
- Bai, S.W.; Chen, C.Y.; Ji, J.; Xie, Q.M.; Ma, Y.; Sun, B.L.; Xue, C.Y.; Cao, Y.C.; Ma, J.Y.; Bi, Y.Z. Inhibition Effect of Alpha-Lipoic Acid on the Propagation of Influenza A Virus in MDCK Cells. Pak. Vet. J. 2012, 32, 101–106. [Google Scholar]
- McGeoch, D.J.; Cook, S.; Dolan, A.; Jamieson, F.E.; Telford, E.A.R. Molecular phylogeny and evolutionary timescale for the family of mammalian herpesviruses. J. Mol. Biol. 1995, 247, 443–458. [Google Scholar] [CrossRef]
- Spisakova, M.; Cizek, Z.; Melkova, Z. Ethacrynic and α-lipoic acids inhibit vaccinia virus late gene expression. Antiviral Res. 2009, 81, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Hepatitis viral load correlates to glutathione levels. Posit. Health News 1998, 17, 14–15.
- Vendemiale, G.; Grattagliano, I.; Portincasa, P.; Serviddio, G.; Palasciamo, G.; Altomare, E. Oxidative stress in symptom-free HCV carriers: Relation with ALT flare-up. Eur. J. Clin. Investig. 2001, 31, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Tanyalcin, T.; Taskiran, D.; Topalak, O.; Batur, Y.; Kutay, F. The effects of chronic hepatitis C and B virus infections on liver reduced and oxidized glutathione concentrations. Hepatol. Res. 2000, 18, 104–109. [Google Scholar] [CrossRef]
- Von Herbay, A.; Stahl, W.; Niederau, C.; Von Laar, J.; Strohmeyer, G.; Sies, H. Diminished plasma levels of vitamin E in patients with severe viral hepatitis. Free Radic. Res. 1996, 25, 461–466. [Google Scholar] [CrossRef]
- Berkson, B.M. A conservative triple antioxidant approach to the treatment of hepatitis C. Combination of alpha lipoic acid (Thioctic acid), silymarin, and selenium: Three case histories. Med. Klin. 1999, 94, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Melhem, A.; Stern, M.; Shibolet, O.; Israeli, E.; Ackerman, Z.; Pappo, O.; Hemed, N.; Rowe, M.; Ohana, H.; Zabrecky, G.; et al. Treatment of chronic hepatitis C virus infection via antioxidants: Results of a phase I clinical trial. J. Clin. Gastroenterol. 2005, 39, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Greenspan, H.C.; Aruoma, O.I. Oxidative stress and apoptosis in HIV infection: A role for plant-derived metabolites with synergistic antioxidant activity. Immunol. Today 1994, 15, 209–213. [Google Scholar] [CrossRef]
- Pace, G.W.; Leaf, C.D. The role of oxidative stress in HIV disease. Free Radic. Biol. Med. 1995, 19, 523–528. [Google Scholar] [CrossRef]
- Sandstrom, P.A.; Tebbey, R.W.; Van Cleave, S.; Buttke, T.M. Lipid hydroperoxides induce apoptosis in T cells displaying a HIV- associated glutathione peroxidase deficiency. J. Biol. Chem. 1994, 269, 798–801. [Google Scholar] [CrossRef]
- Jarstrand, C.; Åkerlund, B. Oxygen radical release by neutrophils of HIV-infected patients. Chem. Biol. Interact. 1994, 91, 141–146. [Google Scholar] [CrossRef]
- Polyakov, V.M.; Shepelev, A.P.; Kokovkina, O.E.; Vtornikova, I.V. B15—Superoxide anion (O2−) production and enzymatic disbalance in peripheral blood cells isolated from HIV-infected children. Free Radic. Biol. Med. 1994, 16, 15. [Google Scholar] [CrossRef]
- Baur, A.; Harrer, T.; Peukert, M.; Jahn, G.; Kalden, J.R.; Fleckenstein, B. Alpha-lipoic acid is an effective inhibitor of human immuno-deficiency virus (HIV-1) replication. Klin. Wochenschr. 1991, 69, 722–724. [Google Scholar] [CrossRef]
- Jariwalla, R.J.; Lalezari, J.; Cenko, D.; Mansour, S.E.; Kumar, A.; Gangapurkar, B.; Nakamura, D. Restoration of blood total glutathione status and lymphocyte function following α-lipoic acid supplementation in patients with HIV infection. J. Altern. Complement. Med. 2008, 14, 139–146. [Google Scholar] [CrossRef]
- Studies on Lipoate Effects on Blood Redox State in Human Immunodeficiency Virus Infected Patients—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/8141828/ (accessed on 9 February 2021).
- Fagone, P.; Ciurleo, R.; Lombardo, S.D.; Iacobello, C.; Palermo, C.I.; Shoenfeld, Y.; Bendtzen, K.; Bramanti, P.; Nicoletti, F. Transcriptional landscape of SARS-CoV-2 infection dismantles pathogenic pathways activated by the virus, proposes unique sex-specific differences and predicts tailored therapeutic strategies. Autoimmun. Rev. 2020, 19, 102571. [Google Scholar] [CrossRef]
- Cavalli, E.; Petralia, M.; Basile, M.; Bramanti, A.; Bramanti, P.; Nicoletti, F.; Spandidos, D.; Shoenfeld, Y.; Fagone, P. Transcriptomic analysis of COVID-19 lungs and bronchoalveolar lavage fluid samples reveals predominant B cell activation responses to infection. Int. J. Mol. Med. 2020, 46, 1266–1273. [Google Scholar] [CrossRef]
- Tancheva, L.; Petralia, M.C.; Miteva, S.; Dragomanova, S.; Solak, A.; Kalfin, R.; Lazarova, M.; Yarkov, D.; Ciurleo, R.; Cavalli, E.; et al. Emerging neurological and psychobiological aspects of COVID-19 infection. Brain Sci. 2020, 10, 852. [Google Scholar] [CrossRef]
- Cavalli, E.; Bramanti, A.; Ciurleo, R.; Tchorbanov, A.I.; Giordano, A.; Fagone, P.; Belizna, C.; Bramanti, P.; Shoenfeld, Y.; Nicoletti, F. Entangling COVID-19 associated thrombosis into a secondary antiphospholipid antibody syndrome: Diagnostic and therapeutic perspectives (Review). Int. J. Mol. Med. 2020, 46, 903–912. [Google Scholar] [CrossRef]
- Sturley, S.L.; Rajakumar, T.; Hammond, N.; Higaki, K.; Márka, Z.; Márka, S.; Munkacsi, A.B. Potential COVID-19 therapeutics from a rare disease: Weaponizing lipid dysregulation to combat viral infectivity. J. Lipid Res. 2020, 61, 972–982. [Google Scholar] [CrossRef]
- Caterino, M.; Gelzo, M.; Sol, S.; Fedele, R.; Annunziata, A.; Calabrese, C.; Fiorentino, G.; D’Abbraccio, M.; Dell’Isola, C.; Fusco, F.M.; et al. Dysregulation of lipid metabolism and pathological inflammation in patients with COVID-19. Sci. Rep. 2021, 11, 2941. [Google Scholar] [CrossRef]
- Abu-Farha, M.; Thanaraj, T.A.; Qaddoumi, M.G.; Hashem, A.; Abubaker, J.; Al-Mulla, F. The Role of Lipid Metabolism in COVID-19 Virus Infection and as a Drug Target. Int. J. Mol. Sci. 2020, 21, 3544. [Google Scholar] [CrossRef] [PubMed]
- Suhail, S.; Zajac, J.; Fossum, C.; Lowater, H.; McCracken, C.; Severson, N.; Laatsch, B.; Narkiewicz-Jodko, A.; Johnson, B.; Liebau, J.; et al. Role of Oxidative Stress on SARS-CoV (SARS) and SARS-CoV-2 (COVID-19) Infection: A Review. Protein J. 2020, 39, 644–656. [Google Scholar] [CrossRef] [PubMed]
- Guloyan, V.; Oganesian, B.; Baghdasaryan, N.; Yeh, C.; Singh, M.; Guilford, F.; Ting, Y.S.; Venketaraman, V. Glutathione supplementation as an adjunctive therapy in COVID-19. Antioxidants 2020, 9, 914. [Google Scholar] [CrossRef]
- Pagano, G.; Manfredi, C.; Pallardó, F.V.; Lyakhovich, A.; Tiano, L.; Trifuoggi, M. Potential roles of mitochondrial cofactors in the adjuvant mitigation of proinflammatory acute infections, as in the case of sepsis and COVID-19 pneumonia. Inflamm. Res. 2021, 70, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Zhong, M.; Sun, A.; Xiao, T.; Yao, G.; Sang, L.; Zheng, X.; Zhang, J.; Jin, X.; Xu, L.; Yang, W.; et al. A Randomized, single-blind, group sequential, active-controlled study to evaluate the clinical efficacy and safety of α-Lipoic acid for critically ill patients with coronavirus disease 2019 (COVID-19). medRxiv 2020. [Google Scholar] [CrossRef]
| Mechanism of Action | References |
|---|---|
| Scavenging reactive oxygen species | [37,38,39] |
| Regeneration other endogenous antioxidants (e.g., vitamins C and E) | [40] |
| Chelation of redox-active metals | [41,42,43,44] |
| Induction of endogenous antioxidants (e.g., ascorbate, GSH) | [45,46,47,48,49,50] |
| Induction of the Nrf2/ARE pathway | [51,52,53,54] |
| Inhibition of NF-κB activation | [55] |
| Activation of: | -- |
| - PKC | [56] |
| - Erk1/2 | [57,58] |
| - p38 MAPK | [59] |
| - PI3K | [59] |
| - Akt | [59,60,61,62] |
| - Inhibition of: | -- |
| - PTEN | [60] |
| - PP2A | [60] |
| - PTP1B | [63] |
| Activation of insulin receptor by direct binding | [64] |
| Activation of AMP-activated protein kinase (AMPK) | [65,66,67] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dragomanova, S.; Miteva, S.; Nicoletti, F.; Mangano, K.; Fagone, P.; Pricoco, S.; Staykov, H.; Tancheva, L. Therapeutic Potential of Alpha-Lipoic Acid in Viral Infections, including COVID-19. Antioxidants 2021, 10, 1294. https://doi.org/10.3390/antiox10081294
Dragomanova S, Miteva S, Nicoletti F, Mangano K, Fagone P, Pricoco S, Staykov H, Tancheva L. Therapeutic Potential of Alpha-Lipoic Acid in Viral Infections, including COVID-19. Antioxidants. 2021; 10(8):1294. https://doi.org/10.3390/antiox10081294
Chicago/Turabian StyleDragomanova, Stela, Simona Miteva, Ferdinando Nicoletti, Katia Mangano, Paolo Fagone, Salvatore Pricoco, Hristian Staykov, and Lyubka Tancheva. 2021. "Therapeutic Potential of Alpha-Lipoic Acid in Viral Infections, including COVID-19" Antioxidants 10, no. 8: 1294. https://doi.org/10.3390/antiox10081294
APA StyleDragomanova, S., Miteva, S., Nicoletti, F., Mangano, K., Fagone, P., Pricoco, S., Staykov, H., & Tancheva, L. (2021). Therapeutic Potential of Alpha-Lipoic Acid in Viral Infections, including COVID-19. Antioxidants, 10(8), 1294. https://doi.org/10.3390/antiox10081294










