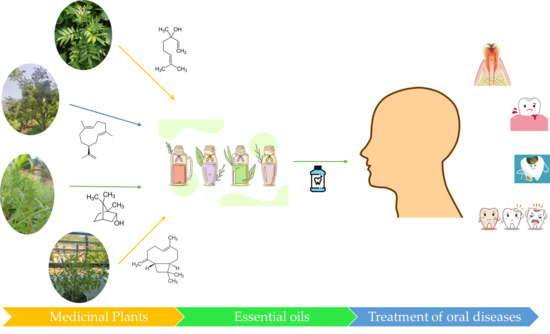
Beneficial Role of Antioxidant Secondary Metabolites from Medicinal Plants in Maintaining Oral Health
Abstract
1. Introduction
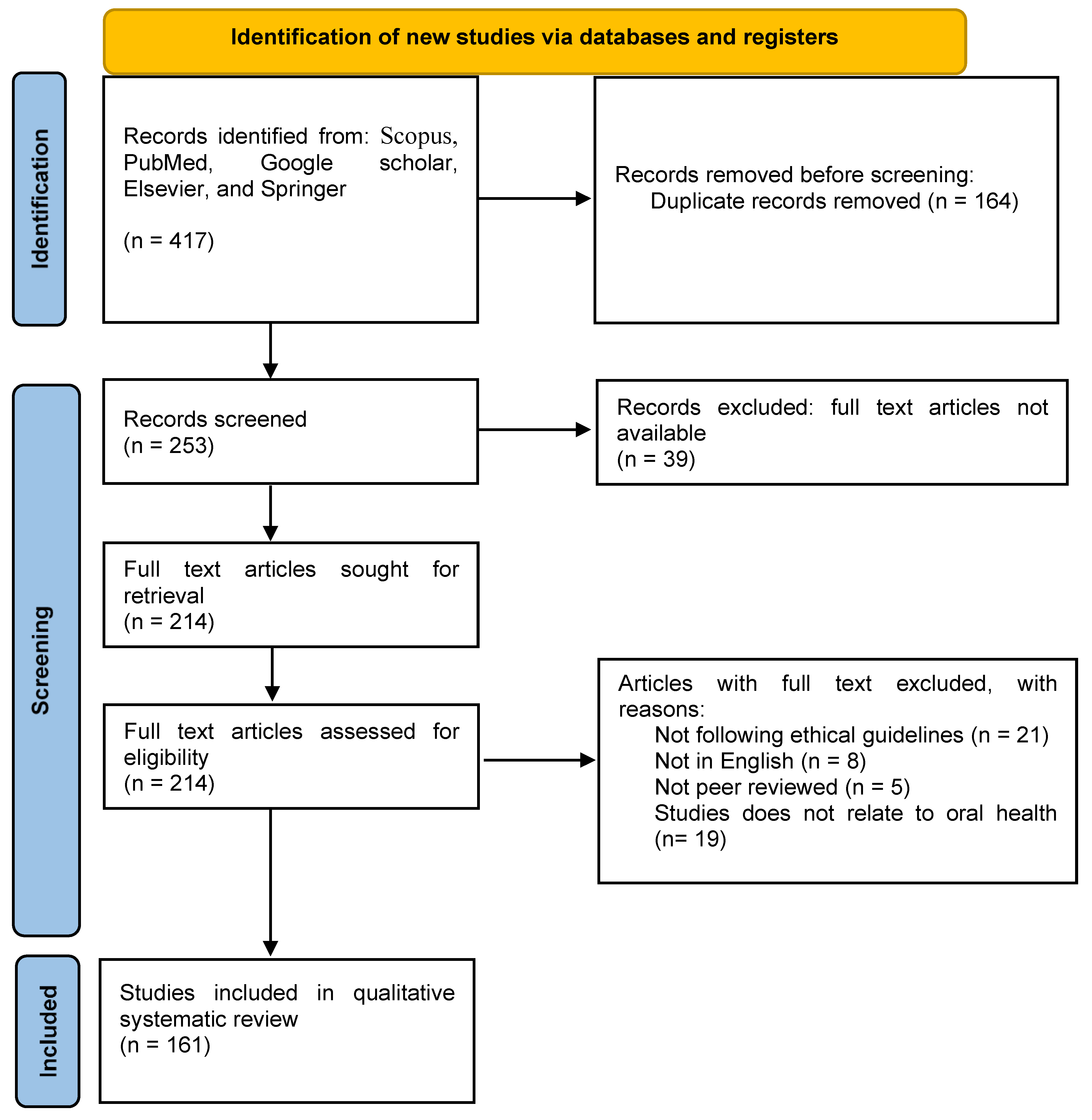
2. Methodology
3. Medicinal Plants and Beneficial Role in Oral Health
3.1. Zanthoxylum armatum DC. (Tejphal)
3.2. Ocimum sanctum L. (Tulsi)
3.3. Salvadora persica L. (Miswak)
3.4. Eucalyptus globulus Labill. (Nilgiri)
3.5. Thymus vulgaris L. (Banajwain)
3.6. Azadirachta indica A. Juss. (Neem)
3.7. Acorus calamus L. (Sweet flag or Vacha)
3.8. Juglans regia L. (Walnut, Akhrot)
3.9. Asparagus racemosus Willd (Satavari)
3.10. Juniperus communis L.
3.11. Melaleuca alternifolia (Maiden and Betche) Cheel
3.12. Acacia nilotica (L.) Delile
3.13. Quercus infectoria G. Oilvier
3.14. Artemisia dracunculus L.
3.15. Streblus asper Lour
3.16. Cichorium intybus L.
3.17. Vitex negundo Linn. (Nirgundi)
3.18. Rosmarinus officinalis L.
3.19. Embelia ribes Burm. f.
3.20. Spilanthes Species
3.21. Nigella sativa L.
4. Antioxidant Extracts from Medicinal Plants in Oral Health: A Clinical Trial Perspective
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- James, S.L.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 Diseases and Injuries for 195 countries and territories, 1990-2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef]
- Petti, S.; Glendor, U.; Andersson, L. World traumatic dental injury prevalence and incidence, a meta-analysis—One billion living people have had traumatic dental injuries. Dent. Traumatol. 2018, 34, 71–86. [Google Scholar] [CrossRef]
- Agbor, M.A.; Naidoo, S. Ethnomedicinal plants used by traditional healers to treat oral health problems in Cameroon. Evid.-Based Complement. Altern. Med. 2015, 2015, 649832. [Google Scholar] [CrossRef] [PubMed]
- Şener, B.; Kiliç, M. Herbal extracts used in dental disorders. J. Sci. Tech. Res. 2019, 19, 14107–14111. [Google Scholar] [CrossRef]
- Martíınez, C.C.; Gómez, M.D.; Oh, M.S. Use of traditional herbal medicine as an alternative in dental treatment in mexican dentistry: A review. Pharm. Biol. 2017, 55, 1992–1998. [Google Scholar] [CrossRef]
- Baratta, M.T.; Dorman, H.J.D.; Deans, S.G.; Figueiredo, A.C.; Barroso, J.G.; Ruberto, G. Antimicrobial and antioxidant properties of some commercial essential oils. Flavour Fragr. J. 1998, 13, 235–244. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antimicrobial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef]
- Guleria, S.; Tiku, A.K.; Koul, A.; Gupta, S.; Singh, G.; Razdan, V.K. Antioxidant and Antimicrobial Properties of the Essential Oil and Extracts of Zanthoxylum alatum Grown in North-Western Himalaya. Sci. World J. 2013, 2013, 790580. [Google Scholar] [CrossRef]
- Thosar, N.; Basak, S.; Bahadure, R.N.; Rajurkar, M. Antimicrobial efficacy of five essential oils against oral pathogens: An in vitro study. Eur. J. Dent. 2013, 7, S071–S077. [Google Scholar] [CrossRef] [PubMed]
- Nordin, A.; Saim, A.B.; Ramli, R.; Hamid, A.A.; Nasri, N.W.M.; Idrus, R.B.H. Miswak and oral health: An evidence-based review. Saudi J. Biol. Sci. 2020, 27, 1801–1810. [Google Scholar] [CrossRef]
- Agarwal, R.; Lakshmi, T. Eucalyptus oil in dentistry: A mini Review. Int. J. Drug Dev. Res. 2013, 5, 58–61. [Google Scholar]
- Fani, M.; Kohanteb, J. In Vitro Antimicrobial Activity of Thymus vulgaris Essential Oil against Major Oral Pathogens. J. Evid.-Based Complement. Altern. Med. 2017, 22, 660–666. [Google Scholar] [CrossRef]
- Jenkinson, H.F.; Lamont, R.J. Oral microbial communities in sickness and in health. Trends Microbiol. 2005, 13, 589–595. [Google Scholar] [CrossRef]
- Tichy, J.; Novak, J. Extraction, assay, and analysis of antimicrobials from plants with activity against dental pathogens (Streptococcus sp.). J. Altern. Complement. Med. 1998, 4, 39–45. [Google Scholar] [CrossRef]
- Badria, F.A.; Zidan, O.A. Natural products for dental caries prevention. J. Med. Food 2004, 7, 381–384. [Google Scholar] [CrossRef] [PubMed]
- Kurose, K.; Yatagai, M. Components of the essential oils of Azadirachta indica A. Juss, Azadirachta siamensis Velton, and Azadirachta excelsa (Jack) Jacobs and their comparison. J. Wood Sci. 2005, 51, 185–188. [Google Scholar] [CrossRef]
- Alok, S.; Jain, S.K.; Verma, A.; Kumar, M.; Mahor, A.; Sabharwal, M. Plant profile, phytochemistry and pharmacology of Asparagus racemosus (Shatavari): A review. Asian Pac. J. Trop. Dis. 2013, 3, 242–251. [Google Scholar] [CrossRef]
- Dash, T.R.; Singh, N.; Gupta, D.; Panwar, E.; Ramisetty, S. Role of medicinal herbs in oral health management. J. Int. Dent. Med. Res. 2014, 1, 113–119. [Google Scholar]
- Rajput, S.B.; Tonge, M.B.; Karuppayil, S.M. An overview on traditional uses and pharmacological profile of Acorus calamus Linn. (Sweet flag) and other Acorus species. Phytomedicine 2014, 21, 268–276. [Google Scholar] [CrossRef]
- Al-Asmari, A.K.; Athar, M.T.; Al-Faraidy, A.A.; Almuhaiza, M.S. Chemical composition of essential oil of Thymus vulgaris collected from Saudi Arabian market. Asian Pac. J. Trop. Biomed. 2017, 7, 147–150. [Google Scholar] [CrossRef]
- Verma, G.; Sharma, V. A scientific update on Juglans regia Linn. Asian J. Pharm. Res. Dev. 2020, 8, 166–175. [Google Scholar] [CrossRef]
- Prabu, G.R.; Gnanamani, A.; Sadulla, S. Guaijaverin-A plant flavonoid as potential antiplaque agent against Streptococcus mutans. J. Appl. Microbiol. 2006, 101, 487–495. [Google Scholar] [CrossRef]
- Bhattacharjee, A.; Kumar, B.D.; Das, B.; Dkhar, D.; Kachari, D. Zanthoxylum Armatum: A systemic review of its ethno-medicinal properties, phytochemistry, pharmacology and toxicology. Adv. Pharmacol. Clin. Trials 2019, 4, 000162. [Google Scholar] [CrossRef]
- Negi, J.S.; Bisht, V.K.; Bhandari, A.K.; Bisht, R.; Negi, S.K. Major constituents, antioxidant and antibacterial activities of Zanthoxylum armatum DC. essential oil. Iran. J. Pharmacol. Ther. 2012, 11, 68–72. [Google Scholar]
- Mehta, D.K.; Das, R.; Bhandari, A. In-vitro anthelmintic activity of seeds of Zanthoxylum armatum DC. against Pheretima Posthuma. Int. J. Green Pharm. 2012, 6. [Google Scholar] [CrossRef]
- Smitha, G.R.; Varghese, T.S.; Manivel, P. Cultivation of Ocimum. Ext. Bull. Dir. Med. Aromat. Plants Res. Anand, Gujarat, India 2014, 30. [Google Scholar]
- Iqbal, Z.; Akhtar, M.; Sabri, M.U.; Altaf, A. Chemical composition of ocimum sanctum essential oil by GC-MS analysis. Nat. Prod. Chem. Res. 2020, 8, 1–5. [Google Scholar] [CrossRef]
- Annigeri, R.G.; Mangala, G.K.; Thimmasetty, J.; Sharma, N.; Kanjani, V.; Sravya, G. Evaluation of tulasi extract mouthwash in the management of oral candidiasis. J. Adv. Clin. Res. Insights 2018, 5, 30–34. [Google Scholar] [CrossRef]
- Saftarasmi; Lakshmi, T. Ocimum Sanctum in Dental Care-A Mini Review. Res. J. Pharm. Technol. 2014, 7, 101–103. [Google Scholar]
- Khatak, M.; Khatak, S.; Siddqui, A.; Vasudeva, N.; Aggarwal, A.; Aggarwal, P. Salvadora persica. Pharmacogn. Rev. 2010, 4, 209–214. [Google Scholar] [CrossRef]
- Alali, F.; Hudaib, M.; Aburjai, T.; Khairallah, K.; Al-Hadidi, N. GC-MS Analysis and Antimicrobial Activity of the Essential Oil from the Stem of the Jordanian Toothbrush Tree Salvadora persica. Pharm. Biol. 2005, 42, 577–580. [Google Scholar] [CrossRef][Green Version]
- Al-Dabbagh, S.A.; Qasim, H.J.; Al-Derzi, N.A. Efficacy of Miswak toothpaste and mouthwash on cariogenic bacteria. Saudi Med. J. 2016, 37, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Mbuya, L.P.; Msanga, H.P.; Ruffo, C.K.; Birnie, A.; Tengnäs, B. Useful Trees and Shrubs for Tanzania: Identification, Propagation and Management for Agricultural and Pastoral Communities; Regional Soil Conservation Unit, Swedish International Development Authority: Nairobi, Kenya, 1994. [Google Scholar]
- Joshi, A.; Sharma, A.; Bachheti, R.K.; Pandey, D.P. A comparative study of the chemical composition of the essential oil from Eucalyptus globulus growing in Dehradun (India) and around the world. Orient. J. Chem. 2016, 32, 331–340. [Google Scholar] [CrossRef]
- Ragul, P.; Dhanraj, M.; Jain, A.R. Efficacy of eucalyptus oil over chlorhexidine mouthwash in dental practice in dental practice. Drug Invent. Today 2018, 10, 638–641. [Google Scholar]
- Reddy, V.P.; Kandisa, R.V.; Varsha, P.V.; Satyam, S. Review on Thymus vulgaris Traditional Uses and Pharmacological Properties. Med. Aromat. Plants 2014, 3, 164. [Google Scholar] [CrossRef]
- Santos, R.I.; Pereira, D.F.; Teodoro, G.R.; Del Ciampo, J.O.; Ogasawara, M.S.; Canettieri, A.C.; Salvador, M.J. Essential oil of Thymus vulgaris: Preparation of pharmaceutical mouthwash formulation and In Vitro evaluation of the bacterial plaque-inhibiting properties. Lat. Am. J. Pharm. 2010, 29, 941–947. [Google Scholar]
- Teklay, A.; Abera, B.; Giday, M. An ethnobotanical study of medicinal plants used in kilte awulaelo district, tigray region of ethiopia. J. Ethnobiol. Ethnomed. 2013, 9, 65. [Google Scholar] [CrossRef] [PubMed]
- Tewari, D.N. Monograph on Neem (Azadirachta Indica A. Juss.); International Book Distributors: Dehradun, India, 1992. [Google Scholar]
- Malik, A.; Shaukat, M.; Qureshi, A.; Abdur, R. Comparative effectiveness of chewing stick and toothbrush: A randomized clinical trial. N. Am. J. Med. Sci. 2014, 6, 333. [Google Scholar] [CrossRef]
- Kaushik, A.; Kaushik, M.; Tanwar, R. Ethnomedicine: Applications of Neem (Azadirachta indica) in dentistry. Dent. Hypotheses 2012, 3, 112. [Google Scholar] [CrossRef]
- Chandra, D.; Prasad, K.; Kohli, G.; Bisht, G.; Punetha, V.D.; Khetwal, K.S.; Pandey, H.K. Essential oil composition of Acorus calamus from District-Pithoragarh, Uttarakhad, India. World J. Pharm. Res. 2015, 4, 1158–1166. [Google Scholar]
- Nanda, B.L.; Sultana, S.N.; Radhakrishnan, T.T. Determination of phytochemicals and antioxidant activity of Acorus calamus rhizome. J. Drug Deliv. Ther. 2014, 4, 117–121. [Google Scholar] [CrossRef]
- Rao, N.S.B.; Rajasekhar, D.; Raju, D.C.; Nagaraju, N. Ethno-medicinal notes on some plants of Tirumala hills for dental disorders. Ethnobotany 1996, 8, 88–91. [Google Scholar]
- Abdallah, I.B.; Baatour, O.; Mechrgui, K.; Herchi, W.; Albouchi, A.; Chalghoum, A.; Boukhchina, S. Essential oil composition of walnut tree (Juglans regia L.) leaves from Tunisia. J. Essent. Oil Res. 2016, 28, 545–550. [Google Scholar] [CrossRef]
- Saimbi, C.S.; Shubh, N.; Kapoor, K.K.; Kaushal, S. Clinical effect of Juglans regia on developing dental plaque. J. Int. Clin. Dent. Res. Organ. 2009, 1, 4. [Google Scholar]
- Gyawali, R.; Kim, K.-S. Bioactive volatile compounds of three medicinal plants from Nepal. Kathmandu Univ. J. Sci. Eng. Technol. 2012, 8, 51–62. [Google Scholar] [CrossRef]
- Singla, R.; Jaitak, V. Shatavari (Asparagus racemosus Wild): A review on its cultivation, morphology, phytochemistry and pharmacological importance. Int. J. Pharm. Sci. Res. 2014, 5, 742–757. [Google Scholar] [CrossRef]
- Karuna, D.S.; Dey, P.; Das, S.; Kundu, A.; Bhakta, T. In vitro antioxidant activities of root extract of Asparagus racemosus Linn. J. Tradit. Complement. Med. 2018, 8, 60–65. [Google Scholar] [CrossRef]
- Vani, S.K.; Sivakumar, G.; Geetha, R.V.; Priya, V.V. Evaluation of antibacterial activity of Asparagus racemosus extract–An in vitro study. Drug Invent. Today 2019, 11, 971–973. [Google Scholar]
- Shah, G.; Baghel, U.S. Melaleuca alternifolia: A review of the medicinal uses, pharmacology and phytochemistry. Int. J. ChemTech Res. 2017, 10, 418–427. [Google Scholar]
- Carson, C.F.; Hammer, K.A.; Riley, T.V. Melaleuca alternifolia (tea tree) oil: A review of antimicrobial and other medicinal properties. Clin. Microbiol. Rev. 2006, 19, 50–62. [Google Scholar] [CrossRef]
- Piekarz, T.; Mertas, A.; Wiatrak, K.; Rój, R.; Kownacki, P.; Śmieszek-Wilczewska, J.; Kopczyńska, E.; Wrzoł, M.; Cisowska, M.; Szliszka, E.; et al. The influence of toothpaste containing australian Melaleuca alternifolia oil and ethanolic extract of polish propolis on oral hygiene and microbiome in patients requiring conservative procedures. Molecules 2017, 22, 1957. [Google Scholar] [CrossRef]
- Elgendy, E.A.; Abdel-Moula Ali, S.; Zineldeen, D.H. Effect of local application of tea tree (Melaleuca alternifolia) oil gel on long pentraxin level used as an adjunctive treatment of chronic periodontitis: A randomized controlled clinical study. J. Indian Soc. Periodontol. 2013, 17, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Casarin, M.; Pazinatto, J.; Santos, R.C.V.; Zanatta, F.B. Melaleuca alternifolia and its application against dental plaque and periodontal diseases: A systematic review. Phyther. Res. 2018, 32, 230–242. [Google Scholar] [CrossRef] [PubMed]
- Rajvaidhya, S.; Nagori, B.P.; Singh, G.K.; Dubey, B.K.; Desai, P.; Jain, S. A review on Acacia arabica-an Indian medicinal plant. Int. J. Pharm. Sci. Res. 2012, 3, 1995–2005. [Google Scholar] [CrossRef]
- Ogunbinu, A.O.; Okeniyi, S.; Flamini, G.; Cioni, P.L.; Ogunwande, I.A.; Babalola, I.T. Essential oil composition of Acacia nilotica linn., and Acacia albida delile (Leguminosae) from Nigeria. J. Essent. Oil Res. 2010, 22, 540–542. [Google Scholar] [CrossRef]
- Badshah, L.; Hussain, F. People preferences and use of local medicinal flora in District Tank, Pakistan. J. Med. Plants Res. 2011, 5, 22–29. [Google Scholar]
- Pote, M.; Hirapure, P. Antimicrobial potential of Acacia nilotica extracts on few dental pathogens. Int. J. Pharm. Sci. Res. 2014, 5, 4759. [Google Scholar] [CrossRef]
- Saeedi, R.; Sultana, A.; Rahman, K. Medicinal properties of different parts of Acacia nilotica Linn. (babul), its phytoconstituents and diverse pharmacological activities. Int. J. Pharm. Pharm. Sci. 2020, 12, 8–14. [Google Scholar] [CrossRef]
- Shariatifar, N.; Fathabad, A.E.; Khaniki, G.J.; Nasrabadi, H.G. Evaluation of the antibacterial activity of essential oil and aqueous and ethanolic extracts of Quercus infectoria leaves on food-borne pathogenic bacteria. Int. J. Pharma Sci. Res. 2014, 5, 709–713. [Google Scholar]
- Basri, D.F.; Tan, L.S.; Shafiei, Z.; Zin, N.M. In vitro antibacterial activity of galls of Quercus infectoria Olivier against oral pathogens. Evid.-Based Complement. Altern. Med. 2012, 2012, 632796. [Google Scholar] [CrossRef]
- Sharopov, F.S.; Salimov, A.; Numonov, S.; Bakri, M.; Sangov, Z.; Habasi, M.; Aisa, H.A.; Setzer, W.N. Phytochemical study on the essential oils of Tarragon (Artemisia dracunculus L.) growing in Tajikistan and its comparison with the essential oil of the species in the rest of the world. Nat. Prod. Commun. 2020, 15, 1934578X20977394. [Google Scholar] [CrossRef]
- Raeisi, M.; Tajik, H.; Razavi roohani, S.M.; Maham, M.; Moradi, M.; Hajimohammadi, B.; Naghili, H.; Hashemi, M.; Mehdizadeh, T. Essential oil of tarragon (Artemisia dracunculus) antibacterial activity on Staphylococcus aureus and Escherichia coli in culture media and Iranian white cheese. Iran. J. Microbiol. 2012, 4, 30–33. [Google Scholar] [PubMed]
- Wongkham, S.; Taweechaisupapong, S. Koi: Medicinal plant for oral hygiene. Thai J. Pharmacol. 2005, 27, 137–145. [Google Scholar]
- Phutdhawong, W.; Donchai, A.; Korth, J.; Pyne, S.G.; Picha, P.; Ngamkham, J.; Buddhasukh, D. The components and anticancer activity of the volatile oil fromStreblus asper. Flavour Fragr. J. 2004, 19, 445–447. [Google Scholar] [CrossRef]
- Kumar, G.; Kanungo, S.; Panigrahi, K. Pharmacology & Clinical Research Antimicrobial Effects of Streblus Asper Leaf Extract: A Randomized Controlled Clinical Trial. J. Pharmacol. Clin. Res. 2020, 8. [Google Scholar] [CrossRef]
- Pope, G.V. Notes on Vernonieae (Compositae) in the Flora Zambesiaca Area. Kew Bull. 1990, 45, 697. [Google Scholar] [CrossRef]
- Haghi, G.; Arshi, R.; Ghazian, F.; Hosseini, H. Chemical composition of essential oil of aerial parts of Cichorium intybus L. from iran. J. Essent. Oil-Bear. Plants 2012, 15, 213–216. [Google Scholar] [CrossRef]
- Al-Snafi, A.E. Medical importance of Cichorium intybus-A review. IOSR J. Pharm. 2016, 6, 41–56. [Google Scholar]
- Street, R.A.; Sidana, J.; Prinsloo, G. Cichorium intybus: Traditional Uses, Phytochemistry, Pharmacology, and Toxicology. Evid.-Based Complement. Altern. Med. 2013, 2013, 579319. [Google Scholar] [CrossRef]
- Venkateswarlu, K. Vitex negundo: Medicinal values, biological activities, toxicity studies and phytopharmacological actions. Int. J. Pharm. Phytopharm. Res. 2012, 2, 126–133. [Google Scholar]
- Singh, A.; Sharma, P.K.; Garg, V.K.; Visht, S. Extraction and analysis of essential oil of Nirgundi (Vitex negundo L.). Der Pharm. Sin. 2011, 2, 262–266. [Google Scholar]
- Suganthi, N.; Dubey, S. Phytochemical constituents and pharmacological activities of Vitex negundo Linn. J. Chem. Pharm. Res. 2016, 8, 800–807. [Google Scholar]
- Sanjay, N.; Tiwari, M.M. Antibacterial activity of Nirgundi (Vitex negundo Linn.). Environ. Conserv. J. 2007, 8, 31–33. [Google Scholar]
- Maurya, H.; Rao, V. Article 1007 The Favorable Role of Alkaloids from Vitex negundo in the Management of Human Ailments. Ann. Clin. Pharmacol. Toxicol. 2019, 1, 1007. [Google Scholar]
- Begum, A.; Sandhya, S.; Ali, S.S.; Vinod, K.R.; Reddy, S.; Banji, D. An in-depth review on the medicinal flora Rosmarinus officinalis (Lamiaceae). Acta Sci. Pol. Technol. Aliment. 2013, 12, 61–73. [Google Scholar]
- de Macedo, L.M.; dos Santos, É.M.; Militão, L.; Tundisi, L.L.; Ataide, J.A.; Souto, E.B.; Mazzola, P.G. Rosemary (Rosmarinus officinalis L., syn Salvia rosmarinus Spenn.) and Its Topical Applications: A Review. Plants 2020, 9, 651. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, J.R.; Camargo, S.E.A.; de Oliveira, L.D. Rosmarinus officinalis L. (rosemary) as therapeutic and prophylactic agent. J. Biomed. Sci. 2019, 26, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Valones, M.A.A.; Silva, I.C.G.; Gueiros, L.A.M.; Leão, J.C.; Caldas, A.F.; Carvalho, A.A.T. Clinical assessment of rosemarybased toothpaste (Rosmarinus officinalis linn.): A randomized controlled double-blind study. Braz. Dent. J. 2019, 30, 146–151. [Google Scholar] [CrossRef]
- Shankar, R.; Lavekar, G.S.; Deb, S.; Sharma, B.K.; Rawat, M.S. Distribution, conservation and folk uses of Vaibidang (Embelia ribes Burm. f.). Int. J. Biodivers. Conserv. 2012, 4, 525–529. [Google Scholar] [CrossRef]
- Radhakrishnan, N.; Gnanamani, A.; Mandal, A.B. A potential antibacterial agent Embelin, a natural benzoquinone extracted from Embelia ribes. Biol. Med. 2011, 3, 1–7. [Google Scholar]
- Radhakrishnan, N.; Gnanamani, A. 2, 5-dihydroxy-3-undecyl-1, 4-benzoquinone (embelin)-A second solid gold of India-A review. Int. J. Pharm. Pharm. Sci. 2014, 6, 2014. [Google Scholar]
- Lal, B.; Mishra, N. Importance of Embelia ribes: An update. Int. J. Pharm. Sci. Res. 2013, 4, 3823. [Google Scholar] [CrossRef]
- Asadulla, S.; Ramandang, R. Pharmacognosy of Embelia ribes Burm. f. Int. J. Res. Pharm. Chem. 2011, 1, 1236–1251. [Google Scholar]
- Paulraj, J.; Govindarajan, R.; Palpu, P. The Genus Spilanthes Ethnopharmacology, Phytochemistry, and Pharmacological Properties: A Review. Adv. Pharmacol. Sci. 2013, 2013, 510298. [Google Scholar] [CrossRef]
- Srinath, J.; Laksmi, T. Therapeutic potential of Spilanthes acmella–A dental note. Int. J. Pharm. Sci. Res. 2014, 25, 151–153. [Google Scholar]
- Chadha, M.L. Indigenous vegetables of India with potentials for improving livelihood. Acta Hortic. 2009, 806, 579–586. [Google Scholar] [CrossRef]
- Nakatani, N.; Nagashima, M. Pungent Alkamides from Spilanthes acmella L. var. oleracea Clarke. Biosci. Biotechnol. Biochem. 1992, 56, 759–762. [Google Scholar] [CrossRef]
- Noumi, E.; Dibakto, T.W. Medicinal plants used for peptic ulcer in the Bangangte region, western Cameroon. Fitoterapia 2000, 71, 406–412. [Google Scholar] [CrossRef]
- Chopra, R.N.; Nayar, S.L.; Chopra, I.C. Glossary of Indian Medicinal Plants. Q. Rev. Biol. 1958, 33, 156. [Google Scholar] [CrossRef]
- Kala, C.P. Ethnomedicinal botany of the Apatani in the Eastern Himalayan region of India. J. Ethnobiol. Ethnomed. 2005, 1, 11. [Google Scholar] [CrossRef] [PubMed]
- Datta, A.K.; Datta, A.K.; Saha, A.; Bhattacharya, A.; Mandal, A.; Paul, R.; Sengupta, S. Black cumin (Nigella sativa L.)-a review Comperative assessment of immediate and heritable effect of nanoparticles and conventional mutagen. View project black cumin (Nigella sativa L.)-A review. J. Plant Dev. Sci. 2012, 4, 1–43. [Google Scholar]
- Ghahramanloo, K.H.; Kamalidehghan, B.; Akbari Javar, H.; Teguh Widodo, R.; Majidzadeh, K.; Noordin, M.I. Comparative analysis of essential oil composition of iranian and indian Nigella sativa L. Extracted using supercritical fluid extraction and solvent extraction. Drug Des. Devel. Ther. 2017, 11, 2221–2226. [Google Scholar] [CrossRef]
- Mekhemar, M.; Hassan, Y.; Dörfer, C. Nigella sativa and thymoquinone: A natural blessing for periodontal therapy. Antioxidants 2020, 9, 1260. [Google Scholar] [CrossRef]
- Nordin, A.; Kamal, H.; Yazid, M.D.; Saim, A.; Idrus, R. Effect of Nigella sativa and its bioactive compound on type 2 epithelial to mesenchymal transition: A systematic review. BMC Complement. Altern. Med. 2019, 19, 290. [Google Scholar] [CrossRef]
- Rivera, D.; Allkin, R.; Obón, C.; Alcaraz, F.; Verpoorte, R.; Heinrich, M. What is in a name? the need for accurate scientific nomenclature for plants. J. Ethnopharmacol. 2014, 152, 393–402. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Gewali, M.B.; Awale, S. Aspects of Traditional Medicine in Nepal; University of Toyama: Toyama, Japan, 2008. [Google Scholar]
- Patino, L.O.J.; Prieto, R.J.A.; Cuc, S.L.E. Zanthoxylum Genus as Potential Source of Bioactive Compounds. In Bioactive Compounds in Phytomedicine; InTech: London, UK, 2012. [Google Scholar]
- Ocheng, F.; Bwanga, F.; Joloba, M.; Borg-Karlson, A.K.; Gustafsson, A.; Obua, C. Antibacterial activities of extracts from Ugandan medicinal plants used for oral care. J. Ethnopharmacol. 2014, 155, 852–855. [Google Scholar] [CrossRef]
- Wang, H.; Ren, D. Controlling Streptococcus mutans and Staphylococcus aureus biofilms with direct current and chlorhexidine. AMB Express 2017, 7, 204. [Google Scholar] [CrossRef]
- Mukhtar, H.M.; Kalsi, V. A review on medicinal properties of zanthoxylum armatum DC. Res. J. Pharm. Technol. 2018, 11, 2131–2138. [Google Scholar] [CrossRef]
- Tsunozaki, M.; Lennertz, R.C.; Vilceanu, D.; Katta, S.; Stucky, C.L.; Bautista, D.M. A “toothache tree” alkylamide inhibits Aδ mechanonociceptors to alleviate mechanical pain. J. Physiol. 2013, 591, 3325–3340. [Google Scholar] [CrossRef] [PubMed]
- Kalra, K.; Vasthare, R.; Shenoy, P.A.; Vishwanath, S.; Singhal, D.K. Antibacterial efficacy of essential oil of two different varieties of ocimum (tulsi) on oral microbiota-an invitro study. Indian J. Public Health Res. Dev. 2019, 10, 188–193. [Google Scholar] [CrossRef]
- Prakash, P.; Gupta, N. Therapeutic uses of Ocimum sanctum Linn (Tulsi) with a note on eugenol and its pharmacological actions: A short review. Indian J. Physiol. Pharmacol. 2005, 49, 125–131. [Google Scholar]
- Frisch, J.; Bhaskar, S.N. Tissue Response to Eugenol-containing Periodontal Dressings. J. Periodontol. 1967, 38, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Ahmad, A.; Manzoor, N.; Khan, L.A. Antifungal Activities of Ocimum sanctum Essential Oil and its Lead Molecules. Nat. Prod. Commun. 2010, 5, 1934578X1000500235. [Google Scholar] [CrossRef]
- Agarwal, P.; Nagesh, L. Murlikrishnan Evaluation of the antimicrobial activity of various concentrations of Tulsi (Ocimum sanctum) extract against Streptococcus mutans: An in vitro study. Indian J. Dent. Res. 2010, 21, 357–359. [Google Scholar] [CrossRef]
- Kamal, S.A.; Hameed, I.H.; Mohammed, G.J. Antimicrobial, Anti-inflammatory, Analgesic Potential and Cytotoxic Activity of Salvadora persica: A Review. Artic. Indian J. Public Health Res. Dev. 2018, 9, 393–398. [Google Scholar] [CrossRef]
- Abhary, M.; Al-Hazmi, A.A. Antibacterial activity of Miswak (Salvadora persica L.) extracts on oral hygiene. J. Taibah Univ. Sci. 2016, 10, 513–520. [Google Scholar] [CrossRef]
- Khan, M.; Alkhathlan, H.Z.; Khan, S.T. Antibiotic and antibiofilm activities of Salvadora persica L. Essential oils against Streptococcus mutans: A detailed comparative study with chlorhexidine digluconate. Pathogens 2020, 9, 66. [Google Scholar] [CrossRef] [PubMed]
- Palanna, R.M. Eucalyptus in India, Conservator of Forests, Kanara Circle, Karnataka, India. Available online: http://www.fao.org/3/AC772E/ac772e06.htm (accessed on 1 May 2021).
- Mayaud, L.; Carricajo, A.; Zhiri, A.; Aubert, G. Comparison of bacteriostatic and bactericidal activity of 13 essential oils against strains with varying sensitivity to antibiotics. Lett. Appl. Microbiol. 2008, 47, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Bachir, R.G.; Benali, M. Antibacterial activity of the essential oils from the leaves of Eucalyptus globulus against Escherichia coli and Staphylococcus aureus. Asian Pac. J. Trop. Biomed. 2012, 2, 739–742. [Google Scholar] [CrossRef]
- Gonçalves, G.M.S.; Bottaro, M.; Nilson, A.C. Effect of the Thymus vulgaris essential oil on the growth of Streptococcus mutans. J. Basic Appl. Pharm. Sci. Rev. Ciênc Farm. Básica Apl 2011, 32, 375–380. [Google Scholar]
- Champion, S.H.; Seth, S.K.; Khattak, G.M. Manual of silviculture for Pakistan. Man. Silvic. 1965, 523–543. [Google Scholar]
- Webb, D.B.; Wood, P.J.; Smith, J.P.; Henman, G.S. A Guide to Species Selection for Tropical and Sub-Tropical Plantations; University of Oxford: Oxford, UK, 1984. [Google Scholar]
- Vennila, K.; Naziya, K.; Elavarasu, S.; Abinaya, P.; Elanchezhiyan, S. Thangakumaran Evaluation of anti-plaque microbial activity of Azadirachta indica (neem oil) in vitro: A pilot study. J. Pharm. Bioallied Sci. 2012, 4, 394. [Google Scholar] [CrossRef] [PubMed]
- Lakshmi, T.; Krishnan, V.; Rajendran, R.; Madhusudhanan, N. Azadirachta indica: A herbal panacea in dentistry-An update. Pharmacogn. Rev. 2015, 9, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Babatunde, D.E.; Otusemade, G.O.; Efeovbokhan, V.E.; Ojewumi, M.E.; Bolade, O.P.; Owoeye, T.F. Chemical composition of steam and solvent crude oil extracts from Azadirachta indica leaves. Chem. Data Collect. 2019, 20, 100208. [Google Scholar] [CrossRef]
- Umamaheshwari, N.; Rekha, A. Sweet flag: (Acarus calamus)-An incredible medicinal herb. J. Pharmacogn. Phytochem. 2018, 7, 15–22. [Google Scholar]
- Shetty, R.N.; Shetty, S.B.; Janardhanan, S.; Shetty, S.; Shetty, S.; Raj, K. Comparative evaluation of effect of use of toothbrush with paste and munident on levels of Streptococcus mutans and gingival health in children: An in vivo study. J. Indian Soc. Pedod. Prev. Dent. 2017, 35, 162–166. [Google Scholar] [CrossRef]
- Deshpande, R.R.; Kale, A.A.; Ruikar, A.D.; Panvalkar, P.S.; Kulkarni, A.A.; Deshpande, N.R.; Salvekar, J.P. Antimicrobial activity of different extracts of Juglans regia L. against oral microflora. Int. J. Pharm. Pharm. Sci. 2011, 3, 200–201. [Google Scholar]
- Zakavi, F.; Golpasand Hagh, L.; Daraeighadikolaei, A.; Farajzadeh Sheikh, A.; Daraeighadikolaei, A.; Leilavi Shooshtari, Z. Antibacterial effect of Juglans regia bark against oral pathologic bacteria. Int. J. Dent. 2013, 2013, 854765. [Google Scholar] [CrossRef]
- Carrol, D.H.; Chassagne, F.; Dettweiler, M.; Quave, C.L. Antibacterial activity of plant species used for oral health against Porphyromonas gingivalis. PLoS ONE 2020, 15, e0239316. [Google Scholar] [CrossRef]
- Nancy, P.; Manasi, M.; Varghese, A. Antiplaque activity of Juglans regia L. and characterization of Juglone from Juglans regia L. Am. J. Biochem. Biotechnol. 2011, 7, 29–31. [Google Scholar] [CrossRef]
- Lohani, H.; Zafar, H.S.; Chauhan, N.K.; Mohan, C.M. Essential oil composition of leaves and berries of Juniperus communis and Juniperus indica from Uttarakhand Himalaya. J. Med. Aromat. Plant. Sci. 2010, 32, 199–201. [Google Scholar]
- Megersa, M.; Jima, T.T.; Goro, K.K. The Use of Medicinal Plants for the Treatment of Toothache in Ethiopia. Evid.-Based Complement. Altern. Med. 2019, 2019, 2645174. [Google Scholar] [CrossRef]
- Jiménez-Gayosso, S.I.; Lara-Carrillo, E.; Scougall-Vilchis, R.J.; Morales-Luckie, R.A.; Medina, S.C.E.; Velázquez-Enríquez, U.; Maupomé, G.; Herrera-Serna, B. Remineralizing effect of xilitol, Juniperus communis and Camellia sinensis added to a toothpaste: An in vitro study. Odovtos-Int. J. Dent. Sci. 2020, 22, 71–79. [Google Scholar] [CrossRef]
- Zhilyakova, E.T.; Novikov, O.O.; Lysykh, E.G.; Zalivskaya, A. V Study of physical, chemical and consumer properties for development of dental gel basis aimed to cure of parodont diseases. J. Int. Pharm. Res. 2018, 45, 416–419. [Google Scholar]
- Raina, R.; Verma, P.K.; Peshin, R.; Kour, H. Potential of Juniperus communis L as a nutraceutical in human and veterinary medicine. Heliyon 2019, 5, e02376. [Google Scholar] [CrossRef]
- Shah, G.; Baghel, U.S. Pharmacognostic standardization of the leaf of Melaleuca alternifolia (Maiden & Betche) Cheel. Afr. J. Tradit. Complement. Altern. Med. AJTCAM 2017, 14, 1–11. [Google Scholar] [CrossRef][Green Version]
- Oliveira, T.R.; Teixeira, A.L.; Barbosa, J.P.; Busato de Feiria, S.N.; Boni, G.C.; Maia, F.; Anibal, P.C.; Wijesinghe, G.K.; Höfling, J.F. Melaleuca spp. essential oil and its medical applicability. A Brief Review. Braz. J. Nat. Sci. 2020, 3, 249. [Google Scholar] [CrossRef]
- Brophy, J.J.; Davies, N.W.; Southwell, I.A.; Stiff, I.A.; Williams, L.R. Gas chromatographic quality control for oil of Melaleuca terpinen-4-ol type (Australian tea tree). J. Agric. Food Chem. 1989, 37, 1330–1335. [Google Scholar] [CrossRef]
- Buck, D.S. Comparison of two topical preparations for the treatment of onyehornycosis: Melaleuca altemifilia (tea tree) oil and clotrimazole. J. Fam. Pract. 1994, 38, 601–605. [Google Scholar] [PubMed]
- Banso A Phytochemical and antibacterial investigation of bark extracts of Acacia nilotica. J. Med. Plants Res. 2009, 3, 82–085.
- Ikram, M.; Nowshad, F. Constituents of Quercus infectoria. Planta Med. 1977, 31, 286–287. [Google Scholar] [CrossRef]
- Machado, T.B.; Pinto, A.V.; Pinto, M.C.F.R.; Leal, I.C.R.; Silva, M.G.; Amaral, A.C.F.; Kuster, R.M.; Netto-dosSantos, K.R. In vitro activity of Brazilian medicinal plants, naturally occurring naphthoquinones and their analogues, against methicillin-resistant Staphylococcus aureus. Int. J. Antimicrob. Agents 2003, 21, 279–284. [Google Scholar] [CrossRef]
- Fogliani, B.; Raharivelomanana, P.; Bianchini, J.P.; Bouraïma-Madjèbi, S.; Hnawia, E. Bioactive ellagitannins from Cunonia macrophylla, an endemic Cunoniaceae from New Caledonia. Phytochemistry 2005, 66, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Hatano, T.; Kusuda, M.; Inada, K.; Ogawa, T.; Shiota, S.; Tsuchiya, T.; Yoshida, T. Effects of tannins and related polyphenols on methicillin-resistant Staphylococcus aureus. Phytochemistry 2005, 66, 2047–2055. [Google Scholar] [CrossRef]
- Greenish, H.G. Materia Medica, 3rd ed.; Scientific Publisher: Jodhpur, India, 1999. [Google Scholar]
- Ahmad, W.; Zeenat, F.; Hasan, A.; Abdullah, A.; Nargis, A.; Tarannum, T. Mazu (Quercus infectoria): An overview. Indian J. Unani Med. 2011, 4, 17–22. [Google Scholar]
- Nigam, M.; Atanassova, M.; Mishra, A.P.; Pezzani, R.; Devkota, H.P.; Plygun, S.; Salehi, B.; Setzer, W.N.; Sharifi-Rad, J. Bioactive compounds and health benefits of Artemisia species. Nat. Prod. Commun. 2019, 14, 1934578X19850354. [Google Scholar]
- Deans, S.G.; Simpson, E.J.M. Artemisia dracunculus. In Artemisia; Wright, C.W., Ed.; Taylor & Francis Ltd.: London, UK; New York, NY, USA, 2001; pp. 91–97. [Google Scholar]
- Wongkham, S.; Laupattarakasaem, P.; Pienthaweechai, K.; Areejitranusorn, P.; Wongkham, C.; Techanitiswad, T. Antimicrobial activity of Streblus asper leaf extract. Phyther. Res. 2001, 15, 119–121. [Google Scholar] [CrossRef]
- Palombo, E.A. Traditional Medicinal Plant Extracts and Natural Products with Activity against Oral Bacteria: Potential Application in the Prevention and Treatment of Oral Diseases. Evid.-Based Complement. Altern. Med. 2011, 2011, 680354. [Google Scholar] [CrossRef] [PubMed]
- Taweechaisupapong, S.; Klanrit, P.; Singhara, S.; Pitiphat, W.; Wongkham, S. Inhibitory effect of Streblus asper leaf-extract on adhesion of Candida albicans to denture acrylic. J. Ethnopharmacol. 2006, 106, 414–417. [Google Scholar] [CrossRef]
- Saeed, M.; Abd El-Hack, M.E.; Alagawany, M.; Arain, M.A.; Arif, M.; Mirza, M.A.; Naveed, M.; Chao, S.; Sarwar, M.; Sayab, M.; et al. Chicory (Cichorium intybus) herb: Chemical composition, pharmacology, nutritional and healthical applications. Int. J. Pharmacol. 2017, 13, 351–360. [Google Scholar] [CrossRef]
- Judžentienė, A.; Būdienė, J. Volatile constituents from aerial parts and roots of Cichorium intybus L. (chicory) grown in Lithuania. Chemija 2008, 19, 25–28. [Google Scholar]
- Huang, H.C.; Chang, T.Y.; Chang, L.Z.; Wang, H.F.; Yih, K.H.; Hsieh, W.Y.; Chang, T.M. Inhibition of melanogenesis Versus antioxidant properties of essential oil extracted from leaves of Vitex negundo linn and chemical composition analysis by GC-MS. Molecules 2012, 17, 3902–3916. [Google Scholar] [CrossRef]
- Ullah, Z.; Ullah, R.; Ahmad, I.; Haider, S. Phytochemical and Biological Evaluation of Vitex Negundo Linn: A Review. Int. J. Pharm. Sci. Res. 2012, 3, 2421–2431. [Google Scholar]
- Patki; sunita amruthesh; Tandur; tandur; Malini Clinical evaluation of a novel herbal dental cream in plaque formation: A double-blind, randomized, controlled clinical trial. J. Exp. Pharmacol. 2010, 2010, 105. [CrossRef]
- Simbo, D.J. An ethnobotanical survey of medicinal plants in Babungo, Northwest Region, Cameroon. J. Ethnobiol. Ethnomed. 2010, 6, 8. [Google Scholar] [CrossRef]
- Tangjang, S.; Namsa, N.D.; Aran, C.; Litin, A. An ethnobotanical survey of medicinal plants in the Eastern Himalayan zone of Arunachal Pradesh, India. J. Ethnopharmacol. 2011, 134, 18–25. [Google Scholar] [CrossRef]
- Nadar, B.; Usha, G.; Lakshminarayan, N. Comparative evaluation of efficacy of 4% tulsi extract (Ocimum sanctum), fluoridated and placebo dentifrices against gingivitis and plaque among 14-15 years school children in Davangere City, India-A triple blinded randomized clinical trial. Contemp. Clin. Dent. 2020, 11, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Malik, A. Comparative clinical effects of Salvadora persica oral rinse and a phenolic commercial mouth wash on human oral health; An Invivo randomized trial. J. Pak. Dent. Assoc. 2021, 30, 87–93. [Google Scholar] [CrossRef]
- Sukhabogi, J.R.; Chandra Shekar, B.R.; Venkata Ramana, I.; Yadav, S.S.; Satish Kumar, G.; Harita, N. Antiplaque efficacy of tooth and gums tonic, Hiora-GA gel, and Spirogyl Gum Paint in comparison with chlorhexidine M gel: A double-blind randomized control trial. Contemp. Clin. Dent. 2017, 8, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Hosny, N.S.; El Khodary, S.A.; El Boghdadi, R.M.; Shaker, O.G. Effect of Neem (Azadirachta indica) versus 2.5% sodium hypochlorite as root canal irrigants on the intensity of post-operative pain and the amount of endotoxins in mandibular molars with necrotic pulps: A randomized controlled trial. Int. Endod. J. [CrossRef]
- Taalab, M.R.; Mahmoud, S.A.; Moslemany, R.M.E.; Abdelaziz, D.M. Intrapocket application of tea tree oil gel in the treatment of stage 2 periodontitis. BMC Oral Health 2021, 21, 239. [Google Scholar] [CrossRef] [PubMed]
- Saliasi, I.; Llodra, J.C.; Bravo, M.; Tramini, P.; Dussart, C.; Viennot, S.; Carrouel, F. Effect of a toothpaste/mouthwash containing carica papaya leaf extract on interdental gingival bleeding: A randomized controlled trial. Int. J. Environ. Res. Public Health 2018, 15, 2660. [Google Scholar] [CrossRef] [PubMed]
- Carrouel, F.; Valette, M.; Gadea, E.; Esparcieux, A.; Illes, G.; Langlois, M.E.; Perrier, H.; Dussart, C.; Tramini, P.; Ribaud, M.; et al. Use of an antiviral mouthwash as a barrier measure in the SARS-CoV-2 transmission in adults with asymptomatic to mild COVID-19: A multicentre, randomized, double-blind controlled trial. Clin. Microbiol. Infect. 2021. [Google Scholar] [CrossRef] [PubMed]

| Sr. No. | Common Name | Kingdom | Phylum | Class | Order | Family | Genus | Species | Binomial Classification |
|---|---|---|---|---|---|---|---|---|---|
| 1. | Tejphal | Plantae | Magnoliophyta | Magnoliopsida | Sapindales | Rutaceae | Zanthoxylum | Zanthoxylum armatum | Zanthoxylum armatum DC. |
| 2. | Tulsi | Plantae | Magnoliophyta | Magnoliopsida | Lamiales | Lamiaceae | Ocimum | Ocimum sanctum | Ocimum sanctum Linn. |
| 3. | Miswak | Plantae | Magnoliophyta | Magnoliopsida | Brassicales | Salvadoraceae | Salvadora | Salvadora persica | Salvadora persica L. |
| 4. | Nilgiri | Plantae | Magnoliophyta | Magnoliopsida | Myrtales | Myrtaceae | Eucalyptus | Eucalyptus globulus | Eucalyptus globulus Labill. |
| 5. | Banajwain | Plantae | Magnoliophyta | Magnoliopsida | Lamiales | Lamiaceae | Thymus | Thymus vulgaris | Thymus vulgaris L. |
| 6. | Neem | Plantae | Magnoliophyta | Magnoliopsida | Sapindales | Meliaceae | Azadirachta | Azadirachta indica | Azadirachta indica A. Juss. |
| 7. | Vacha | Plantae | Magnoliophyta | Liliopsida | Arales | Acoraceae | Acorus | Acorus calamus | Acorus calamus L. |
| 8. | Akhrot | Plantae | Magnoliophyta | Magnoliopsida | Juglandales | Juglandaceae | Juglans | Juglans regia | Juglans regia L. |
| 9. | Satavari | Plantae | Magnoliophyta | Liliopsida | Asparagales | Asparagaceae | Asparagus | Asparagus racemosus | Asparagus racemosus Willd. |
| 10. | Aaraar | Plantae | Coniferophyta | Pinopsida | Pinales | Cupressaceae | Juniperus | Juniperus communis | Juniperus communis L. |
| 11. | Tea tree | Plantae | Magnoliophyta | Magnoliopsida | Myrtales | Myrtaceae | Melaleuca | Melaleuca alternifolia | Melaleuca alternifolia (Maiden and Betche) Cheel |
| 12. | Babul | Plantae | Magnoliophyta | Magnoliopsida | Fabales | Fabaceae | Acacia | Acacia nilotica | Acacia nilotica (L.) Delile |
| 13. | Baloot | Plantae | Magnoliophyta | Magnoliopsida | Fagales | Fagaceae | Quercus | Quercus infectoria | Quercus infectoria G. Olivier |
| 14. | Estragon | Plantae | Magnoliophyta | Magnoliopsida | Asterales | Asteraceae | Artemisia | Artemisia dracunculus | Artemisia dracunculus L. |
| 15. | Khoi | Plantae | Magnoliophyta | Magnoliopsida | Urticales | Moraceae | Streblus | Streblus asper | Streblus asper Lour. |
| 16. | Chicory | Plantae | Magnoliophyta | Magnoliopsida | Asterales | Asteraceae | Cichorium | Cichorium intybus | Cichorium intybus L. |
| 17. | Nirgundi | Plantae | Magnoliophyta | Magnoliopsida | Lamiales | Verbenaceae | Vitex | Vitex negundo | Vitex negundo L. |
| 18. | Rosemary | Plantae | Magnoliophyta | Magnoliopsida | Lamiales | Lamiaceae | Rosmarinus | Rosmarinus officinalis | Rosmarinus officinalis L. |
| 19. | Vaibidang | Plantae | Magnoliophyta | Magnoliopsida | Ericales | Primulaceae | Embelia | Embelia ribes Burm.f. | Embelia ribes Burm.f. |
| 20. | Akalkara | Plantae | Magnoliophyta | Magnoliopsida | Asterales | Asteraceae | Spilanthes | Spilanthes acmella | Spilanthes acmella (L.) L. |
| 21. | Kalonji | Plantae | Magnoliophyta | Magnoliopsida | Ranunculales | Ranunculaceae | Nigella | Nigella Sativa | Nigella sativa L. |
| Botanical Name (Common Name) | Location | Extraction Method or Type of Solvent | Essential Oil Components | Study Type (In Vitro/In Vivo/Clinical Trial) and Dose of the Extract | Role in Oral Health |
|---|---|---|---|---|---|
| Zanthoxylum armatum DC. (Tejphal, Tumbru) | India: Kashmir to Bhutan, China, Taiwan, Malaysia, Japan [23] | Hydrodistillation method, Analyzed-GC-MS | Linalool (53.05%), Limonene (11.39%), Myrcene (3.69%), α-pinene (4.08%), Bergamot mint oil (12.73%) [23] | Study—In vitro (antibacterial-) on Streptococcus faecalis, S. aureus, Proteus vulgaris, Klebsiella pneumoniae | |
| Dose: 10 mg/well | |||||
| Essential oil extracted from seeds [23] | Application: Essential oil | ||||
| [24] | Gum bleeding, Mouth Freshener, Toothache, Toothpowder, Tooth Cleaning [25] | ||||
| Ocimum sanctum Linn. (Holy Basil, Tulsi) | India (Uttar Pradesh) Andaman and Nicobar, Africa, South America, Brazil [26] | Hydrodistillation method, Analyzed-GC-MS | Caryophyllene (22.265%), α-caryophyllene (2.071%), α-pinene (0.125%), copaene (1.637%) and eugenol (15.906%) [27] | In vivo (clinical trial) on humans for efficiency of mouth wash containing tulsi, VAS score for burning sensation—Pre-treatment (5.33 ± 1.80), Post-treatment (2.44 ± 2.10) | Oil extract used to treat toothache |
| Essential oil extracted from dried leaves [27] | Dose: 10 mL (thrice)/day–one week), | ||||
| Application: Mouthwash [28] | Dried leaves used to treat gingival and periodontal diseases [29] | ||||
| Salvadora persica L. (Miswak) | India, East, Southern and North Africa, South and West Asia, Arabic Peninsula [30] | Hydrodistillation method, Analyzed-GC-MS | α-caryophellene (13.4%), 1,8-cineole (eucalyptol) (46%), 9-epi. -(E.)-caryophellene, β-pinene (6.3%) [31] | In vivo (clinical trial) on humans for efficiency of miswak toothpaste against cariogenic bacteria, | Antigingivitis, anti-cariogenic, antiplaque, whitening properties, orthodontic chain preservation and promotion of gingival wound healing [10] |
| Essential oil extracted from stem [31] | Dose: twice/day (2 weeks) | ||||
| Application: Toothpaste [32] | |||||
| Eucalyptus globulus Labill. (Nilgiri) | India: Goa, Gujrat, Haryana, Punjab, Uttar Pradesh. Albina, Spain, Uganda, Cambodia [33] | Analyzed by GC-MS, hydrodistillation extraction method, essential oil extracted from leaves [34] | β-pinene 18.54%, eucalyptol (1,8-cineole) 54.79%, para cymene 1.60%,β-eudesmol 4.68%, α-phellandrene 2.06%, α-pinene 11.46% and gamma-eudesmol 1.20% [34] | In vivo (clinical trial) on humans, | For treatment of toothache, sore throat, halitosis in Cameroon mouthwash gargle of Eucalyptus saligna is used [3] |
| Plaque index score—Baseline (1.485 ± 0.34), After 14 days (1.254 ± 0.58). | |||||
| Dose: 10 mL twice/day (14 days) | |||||
| Application—Gargle, Mouthwash [35] | |||||
| Thymus vulgaris L. (Thyme, Banajwain) | India (Western Himalayas and Nilgiris), Spain, European countries, Svizzera, France, Italy, Portuguese Republic, Bulgaria, and Ellas [36] | Steam distillation method, Analyzed- GC-MS | Thymol (3.82%), α-thymol (38.71%) camphene (0.13%), caryophyllene (0.915), humulene (0.22%), α-terpineol (0.285) and ρ-cymene (2.77%) [20] | In vitro antimicrobial effect against Streptococcus mutans (ATCC 25175), | |
| Essential oil extracted from leaves [20] | MIC value (essential oil)—100 μg/mL (1%), | ||||
| Application: Mouthwash [37] | Used in toothpaste, mouth rinse, and aromatherapy for prevention and treatment of oral infection [12,38] | ||||
| Azadirachta indica (Neem) | Afghanistan, Pakistan, India, Sri Lanka, Bangladesh, Myanmar, and China [39] | Hydrodistillation method, Analyzed-GC-MS | Hexadecanoic acid (34.0%), oleic acid (15.7%), 5,6-dihydro-2,4,6-triethyl-(4H)-1,3,5-dithiazine (11.7%), methyl oleate (3.8%), and eudesm-7(11)-en-4-ol (2.7%) [16] | In vivo (clinical trial) on humans, | |
| Gingival index score (Chewing stick)—Pre-intervention (0.31 ± 0.44), Post-intervention (0.16 ± 0.29), | |||||
| Essential oil extracted from seeds [16] | Dose—Neem (chewing stick) 20 cm × 20 mm, | ||||
| Application: Chewing | |||||
| [40] | Neem bark extract used in toothpaste or tooth powder. Leaf extract used in mouth rinses [41] | ||||
| Acorus calamus L. (Sweet flag, Vacha) | India, Central Asia, Eastern Europe, Jammu Kashmir, Himachal Pradesh, Manipur, Naga land, Uttarakhand [19] | Steam distillation method, Analyzed- GC-MS | |||
| Essential oil extracted from leaves [42] | α-Asarone (16.54%), (E)-Methyl isoeugenol (5.06%), γ-Cadinene (3.00)%, α-pinene (2.96%) and Citronellal (2.82%) [42] | In vitro antioxidant activity of Acorus calamus (rhizome). DPPH method: IC50 value (acetone extract of rhizome)—5 μg/mL [43] | Rhizome part is used for the treatment of dental disorders [44] | ||
| Juglans regia L. (Walnut, Akhrot) | China, United State, Jammu and Kashmir, Himachal Pradesh, Arunachal Pradesh, Uttarakhand [21] | Hydrodistillation method, Analyzed GC-MS, GC-FIDEssential oil extracted from leaves [45] | Caryophyllene oxide (16.9 to 27.4%), | In vivo (clinical trial) effect of Juglans regia dental plaque in humans, Dose: twice/day (3 days), | Bark extract used in oral cavity hygiene, treatment of gingivitis, dental plaque, cleaning of teeth [21] |
| β-Caryophyllene (4.0 to 22.5%), Germacrene (1.2 to 9.4%) and | 2% ether extract (bark) reported maximum plaque inhibition (32%), | ||||
| β-Pinene (2.8 to 9.5%) [45] | Application: extract directly applied on tooth surface [46] | ||||
| Asparagus racemosus (Satavari) | Sri Lanka, India, Himalayas, Australia, Africa [17] | Solvent extraction method, Analyzed by GC-MS | Borneol (26.40%), myrtanol (13.72%), pinocarveol (2.37%), 2-ethylhexanol (1.76%) perillaldehyde (8.97%) [47,48] | In vitro antioxidant activity of root extract, | |
| Essential oil extracted from aerial parts [47,48] | DPPH method: IC50 value (ethanolic extract of root)—468.57 ± 3.002 μg/mL | ||||
| [49] | Antibacterial properties against caries causing oral pathogens [50] | ||||
| Melaleuca alternifolia (Tea tree oil) | India (Ansari et al., 2006), Australia [51] | Steam distillation method, analyzed by GC and GC-MS, essential oil extracted from leaves and terminal branches [52] | Terpinen-4-ol, p-cymene, α-terpinene, γ-terpinene, 1,8-cineole, α-pinene and α-terpinol [52] | In vivo (clinical trial) effect of Melaleuca alternifolia essential oil on dental plaque in humans in the form of toothpaste along with ethanolic extract of Polish propolis. | |
| Comparison after 7 and 28 days of using toothpaste. | |||||
| Result: Approximal plaque index (API)—Before treatment; 64.58 ± 22.38%. After treatment, | |||||
| 7 days—(49.00 ± 25.32%, p < 0.006) and after 28 days—(39.39 ± 20.60%, p < 0.0002) [53] | Periodontitis [54], Relieve from bad breath, bleeding gums, and plaque [55] | ||||
| Acacia nilotica (Babul) | India, Nepal, Pakistan, Arabian Peninsula, Africa, South Africa, Egypt [56] | Hydrodistillation method and analyzed by GC-FID and GC/MS. Essential oil extracted from the bark, leaves [57] | Menthol (34.9%), limonene (15.3%), α-Curcumene (6.9%) and carvacrol (4.1%) [57] | In vitro (antibacterial) on Lactobacillus acidophilus, Streptococcus sanguinis, S. salivarius, and Aggregatibacter actinomycetemcomitans. | To cure mouth ulcers [56], |
| Dosage: Concentration of extract ranging between 5 and 30 mL in different test tubes. Incubated at 37 °C for 24 h. | To treat toothache and for cleaning teeth [58], | ||||
| MIC value of bark extract of Lactobacillus acidophilus, Streptococcus sanguinis, S. salivarius, Aggregatibacter actinomycetemcomitans is 40,35,35, and 45, respectively [59] | and for sore throat [60] | ||||
| Quercus infectoria (Baloot) | India, Nepal, Iran, Greece, Syria [61] | Steam distillation (Clevenger apparatus)-Aqueous and ethanolic extract of essential oil from the galls [61] | Tannins 50–70%, gallic acid (2–4%), ellagic acid [61] | In vitro study for dental caries and plaque. Methanol and acetone extracts were screened against bacteria Streptococcus mutans ATCC 25175, Streptococcus salivarius ATCC 13419, Porphyromonas gingivalis ATCC 33277, and Fusobacterium nucleatum ATCC 25586. | Used to treat gum infections, gingivitis, and toothache [62] |
| MIC value of methanol and acetone extract is 0.16 and 0.63 mg/mL, respectively, while MBC value for methanol and acetone extract is 0.31–1.25 mg/mL and 0.31–2.50 mg/mL, respectively [62] | |||||
| Artemisia dracunculus (Estragon) | Asia and central Europe | Hydrodistillation method for extraction and GC-MS method for the identification. Extraction of essential oil from aerial parts [61] | Estragol (methyl chavicol), (E)- anethole, capillene, methyl eugenol, (E)-β-ocimene, (E)-α-ocimene, (Z)-β-ocimene limonene, α-pinene, α-terpinolene, isoelemicin, elemicin 5-phenyl-1,3- pentadiyne, α-phellandrene, β-phelland-rene, pulegone, (Z)-artemidin, hinokitiol, and acenaphthene [63] | In vitro(antibacterial) on Staphylococcus aureus (ATCC 23235). Concentration—10 µL of tarragon oil tested on agar plate. MIC value is 1250 µg/mL after 24 h of incubation period and MBC value is 2500 µg/mL [63,64] | To treat bleeding gums (gingiva) and bad breath [63] |
| Streblus asper (Koi) | Southern China, India, Sri Lanka, Malaysia, The Philippines, Malaysia, Thailand [65] | Extraction by Hydrodistillation and analyzed by GC-MS method and GC-FID method [66].Essential oil extracted from aerial parts | Leaves: phytol (45.1%), trans-farnesyl acetate (5.8%), α-farnesene (6.4%), trans-trans-α-farnesene (2.0%) and caryophyllene (4.9%) [66] | Leaf extract tested for plaque formation and gingivitis caused by Streptococcus mutans and Actinomycetemcomitans by using disc diffusion method on agar surface. | Dental caries (Wongkhan et al., 2001), strengthening gums, toothache, and gingivitis [65] |
| Stem bark: α-amyrin acetate, β-sitosterol, Strebloside, lupeol acetate, diol, Sioraside, α-amyrin, mansonin, (7’S, 8’S)-trans-streblusol A, (7’S, 8’S)-threo-streblusol B, streblusquinone, 8’R-streblusol C, streblusol E and (8R, 8’R)-streblusol D [65] | The baseline mean of the plaque index is 2.42 in the chlorohexinde group, 1.25 in the placebo group, 2.22 Streblus asper alcoholic extract group, and 2.31 in Streblus asper aqueous extract group. The baseline mean of the gingival index is 2.12 in the chlorohexidine group, 2.23 in the Streblus asper alcoholic extract group, and 2.13 in the Streblus asper aqueous extract group was found to be statistically significant p ≥ 0.001. Swab from mouth is collected. Duration of test is 21 days [67]. | ||||
| Aerial bark: n-Triacontane, β-sitosterol, Stigmasterol, tetraiacontan-3-one, oleanolic acid and botulin [65] | |||||
| Cichorium intybus (Chicory) | Afghanistan, India, Bulgaria, Italy, Morocco, Iran, Serbia, Jordan, Poland, Serbia [68] | Hydrodistillation method for extraction and analysis is performed by GC-FID method. Essential oil extracted from aerial part [69] | Carvacrol (50.1%), cinnamic aldehyde (12.4%), thymol (13.3%), camphor (4.4%), linalool (3.9%), carvone (4.1%) and terpineol (2.1%) [69] | In vitro agar diffusion method (antibacterial) on Staphylococcus aureus, Bacillus subtills, Escherichia coli, and Salmonella typhi causing plaque formation, tooth caries, and gingivitis. Methanolic extract of leaf and root show maximum inhibition at 200 mg/mL concentration [70] | To break up molars (with cavities), plaque, gingivitis, and tooth decay [71] |
| Vitex negundo (Nirgundi) | Afghanistan, India, Sri Lanka, Pakistan, Thailand, eastern Africa, Malaysia, Madagascar [72] | Hydrodistillation method for extraction and analysis by GC and GC-MS method. Essential oil extracted from leaves, flowers and dried fruits [73] | δ-guaiene, epoxide, ethyl-hexadecenoate, guaia-3,7-dienecaryophyllene epoxide, α-selinene, caryophyllene epoxide, germacren-4-ol β-selinene, (E)-nerolidol, α-cedrene, germacrene D, hexadecanoic acid, p-cymene, valencene, germacrene, D viridiflorol (19.55%), β-caryophyllene (16.59%), sabinene (12.07%), γ-terpinene (2.21%), 4-terpineol (9.65%), caryophyllene oxide (1.75%), 1-oceten-3-ol (1.59%), 1-oceten-3-ol (1.59%) and globulol (1.05%) [74] | In vitro study on Streptococcus mutans, Streptococcus sanguis, and Staphylococcus aureus. The aqueous, methanolic, and petroleum ether extract of Vitex negundo were tested for their antibacterial activity using well diffusion method. Concentration: 200 mg/mL. Maximum inhibition zone is shown by methanolic extract is 23 mm [75] | Toothache, throat pain, mouth ulcers (Ullah et al., 2012). The decoction prepared from the leaves of Vitex negundo is used for gargling in the treatment of mouth ulcers [76] |
| Rosmarinus officinalis (Rosemary) | South Europe, India, Mediterranean basin [77] | Hydrodistillation for extraction (Elyemni et al., 2019) and analysis by HPLC and gas chromatography. Extraction of essential oil from leaves [78] | Borneol (1.5–5.0%), camphor (5–31%), pinene (9–26%), 1,8-cineol (15–55%), camphene (2.5–12.0%), pinene (2.0–9.0%), limonene (1.5–5.0%), myrcene (0.9–4.5%), verbenone (2.2–11.1%) and caryophyllene (1.8–5.1%) [79] | Clinical trial on the action of toothpaste made from the extract of Rosmarinus officinalis on humans divided into two groups (experimental and controlled), assessed at baseline and 30 days after the study using the gingival bleeding index (GBI) and the plaque index (PI). | Plaque [79], dental caries [80] |
| Results: reduction of 38% in the risk of gingival bleeding (relative and absolute) | |||||
| And reductions in bacterial plaque is 22.7% [80] | |||||
| Embelia ribes (Vaibidang) | Sri Lanka, China, India, Malaysia [81] | Soxhlet extraction and analysis by FT-IR, DSC, UV-visible, NMR, X-ray diffraction, and TGA method. Extraction of essential oil from berries [82] | Embelin, embolic acid, rapanone [81] and vilangin [83] | The extract of Embelia ribes at a concentration of 500 mg/50 mL reported 12 mm diameter of zone of inhibition against test organism Bacillus subtilis, causing periodontitis and tooth decay [84]. | Dental cavities, as mouthwash, gum infection, and tooth decay [85] |
| Spilanthes species | Tropical Africa, South America, Tropical America, North Australia, Africa, Malaya, Borneo, India, Sri Lanka [86] | Simultaneous distillation extraction method for isolation and GC-MS method for analysis. Essential oil extracted from stem, leaves, and flowers [86] | α-and β-bisabolenes, α-caryophyllene, β-caryophyllene, cadinene, N-(isobutyl)-2E,6Z,8E-decatrienamide, N-(isobutyl)-6Z,8E-decadienamide, N-(2-methylbutyl)-2E,6Z,8E-decatrienamide, decatrienamide, N-(2-phenylethyl)-2E,6Z,8E-decatrienamide [86] | Chewing on the flower heads and roots has shown to decrease gum inflammation and have been used in the treatment of periodontitis [87] | Toothache, throat complaints [88,89], Tooth decay [90], Sore throat, gum infection [91], Gingivitis [92] |
| Nigella sativa L. (Kalonji) | The Middle East, southern European continent, North Africa, India: Bengal, Bihar, Gangetic plains, Himachal Pradesh, Assam, Maharashtra, and Punjab [93] | Essential oil from seeds extracted using solvent extraction and SFE method and analyzed by GC-MS [94] | Carvacrol (5.8–11.6%), longifolene (1.0–8.0%), ρ-cymene (7.1–15.5%), t-anethole (0.25–2.3%), 4-terpineol (2–6.6%) and thymoquinone (27.8–57.0%) [94] | Clinical trial on efficacy of 0.2% thymoquinone oral gel (topical) in treatment of periodontitis: heathy female and male patients with at least 2 periodontally involved sites (≥5 mm), n = 20. | |
| Dose: repeated from baseline up to 4 weeks. | |||||
| Result showed reduction in GI, PI and PPD levels [95] | Essential oil having anticarcinogenic, antioxidant and antimicrobial properties [96] |
| Title | Extract and Dose Used | Objective | Location | Main Finding of the Study | Reference |
|---|---|---|---|---|---|
| Comparative evaluation of efficacy of 4% tulsi extract fluoridated and placebo dentifrices against gingivitis and plaque: a triple-blind RCT | 4% ethanolic extract (tulsi dry leaves), Dose: twice/day (21 days), Application:Toothpaste | To assess and compare the antigingivitis and antiplaque effect of fluoridated, placebo dentifrice (PD) and 4% tulsi leaf extract dentifrice among 14–15-year-old school children. | Davangere city, India | Maximum reduction in dental plaque (p = 0.01) and gingivitis score (p = 0.001) in 4% tulsi dentifrice compared to PD. | [156] |
| Comparative clinical effects of Salvadora Persica oral rinse and phenolic commercial mouth wash on human oral health: a triple-blind RCT | Salvadora persica oral rinse 50% conc., Dose: 15 mL twice/day (6 months), Application: gargle, mouthwash | To compare the clinical effects of Salvadora persica oral rinse and commercial phenolic mouth wash on oral health status of socially deprived madrasa girls 18–22 years old. | Multan city, Pakistan | With no statistical difference in gignival and plaque scores, Salvadora persica oral rinse is equally effective as phenolic mouth wash. | [157] |
| Antiplaque effect of hiora-GA gel, spirogyl gum paint, and tooth and gums tonic in comparison with chlorhexidine M gel: a double-blind RCT | Eucalyptus globulus extract (tooth and gums tonic), Dose: twice/day (90 days), Application: gel directly applied on tooth surface | To compare the efficacy of three different herbal products in gingival inflammation, bacterial count, and reducing plaque in comparison with chlorhexidine M gel among participants with moderate to severe periodontitis. | Osmania Dental College and Hospital, Hyderabad, India | The mean gignival and plaque scores were decreased at different intervals, and no significant difference is oserved in efficacy of gel compared to chlorhexidine. | [158] |
| Effect of 2.5% sodium hypochlorite versus neem as root canal irrigants on the intensity of post-operative pain and the amount of endotoxins in mandibular molars with necrotic pulps: RCT | Neem (root canal irrigant), Dose: one time each followed by two root canal treatments | To assess the efficacy of 2.5% NaOCl versus neem as root canal irrigants on amount of endotoxins and intensity of post-operative pain following root canal treatment of mandibular molars with necrotic pulps | Cairo University, Egypt | In neem group, mean pain scores were lower as compared to 2.5% NaOCl, and neem group reduced endotoxin level by 18% in comparison with pre-instrumentation samples. | [159] |
| Clinical effect of Juglans regia on the developing dental plaque: RCT | 2% ether extract (bark), | To assess the clinical effect of 2% aqueous extract, 2% and 3% concentration of ether fractions in propylene glycol and petrol-ether extract of bark of Juglans regia against developing plaque | Faculty of Dental Sciences, C. S. M. Medical University, Lucknow, India | 2% ether extract of Juglans regia showed maximum antiplaque activity of 32.12% (p < 0.001). | [46] |
| Dose: twice/day (3 days), | |||||
| Application: extract directly applied on tooth surface | |||||
| Intrapocket application of Melaleuca alternifolia tea tree oil (TTO) gel in the treatment of stage II periodontitis: a phase 2 clinical trial | 5% TTO gel and SRP, Dose: 0.5 mL gel, Application: gel directly applied on dental pockets | To assess biochemically and clinical the effect of intrapocket application of TTO gel and scaling and root planing (SRP) in the treatment of stage II periodontitis and to correlate biochemical levels with clinical response | Faculty of Dentistry, Alexandria University | TTO gel adjunctive to SRP is found to be effective in treatment of stage II periodontitis. | [160] |
| Alexandria, Egypt | |||||
| Effect of a Toothpaste/Mouthwash Containing Carica papaya Leaf Extract on Interdental Gingival Bleeding: A Randomized Controlled Trial | Carica papaya leaf extract | To study the comapartive effectiveness of dentifrice having papaya leaf extract to a commercially available sodium lauryl sulfate-free enzyme-containing dentifrice in management of gingival bleeding | Dental Faculty, University of Granada, Spain | Papaya leaf extract dentifrice/mouthwash provides an efficacious and natural alternative to sodium lauryl sulfate-free dentifrice and reduces gingival bleeding. | [161] |
| Application: Mouthwash and tooth paste | |||||
| Use of an antiviral mouthwash as a barrier measure in the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) transmission in adults with asymptomatic to mild COVID-19: a multicenter, randomized, double-blind controlled trial | ß-cyclodextrin and citrox (bioflavonoids) (CDCM) | To determine if commercially available mouthwash with CDCM could decrease the SARS-CoV-2 load from saliva | Hospital Centers, France | CDCM had a significant beneficial effect on reducing SARS-CoV-2 salivary viral load in 280 adults with asymptomatic or mild COVID-19, 4 h after the initial dose. | [162] |
| Application: Mouthwash |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, M.; Prakash, S.; Radha; Kumari, N.; Pundir, A.; Punia, S.; Saurabh, V.; Choudhary, P.; Changan, S.; Dhumal, S.; et al. Beneficial Role of Antioxidant Secondary Metabolites from Medicinal Plants in Maintaining Oral Health. Antioxidants 2021, 10, 1061. https://doi.org/10.3390/antiox10071061
Kumar M, Prakash S, Radha, Kumari N, Pundir A, Punia S, Saurabh V, Choudhary P, Changan S, Dhumal S, et al. Beneficial Role of Antioxidant Secondary Metabolites from Medicinal Plants in Maintaining Oral Health. Antioxidants. 2021; 10(7):1061. https://doi.org/10.3390/antiox10071061
Chicago/Turabian StyleKumar, Manoj, Suraj Prakash, Radha, Neeraj Kumari, Ashok Pundir, Sneh Punia, Vivek Saurabh, Poonam Choudhary, Sushil Changan, Sangram Dhumal, and et al. 2021. "Beneficial Role of Antioxidant Secondary Metabolites from Medicinal Plants in Maintaining Oral Health" Antioxidants 10, no. 7: 1061. https://doi.org/10.3390/antiox10071061
APA StyleKumar, M., Prakash, S., Radha, Kumari, N., Pundir, A., Punia, S., Saurabh, V., Choudhary, P., Changan, S., Dhumal, S., Pradhan, P. C., Alajil, O., Singh, S., Sharma, N., Ilakiya, T., Singh, S., & Mekhemar, M. (2021). Beneficial Role of Antioxidant Secondary Metabolites from Medicinal Plants in Maintaining Oral Health. Antioxidants, 10(7), 1061. https://doi.org/10.3390/antiox10071061