Effects of Dietary Strategies on Exercise-Induced Oxidative Stress: A Narrative Review of Human Studies
Abstract
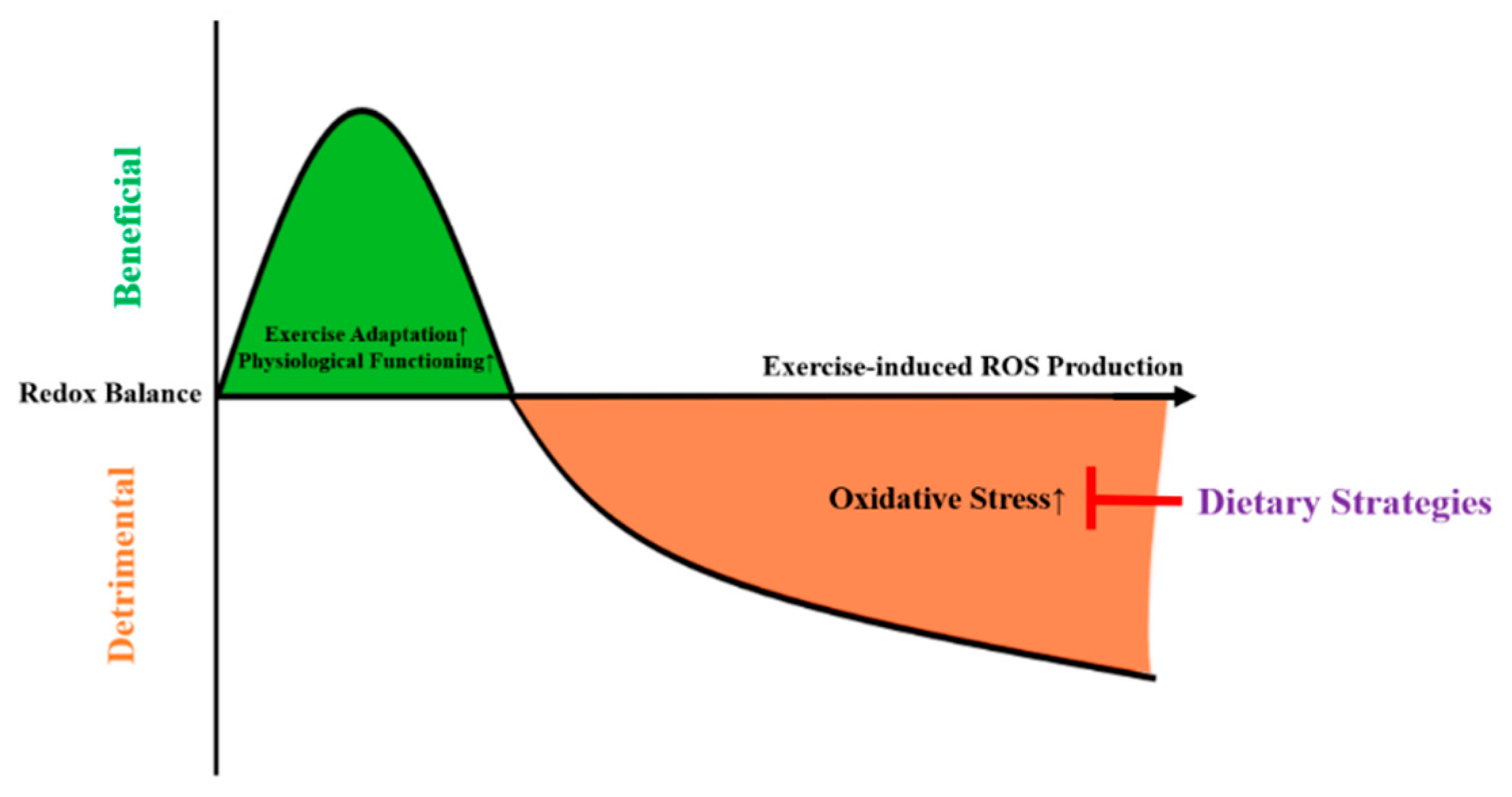
1. Background
2. Methods
3. Dietary Strategies
4. Effects on Biomarkers of Exercise-Induced Oxidative Stress
4.1. Effects of Dietary Interventions on Direct ROS Generation
4.2. Effects of Dietary Interventions on ROS-Induced Macromolecule Damage
4.3. Effects of Dietary Interventions on Inflammatory Markers
4.4. Effects of Dietary Interventions on Antioxidant Activity
5. Perspectives
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Halliwell, B. Free Radicals, Antioxidants, and Human Disease: Curiosity, Cause, or Consequence? Lancet 1994, 344, 721–724. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 5th ed.; Oxford University Press: New York, NY, USA, 2015; ISBN 978-0198717485. [Google Scholar]
- Dröge, W. Free Radicals in the Physiological Control of Cell Function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef] [PubMed]
- Peternelj, T.T.; Coombes, J.S. Antioxidant Supplementation during Exercise Training: Beneficial or Detrimental? Sports Med. 2011, 41, 1043–1069. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free Radicals and Antioxidants in Normal Physiological Functions and Human Disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.K.; Duarte, J.; Kavazis, A.N.; Talbert, E.E. Reactive Oxygen Species Are Signalling Molecules for Skeletal Muscle Adaptation. Exp. Physiol. 2010, 95, 1–9. [Google Scholar] [CrossRef]
- Powers, S.K.; Ji, L.L.; Kavazis, A.N.; Jackson, M.J. Reactive Oxygen Species: Impact on Skeletal Muscle. Compr. Physiol. 2011, 1, 941–969. [Google Scholar] [CrossRef]
- Waris, G.; Ahsan, H. Reactive Oxygen Species: Role in the Development of Cancer and Various Chronic Conditions. J. Carcinog. 2006, 5, 14. [Google Scholar] [CrossRef]
- Maritim, A.C.; Sanders, R.A.; Watkins, J.B. Diabetes, Oxidative Stress, and Antioxidants: A Review. J. Biochem. Mol. Toxicol. 2003, 17, 24–38. [Google Scholar] [CrossRef]
- Ozkul, A.; Akyol, A.; Yenisey, C.; Arpaci, E.; Kiylioglu, N.; Tataroglu, C. Oxidative Stress in Acute Ischemic Stroke. J. Clin. Neurosci. 2007, 14, 1062–1066. [Google Scholar] [CrossRef]
- Allen, C.L.; Bayraktutan, U. Oxidative Stress and Its Role in the Pathogenesis of Ischaemic Stroke. Int. J. Stroke 2009, 4, 461–470. [Google Scholar] [CrossRef]
- Bloomer, R.J.; Schilling, B.K.; Karlage, R.E.; Ledoux, M.S.; Pfeiffer, R.F.; Callegari, J. Effect of Resistance Training on Blood Oxidative Stress in Parkinson Disease. Med. Sci. Sports Exerc. 2008, 40, 1385–1389. [Google Scholar] [CrossRef] [PubMed]
- Brieger, K.; Schiavone, S.; Miller, F.J.; Krause, K.H. Reactive Oxygen Species: From Health to Disease. Swiss Med. Wkly. 2012, 142, w13659. [Google Scholar] [CrossRef] [PubMed]
- Alessio, H.M.; Hagerman, A.E.; Fulkerson, B.K.; Ambrose, J.; Rice, R.E.; Wiley, R.L. Generation of Reactive Oxygen Species after Exhaustive Aerobic and Isometric Exercise. Med. Sci. Sports Exerc. 2000, 32, 1576–1581. [Google Scholar] [CrossRef] [PubMed]
- Finaud, J.; Lac, G.; Filaire, E. Oxidative Stress: Relationship with Exercise and Training. Sport. Med. 2006, 36, 327–358. [Google Scholar] [CrossRef] [PubMed]
- Tapia, P.C. Sublethal Mitochondrial Stress with an Attendant Stoichiometric Augmentation of Reactive Oxygen Species May Precipitate Many of the Beneficial Alterations in Cellular Physiology Produced by Caloric Restriction, Intermittent Fasting, Exercise and Dietary. Med. Hypotheses 2006, 66, 832–843. [Google Scholar] [CrossRef]
- Merry, T.L.; Ristow, M. Mitohormesis in Exercise Training. Free Radic. Biol. Med. 2016, 98, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Pinckard, K.; Baskin, K.K.; Stanford, K.I. Effects of Exercise to Improve Cardiovascular Health. Front. Cardiovasc. Med. 2019, 6, 69. [Google Scholar] [CrossRef] [PubMed]
- Russomanno, G.; Corbi, G.; Manzo, V.; Ferrara, N.; Rengo, G.; Puca, A.A.; Latte, S.; Carrizzo, A.; Calabrese, M.C.; Andriantsitohaina, R.; et al. The Anti-Ageing Molecule Sirt1 Mediates Beneficial Effects of Cardiac Rehabilitation. Immun. Ageing 2017, 14, 1–9. [Google Scholar] [CrossRef]
- Korsager Larsen, M.; Matchkov, V.V. Hypertension and Physical Exercise: The Role of Oxidative Stress. Med. 2016, 52, 19–27. [Google Scholar] [CrossRef]
- Sun, M.-W.; Zhong, M.-F.; Gu, J.; Qian, F.-L.; Gu, J.-Z.; Chen, H. Effects of Different Levels of Exercise Volume on Endothelium-Dependent Vasodilation: Roles of Nitric Oxide Synthase and Heme Oxygenase. Hypertens. Res. 2008, 31, 805–816. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pingitore, A.; Lima, G.P.P.; Mastorci, F.; Quinones, A.; Iervasi, G.; Vassalle, C. Exercise and Oxidative Stress: Potential Effects of Antioxidant Dietary Strategies in Sports. Nutrition 2015, 31, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, G.; Hamarsland, H.; Cumming, K.T.; Johansen, R.E.; Hulmi, J.J.; Børsheim, E.; Wiig, H.; Garthe, I.; Raastad, T. Vitamin C and E Supplementation Alters Protein Signalling after a Strength Training Session, but Not Muscle Growth during 10 Weeks of Training. J. Physiol. 2014, 592, 5391–5408. [Google Scholar] [CrossRef]
- Gomez-Cabrera, M.C.; Domenech, E.; Romagnoli, M.; Arduini, A.; Borras, C.; Pallardo, F.V.; Sastre, J.; Viña, J. Oral Administration of Vitamin C Decreases Muscle Mitochondrial Biogenesis and Hampers Training-Induced Adaptations in Endurance Performance. Am. J. Clin. Nutr. 2008, 87, 142–149. [Google Scholar] [CrossRef]
- Stepanyan, V.; Crowe, M.; Haleagrahara, N.; Bowden, B. Effects of Vitamin E Supplementation on Exercise-Induced Oxidative Stress: A Meta-Analysis. Appl. Physiol. Nutr. Metab. 2014, 39, 1029–1037. [Google Scholar] [CrossRef]
- Bruce, B.; Spiller, G.A.; Klevay, L.M.; Gallagher, S.K. A Diet High in Whole and Unrefined Foods Favorably Alters Lipids, Antioxidant Defenses, and Colon Function. J. Am. Coll. Nutr. 2000, 19, 61–67. [Google Scholar] [CrossRef]
- Ristow, M.; Zarse, K.; Oberbach, A.; Klöting, N.; Birringer, M.; Kiehntopf, M.; Stumvoll, M.; Kahn, C.R.; Blüher, M. Antioxidants Prevent Health-Promoting Effects of Physical Exercise in Humans. Proc. Natl. Acad. Sci. USA 2009, 106, 8665–8670. [Google Scholar] [CrossRef]
- Higgins, M.R.; Izadi, A.; Kaviani, M. Antioxidants and Exercise Performance: With a Focus on Vitamin e and c Supplementation. Int. J. Environ. Res. Public Health 2020, 17, 8452. [Google Scholar] [CrossRef] [PubMed]
- Davison, G.; Callister, R.; Williamson, G.; Cooper, K.A.; Gleeson, M. The Effect of Acute Pre-Exercise Dark Chocolate Consumption on Plasma Antioxidant Status, Oxidative Stress and Immunoendocrine Responses to Prolonged Exercise. Eur. J. Nutr. 2012, 51, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Allgrove, J.; Farrell, E.; Gleeson, M.; Williamson, G.; Cooper, K. Regular Dark Chocolate Consumption’s Reduction of Oxidative Stress and Increase of Free-Fatty-Acid Mobilization in Response to Prolonged Cycling. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 113–123. [Google Scholar] [CrossRef]
- Taub, P.R.; Ramirez-Sanchez, I.; Patel, M.; Higginbotham, E.; Moreno-Ulloa, A.; Román-Pintos, L.M.; Phillips, P.; Perkins, G.; Ceballos, G.; Villarreal, F. Beneficial Effects of Dark Chocolate on Exercise Capacity in Sedentary Subjects: Underlying Mechanisms. A Double Blind, Randomized, Placebo Controlled Trial. Food Funct. 2016, 7, 3686–3693. [Google Scholar] [CrossRef]
- Wiswedel, I.; Hirsch, D.; Kropf, S.; Gruening, M.; Pfister, E.; Schewe, T.; Sies, H. Flavanol-Rich Cocoa Drink Lowers Plasma F2-Isoprostane Concentrations in Humans. Free Radic. Biol. Med. 2004, 37, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Panza, V.S.P.; Wazlawik, E.; Ricardo Schütz, G.; Comin, L.; Hecht, K.C.; da Silva, E.L. Consumption of Green Tea Favorably Affects Oxidative Stress Markers in Weight-Trained Men. Nutrition 2008, 24, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Panza, V.P.; Diefenthaeler, F.; Tamborindeguy, A.C.; Camargo, C.D.Q.; De Moura, B.M.; Brunetta, H.S.; Sakugawa, R.L.; De Oliveira, M.V.; Puel, E.D.O.; Nunes, E.A.; et al. Effects of Mate Tea Consumption on Muscle Strength and Oxidative Stress Markers after Eccentric Exercise. Br. J. Nutr. 2016, 115, 1370–1378. [Google Scholar] [CrossRef]
- McLeay, Y.; Barnes, M.J.; Mundel, T.; Hurst, S.M.; Hurst, R.D.; Stannard, S.R. Effect of New Zealand Blueberry Consumption on Recovery from Eccentric Exercise-Induced Muscle Damage. J. Int. Soc. Sports Nutr. 2012, 9, 19. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Kwak, Y.S.; Seo, H.K.; Kim, H.Y. Assessing the Values of Blueberries Intake on Exercise Performance, TAS, and Inflammatory Factors. Iran J. Public Health 2018, 47, 27–32. [Google Scholar] [PubMed]
- McAnulty, S.R.; McAnulty, L.S.; Nieman, D.C.; Dumke, C.L.; Morrow, J.D.; Utter, A.C.; Henson, D.A.; Proulx, W.R.; George, G.L. Consumption of Blueberry Polyphenols Reduces Exercise-Induced Oxidative Stress Compared to Vitamin C. Nutr. Res. 2004, 24, 209–221. [Google Scholar] [CrossRef]
- de Lima Tavares Toscano, L.; Silva, A.S.; de França, A.C.L.; de Sousa, B.R.V.; de Almeida Filho, E.J.B.; da Silveira Costa, M.; Marques, A.T.B.; da Silva, D.F.; de Farias Sena, K.; Cerqueira, G.S.; et al. A Single Dose of Purple Grape Juice Improves Physical Performance and Antioxidant Activity in Runners: A Randomized, Crossover, Double-Blind, Placebo Study. Eur. J. Nutr. 2020, 59, 2997–3007. [Google Scholar] [CrossRef]
- Toscano, L.T.; Tavares, R.L.; Toscano, L.T.; da Silva, C.S.O.; de Almeida, A.E.M.; Biasoto, A.C.T.; Gonçalves, M.d.C.R.; Silva, A.S. Potential Ergogenic Activity of Grape Juice in Runners. Appl. Physiol. Nutr. Metab. 2015, 40, 899–906. [Google Scholar] [CrossRef]
- Bowtell, J.L.; Sumners, D.P.; Dyer, A.; Fox, P.; Mileva, K.N. Montmorency Cherry Juice Reduces Muscle Damage Caused by Intensive Strength Exercise. Med. Sci. Sports Exerc. 2011, 43, 1544–1551. [Google Scholar] [CrossRef]
- Howatson, G.; McHugh, M.P.; Hill, J.A.; Brouner, J.; Jewell, A.P.; Van Someren, K.A.; Shave, R.E.; Howatson, S.A. Influence of Tart Cherry Juice on Indices of Recovery Following Marathon Running. Scand. J. Med. Sci. Sport. 2010, 20, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Jendricke, P.; Centner, C.; Storck, H.; Gollhofer, A.; König, D. Acute Effects of Oatmeal on Exercise-Induced Reactive Oxygen Species Production Following High-Intensity Interval Training in Women: A Randomized Controlled Trial. Antioxidants 2021, 10, 3. [Google Scholar] [CrossRef]
- Koenig, R.T.; Dickman, J.R.; Kang, C.H.; Zhang, T.; Chu, Y.F.; Ji, L.L. Avenanthramide Supplementation Attenuates Eccentric Exercise-Inflicted Blood Inflammatory Markers in Women. Eur. J. Appl. Physiol. 2016, 116, 67–76. [Google Scholar] [CrossRef]
- Zhang, T.; Zhao, T.; Zhang, Y.; Liu, T.; Gagnon, G.; Ebrahim, J.; Johnson, J.; Chu, Y.F.; Ji, L.L. Avenanthramide Supplementation Reduces Eccentric Exercise-Induced Inflammation in Young Men and Women. J. Int. Soc. Sports Nutr. 2020, 17. [Google Scholar] [CrossRef]
- Copetti, C.L.K.; Orssatto, L.B.R.; Diefenthaeler, F.; Silveira, T.T.; da Silva, E.L.; de Liz, S.; Mendes, B.C.; Rieger, D.K.; Vieira, F.G.K.; Hinnig, P.F.; et al. Acute Effect of Juçara Juice (Euterpe Edulis Martius) on Oxidative Stress Biomarkers and Fatigue in a High-Intensity Interval Training Session: A Single-Blind Cross-over Randomized Study. J. Funct. Foods 2020, 67, 103835. [Google Scholar] [CrossRef]
- Pittaluga, M.; Sgadari, A.; Tavazzi, B.; Fantini, C.; Sabatini, S.; Ceci, R.; Amorini, A.M.; Parisi, P.; Caporossi, D. Exercise-Induced Oxidative Stress in Elderly Subjects: The Effect of Red Orange Supplementation on the Biochemical and Cellular Response to a Single Bout of Intense Physical Activity. Free Radic. Res. 2013, 47, 202–211. [Google Scholar] [CrossRef]
- Chang, W.H.; Hu, S.P.; Huang, Y.F.; Yeh, T.S.; Liu, J.F. Effect of Purple Sweet Potato Leaves Consumption on Exercise-Induced Oxidative Stress and IL-6 and HSP72 Levels. J. Appl. Physiol. 2010, 109, 1710–1715. [Google Scholar] [CrossRef]
- Mazani, M.; Nemati, A.; Baghi, A.N.; Amani, M.; Haedari, K.; Alipanah-Mogadam, R. The Effect of Probiotic Yoghurt Consumption on Oxidative Stress and Inflammatory Factors in Young Females after Exhaustive Exercise. J. Pak. Med. Assoc. 2018, 68, 1748–1754. [Google Scholar] [PubMed]
- Harms-Ringdahl, M.; Jenssen, D.; Haghdoost, S. Tomato Juice Intake Suppressed Serum Concentration of 8-OxodG after Extensive Physical Activity. Nutr. J. 2012, 11, 29. [Google Scholar] [CrossRef]
- Kawamura, A.; Aoi, W.; Abe, R.; Kobayashi, Y.; Kuwahata, M.; Higashi, A. Astaxanthin-, β-Carotene-, and Resveratrol-Rich Foods Support Resistance Training-Induced Adaptation. Antioxidants 2021, 10, 113. [Google Scholar] [CrossRef]
- Sureda, A.; Tauler, P.; Aguiló, A.; Cases, N.; Llompart, I.; Tur, J.A.; Pons, A. Antioxidant Supplementation Influences the Neutrophil Tocopherol Associated Protein Expression, but Not the Inflammatory Response to Exercise. Cent. Eur. J. Biol. 2007, 2, 56–70. [Google Scholar] [CrossRef]
- Carrera-Quintanar, L.; Funes, L.; Herranz-López, M.; Martínez-Peinado, P.; Pascual-García, S.; Sempere, J.M.; Boix-Castejón, M.; Córdova, A.; Pons, A.; Micol, V.; et al. Antioxidant Supplementation Modulates Neutrophil Inflammatory Response to Exercise-Induced Stress. Antioxidants 2020, 9, 1242. [Google Scholar] [CrossRef]
- Daly, R.M.; Gianoudis, J.; de Ross, B.; O’Connell, S.L.; Kruger, M.; Schollum, L.; Gunn, C. Effects of a Multinutrient-Fortified Milk Drink Combined with Exercise on Functional Performance, Muscle Strength, Body Composition, Inflammation, and Oxidative Stress in Middle-Aged Women: A 4-Month, Double-Blind, Placebo-Controlled, Randomized Trial. Am. J. Clin. Nutr. 2020, 112, 427–446. [Google Scholar] [CrossRef]
- Prasertsri, P.; Roengrit, T.; Kanpetta, Y.; Tong-Un, T.; Muchimapura, S.; Wattanathorn, J.; Leelayuwat, N. Cashew Apple Juice Supplementation Enhances Leukocyte Count by Reducing Oxidative Stress after High-Intensity Exercise in Trained and Untrained Men. J. Int. Soc. Sports Nutr. 2019, 16, 31. [Google Scholar] [CrossRef]
- Iwasa, M.; Aoi, W.; Mune, K.; Yamauchi, H.; Furuta, K.; Sasaki, S.; Takeda, K.; Harada, K.; Wada, S.; Nakamura, Y.; et al. Fermented Milk Improves Glucose Metabolism in Exercise-Induced Muscle Damage in Young Healthy Men. Nutr. J. 2013, 12, 83. [Google Scholar] [CrossRef]
- Beavers, K.M.; Serra, M.C.; Beavers, D.P.; Cooke, M.B.; Willoughby, D.S. Soy and the Exercise-Induced Inflammatory Response in Postmenopausal Women. Appl. Physiol. Nutr. Metab. 2010, 35, 261–269. [Google Scholar] [CrossRef]
- Dikalov, S.I.; Polienko, Y.F.; Kirilyuk, I. Electron Paramagnetic Resonance Measurements of Reactive Oxygen Species by Cyclic Hydroxylamine Spin Probes. Antioxid. Redox Signal. 2018, 28, 1433–1443. [Google Scholar] [CrossRef]
- Suzen, S.; Gurer-Orhan, H.; Saso, L. Detection of Reactive Oxygen and Nitrogen Species by Electron Paramagnetic Resonance (EPR) Technique. Molecules 2017, 22, 181. [Google Scholar] [CrossRef]
- Collins, F.W. Oat Phenolics: Avenanthramides, Novel Substituted N-Cinnamoylanthranilate Alkaloids from Oat Groats and Hulls. J. Agric. Food Chem. 1989, 37, 60–66. [Google Scholar] [CrossRef]
- Bratt, K.; Sunnerheim, K.; Bryngelsson, S.; Fagerlund, A.; Engman, L.; Andersson, R.E.; Dimberg, L.H. Avenanthramides in Oats (Avena Sativa L.) and Structure-Antioxidant Activity Relationships. J. Agric. Food Chem. 2003, 51, 594–600. [Google Scholar] [CrossRef]
- Lee-Manion, A.M.; Price, R.K.; Strain, J.J.; Dimberg, L.H.; Sunnerheim, K.; Welch, R.W. In Vitro Antioxidant Activity and Antigenotoxic Effects of Avenanthramides and Related Compounds. J. Agric. Food Chem. 2009, 57, 10619–10624. [Google Scholar] [CrossRef]
- Fu, J.; Zhu, Y.; Yerke, A.; Wise, M.L.; Johnson, J.; Chu, Y.; Sang, S. Oat Avenanthramides Induce Heme Oxygenase-1 Expression via Nrf2-Mediated Signaling in HK-2 Cells. Mol. Nutr. Food Res. 2015, 59, 2471–2479. [Google Scholar] [CrossRef] [PubMed]
- Andújar, I.; Recio, M.C.; Giner, R.M.; Ríos, J.L. Cocoa Polyphenols and Their Potential Benefits for Human Health. Oxid. Med. Cell. Longev. 2012, 2012, 906252. [Google Scholar] [CrossRef]
- Yan-Hwa, C.; Chang, C.L.; Hsu, H.F. Flavonoid Content of Several Vegetables and Their Antioxidant Activity. J. Sci. Food Agric. 2000, 80, 561–566. [Google Scholar] [CrossRef]
- Chang, W.H.; Chen, C.M.; Hu, S.P.; Kan, N.W.; Chiu, C.C.; Liu, J.F. Effect of Purple Sweet Potato Leaf Consumption on the Modulation of the Antioxidative Status in Basketball Players during Training. Asia Pac. J. Clin. Nutr. 2007, 16, 455–461. [Google Scholar] [CrossRef]
- Chang, W.H.; Chen, C.M.; Hu, S.P.; Kan, N.W.; Chiu, C.C.; Liu, J.F. Effect of Purple Sweet Potato Leaves Consumption on the Modulation of the Immune Response in Basketball Players during the Training Period. Asia Pac. J. Clin. Nutr. 2007, 16, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Mikelsaar, M.; Zilmer, M. Lactobacillus Fermentum ME-3—An Antimicrobial and Antioxidative Probiotic. Microb. Ecol. Health Dis. 2009, 21, 1–27. [Google Scholar] [CrossRef]
- Uskova, M.A.; Kravchenko, L.V. Antioxidant properties of lactic acid bacteria—Probiotic and yogurt strains. Vopr. Pitan. 2009, 78, 18–23. [Google Scholar] [PubMed]
- Abegaz, E.G.; Tandon, K.S.; Scott, J.W.; Baldwin, E.A.; Shewfelt, R.L. Partitioning Taste from Aromatic Flavor Notes of Fresh Tomato (Lycopersicon Esculentum, Mill) to Develop Predictive Models as a Function of Volatile and Nonvolatile Components. Postharvest Biol. Technol. 2004, 34, 227–235. [Google Scholar] [CrossRef]
- Di Mascio, P.; Kaiser, S.; Sies, H. Lycopene as the Most Efficient Biological Carotenoid Singlet Oxygen Quencher. Arch. Biochem. Biophys. 1989, 274, 532–538. [Google Scholar] [CrossRef]
- Carrera-Quintanar, L.; Funes, L.; Vicente-Salar, N.; Blasco-Lafarga, C.; Pons, A.; Micol, V.; Roche, E. Effect of Polyphenol Supplements on Redox Status of Blood Cells: A Randomized Controlled Exercise Training Trial. Eur. J. Nutr. 2015, 54, 1081–1093. [Google Scholar] [CrossRef]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.B.; Rahu, N. Oxidative Stress and Inflammation: What Polyphenols Can Do for Us? Oxid. Med. Cell. Longev. 2016, 2016, 7432797. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, T.; Muraoka, I. Exercise-Induced Oxidative Stress and the Effects of Antioxidant Intake from a Physiological Viewpoint. Antioxidants 2018, 7, 119. [Google Scholar] [CrossRef] [PubMed]
- Wintergerst, E.S.; Maggini, S.; Hornig, D.H. Immune-Enhancing Role of Vitamin C and Zinc and Effect on Clinical Conditions. Ann. Nutr. Metab. 2006, 50, 85–94. [Google Scholar] [CrossRef]
- Kubo, I.; Masuoka, N.; Ha, T.J.; Tsujimoto, K. Antioxidant Activity of Anacardic Acids. Food Chem. 2006, 99, 555–562. [Google Scholar] [CrossRef]
- Si, W.; Gong, J.; Tsao, R.; Kalab, M.; Yang, R.; Yin, Y. Bioassay-Guided Purification and Identification of Antimicrobial Components in Chinese Green Tea Extract. J. Chromatogr. 2006, 1125, 204–210. [Google Scholar] [CrossRef]
- Schulz, M.; da Silva Campelo Borges, G.; Gonzaga, L.V.; Oliveira Costa, A.C.; Fett, R. Juçara Fruit (Euterpe Edulis Mart.): Sustainable Exploitation of a Source of Bioactive Compounds. Food Res. Int. 2016, 89, 14–26. [Google Scholar] [CrossRef]
- Tsao, R. Chemistry and Biochemistry of Dietary Polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef]
- Elejalde, E.; Villarán, M.C.; Alonso, R.M. Grape Polyphenols Supplementation for Exercise-Induced Oxidative Stress. J. Int. Soc. Sports Nutr. 2021, 18, 1–12. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Neveu, V.; Vos, F.; Scalbert, A. Identification of the 100 Richest Dietary Sources of Polyphenols: An Application of the Phenol-Explorer Database. Eur. J. Clin. Nutr. 2010, 64, 112–120. [Google Scholar] [CrossRef]
- Kim, K.H.; Tsao, R.; Yang, R.; Cui, S.W. Phenolic Acid Profiles and Antioxidant Activities of Wheat Bran Extracts and the Effect of Hydrolysis Conditions. Food Chem. 2006, 95, 466–473. [Google Scholar] [CrossRef]
- Adom, K.K.; Liu, R.H. Antioxidant Activity of Grains. J. Agric. Food Chem. 2002, 50, 6182–6187. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.B.; Markey, C.E.; Busch, M.A.; Busch, K.W. Determination of Capsaicinoids in Habanero Peppers by Chemometric Analysis of UV Spectral Data. J. Agric. Food Chem. 2007, 55, 5925–5933. [Google Scholar] [CrossRef]
- Suhett, L.G.; de Miranda Monteiro Santos, R.; Silveira, B.K.S.; Leal, A.C.G.; de Brito, A.D.M.; de Novaes, J.F.; Lucia, C.M. Effects of Curcumin Supplementation on Sport and Physical Exercise: A Systematic Review. Crit. Rev. Food Sci. Nutr. 2021, 61, 946–958. [Google Scholar] [CrossRef]
- Schmutz, S.; Däpp, C.; Wittwer, M.; Vogt, M.; Hoppeler, H.; Flück, M. Endurance Training Modulates the Muscular Transcriptome Response to Acute Exercise. Pflugers Arch. Eur. J. Physiol. 2006, 451, 678–687. [Google Scholar] [CrossRef] [PubMed]
- Nocella, C.; Cammisotto, V.; Pigozzi, F.; Borrione, P.; Fossati, C.; D’Amico, A.; Cangemi, R.; Peruzzi, M.; Gobbi, G.; Ettorre, E.; et al. Impairment between Oxidant and Antioxidant Systems: Short- and Long-term Implications for Athletes’ Health. Nutrients 2019, 11, 1353. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Whiteman, M. Measuring Reactive Species and Oxidative Damage in Vivo and in Cell Culture: How Should You Do It and What Do the Results Mean? Br. J. Pharmacol. 2004, 142, 231–255. [Google Scholar] [CrossRef]
- Cobley, J.N.; Close, G.L.; Bailey, D.M.; Davison, G.W. Exercise Redox Biochemistry: Conceptual, Methodological and Technical Recommendations. Redox Biol. 2017, 12, 540–548. [Google Scholar] [CrossRef] [PubMed]
- Arts, M.J.T.J.; Haenen, G.R.M.M.; Voss, H.-P.; Bast, A. Antioxidant Capacity of Reaction Products Limits the Applicability of the Trolox Equivalent Antioxidant Capacity (TEAC) Assay. Food Chem. Toxicol. 2004, 42, 45–49. [Google Scholar] [CrossRef]
| Main Result Category | Study | Type of Diet | Nutritional Protocol | Type and Intensity of Exercise | Detection Method | |||
|---|---|---|---|---|---|---|---|---|
| ROS Generation | Oxidative Stress Marker | Inflammation Marker | Antioxidant Activity | |||||
| ROS Generation | Zeng et al. [42] | Oatmeal | Oat flake + skim milk versus Fasting; 2 h before exercise | Body weight HIIT, 30 min | ↓ | N/A | N/A | N/A |
| ROS-induced Macromolecule Damage | Davison et al. [29] | Dark chocolate | Dark chocolate versus cocoa-liquor-free control bar versus neither, 2 h before exercise | Cycling, 2.5 h | N/A | F2-isoprostane↓ | Circulating leucocyte↔, IL-6↔ | N/A |
| Wiswedel et al. [32] | High-flavanol cocoa drink (HFCD) | HFCD versus low-flavanol cocoa drink (LFCD), 2 h before exercise | Cycling, 29 min | N/A | F2-isoprostane↓ | N/A | N/A | |
| Allgrove et al. [30] | Dark chocolate | Dark chocolate versus isocarbohydrate-fat control cocoa-liquor-free chocolate, twice/d, 2 weeks | Cycling for 90 min followed by 25 min exhaustion time trial | N/A | F2-isoprostane↓ | Circulating leucocyte↔, IL-6↔, IL-10↔, IL-1Ra↔ | N/A | |
| Taub et al. [31] | High-flavanol dark chocolate (HFCHO) | HFCHO versus Low-flavanol dark chocolate (LFCHO), 3 months | Ramped exercise on stationary bicycle (Cardiopulmonary exercise testing), ~10 min | N/A | PC↓ | N/A | GSH/GSSH↑ | |
| McAnulty et al. [37] | Blueberry | Blueberries versus blueberry-flavored shake, 7 days | Running, until a core temperature of 39.5 °C was reached | N/A | LH↓, F2-isoprostanes↔ | IL-6↔, IL-8↔, IL-10↔ | FRAP↔ | |
| Bowtell et al. [40] | Montmorency cherry juice | Montmorency cherry juice versus isoenergetic fruit concentrate, 7 d before and 48 h after exercise | Two trials of 10 sets of 10 single-leg knee extensions | N/A | PC↓ | N/A | N/A | |
| Pittaluga et al. [46] | Fresh red orange juice (ROJ) | ROJ versus nothing extra, thrice/day, 4 weeks | A single bout of exhaustive exercise by cycle ergometer (3 min warm-up, an initial load of 25 W, and further increments of 15 W/3 min) | N/A | MDA↓, ascorbic acid↓, hypoxanthine/xanthine↓ | N/A | N/A | |
| Chang et al. [47] | Purple sweet potato leaves (PSPL) | Standard cooked PSPL versus low-polyphenols diet, 7 days | Treadmill running at 70% VO2max, 1 h | N/A | PC↓ | IL-6↓, HSP72↔ | TAC (FRAP assay)↑, polyphenols↑ | |
| Mazani et al. [48] | Probiotic yoghurt | Probiotic yoghurt versus ordinary yoghurt, 2 weeks | Exhaustive exercise (Bruce test) | N/A | MDA↓ | TNF-α↓, MMP2↓, MMP9↓ | SOD↑, GPX↑, TAC↑, | |
| Harms-Ringdahl et al. [49] | Tomato juice | Tomato juice versus nothing extra, 5 weeks | Cycle ergometer at 80% of HRmax, 20 min | N/A | 8-oxodG↓ | N/A | N/A | |
| Kawamura et al. [50] | Mixed diet | Salmon flakes + green and yellow vegetable juice + lingonberry jam versus normal diet, 10 weeks | Resistance training twice/week, 10 weeks | N/A | PC↓ | N/A | N/A | |
| Sureda et al. [51] | Mixed beverage | Almond-based isotonic and energetic beverage with vitamin C and E versus Nonenriched beverage, 1 month | A half-marathon race (21 km-run) | N/A | MDA↓ | N/A | N/A | |
| Carrera-Quintanar et al. [52] | Mixed beverage | Mixed beverage with extra Lippia citriodora extract versus mixed beverage enriched with vitamins C and E, 22 days | 2000-m running exercise trial | N/A | PC↑ | N/A | SOD↓, GRD↓ | |
| M Daly et al. [53] | Multinutrient-fortified milk (MFMD) | MFMD versus placebo milk, twice/d, 4 months | Resistance exercise 3 d/week, 4 months | N/A | PC↔, 8-isoprostane↔ | N/A | N/A | |
| Inflammatory Markers | Koenig et al. [43], Zhang et al. [44] | AVA-enrich cookies | 4.6 mg AVA/cookie versus 0.2 mg AVA/cookie, 2 cookies/day, 8 weeks | Downhill running, 1 h | N/A | N/A | NRB [43]; NF-κB↓ and IL-6 [43]; G-CSF, IL-1Ra and sVCAM-1 [44] | N/A |
| Prasertsri et al. [54] | Cashew apple juice (CAJ) | CAJ versus placebo (isocaloric), 4 weeks | Cycling at 85% of VO2max, 20 min | N/A | MDA↓, 8-isoprostane↓, | Leukocyte count↑ | N/A | |
| Antioxidant Activity | Panza et al. [34] | Mate tea | Mate tea versus water, 11days, exercise and blood test were performed at 8th day | Three sets of twenty maximal eccentric elbow flexion exercises | N/A | N/A | N/A | GSH↑, GSSG↔, GSH/GSSG↔, LOOH↔ |
| Panze et al. [33] | Green tea | Green tea versus Water, three times/day, 7 days | A bench press exercise, four sets, 10 to 4 repetitions | N/A | LH↓ | N/A | TAC (FRAP assay)↑, total polyphenol↑, GSH↑ | |
| McLeay et al. [35] | New Zealand blueberry | Blueberry + banana + commercial apple juice versus Shake dextrose + banana + commercial apple juice (isocaloric); 5 and 10 h pre, immediately, 12 and 36 h after exercise | 3 sets × 100 eccentric repetitions of quadriceps muscle | N/A | PC↓ | IL-6↓ | TAC (FRAP assay)↑, ROS-GP↓ | |
| Park et al. [36] | Blueberry | Blueberry + aronia + sugar + refined water versus nothing extra, before exercise | Treadmill exercise (Bruce test) | N/A | N/A | IL-6↓, CRP↓ | TAS↑ | |
| Toscano et al. [38,39] | Grape | Integral grape juice versus isocaloric, isoglycemic and isovolumetric control beverage, 10 mL/kg/day, 2 h before exercise [38], or for 28 days [39] | Time-to exhaustion exercise test, anaerobic threshold test and aerobic capacity test | N/A | N/A | N/A | TAC↑ [38,39], UA↑ and vitamin A↑ [39] | |
| Howatson et al. [41] | Tart cherry | Tart cherry juice versus control, before, on the day of, and 48 h following exercise | A marathon run | N/A | MDA↓ | IL-6↓, CRP↓, UA↓ | TAS↑ | |
| Copetti et al. [45] | Juçara (Euterpe edulis Martius) | Juçara juice versus Water, 1 h before exercise | HIIT, 17 min | N/A | N/A | N/A | OSI↓ | |
| Iwasa et al. [55] | Fermented milk | Fermented milk (Lactobacillus helveticus) versus equivalent dose of unfermented milk, 1 h before and 2 h after exercise | Resistance exercise consisting of five sets of leg and bench presses | N/A | N/A | hsCRP↓, TNF-a↔ | TAC (ORAC assay)↑ | |
| Beavers et al. [56] | Soy | Soy versus dairy milk, 3 serving/d, 4 weeks | Downhill-running at 60% VO2max and −10% grade, 45 min | N/A | N/A | TNF-α↔, IL-1β↔, IL-6↔ | GPx↔, COX-2↔ | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeng, Z.; Centner, C.; Gollhofer, A.; König, D. Effects of Dietary Strategies on Exercise-Induced Oxidative Stress: A Narrative Review of Human Studies. Antioxidants 2021, 10, 542. https://doi.org/10.3390/antiox10040542
Zeng Z, Centner C, Gollhofer A, König D. Effects of Dietary Strategies on Exercise-Induced Oxidative Stress: A Narrative Review of Human Studies. Antioxidants. 2021; 10(4):542. https://doi.org/10.3390/antiox10040542
Chicago/Turabian StyleZeng, Zhen, Christoph Centner, Albert Gollhofer, and Daniel König. 2021. "Effects of Dietary Strategies on Exercise-Induced Oxidative Stress: A Narrative Review of Human Studies" Antioxidants 10, no. 4: 542. https://doi.org/10.3390/antiox10040542
APA StyleZeng, Z., Centner, C., Gollhofer, A., & König, D. (2021). Effects of Dietary Strategies on Exercise-Induced Oxidative Stress: A Narrative Review of Human Studies. Antioxidants, 10(4), 542. https://doi.org/10.3390/antiox10040542







