Modulation of the Visual to Auditory Human Inhibitory Brain Network: An EEG Dipole Source Localization Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Design
2.3. Acquisition and Pre-Processing of EEG Signals
2.4. Behavioral Analysis
2.5. Independent Component and Dipole Clusters Used as Regions of Interest (ROI)
2.6. Analysis of Brain Connectivity under Human Inhibitory Control
2.7. Statistical Analysis
3. Results
3.1. Behavioral Results
3.2. EEG Results
3.2.1. EEG-Scalp Maps and Dipole Source Locations
3.2.2. Event Related Spectral Perturbation (ERSP) Analysis
3.2.3. The Neural Connectivity of Visual and Auditory Modalities in Human Inhibitory Control
3.2.4. Change of Visual to Auditory Neural Networks
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Logan, G.D. On the ability to inhibit thought and action: A users’ guide to the stop-signal paradigm. In Inhibitory Processes in Attention, Memory, and Language; Dagenbach, D., Carr, T.H., Eds.; Academic Press: San Diego, CA, USA, 1994; pp. 188–239. [Google Scholar]
- Serrien, D.J.; Orth, M.; Evans, A.H.; Lees, A.J.; Brown, P. Motor inhibition in patients with Gilles de la Tourette syndrome: Functional activation patterns as revealed by EEG coherence. Brain 2005, 128, 116–125. [Google Scholar] [CrossRef]
- Chambers, C.D.; Garavan, H.; Bellgrove, M.A. Insights into the neural basis of response inhibition from cognitive and clinical neuroscience. Neurosci. Biobehav. Rev. 2009, 33, 631–646. [Google Scholar] [CrossRef]
- Aron, A.R.; Dowson, J.H.; Sahakian, B.J.; Robbins, T.W. Methylphenidate improves response inhibition in adults with attention-deficit/hyperactivity disorder. Biol. Psychiatry 2003, 54, 1465–1468. [Google Scholar] [CrossRef]
- Elchlepp, H.; Lavric, A.; Chambers, C.D.; Verbruggena, F. Proactive inhibitory control: A general biasing account. Cogn. Psychol. 2016, 86, 27–61. [Google Scholar] [CrossRef] [Green Version]
- Vingerhoets, G.; Acke, F.; Alderweireldt, A.S.; Nys, J.; Vandemaele, P.; Achten, E. Cerebral lateralization of praxis in right- and left-handedness: Same pattern, different strength. Hum. Brain Mapp. 2012, 33, 763–777. [Google Scholar] [CrossRef]
- Reid, C.S.; Serrien, D.J. Handedness and the excitability of cortical inhibitory circuits. Behav. Brain Res. 2012, 230, 144–148. [Google Scholar] [CrossRef]
- Kloppel, S.; van Eimeren, T.; Glauche, V.; Vongerichten, A.; Munchau, A.; Frackowiak, R.S. The effect of handedness on cortical motor activation during simple bilateral movements. Neuroimage 2007, 34, 274–280. [Google Scholar] [CrossRef]
- Busch, N.A.; Dubois, J.; Van Rullen, R. The phase of ongoing EEG oscillations predicts visual perception. J. Neurosci. 2009, 29, 7869–7876. [Google Scholar] [CrossRef]
- Mathewson, K.E.; Gratton, G.; Fabiani, M.; Beck, D.M.; Ro, T. To see or not to see: Prestimulus alpha phase predicts visual awareness. J. Neurosci. 2009, 29, 2725–2732. [Google Scholar] [CrossRef]
- King, A.J. Visual influences on auditory spatial learning. Philos. Trans. R Soc. Lond B Biol. Sci. 2009, 364, 331–339. [Google Scholar] [CrossRef]
- Martini, F.; Nath, J. Anatomy & Physiology, 2nd ed.; Benjamin Cummings: San Frascisco, CA, USA, 2010. [Google Scholar]
- Nicholls, J.; Martin, A.R.; Wallace, B.; Fuchs, P. From Neuron to Brain, 4th ed.; Sinauer: Sunderland, MA, USA, 2001. [Google Scholar]
- Senkowski, D.; Schneider, T.R.; Foxe, J.J.; Engel, A.K. Crossmodal binding by neural coherence: Implications for multisensory processing. Trends Neurosci. 2008, 31, 401–409. [Google Scholar] [CrossRef]
- Kaiser, J.; Naumer, M.J. Cortical oscillations and multisensory interactions in humans. In Multisensory Object Perception in the Primate Brain; Naumer, M.J., Kaiser, J., Eds.; Springer: Heidelberg, Germany, 2010; pp. 71–82. [Google Scholar]
- Verbruggen, F.; Logan, G.D.; Stevens, M.A. STOP-IT: Windows executable software for the stop-signal paradigm. Behav. Res. Methods 2008, 40, 479–483. [Google Scholar] [CrossRef] [Green Version]
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef]
- Jung, T.P.; Makeig, S.; Westerfield, M.; Townsend, J.; Courchesne, E.; Sejnowski, T.J. Removal of eye activity artifacts from visual event-related potentials in normal and clinical subjects. Clin. Neurophysiol. 2000, 111, 1745–1758. [Google Scholar] [CrossRef]
- Jung, T.P.; Makeig, S.; McKeown, M.J.; Bell, A.J.; Lee, T.W.; Sejnowski, T.J. Imaging brain dynamics using independent component analysis. Proc. IEEE. 2001, 89, 1107–1122. [Google Scholar] [CrossRef]
- Makeig, S.; Debener, S.; Onton, J.; Delorme, A. Mining event-related brain dynamics. Trends. Cogn. Sci. 2004, 8, 204–210. [Google Scholar] [CrossRef] [Green Version]
- Palva, J.M.; Palva, S.; Kaila, K. Phase synchrony among neuronal oscillations in the human cortex. J. Neurosci. 2005, 25, 3962–3972. [Google Scholar] [CrossRef]
- Lakatos, P.; O’Connell, M.N.; Barczak, A.; Mills, A.; Javitt, D.C.; Schroeder, C.E. The leading sense: Supramodal control of neurophysiological context by attention. Neuron 2009, 64, 419–430. [Google Scholar] [CrossRef]
- Thorne, J.D.; De vos, M.; Viola, F.C.; Debener, S. Cross-modal phase reset predicts auditory task performance in humans. J. Neurosci. 2011, 31, 3853–3861. [Google Scholar] [CrossRef]
- Bowyer, S.M. Coherence a measure of the brain networks: Past and present. Bowyer Neuropsychiatr. Electrophysiol. 2016, 2, 1. [Google Scholar] [CrossRef]
- Tiesinga, P.H.; Sejnowski, T.J. Mechanisms for phase shifting in cortical networks and their role in communication through coherence. Front. Hum. Neurosci. 2010, 4, 1–14. [Google Scholar] [CrossRef]
- Thatcher, R.W. Coherence, Phase Differences, Phase Shift, and Phase Lock in EEG/ERP Analyses. Dev. Neuropsychol. 2012, 37, 476–496. [Google Scholar] [CrossRef]
- Riddle, C.N.; Baker, S.N. Manipulation of peripheral neural feedback loops alters human corticomuscular coherence. J. Physiol. 2005, 566, 625–639. [Google Scholar] [CrossRef] [Green Version]
- Thatcher, R.W.; North, D.; Biver, C. Development of cortical connectivity as measured by EEG coherence and phase. Hum. Brain Mapp. 2008, 29, 1400–1415. [Google Scholar] [CrossRef]
- Jung-Beeman, M.; Bowden, E.M.; Haberman, J.; Frymiare, J.L.; Arambel-Liu, S.; Greenblatt, R.; Reber, P.J.; Kounios, J. Neural activity when people solve verbal problems with insight. PLoS Biol. 2004, 2, E97. [Google Scholar] [CrossRef]
- Vander Ghinst, M.; Bourguignon, M.; Op de Beeck, M.; Wens, V.; Marty, B.; Hassid, S.; Choufani, G.; Jousmäki, V.; Hari, R.; Van Bogaert, P.; et al. Left Superior Temporal Gyrus Is Coupled to Attended Speech in a Cocktail-Party Auditory Scene. J. Neurosci. 2016, 36, 1596–1606. [Google Scholar] [CrossRef]
- Silvia, P.G.; Rebecca, E.; Millman, M.H.; Sven, L.M. Anterior paracingulate and cingulate cortex mediates the effects of cognitiveload on speech sound discrimination. NeuroImage 2018, 178, 735–743. [Google Scholar]
- Drevets, W.C.; Savitz, J.; Trimble, M. The subgenual anterior cingulate cortex in mood disorders. CNS Spectr. 2008, 13, 663–681. [Google Scholar] [CrossRef]
- Adams, R.; David, A.S. Patterns of anterior cingulate activation in schizophrenia: A selective review. Neuropsychiatr. Dis. Treat. 2007, 3, 87–101. [Google Scholar] [CrossRef]
- Ko, L.-W.; Shih, Y.-C.; Chikara, R.K.; Chuang, Y.-T.; Chang, E.C. Neural Mechanisms of Inhibitory Response in a Battlefield Scenario: A Simultaneous fMRI-EEG Study. Front. Hum. Neurosci. 2016, 10, 185. [Google Scholar] [CrossRef] [Green Version]
- Chikara, R.K.; Chang, E.C.; Lu, Y.C.; Lin, D.S.; Lin, C.T.; Ko, L.W. Monetary Reward and Punishment to Response Inhibition Modulate Activation and Synchronization Within the Inhibitory Brain Network. Front. Hum. Neurosci. 2018, 12, 27. [Google Scholar] [CrossRef] [Green Version]
- Mishkin, M.; Ungerleider, L.G. Contribution of striate inputs to the visuospatial functions of parieto-preoccipital cortex in monkeys. Behav. Brain Res. 1982, 6, 57–77. [Google Scholar] [CrossRef]
- Goodale, M.A.; Milner, A.D. Separate visual pathways for perception and action. Trends Neurosci. 1992, 15, 20–25. [Google Scholar] [CrossRef]
- Fogassi, L.; Luppino, G. Motor functions of the parietal lobe. Curr. Opin. Neurobiol. 2005, 15, 626–631. [Google Scholar] [CrossRef]
- Tabu, H.; Mima, T.; Aso, T.; Takahashi, R.; Fukuyama, H. Common inhibitory prefrontal activation during inhibition of hand and foot responses. Neuroimage 2012, 59, 3373–3378. [Google Scholar] [CrossRef]
- Bell, A.J.; Sejnowski, T.J. An information-maximization approach to blind separation and blind deconvolution. Neural. Comput. 1995, 7, 1129–1159. [Google Scholar] [CrossRef]
- Challis, R.E.; Kitney, R.I. Biomedical signal processing (in four parts). Part 3: The power spectrum and coherence function. Med. Biol. Eng. Comput. 1991, 29, 225–241. [Google Scholar] [CrossRef]
- Delorme, A.; Mullen, T.; Kothe, C.; Akalin Acar, Z.; Bigdely-Shamlo, N.; Vankov, A.; Makeig, S. EEGLAB, SIFT, NFT, BCILAB, and ERICA: New tools for advanced EEG processing. Comput. Intell. Neurosci. 2011, 1–12. [Google Scholar] [CrossRef]
- Efron, B.; Tibshirani, R.J. An Introduction to the Bootstrap; Monographs on Statistics & Applied Probability; Chapman Hall/CRC: Boca Raton, FL, USA, 1994; Volume 1, 456p. [Google Scholar]
- Duann, J.R.; Ide, J.S.; Luo, X.; Li, C.S. Functional connectivity delineates distinct roles of the inferior frontal cortex and presupplementary motor area in stop signal inhibition. J. Neurosci. 2009, 29, 10171–10179. [Google Scholar] [CrossRef]
- Pain, T.G.M.; Hibbs, A. Sprint Starts and the Minimum Auditory Reaction Time. J. Sports Sci. 2007, 25, 79–86. [Google Scholar] [CrossRef]
- Shelton, J.; Kumar, G.P. Comparison between Auditory and Visual Simple Reaction Times. Neurosci. Med. 2010, 1, 30–32. [Google Scholar] [CrossRef] [Green Version]
- Ko, L.W.; Komarov, O.; Hairston, W.D.; Jung, T.P.; Lin, C.T. Sustained Attention in Real Classroom Settings: An EEG Study. Front. Hum. Neurosci. 2017, 11, 388. [Google Scholar] [CrossRef]
- Naue, N.; Rach, S.; Struber, D.; Huster, R.J.; Zaehle, T.; Korner, U.; Herrmann, C.S. Auditory event-related response in visual cortex modulates subsequent visual responses in humans. J. Neurosci. 2011, 21, 7729–7736. [Google Scholar] [CrossRef]
- Sumby, W.H.; Pollack, I. Visual contributions to speech intelligibility in noise. J. Acoust. Soc. Am. 1954, 26, 212–215. [Google Scholar] [CrossRef]
- Fehr, T.; Milz, P. The individuality index: A measure to quantify the degree of interindividual, spatial variability in intra-cerebral brain electric and metabolic activity. Cogn. Neurodyn. 2019. [Google Scholar] [CrossRef]
- Fuster, J.M. Cortex and Memory: Emergence of a New Paradigm. J. Cogn. Neurosci. 2009, 21, 2047–2072. [Google Scholar] [CrossRef]
- McGurk, H.; MacDonald, J. Hearing lips and seeing voices. Nature 1976, 264, 746–748. [Google Scholar] [CrossRef]
- Mowery, T.M.; Kotak, V.C.; Sanes, D.H. The onset of visual experience gates auditory cortex critical periods. Nat. Commun. 2016, 7, 10416. [Google Scholar] [CrossRef]
- Molholm, S.; Ritter, W.; Murray, M.M.; Javitt, D.C.; Schroeder, C.E.; Foxe, J.J. Multisensory auditory–visual interactions during early sensory processing in humans: A high-density electrical mapping study. Brain Res. Cogn. 2002, 14, 115–128. [Google Scholar] [CrossRef]
- Giard, M.H.; Peronnet, F. Auditory-visual integration during multimodal object recognition in humans: A behavioral and electrophysiological study. J. Cogn. Neurosci. 1999, 11, 473–490. [Google Scholar] [CrossRef]
- Schroeder, C.E.; Lakatos, P.; Kajikawa, Y.; Partan, S.; Puce, A. Neuronal oscillations and visual amplification of speech. Trends Cogn. Sci. 2008, 12, 106–113. [Google Scholar] [CrossRef] [Green Version]
- Schroeder, C.E.; Foxe, J. Multisensory contributions to low-level, ‘unisensory’ processing. Curr. Opin. Neurobiol. 2005, 15, 454–458. [Google Scholar] [CrossRef]
- Bizley, J.K.; King, A.J. Visual-auditory spatial processing in auditory cortical neurons. Brain Res. 2008, 1242, 24–36. [Google Scholar] [CrossRef]
- King, A.J. The superior colliculus. Curr. Biol. 2004, 14, R335–R338. [Google Scholar] [CrossRef] [Green Version]
- Stein, B.E.; Stanford, T.R. Multisensory integration: Current issues from the perspective of the single neuron. Nat. Rev. Neurosci. 2008, 9, 255–266. [Google Scholar] [CrossRef]
- Stein, B.E.; Stanford, T.R.; Ramachandran, R.; Perrault, T.J.J.; Rowland, B.A. Challenges in quantifying multisensory integration: Alternative criteria, models, and inverse effectiveness. Exp. Brain Res. 2009, 198, 113–126. [Google Scholar] [CrossRef]
- Sakowitz, O.W.; Quian, Q.R.; Schurmann, M.; Basar, E. Spatio-temporal frequency characteristics of intersensory components in audiovisually evoked potentials. Brain Res. Cogn. Brain Res. 2005, 23, 316–326. [Google Scholar] [CrossRef]
- Demiralp, T.; Bayraktaroglu, Z.; Lenz, D.; Junge, S.; Busch, N.A.; Maess, B.; Ergen, M.; Herrmann, C.S. Gamma amplitudes are coupled to theta phase in human EEG during visual perception. Int. J. Psychophysiol. 2007, 64, 24–30. [Google Scholar] [CrossRef]
- Lakatos, P.; Chen, C.M.; O’Connell, M.N.; Mills, A.; Schroeder, C.E. Neuronal oscillations and multisensory interaction in primary auditory cortex. Neuron 2007, 53, 279–292. [Google Scholar] [CrossRef]
- Driver, J.; Noesselt, T. Multisensory interplay reveals crossmodal influences on ‘sensory-specific’ brain regions, neural responses, and judgments. Neuron 2008, 57, 11–23. [Google Scholar] [CrossRef]


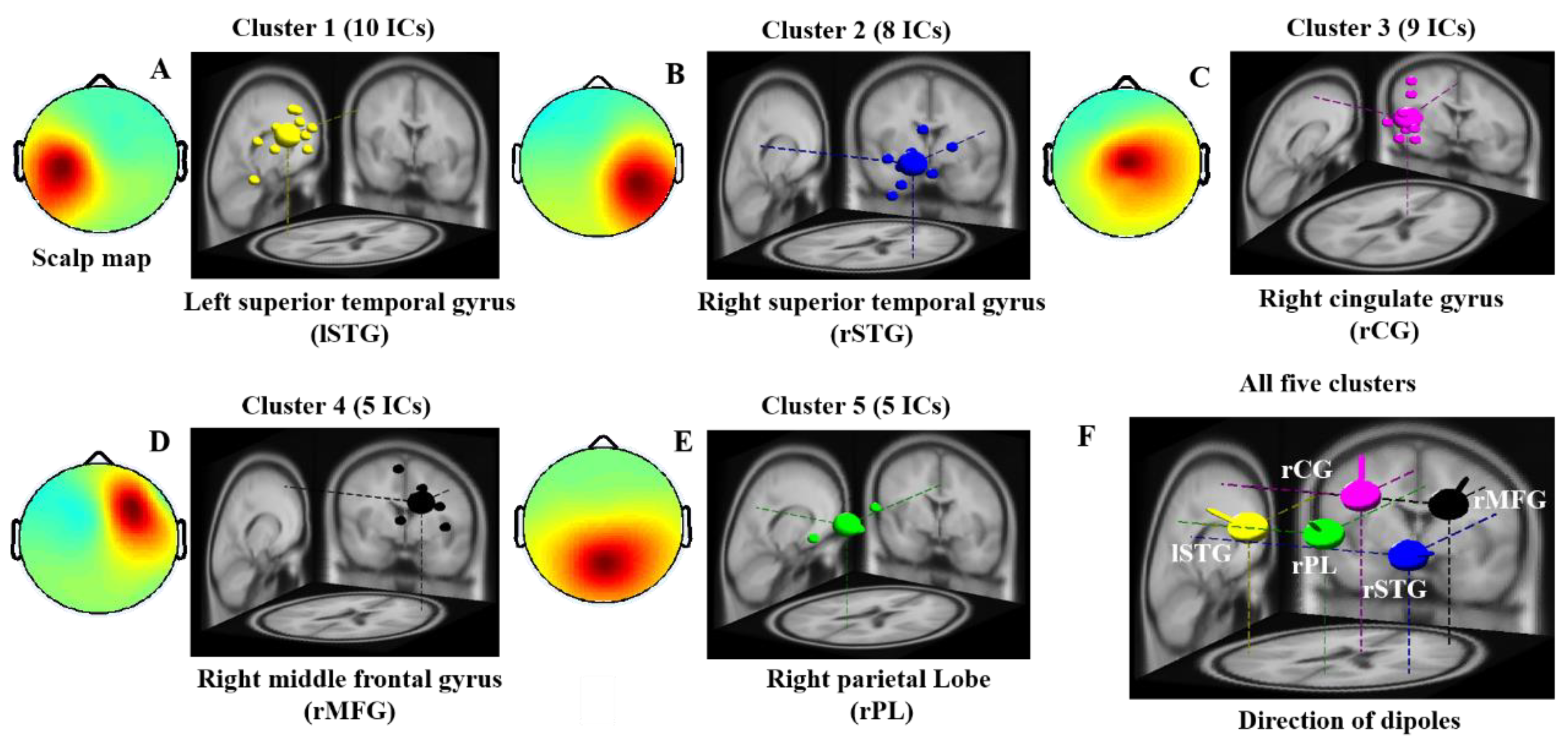







| Component Clusters | Side | Brain Regions | MNI Coordinates (mm) | Cluster Size (voxels) | ||
|---|---|---|---|---|---|---|
| X | Y | Z | ||||
| 1 | Left | Superior Temporal Gyrus | −59 | −25 | 30 | 18 |
| 2 | Right | Superior Temporal Gyrus | 51 | −40 | 24 | 12 |
| 3 | Right | Cingulate Gyrus | 0 | 4 | 56 | 15 |
| 4 | Right | Middle Frontal Gyrus | 43 | 36 | 45 | 59 |
| 5 | Right | Parietal Lobe | 5 | −60 | 42 | 55 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chikara, R.K.; Ko, L.-W. Modulation of the Visual to Auditory Human Inhibitory Brain Network: An EEG Dipole Source Localization Study. Brain Sci. 2019, 9, 216. https://doi.org/10.3390/brainsci9090216
Chikara RK, Ko L-W. Modulation of the Visual to Auditory Human Inhibitory Brain Network: An EEG Dipole Source Localization Study. Brain Sciences. 2019; 9(9):216. https://doi.org/10.3390/brainsci9090216
Chicago/Turabian StyleChikara, Rupesh Kumar, and Li-Wei Ko. 2019. "Modulation of the Visual to Auditory Human Inhibitory Brain Network: An EEG Dipole Source Localization Study" Brain Sciences 9, no. 9: 216. https://doi.org/10.3390/brainsci9090216




