Comparison of Transcranial Direct Current Stimulation Electrode Montages for the Lower Limb Motor Cortex
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Study Design
2.3. Electromyography and Maximum Voluntary Contractions
2.4. Transcranial Magnetic Stimulation
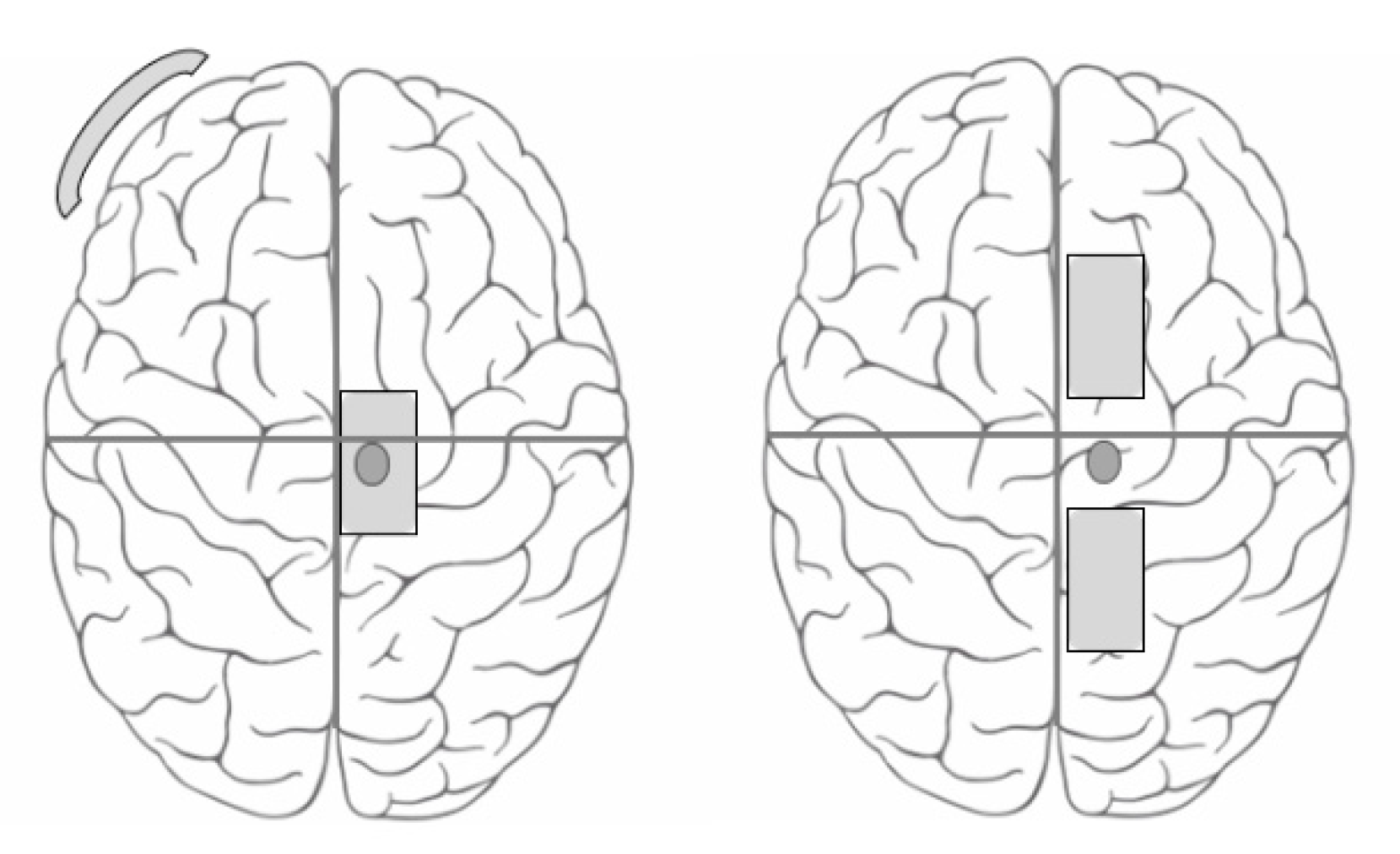
2.5. Transcranial Direct Current Stimulation
- (a)
- Traditional Electrode Montage (TRAD): The conventional electrode placement with the active electrode over the TA hotspot and the reference electrode over the contralateral supra-orbital region [19,20,21,33]. The stimulating electrode was a 13 cm2 (5 cm × 2.6 cm) butterfly saline-soaked electrode and the reference electrode was a 35 cm2 (7 cm × 5 cm) self-adhesive carbonized electrode.
- (b)
- Anterior Posterior Montage (AP): The active electrode was placed 5 cm posterior to the tibialis anterior hotspot and the reference electrode was placed 5 cm anterior to the hotspot [18]. Both the stimulating and reference electrodes were 13 cm2 (5 cm × 2.6 cm) butterfly saline soaked electrodes, as it was not possible to fix the self-adhesive carbonized electrode (that was used as a reference electrode in the TRAD montage) to the scalp. The sponge electrodes were held on the scalp using a comfortably wrapped elastic bandage wrap.
2.6. Motor Control Task
2.7. Data Analyses
2.8. TMS
2.9. Statistical Analyses
3. Results
3.1. CME Changes
3.2. Ankle Motor Control
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Priori, A. Brain polarization in humans: A reappraisal of an old tool for prolonged non-invasive modulation of brain excitability. Clin. Neurophysiol. 2003, 114, 589–595. [Google Scholar] [CrossRef]
- Madhavan, S.; Shah, B. Enhancing Motor Skill Learning with Transcranial Direct Current Stimulation—A concise review with applications to stroke. Front. Psychiatry 2012, 3, 66. [Google Scholar] [CrossRef]
- Lefaucheur, J.P.; Antal, A.; Ayache, S.S.; Benninger, D.H.; Brunelin, J.; Cogiamanian, F.; Cotelli, M.; De Ridder, D.; Ferrucci, R.; Langguth, B.; et al. Evidence-based guidelines on the therapeutic use of transcranial direct current stimulation (tDCS). Clin. Neurophysiol. 2017, 128, 56–92. [Google Scholar] [CrossRef]
- Elsner, B.; Kugler, J.; Pohl, M.; Mehrholz, J. Transcranial direct current stimulation (tDCS) for improving activities of daily living, and physical and cognitive functioning, in people after stroke. Cochrane Database Syst. Rev. 2016, 3, CD009645. [Google Scholar] [CrossRef]
- Marquez, J.; van Vliet, P.; McElduff, P.; Lagopoulos, J.; Parsons, M. Transcranial direct current stimulation (tDCS): Does it have merit in stroke rehabilitation? A systematic review. Int. J. Stroke 2015, 10, 306–316. [Google Scholar] [CrossRef]
- Horvath, J.C.; Forte, J.D.; Carter, O. Evidence that transcranial direct current stimulation (tDCS) generates little-to-no reliable neurophysiologic effect beyond MEP amplitude modulation in healthy human subjects: A systematic review. Neuropsychologia 2015, 66, 213–236. [Google Scholar] [CrossRef]
- Chew, T.; Ho, K.A.; Loo, C.K. Inter-and Intra-individual Variability in Response to Transcranial Direct Current Stimulation (tDCS) at Varying Current Intensities. Brain Stimul. 2015, 8, 1130–1137. [Google Scholar] [CrossRef]
- Lopez-Alonso, V.; Cheeran, B.; Rio-Rodriguez, D.; Fernández-Del-Olmo, M. Inter-individual variability in response to non-invasive brain stimulation paradigms. Brain Stimul. 2014, 7, 372–380. [Google Scholar] [CrossRef]
- Wiethoff, S.; Hamada, M.; Rothwell, J.C. Variability in response to transcranial direct current stimulation of the motor cortex. Brain Stimul. 2014, 7, 468–475. [Google Scholar] [CrossRef]
- Madhavan, S.; Sriraman, A.; Freels, S. Reliability and Variability of tDCS Induced Changes in the Lower Limb Motor Cortex. Brain Sci. 2016, 6, 26. [Google Scholar] [CrossRef]
- Bikson, M.; Datta, A.; Rahman, A.; Scaturro, J. Electrode montages for tDCS and weak transcranial electrical stimulation: Role of “return” electrode’s position and size. Clin. Neurophysiol. 2010, 121, 1976–1978. [Google Scholar] [CrossRef] [PubMed]
- Cancelli, A.; Cottone, C.; Parazzini, M.; Fiocchi, S.; Truong, D.; Bikson, M.; Tecchio, F.; Parazzini, M. Transcranial Direct Current Stimulation: Personalizing the neuromodulation. In Proceedings of the 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015; pp. 234–237. [Google Scholar] [CrossRef]
- Saturnino, G.B.; Antunes, A.; Thielscher, A. On the importance of electrode parameters for shaping electric field patterns generated by tDCS. Neuroimage 2015, 120, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Guerra, A.; Lopez-Alonso, V.; Cheeran, B.; Suppa, A. Solutions for managing variability in non-invasive brain stimulation studies. Neurosci. Lett. 2017. [Google Scholar] [CrossRef] [PubMed]
- Guerra, A.; Lopez-Alonso, V.; Cheeran, B.; Suppa, A. Variability in non-invasive brain stimulation studies: Reasons and results. Neurosci. Lett. 2017. [Google Scholar] [CrossRef] [PubMed]
- Lefaucheur, J.P. A comprehensive database of published tDCS clinical trials (2005–2016). Neurophysiol. Clin. 2016, 46, 319–398. [Google Scholar] [CrossRef] [PubMed]
- Miranda, P.C.; Lomarev, M.; Hallett, M. Modeling the current distribution during transcranial direct current stimulation. Clin. Neurophysiol. 2006, 117, 1623–1629. [Google Scholar] [CrossRef] [PubMed]
- Rampersad, S.M.; Janssen, A.M.; Lucka, F.; Aydin, Ü.; Lanfer, B.; Lew, S.; Wolters, C.H.; Stegeman, D.F.; Oostendorp, T.F. Simulating transcranial direct current stimulation with a detailed anisotropic human head model. IEEE Trans. Neural Syst. Rehabil. Eng. 2014, 22, 441–452. [Google Scholar] [CrossRef]
- Madhavan, S.; Stinear, J.W. Focal and bidirectional modulation of lower limb motor cortex using anodal transcranial direct current stimulation. Brain Stimul. 2010, 3, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Sriraman, A.; Oishi, T.; Madhavan, S. Timing-dependent priming effects of tDCS on ankle motor skill learning. Brain Res. 2014, 1581, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Devanathan, D.; Madhavan, S. Effects of anodal tDCS of the lower limb M1 on ankle reaction time in young adults. Exp. Brain Res. 2016, 234, 377–385. [Google Scholar] [CrossRef]
- Foerster, A.S.; Rezaee, Z.; Paulus, W.; Nitsche, M.A.; Dutta, A. Effects of Cathode Location and the Size of Anode on Anodal Transcranial Direct Current Stimulation Over the Leg Motor Area in Healthy Humans. Front. Neurosci. 2018, 12, 443. [Google Scholar] [CrossRef] [PubMed]
- Keel, J.C.; Smith, M.J.; Wassermann, E.M. A safety screening questionnaire for transcranial magnetic stimulation. Clin. Neurophysiol. 2001, 112, 720. [Google Scholar] [CrossRef]
- Cerqueira, V.; de Mendonca, A.; Minez, A.; Dias, A.R.; de Carvalho, M. Does caffeine modify corticomotor excitability? Neurophysiol. Clin. 2006, 36, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Tamm, A.S.; Lagerquist, O.; Ley, A.L.; Collins, D.F. Chronotype influences diurnal variations in the excitability of the human motor cortex and the ability to generate torque during a maximum voluntary contraction. J. Biol. Rhythms. 2009, 24, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Stagg, C.J.; Jayaram, G.; Pastor, D.; Kincses, Z.T.; Matthews, P.M.; Johansen-Berg, H. Polarity and timing-dependent effects of transcranial direct current stimulation in explicit motor learning. Neuropsychologia 2011, 49, 800–804. [Google Scholar] [CrossRef] [PubMed]
- Shah, B.; Nguyen, T.T.; Madhavan, S. Polarity independent effects of cerebellar tDCS on short term ankle visuomotor learning. Brain Stimul. 2013, 6, 966–968. [Google Scholar] [CrossRef] [PubMed]
- Madhavan, S.; Rogers, L.M.; Stinear, J.W. A paradox: After stroke, the non-lesioned lower limb motor cortex may be maladaptive. Eur. J. Neurosci. 2010, 32, 1032–1039. [Google Scholar] [CrossRef] [PubMed]
- Sivaramakrishnan, A.; Tahara-Eckl, L.; Madhavan, S. Spatial localization and distribution of the TMS-related ‘hotspot’ of the tibialis anterior muscle representation in the healthy and post-stroke motor cortex. Neurosci. Lett. 2016, 627, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Bikson, M.; Datta, A.; Elwassif, M. Establishing safety limits for transcranial direct current stimulation. Clin. Neurophysiol. 2009, 120, 1033–1034. [Google Scholar] [CrossRef] [PubMed]
- Nitsche, M.A.; Liebetanz, D.; Antal, A.; Lang, N.; Tergau, F.; Paulus, W. Modulation of cortical excitability by weak direct current stimulation—Technical, safety and functional aspects. Suppl. Clin. Neurophysiol. 2003, 56, 255–276. [Google Scholar]
- McCreery, D.B.; Agnew, W.F.; Yuen, T.G.; Bullara, L. Charge density and charge per phase as cofactors in neural injury induced by electrical stimulation. IEEE Trans. Biomed. Eng. 1990, 37, 996–1001. [Google Scholar] [CrossRef] [PubMed]
- Madhavan, S.; Weber, K.A.; Stinear, J.W., 2nd. Non-invasive brain stimulation enhances fine motor control of the hemiparetic ankle: Implications for rehabilitation. Exp. Brain Res. 2011, 209, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Pukelsheim, F. The Three Sigma Rule. Am. Stat. 1994, 48, 88–91. [Google Scholar]
- Labruna, L.; Jamil, A.; Fresnoza, S.; Batsikadze, G.; Kuo, M.F.; Vanderschelden, B.; Ivry, R.B.; Nitsche, M.A. Efficacy of Anodal Transcranial Direct Current Stimulation is Related to Sensitivity to Transcranial Magnetic Stimulation. Brain Stimul. 2016, 9, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Kim, Y.H.; Im, C.H.; Kim, J.H.; Park, C.H.; Chang, W.H.; Lee, A. What is the optimal anodal electrode position for inducing corticomotor excitability changes in transcranial direct current stimulation? Neurosci. Lett. 2015, 584, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Dmochowski, J.P.; Datta, A.; Bikson, M.; Su, Y.; Parra, L.C. Optimized multi-electrode stimulation increases focality and intensity at target. J. Neural Eng. 2011, 8, 046011. [Google Scholar] [CrossRef] [PubMed]
- Faria, P.; Hallett, M.; Miranda, P.C. A finite element analysis of the effect of electrode area and inter-electrode distance on the spatial distribution of the current density in tDCS. J. Neural Eng. 2011, 8, 066017. [Google Scholar] [CrossRef]
- Nitsche, M.A.; Doemkes, S.; Karakose, T.; Antal, A.; Liebetanz, D.; Lang, N.; Tergau, F.; Paulus, W. Shaping the effects of transcranial direct current stimulation of the human motor cortex. J. Neurophysiol. 2007, 97, 3109–3117. [Google Scholar] [CrossRef]
- Moliadze, V.; Antal, A.; Paulus, W. Electrode-distance dependent after-effects of transcranial direct and random noise stimulation with extracephalic reference electrodes. Clin. Neurophysiol. 2010, 121, 2165–2171. [Google Scholar] [CrossRef]
- Smith, M.J.; Keel, J.C.; Greenberg, B.D.; Adams, L.F.; Schmidt, P.J.; Rubinow, D.A.; Wassermann, E.M. Menstrual cycle effects on cortical excitability. Neurology 1999, 53, 2069–2072. [Google Scholar] [CrossRef]
- Inghilleri, M.; Conte, A.; Curra, A.; Frasca, V.; Lorenzano, C.; Berardelli, A. Ovarian hormones and cortical excitability. An rTMS study in humans. Clin. Neurophysiol. 2004, 115, 1063–1068. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, S.; Liew, S.L. Anatomical Parameters of tDCS to Modulate the Motor System after Stroke: A Review. Front. Neurol. 2017, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Jedlicka, P. Synaptic plasticity, metaplasticity and BCM theory. Bratisl. Lek. Listy. 2002, 103, 137–143. [Google Scholar] [PubMed]
- Sadleir, R.J.; Vannorsdall, T.D.; Schretlen, D.J.; Gordon, B. Target optimization in transcranial direct current stimulation. Front. Psychiatry 2012, 3, 90. [Google Scholar] [CrossRef] [PubMed]
- Im, C.H.; Park, J.H.; Shim, M.; Chang, W.H.; Kim, Y.H. Evaluation of local electric fields generated by transcranial direct current stimulation with an extracephalic reference electrode based on realistic 3D body modeling. Phys. Med. Biol. 2012, 57, 2137–2150. [Google Scholar] [CrossRef]
| Traditional | Anterior–Posterior | |||
|---|---|---|---|---|
| N | Change in CME | N | Change in CME | |
| All | 17 | 9.4 ± 7.0% | 17 | −0.6 ± 3.0% |
| Responders | 10 | 36 ± 10.0% * | 6 | 18 ± 3.2% * |
| Non-responders | 7 | −12 ± 4.3% | 11 | −11 ± 2.0% |
| Traditional | Anterior–Posterior | |||
|---|---|---|---|---|
| N | Change in AI | N | Change in AI | |
| All | 17 | 6.4 ± 2.85% | 17 | 7.19 ± 1.37% |
| Responders | 10 | 14.6 ± 4.9% * | 6 | 8 ± 3.9% |
| Non-responders | 7 | 0.09 ± 3.4% | 11 | 6.6 ± 1.3% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patel, R.; Madhavan, S. Comparison of Transcranial Direct Current Stimulation Electrode Montages for the Lower Limb Motor Cortex. Brain Sci. 2019, 9, 189. https://doi.org/10.3390/brainsci9080189
Patel R, Madhavan S. Comparison of Transcranial Direct Current Stimulation Electrode Montages for the Lower Limb Motor Cortex. Brain Sciences. 2019; 9(8):189. https://doi.org/10.3390/brainsci9080189
Chicago/Turabian StylePatel, Radhika, and Sangeetha Madhavan. 2019. "Comparison of Transcranial Direct Current Stimulation Electrode Montages for the Lower Limb Motor Cortex" Brain Sciences 9, no. 8: 189. https://doi.org/10.3390/brainsci9080189
APA StylePatel, R., & Madhavan, S. (2019). Comparison of Transcranial Direct Current Stimulation Electrode Montages for the Lower Limb Motor Cortex. Brain Sciences, 9(8), 189. https://doi.org/10.3390/brainsci9080189





