Target Size Manipulations Affect Error-Processing Duration and Success Perceptions but not Behavioural Indices of Learning
Abstract
:1. Introduction
1.1. Evidence that Success-Enhancing Manipulations Benefit Learning
1.2. Potential Mechanisms for Success Perception Effects on Motor Learning
1.3. Evidence that Success-Enhancing Manipulations Do Not Benefit Motor Learning
1.4. Aims of Present Study
1.5. Hypotheses
1.5.1. Behavioural and Psychological Measures
1.5.2. Transfer and Process Measures
2. Materials and Methods
2.1. Participants and Groups
2.2. Task and Apparatus
2.3. Procedure
2.3.1. Pre-Test
2.3.2. Acquisition
2.3.3. Delayed Tests of Learning
2.4. Data Collection and Analysis
2.4.1. Outcome Variables
2.4.2. Measures of Success, Competence, and Motivation
2.4.3. Process and Motor-Control Measures
3. Results
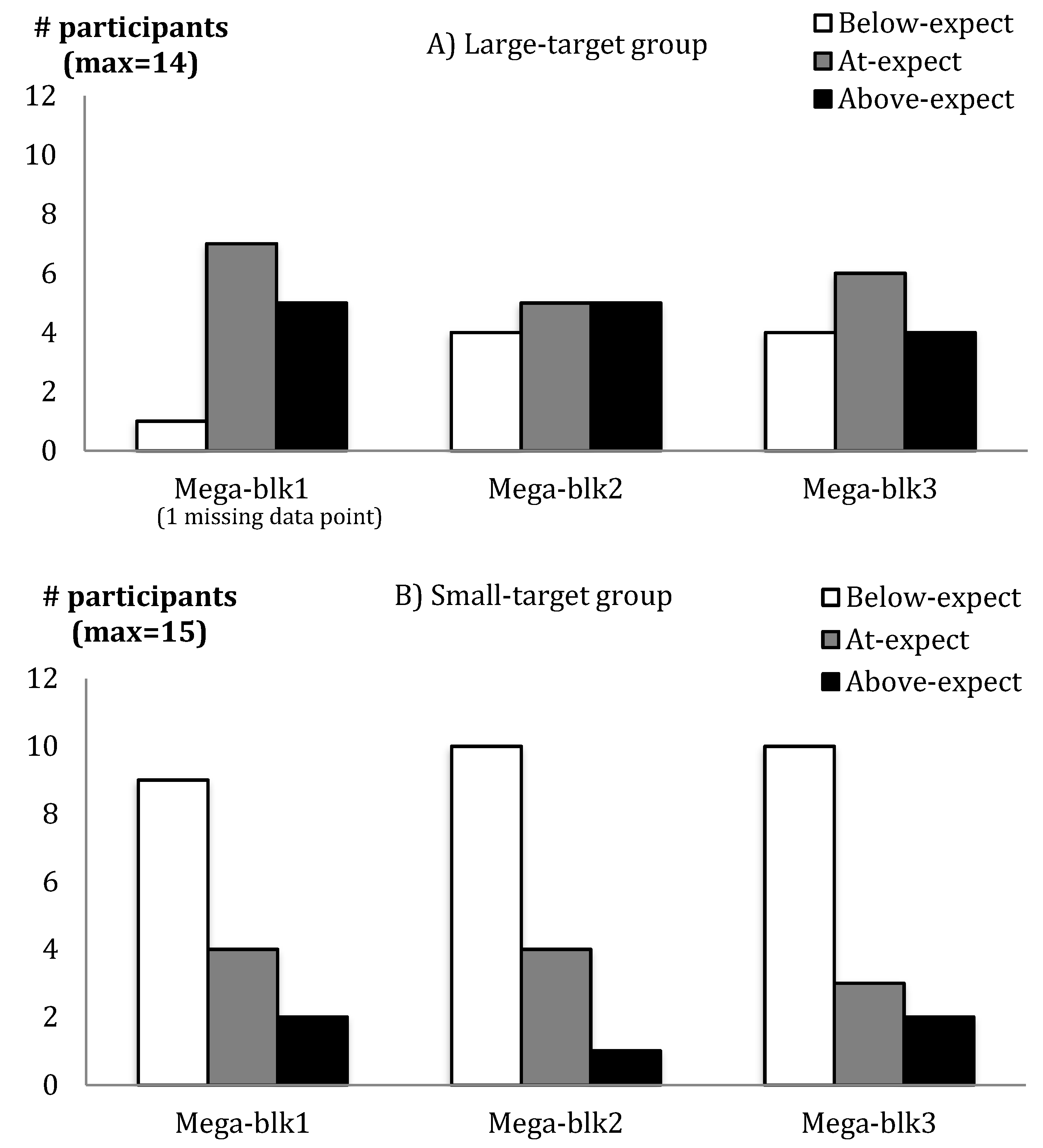
3.1. Success Rates and Success Perceptions Were Affected by Target Size Manipulations
3.2. Success Rates and Perceptions Did not Impact Outcome Measures
3.2.1. No Vision Pre-Test
3.2.2. Acquisition
3.2.3. Retention Tests
3.2.4. Other Delayed Tests of Learning
3.3. Success Did Not Impact Motivation
3.4. Processing Durations and Movement Kinematics Were Affected by Target Size Manipulation
3.4.1. Pre-Throw and Post-Throw Duration
3.4.2. Explicit Knowledge and Strategies
3.4.3. EDA (Electrodermal), EMG (Electromyography), and Joint Kinematics
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wulf, G.; Lewthwaite, R. Optimizing performance through intrinsic motivation and attention for learning: The OPTIMAL theory of motor learning. Psychon. Bull. Rev. 2016, 23, 1382–1414. [Google Scholar] [CrossRef]
- Ávila, L.T.G.; Chiviacowsky, S.; Wulf, G.; Lewthwaite, R. Positive social-comparative feedback enhances motor learning in children. Psychol. Sport Exerc. 2012, 13, 849–853. [Google Scholar] [CrossRef]
- Chiviacowsky, S.; Drews, R. Temporal-comparative feedback affects motor learning. J. Mot. Learn. Dev. 2016, 4, 208–218. [Google Scholar] [CrossRef]
- Lewthwaite, R.; Wulf, G. Social-comparative feedback affects motor skill learning. Q. J. Exp. Psychol. 2010, 63, 738–749. [Google Scholar] [CrossRef]
- Wulf, G.; Chiviacowsky, S.; Lewthwaite, R. Normative feedback effects on learning a timing task. Res. Q. Exerc. Sport 2010, 81, 425–431. [Google Scholar] [CrossRef]
- Wulf, G.; Chiviacowsky, S.; Lewthwaite, R. Altering mindset can enhance motor learning in older adults. Psychol. Aging 2012, 27, 14–21. [Google Scholar] [CrossRef]
- Abbas, Z.A.; North, J.S. Good-vs. Poor-trial feedback in motor learning: The role of self-efficacy and intrinsic motivation across levels of task difficulty. Learn. Instr. 2018, 55, 105–112. [Google Scholar] [CrossRef]
- Chiviacowsky, S.; Wulf, G. Feedback after good trials enhances learning. Res. Q. Exerc. Sport 2007, 78, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Saemi, E.; Porter, J.M.; Ghotbi-Varzaneh, A.; Zarghami, M.; Maleki, F. Knowledge of Results After Relatively Good Trials Enhances Self-Efficacy And Motor Learning. Psychol. Sport Exerc. 2012, 13, 378–382. [Google Scholar] [CrossRef]
- Chiviacowsky, S.; Wulf, G.; Wally, R.; Borges, T. Knowledge of results after good trials enhances learning in older adults. Res. Q. Exerc. Sport 2009, 80, 663–668. [Google Scholar] [CrossRef]
- Palmer, K.; Chiviacowsky, S.; Wulf, G. Enhanced expectancies facilitate golf putting. Psychol. Sport Exerc. 2016, 22, 229–232. [Google Scholar] [CrossRef]
- Chiviacowsky, S.; Wulf, G.; Lewthwaite, R. Self-controlled learning: The importance of protecting perceptions of competence. Front. Psychol. 2012, 3, 458. [Google Scholar] [CrossRef]
- Trempe, M.; Sabourin, M.; Proteau, L. Success modulates consolidation of a visuomotor adaptation task. J. Exp. Psychol. Learn. Mem. Cogn. 2012, 38, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Masters, R.; Maxwell, J. The theory of reinvestment. Int. Rev. Sport Exerc. Psychol. 2008, 1, 160–183. [Google Scholar] [CrossRef]
- Masters, R.S.W. Knowledge, knerves and know-how: The role of explicit versus implicit knowledge in the breakdown of a complex motor skill under pressure. Br. J. Psychol. 1992, 83, 343–358. [Google Scholar] [CrossRef]
- Ford, P.; Hodges, N.J.; Williams, A.M. Online attentional-focus manipulations in a soccer-dribbling task: Implications for the proceduralization of motor skills. J. Mot. Behav. 2005, 37, 386–394. [Google Scholar] [CrossRef]
- McGaugh, J.L. Memory—A century of consolidation. Science 2000, 287, 248–251. [Google Scholar] [CrossRef] [PubMed]
- Robertson, E.M.; Press, D.Z.; Pascual-Leone, A. Off-line learning and the primary motor cortex. J. Neurosci. 2005, 25, 6372–6378. [Google Scholar] [CrossRef]
- Carter, M.J.; Smith, V.; Ste-Marie, D.M. Judgments of learning are significantly higher following feedback on relatively good versus relatively poor trials despite no actual learning differences. Hum. Mov. Sci. 2016, 45, 63–70. [Google Scholar] [CrossRef]
- Patterson, J.T.; Azizieh, J. Knowing the good from the bad: Does being aware of kr content matter? Hum. Mov. Sci. 2012, 31, 1449–1458. [Google Scholar] [CrossRef]
- Ong, N.T.; Lohse, K.R.; Hodges, N.J. Manipulating target size influences perceptions of success when learning a dart-throwing skill but does not impact retention. Front. Psychol. 2015, 6, 1378. [Google Scholar] [CrossRef]
- Schmidt, R.A.; Zelaznik, H.; Hawkins, B.; Frank, J.S.; Quinn, J.T., Jr. Motor-output variability: A theory for the accuracy of rapid motor acts. Psychol. Rev. 1979, 86, 415–451. [Google Scholar] [CrossRef]
- Holroyd, C.B.; Coles, M.G.H. The neural basis of human error processing: Reinforcement learning, dopamine, and the error-related negativity. Psychol. Rev. 2002, 109, 679–709. [Google Scholar] [CrossRef] [PubMed]
- Schultz, W. Book review: Reward signaling by dopamine neurons. Neuroscientist 2001, 7, 293–302. [Google Scholar] [CrossRef]
- Lohse, K.R.; Sherwood, D.E. Thinking about muscles: The neuromuscular effects of attentional focus on accuracy and fatigue. Acta Psychol. (Amst). 2012, 140, 236–245. [Google Scholar] [CrossRef]
- Brehm, J.; Self, E.A. The intensity of motivation. Annu. Rev. Psychol. 1989, 40, 109–131. [Google Scholar] [CrossRef]
- Kahneman, D. Attention and Effort; Prentice-Hall: Englewood Cliffs, NJ, USA, 1973. [Google Scholar]
- Schmidt, R.A.; Lee, T.D. Motor control and learning: A behavioral emphasis, 5th ed.; Human Kinetics: Champaigne, IL, USA, 2011. [Google Scholar] [CrossRef]
- Hauptmann, B.; Reinhart, E.; Brandt, S.A.; Karni, A. The predictive value of the leveling off of within session performance for procedural memory consolidation. Cogn. Brain Res. 2005, 24, 181–189. [Google Scholar] [CrossRef]
- McAuley, E.; Duncan, T.; Tammen, V.V. Psychometric properties of the intrinsic motivation inventory in a competitive sport setting: A confirmatory factor analysis. Res. Q. Exerc. Sport 1989, 60, 48–58. [Google Scholar] [CrossRef]
- Guay, F.; Vallerand, R.J.; Blanchard, C. On the assessment of situational intrinsic and extrinsic motivation: The situational motivation scale (sims). Motiv. Emot. 2000, 24, 175–213. [Google Scholar] [CrossRef]
- Zimmerman, B.J.; Kitsantas, A. Developmental phases in self-regulation: Shifting from process goals to outcome goals. J. Educ. Psychol. 1997, 89, 29–36. [Google Scholar] [CrossRef]
- De Luca, C.J. The use of surface electromyography in biomechanics. J. Appl. Biomech. 1997, 13, 135–163. [Google Scholar] [CrossRef]
- Ong, N.T.; Hodges, N.J. Balancing our perceptions of the efficacy of success-based feedback manipulations on motor learning. J. Mot. Behav. 2018, 50, 614–630. [Google Scholar] [CrossRef] [PubMed]
- Bandura, A. Self-Efficacy: The Exercise of Control; W.H. Freeman: New York, NY, USA, 1997. [Google Scholar]
- Lewthwaite, R.; Wulf, G. Grand challenge for movement science and sport psychology: Embracing the social-cognitive–affective–motor nature of motor behavior. Front. Psychol. 2010, 1, 42. [Google Scholar] [CrossRef] [PubMed]
- Grand, K.F.; Daou, M.; Lohse, K.R.; Miller, M.W. Investigating the mechanisms underlying the effects of an incidental choice on motor learning. J. Mot. Learn. Dev. 2017, 5, 207–226. [Google Scholar] [CrossRef]
- Levac, D.; Driscoll, K.; Galvez, J.; Mercado, K.; O’Neil, L. OPTIMAL Practice Conditions Enhance The Benefits Of Gradually Increasing Error Opportunities On Retention Of A Stepping Sequence Task. Hum. Mov. Sci. 2017, 56, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Daou, M.; Lohse, K.R.; Miller, M.W. Expecting to teach enhances motor learning and information processing during practice. Hum. Mov. Sci. 2016, 49, 336–345. [Google Scholar] [CrossRef]
- Wilson, M.; Smith, N.C.; Holmes, P.S. The role of effort in influencing the effect of anxiety on performance: Testing the conflicting predictions of processing efficiency theory and the conscious processing hypothesis. Br. J. Psychol. 2007, 98, 411–428. [Google Scholar] [CrossRef]
- Fitts, P.M.; Peterson, J.R. Information capacity of discrete motor responses. J. Exp. Psychol. 1964, 67, 103–112. [Google Scholar] [CrossRef]
- Powers, W.T. Commentary on bandura’s “human agency.”. Am. Psychol. 1991, 46, 151–153. [Google Scholar] [CrossRef]
- Ilgen, D.; Davis, C. Bearing bad news: Reactions to negative performance feedback. Appl. Psychol. 2000, 49, 550–565. [Google Scholar] [CrossRef]
- Yeo, G.; Neal, A. Subjective Cognitive Effort: A Model of States, Traits, And Time. J. Appl. Psychol. 2008, 93, 617–631. [Google Scholar] [CrossRef] [PubMed]
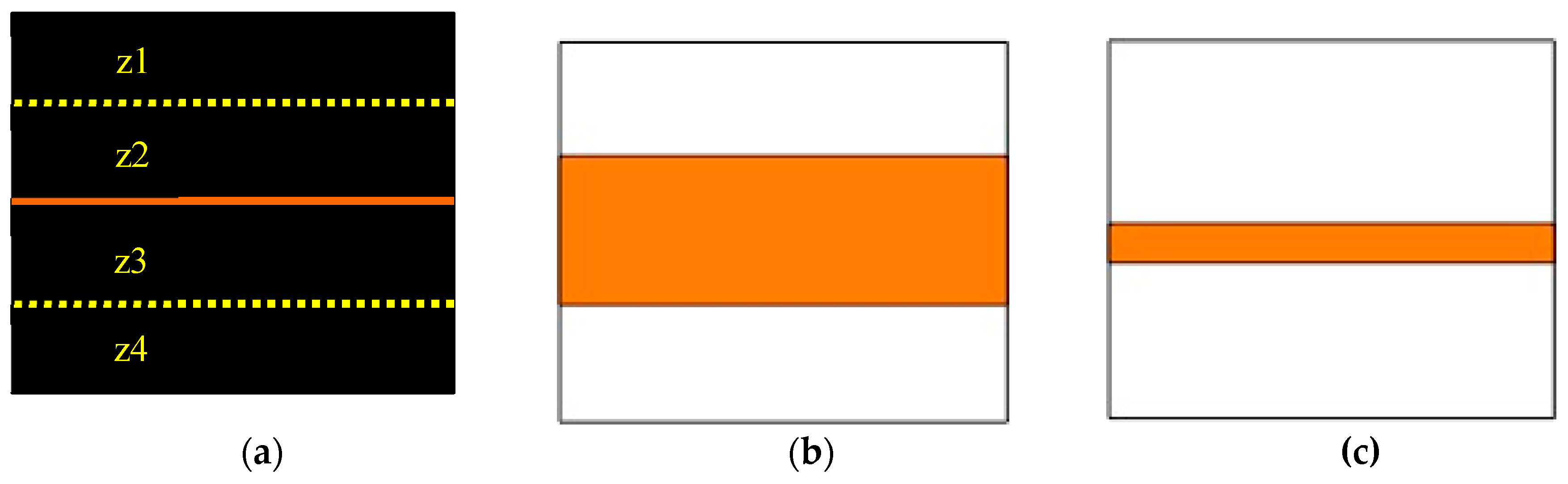
| Task and Procedures (# Trials) Day 1 (# Trials) | Day 2 (# Trials) |
|---|---|
| 1. Familiarisation (1) | 1. Familiarisation (1) |
| 2. No vision pre-test (6) | 2. No vision retention (6) |
| 3. Acquisition (90) | 3. Vision retention (9) |
| 4. No vision plus 5. Prediction (9) | |
| 6. Secondary task (9) | |
| 7. Slowed + speeded (9 + 9) | |
| Dependent Measures | |
| Behavioural: | |
| 1. # Target hits and outcome error (absolute error, constant error, variable error) | |
| 2. Secondary tone-counting response | |
| 3. Outcome-prediction response | |
| Questionnaire: | |
| 1. Self-efficacy rating: 3 × acquisition | |
| 2. Outcome expectation: 3 × acquisition | |
| 3. IMI (competency): Pre and post Day 1, Pre Day 2 | |
| 4. IMI (interest/enjoyment): Post-Day 1 | |
| 5. SMS: Post Day 1 | |
| 6. Success perception check: Post Day 1 | |
| 7. Custom motivation items: Post Day 1 | |
| 8. Explicit knowledge: Post Day 2 | |
| Process: | |
| 1. EMG (electromyography), EDA (electrodermal), kinematics | |
| 2. Trial or movement durations |
| Measure | Phase/Condition | Mean (SD) | Mean (SD) | Mean (SD) |
|---|---|---|---|---|
| Target hits (%) | Acq | Large = 44.1 (7.8) | Small = 11.3 (3.6) | |
| B1 = 25.5 (16.9) | B2 = 27.0 (17.7) | B3 = 28.7 (21.1) | ||
| Self-efficacy probe (%) | Acq | Large = 63.33 (13.22) | Small = 29.78 (19.59) | |
| B1 = 32.32 (22.09) | B2 = 49.29 (26.76) | B3 = 55.36 (28.89) | ||
| Acq (Large) | B1 = 45.8 (19.8) | B2 = 70.8 (13.8) | B3 = 75.4 (16.6) | |
| Acq (Small) | B1 = 20.7 (17.1) | B2 = 30.7 (20.4) | B3 = 38.0 (26.0) | |
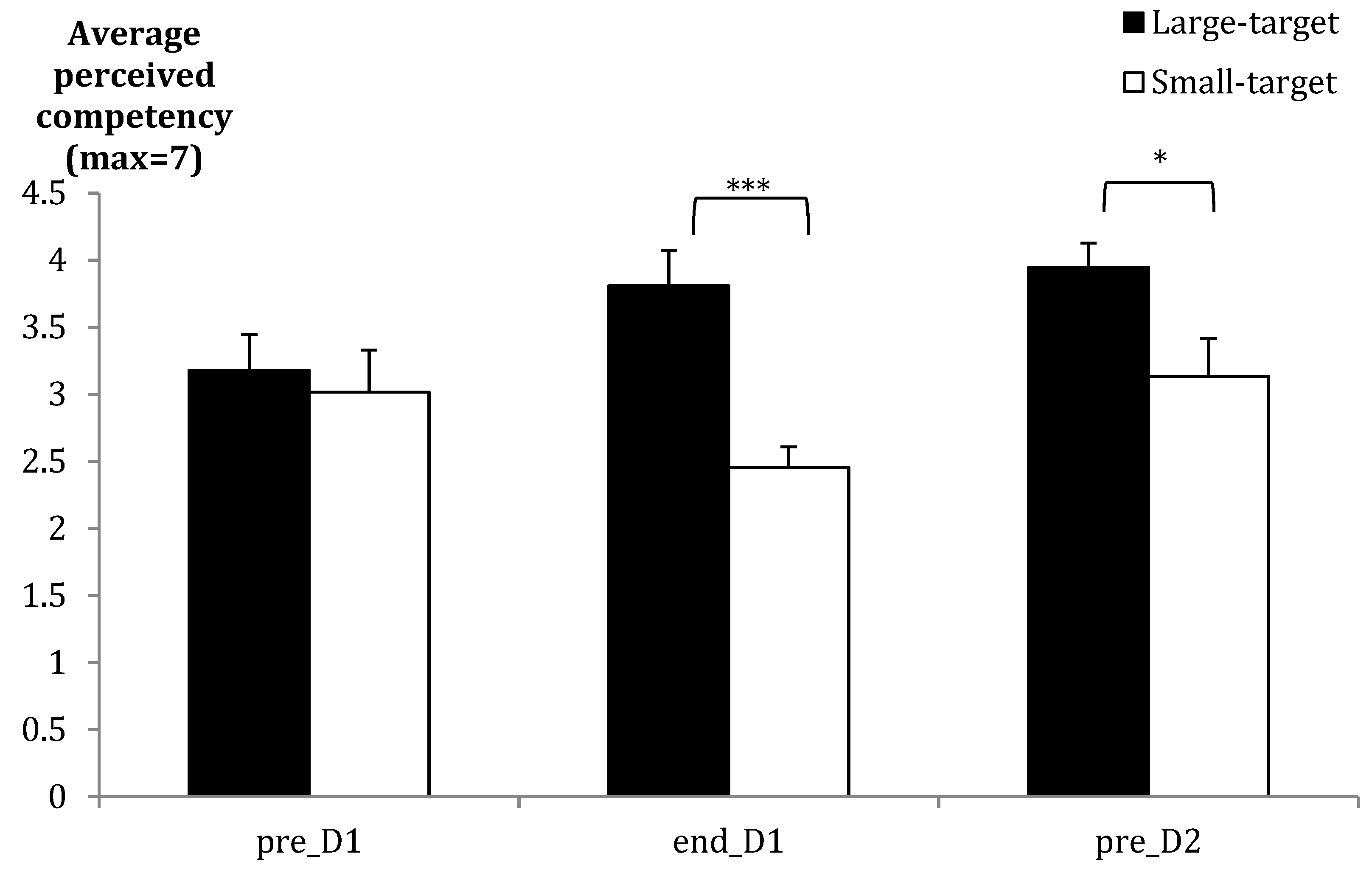
| Perceived competency (1–7 scale) | Start Acq | Large = 3.2 (1.0) (mdn = 3.1) | Small = 3.0 (1.2) (mdn = 3.3) | |
| End Acq | Large = 3.8 (1.0) (mdn = 3.8) | Small = 2.5 (0.6) (mdn = 2.2) | ||
| Ret | Large = 3.9 (0.7) (mdn = 4.0) | Small = 3.1 (1.1) (mdn = 3.0) | ||
| Perceived success (1–7 scale) | End Acq | Large = 4.6 (0.8) (mdn = 5) | Small = 3.5 (1.1) (mdn = 3) | |
| Interest/enjoyment (1–7 scale) | End Acq | Large = 5.6 (0.6) (mdn = 5.7) | Small = 5.7 (0.6) (mdn = 5.9) | |
| Intrinsic motivation (1–7 scale) | End Acq | Large = 5.6 (0.6) (mdn = 5.5) | Small = 5.5 (0.7) (mdn = 5.8) | |
| Customised motivation (further practice), 1–7 scale | End Acq | Large = 5.5 (1.3) (mdn = 6.0) | Small = 5.5 (1.0) (mdn = 5.0) | |
| Customised motivation (do well in practice), 1–7 scale | End Acq | Large = 5.6 (1.1) (mdn = 6.0) | Small = 6.3 (0.8) (mdn = 6.0) | |
| Amotivation (1–7 scale) | End Acq | Large = 2.6 (0.6) (mdn = 2.5) | Small = 2.4 (0.9) (mdn = 2.3) | |
| #Rules or strategies accumulated | End Ret | Large = 1.9 (1.1) (mdn = 2.0) | Small = 2.3 (1.1) (mdn = 2.0) | |
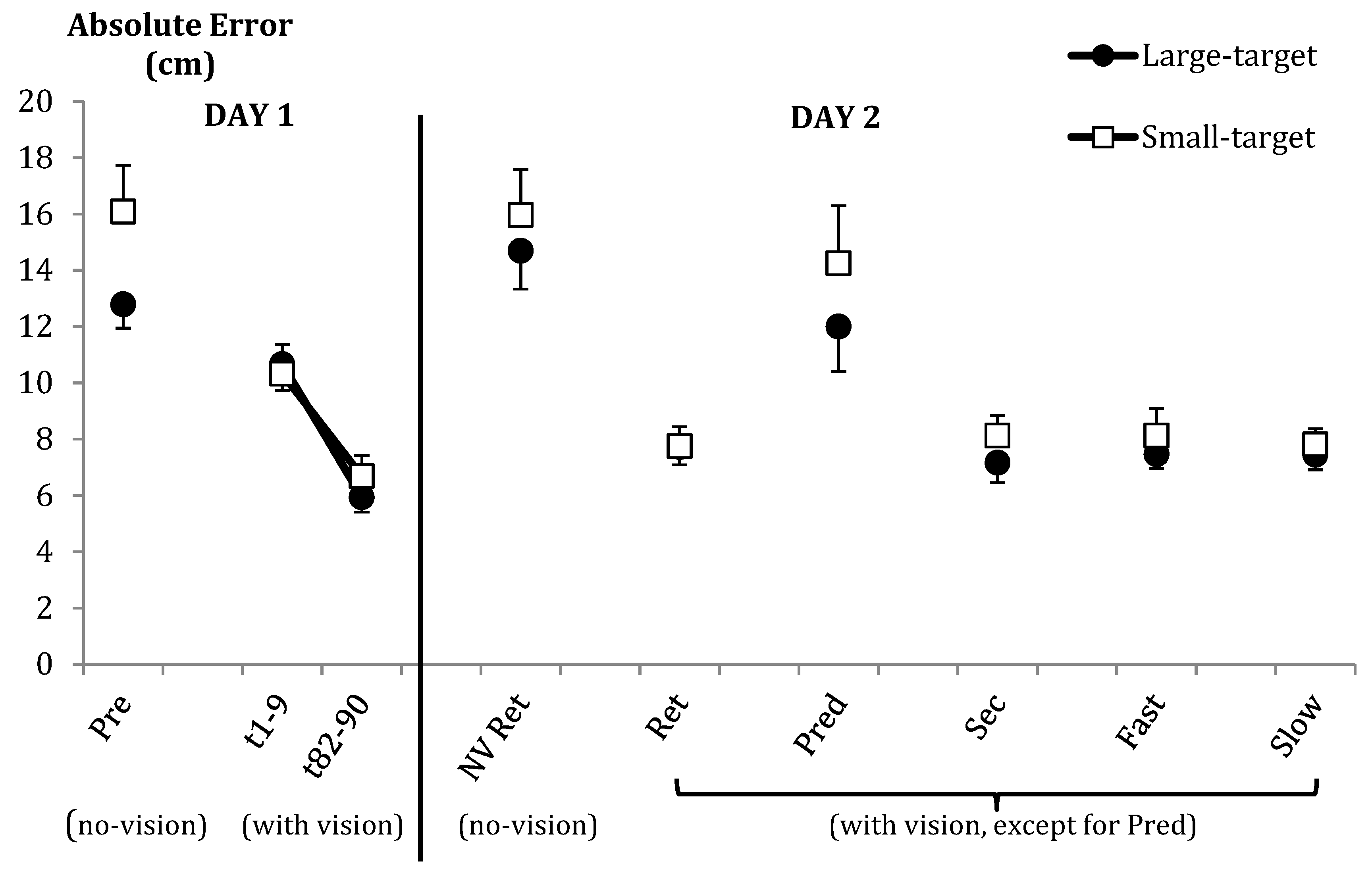
| Outcome error: Absolute error (cm) | Pre | Large = 12.79 (3.16) | Small = 16.08 (6.39) | |
| Acq | Large = 8.30 (3.68) | Small = 8.51 (3.88) | ||
| B1 = 10.49 (3.71) | B10 = 6.33 (2.44) | |||
| Pre vs. Ret | Large = 13.74 (4.26) | Small = 16.02 (6.22) | ||
| Pre = 14.49 (5.28) | Ret = 15.35 (5.66) | |||
| Ret V vs. NV | Large = 11.20 (5.27) | Small = 11.85 (6.31) | ||
| Vis = 7.73 (2.49) | NV = 15.35 (5.66) | |||
| Ret (V) | Large = 7.72 (2.37) | Small = 7.74 (2.68) | ||
| Prediction | Large = 11.99 (5.98) | Small = 14.25 (7.94) | ||
| Secondary | Large = 7.16 (2.67) | Small = 8.12 (2.77) | ||
| Fast | Large = 7.46 (1.89) | Small = 8.13 (3.72) | ||
| Slow | Large = 7.44 (1.98) | Small = 7.81 (2.15) | ||
| Constant error (cm) | Pre | Large = –10.08 (6.55) | Small = –15.04 (7.41) | |
| Acq | Large = –4.64 (4.29) | Small = –5.38 (5.13) | ||
| B1 = –7.63 (4.84) | B10 = –2.41 (2.77) | |||
| Pre vs. Ret | Large = –11.48 (6.51) Pre = –12.64 (7.33) | Small = –14.64 (7.37) Ret = –13.59 (6.94) | ||
| Ret V vs. NV | Large = –8.34 (6.68) Vis = –4.68 (3.00) | Small = –9.87 (7.23) NV = –13.59 (6.94) | ||
| Ret (V) | Large = –3.81 (2.72) | Small = –5.49 (3.12) | ||
| Prediction | Large = –9.58 (8.52) | Small = –11.79 (10.45) | ||
| Secondary | Large = –2.56 (3.94) | Small = –4.91 (3.89) | ||
| Fast | Large = –1.02 (4.30) | Small = –3.80 (3.04) | ||
| Slow | Large = –4.37 (2.90) | Small = –3.86 (2.95) | ||
| Variable error (cm) | Pre | Large = 9.05 (3.30) | Small = 9.30 (3.24) | |
| Acq | Large = 8.61 (3.08) | Small = 8.38 (3.04) | ||
| B1 = 10.14 (2.34) | B10 = 6.84 (2.77) | |||
| Pre vs. Ret | Large = 9.34 (3.63) | Small = 10.03 (3.51) | ||
| Pre = 9.18 (3.21) | Ret = 10.21 (3.85) | |||
| Ret V vs. NV | Large = 9.01 (3.42) | Small = 9.41 (3.34) | ||
| Vis = 8.22 (2.45) | NV = 10.21 (3.85) | |||
| Ret (V) | Large = 8.39 (2.66) | Small = 8.07 (2.31) | ||
| Prediction | Large = 8.13 (3.39) | Small = 9.35 (4.06) | ||
| Secondary | Large = 8.07 (2.72) | Small = 7.78 (2.64) | ||
| Fast | Large = 7.92 (2.56) | Small = 8.40 (3.42) | ||
| Slow | Large = 7.58 (2.11) | Small = 8.00 (1.95) | ||
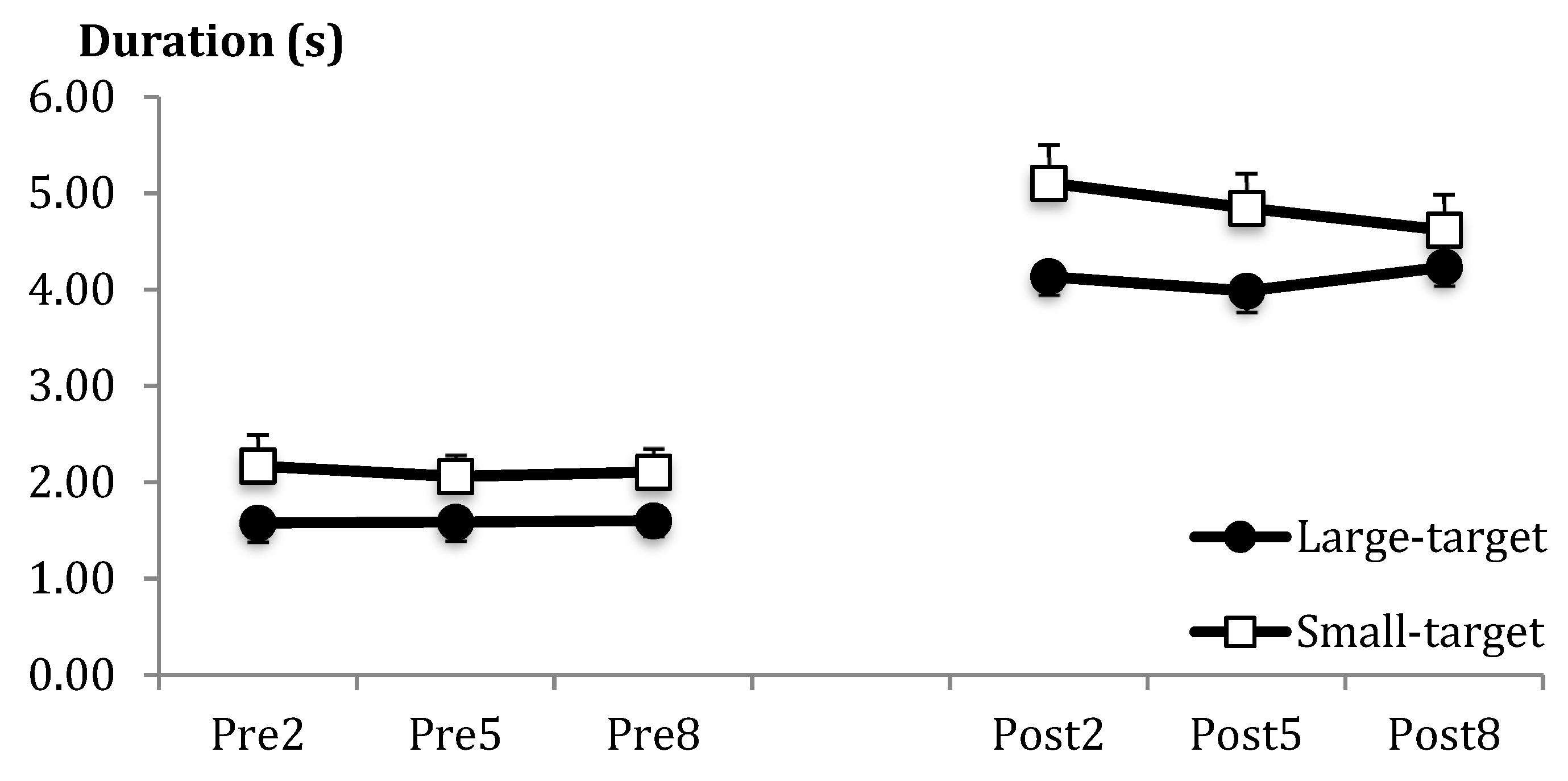
| Processing duration (s) | Acq | Large = 2.85 (1.43) | Small = 3.48 (1.82) | |
| B2 = 3.28 (1.79) | B5 = 3.14 (1.64) | B8 = 3.16 (1.61) | ||
| PreT = 1.87 (0.86) | PostT = 4.51 (1.16) | |||
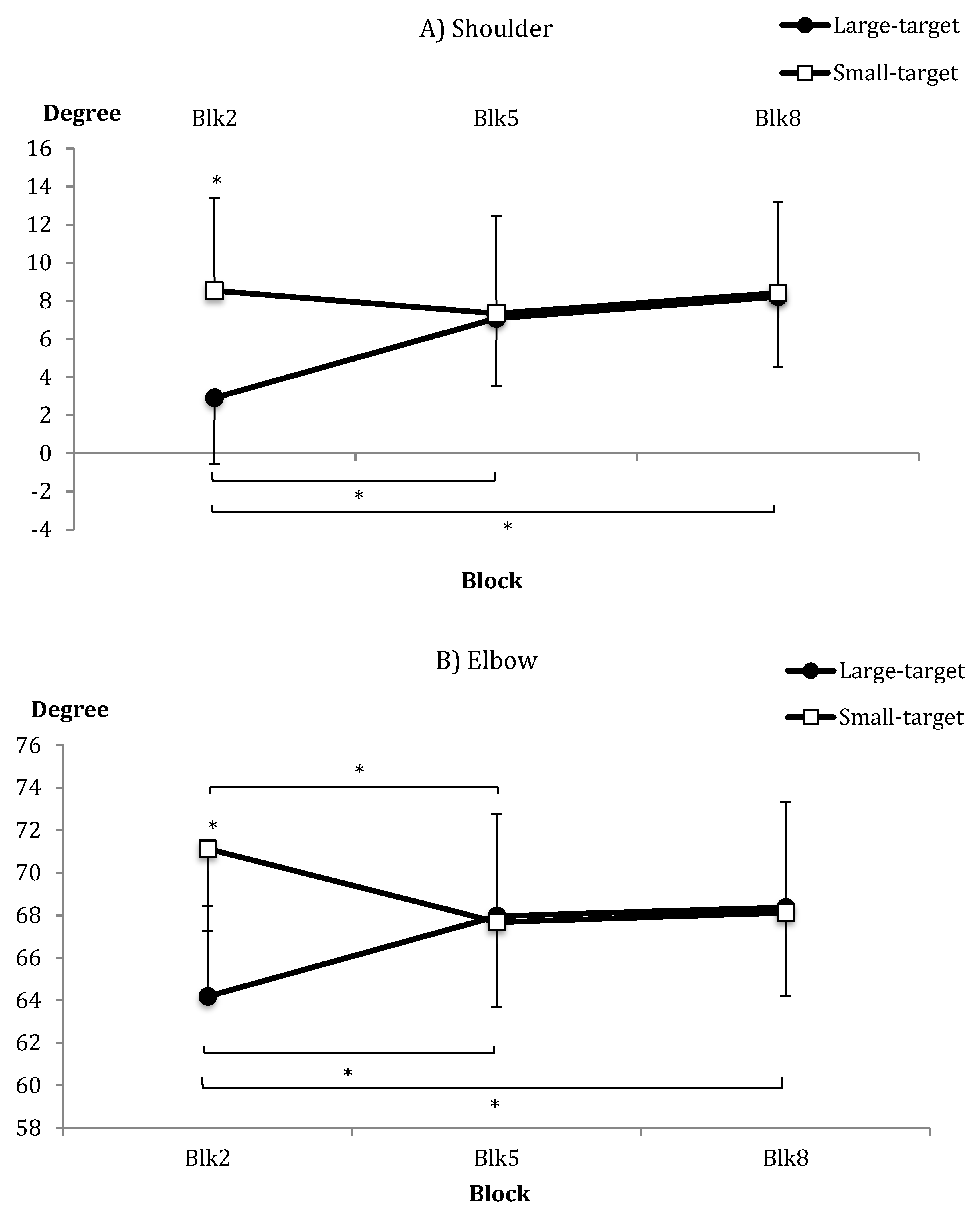
| Joint amplitude | Acq | Large = 6.07 (12.19) | Small = 8.09 (18.04) | |
| (shoulder) (°) | B2 = 5.94 (15.63) | B5 = 7.22 (16.05) | B8 = 8.32 (15.48) | |
| Acq/B2 | Large = 2.91 (11.94) | Small = 8.53 (18.26) | ||
| Joint amplitude | Acq | Large = 66.84 (15.87) | Small = 68.98 (14.38) | |
| (elbow) (°) | B2 = 67.92 (14.70) | B5 = 67.81 (15.44) | B8 = 68.24 (15.52) | |
| Acq/B2 | Large = 64.19 (14.69) | Small = 71.13 (14.46) | ||
| SD joint amplitude | Acq | Large = 3.60 (1.93) | Small = 4.23 (2.43) | |
| (shoulder) (°) | B2 = 4.24 (2.53) | B5 = 3.76 (1.90) | B8 = 3.82 (2.25) | |
| Acq/B2 | Large = 3.37 (1.91) | Small = 4.98 (2.81) | ||
| Acq/B8 | Large = 4.12 (2.57) | Small = 3.56 (2.00) | ||
| SD joint amplitude | Acq | Large = 7.80 (2.78) | Small = 7.16 (2.69) | |
| (elbow) (°) | B2 = 7.86 (2.67) | B5 = 7.65 (2.94) | B8 = 6.85 (2.58) | |
| Movement time (ms) | Acq | Large = 199.1 (93.2) | Small = 186.5 (50.0) | |
| B2 = 188.0 (64.6) | B5 = 192.5 (63.7) | B8 = 196.6 (90.1) | ||
| SD movement time | Acq | Large = 43.7 (72.8) | Small = 37.7 (33.2) | |
| (ms) | B2 = 37.7 (38.0) | B5 = 45.2 (71.8) | B8 = 38.5 (51.2) | |
| Angular velocity (°/s) | Acq | Large = 366.8 (130.1) | Small = 371.4 (93.9) | |
| B2 = 371.2 (112.5) | B5 = 365.0 (107.0) | B8 = 371.0 (118.6) | ||
| SD angular velocity | Acq | Large = 54.6 (25.9) | Small = 60.9 (28.0) | |
| (°/s) | B2 = 62.7 (25.7) | B5 = 56.0 (32.4) | B8 = 55.2 (22.5) | |
| Electrodermal activity * | Acq | Large = 6.30 (1.93) | Small = 5.09 (2.10) | |
| (μS) | B2 = 5.63 (2.00) | B5 = 5.55 (1.96) | B8 = 5.75 (2.37) | |
| PreT = 5.54 (2.09) | PostT= 5.75 (2.13) | |||
| Electromyography co-contraction (ratio) | Acq | Large = 1.62 (.88) B2 = 1.77 (.76) | Small = 1.92 (.72) B5 = 1.83 (.83) | B8 = 1.72 (.87) |
| Measures | Effects | Test Statistic | p-Value | Effect Size |
|---|---|---|---|---|
| Target Hits: | ||||
| Acq | Group | F(1,27) = 218.02 | *** | η2p = 0.89 |
| Mega-blk | F(2,54) = 1.61 | 0.21 | η2p = 0.06 | |
| Group × Mega-block | F(2,54) = 2.33 | 0.11 | η2p = 0.08 | |
| Self-Efficacy Probes: | ||||
| Acq | Group | F(1,26) = 27.98 | *** | η2p = 0.52 |
| Mega-blk | F(2,52) = 30.90 | *** | η2p = 0.54 | |
| Group × Mega-blk | F(2,52) = 3.32 | * | η2p = 0.11 | |
| Outcome Expectation Error: | ||||
| Mega-Blk 1 | Group | Fisher’s exact test | * | φ = 0.55 |
| Perceived Competency: | ||||
| End Acq | Group | U = 22.50 | *** | r = 0.67 |
| Ret | Group | U = 52.00 | * | r = 0.43 |
| Perceived Success: | ||||
| End Acq | Group | U = 43.50 | ** | r = 0.53 |
| Customised Motivation (Do Well in Practice): | ||||
| End Acq | Group | U = 66.50 | 0.09 | r = 0.33 |
| Absolute Error (AE): | ||||
| Pre | Group | t (20.8) = 1.78 | 0.09 | d = 0.65 |
| Acq | Blk | F (1,27) = 47.26 | *** | η2p = 0.64 |
| Ret | Vision | F (1,27) = 58.19 | *** | η2p = 0.68 |
| Pre vs. NV Ret | Group | F (1,27) = 2.27 | 0.14 | η2p = 0.08 |
| Constant Error (CE): | ||||
| Pre | Group | t (27) = 1.90 | 0.07 | d = 0.71 |
| Pre vs. NV Ret | Group | F(1,27) = 2.66 | 0.12 | η2p = 0.09 |
| Acq Blk1 | Group | t (27) = 1.11 | 0.28 | d = 0.41 |
| Acq | Blk | F(1,27) = 44.79 | *** | η2p = 0.62 |
| Group × Blk | F(1,27) = 2.60 | 0.12 | η2p = 0.09 | |
| Ret | Vision | F (1,27) = 57.10 | *** | η2p = 0.68 |
| Sec | Group | t (27) = 1.61 | 0.12 | d = 0.60 |
| Fast | Group | t (27) = 2.03 | 0.05 | d = 0.75 |
| Variable Error (VE): | ||||
| Acq Blk1 | Group | t (27) = 1.53 | 0.14 | d = 0.57 |
| Acq | Blk | F (1,27) = 38.07 | *** | η2p = 0.59 |
| Group × Blk | F (1,27) = 3.96 | 0.06 | η2p = 0.13 | |
| Ret | Vision | F (1,27) = 5.19 | * | η2p = 0.16 |
| Processing Duration: | ||||
| Acq | Group | F(1,24) = 4.83 | * | η2p = 0.17 |
| Throw Period | F(1,24) = 126.09 | *** | η2p = 0.84 | |
| Group × Blk | F(2,48) = 2.15 | 0.13 | η2p = 0.08 | |
| Group × Blk × Period | F(2,48) = 2.02 | 0.14 | η2p = 0.08 | |
| Joint Amplitudes: | ||||
| Shoulder | Blk | F(2,48) = 4.29 | * | η2p = 0.15 |
| Group × Blk | F(2,48) = 6.14 | ** | η2p = 0.20 | |
| Elbow | Group × Blk | F(2,48) = 6.94 | ** | η2p = 0.22 |
| Variability (SD) of Joint Amplitudes: | ||||
| Shoulder | Group × Blk | F(2,48) = 4.43 | * | η2p = 0.16 |
| Elbow | Blk | F(2,48) = 1.54 | 0.23 | η2p = 0.06 |
| Electrodermal Activity (EDA): | ||||
| Acq | Period | F(1,24) = 15.24 | ** | η2p = 0.39 |
| Acq | Group × Period | F(1,24) = 3.46 | 0.08 | η2p = 0.13 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ong, N.T.; Hawke, J.; Hodges, N.J. Target Size Manipulations Affect Error-Processing Duration and Success Perceptions but not Behavioural Indices of Learning. Brain Sci. 2019, 9, 119. https://doi.org/10.3390/brainsci9050119
Ong NT, Hawke J, Hodges NJ. Target Size Manipulations Affect Error-Processing Duration and Success Perceptions but not Behavioural Indices of Learning. Brain Sciences. 2019; 9(5):119. https://doi.org/10.3390/brainsci9050119
Chicago/Turabian StyleOng, Nicole T., Jamie Hawke, and Nicola J. Hodges. 2019. "Target Size Manipulations Affect Error-Processing Duration and Success Perceptions but not Behavioural Indices of Learning" Brain Sciences 9, no. 5: 119. https://doi.org/10.3390/brainsci9050119
APA StyleOng, N. T., Hawke, J., & Hodges, N. J. (2019). Target Size Manipulations Affect Error-Processing Duration and Success Perceptions but not Behavioural Indices of Learning. Brain Sciences, 9(5), 119. https://doi.org/10.3390/brainsci9050119





