Superior Effects of Modified Chen-Style Tai Chi versus 24-Style Tai Chi on Cognitive Function, Fitness, and Balance Performance in Adults over 55
Abstract
1. Introduction
2. Methods
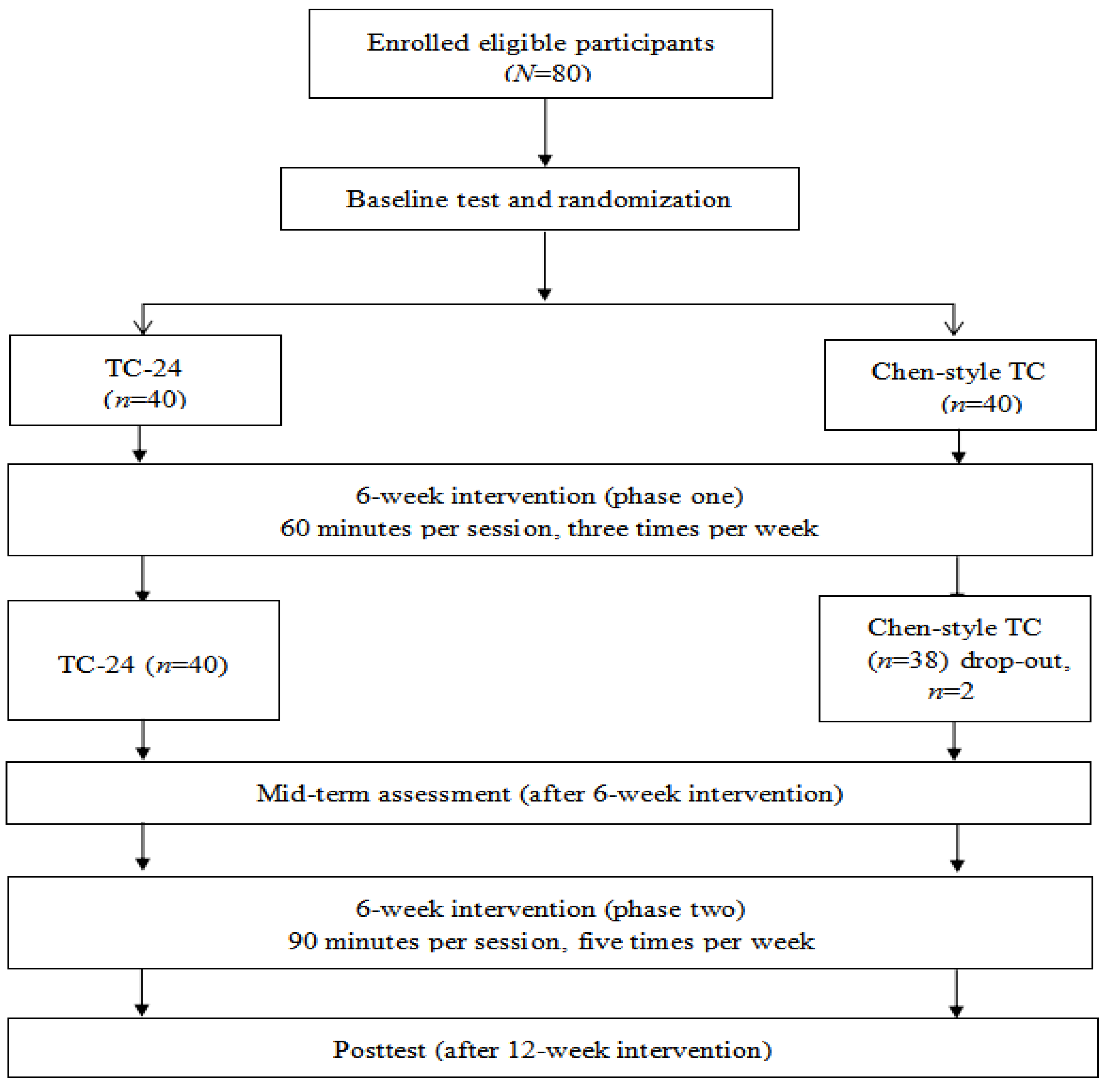
2.1. Study Participants
2.2. Intervention
2.3. Outcome Measures
2.3.1. Demographic Information
2.3.2. Cognitive Function
2.3.3. Balance
2.3.4. Functional Fitness of Lower Limbs
2.3.5. Aerobic Exercise Capacity
2.4. Statistical Analysis
3. Results
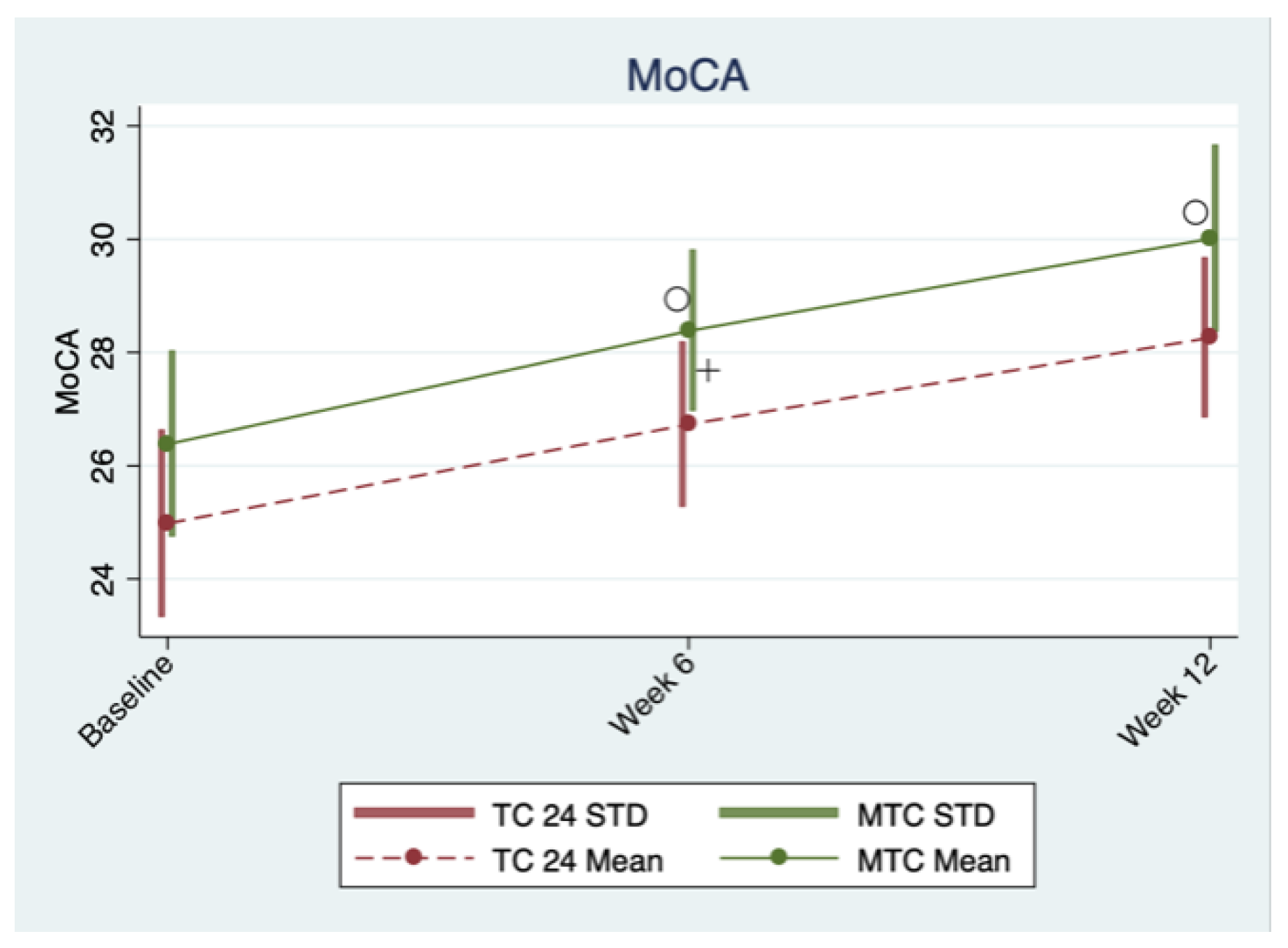
3.1. Cognitive Function
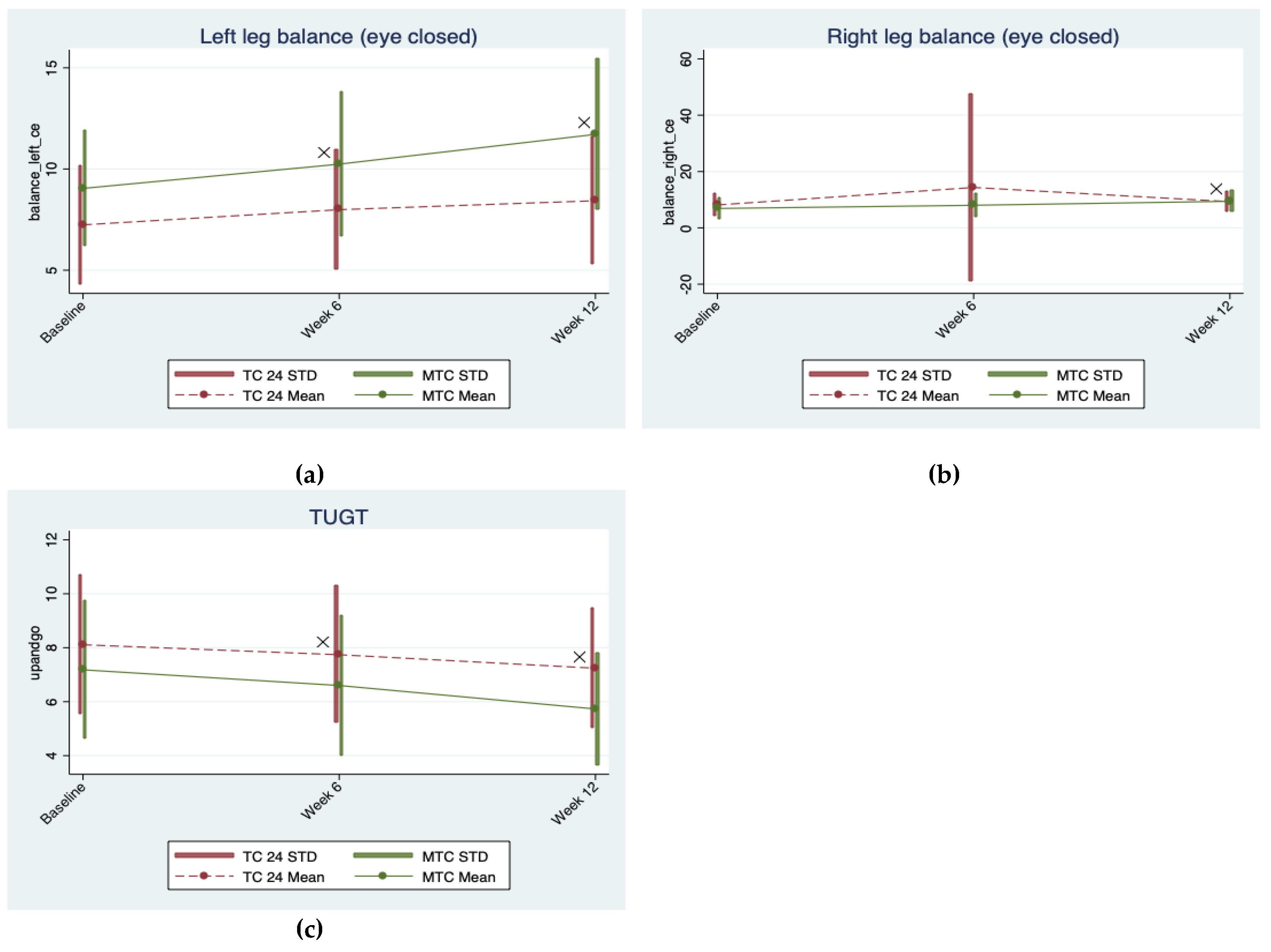
3.2. Balance Performance
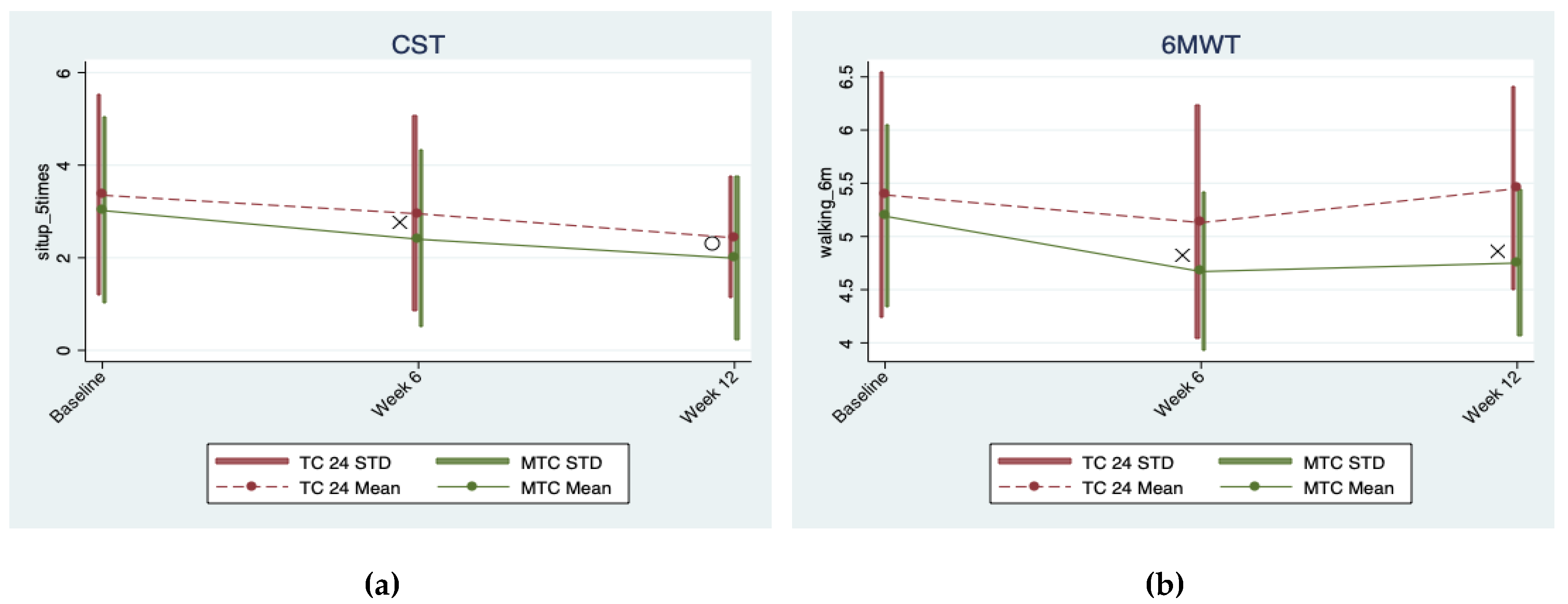
3.3. Aerobic Exercise Capacity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- The World Factbook. China Demographics Profile 2018. Available online: https://www.indexmundi.com/china/demographics_profile.html (accessed on 10 April 2019).
- Chang, Y.K.; Nien, Y.H.; Tsai, C.L.; Etnier, J.L. Physical activity and cognition in older adults: The potential of Tai Chi Chuan. J. Aging Phys. Act. 2010, 18, 451–472. [Google Scholar] [CrossRef] [PubMed]
- Ian, J.D.; Janie, C.; Alan, J.; Gow, S.E.; Harris, L.M.; Houlihan, R.E.; Marioni, L.P.; Snorri, B.R.; John, M.S. Age-associated cognitive decline. Brit. Medic. Bull. 2009, 1, 135–152. [Google Scholar] [CrossRef]
- Fissler, P.; Küster, O.; Schlee, W.; Kolassa, I. Novelty Intervention to enhance broad cognitive abilities and prevent dementia: Synergistic approaches for the facilitation of positive plastic change. Prog. Brain Res. 2013, 207, 403–434. [Google Scholar]
- Erickson, K.L.; Kramer, A.F. Aerobic exercise effects on cognitive and neural plasticity in older adults. Br. J. Sports Med. 2009, 43, 22–24. [Google Scholar] [CrossRef]
- Wayne, P.M.; Yeh, G.; Mehta, D.H. Minding the Mind–Body Literature: Aging and Cognitive Decline. J. Altern. Complement. Med. 2018, 24, 196–199. [Google Scholar] [CrossRef]
- Petersen, R.C.; Lopez, O.; Armstrong, M.J.; Getchius, T.D.; Ganguli, M.; Gloss, D. Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology. Neurology 2018, 91, 373–374. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Loprinzi, P.D.; Yeung, A.S.; Zeng, N.; Huang, T. The beneficial effects of mind-body exercises for people with mild cognitive impairment: A systematic review with meta-analysis. Arch. Phys. Med. Rehabil. 2019. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, C.; Zou, L.; Liu, X.; Song, W. The Effects of Mind-Body Exercise on Cognitive Performance in Elderly: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2018, 15, 2791. [Google Scholar] [CrossRef]
- Zou, L.; SasaKi, J.; Wang, H.; Xiao, Z.; Fang, Q.; Zhang, M. A Systematic Review and Meta—Analysis Baduanjin Qigong for Health Benefits: Randomized Controlled Trials. Evid. Based Complement. Altern. Med. 2017, 1, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Wang, C.; Yeung, A.; Liu, Y.; Pan, Z. A Review Study on the beneficial effects of Baduanjin. J. Altern. Complement. Med. 2018, 24, 324–335. [Google Scholar] [CrossRef]
- Zhou, S.; Zhang, Y.; Kong, Z.; Loprinzi, P.D.; Hu, Y.; Ye, J.; Liu, S.; Yu, J.J.; Zou, L. The Effects of Tai Chi on Markers of Atherosclerosis, Lower-limb Physical Function, and Cognitive Ability in Adults Aged Over 60: A Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2019, 16, 753. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Wang, H.; Xiao, Z.; Fang, Q.; Zhang, M.; Li, T.; Du, G.; Liu, Y. Tai chi for health benefits in patients with multiple sclerosis: A systematic review. PLoS ONE 2017, 12, e0170212. [Google Scholar] [CrossRef]
- Kong, Z.; Sze, T.-M.; Yu, J.J.; Loprinzi, P.D.; Xiao, T.; Yeung, A.S.; Li, C.; Zhang, H.; Zou, L. Tai Chi as an Alternative Exercise to Improve Physical Fitness for Children and Adolescents with Intellectual Disability. Int. J. Environ. Res. Public Health 2019, 16, 1152. [Google Scholar] [CrossRef]
- Zou, L.; Wang, C.; Tian, Z.; Wang, H.; Shu, Y. Effect of Yang-Style Tai Chi on Gait Parameters and Musculoskeletal Flexibility in Healthy Chinese Older Women. Sports 2017, 5, 52. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Wang, H.; Yu, D. Effect of a long-term modified Tai Chi-based intervention in attenuating bone mineral density in postmenopausal women in southeast China: Study protocol for arandomized controlled trial. Clin. Trials Degener. Dis. 2017, 2, 46–52. [Google Scholar] [CrossRef]
- Zou, L.; Wang, C.; Chen, K.; Shu, Y.; Chen, X.; Luo, L.; Zhao, X. The Effect of Taichi Practice on Attenuating Bone Mineral Density Loss: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Int. J. Environ. Res. Public Health 2017, 14, 1000. [Google Scholar] [CrossRef]
- Zou, L.; Sasaki, J.; Zeng, N.; Wang, C.; Sun, L.A. Systematic Review with Meta-Analysis of Mindful Exercises on Rehabilitative Outcomes among post-stroke patients. Arch. Phys. Med. Rehabil. 2018, 9, 2355–2364. [Google Scholar] [CrossRef]
- Zou, L.; Huang, T.; Tsang, T.; Pan, Z.; Wang, C. Hard Martial Arts for Cognitive Function across the Lifespan: A Systematic Review. Arch. Budo 2018, 14, 41–58. [Google Scholar]
- Zhang, Y.; Loprinzi, P.D.; Yang, L.; Liu, J.; Liu, S.; Zou, L. The Beneficial Effects of Traditional Chinese Exercises for Adults with Low Back Pain: A Meta-Analysis of Randomized Controlled Trials. Medicina 2019, 55, 118. [Google Scholar] [CrossRef]
- Zou, L.; Han, J.; Tsang, W.; Yeung, A. Effects of Tai Chi on lower limb proprioception in adults aged over 55: A systematic review ad meta-analysis. Arch. Phys. Med. Rehabil. 2018, in press. [Google Scholar] [CrossRef]
- Wayne, P.M.; Walsh, J.N.; Wells, R.E.; Papp, K.V.; Donovan, N.J.; Yeh, G.Y.; Taylor-Piliae, R.E.; Taylor-Piliae, R.E. The Impact of Tai Chi on Cognitive Performance in Older Adults: A Systematic Review and Meta-Analysis. J. Am. Geriatr. Soc. 2014, 62, 25–39. [Google Scholar] [CrossRef]
- Wayne, P.M.; Gagnon, M.M.; Macklin, E.A.; Travison, T.G.; Manor, B.; Lachman, M.; Thomas, C.P.; Lipsitz, L.A. The Mind Body-Wellness in Supportive Housing (Mi-WiSH) Study: Design and Rationale of a Cluster Randomized Controlled Trial of Tai Chi in Senior Housing. Contemp. Clin. Trials 2017, 60, 96–104. [Google Scholar] [CrossRef]
- Wayne, P.M.; Kaptchuk, T.J. Challenges inherent to Tai Chi research: Part I—Tai Chi as a complex multicomponent intervention. J. Altern. Complement. Med. 2008, 14, 95–102. [Google Scholar] [CrossRef]
- Mortimer, J.A.; Ding, D.; Borenstein, A.R.; DeCarli, C.; Guo, Q.H.; Wu, Y.G.; Zhao, Q.H.; Chu, S.G. Changes in brain volume and cognition in a randomized trial of exercise and social interaction in a community-based sample of non-demented Chinese elders. Alzheihmer Dis. 2012, 30, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Hui, S.S.C.; Woo, J.; Kwok, T. Evaluation of energy expenditure and cardiovascular health effects from Tai Chi and walking exercise. Hong Kong Med. J. 2009, 15, 4–7. [Google Scholar]
- Hui, S.S.-C.; Xie, Y.J.; Woo, J.; Kwok, T.C.-Y. Effects of Tai Chi and Walking Exercises on Weight Loss, Metabolic Syndrome Parameters, and Bone Mineral Density: A Cluster Randomized Controlled Trial. Evid.-Based Complement. Altern. Med. 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Goto, S.; Sasaki, A.; Takahashi, I.; Mitsuhashi, Y.; Nakaji, S.; Matsubara, A. Relationship between cognitive function and balance in a community-dwelling population in Japan. Acta Otolaryngol. 2018, 138, 471–474. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Yeung, A.; Zeng, N.; Wang, C.; Sun, L.; Thomas, G.; Wang, H. Effects of Mind-Body Exercises for Mood and Functional Capabilities in Post-Stroke Patients: An Analytical Review of Randomized Controlled Trials. Int. J. Environ. Res. Public Health 2018, 15, 721. [Google Scholar] [CrossRef]
- Wetherell, J.L.; Hershey, T.; Hickman, S.; Tate, S.R.; Dixon, D.; Bower, E.S.; Lenze, E.J. Mindfulness-Based Stress Reduction for Older Adults With Stress Disorders and Neurocognitive Difficulties: A Randomized Controlled Trial. J. Clin. Psychiatry 2017, 78, e734–e743. [Google Scholar] [CrossRef]
- Zou, L.; Yeung, A.; Li, C.; Wei, G.-X.; Chen, K.W.; Kinser, P.A.; Chan, J.S.M.; Ren, Z. Effects of Meditative Movements on Major Depressive Disorder: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Clin. Med. 2018, 7, 195. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Yeung, A.; Quan, X.; Hui, S.S.-C.; Hu, X.; Chan, J.S.M.; Wang, C.; Boyden, S.D.; Sun, L.; Wang, H. Mindfulness-Based Baduanjin Exercise for Depression and Anxiety in People with Physical or Mental Illnesses: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2018, 15, 321. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Sasaki, J.E.; Wei, G.X.; Huang, T.; Yeung, A.S.; Neto, O.B.; Chen, K.W.; Hui, S.C. Effects of Mind–Body Exercises (Tai Chi/Yoga) on Heart Rate Variability Parameters and Perceived Stress: A Systematic Review with Meta-Analysis of Randomized Controlled Trials. J. Clin. Med. 2018, 7, 404. [Google Scholar] [CrossRef] [PubMed]
- Weuve, J.; Barnes, L.L.; De Leon, C.F.M.; Rajan, K.B.; Beck, T.; Aggarwal, N.T.; Hebert, L.E.; Bennett, D.A.; Wilson, R.S.; Evans, D.A. Cognitive aging in black and white Americans: Cognition, cognitive decline, and incidence of Alzheimer disease dementia. Epidemiology 2018, 29, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Yeung, A.; Li, C.; Chiou, S.-Y.; Zeng, N.; Tzeng, H.-M.; Wang, L.; Ren, Z.; Dean, T.; Thomas, G.A. Effects of Mind–Body Movements on Balance Function in Stroke Survivors: A Meta-Analysis of Randomized Controlled Trials. Int. J. Environ. Res. Public Health 2018, 15, 1292. [Google Scholar] [CrossRef] [PubMed]
- Hamacher, D.; Herold, F.; Wiegel, P.; Hamacher, D.; Schega, L. Brain activity during walking: A systematic review. Neurosci. Biobehav. Rev. 2015, 57, 310–327. [Google Scholar] [CrossRef] [PubMed]
- Hartholt, K.A.; Olinder, S.; Vander-Cammen, T.J.; Panneman, M.J.; Vander-Velde, N.; Van-Lieshout, E.M.; Patka, P.; Van-Beeck, E.F. Costs of falls in an ageing population: A nationwide study from the Netherlands (2007–2009). Injury 2012, 43, 1199–1203. [Google Scholar] [CrossRef]
- Cali, C.M.; Kiel, D.P. An Epidemiologic Study of Fall-Related Fractures Among Institutionalized Older People. J. Am. Geriatr. Soc. 1995, 43, 1336–1340. [Google Scholar] [CrossRef]
- Wayne, P.M.; Yeh, G.; Mehta, D. Minding the Mind-Body Literature. J. Altern. Complement. Med. 2016, 22, 849–852. [Google Scholar] [CrossRef]
- Demnitz, N.; Esser, P.; Dawes, H.; Valkanova, V.; Johansen-Berg, H.; Ebmeier, K.P.; Sexton, C. A systematic review and meta-analysis of cross-sectional studies examining the relationship between mobility and cognition in healthy older adults. Gait Posture 2016, 50, 164–174. [Google Scholar] [CrossRef]
- Clouston, S.A.P.; Brewster, P.; Kuh, D.; Richards, M.; Cooper, R.; Hardy, R.; Rubin, M.S.; Hofer, S.M. The Dynamic Relationship Between Physical Function and Cognition in Longitudinal Aging Cohorts. Epidemiol. Rev. 2013, 35, 33–50. [Google Scholar] [CrossRef]
- Renae, L.; Smith-Ray, S.L.; Hughes, T.R.; Prohaska, D.M.; Little, D.A.; Jurivich, D. Hedeker, Impact of Cognitive Training on Balance and Gait in Older Adults. J. Gerontol. B Psychol. Sci. Soc. Sci. 2015, 70, 357–366. [Google Scholar] [CrossRef]
- Zou, L.; Zhang, Y.; Liu, Y.; Tian, X.; Xiao, T.; Liu, X.; Yeung, A.S.; Liu, J.; Wang, X.; Yang, Q. The Effects of Tai Chi Chuan Versus Core Stability Training on Lower-Limb Neuromuscular Function in Aging Individuals with Non-Specific Chronic Lower Back Pain. Medicina 2019, 55, 60. [Google Scholar] [CrossRef]
- Liu, J.; Yeung, A.; Xiao, T.; Tian, X.; Kong, Z.; Zou, L.; Wang, X. Chen-Style Tai Chi for Individuals (Aged 50 Years Old or Above) with Chronic Non-Specific Low Back Pain: A Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2019, 16, 517. [Google Scholar] [CrossRef]
- Lu, J.; Li, D.; Li, F.; Zhou, A.; Wang, F.; Zuo, X.; Jia, X.-F.; Song, H.; Jia, J. Montreal Cognitive Assessment in Detecting Cognitive Impairment in Chinese Elderly Individuals: A Population-Based Study. J. Geriatr. Psychiatry Neurol. 2011, 24, 184–190. [Google Scholar] [CrossRef]
- Chen, K.L.; Xu, Y.; Chu, A.Q.; Ding, D.; Liang, X.N.; Nasreddine, Z.S.; Dong, Q.; Hong, Z.; Zhao, Q.H.; Guo, Q.H. Validation of the Chinese Version of Montreal Cognitive Assessment Basic for Screening Mild Cognitive Impairment. J. Am. Geriatr. Soc. 2016, 64, e285–e290. [Google Scholar] [CrossRef]
- Michikawa, T.; Nishiwaki, Y.; Takebayashi, T.; Toyama, Y. One-leg standing test for elderly populations. J. Orthop. Sci. 2009, 14, 675–685. [Google Scholar] [CrossRef]
- Chan, P.P.; Tou, J.I.S.; Tse, M.M.; Ng, S.S. Reliability and Validity of the Timed Up and Go Test With a Motor Task in People With Chronic Stroke. Arch. Phys. Med. Rehabil. 2017, 98, 2213–2220. [Google Scholar] [CrossRef]
- Hardy, R.; Cooper, R.; Shah, I.; Harridge, S.; Guralnik, J.; Kuh, D. Is chair rise performance a useful measure of leg power? Aging Clin. Exp. Res. 2010, 22, 4128. [Google Scholar] [CrossRef]
- Kathrin, R.; Angie, L.; Anita, H.; Volkmar, L.; Joern, K.; Tanja, B.; Patrick, M.; Notger, G. Dance training is superioer to repetitive physical exercise in inducing brain plasticity in the elderly. PLos ONE 2018, 13, e0196636. [Google Scholar]
- Catherine-Alexandra, G.; Nicolas, B.; Floerence, S.; Thien, T.; Laurent, B.; Nathalie, A.; Louis, B. Gross motor skills training leads to increase brain-derived neurothrophic factor levels in healthy older adults: A pilot study. Front. Physiol. 2018, 10, e410. [Google Scholar]
- Herold, F.; Hamacher, D.; Schega, L.; Müller, N.G. Thinking While Moving or Moving While Thinking—Concepts of Motor-Cognitive Training for Cognitive Performance Enhancement. Front. Aging Neurosci. 2018, 10, 288. [Google Scholar] [CrossRef]
- Lyons, J.G.; Heeren, T.; Stuver, S.O.; Fredman, L. Assessing the agreement between 3-meter and 6-meter walk tests in 136 community-dwelling older adults. J. Aging Health 2015, 27, 594–605. [Google Scholar] [CrossRef]
- Z-gambo, J.; Siulapwa, Y.; Michelo, C. Pattern of acute poisoning at two urban referral hospitals in Lusaka, Zambia. BMC Emerg. Med. 2016, 16, 2. [Google Scholar] [CrossRef]
- Liang, K.-Y.; Zeger, S.L. Longitudinal data analysis using generalized linear models. Biometrika 1986, 73, 13–22. [Google Scholar] [CrossRef]
- Alan, E.; Hubbard, J.A.; Nancy, L.; Fleischer, M.V.; Laan, S.A.; Lippman, N.J.; Tim, B.; William, A. Satariano To GEE or Not to GEE Comparing Population Average and Mixed Models for Estimating the Associations Between Neighborhood Risk Factors and Health. Epidemiology 2010, 21, 4. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Crush, E. Sensory Impairment, Functional Balance and Physical Activity with All-Cause Mortality. J. Phys. Act. Health 2016, 13, 980–987. [Google Scholar] [CrossRef]
- Firth, E.; Addoh, O.; Mann, J.; Windham, B.W.; Loprinzi, P.D. Individual and Combined Associations of Cognitive and Mobility Limitations on Mortality Risk in Older Adults. Mayo Clin. Proc. 2018, 92, 1494–1501. [Google Scholar] [CrossRef]
- Hung, C.-L.; Tseng, J.-W.; Chao, H.-H.; Hung, T.-M.; Wang, H.-S. Effect of Acute Exercise Mode on Serum Brain-Derived Neurotrophic Factor (BDNF) and Task Switching Performance. J. Clin. Med. 2018, 7, 301. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Frith, E.; Edwards, M.K.; Sng, E.; Ashpole, N. The Effects of Exercise on Memory Function Among Young to Middle-Aged Adults: Systematic Review and Recommendations for Future Research. Am. J. Health Promot. 2018, 32, 691–704. [Google Scholar] [CrossRef]
- Douris, P.; Douris, C.; Balder, N.; Lacasse, M.; Rand, A.; Tarapore, F.; Zhuchkan, A.; Handrakis, J. Martial Art Training and Cognitive Performance in Middle-Aged Adults. J. Hum. Kinet. 2015, 47, 277–283. [Google Scholar] [CrossRef]
- Moreau, D.; Morrison, A.B.; Conway, A.R. An ecological approach to cognitive enhancement: Complex motor training. Acta Psychol. 2015, 157, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Winstein, C.J.; Grafton, S.T.; Pohl, P.S. Motor task difficulty and brain activity: investigation of goal-directed reciprocal aiming using positron emission tomography. J. Neurophysiol. 1997, 77, 1581–1594. [Google Scholar] [CrossRef] [PubMed]
- Carey, J.R.; Bhatt, E.; Nagpal, A. Neuroplasticity promoted by task complexity. Exerc. Sport Sci. Rev. 2005, 33, 24–31. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Loprinzi, P.D. Brief Walking Test and Cognitive Function among Congestive Heart Failure Patients: Effect Modification by Duration of Congestive Heart Failure. Int. Cardiovasc. Forum J. 2016, 22, 346. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Herod, S.M.; Cardinal, B.J.; Noakes, T.D. Physical activity and the brain: A review of this dynamic, bi-directional relationship. Brain Res. 2013, 1539, 95–104. [Google Scholar] [CrossRef]
- Herold, F.; Wiegel, P.; Scholkmann, F.; Müller, N.G. Applications of Functional Near-Infrared Spectroscopy (fNIRS) Neuroimaging in Exercise–Cognition Science: A Systematic, Methodology-Focused Review. J. Clin. Med. 2018, 7, 466. [Google Scholar] [CrossRef]
- Herold, F.; Wiegel, P.; Scholkmann, F.; Thiers, A.; Hamacher, D.; Schega, L. Functional near-infrared spectroscopy in movement science: a systematic review on cortical activity in postural and walking tasks. Neurophotonics 2017, 4, 41403. [Google Scholar] [CrossRef] [PubMed]
| TC-24 Group | MTC Group | |
|---|---|---|
| Gender | ||
| Male (n) | 18 | 19 |
| Female (n) | 22 | 19 |
| Educational level * | ||
| Middle school or below (n) | 1 | 19 |
| Associate degree (n) | 21 | 13 |
| College or above (n) | 18 | 6 |
| Age (years) | 59.55 ± 8.91 | 58.26 ± 7.05 |
| BMI * (kg/m2) | 24.04 ± 2.66 | 25.21 ± 2.00 |
| Heart rate (beats/minute) | 73.55 ± 8.30 | 75.53 ± 5.56 |
| Variable | Time Effect | Group Effect | Interaction Effect | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Time 2 | Time 3 | MTC | Time2 × MTC | Time3 × MTC | ||||||
| Coef. (Cohen’s d) | p | Coef. (Cohen’s d) | p | Coef. (Cohen’s d) | p | Coef. (Cohen’s d) | p | Coef. (Cohen’s d) | p | |
| MoCA | 1.87 (1.091) | <0.0005 | 3.449 (1.951) | <0.0005 | 0.487 (0.769) | 0.031 | ||||
| Left leg balance (eyes closed) | 0.754 (0.3) | <0.0005 | 1.189 (0.558) | <0.0005 | 0.633 (0.742) | 0.396 | 0.446 (0.457) | 0.044 | 1.485 (1.203) | <0.0005 |
| Right leg balance (eyes closed) | 6.199 (0.216) | 0.242 | 1.181 (0.483) | <0.0005 | 0.057 (0.176) | 0.945 | −5.065 (0.212) | 0.34 | 1.385 (0.918) | <0.0005 |
| TUGT | −0.370 (0.181) | <0.0005 | −0.874 (0.471) | <0.0005 | −0.366 (0.48) | 0.578 | −0.213 (0.558) | 0.013 | −0.576 (0.523) | 0.02 |
| CST | −0.401 (0.248) | <0.0005 | −0.922 (0.531) | <0.0005 | −0.022 (0.228) | 0.971 | −0.221 (0.555) | 0.014 | −0.116 (0.104) | 0.642 |
| 6-MWT | −0.266 (0.395) | 0.002 | 0.062 (0.188) | 0.681 | −0.051 (0.483) | 0.842 | −0.256 (0.529) | 0.018 | −0.498 (0.661) | 0.003 |
| Variable | TC-24 Group | MTC Group | ||||
|---|---|---|---|---|---|---|
| Baseline | Week 6 | Week 12 | Baseline | Week 6 | Week 12 | |
| The Montreal Cognitive Assessment | 26.62 ± 1.39 (24.98 ± 1.65) | 28.38 ± 1.27 (26.73 ± 1.45) | 29.90 ± 0.38 (28.26 ± 1.4) | 26.18 ± 1.39 (26.38 ± 1.64) | 28.18 ± 1.43 (28.38 ± 1.42) | 29.82 ± 0.51 (30.01 ± 1.65) |
| Left leg balance (eyes closed) | 3.86 ± 2.55 (7.24 ± 2.93) | 4.61 ± 2.65 (7.99 ± 2.95) | 5.04 ± 2.74 (8.43 ± 3.13) | 3.58 ± 2.55 (9.04 ± 2.87) | 4.78 ± 3.32 (10.24 ± 3.57) | 6.25 ± 3.44 (11.71 ± 3.73) |
| Right leg balance (eyes closed) | 3.29 ± 2.23 (8.13 ± 3.87) | 9.48 ± 33.56 (14.33 ± 33.3) | 4.47 ± 2.05 (9.31 ± 3.61) | 3.95 ± 3.32 (6.88 ± 3.9) | 5.08 ± 3.59 (8.02 ± 4.14) | 6.52 ± 3.33 (9.45 ± 3.97) |
| TUGT | 10.10 ± 2.59 (8.11 ± 2.59) | 9.73 ± 2.5 (7.74 ± 2.54) | 9.22 ± 1.9 (7.24 ± 2.24) | 10.11 ± 2.33 (7.18 ± 2.57) | 9.52 ± 2.29 (6.6 ± 2.6) | 8.66 ± 1.66 (5.73 ± 2.08) |
| CST | 9.15 ± 2.20 (3.35 ± 2.17) | 8.75 ± 2.12 (2.95 ± 2.11) | 8.23 ± 1.46 (2.43 ± 1.31) | 9.59 ± 2.08 (3.02 ± 2.01) | 8.97 ± 1.95 (2.4 ± 1.91) | 8.55 ± 1.76 (1.99 ± 1.77) |
| 6-MWT | 5.10 ± 1.17 (5.39 ± 1.15) | 4.83 ± 1.12 (5.13 ± 1.1) | 5.16 ± 0.93 (5.45 ± 0.95) | 5.13 ± 0.78 (5.19 ± 0.86) | 4.61 ± 0.70 (4.67 ± 0.74) | 4.70 ± 0.63 (4.75 ± 0.69) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zou, L.; Loprinzi, P.D.; Yu, J.J.; Yang, L.; Li, C.; Yeung, A.S.; Kong, Z.; Chiou, S.-Y.; Xiao, T. Superior Effects of Modified Chen-Style Tai Chi versus 24-Style Tai Chi on Cognitive Function, Fitness, and Balance Performance in Adults over 55. Brain Sci. 2019, 9, 102. https://doi.org/10.3390/brainsci9050102
Zou L, Loprinzi PD, Yu JJ, Yang L, Li C, Yeung AS, Kong Z, Chiou S-Y, Xiao T. Superior Effects of Modified Chen-Style Tai Chi versus 24-Style Tai Chi on Cognitive Function, Fitness, and Balance Performance in Adults over 55. Brain Sciences. 2019; 9(5):102. https://doi.org/10.3390/brainsci9050102
Chicago/Turabian StyleZou, Liye, Paul D. Loprinzi, Jane Jie Yu, Lin Yang, Chunxiao Li, Albert S. Yeung, Zhaowei Kong, Shin-Yi Chiou, and Tao Xiao. 2019. "Superior Effects of Modified Chen-Style Tai Chi versus 24-Style Tai Chi on Cognitive Function, Fitness, and Balance Performance in Adults over 55" Brain Sciences 9, no. 5: 102. https://doi.org/10.3390/brainsci9050102
APA StyleZou, L., Loprinzi, P. D., Yu, J. J., Yang, L., Li, C., Yeung, A. S., Kong, Z., Chiou, S.-Y., & Xiao, T. (2019). Superior Effects of Modified Chen-Style Tai Chi versus 24-Style Tai Chi on Cognitive Function, Fitness, and Balance Performance in Adults over 55. Brain Sciences, 9(5), 102. https://doi.org/10.3390/brainsci9050102








