Ayres Theories of Autism and Sensory Integration Revisited: What Contemporary Neuroscience Has to Say
Abstract
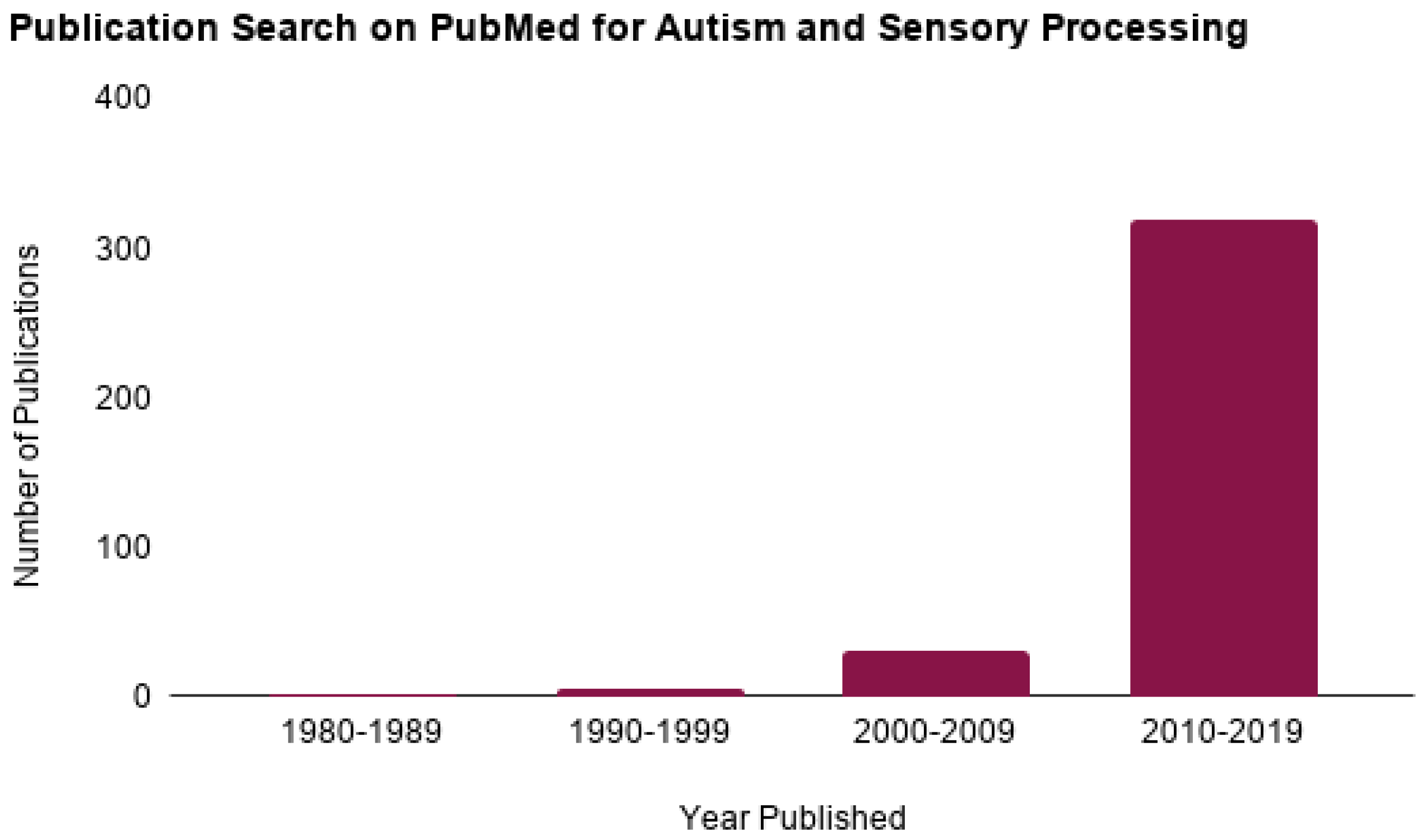
1. Introduction
2. Background
3. Current Neuroscience Evidence
3.1. Registration and Modulation: Emotion-Related Brain Regions (Previously Referred to as the Limbic System)
3.1.1. Brain Structure and Function
3.1.2. Functional Connectivity
3.1.3. Neural Responses to Aversive or Pleasant Sensory Stimulation
3.2. Motivation: Attraction and Reward in the “I Want to Do It Part of the Brain”
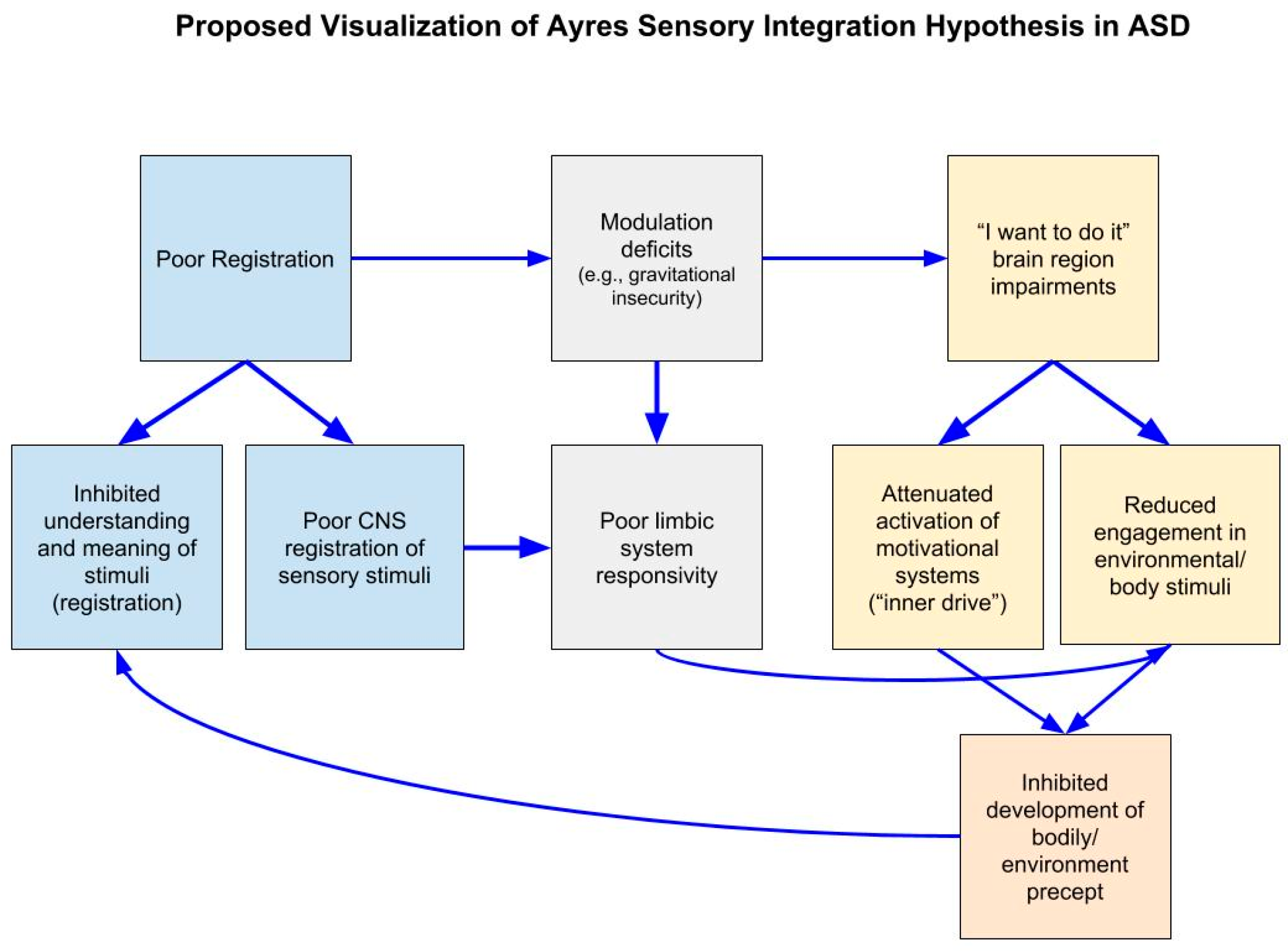
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; DSM-5; American Psychiatric Association: Philadelphia, PA, USA, 2013; ISBN 978-0-89-042555-8. [Google Scholar]
- Baio, J.; Wiggins, L.; Christensen, D.L.; Maenner, M.J.; Daniels, J.; Warren, Z.; Kurzius-Spencer, M.; Zahorodny, W.; Rosenberg, C.R.; White, T.; et al. Prevalence of Autism Spectrum Disorder among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2014. MMWR Surveill. Summ. 2018, 67, 1–23. [Google Scholar] [CrossRef]
- Geschwind, D.H. Advances in Autism. Annu. Rev. Med. 2009, 60, 367–380. [Google Scholar] [CrossRef] [PubMed]
- Marco, E.J.; Hinkley, L.B.N.; Hill, S.S.; Nagarajan, S.S. Sensory Processing in Autism: A Review of Neurophysiologic Findings. Pediatr. Res. 2011, 69, 48R–54R. [Google Scholar] [CrossRef] [PubMed]
- Jasmin, E.; Couture, M.; McKinley, P.; Reid, G.; Fombonne, E.; Gisel, E. Sensori-Motor and Daily Living Skills of Preschool Children with Autism Spectrum Disorders. J. Autism Dev. Disord. 2009, 39, 231–241. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, M.; Lord, C.; Ulrich, D. The Relationship of Motor Skills and Adaptive Behavior Skills in Young Children with Autism Spectrum Disorders. Res. Autism Spectr. Disord. 2013, 7, 1383–1390. [Google Scholar] [CrossRef]
- Matsushima, K.; Kato, T. Social Interaction and Atypical Sensory Processing in Children with Autism Spectrum Disorders. Hong Kong J. Occup. Ther. 2013, 23, 89–96. [Google Scholar] [CrossRef]
- Ayres, J. Sensory integration therapy. In Sensory Integration and the Child; Western Psychological Services: Los Angeles, CA, USA, 1979; p. 1352156. [Google Scholar]
- Wing, L.; Yeates, S.R.; Brierley, L.M.; Gould, J. The Prevalence of Early Childhood Autism: Comparison of Administrative and Epidemiological Studies. Psychol. Med. 1976, 6, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Ayres, A.J.; Tickle, L.S. Hyper-Responsivity to Touch and Vestibular Stimuli as a Predictor of Positive Response to Sensory Integration Procedures by Autistic Children. Am. J. Occup. Ther. 1980, 34, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Lectures and Speeches (1964–1985), Dr. A. Jean Ayres Archive, Collection no. 0317, Special Collections, USC Libraries, University of Southern California. Available online: https://archives.usc.edu/repositories/3/resources/2321 (accessed on 20 March 2019).
- Koegel, R.L.; Mentis, M. Motivation in Childhood Autism: Can They or Won’t They? J. Child Psychol. Psychiatry 1985, 26, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Dawson, G.; Webb, S.J.; McPartland, J. Understanding the Nature of Face Processing Impairment in Autism: Insights from Behavioral and Electrophysiological Studies. Dev. Neuropsychol. 2005, 27, 403–424. [Google Scholar] [CrossRef] [PubMed]
- Harms, M.B.; Martin, A.; Wallace, G.L. Facial Emotion Recognition in Autism Spectrum Disorders: A Review of Behavioral and Neuroimaging Studies. Neuropsychol. Rev. 2010, 20, 290–322. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.; Carper, R.A.; Abbott, A.E.; Chen, C.P.; Solders, S.; Nakutin, S.; Datko, M.C.; Fishman, I.; Müller, R.-A. Regional Specificity of Aberrant Thalamocortical Connectivity in Autism. Hum. Brain Mapp. 2015, 36, 4497–4511. [Google Scholar] [CrossRef] [PubMed]
- Gibbard, C.R.; Ren, J.; Skuse, D.H.; Clayden, J.D.; Clark, C.A. Structural Connectivity of the Amygdala in Young Adults with Autism Spectrum Disorder. Hum. Brain Mapp. 2018, 39, 1270–1282. [Google Scholar] [CrossRef] [PubMed]
- Courchesne, E.; Pierce, K.; Schumann, C.M.; Redcay, E.; Buckwalter, J.A.; Kennedy, D.P.; Morgan, J. Mapping Early Brain Development in Autism. Neuron 2007, 56, 399–413. [Google Scholar] [CrossRef] [PubMed]
- Buxbaum, J.D.; Hof, P.R.; Morgan, J.T.; Nordahl, C.W.; Schumann, C.M. The Amygdala in Autism Spectrum Disorders. Neurosci. Autism Spectr. Disord. 2013, 297–312. [Google Scholar] [CrossRef]
- Pfeiffer, B.A.; Koenig, K.; Kinnealey, M.; Sheppard, M.; Henderson, L. Effectiveness of Sensory Integration Interventions in Children with Autism Spectrum Disorders: A Pilot Study. Am. J. Occup. Ther. 2011, 65, 76–85. [Google Scholar] [CrossRef]
- Schaaf, R.C.; Hunt, J.; Benevides, T. Occupational Therapy Using Sensory Integration to Improve Participation of a Child with Autism: A Case Report. Am. J. Occup. Ther. 2012, 66, 547–555. [Google Scholar] [CrossRef]
- Schaaf, R.C.; Benevides, T.; Mailloux, Z.; Faller, P.; Hunt, J.; van Hooydonk, E.; Freeman, R.; Leiby, B.; Sendecki, J.; Kelly, D. An Intervention for Sensory Difficulties in Children with Autism: A Randomized Trial. J. Autism Dev. Disord. 2014, 44, 1493–1506. [Google Scholar] [CrossRef] [PubMed]
- Watling, R.; Hauer, S. Effectiveness of Ayres Sensory Integration® and Sensory-Based Interventions for People with Autism Spectrum Disorder: A Systematic Review. Am. J. Occup. Ther. 2015, 69. [Google Scholar] [CrossRef]
- Kandel, E.R.; Schwartz, J.H.; James, H.; Jessell, T.M. (Eds.) Principles of Neural Science; Elsevier: New York, NY, USA, 1991; ISBN 0838580688. [Google Scholar]
- Damasio, A.R. Emotion in the Perspective of an Integrated Nervous System. Brain Res. Rev. 1998, 26, 83–86. [Google Scholar] [CrossRef]
- Rolls, E.T. Limbic Systems for Emotion and For Memory but No Single Limbic System. Cortex 2015, 62, 119–157. [Google Scholar] [CrossRef] [PubMed]
- Herrington, J.D.; Maddox, B.B.; Kerns, C.M.; Rump, K.; Worley, J.A.; Bush, J.C.; McVey, A.J.; Schultz, R.T.; Miller, J.S. Amygdala Volume Differences in Autism Spectrum Disorder Are Related to Anxiety. J. Autism Dev. Disord. 2017, 47, 3682–3691. [Google Scholar] [CrossRef] [PubMed]
- Radeloff, D.; Ciaramidaro, A.; Siniatchkin, M.; Hainz, D.; Schlitt, S.; Weber, B.; Poustka, F.; Bölte, S.; Walter, H.; Freitag, C.M. Structural Alterations of The Social Brain: A Comparison between Schizophrenia and Autism. PLoS ONE 2014, 9, e106539. [Google Scholar] [CrossRef]
- Dalton, K.M.; Nacewicz, B.M.; Alexander, A.L.; Davidson, R.J. Gaze-Fixation, Brain Activation and Amygdala Volume in Unaffected Siblings of Individuals with Autism. Biol. Psychiatry 2007, 61, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Libero, L.E.; Burge, W.K.; Deshpande, H.D.; Pestilli, F.; Kana, R.K. White Matter Diffusion of Major Fiber Tracts Implicated in Autism Spectrum Disorder. Brain Connect. 2016, 6, 691–699. [Google Scholar] [CrossRef] [PubMed]
- Schumann, C.M.; Hamstra, J.; Goodlin-Jones, B.L.; Lotspeich, L.J.; Kwon, H.; Buonocore, M.H.; Lammers, C.R.; Reiss, A.L.; Amaral, D.G. The Amygdala Is Enlarged in Children but Not Adolescents with Autism; The Hippocampus Is Enlarged at All Ages. J. Neurosci. 2004, 24, 6392–6401. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Qiu, T.; Ke, X.; Xiao, X.; Xiao, T.; Liang, F.; Zou, B.; Huang, H.; Fang, H.; Chu, K.; et al. Autism Spectrum Disorder as Early Neurodevelopmental Disorder: Evidence from the Brain Imaging Abnormalities in 2–3 Years Old Toddlers. J. Autism Dev. Disord. 2014, 44, 1633–1640. [Google Scholar] [CrossRef] [PubMed]
- Courchesne, E.; Campbell, K.; Solso, S. Brain Growth across the Life Span in Autism: Age-Specific Changes in Anatomical Pathology. Brain Res. 2011, 1380, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Schumann, C.M.; Amaral, D.G. Stereological Analysis of Amygdala Neuron Number in Autism. J. Neurosci. 2006, 26, 7674–7679. [Google Scholar] [CrossRef]
- Aylward, E.H.; Minshew, N.J.; Goldstein, G.; Honeycutt, N.A.; Augustine, A.M.; Yates, K.O.; Barta, P.E.; Pearlson, G.D. MRI Volumes of Amygdala and Hippocampus in Non-Mentally Retarded Autistic Adolescents and Adults. Neurology 1999, 53, 2145–2150. [Google Scholar] [CrossRef] [PubMed]
- Braden, B.B.; Smith, C.J.; Thompson, A.; Glaspy, T.K.; Wood, E.; Vatsa, D.; Abbott, A.E.; McGee, S.C.; Baxter, L.C. Executive Function and Functional and Structural Brain Differences in Middle-Age Adults with Autism Spectrum Disorder. Autism Res. 2017, 10, 1945–1959. [Google Scholar] [CrossRef] [PubMed]
- Eilam-Stock, T.; Wu, T.; Spagna, A.; Egan, L.J.; Fan, J. Neuroanatomical Alterations in High-Functioning Adults with Autism Spectrum Disorder. Front. Neurosci. 2016, 10, 237. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.H.; Greenspan, K.S.; van Erp, T.G.M. Pallidum and Lateral Ventricle Volume Enlargement in Autism Spectrum Disorder. Psychiatry Res. Neuroimaging 2016, 252, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Groen, W.; Mennes, M.; Greven, C.; Buitelaar, J.; Rommelse, N. Revisiting Subcortical Brain Volume Correlates of Autism in the ABIDE Dataset: Effects of Age and Sex. Psychol. Med. 2018, 48, 654–668. [Google Scholar] [CrossRef]
- Mosconi, M.W.; Cody-Hazlett, H.; Poe, M.D.; Gerig, G.; Gimpel-Smith, R.; Piven, J. Longitudinal Study of Amygdala Volume and Joint Attention in 2- to 4-Year-Old Children with Autism. Arch. Gen. Psychiatry 2009, 66, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Nacewicz, B.M.; Dalton, K.M.; Johnstone, T.; Long, M.T.; McAuliff, E.M.; Oakes, T.R.; Alexander, A.L.; Davidson, R.J. Amygdala Volume and Nonverbal Social Impairment in Adolescent and Adult Males with Autism. Arch. Gen. Psychiatry 2006, 63, 1417–1428. [Google Scholar] [CrossRef]
- Lopez-Larson, M.P.; King, J.B.; Terry, J.; McGlade, E.C.; Yurgelun-Todd, D. Reduced Insular Volume in Attention Deficit Hyperactivity Disorder. Psychiatry Res. Neuroimaging 2012, 204, 32–39. [Google Scholar] [CrossRef]
- Pagnozzi, A.M.; Conti, E.; Calderoni, S.; Fripp, J.; Rose, S.E. A Systematic Review of Structural MRI Biomarkers in Autism Spectrum Disorder: A Machine Learning Perspective. Int. J. Dev. Neurosci. 2018, 71, 68–82. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-S.; Owen, J.P.; Desai, S.S.; Hill, S.S.; Arnett, A.B.; Harris, J.; Marco, E.J.; Mukherjee, P. Autism and Sensory Processing Disorders: Shared White Matter Disruption in Sensory Pathways But Divergent Connectivity in Social-Emotional Pathways. PLoS ONE 2014, 9, e103038. [Google Scholar] [CrossRef]
- Dziuk, M.A.; Larson, J.C.G.; Apostu, A.; Mahone, E.M.; Denckla, M.B.; Mostofsky, S.H. Dyspraxia in Autism: Association with Motor, Social and Communicative Deficits. Dev. Med. Child Neurol. 2007, 49, 734–739. [Google Scholar] [CrossRef]
- Mostofsky, S.H.; Burgess, M.P.; Gidley Larson, J.C. Increased Motor Cortex White Matter Volume Predicts Motor Impairment in Autism. Brain 2007, 130, 2117–2122. [Google Scholar] [CrossRef]
- Hanaie, R.; Mohri, I.; Kagitani-Shimono, K.; Tachibana, M.; Matsuzaki, J.; Hirata, I.; Nagatani, F.; Watanabe, Y.; Fujita, N.; Taniike, M. White Matter Volume in the Brainstem and Inferior Parietal Lobule Is Related to Motor Performance in Children with Autism Spectrum Disorder: A Voxel-Based Morphometry Study. Autism Res. 2016, 9, 981–992. [Google Scholar] [CrossRef] [PubMed]
- Fuster, J.M.; Uyeda, A.A. Reactivity of Limbic Neurons of the Monkey to Appetitive and Aversive Signals. Electroencephalogr. Clin. Neurophysiol. 1971, 30, 281–293. [Google Scholar] [CrossRef]
- Adolphs, R.; Tranel, D.; Hamann, S.; Young, A.W.; Calder, A.J.; Phelps, E.A.; Anderson, A.; Lee, G.P.; Damasio, A.R. Recognition of Facial Emotion in Nine Individuals with Bilateral Amygdala Damage. Neuropsychologia 1999, 37, 1111–1117. [Google Scholar] [CrossRef]
- Peck, C.J.; Salzman, C.D. The Amygdala and Basal Forebrain as a Pathway for Motivationally Guided Attention. J. Neurosci. 2014, 34, 13757–13767. [Google Scholar] [CrossRef] [PubMed]
- Pierce, K.; Haist, F.; Sedaghat, F.; Courchesne, E. The Brain Response to Personally Familiar Faces in Autism: Findings of Fusiform Activity and Beyond. Brain 2004, 127, 2703–2716. [Google Scholar] [CrossRef]
- Rudie, J.D.; Brown, J.A.; Beck-Pancer, D.; Hernandez, L.M.; Dennis, E.L.; Thompson, P.M.; Bookheimer, S.Y.; Dapretto, M. Altered Functional and Structural Brain Network Organization in Autism. NeuroImage Clin. 2012, 2, 79–94. [Google Scholar] [CrossRef]
- Iacoboni, M.; Dapretto, M. The Mirror Neuron System and the Consequences of Its Dysfunction. Nat. Rev. Neurosci. 2006, 7, 942–951. [Google Scholar] [CrossRef]
- Tottenham, N.; Hertzig, M.E.; Gillespie-Lynch, K.; Gilhooly, T.; Millner, A.J.; Casey, B.J. Elevated Amygdala Response to Faces and Gaze Aversion in Autism Spectrum Disorder. Soc. Cogn. Affect. Neurosci. 2014, 9, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Dalton, K.M.; Nacewicz, B.M.; Johnstone, T.; Schaefer, H.S.; Gernsbacher, M.A.; Goldsmith, H.H.; Alexander, A.L.; Davidson, R.J. Gaze Fixation and The Neural Circuitry of Face Processing in Autism. Nat. Neurosci. 2005, 8, 519–526. [Google Scholar] [CrossRef]
- Kleinhans, N.M.; Richards, T.; Weaver, K.; Johnson, L.C.; Greenson, J.; Dawson, G.; Aylward, E. Association Between Amygdala Response to Emotional Faces and Social Anxiety in Autism Spectrum Disorders. Neuropsychologia 2010, 48, 3665–3670. [Google Scholar] [CrossRef] [PubMed]
- Kylliäinen, A.; Wallace, S.; Coutanche, M.N.; Leppänen, J.M.; Cusack, J.; Bailey, A.J.; Hietanen, J.K. Affective-Motivational Brain Responses to Direct Gaze in Children with Autism Spectrum Disorder. J. Child Psychol. Psychiatry 2012, 53, 790–797. [Google Scholar] [CrossRef]
- Pelphrey, K.A.; Morris, J.P.; McCarthy, G. Neural Basis of Eye Gaze Processing Deficits in Autism. Brain 2005, 128, 1038–1048. [Google Scholar] [CrossRef] [PubMed]
- Klin, A.; Jones, W.; Schultz, R.; Volkmar, F.; Cohen, D. Visual Fixation Patterns during Viewing of Naturalistic Social Situations as Predictors of Social Competence in Individuals with Autism. Arch. Gen. Psychiatry 2002, 59, 809–816. [Google Scholar] [CrossRef]
- Moriuchi, J.M.; Klin, A.; Jones, W. Mechanisms of Diminished Attention to Eyes in Autism. Am. J. Psychiatry 2017, 174, 26–35. [Google Scholar] [CrossRef]
- Schieber, M.H.; Baker, J.F. Control of Movement. In Fundamental Neuroscience; Academic Press: Cambridge, MA, USA, 2013; pp. 631–651. [Google Scholar]
- Odriozola, P.; Uddin, L.Q.; Lynch, C.J.; Kochalka, J.; Chen, T.; Menon, V. Insula Response and Connectivity During Social and Non-Social Attention in Children with Autism. Soc. Cogn. Affect. Neurosci. 2016, 11, 433–444. [Google Scholar] [CrossRef]
- Menon, V.; Uddin, L.Q. Saliency, Switching, Attention and Control: A Network Model of Insula Function. Brain Struct. Funct. 2010, 214, 655–667. [Google Scholar] [CrossRef] [PubMed]
- DuBois, D.; Ameis, S.H.; Lai, M.-C.; Casanova, M.F.; Desarkar, P. Interoception in Autism Spectrum Disorder: A Review. Int. J. Dev. Neurosci. 2016, 52, 104–111. [Google Scholar] [CrossRef]
- Abbott, A.E.; Nair, A.; Keown, C.L.; Datko, M.; Jahedi, A.; Fishman, I.; Müller, R.-A. Patterns of Atypical Functional Connectivity and Behavioral Links in Autism Differ Between Default, Salience and Executive Networks. Cereb. Cortex 2016, 26, 4034–4045. [Google Scholar] [CrossRef]
- Delmonte, S.; Gallagher, L.; O’Hanlon, E.; McGrath, J.; Balsters, J.H. Functional and Structural Connectivity of Frontostriatal Circuitry in Autism Spectrum Disorder. Front. Hum. Neurosci. 2013, 7, 430. [Google Scholar] [CrossRef]
- Uddin, L.Q.; Menon, V. The Anterior Insula in Autism: Under-Connected and Under-Examined. Neurosci. Biobehav. Rev. 2009, 33, 1198–1203. [Google Scholar] [CrossRef]
- Haist, F.; Adamo, M.; Westerfield, M.; Courchesne, E.; Townsend, J. The Functional Neuroanatomy of Spatial Attention in Autism Spectrum Disorder. Dev. Neuropsychol. 2005, 27, 425–458. [Google Scholar] [CrossRef]
- Di Martino, A.; Ross, K.; Uddin, L.Q.; Sklar, A.B.; Castellanos, F.X.; Milham, M.P. Functional Brain Correlates of Social and Nonsocial Processes in Autism Spectrum Disorders: An Activation Likelihood Estimation Meta-Analysis. Biol. Psychiatry 2009, 65, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Shafritz, K.M.; Dichter, G.S.; Baranek, G.T.; Belger, A. The Neural Circuitry Mediating Shifts in Behavioral Response and Cognitive Set in Autism. Biol. Psychiatry 2008, 63, 974–980. [Google Scholar] [CrossRef] [PubMed]
- Kana, R.K.; Keller, T.A.; Minshew, N.J.; Just, M.A. Inhibitory Control in High-Functioning Autism: Decreased Activation and Underconnectivity in Inhibition Networks. Biol. Psychiatry 2007, 62, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, N.; Rubia, K.; Daly, E.; Smith, A.; Williams, S.; Murphy, D.G.M. Neural Correlates of Executive Function in Autistic Spectrum Disorders. Biol. Psychiatry 2006, 59, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Power, J.D.; Barnes, K.A.; Snyder, A.Z.; Schlaggar, B.L.; Petersen, S.E. Spurious But Systematic Correlations in Functional Connectivity MRI Networks Arise From Subject Motion. Neuroimage 2012, 59, 2142–2154. [Google Scholar] [CrossRef] [PubMed]
- Supekar, K.; Uddin, L.Q.; Khouzam, A.; Phillips, J.; Gaillard, W.D.; Kenworthy, L.E.; Yerys, B.E.; Vaidya, C.J.; Menon, V. Brain Hyperconnectivity in Children with Autism and Its Links to Social Deficits. Cell Rep. 2013, 5, 738–747. [Google Scholar] [CrossRef]
- Uddin, L.Q.; Supekar, K.; Menon, V. Reconceptualizing Functional Brain Connectivity in Autism from a Developmental Perspective. Front. Hum. Neurosci. 2013, 7, 458. [Google Scholar] [CrossRef] [PubMed]
- Rudie, J.D.; Shehzad, Z.; Hernandez, L.M.; Colich, N.L.; Bookheimer, S.Y.; Iacoboni, M.; Dapretto, M. Reduced Functional Integration and Segregation of Distributed Neural Systems Underlying Social and Emotional Information Processing in Autism Spectrum Disorders. Cereb. Cortex 2012, 22, 1025–1037. [Google Scholar] [CrossRef] [PubMed]
- Noonan, S.K.; Haist, F.; Müller, R.-A. Aberrant Functional Connectivity in Autism: Evidence from Low-Frequency BOLD Signal Fluctuations. Brain Res. 2009, 1262, 48–63. [Google Scholar] [CrossRef] [PubMed]
- Stevens, W.D.; Spreng, R.N. Resting-State Functional Connectivity MRI Reveals Active Processes Central to Cognition. Wiley Interdiscip. Rev. Cogn. Sci. 2014, 5, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Raichle, M.E. Disease and The Brain’s Dark Energy. Nat. Rev. Neurol. 2010, 6, 15–28. [Google Scholar] [CrossRef]
- Maximo, J.O.; Kana, R.K. Aberrant “Deep Connectivity” in Autism: A Cortico-Subcortical Functional Connectivity Magnetic Resonance Imaging Study. Autism Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Takarae, Y.; Luna, B.; Minshew, N.J.; Sweeney, J.A. Visual Motion Processing and Visual Sensorimotor Control in Autism. J. Int. Neuropsychol. Soc. 2014, 20, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Linke, A.C.; Jao Keehn, R.J.; Pueschel, E.B.; Fishman, I.; Müller, R.-A. Children with ASD Show Links between Aberrant Sound Processing, Social Symptoms and Atypical Auditory Interhemispheric and Thalamocortical Functional Connectivity. Dev. Cogn. Neurosci. 2018, 29, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Cerliani, L.; Mennes, M.; Thomas, R.M.; Di Martino, A.; Thioux, M.; Keysers, C. Increased Functional Connectivity Between Subcortical and Cortical Resting-State Networks in Autism Spectrum Disorder. JAMA Psychiatry 2015, 72, 767–777. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.D.; Yang, D.Y.-J.; Voos, A.C.; Bennett, R.H.; Gordon, I.; Pretzsch, C.; Beam, D.; Keifer, C.; Eilbott, J.; McGlone, F.; et al. Brain Mechanisms for Processing Affective (And Nonaffective) Touch Are Atypical in Autism. Cereb. Cortex 2016, 26, 2705–2714. [Google Scholar] [CrossRef]
- Keown, C.L.; Shih, P.; Nair, A.; Peterson, N.; Mulvey, M.E.; Müller, R.-A. Local Functional Overconnectivity in Posterior Brain Regions Is Associated with Symptom Severity in Autism Spectrum Disorders. Cell Rep. 2013, 5, 567–572. [Google Scholar] [CrossRef]
- Acevedo, B.; Aron, E.; Pospos, S.; Jessen, D. The Functional Highly Sensitive Brain: A Review of the Brain Circuits Underlying Sensory Processing Sensitivity and Seemingly Related Disorders. Philos. Trans. R. Soc. B Biol. Sci. 2018, 373, 20170161. [Google Scholar] [CrossRef]
- Green, S.A.; Hernandez, L.M.; Bowman, H.C.; Bookheimer, S.Y.; Dapretto, M. Sensory Over-Responsivity and Social Cognition in ASD: Effects of Aversive Sensory Stimuli and Attentional Modulation on Neural Responses to Social Cues. Dev. Cogn. Neurosci. 2018, 29, 127–139. [Google Scholar] [CrossRef]
- Green, S.A.; Ben-Sasson, A.; Soto, T.W.; Carter, A.S. Anxiety and Sensory Over-Responsivity in Toddlers with Autism Spectrum Disorders: Bidirectional Effects across Time. J. Autism Dev. Disord. 2012, 42, 1112–1119. [Google Scholar] [CrossRef] [PubMed]
- Green, S.A.; Hernandez, L.; Bookheimer, S.Y.; Dapretto, M. Salience Network Connectivity in Autism Is Related to Brain and Behavioral Markers of Sensory Overresponsivity. J. Am. Acad. Child Adolesc. Psychiatry 2016, 55, 618–626. [Google Scholar] [CrossRef]
- Cascio, C.J.; Moana-Filho, E.J.; Guest, S.; Nebel, M.B.; Weisner, J.; Baranek, G.T.; Essick, G.K. Perceptual and Neural Response to Affective Tactile Texture Stimulation in Adults with Autism Spectrum Disorders. Autism Res. 2012, 5, 231–244. [Google Scholar] [CrossRef]
- Le Couteur, A.; Lord, C.; Rutter, M. Autism Diagnostic Interview–Revised (ADI-R); Western Psychological Services: Los Angeles, CA, USA, 2003. [Google Scholar]
- Green, S.A.; Hernandez, L.; Bookheimer, S.Y.; Dapretto, M. Reduced Modulation of Thalamocortical Connectivity during Exposure to Sensory Stimuli in ASD. Autism Res. 2017, 10, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Green, S.A.; Rudie, J.D.; Colich, N.L.; Wood, J.J.; Shirinyan, D.; Hernandez, L.; Tottenham, N.; Dapretto, M.; Bookheimer, S.Y. Overreactive Brain Responses to Sensory Stimuli in Youth with Autism Spectrum Disorders. J. Am. Acad. Child Adolesc. Psychiatry 2013, 52, 1158–1172. [Google Scholar] [CrossRef]
- Green, S.A.; Hernandez, L.M.; Tottenham, N.; Krasileva, K.; Bookheimer, S.Y.; Dapretto, M. Neurobiology of Sensory Overresponsivity in Youth with Autism Spectrum Disorders. JAMA Psychiatry 2015, 72, 778–786. [Google Scholar] [CrossRef]
- Schoen, S.A.; Miller, L.J.; Green, K.E. Pilot Study of the Sensory over-Responsivity Scales: Assessment and Inventory. Am. J. Occup. Ther. 2008, 62, 393–406. [Google Scholar] [CrossRef]
- Wang, A.T.; Lee, S.S.; Sigman, M.; Dapretto, M. Neural Basis of Irony Comprehension in Children with Autism: The Role of Prosody and Context. Brain 2006, 129, 932–943. [Google Scholar] [CrossRef]
- Fang, Z.; Li, H.; Chen, G.; Yang, J. Unconscious Processing of Negative Animals and Objects: Role of the Amygdala Revealed by FMRI. Front. Hum. Neurosci. 2016, 10, 146. [Google Scholar] [CrossRef]
- Morris, J.S.; Öhman, A.; Dolan, R.J. Conscious and Unconscious Emotional Learning in the Human Amygdala. Nature 1998, 393, 467–470. [Google Scholar] [CrossRef] [PubMed]
- Schultz, W. Reward Processing in Primate Orbitofrontal Cortex and Basal Ganglia. Cereb. Cortex 2000, 10, 272–283. [Google Scholar] [CrossRef] [PubMed]
- Caston, J.; Chianale, C.; Delhaye-Bouchaud, N.; Mariani, J. Role of the Cerebellum in Exploration Behavior. Brain Res. 1998, 808, 232–237. [Google Scholar] [CrossRef]
- Chevallier, C.; Kohls, G.; Troiani, V.; Brodkin, E.S.; Schultz, R.T. The Social Motivation Theory. Trends Cogn. Sci. 2012, 16, 231–239. [Google Scholar] [CrossRef]
- Scott-Van Zeeland, A.A.; Dapretto, M.; Ghahremani, D.G.; Poldrack, R.A.; Bookheimer, S.Y. Reward Processing in Autism. Autism Res. 2010, 3, 53–67. [Google Scholar] [CrossRef]
- Kuhl, P.K.; Coffey-Corina, S.; Padden, D.; Dawson, G. Links between Social and Linguistic Processing of Speech in Preschool Children with Autism: Behavioral and Electrophysiological Measures. Dev. Sci. 2005, 8, F1–F12. [Google Scholar] [CrossRef]
- Abrams, D.A.; Lynch, C.J.; Cheng, K.M.; Phillips, J.; Supekar, K.; Ryali, S.; Uddin, L.Q.; Menon, V. Underconnectivity between Voice-Selective Cortex and Reward Circuitry in Children with Autism. Proc. Natl. Acad. Sci. USA 2013. [Google Scholar] [CrossRef] [PubMed]
- Nomi, J.S.; Uddin, L.Q. Face processing in autism spectrum disorders: From brain regions to brain networks. Neuropsychologia 2015, 71, 201–216. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.H.; Cross, E.S. Decreased Reward Value of Biological Motion among Individuals with Autistic Traits. Cognition 2018, 171, 1–9. [Google Scholar] [CrossRef]
- Delmonte, S.; Balsters, J.H.; McGrath, J.; Fitzgerald, J.; Brennan, S.; Fagan, A.J.; Gallagher, L. Social and Monetary Reward Processing in Autism Spectrum Disorders. Mol. Autism 2012, 3, 7. [Google Scholar] [CrossRef]
- Kohls, G.; Schulte-Rüther, M.; Nehrkorn, B.; Müller, K.; Fink, G.R.; Kamp-Becker, I.; Herpertz-Dahlmann, B.; Schultz, R.T.; Konrad, K. Reward System Dysfunction in Autism Spectrum Disorders. Soc. Cogn. Affect. Neurosci. 2013, 8, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Fletcher-Watson, S.; Leekam, S.R.; Benson, V.; Frank, M.C.; Findlay, J.M. Eye-Movements Reveal Attention to Social Information in Autism Spectrum Disorder. Neuropsychologia 2009, 47, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Sepeta, L.; Tsuchiya, N.; Davies, M.S.; Sigman, M.; Bookheimer, S.Y.; Dapretto, M. Abnormal Social Reward Processing in Autism as Indexed by Pupillary Responses to Happy Faces. J. Neurodev. Disord. 2012, 4. [Google Scholar] [CrossRef] [PubMed]
- Martineau, J.; Aguillon-Hernandez, N.; Roche, L.; Malvy, J.; Mofid, Y.; Barthelemy, C.; Elian, J.-C.; Bonnet-Brilhault, F. Interactions Sociales: Explorations Oculaires Et Pupillométrie Dans L’autisme De V Enfant [Social Interactions: Visual Exploration Behavior and Pupillary Reactivity in The Case of Children with Autism]. ANAE Approche Neuropsychol. Apprentiss. l’Enfant 2016, 28, 333–338. [Google Scholar]
- Jones, W.; Carr, K.; Klin, A. Absence of Preferential Looking to the Eyes of Approaching Adults Predicts Level of Social Disability in 2-Year-Old Toddlers with Autism Spectrum Disorder. Arch. Gen. Psychiatry 2008, 65, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Sterling, L.; Dawson, G.; Webb, S.; Murias, M.; Munson, J.; Panagiotides, H.; Aylward, E. The Role of Face Familiarity in Eye Tracking of Faces by Individuals with Autism Spectrum Disorders. J. Autism Dev. Disord. 2008, 38, 1666–1675. [Google Scholar] [CrossRef]
- Pelphrey, K.A.; Sasson, N.J.; Reznick, J.S.; Paul, G.; Goldman, B.D.; Piven, J. Visual Scanning of Faces in Autism. J. Autism Dev. Disord. 2002, 32, 249–261. [Google Scholar] [CrossRef]
- Critchley, H.D.; Daly, E.M.; Bullmore, E.T.; Williams, S.C.; Van Amelsvoort, T.; Robertson, D.M.; Rowe, A.; Phillips, M.; McAlonan, G.; Howlin, P.; et al. The Functional Neuroanatomy of Social Behaviour: Changes in Cerebral Blood Flow When People with Autistic Disorder Process Facial Expressions. Brain 2000, 123, 2203–2212. [Google Scholar] [CrossRef]
- Dichter, G.S.; Felder, J.N.; Green, S.R.; Rittenberg, A.M.; Sasson, N.J.; Bodfish, J.W. Reward Circuitry Function in Autism Spectrum Disorders. Soc. Cogn. Affect. Neurosci. 2012, 7, 160–172. [Google Scholar] [CrossRef]
- Assaf, M.; Hyatt, C.J.; Wong, C.G.; Johnson, M.R.; Schultz, R.T.; Hendler, T.; Pearlson, G.D. Mentalizing and Motivation Neural Function during Social Interactions in Autism Spectrum Disorders. NeuroImage Clin. 2013, 3, 321–331. [Google Scholar] [CrossRef]
- Pelphrey, K.A.; Michelich, C.R.; Viola, R.J.; Mack, P.B.; Allison, T.; Mccarthy, G. Neurofunctional Organization of Biological Motion Perception: An FMRI Study of Eye, Hand and Mouth Movements. Hand 2003, 2, 6. [Google Scholar]
- Klin, A.; Lin, D.J.; Gorrindo, P.; Ramsay, G.; Jones, W. Two-Year-Olds with Autism Orient to Non-Social Contingencies Rather Than Biological Motion. Nature 2009, 459, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Ventola, P.; Yang, D.Y.J.; Friedman, H.E.; Oosting, D.; Wolf, J.; Sukhodolsky, D.G.; Pelphrey, K.A. Heterogeneity of Neural Mechanisms of Response to Pivotal Response Treatment. Brain Imaging Behav. 2015, 9, 74–88. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Hofmann, J. Action Observation and Imitation in Autism Spectrum Disorders: An ALE Meta-Analysis of FMRI Studies. Brain Imaging Behav. 2016, 10, 960–969. [Google Scholar] [CrossRef] [PubMed]
- Nair, G.; Schmidt, K.; Febo, M.; Segarra, A.C.; Ferris, C.F.; Duong, T.Q. The Neural Consequences of Repeated Cocaine Exposure Revealed by Functional MRI in Awake Rats. Neuropsychopharmacology 2005, 30, 936–943. [Google Scholar] [CrossRef]
- Richard, J.M.; Castro, D.C.; DiFeliceantonio, A.G.; Robinson, M.J.F.; Berridge, K.C. Mapping Brain Circuits of Reward and Motivation: in The Footsteps of Ann Kelley. Neurosci. Biobehav. Rev. 2013, 37, 1919–1931. [Google Scholar] [CrossRef]
- Wittmann, M.; Lovero, K.L.; Lane, S.D.; Paulus, M.P. Now or Later? Striatum and Insula Activation to Immediate Versus Delayed Rewards. J. Neurosci. Psychol. Econ. 2010, 3, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Clements, C.C.; Zoltowski, A.R.; Yankowitz, L.D.; Yerys, B.E.; Schultz, R.T.; Herrington, J.D. Evaluation of The Social Motivation Hypothesis of Autism. JAMA Psychiatry 2018, 75, 797–808. [Google Scholar] [CrossRef] [PubMed]
- O’Doherty, J.P.; Deichmann, R.; Critchley, H.D.; Dolan, R.J. Neural Responses during Anticipation of a Primary Taste Reward. Neuron 2002, 33, 815–826. [Google Scholar] [CrossRef]
- O’Doherty, J.; Rolls, E.T.; Francis, S.; Bowtell, R.; McGlone, F. Representation of Pleasant and Aversive Taste in the Human Brain. J. Neurophysiol. 2001, 85, 1315–1321. [Google Scholar] [CrossRef]
- Thut, G.; Schultz, W.; Roelcke, U.; Nienhusmeier, M.; Missimer, J.; Maguire, R.P.; Leenders, K.L. Activation of the Human Brain by Monetary Reward. Neuroreport 1997, 8, 1225–1228. [Google Scholar] [CrossRef]
- Phillips, M.L.; Bullmore, E.T.; Howard, R.; Woodruff, P.W.; Wright, I.C.; Williams, S.C.; Simmons, A.; Andrew, C.; Brammer, M.; David, A.S. Investigation of Facial Recognition Memory and Happy and Sad Facial Expression Perception: An FMRI Study. Psychiatry Res. Neuroimaging 1998, 83, 127–138. [Google Scholar] [CrossRef]
- Haber, S.N.; Knutson, B. The Reward Circuit: Linking Primate Anatomy and Human Imaging. Neuropsychopharmacology 2010, 35, 4–26. [Google Scholar] [CrossRef]
- O’Connell, L.A.; Hofmann, H.A. The Vertebrate Mesolimbic Reward System and Social Behavior Network: A Comparative Synthesis. J. Comp. Neurol. 2011, 519, 3599–3639. [Google Scholar] [CrossRef] [PubMed]
- Berridge, K.C.; Kringelbach, M.L. Pleasure Systems in the Brain. Neuron 2015, 86, 646–664. [Google Scholar] [CrossRef]
- Supekar, K.; Kochalka, J.; Schaer, M.; Wakeman, H.; Qin, S.; Padmanabhan, A.; Menon, V. Deficits in Mesolimbic Reward Pathway Underlie Social Interaction Impairments in Children with Autism. Brain 2018, 141, 2795–2805. [Google Scholar] [CrossRef] [PubMed]
- Pierce, K.; Courchesne, E. Evidence for a Cerebellar Role in Reduced Exploration and Stereotyped Behavior in Autism. Biol. Psychiatry 2001, 49, 655–664. [Google Scholar] [CrossRef]
- Schmahmann, J.D. Disorders of the Cerebellum: Ataxia, Dysmetria of Thought and the Cerebellar Cognitive Affective Syndrome. J. Neuropsychiatry Clin. Neurosci. 2004, 16, 367–378. [Google Scholar] [CrossRef]
- Schutter, D.J.L.G. Human Cerebellum in Motivation and Emotion. In Handbook of the Cerebellum and Cerebellar Disorders; Springer: Dordrecht, The Netherlands, 2013; pp. 1771–1782. [Google Scholar]
- Wise, R.A. Roles for Nigrostriatal-Not Just Mesocorticolimbic-Dopamine in Reward and Addiction. Trends Neurosci. 2009. [Google Scholar] [CrossRef]
- Schmahmann, J.D. The Role of the Cerebellum in Affect and Psychosis. J. Neurolinguist. 2000, 13, 189–214. [Google Scholar] [CrossRef]
- Ebner, T.J.; Pasalar, S. Cerebellum Predicts the Future Motor State. Cerebellum 2008, 7, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Courchesne, E. Brainstem, Cerebellar and Limbic Neuroanatomical Abnormalities in Autism. Curr. Opin. Neurobiol. 1997, 7, 269–278. [Google Scholar] [CrossRef]
- Stanfield, A.C.; McIntosh, A.M.; Spencer, M.D.; Philip, R.; Gaur, S.; Lawrie, S.M. Towards a Neuroanatomy of Autism: A Systematic Review and Meta-Analysis of Structural Magnetic Resonance Imaging Studies. Eur. Psychiatry 2008, 23, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Keown, C.L.; Datko, M.C.; Chen, C.P.; Maximo, J.O.; Jahedi, A.; Müller, R.-A. Network Organization Is Globally Atypical in Autism: A Graph Theory Study of Intrinsic Functional Connectivity. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2017, 2, 66–75. [Google Scholar] [CrossRef]
- Ornitz, E.M. Vestibular Dysfunction in Schizophrenia and Childhood Autism. Compr. Psychiatry 1970, 11, 159–173. [Google Scholar] [CrossRef]
- Ritvo, E.R.; Ornitz, E.M.; Eviatar, A.; Markham, C.H.; Brown, M.B.; Mason, A. Decreased Postrotatory Nystagmus in Early Infantile Autism. Neurology 1969, 19, 653–658. [Google Scholar] [CrossRef]
- Maurer, R.G.; Damasio, A.R. Vestibular Dysfunction in Autistic Children. Dev. Med. Child Neurol. 1979, 21, 656–659. [Google Scholar] [CrossRef] [PubMed]
- Ornitz, E.M.; Brown, M.B.; Mason, A.; Putnam, N.H. Effect of Visual Input on Vestibular Nystagmus in Autistic Children. Arch. Gen. Psychiatry 1974. [Google Scholar] [CrossRef]
- Egawa, J.; Watanabe, Y.; Kitamura, H.; Endo, T.; Tamura, R.; Hasegawa, N.; Someya, T. Reduced Thalamus Volume in Non-Right-Handed Male Patients with Autism Spectrum Disorders. Psychiatry Clin. Neurosci. 2011, 65, 395. [Google Scholar] [CrossRef]
- McAlonan, G.M.; Suckling, J.; Wong, N.; Cheung, V.; Lienenkaemper, N.; Cheung, C.; Chua, S.E. Distinct Patterns of Grey Matter Abnormality in High-Functioning Autism and Asperger’s Syndrome. J. Child Psychol. Psychiatry 2008, 49, 1287–1295. [Google Scholar] [CrossRef]
- Spencer, J.V.; O’Brien, J.M.D. Visual Form-Processing Deficits in Autism. Perception 2006, 35, 1047–1055. [Google Scholar] [CrossRef]
- Waiter, G.D.; Williams, J.H.; Murray, A.D.; Gilchrist, A.; Perrett, D.I.; Whiten, A. A Voxel-Based Investigation of Brain Structure in Male Adolescents with Autistic Spectrum Disorder. Neuroimage 2004, 22, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Carson, T.B.; Wilkes, B.J.; Patel, K.; Pineda, J.L.; Ko, J.H.; Newell, K.M.; Bodfish, J.W.; Schubert, M.C.; Radonovich, K.; White, K.D.; et al. Vestibulo-Ocular Reflex Function in Children with High-Functioning Autism Spectrum Disorders. Autism Res. 2017, 10, 251–266. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, M.C.; Landa, R.; Lasker, A.; Cooper, L.; Zee, D.S. Evidence of Normal Cerebellar Control of the Vestibulo-Ocular Reflex (VOR) in Children with High-Functioning Autism. J. Autism Dev. Disord. 2000, 30, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Widiger, T.A. Diagnostic and Statistical Manual of Mental Disorders. In Encyclopedia of Psychology; American Psychological Association: Washington, DC, USA, 2000; Volume 3, pp. 32–35. [Google Scholar]
- Ben-Sasson, A.; Hen, L.; Fluss, R.; Cermak, S.A.; Engel-Yeger, B.; Gal, E. A Meta-Analysis of Sensory Modulation Symptoms in Individuals with Autism Spectrum Disorders. J. Autism Dev. Disord. 2009, 39, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Salimpoor, V.N.; van den Bosch, I.; Kovacevic, N.; McIntosh, A.R.; Dagher, A.; Zatorre, R.J. Interactions Between the Nucleus Accumbens and Auditory Cortices Predict Music Reward Value. Science 2013, 340, 216–219. [Google Scholar] [CrossRef] [PubMed]
- Gordon, I.; Jack, A.; Pretzsch, C.M.; Vander Wyk, B.; Leckman, J.F.; Feldman, R.; Pelphrey, K.A. Intranasal Oxytocin Enhances Connectivity in the Neural Circuitry Supporting Social Motivation and Social Perception in Children with Autism. Sci. Rep. 2016, 6, 35054. [Google Scholar] [CrossRef] [PubMed]
- Toback, M.; Zangeneh, K.; Siddiqui, T.J.; Marzban, H. Neurodevelopmental Disorders of the Cerebellum: Autism Spectrum Disorder. In Development of the Cerebellum from Molecular Aspects to Diseases; Springer: Cham, Switzerland, 2017; pp. 369–387. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kilroy, E.; Aziz-Zadeh, L.; Cermak, S. Ayres Theories of Autism and Sensory Integration Revisited: What Contemporary Neuroscience Has to Say. Brain Sci. 2019, 9, 68. https://doi.org/10.3390/brainsci9030068
Kilroy E, Aziz-Zadeh L, Cermak S. Ayres Theories of Autism and Sensory Integration Revisited: What Contemporary Neuroscience Has to Say. Brain Sciences. 2019; 9(3):68. https://doi.org/10.3390/brainsci9030068
Chicago/Turabian StyleKilroy, Emily, Lisa Aziz-Zadeh, and Sharon Cermak. 2019. "Ayres Theories of Autism and Sensory Integration Revisited: What Contemporary Neuroscience Has to Say" Brain Sciences 9, no. 3: 68. https://doi.org/10.3390/brainsci9030068
APA StyleKilroy, E., Aziz-Zadeh, L., & Cermak, S. (2019). Ayres Theories of Autism and Sensory Integration Revisited: What Contemporary Neuroscience Has to Say. Brain Sciences, 9(3), 68. https://doi.org/10.3390/brainsci9030068




