Individual Differences in Ethanol Drinking and Seeking Behaviors in Rats Exposed to Chronic Intermittent Ethanol Vapor Exposure is Associated with Altered CaMKII Autophosphorylation in the Nucleus Accumbens Shell
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Ethanol Self-Administration
2.3. Chronic Intermittent Ethanol Vapor Exposure (CIE)
2.4. Tail Bleeding for Determination of BEL
2.5. Drinking During Abstinence (DDA)
2.6. Extinction
2.7. Cue-Induced Reinstatement
2.8. Plasma Corticosterone Quantification
2.9. Brain Tissue Collection and Western Blotting
2.10. Statistical Analysis
3. Results
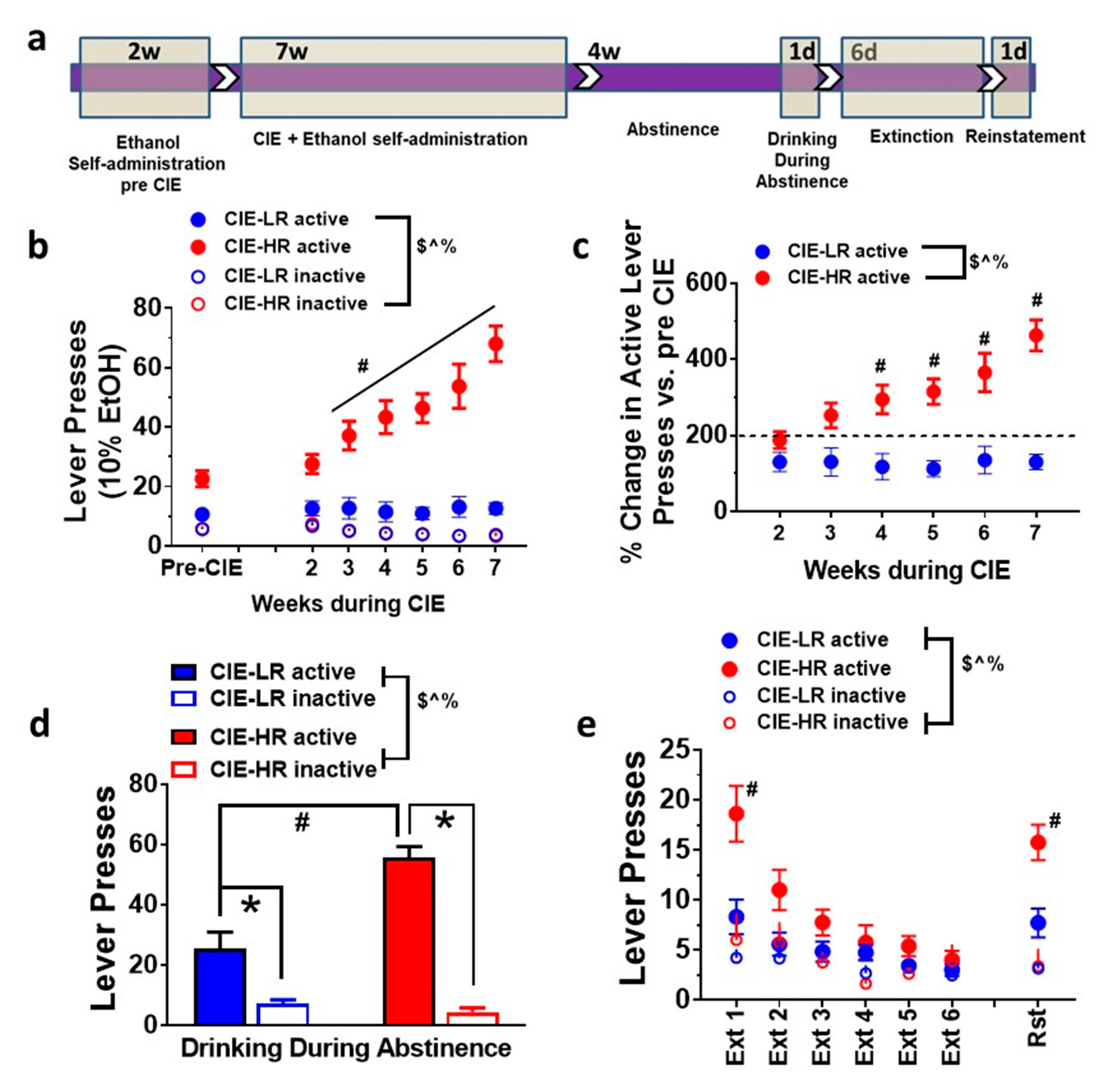
3.1. Escalation of Ethanol Drinking
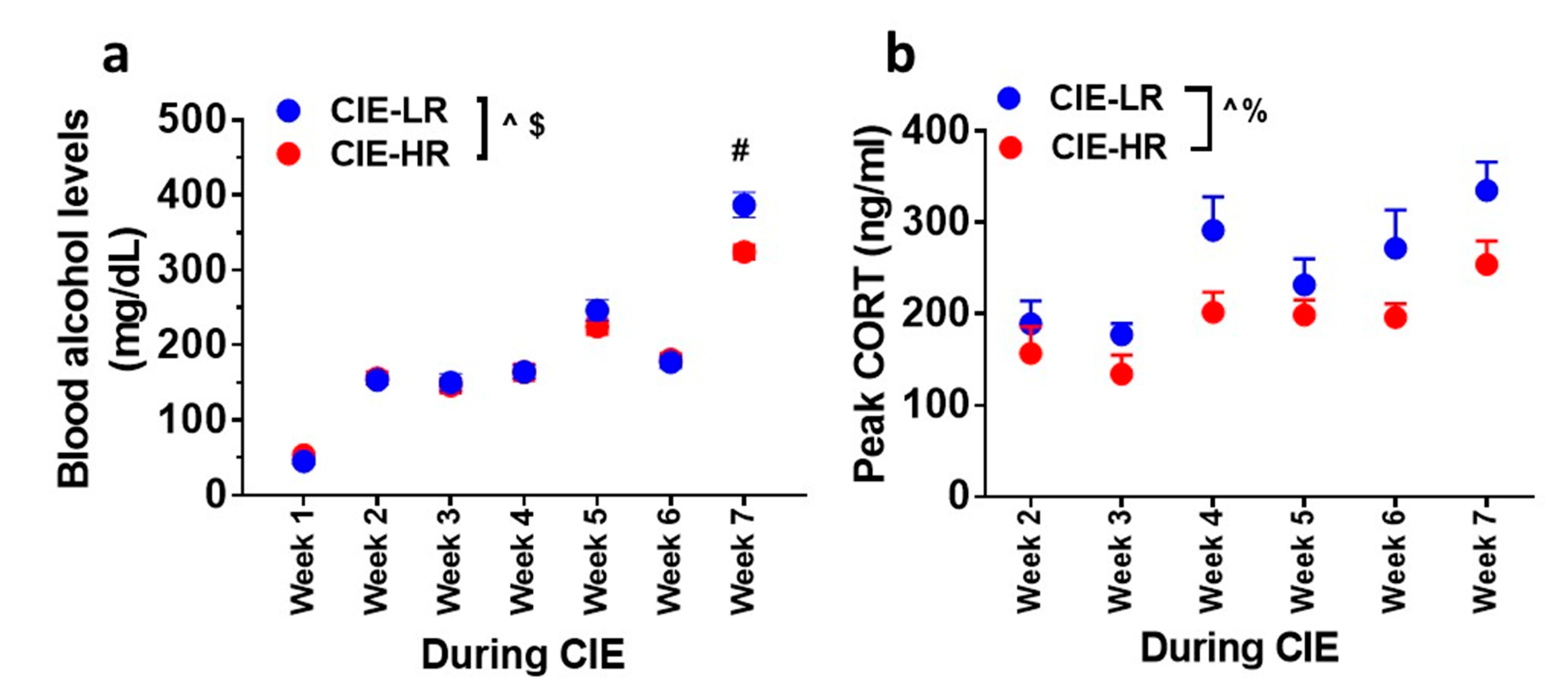
3.2. Blood Ethanol Levels during CIE
3.3. Plasma Corticosterone Levels during CIE
3.4. Drinking during Abstinence
3.5. Extinction and Contextual Cued Reinstatement of Ethanol Seeking
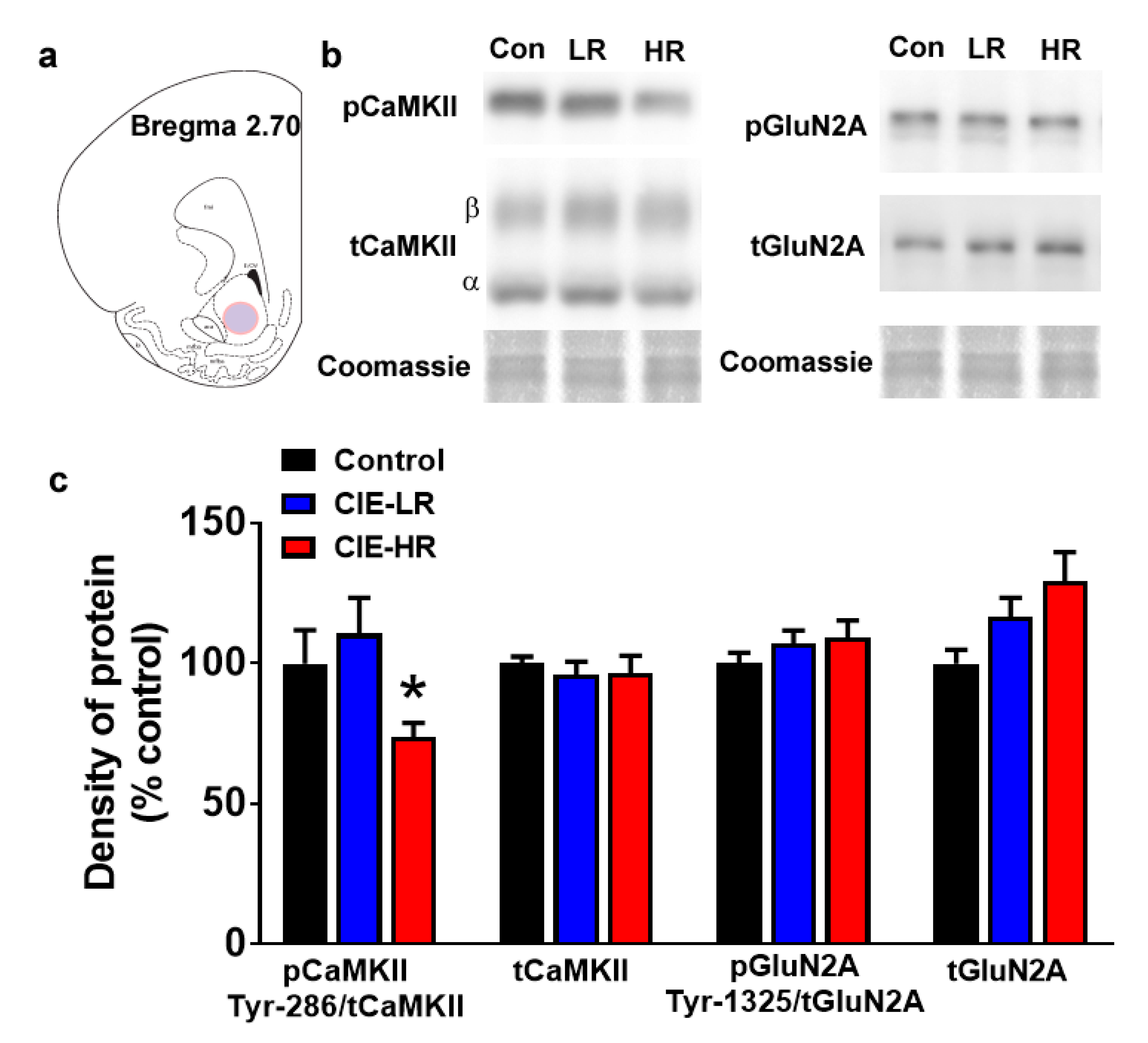
3.6. Expression of Plasticity-Related Proteins in the Nucleus Accumbens Shell of the Ventral Striatum
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- APA, American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; APA: Richmond, VA, USA, 2013. [Google Scholar]
- Ciccocioppo, R. Genetically selected alcohol preferring rats to model human alcoholism. In Behavioral Neurobiology of Alcohol Addiction; Springer: Berlin/Heidelberg, Germany, 2013; Volume 13, pp. 251–269. [Google Scholar]
- Fan, A.Z.; Chou, S.P.; Zhang, H.; Jung, J.; Grant, B.F. Prevalence and correlates of past-year recovery from DSM-5 alcohol use disorder: Results from national epidemiologic survey on alcohol and related conditions-III. Alcohol. Clin. Exp. Res. 2019, 43, 2406–2420. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P. Global use of alcohol, drugs and tobacco. Drug. Alcohol. Rev. 2006, 25, 489–502. [Google Scholar] [CrossRef] [PubMed]
- Rehm, J.; Mathers, C.; Popova, S.; Thavorncharoensap, M.; Teerawattananon, Y.; Patra, J. Global burden of disease and injury and economic cost attributable to alcohol use and alcohol-use disorders. Lancet 2009, 373, 2223–2233. [Google Scholar] [CrossRef]
- Chassin, L.; Pitts, S.C.; Prost, J. Binge drinking trajectories from adolescence to emerging adulthood in a high-risk sample: Predictors and substance abuse outcomes. J. Consult. Clin. Psychol. 2002, 70, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Enoch, M.A. Genetic influences on the development of alcoholism. Curr. Psychiatry Rep. 2013, 15, 412. [Google Scholar] [CrossRef]
- Goudriaan, A.E.; Grekin, E.R.; Sher, K.J. Decision making and response inhibition as predictors of heavy alcohol use: A prospective study. Alcohol. Clin. Exp. Res. 2011, 35, 1050–1057. [Google Scholar] [CrossRef]
- Fuehrlein, B.S.; Mota, N.; Arias, A.J.; Trevisan, L.A.; Kachadourian, L.K.; Krystal, J.H.; Pietrzak, R.H. The burden of alcohol use disorders in US military veterans: Results from the National Health and Resilience in Veterans Study. Addiction 2016, 111, 1786–1794. [Google Scholar] [CrossRef]
- Mason, B.J. Emerging pharmacotherapies for alcohol use disorder. Neuropharmacology 2017, 122, 244–253. [Google Scholar] [CrossRef]
- Colombo, G.; Agabio, R.; Lobina, C.; Reali, R.; Zocchi, A.; Fadda, F.; Gessa, G.L. Sardinian alcohol-preferring rats: A genetic animal model of anxiety. Physiol. Behav. 1995, 57, 1181–1185. [Google Scholar] [CrossRef]
- Crabbe, J.C.; Metten, P.; Rhodes, J.S.; Yu, C.H.; Brown, L.L.; Phillips, T.J.; Finn, D.A. A line of mice selected for high blood ethanol concentrations shows drinking in the dark to intoxication. Biol. Psychiatry 2009, 65, 662–670. [Google Scholar] [CrossRef]
- Le, A.D.; Israel, Y.; Juzytsch, W.; Quan, B.; Harding, S. Genetic selection for high and low alcohol consumption in a limited-access paradigm. Alcohol. Clin. Exp. Res. 2001, 25, 1613–1620. [Google Scholar] [CrossRef]
- Sinclair, J.D.; Le, A.D.; Kiianmaa, K. The AA and ANA rat lines, selected for differences in voluntary alcohol consumption. Experientia 1989, 45, 798–805. [Google Scholar] [CrossRef]
- Spoelder, M.; Hesseling, P.; Baars, A.M.; Lozeman van ‘t Klooster, J.G.; Rotte, M.D.; Vanderschuren, L.J.; Lesscher, H.M. Individual variation in alcohol intake predicts reinforcement, motivation, and compulsive alcohol use in rats. Alcohol. Clin. Exp. Res. 2015, 39, 2427–2437. [Google Scholar] [CrossRef]
- Moorman, D.E.; James, M.H.; Kilroy, E.A.; Aston-Jones, G. Orexin/hypocretin-1 receptor antagonism reduces ethanol self-administration and reinstatement selectively in highly-motivated rats. Brain Res. 2017, 1654, 34–42. [Google Scholar] [CrossRef]
- Gilpin, N.W.; Smith, A.D.; Cole, M.; Weiss, F.; Koob, G.F.; Richardson, H.N. Operant behavior and alcohol levels in blood and brain of alcohol-dependent rats. Alcohol. Clin. Exp. Res. 2009, 33, 2113–2123. [Google Scholar] [CrossRef]
- Fukunaga, K.; Muller, D.; Miyamoto, E. Increased phosphorylation of Ca2+/calmodulin-dependent protein kinase II and its endogenous substrates in the induction of long-term potentiation. J. Biol. Chem. 1995, 270, 6119–6124. [Google Scholar] [CrossRef]
- Lengyel, I.; Voss, K.; Cammarota, M.; Bradshaw, K.; Brent, V.; Murphy, K.P.S.J.; Bliss, T.V.P. Autonomous activity of CaMKII is only transiently increased following the induction of long-term potentiation in the rat hippocampus. Eur. J. Neurosci. 2004, 20, 3063–3072. [Google Scholar] [CrossRef]
- Colbran, R.J.; Brown, A.M. Calcium/calmodulin-dependent protein kinase II and synaptic plasticity. Curr. Opin. Neurobiol. 2004, 14, 318–327. [Google Scholar] [CrossRef]
- Zhao, B.; Wang, Y.; Li, Y.; Qiao, X.; Yan, P.; Zhu, Y.; Lai, J. Differential phosphorylation of NMDAR1-CaMKII-MAPKs in the rat nucleus accumbens following chronic ethanol exposure. Neurosci. Lett. 2015, 597, 60–65. [Google Scholar] [CrossRef]
- Salling, M.C.; Hodge, C.J.; Psilos, K.E.; Eastman, V.R.; Faccidomo, S.P.; Hodge, C.W. Cue-induced reinstatement of alcohol-seeking behavior is associated with increased CaMKII T286 phosphorylation in the reward pathway of mice. Pharmacol. Biochem. Behav. 2017, 163, 20–29. [Google Scholar] [CrossRef]
- Mijakowska, Z.; Łukasiewicz, K.; Ziółkowska, M.; Lipiński, M.; Trąbczyńska, A.; Matuszek, Ż.; Radwanska, K. Autophosphorylation of alpha isoform of calcium/calmodulin-dependent kinase II regulates alcohol addiction-related behaviors. Addict. Biol. 2017, 22, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Easton, A.C.; Lucchesi, W.; Lourdusamy, A.; Lenz, B.; Solati, J.; Golub, Y.; Moll, G.H. alphacamkii autophosphorylation controls the establishment of alcohol drinking behavior. Neuropsychopharmacology 2013, 38, 1636–1647. [Google Scholar] [CrossRef] [PubMed]
- Easton, A.C.; Lucchesi, W.; Mizuno, K.; Fernandes, C.; Schumann, G.; Giese, K.P.; Müller, C.P. AlphaCaMKII autophosphorylation controls the establishment of alcohol-induced conditioned place preference in mice. Behav. Brain Res. 2013, 252, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Wscieklica, T.; de Barros Viana, M.; Maluf, L.L.S.; Pouza, K.C.P.; Spadari, R.C.; Céspedes, I.C. Alcohol consumption increases locomotion in an open field and induces Fos-immunoreactivity in reward and approach/withdrawal-related neurocircuitries. Alcohol 2016, 50, 73–82. [Google Scholar] [CrossRef]
- Liu, W.; Crews, F.T. Adolescent intermittent ethanol exposure enhances ethanol activation of the nucleus accumbens while blunting the prefrontal cortex responses in adult rat. Neuroscience 2015, 293, 92–108. [Google Scholar] [CrossRef]
- Littleton, J.M.; Lovinger, D.; Liljequist, S.; Ticku, R.; Matsumoto, I.; Barron, S. Role of polyamines and NMDA receptors in ethanol dependence and withdrawal. Alcohol. Clin. Exp. Res. 2001, 25 (Suppl. 5 ISBRA), 132S–136S. [Google Scholar] [CrossRef]
- Chandrasekar, R. Alcohol and NMDA receptor: Current research and future direction. Front. Mol. Neurosci. 2013, 6, 14. [Google Scholar] [CrossRef]
- Chandler, L.J. Ethanol and brain plasticity: Receptors and molecular networks of the postsynaptic density as targets of ethanol. Pharmacol. Ther. 2003, 99, 311–326. [Google Scholar] [CrossRef]
- Hoffman, P.L. NMDA receptors in alcoholism. Int. Rev. Neurobiol. 2003, 56, 35–82. [Google Scholar]
- Suvarna, N.; Borgland, S.L.; Wang, J.; Phamluong, K.; Auberson, Y.P.; Bonci, A.; Ron, D. Ethanol alters trafficking and functional N-methyl-D-aspartate receptor NR2 subunit ratio via H-Ras. J. Biol. Chem. 2005, 280, 31450–31459. [Google Scholar] [CrossRef]
- Salter, W.M.; Kalia, L.V. Src kinases: A hub for NMDA receptor regulation. Nat. Rev. Neurosci. 2004, 5, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zheng, F.; Moon, C.; Schlüter, O.M.; Wang, H. Bi-directional regulation of CaMKIIalpha phosphorylation at Thr286 by NMDA receptors in cultured cortical neurons. J. Neurochem. 2012, 122, 295–307. [Google Scholar] [CrossRef] [PubMed]
- Somkuwar, S.S.; Fannon-Pavlich, M.J.; Ghofranian, A.; Quigley, J.A.; Dutta, R.R.; Galinato, M.H.; Mandyam, C.D. Wheel running reduces ethanol seeking by increasing neuronal activation and reducing oligodendroglial/neuroinflammatory factors in the medial prefrontal cortex. Brain Behav. Immun. 2016, 58, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Somkuwar, S.S.; Vendruscolo, L.F.; Fannon, M.J.; Schmeichel, B.E.; Nguyen, T.B.; Guevara, J.; Mandyam, C.D. Abstinence from prolonged ethanol exposure affects plasma corticosterone, glucocorticoid receptor signaling and stress-related behaviors. Psychoneuroendocrinology 2017, 84, 17–31. [Google Scholar] [CrossRef]
- Kim, A.; Zamora-Martinez, E.R.; Edwards, S.; Mandyam, C.D. Structural reorganization of pyramidal neurons in the medial prefrontal cortex of alcohol dependent rats is associated with altered glial plasticity. Brain Struct. Funct. 2015, 220, 1705–1720. [Google Scholar] [CrossRef]
- Galinato, M.H.; Orio, L.; Mandyam, C.D. Methamphetamine differentially affects BDNF and cell death factors in anatomically defined regions of the hippocampus. Neuroscience 2015, 286, 97–108. [Google Scholar] [CrossRef]
- Welinder, C.; Ekblad, L. Coomassie staining as loading control in Western blot analysis. J. Proteome Res. 2011, 10, 1416–1419. [Google Scholar] [CrossRef]
- Thacker, J.S.; Yeung, D.H.; Staines, W.R.; Mielke, J.G. Total protein or high-abundance protein: Which offers the best loading control for Western blotting? Anal. Biochem. 2016, 496, 76–78. [Google Scholar] [CrossRef]
- Gilpin, N.W.; Richardson, H.N.; Cole, M.; Koob, G.F. Vapor inhalation of alcohol in rats. Curr. Protoc. Neurosci. 2008, 44, 9–29. [Google Scholar] [CrossRef]
- Rivier, C.; Rivest, S.; Vale, W. Alcohol-induced inhibition of LH secretion in intact and gonadectomized male and female rats: Possible mechanisms. Alcohol. Clin. Exp. Res. 1992, 16, 935–941. [Google Scholar] [CrossRef]
- Erickson, C.K. Ethanol clearance in nine inbred rat strains. Alcohol. Clin. Exp. Res. 1984, 8, 491–494. [Google Scholar] [CrossRef] [PubMed]
- Bejanian, M.; Finn, D.A.; Syapin, P.J.; Alkana, R.L. Body temperature and ethanol pharmacokinetics in temperature-challenged mice. Alcohol 1990, 7, 331–337. [Google Scholar] [CrossRef]
- Finn, D.A.; Bejanian, M.; Jones, B.L.; McGivern, R.F.; Syapin, P.J.; Crabbe, J.C.; Alkana, R.L. Body temperature differentially affects ethanol sensitivity in both inbred strains and selected lines of mice. J. Pharmacol. Exp. Ther. 1990, 253, 1229–1235. [Google Scholar] [PubMed]
- Romm, E.; Collins, A.C. Body temperature influences on ethanol elimination rate. Alcohol 1987, 4, 189–198. [Google Scholar] [CrossRef]
- Richardson, H.N.; Lee, S.Y.; O’Dell, L.E.; Koob, G.F.; Rivier, C.L. Alcohol self-administration acutely stimulates the hypothalamic-pituitary-adrenal axis, but alcohol dependence leads to a dampened neuroendocrine state. Eur. J. Neurosci. 2008, 28, 1641–1653. [Google Scholar] [CrossRef] [PubMed]
- Vendruscolo, L.F.; Barbier, E.; Schlosburg, J.E.; Misra, K.K.; Whitfield, T.W., Jr.; Logrip, M.L.; Rivier, C.; Repunte-Canonigo, V.; Zorrilla, E.P.; Sanna, P.P.; et al. Corticosteroid-dependent plasticity mediates compulsive alcohol drinking in rats. J. Neurosci. 2012, 32, 7563–7571. [Google Scholar] [CrossRef]
- Vendruscolo, L.F.; Estey, D.; Goodell, V.; Macshane, L.G.; Logrip, M.L.; Schlosburg, J.E.; McGinn, M.A.; Zamora-Martinez, E.R.; Belanoff, J.K.; Hunt, H.J.; et al. Glucocorticoid receptor antagonism decreases alcohol seeking in alcohol-dependent individuals. J. Clin. Investig. 2015, 125, 3193–3197. [Google Scholar] [CrossRef]
- Rasmussen, D.D.; Boldt, B.M.; Bryant, C.A.; Mitton, D.R.; Larsen, S.A.; Wilkinson, C.W. Chronic daily ethanol and withdrawal: 1. Long-term changes in the hypothalamo-pituitary-adrenal axis. Alcohol. Clin. Exp. Res. 2000, 24, 1836–1849. [Google Scholar] [CrossRef]
- Engleman, E.A.; Ding, Z.M.; Oster, S.M.; Toalston, J.E.; Bell, R.L.; Murphy, J.M.; McBride, W.J.; Rodd, Z.A. Ethanol is self-administered into the nucleus accumbens shell, but not the core: Evidence of genetic sensitivity. Alcohol. Clin. Exp. Res. 2009, 33, 2162–2171. [Google Scholar] [CrossRef]
- Chaudhri, N.; Sahuque, L.L.; Schairer, W.W.; Janak, P.H. Separable roles of the nucleus accumbens core and shell in context- and cue-induced alcohol-seeking. Neuropsychopharmacology 2010, 35, 783–791. [Google Scholar] [CrossRef]
- Dayas, C.V.; Liu, X.; Simms, J.A.; Weiss, F. Distinct patterns of neural activation associated with ethanol seeking: Effects of naltrexone. Biol. Psychiatry 2007, 61, 979–989. [Google Scholar] [CrossRef] [PubMed]
- Hein, A.M.; Sridharan, A.; Nordeen, K.W.; Nordeen, E.J. Characterization of CaMKII-expressing neurons within a striatal region implicated in avian vocal learning. Brain Res. 2007, 1155, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Pasek, J.G.; Wang, X.; Colbran, R.J. Differential CaMKII regulation by voltage-gated calcium channels in the striatum. Mol. Cell Neurosci. 2015, 68, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Picconi, B.; Gardoni, F.; Centonze, D.; Mauceri, D.; Cenci, M.A.; Bernardi, G.; Calabresi, P.; Di Luca, M. Abnormal Ca2+-calmodulin-dependent protein kinase II function mediates synaptic and motor deficits in experimental parkinsonism. J. Neurosci. 2004, 24, 5283–5291. [Google Scholar] [CrossRef] [PubMed]
- Wanjerkhede, M.S.; Bapi, R.S. Role of CAMKII in reinforcement learning: A computational model of glutamate and dopamine signaling pathways. Biol. Cybern. 2011, 104, 397–424. [Google Scholar] [CrossRef] [PubMed]
- Yabuki, Y.; Nakagawasai, O.; Moriguchi, S.; Shioda, N.; Onogi, H.; Tan-No, K.; Tadano, T.; Fukunaga, K. Decreased CaMKII and PKC activities in specific brain regions are associated with cognitive impairment in neonatal ventral hippocampus-lesioned rats. Neuroscience 2013, 234, 103–115. [Google Scholar] [CrossRef]
- Hayashi, Y.; Nishio, M.; Naito, Y.; Yokokura, H.; Nimura, Y.; Hidaka, H.; Watanabe, Y. Regulation of neuronal nitric-oxide synthase by calmodulin kinases. J. Biol. Chem. 1999, 274, 20597–20602. [Google Scholar] [CrossRef]
- Steiner, P.; Higley, M.J.; Xu, W.; Czervionke, B.L.; Malenka, R.C.; Sabatini, B.L. Destabilization of the postsynaptic density by PSD-95 serine 73 phosphorylation inhibits spine growth and synaptic plasticity. Neuron 2008, 60, 788–802. [Google Scholar] [CrossRef]
- Agoglia, A.E.; Holstein, S.E.; Reid, G.; Hodge, C.W. CaMKIIalpha-GluA1 activity underlies vulnerability to adolescent binge alcohol drinking. Alcohol. Clin. Exp. Res. 2015, 39, 1680–1690. [Google Scholar] [CrossRef]
- Cannady, R.; Fisher, K.R.; Graham, C.; Crayle, J.; Besheer, J.; Hodge, C.W. Potentiation of amygdala AMPA receptor activity selectively promotes escalated alcohol self-administration in a CaMKII-dependent manner. Addict. Biol. 2017, 22, 652–664. [Google Scholar] [CrossRef]
- Wang, Y.; Cui, H.; Wang, W.; Zhao, B.; Lai, J. The region-specific activation of Ca2+/calmodulin dependent protein kinase II and extracellular signal-regulated kinases in hippocampus following chronic alcohol exposure. Brain Res. Bull. 2012, 89, 191–196. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Somkuwar, S.S.; Mandyam, C.D. Individual Differences in Ethanol Drinking and Seeking Behaviors in Rats Exposed to Chronic Intermittent Ethanol Vapor Exposure is Associated with Altered CaMKII Autophosphorylation in the Nucleus Accumbens Shell. Brain Sci. 2019, 9, 367. https://doi.org/10.3390/brainsci9120367
Somkuwar SS, Mandyam CD. Individual Differences in Ethanol Drinking and Seeking Behaviors in Rats Exposed to Chronic Intermittent Ethanol Vapor Exposure is Associated with Altered CaMKII Autophosphorylation in the Nucleus Accumbens Shell. Brain Sciences. 2019; 9(12):367. https://doi.org/10.3390/brainsci9120367
Chicago/Turabian StyleSomkuwar, Sucharita S., and Chitra D. Mandyam. 2019. "Individual Differences in Ethanol Drinking and Seeking Behaviors in Rats Exposed to Chronic Intermittent Ethanol Vapor Exposure is Associated with Altered CaMKII Autophosphorylation in the Nucleus Accumbens Shell" Brain Sciences 9, no. 12: 367. https://doi.org/10.3390/brainsci9120367
APA StyleSomkuwar, S. S., & Mandyam, C. D. (2019). Individual Differences in Ethanol Drinking and Seeking Behaviors in Rats Exposed to Chronic Intermittent Ethanol Vapor Exposure is Associated with Altered CaMKII Autophosphorylation in the Nucleus Accumbens Shell. Brain Sciences, 9(12), 367. https://doi.org/10.3390/brainsci9120367




