Abstract
Objective: Providing melatonin in animal models with spontaneous intracerebral hemorrhage (SIH) has been associated with beneficial effects. However, to our knowledge, there are no published data on circulating melatonin levels regarding the prognosis of SIH patients. Therefore, the objectives of this study were to determine whether serum melatonin levels in SIH patients were associated with early mortality and whether they could be used as prognostic biomarkers. Methods: This observational and prospective study included patients with supratentorial and clinically severe SIH (defined as Glasgow Coma Scale GCS <9) admitted to the Intensive Care Units of six Spanish hospitals. Serum melatonin levels were determined at the time of severe SIH diagnosis. Mortality at 30 days was the study end-point. Results: Non-surviving patients (n = 46) showed higher serum melatonin levels (p < 0.001) than surviving (n = 54) patients. An area under the curve was found for the prediction of 30-day mortality by serum melatonin levels of 0.89 (95% CI = 0.81–0.94; p < 0.001). Multiple logistic regression analysis showed an association of serum melatonin levels with 30-day mortality (Odds Ratio = 8.16; 95% CI = 2.30–28.95; p = 0.001) after controlling for midline shift, glycemia, early evacuation of SIH, and Intracerebral hemorrhage (ICH) score. Conclusions: The novel findings by our study were the presence of higher serum melatonin levels in non-surviving patients than in surviving patients and the association of these levels with mortality.
1. Introduction
A large number of deaths and disabilities as well as the need for a high consumption of care resources are caused by spontaneous intracerebral hemorrhage (SIH) [1]. Oxidative stress has been implicated in the secondary brain injury of SIH [2,3,4].
Melatonin, a lipophilic amino acid derived from tryptophan, is synthesized in the pineal gland and also on other organs such as the retina, thymus, bone marrow, and gastrointestinal tract [5,6]. Melatonin has a role in sleep regulation, shows antioxidant effects due to upregulation of some antioxidant enzymes, and is a potent reactive oxygen species scavenger [7,8]. The administration of melatonin in animal models with SIH has been associated with beneficial effects, such as the reduction of oxidative damage and motor dysfunction [9,10]. Previously, we found a higher oxidative state in non-survivor SIH patients than in survivor patients, as assessed by serum concentrations of malondialdehyde [11] (biomarker of lipid peroxidation) [12,13]. However, to our knowledge, there are no published data about circulating melatonin levels regarding the prognosis of SIH patients. Therefore, the objectives of this study were to determine whether serum melatonin levels in SIH patients were associated with early mortality and whether they could be used as prognostic biomarkers.
2. Methods
2.1. Design and Subjects
This observational and prospective study was made with the approval by the Review Board of the six Spanish hospitals participating: H. Clínico Universitario de Valencia, Universitario Nuestra Señora de Candelaria (Santa Cruz de Tenerife), H. General de La Palma, H. Universitario Dr. Negrín (Las Palmas de Gran Canaria), H. Universitario de Canarias (San Cristóbal de La Laguna), and H. Insular de Las Palmas de Gran Canaria. Written and signed informed consent was obtained by a family member of each patient.
Patients with supratentorial SIH and clinically severe (defined as Glasgow Coma Scale (GCS) [14] <9) admitted to Intensive Care Units were included in the study. Patients with infratentorial hemorrhage, traumatic hemorrhage, hemorrhagic transformation of brain infarction, age <18 years, pregnancy, inflammatory disease, or malignant disease were excluded from the study.
2.2. Variables Recorded
Epidemiological variables such as age and sex were collected. Blood variables were also collected such as the international normalized ratio (INR), activated partial thromboplastin time (aPTT), platelets, fibrinogen, sodium, lactic acid, creatinine, glycemia, and the arterial oxygen pressure/fraction of inspired oxygen ratio (PaO2/FIO2). The following scores were recorded: the Acute Physiology and Chronic Health Evaluation II (APACHE II) score [15], GCS, and the intracerebral hemorrhage (ICH) score [16]. Additionally, the site, volume, and cause of SIH, as well as the presence of intraventricular hemorrhage, hydrocephalus, transtentorial herniation, or midline shift were also recorded. In addition, we registered early SIH evacuation (within first 24 hours of SIH diagnosis), and mortality at 30 days (which was considered the study end-point).
2.3. Blood Samples and Determination of Serum Melatonin Levels
Serum samples at moment of severe SIH diagnosis were taken and frozen at −80 °C until its analysis in the Physiology Department at Faculty of Medicine of University of La Laguna (Tenerife, Spain). Serum melatonin determinations were made using a kit from Immuno Biological Laboratories (IBL Hamburg GmbH, Hamburg, Germany). The assay had a detection limit of 0.13 pg/mL, an intra-assay variation coefficient of 6.4%, and an inter-assay variation coefficient of 11.1%.
2.4. Statistical Methods
Frequencies and percentages were used to report categorical variables, and medians and interquartile ranges were used to report continuous variables. The Chi-square test was used to compare categorical variables between surviving and non-surviving patients, and the Wilcoxon-Mann-Whitney test was used to compare continuous variables. The prediction capacity of mortality at 30 days involving serum melatonin levels was estimated by performing a receiver operating characteristic (ROC) analysis. To determine the association of serum melatonin levels with 30 day-mortality, a multiple logistic regression analysis was performed controlling for midline shift, glycemia, and early evacuation of SIH and ICH scores; while an odds ratio and its 95% confidence intervals (CI) were used to estimate the clinical impact of the variables included in the analysis. Kaplan-Meier curves were constructed using serum melatonin levels > 3.94 pg/mL (because it was the optimal cut-off according to the Youden J index) and mortality at 30 days. To do the statistical analyses, the programs NCSS 2000 (Kaysville, UT, USA), LogXact 4.1 (Cytel Co., Cambridge, MA), and SPSS 17.0 (SPSS Inc., Chicago, IL, USA) were used. p-values < 0.05 were considered statistically significant.
3. Results
Non-surviving (n = 46) patients showed higher glycemia (p = 0.01), older age (p = 0.006), ICH score (p < 0.001), APACHE-II score (p < 0.001), midline shift (p = 0.005) and intracerebral hemorrhage volume (p = 0.02), and lower GCS (p < 0.001) than surviving SIH patients (n = 54). In addition, non-surviving patients had higher serum melatonin levels than surviving patients (p < 0.001) (Table 1).

Table 1.
Clinical and biochemical characteristics of 30-day surviving and non-surviving patients with spontaneous intracerebral hemorrhage (SIH).
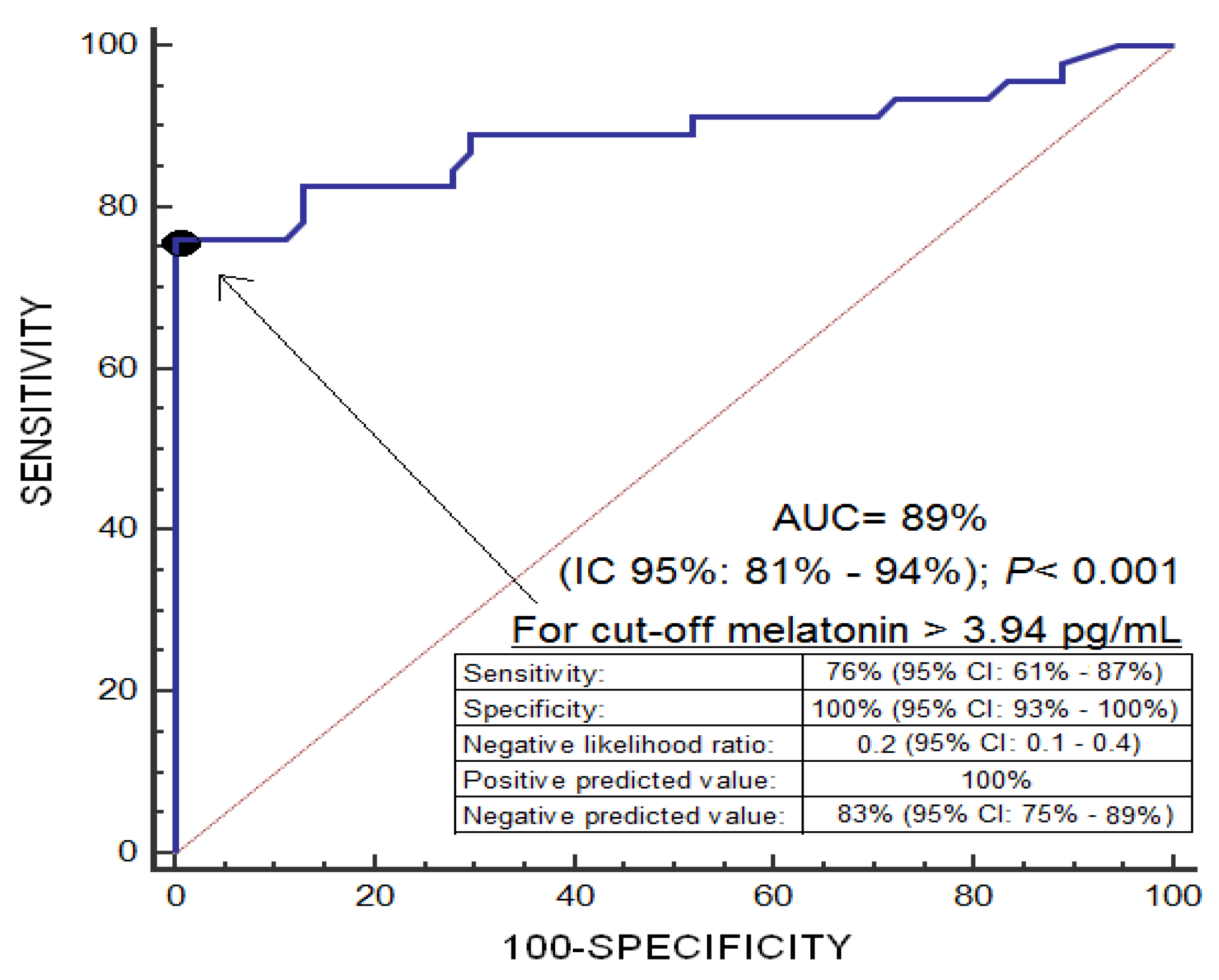
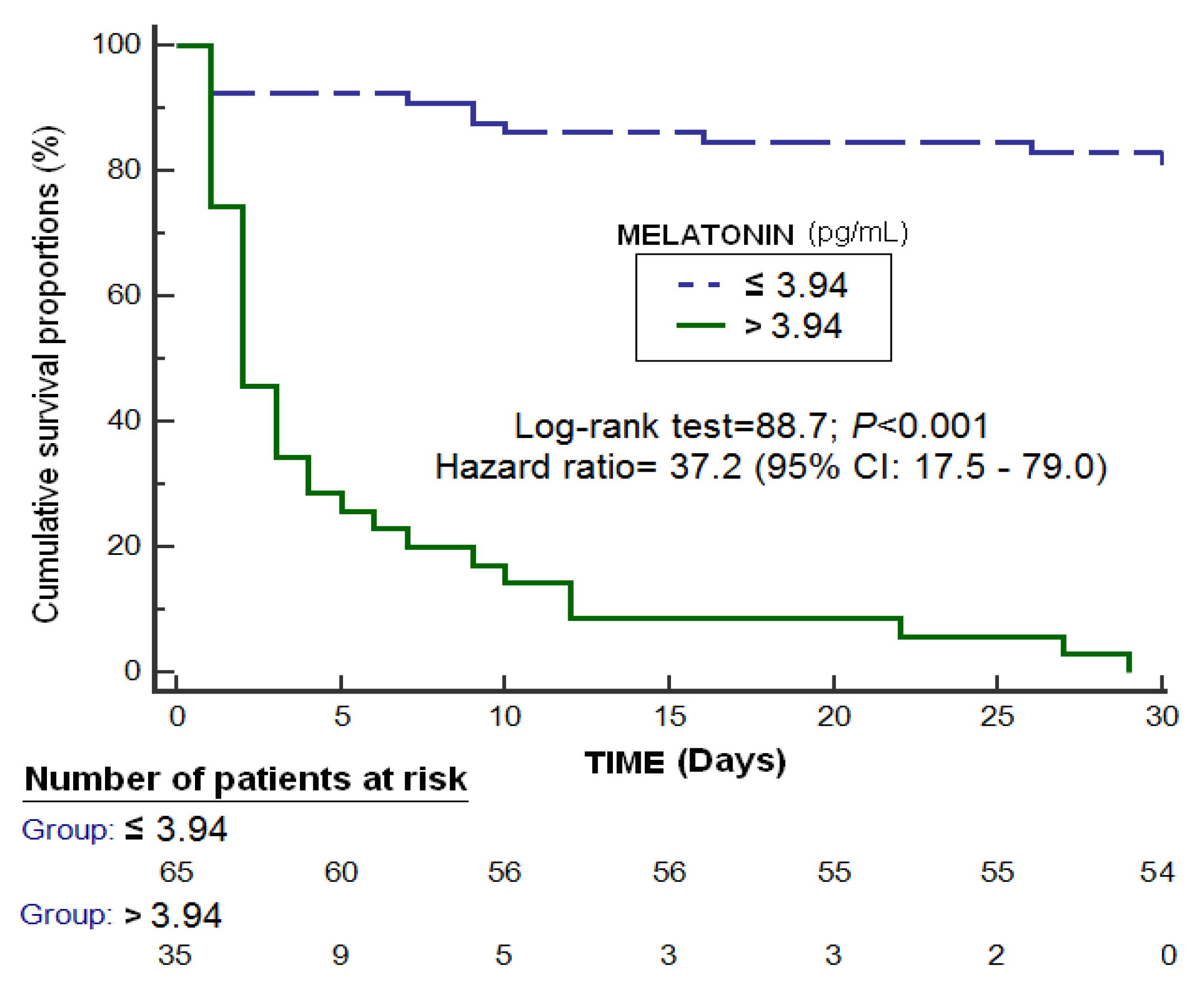
An area under the curve was found for the prediction of 30-day mortality by serum melatonin levels of 0.89 (95% CI = 0.81–0.94; p < 0.001) (Figure 1). Kaplan-Meier analysis showed higher mortality at 30 days in patients with serum melatonin levels > 3.94 pg/mL (Hazard ratio = 37.2; 95% CI = 17.5–79.0; p < 0.001) (Figure 2). Multiple logistic regression analysis showed an association of serum melatonin levels with 30-day mortality after controlling for midline shift, glycemia, early evacuation of SIH, and the ICH score (Odds Ratio = 8.16; 95% CI = 2.30–28.95; p = 0.001), and after controlling for age, GCS, volume of SIH and intraventricular hemorrhage (Odds Ratio = 7.75; 95% CI = 2.05–29.27; p = 0.003) (Table 2). A positive association between serum levels of melatonin and malondialdehyde was found (rho = 0.43; p < 0.001); but not between serum melatonin levels with age (rho = 0.19; p = 0.06) and the SIH volume (rho = 0.11; p = 0.31).
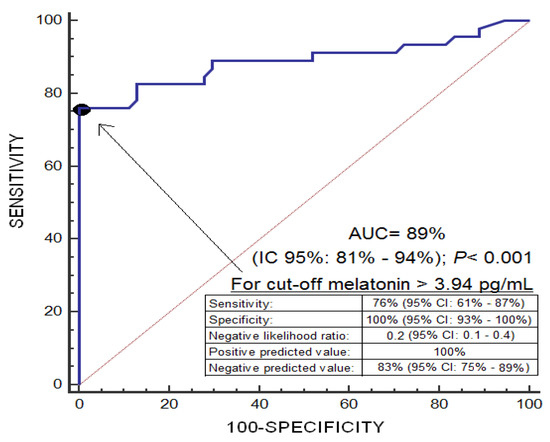
Figure 1.
Receiver operation characteristic analysis using serum melatonin levels as a predictor of mortality at 30 days.
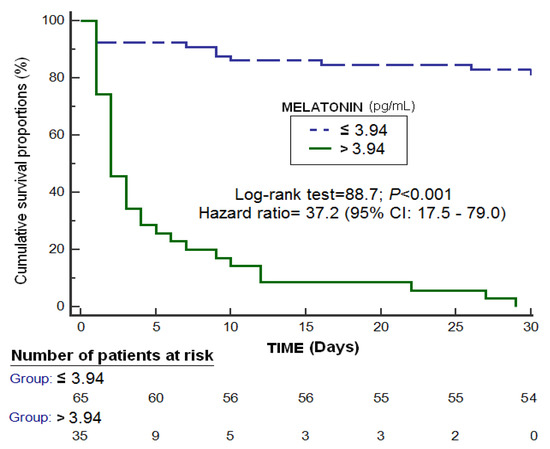
Figure 2.
Survival curves at 30 days using serum melatonin levels >3.94 pg/mL as the cut-off.

Table 2.
Logistic regression analysis to predict 30-day mortality.
4. Discussion
To our knowledge, our study showed serum melatonin levels in SIH patients for first time. The novel findings of our study were the presence of higher serum melatonin levels in non-surviving than in surviving patients and the association of those levels with lipid oxidation and mortality. Two regression models with five variables were constructed to avoid an over-fitting effect because 46 non-survivor patients were recorded in our study. The variables included in the first model were serum melatonin levels, glycemia, early SIH evacuation, ICH score, and midline shift, and the variables associated with mortality were the serum melatonin levels and midline shift. The variables included in the first model were age, volume of SIH, GCS, and intraventricular haemorrhage because those variables are included in the ICH score. This model also included serum melatonin levels, and the variables associated with mortality were serum melatonin levels, age and GCS. Our findings are consistent with the findings from previous studies showing an association between high serum melatonin concentrations and mortality in patients with traumatic brain injury [17] or brain infarction [18].
Previously, serum malondialdehyde were determined in these patients and higher levels were found in non-surviving patients than in surviving patients [11]. In our current study, a positive association was found between circulating levels of melatonin and malondialdehyde. Malondialdehyde is a lipid peroxidation product that appears due to oxidation of phospholipids from cell membranes, which is released to extracellular space and finally appears in blood [12,13]. In addition, this association between high circulating malondialdehyde levels and mortality has been found in patients with traumatic brain injury [19] or cerebral infarction [20].
Another interesting point is that the administration of melatonin in SIH animal models have reduced oxidative damage [21,22,23,24,25] and motor deficit [21]. We believe that higher serum levels of melatonin and malondialdehyde in non-surviving patients compared to surviving SIH patients, and the association between serum levels of melatonin and malondialdehyde could mean that the non-survivors have higher oxidants species production, which is the origin of a higher oxidative damage (with higher serum malondialdehyde levels) and higher serum melatonin levels (attempting to maintain a balance between antioxidant and oxidant states). However, those higher serum melatonin levels in non-surviving patients are not able to compensate for the high production of oxidant species, and they contribute to brain damage and ultimately to patient death.
We must recognize some limitations of our study, such as the fact that we have not assessed circulating melatonin levels during the evolution of the patient and other oxidative damage products. In addition, we have not determined blood melatonin levels in healthy subjects; although the levels found in other series were 15.15 ± 1.65 pg/mL [26]. However, we believe that our findings in SIH patients could motivate research on melatonin in SIH patients.
5. Conclusions
The novel findings of our study were the presence of higher serum melatonin levels in non-surviving patients than in surviving patients and the association of these levels with mortality.
Author Contributions
L.L. was responsible of conceive, design and coordinate the study, made substantial contributions to acquisition of data, analysis and interpretation of data, and drafted the manuscript. M.M.M., L.R., M.A., J.S.-V., J.J.C., and V.G.-M. have made substantial contributions to acquisition of data and provided useful suggestions. P.A.-G. determined blood concentrations. A.J. have made substantial contributions to analysis and interpretation of data. All authors read critically and approved the manuscript, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved
Funding
This study was supported by a grant (OA18/011) from Fundación DISA a la Investigación Médica 2017 (Santa Cruz de Tenerife. Spain) and by a grant from Grupo de Expertos Neurológicos de Canarias (GEN-Canarias. Santa Cruz de Tenerife. Spain). Funding did not influence in the study design, the collection, analysis, and interpretation of data, the manuscript writing, and the decision to submit it for publication.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Hemphill, J.C., 3rd; Greenberg, S.M.; Anderson, C.S.; Becker, K.; Bendok, B.R.; Cushman, M.; Fung, G.L.; Goldstein, J.N.; Macdonald, R.L.; Mitchell, P.H.; et al. American Heart Association Stroke Council; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology. Guidelines for the Management of Spontaneous Intracerebral Hemorrhage: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association. Stroke 2015, 46, 2032–2060. [Google Scholar]
- Aronowski, J.; Zhao, X. Molecular pathophysiology of cerebral hemorrhage: Secondary brain injury. Stroke 2011, 42, 1781–1786. [Google Scholar] [CrossRef] [PubMed]
- Aronowski, J.; Hall, C.E. New horizons for primary intracerebral hemorrhage treatment: Experience from preclinical studies. Neurol. Res. 2005, 27, 268–279. [Google Scholar] [CrossRef] [PubMed]
- Katsuki, H. Exploring neuroprotective drug therapies for intracerebral hemorrhage. J. Pharmacol. Sci. 2010, 114, 366–378. [Google Scholar] [CrossRef] [PubMed]
- Cagnacci, A. Melatonin in relation to physiology in adult humans. J. Pineal Res. 1996, 21, 200–213. [Google Scholar] [CrossRef] [PubMed]
- Dawson, D.; Encel, N. Melatonin and sleep in humans. J. Pineal Res. 1993, 15, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.X.; Reiter, R.J. Melatonin as a natural ally against oxidative stress: A physicochemical examination. J. Pineal Res. 2011, 51, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Paredes, S.D.; Manchester, L.C.; Tan, D.X. Reducing oxidative/nitrosative stress: A newly-discovered genre for melatonin. Crit. Rev. Biochem. Mol. Biol. 2009, 44, 175–200. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.J.; Wu, C.; Niu, H.J.; Wang, K.; Mo, L.J.; Shao, A.W.; Dixon, B.J.; Zhang, J.M.; Yang, S.X.; Wang, Y.R. Neuroprotective Mechanisms of Melatonin in Hemorrhagic Stroke. Cell. Mol. Neurobiol. 2017, 37, 1173–1185. [Google Scholar] [CrossRef]
- Katsuki, H.; Hijioka, M. Intracerebral Hemorrhage as an Axonal Tract Injury Disorder with Inflammatory Reactions. Biol. Pharm. Bull. 2017, 40, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Lorente, L.; Martín, M.M.; Abreu-González, P.; Sabatel, R.; Ramos, L.; Argueso, M.; Solé-Violán, J.; Riaño-Ruiz, M.; Jiménez, A.; García-Marín, V. Serum Malondialdehyde Levels and Mortality in Patients with Spontaneous Intracerebral Hemorrhage. World Neurosurg. 2018, 113, e542–e547. [Google Scholar] [CrossRef] [PubMed]
- Draper, H.H.; Hadley, M. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 1990, 186, 421–431. [Google Scholar]
- Dalle-Donne, I.; Rossi, R.; Colombo, R.; Giustarini, D.; Milzani, A. Biomarkers of oxidative damage in human disease. Clin. Chem. 2006, 52, 601–623. [Google Scholar] [CrossRef] [PubMed]
- Teasdale, G.; Jennett, B. Assessment of coma and impaired consciousness. A practical scale. Lancet 1974, 2, 81–84. [Google Scholar] [CrossRef]
- Knaus, W.A.; Draper, E.A.; Wagner, D.P.; Zimmerman, J.E. APACHE II: A severity of disease classification system. Crit. Care Med. 1985, 13, 818–829. [Google Scholar] [CrossRef]
- Hemphill, J.C., 3rd; Bonovich, D.C.; Besmertis, L.; Manley, G.T.; Johnston, S.C. The ICH score: A simple, reliable grading scale for intracerebral hemorrhage. Stroke 2001, 32, 891–897. [Google Scholar] [CrossRef] [PubMed]
- Lorente, L.; Martín, M.M.; Abreu-González, P.; Pérez-Cejas, A.; Ramos, L.; Argueso, M.; Solé-Violán, J.; Cáceres, J.J.; Jiménez, A.; García-Marín, V. Serum melatonin levels in survivor and non-survivor patients with traumatic brain injury. BMC Neurol. 2017, 17, 138. [Google Scholar] [CrossRef]
- Lorente, L.; Martín, M.M.; Abreu-González, P.; Pérez-Cejas, A.; Ramos, L.; Argueso, M.; Solé-Violán, J.; Cáceres, J.J.; Jiménez, A.; García-Marín, V. Serum melatonin levels are associated with mortality in patients with malignant middle cerebral artery infarction. J. Int. Med. Res. 2018, 46, 3268–3277. [Google Scholar] [CrossRef]
- Lorente, L.; Martín, M.M.; Abreu-González, P.; Ramos, L.; Argueso, M.; Cáceres, J.J.; Solé-Violán, J.; Lorenzo, J.M.; Molina, I.; Jiménez, A. Association between serum malondialdehyde levels and mortality in patients with severe brain trauma injury. J. Neurotrauma 2015, 32, 1–6. [Google Scholar] [CrossRef]
- Lorente, L.; Martín, M.M.; Abreu-González, P.; Ramos, L.; Argueso, M.; Solé-Violán, J.; Riaño-Ruiz, M.; Jiménez, A. Serum malondialdehyde levels in patients with malignant middle cerebral artery infarction are associated with mortality. PLoS ONE 2015, 10, e0125893. [Google Scholar] [CrossRef]
- Ueda, Y.; Masuda, T.; Ishida, A.; Misumi, S.; Shimizu, Y.; Jung, C.G.; Hida, H. Enhanced electrical responsiveness in the cerebral cortex with oral melatonin administration after a small hemorrhage near the internal capsule in rats. J. Neurosci. Res. 2014, 92, 1499–1508. [Google Scholar] [CrossRef] [PubMed]
- Lekic, T.; Hartman, R.; Rojas, H.; Manaenko, A.; Chen, W.; Ayer, R.; Tang, J.; Zhang, J.H. Protective effect of melatonin upon neuropathology, striatal function, and memory ability after intracerebral hemorrhage in rats. J. Neurotrauma 2010, 27, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.Q.; Liang, G.B.; Xue, Y.X.; Liu, Y.H. Effects of combination treatment of dexamethasone and melatonin on brain injury in intracerebral hemorrhage model in rats. Brain Res. 2009, 1264, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Rojas, H.; Lekic, T.; Chen, W.; Jadhav, V.; Titova, E.; Martin, R.D.; Tang, J.; Zhang, J. The antioxidant effects of melatonin after intracerebral hemorrhage in rats. Acta Neurochir. Suppl. 2008, 105, 19–21. [Google Scholar] [PubMed]
- Wang, Z.; Zhou, F.; Dou, Y.; Tian, X.; Liu, C.; Li, H.; Shen, H.; Chen, G. Melatonin Alleviates Intracerebral Hemorrhage-Induced Secondary Brain Injury in Rats via Suppressing Apoptosis, Inflammation, Oxidative Stress, DNA Damage, and Mitochondria Injury. Transl. Stroke Res. 2018, 9, 74–91. [Google Scholar] [CrossRef]
- Seifman, M.A.; Gomes, K.; Nguyen, P.N.; Bailey, M.; Rosenfeld, J.V.; Cooper, D.J.; Morganti-Kossmann, M.C. Measurement of serum melatonin in intensive care unit patients: Changes in traumatic brain injury, trauma, and medical conditions. Front. Neurol. 2014, 5, 237. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).