Exercise Strengthens Central Nervous System Modulation of Pain in Fibromyalgia
Abstract
:1. Introduction
2. Experimental Section
2.1. Participants
2.2. Procedures
2.2.1. Condition Descriptions
2.2.2. fMRI Procedures
2.3. Data Processing and Analyses
2.3.1. Corrections for Physiological Noise
2.3.2. Group Level Analyses
3. Results and Discussion
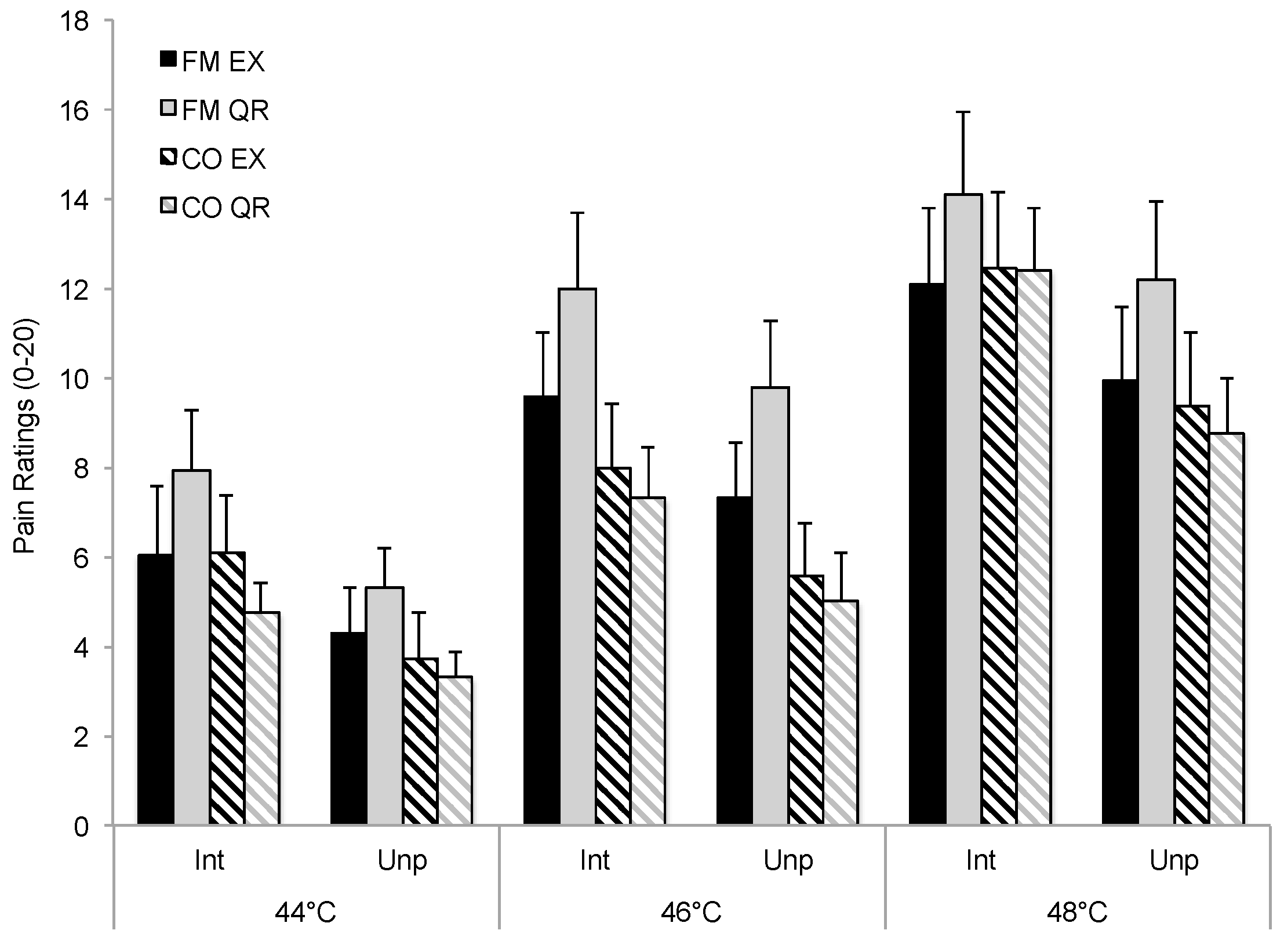
3.1. Influence of Exercise on Pain Ratings and Symptoms
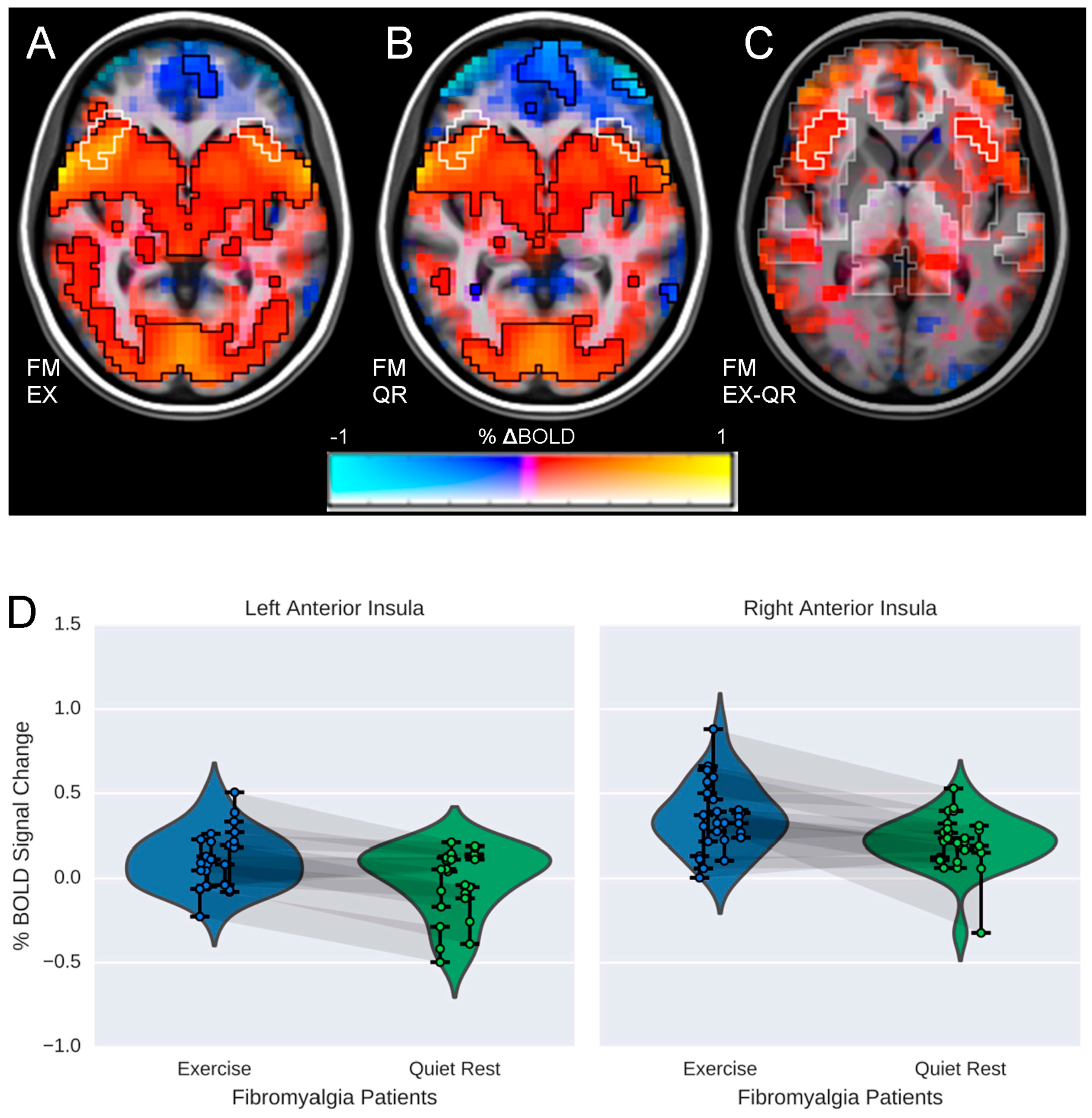
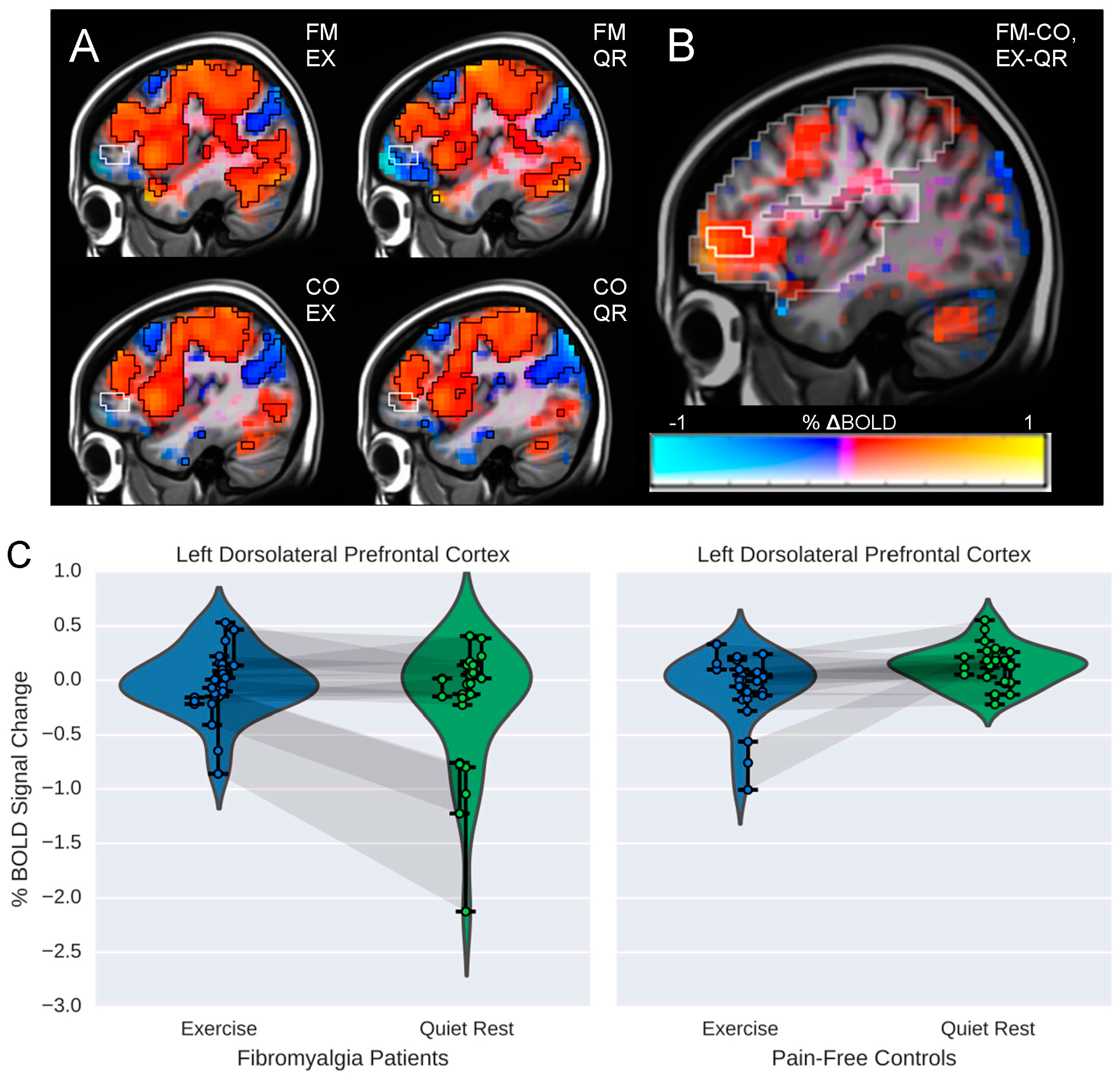
3.2. Influence of Exercise on Brain Processing of Pain
3.3. Relationship between Brain Responses and Pain Ratings
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Disclaimer
References
- Gracely, R.H.; Ambrose, K.R. Neuroimaging of fibromyalgia. Best Pract. Res. Clin. Rheumatol. 2011, 25, 271–284. [Google Scholar] [CrossRef] [PubMed]
- Ossipov, M.H.; Morimura, K.; Porreca, F. Descending pain modulation and chronification of pain. Curr. Opin. Support. Palliat. Care 2014, 8, 143–151. [Google Scholar] [PubMed]
- Koltyn, K.F. Analgesia following exercise: A review. Sports Med. 2000, 29, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Bidonde, J.; Busch, A.J.; Bath, B.; Milosavljevic, S. Exercise for Adults with Fibromyalgia: An Umbrella Systematic Review with Synthesis of Best Evidence. Curr. Rheumatol. Rev. 2014, 10, 45–79. [Google Scholar] [CrossRef] [PubMed]
- Newcomb, L.W.; Koltyn, K.F.; Morgan, W.P.; Cook, D.B. Influence of preferred versus prescribed exercise on pain in fibromyalgia. Med. Sci. Sports Exerc. 2011, 43, 1106–1113. [Google Scholar] [CrossRef] [PubMed]
- Nijs, J.; Kosek, E.; Van Oosterwijck, J.; Meeus, M. Dysfunctional endogenous analgesia during exercise in patients with chronic pain: To exercise or not to exercise? Pain Physician 2012, 15, ES205–ES213. [Google Scholar] [PubMed]
- Physician’s Desk Reference, 65th ed.; Thomson Press: Montvale, NJ, USA, 2011.
- Burckhardt, C.S.; Clark, S.R.; Bennett, R.M. The fibromyalgia impact questionnaire: Development and validation. J. Rheumatol. 1991, 18, 728–733. [Google Scholar] [PubMed]
- Melzack, R. The short-form McGill Pain Questionnaire. Pain 1987, 30, 191–197. [Google Scholar] [CrossRef]
- Borg, G. Borg’s Perceived Exertion and Pain Scales; Human Kinetics: Champaign, IL, USA, 1998. [Google Scholar]
- Gracely, R.H.; McGrath, F.; Dubner, R. Ratio scales of sensory and affective verbal pain descriptors. Pain 1978, 5, 5–18. [Google Scholar] [CrossRef]
- Freedson, P.S.; Melanson, E.; Sirard, J. Calibration of the Computer Science and Applications, Inc. accelerometer. Med. Sci. Sports Exerc. 1998, 30, 777–781. [Google Scholar] [CrossRef] [PubMed]
- Matthew, C.E. Calibration of accelerometer output for adults. Med. Sci. Sports Exerc. 2005, 37, S512–S522. [Google Scholar] [CrossRef] [PubMed]
- Cox, R.W. AFNI: Software for Analysis and Visualization of Functional Magnetic Resonance Neuroimages. Comput. Biomed. Res. 1996, 29, 162–173. [Google Scholar] [CrossRef] [PubMed]
- AFNI. Available online: https://afni.nimh.nih.gov/ (accessed on 24 February 2016).
- Mazziotta, J.C.; Toga, A.W.; Evans, A.; Fox, P.; Lancaster, J. A probabilistic atlas of the human brain: Theory and rationale for its development. The International Consortium for Brain Mapping (ICBM). NeuroImage 1995, 2, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Beall, E.B. Adaptive cyclic physiologic noise modeling and correction in functional MRI. J. Neurosci. Methods 2010, 187, 216–228. [Google Scholar] [CrossRef] [PubMed]
- Beall, E.B.; Lowe, M.J. Isolating physiologic noise sources with independently determined spatial measures. NeuroImage 2007, 37, 1286–1300. [Google Scholar] [CrossRef] [PubMed]
- Birn, R.M.; Smith, M.A.; Jones, T.B.; Bandettini, P.A. The Respiration Response Function: The temporal dynamics of fMRI signal fluctuations related to changes in respiration. NeuroImage 2008, 40, 644–654. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Cunningham, J.P.; Glover, G.H. Influence of heart rate on the BOLD signal: The cardiac response function. NeuroImage 2009, 44, 857–869. [Google Scholar] [CrossRef] [PubMed]
- Tracey, I.; Mantyh, P.W. The cerebral signature for pain perception and its modulation. Neuron 2007, 55, 377–391. [Google Scholar] [CrossRef] [PubMed]
- Cook, D.B.; Stegner, A.J.; McLoughlin, M.J. Imaging pain of fibromyalgia. Curr. Pain Headache Rep. 2007, 11, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Wiech, K.; Lin, C.; Brodersen, K.H.; Bingel, U.; Ploner, M.; Tracey, I. Anterior Insula Integrates Information about Salience into Perceptual Decisions about Pain. J. Neurosci. 2010, 30, 16324–16331. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, J.; Minoshima, S.; Casey, K.L. Keeping pain out of mind: The role of the dorsolateral prefrontal cortex in pain modulation. Brain J. Neurol. 2003, 126, 1079–1091. [Google Scholar] [CrossRef]
- Ellingson, L.D.; Shields, M.R.; Stegner, A.J.; Cook, D.B. Physical activity, sustained sedentary behavior, and pain modulation in women with fibromyalgia. J. Pain 2012, 13, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Kadetoff, D.; Kosek, E. The effects of static muscular contraction on blood pressure, heart rate, pain ratings and pressure pain thresholds in healthy individuals and patients with fibromyalgia. Eur. J. Pain Lond. Engl. 2007, 11, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Staud, R.; Robinson, M.E.; Price, D.D. Isometric exercise has opposite effects on central pain mechanisms in fibromyalgia patients compared to normal controls. Pain 2005, 118, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Kosek, E.; Ekholm, J.; Hansson, P. Modulation of pressure pain thresholds during and following isometric contraction in patients with fibromyalgia and in healthy controls. Pain 1996, 64, 415–423. [Google Scholar] [CrossRef]
- Hoeger Bement, M.K.; Weyer, A.; Hartley, S.; Drewek, B.; Harkins, A.L.; Hunter, S.K. Pain perception after isometric exercise in women with fibromyalgia. Arch. Phys. Med. Rehabil. 2011, 92, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.D.; Booth, J.; Taylor, J.L.; Barry, B.K. Aerobic training increases pain tolerance in healthy individuals. Med. Sci. Sports Exerc. 2014, 46, 1640–1647. [Google Scholar] [CrossRef] [PubMed]
- Ellingson, L.D.; Colbert, L.H.; Cook, D.B. Physical activity is related to pain sensitivity in healthy women. Med. Sci. Sports Exerc. 2012, 44, 1401–1406. [Google Scholar] [CrossRef] [PubMed]
- Umeda, M.; Corbin, L.W.; Maluf, K.S. Examination of contraction-induced muscle pain as a behavioral correlate of physical activity in women with and without fibromyalgia. Disabil. Rehabil. 2015, 37, 1864–1869. [Google Scholar] [CrossRef] [PubMed]
- Bingel, U.; Lorenz, J.; Glauche, V.; Knab, R.; Gläscher, J.; Weiller, C.; Büchel, C. Somatotopic organization of human somatosensory cortices for pain: A single trial fMRI study. NeuroImage 2004, 23, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, Y.; Kan, S.; Uematsu, H.; Shibata, M.; Fujino, Y. Pain-Related Brain Activity Evoked by Active and Dynamic Arm Movement: Delayed-Onset Muscle Soreness as a Promising Model for Studying Movement-Related Pain in Humans. Pain Med. Malden Mass 2015, 16, 1528–1539. [Google Scholar] [CrossRef] [PubMed]
- Koltyn, K.F. Exercise-induced hypoalgesia and intensity of exercise. Sports Med. 2002, 32, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Koltyn, D.K.F.; Umeda, M. Exercise, Hypoalgesia and Blood Pressure. Sports Med. 2012, 36, 207–214. [Google Scholar] [CrossRef]
- Ruiz, J.R.; Segura-Jiménez, V.; Ortega, F.B.; Álvarez-Gallardo, I.C.; Camiletti-Moirón, D.; Aparicio, V.A.; Carbonell-Baeza, A.; Femia, P.; Munguía-Izquierdo, D.; Delgado-Fernández, M. Objectively measured sedentary time and physical activity in women with fibromyalgia: A cross-sectional study. BMJ Open 2013, 3, e002722. [Google Scholar] [CrossRef] [PubMed]
- Segura-Jiménez, V.; Álvarez-Gallardo, I.C.; Estévez-López, F.; Soriano-Maldonado, A.; Delgado-Fernández, M.; Ortega, F.B.; Aparicio, V.A.; Carbonell-Baeza, A.; Mota, J.; Silva, P.; et al. Differences in sedentary time and physical activity between women with fibromyalgia and healthy controls: The al-Ándalus project. Arthritis Rheumatol. 2015, 67, 3047–3057. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.C.; Nielson, K.A.; Antuono, P.; Lyons, J.-A.; Hanson, R.J.; Butts, A.M.; Hantke, N.C.; Verber, M.D. Semantic memory functional MRI and cognitive function after exercise intervention in mild cognitive impairment. J. Alzheimers Dis. 2013, 37, 197–215. [Google Scholar] [PubMed]
- Smith, J.C.; Nielson, K.A.; Woodard, J.L.; Seidenberg, M.; Durgerian, S.; Hazlett, K.E.; Figueroa, C.M.; Kandah, C.C.; Kay, C.D.; Matthews, M.A.; et al. Physical activity reduces hippocampal atrophy in elders at genetic risk for Alzheimer’s disease. Front. Aging Neurosci. 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Lattari, E.; Portugal, E.; Moraes, H.; Machado, S.; Santos, T.M.; Deslandes, A.C. Acute effects of exercise on mood and EEG activity in healthy young subjects: A systematic review. CNS Neurol. Disord. Drug Targets 2014, 13, 972–980. [Google Scholar] [CrossRef] [PubMed]
- Boecker, H.; Sprenger, T.; Spilker, M.E.; Henriksen, G.; Koppenhoefer, M.; Wagner, K.J.; Valet, M.; Berthele, A.; Tolle, T.R. The runner’s high: Opioidergic mechanisms in the human brain. Cereb. Cortex 2008, 18, 2523–2531. [Google Scholar] [CrossRef] [PubMed]
- Janse Van Rensburg, K.; Taylor, A.; Hodgson, T.; Benattayallah, A. Acute exercise modulates cigarette cravings and brain activation in response to smoking-related images: An fMRI study. Psychopharmacology 2009, 203, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Janse Van Rensburg, K.; Taylor, A.; Benattayallah, A.; Hodgson, T. The effects of exercise on cigarette cravings and brain activation in response to smoking-related images. Psychopharmacology 2012, 221, 659–666. [Google Scholar] [CrossRef] [PubMed]
| FM (n = 11) Mean (SD) | CO (n = 12) Mean (SD) | ||
|---|---|---|---|
| Age (years) | 38.58 (11.17) | 43.67 (7.02) | |
| Height (cm) | 165.18 (6.77) | 165.92 (5.77) | |
| Weight (kg) | 65.55 (11.17) | 68.41 (10.20) | |
| FIQ | 52.95 (13.06) | NA | |
| MPQ VAS | Pre-Exercise | 57.82 (29.10) * | 4.83 (10.18) |
| Pre-Quiet Rest | 50.50 (18.24) * | 1.33 (2.77) | |
| Physical Activity Data (minutes/day) | Sedentary | 624.40 (78.69) | 625.88 (81.35) |
| Light | 164.77 (48.17) | 158.43 (37.42) | |
| Moderate | 82.15 (31.23) | 108.04 (39.8) | |
| Vigorous | 0.28 (0.55) | 3.58 (9.27) | |
| FM Patients (n = 11) | CO (n = 12) | ||||
|---|---|---|---|---|---|
| PI | PU | PI | PU | ||
| EX | Run 1 | 9.31 (5.07) | 7.30 (4.36) | 8.83 (4.42) | 6.24 (3.74) |
| Run 2 | 10.03 (4.49) | 7.97 (4.14) | 8.51 (4.06) | 5.42 (3.73) | |
| Run 3 | 9.88 (4.86) | 7.80 (4.39) | 8.47 (4.60) | 5.76 (3.70) | |
| QR | Run 1 | 11.33 (5.21) | 9.09 (4.30) | 8.15 (3.44) | 5.68 (3.01) |
| Run 2 | 9.60 (3.96) | 7.47 (3.21) | 7.31 (3.50) | 4.96 (2.39) | |
| Run 3 | 9.50 (3.60) | 7.53 (3.40) | 7.79 (3.16) | 5.35 (2.40) | |
| Direction | Peak X,Y,Z | Volume (mm3) | Peak t-Statistic | α | |
|---|---|---|---|---|---|
| FM EX-QR | |||||
| Left Anterior Insula | + | 32, −20, −18 | 4608 | 2.83 | <0.01 |
| Right Anterior Insula | + | −52, −20, −6 | 4032 | 2.88 | <0.01 |
| CO EX-QR | |||||
| Right parietal operculum | + | −64, 28, 14 | 4416 | 5.32 | <0.01 |
| Right pre/postcentral gyrus | + | −36, 24, 50 | 2816 | 3.57 | <0.05 |
| FM-CO × EX-QR | |||||
| Left DLPFC | + | 28, −56, −14 | 3136 | 2.91 | <0.03 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ellingson, L.D.; Stegner, A.J.; Schwabacher, I.J.; Koltyn, K.F.; Cook, D.B. Exercise Strengthens Central Nervous System Modulation of Pain in Fibromyalgia. Brain Sci. 2016, 6, 8. https://doi.org/10.3390/brainsci6010008
Ellingson LD, Stegner AJ, Schwabacher IJ, Koltyn KF, Cook DB. Exercise Strengthens Central Nervous System Modulation of Pain in Fibromyalgia. Brain Sciences. 2016; 6(1):8. https://doi.org/10.3390/brainsci6010008
Chicago/Turabian StyleEllingson, Laura D., Aaron J. Stegner, Isaac J. Schwabacher, Kelli F. Koltyn, and Dane B. Cook. 2016. "Exercise Strengthens Central Nervous System Modulation of Pain in Fibromyalgia" Brain Sciences 6, no. 1: 8. https://doi.org/10.3390/brainsci6010008
APA StyleEllingson, L. D., Stegner, A. J., Schwabacher, I. J., Koltyn, K. F., & Cook, D. B. (2016). Exercise Strengthens Central Nervous System Modulation of Pain in Fibromyalgia. Brain Sciences, 6(1), 8. https://doi.org/10.3390/brainsci6010008






