Attention and Default Mode Network Assessments of Meditation Experience during Active Cognition and Rest
Abstract
1. Introduction
2. Materials and Methods
2.1. Participant Recruitment
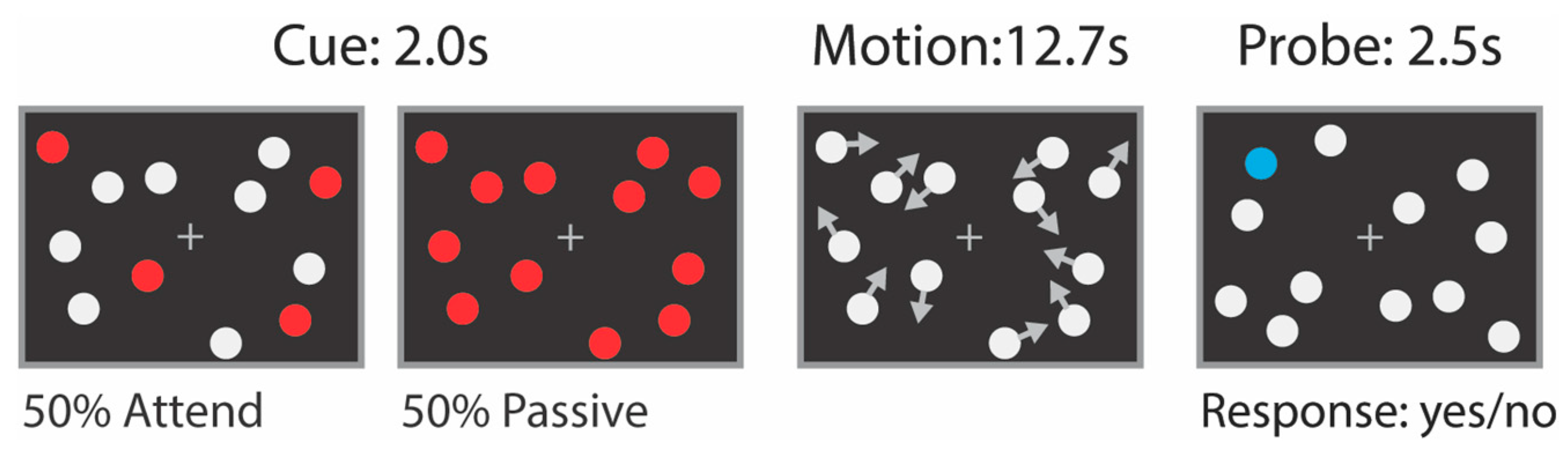
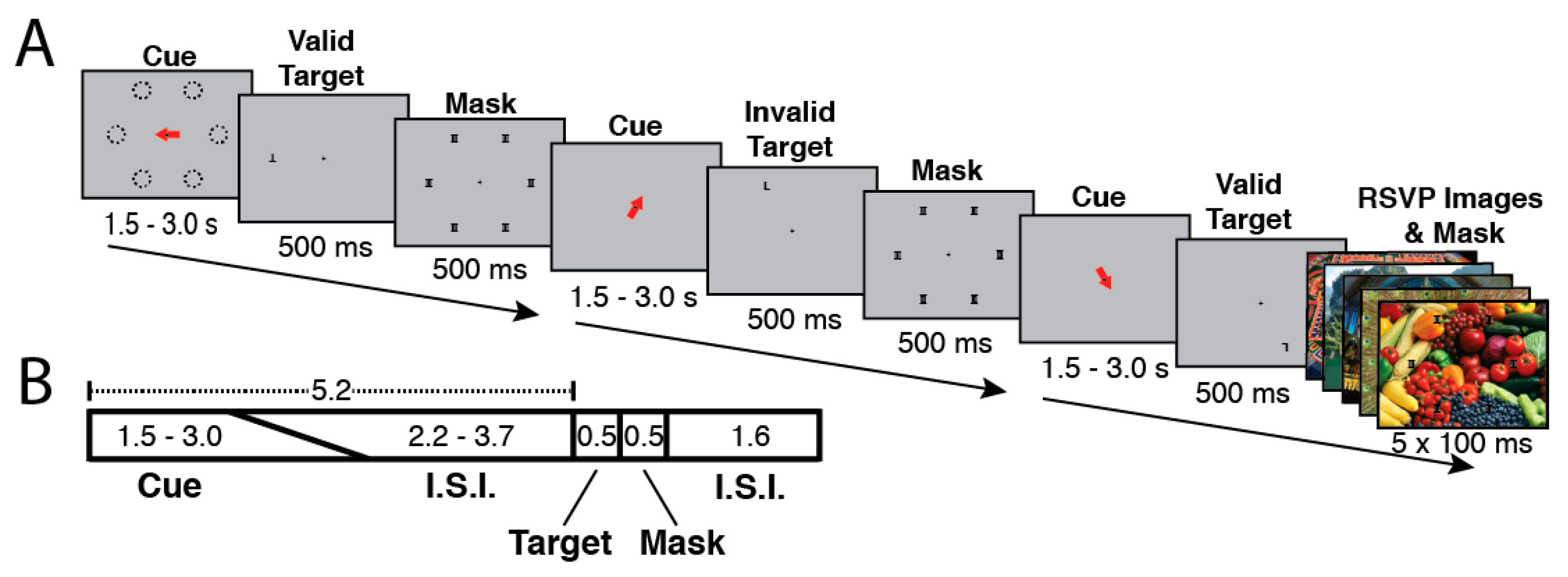
2.2. Attention Tasks Performed during fMRI Scanning
2.3. MRI Scanning Parameters
2.4. Data Analysis
3. Results
3.1. Multiple Object Tracking: Behavior
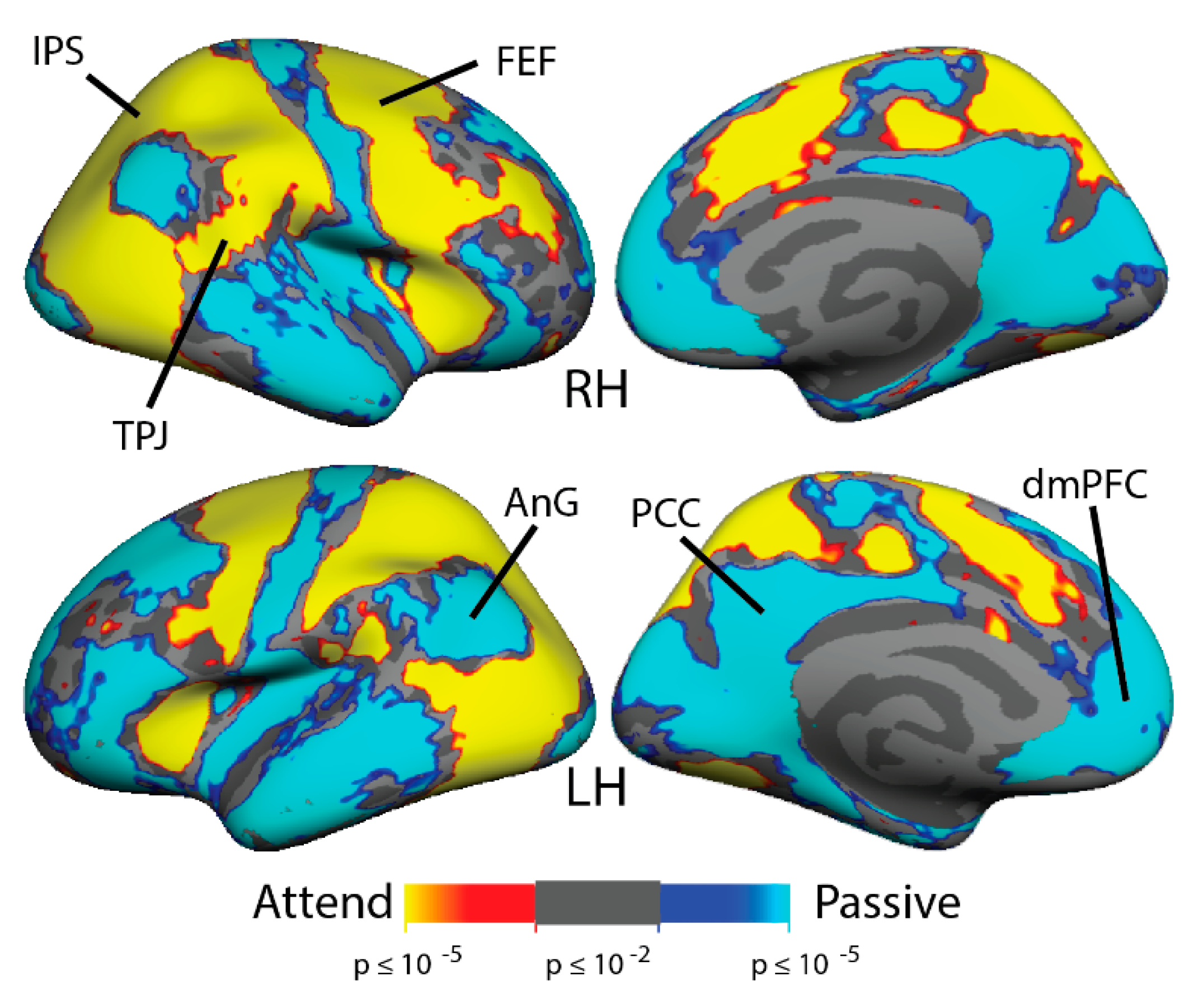
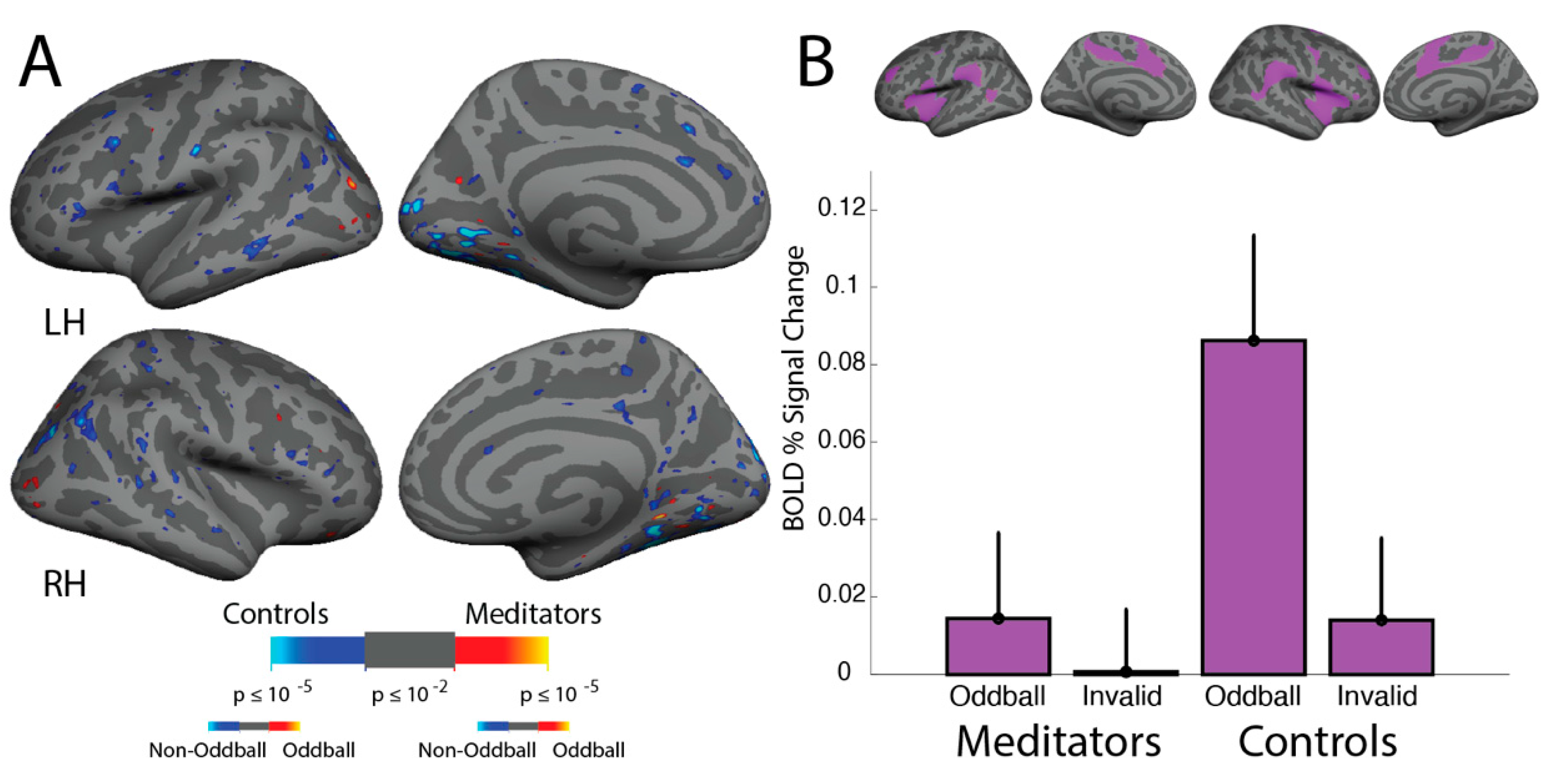
3.2. Multiple Object Tracking: fMRI Activation
3.3. Multiple Object Tracking: Region of Interest Analyses
3.4. Relationship between Behavior, Meditation Experience, and BOLD Activation
3.5. ViNO: Behavior
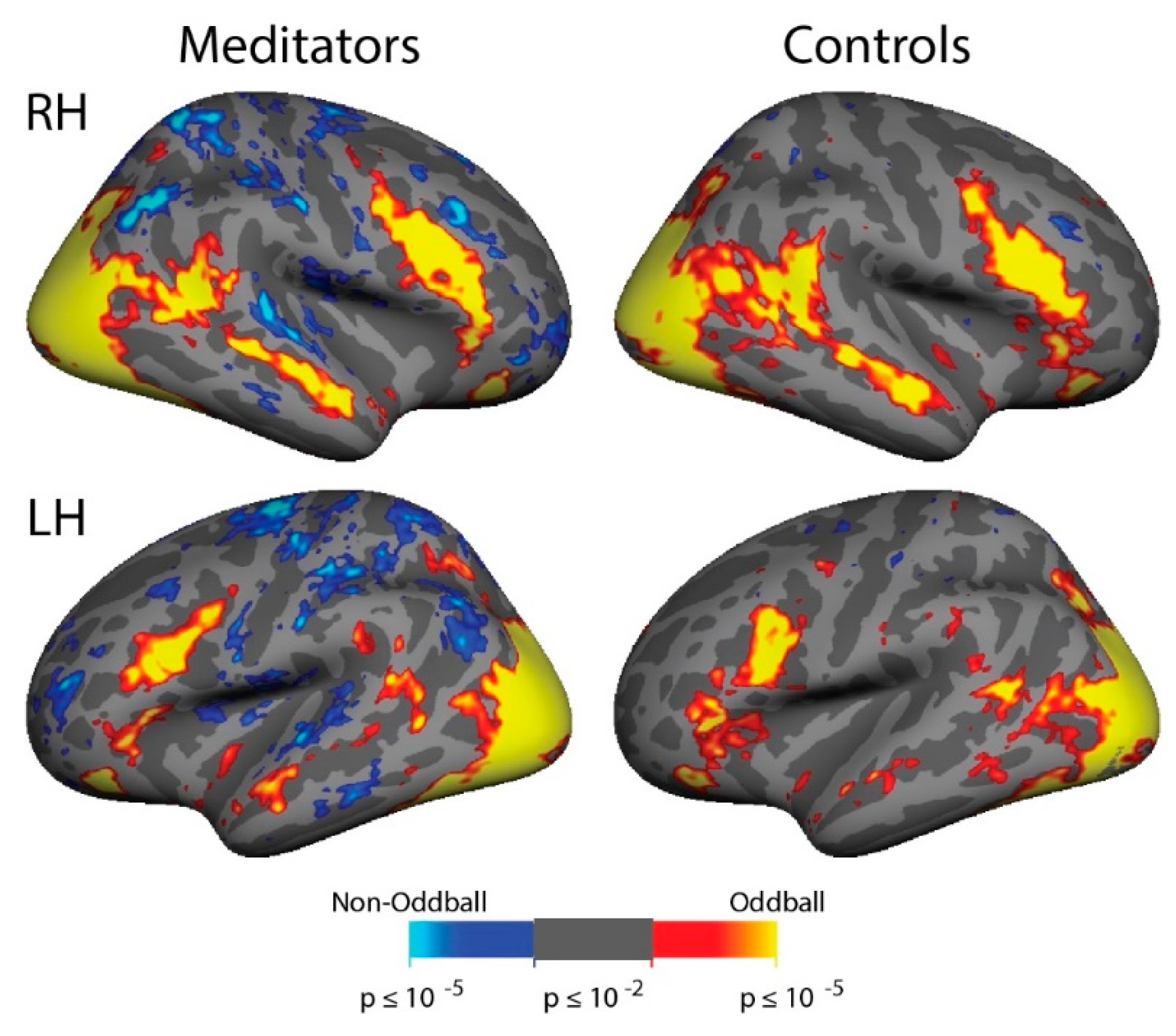
3.6. ViNO: fMRI Activation
3.7. ViNO: ROI Analyses
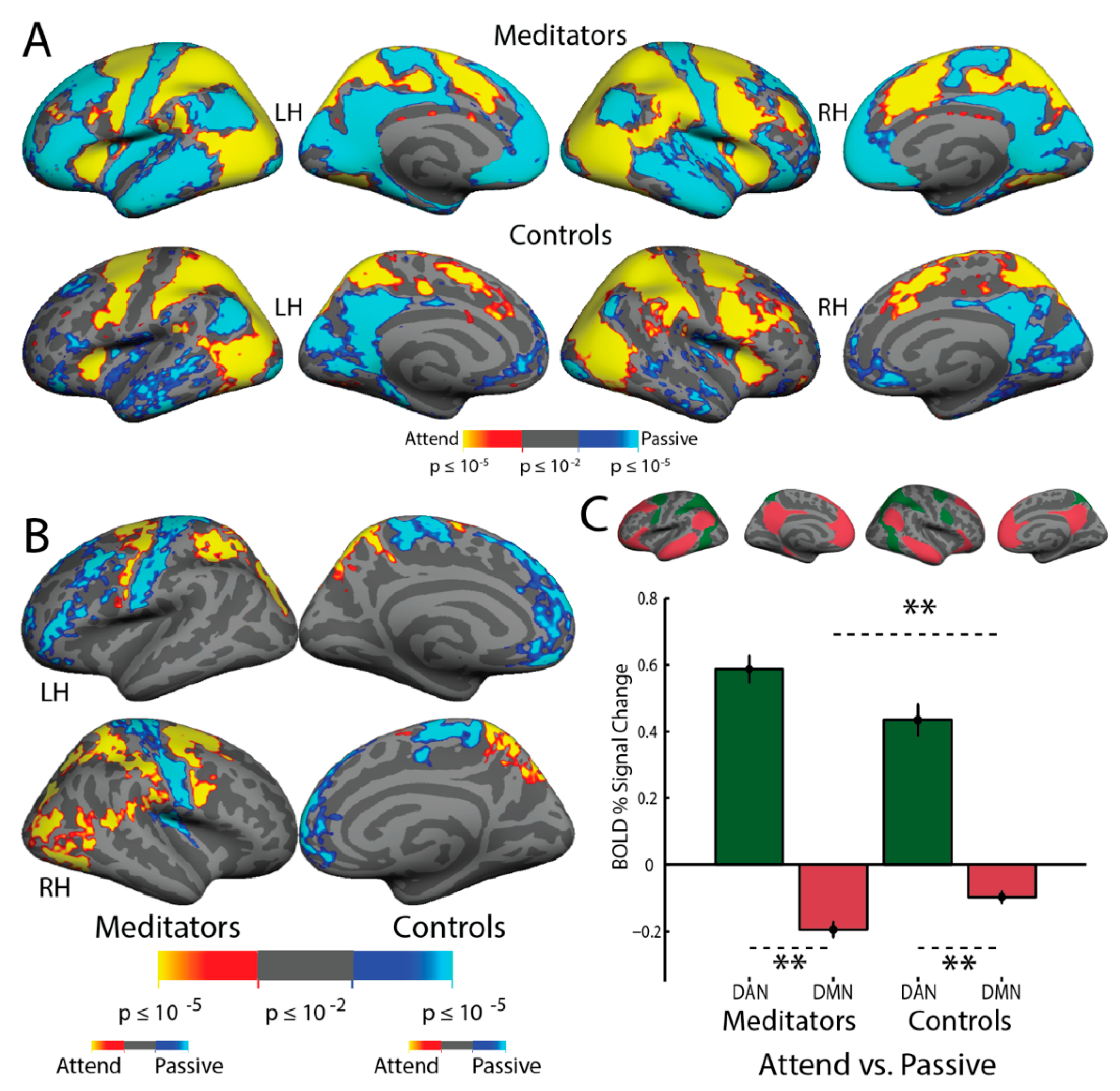
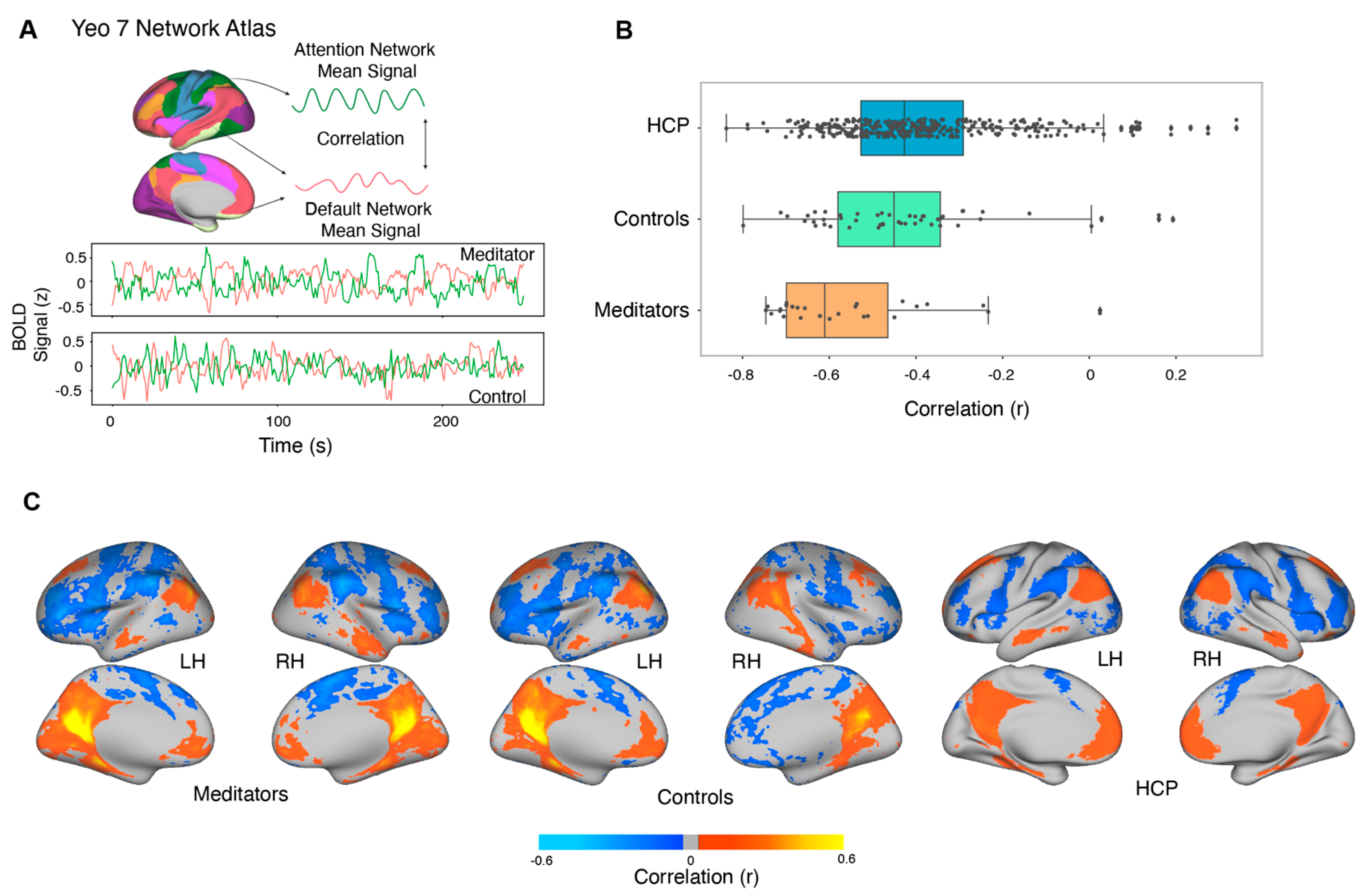
3.8. Resting-State Functional Connectivity
4. Discussion
4.1. MOT: Greater DMN Suppression in Meditators While Attending
4.2. ViNO: No Differences in Attentional Capture Associated with Meditation
4.3. Resting-State Functional Connectivity: Experienced Meditators Exhibit Greater DAN–DMN Opponency
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Kabat-Zinn, J. Mindfulness-Based Interventions in Context: Past, Present, and Future. Clin. Psychol. Sci. Pr. 2003, 10, 144–156. [Google Scholar] [CrossRef]
- Tang, Y.-Y.; Ma, Y.; Wang, J.; Fan, Y.; Feng, S.; Lu, Q.; Yu, Q.; Sui, D.; Rothbart, M.K.; Fan, M.; et al. Short-term meditation training improves attention and self-regulation. Proc. Natl. Acad. Sci. USA 2007, 104, 17152–17156. [Google Scholar] [CrossRef]
- Lazar, S.W.; Bush, G.; Gollub, R.L.; Fricchione, G.L.; Khalsa, G.; Benson, H. Functional brain mapping of the relaxation response and meditation. NeuroReport 2000, 11, 1581–1585. [Google Scholar] [CrossRef]
- Manna, A.; Raffone, A.; Perrucci, M.G.; Nardo, D.; Ferretti, A.; Tartaro, A.; Londei, A.; Del Gratta, C.; Belardinelli, M.O.; Romani, G.L. Neural correlates of focused attention and cognitive monitoring in meditation. Brain Res. Bull. 2010, 82, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Brefczynski-Lewis, J.A.; Lutz, A.; Schaefer, H.S.; Levinson, D.B.; Davidson, R.J. Neural correlates of attentional expertise in long-term meditation practitioners. Proc. Natl. Acad. Sci. USA 2007, 104, 11483–11488. [Google Scholar] [CrossRef]
- Valentine, E.R.; Sweet, P.L.G. Meditation and attention: A comparison of the effects of concentrative and mindfulness meditation on sustained attention. Ment. Health Relig. Cult. 1999, 2, 59–70. [Google Scholar] [CrossRef]
- Lutz, A.; Slagter, H.A.; Rawlings, N.B.; Francis, A.D.; Greischar, L.L.; Davidson, R.J. Mental Training Enhances Attentional Stability: Neural and Behavioral Evidence. J. Neurosci. 2009, 29, 13418–13427. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.; Malinowski, P. Meditation, mindfulness and cognitive flexibility. Conscious. Cogn. 2009, 18, 176–186. [Google Scholar] [CrossRef] [PubMed]
- MacLean, K.A.; Ferrer, E.; Aichele, S.R.; Bridwell, D.A.; Zanesco, A.P.; Jacobs, T.L.; King, B.G.; Rosenberg, E.L.; Sahdra, B.K.; Shaver, P.R.; et al. Intensive Meditation Training Improves Perceptual Discrimination and Sustained Attention. Psychol. Sci. 2010, 21, 829–839. [Google Scholar] [CrossRef]
- Izzetoglu, M.; Shewokis, P.A.; Tsai, K.; Dantoin, P.; Sparango, K.; Min, K. Short-Term Effects of Meditation on Sustained Attention as Measured by fNIRS. Brain Sci. 2020, 10, 608. [Google Scholar] [CrossRef] [PubMed]
- Corbetta, M.; Shulman, G.L. Control of goal-directed and stimulus-driven attention in the brain. Nat. Rev. Neurosci. 2002, 3, 201–215. [Google Scholar] [CrossRef] [PubMed]
- Serences, J.T.; Shomstein, S.; Leber, A.B.; Golay, X.; Egeth, H.E.; Yantis, S. Coordination of Voluntary and Stimulus-Driven Attentional Control in Human Cortex. Psychol. Sci. 2005, 16, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Corbetta, M.; Patel, G.; Shulman, G.L. The Reorienting System of the Human Brain: From Environment to Theory of Mind. Neuron 2008, 58, 306–324. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, A.S.; Esterman, M.; Wilson, D.; Serences, J.T.; Yantis, S. Control of Spatial and Feature-Based Attention in Frontoparietal Cortex. J. Neurosci. 2010, 30, 14330–14339. [Google Scholar] [CrossRef]
- Somers, D.C.; Sheremata, S.L. Attention maps in the brain. Wiley Interdiscip. Rev. Cogn. Sci. 2013, 4, 327–340. [Google Scholar] [CrossRef]
- Michalka, S.W.; Kong, L.; Rosen, M.L.; Shinn-Cunningham, B.G.; Somers, D.C. Short-Term Memory for Space and Time Flexibly Recruit Complementary Sensory-Biased Frontal Lobe Attention Networks. Neuron 2015, 87, 882–892. [Google Scholar] [CrossRef] [PubMed]
- Jonides, J.; Yantis, S. Uniqueness of abrupt visual onset in capturing attention. Percept. Psychophys. 1988, 43, 346–354. [Google Scholar] [CrossRef]
- Corbetta, M.; Kincade, J.M.; Ollinger, J.M.; McAvoy, M.P.; Shulman, G.L. Voluntary orienting is dissociated from target detection in human posterior parietal cortex. Nat. Neurosci. 2000, 3, 292–297. [Google Scholar] [CrossRef]
- Fox, M.D.; Corbetta, M.; Snyder, A.Z.; Vincent, J.L.; Raichle, M.E. Spontaneous neuronal activity distinguishes human dorsal and ventral attention systems. Proc. Natl. Acad. Sci. USA 2006, 103, 10046–10051. [Google Scholar] [CrossRef]
- Weissman, D.H.; Prado, J. Heightened activity in a key region of the ventral attention network is linked to reduced activity in a key region of the dorsal attention network during unexpected shifts of covert visual spatial attention. NeuroImage 2012, 61, 798–804. [Google Scholar] [CrossRef]
- Patel, G.H.; Yang, D.; Jamerson, E.C.; Snyder, L.H.; Corbetta, M.; Ferrera, V.P. Functional evolution of new and expanded attention networks in humans. Proc. Natl. Acad. Sci. USA 2015, 112, 9454–9459. [Google Scholar] [CrossRef] [PubMed]
- Theeuwes, J. Endogenous and Exogenous Control of Visual Selection. Perception 1994, 23, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Shulman, G.L.; Fiez, J.A.; Corbetta, M.; Buckner, R.L.; Miezin, F.M.; Raichle, M.E.; Petersen, S.E. Common Blood Flow Changes across Visual Tasks: II. Decreases in Cerebral Cortex. J. Cogn. Neurosci. 1997, 9, 648–663. [Google Scholar] [CrossRef]
- Raichle, M.E.; MacLeod, A.M.; Snyder, A.Z.; Powers, W.J.; Gusnard, D.A.; Shulman, G.L. A default mode of brain function. Proc. Natl. Acad. Sci. USA 2001, 98, 676–682. [Google Scholar] [CrossRef]
- Greicius, M.D.; Menon, V. Default-Mode Activity during a Passive Sensory Task: Uncoupled from Deactivation but Impacting Activation. J. Cogn. Neurosci. 2004, 16, 1484–1492. [Google Scholar] [CrossRef] [PubMed]
- Buckner, R.L.; Andrews-Hanna, J.R.; Schacter, D.L. The Brain’s Default Network. Ann. N. Y. Acad. Sci. 2008, 1124, 1–38. [Google Scholar] [CrossRef]
- Power, J.D.; Schlaggar, B.L.; Petersen, S.E. Studying Brain Organization via Spontaneous fMRI Signal. Neuron 2014, 84, 681–696. [Google Scholar] [CrossRef]
- Yeo, B.T.T.; Krienen, F.M.; Sepulcre, J.; Sabuncu, M.R.; Lashkari, D.; Hollinshead, M.; Roffman, J.L.; Smoller, J.W.; Zöllei, L.; Polimeni, J.R.; et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J. Neurophysiol. 2011, 106, 1125–1165. [Google Scholar] [CrossRef]
- Power, J.D.; Cohen, A.L.; Nelson, S.M.; Wig, G.S.; Barnes, K.A.; Church, J.A.; Vogel, A.C.; Laumann, T.O.; Miezin, F.M.; Schlaggar, B.L.; et al. Functional Network Organization of the Human Brain. Neuron 2011, 72, 665–678. [Google Scholar] [CrossRef]
- Cole, M.W.; Reynolds, J.R.; Power, J.D.; Repovs, G.; Anticevic, A.; Braver, T.S. Multi-task connectivity reveals flexible hubs for adaptive task control. Nat. Neurosci. 2013, 16, 1348–1355. [Google Scholar] [CrossRef]
- Gonzalez-Castillo, J.; Bandettini, P.A. Task-based dynamic functional connectivity: Recent findings and open questions. NeuroImage 2018, 180, 526–533. [Google Scholar] [CrossRef]
- Spreng, R.N.; Stevens, W.D.; Viviano, J.D.; Schacter, D.L. Attenuated anticorrelation between the default and dorsal attention networks with aging: Evidence from task and rest. Neurobiol. Aging 2016, 45, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Barrantes, R.; Arancibia, M.; Stojanova, J.; Aspé-Sánchez, M.; Córdova, C.; Henríquez-Ch, R.A. Default Mode Network, Meditation, and Age-Associated Brain Changes: What Can We Learn from the Impact of Mental Training on Well-Being as a Psychotherapeutic Approach? Neural Plast. 2019, 2019, 7067592. [Google Scholar] [CrossRef] [PubMed]
- Esterman, M.; Rosenberg, M.D.; Noonan, S.K. Intrinsic Fluctuations in Sustained Attention and Distractor Processing. J. Neurosci. 2014, 34, 1724–1730. [Google Scholar] [CrossRef]
- Lo, O.-Y.; Halko, M.A.; Zhou, J.; Harrison, R.; Lipsitz, L.A.; Manor, B. Gait Speed and Gait Variability Are Associated with Different Functional Brain Networks. Front. Aging Neurosci. 2017, 9, 390. [Google Scholar] [CrossRef] [PubMed]
- Esposito, R.; Cieri, F.; Chiacchiaretta, P.; Cera, N.; Lauriola, M.; Di Giannantonio, M.; Tartaro, A.; Ferretti, A. Modifications in resting state functional anticorrelation between default mode network and dorsal attention network: Comparison among young adults, healthy elders and mild cognitive impairment patients. Brain Imaging Behav. 2017, 12, 127–141. [Google Scholar] [CrossRef]
- Weathersby, F.L.; King, J.B.; Fox, J.C.; Loret, A.; Anderson, J.S. Functional connectivity of emotional well-being: Overconnectivity between default and attentional networks is associated with attitudes of anger and aggression. Psychiatry Res. Neuroimaging 2019, 291, 52–62. [Google Scholar] [CrossRef]
- Brewer, J.A.; Worhunsky, P.D.; Gray, J.R.; Tang, Y.-Y.; Weber, J.; Kober, H. Meditation experience is associated with differences in default mode network activity and connectivity. Proc. Natl. Acad. Sci. USA 2011, 108, 20254–20259. [Google Scholar] [CrossRef]
- Lazar, S.W.; Kerr, C.E.; Wasserman, R.H.; Gray, J.R.; Greve, D.N.; Treadway, M.T.; McGarvey, M.; Quinn, B.T.; Dusek, J.A.; Benson, H.; et al. Meditation experience is associated with increased cortical thickness. NeuroReport 2005, 16, 1893–1897. [Google Scholar] [CrossRef] [PubMed]
- Taylor, V.A.; Daneault, V.; Grant, J.; Scavone, G.; Breton, E.; Roffe-Vidal, S.; Courtemanche, J.; Lavarenne, A.S.; Marrelec, G.; Benali, H.; et al. Impact of meditation training on the default mode network during a restful state. Soc. Cogn. Affect. Neurosci. 2013, 8, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Killingsworth, M.A.; Gilbert, D.T. A Wandering Mind Is an Unhappy Mind. Science 2010, 330, 932. [Google Scholar] [CrossRef] [PubMed]
- Travis, F.; Shear, J. Focused attention, open monitoring and automatic self-transcending: Categories to organize meditations from Vedic, Buddhist and Chinese traditions. Conscious. Cogn. 2010, 19, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Vago, D.R.; Zeidan, F. The brain on silent: Mind wandering, mindful awareness, and states of mental tranquility. Ann. N. Y. Acad. Sci. 2016, 1373, 96–113. [Google Scholar] [CrossRef] [PubMed]
- Goenka, S.N. The Discourse Summaries of SN Goenka; Pariyatti Publishing: Onalaska, WA, USA, 1987. [Google Scholar]
- Pylyshyn, Z.W.; Storm, R.W. Tracking multiple independent targets: Evidence for a parallel tracking mechanism. Spat. Vis. 1988, 3, 179–197. [Google Scholar] [CrossRef] [PubMed]
- Bettencourt, K.C.; Somers, D.C. Effects of target enhancement and distractor suppression on multiple object tracking capacity. J. Vis. 2009, 9, 9. [Google Scholar] [CrossRef]
- Devaney, K.J.; Rosen, M.L.; Levin, E.J.; Somers, D.C. Identification of Visual Attentional Regions of the Temporoparietal Junction in Individual Subjects using a Vivid, Novel Oddball Paradigm. Front. Hum. Neurosci. 2019, 13, 424. [Google Scholar] [CrossRef]
- Goodman, L.A. Snowball Sampling. Ann. Math. Stat. 1961, 32, 148–170. [Google Scholar] [CrossRef]
- Biernacki, P.; Waldorf, D. Snowball Sampling: Problems and Techniques of Chain Referral Sampling. Sociol. Methods Res. 1981, 10, 141–163. [Google Scholar] [CrossRef]
- Boot, W.R.; Blakely, D.P.; Simons, D.J. Do Action Video Games Improve Perception and Cognition? Front. Psychol. 2011, 2, 226. [Google Scholar] [CrossRef]
- Van Essen, D.C.; Smith, S.M.; Barch, D.M.; Behrens, T.E.; Yacoub, E.; Ugurbil, K. The WU-Minn Human Connectome Project: An overview. NeuroImage 2013, 80, 62–79. [Google Scholar] [CrossRef]
- Scholl, B.J.; Pylyshyn, Z.W.; Feldman, J. What is a visual object? Evidence from target merging in multiple object tracking. Cognition 2001, 80, 159–177. [Google Scholar] [CrossRef]
- Culham, J.C.; Cavanagh, P.; Kanwisher, N.G. Attention Response Functions. Neuron 2001, 32, 737–745. [Google Scholar] [CrossRef]
- Engell, A.D.; Haxby, J.V. Facial expression and gaze-direction in human superior temporal sulcus. Neuropsychologia 2007, 45, 3234–3241. [Google Scholar] [CrossRef] [PubMed]
- Tsao, D.Y.; Livingstone, M.S. Mechanisms of Face Perception. Annu. Rev. Neurosci. 2008, 31, 411–437. [Google Scholar] [CrossRef]
- Jovicich, J.; Czanner, S.; Greve, D.; Haley, E.; van der Kouwe, A.; Gollub, R.; Kennedy, D.; Schmitt, F.; Brown, G.; MacFall, J.; et al. Reliability in multi-site structural MRI studies: Effects of gradient non-linearity correction on phantom and human data. NeuroImage 2006, 30, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Ségonne, F.; Dale, A.; Busa, E.; Glessner, M.; Salat, D.; Hahn, H.; Fischl, B. A hybrid approach to the skull stripping problem in MRI. NeuroImage 2004, 22, 1060–1075. [Google Scholar] [CrossRef] [PubMed]
- Dale, A.M.; Fischla, B.; Sereno, M.I. Cortical Surface-Based Analysis: I. Segmentation and Surface Reconstruction. Neuroimage 1999, 9, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Fischl, B.; Dale, A.M. Measuring the thickness of the human cerebral cortex from magnetic resonance images. Proc. Natl. Acad. Sci. USA 2000, 97, 11050–11055. [Google Scholar] [CrossRef]
- Fischl, B.; Liu, A.; Dale, A. Automated manifold surgery: Constructing geometrically accurate and topologically correct models of the human cerebral cortex. IEEE Trans. Med. Imaging 2001, 20, 70–80. [Google Scholar] [CrossRef]
- Fischl, B.; Salat, D.H.; Busa, E.; Albert, M.; Dieterich, M.; Haselgrove, C.; Van Der Kouwe, A.; Killiany, R.; Kennedy, D.; Klaveness, S.; et al. Whole brain segmentation: Automated labeling of neuroanatomical structures in the human brain. Neuron 2002, 33, 341–355. [Google Scholar] [CrossRef]
- Fischl, B.; Salat, D.H.; van der Kouwe, A.J.; Makris, N.; Ségonne, F.; Quinn, B.T.; Dale, A.M. Sequence-independent segmentation of magnetic resonance images. Neuroimage 2004, 23, S69–S84. [Google Scholar] [CrossRef]
- Fischl, B.; Sereno, M.I.; Tootell, R.; Dale, A.M. High-resolution intersubject averaging and a coordinate system for the cortical surface. Hum. Brain Mapp. 1999, 8, 272–284. [Google Scholar] [CrossRef]
- Fischl, B.; Sereno, M.I.; Dale, A.M. Cortical Surface-Based Analysis. NeuroImage 1999, 9, 195–207. [Google Scholar] [CrossRef] [PubMed]
- Fischl, B.; Van Der Kouwe, A.; Destrieux, C.; Halgren, E.; Ségonne, F.; Salat, D.H.; Busa, E.; Seidman, L.J.; Goldstein, J.; Kennedy, D.; et al. Automatically Parcellating the Human Cerebral Cortex. Cereb. Cortex 2004, 14, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Greve, D.N.; Fischl, B. Accurate and robust brain image alignment using boundary-based registration. Neuroimage 2009, 48, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Hagler, D.J.; Saygin, A.P.; Sereno, M.I. Smoothing and cluster thresholding for cortical surface-based group analysis of fMRI data. Neuroimage 2006, 33, 1093–1103. [Google Scholar] [CrossRef]
- Eklund, A.; Nichols, T.E.; Knutsson, H. Cluster failure: Why fMRI inferences for spatial extent have inflated false-positive rates. Proc. Natl. Acad. Sci. USA 2016, 113, 7900–7905. [Google Scholar] [CrossRef] [PubMed]
- MathWorks Inc. MATLAB: The Language of Technical Computing; Desktop Tools and Development Environment, Version 7; MathWorks Inc.: Natick, MA, USA, 2005. [Google Scholar]
- Van Dijk, K.R.A.; Hedden, T.; Venkataraman, A.; Evans, K.C.; Lazar, S.W.; Buckner, R.L. Intrinsic Functional Connectivity as a Tool for Human Connectomics: Theory, Properties, and Optimization. J. Neurophysiol. 2010, 103, 297–321. [Google Scholar] [CrossRef]
- Power, J.D.; Barnes, K.A.; Snyder, A.Z.; Schlaggar, B.L.; Petersen, S.E. Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. Neuroimage 2012, 59, 2142–2154. [Google Scholar] [CrossRef]
- Carp, J. The secret lives of experiments: Methods reporting in the fMRI literature. Neuroimage 2012, 63, 289–300. [Google Scholar] [CrossRef]
- Schaefer, A.; Kong, R.; Gordon, E.M.; O Laumann, T.; Zuo, X.-N.; Holmes, A.J.; Eickhoff, S.B.; Yeo, B.T.T. Local-Global Parcellation of the Human Cerebral Cortex from Intrinsic Functional Connectivity MRI. Cereb. Cortex 2018, 28, 3095–3114. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, P.; Gommers, R.; Oliphant, T.E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Burovski, E.; Peterson, P.; Weckesser, W.; Bright, J.; et al. SciPy 1.0: Fundamental algorithms for scientific computing in Python. Nat. Methods 2020, 17, 261–272. [Google Scholar] [CrossRef]
- Seabold, S.; Perktold, J. Statsmodels: Econometric and statistical modeling with python. In Proceedings of the 9th Python in Science Conference, Austin, TX, USA, 28–30 June 2010. [Google Scholar]
- Cowan, N. The magical number 4 in short-term memory: A reconsideration of mental storage capacity. Behav. Brain Sci. 2001, 24, 87–114. [Google Scholar] [CrossRef]
- Brissenden, J.A.; Levin, E.J.; Osher, D.E.; Halko, M.A.; Somers, D.C. Functional Evidence for a Cerebellar Node of the Dorsal Attention Network. J. Neurosci. 2016, 36, 6083–6096. [Google Scholar] [CrossRef]
- Glasser, M.F.; Sotiropoulos, S.N.; Wilson, J.A.; Coalson, T.S.; Fischl, B.; Andersson, J.L.; Xu, J.; Jbabdi, S.; Webster, M.; Polimeni, J.R.; et al. The minimal preprocessing pipelines for the Human Connectome Project. Neuroimage 2013, 80, 105–124. [Google Scholar] [CrossRef]
- Whitfield-Gabrieli, S.; Ford, J.M. Default Mode Network Activity and Connectivity in Psychopathology. Annu. Rev. Clin. Psychol. 2012, 8, 49–76. [Google Scholar] [CrossRef]
- James, W. Psychology, Briefer Course; Harvard University Press: Cambridge, MA, USA, 1892. [Google Scholar]
- Smallwood, J.; Schooler, J.W. The restless mind. Psychol. Bull. 2006, 132, 946–958. [Google Scholar] [CrossRef] [PubMed]
- Gruberger, M.; Ben Simon, E.; Levkovitz, Y.; Zangen, A.; Hendler, T. Towards a Neuroscience of Mind-Wandering. Front. Hum. Neurosci. 2011, 5, 56. [Google Scholar] [CrossRef] [PubMed]
- Gusnard, D.A.; Raichle, M.E. Searching for a baseline: Functional imaging and the resting human brain. Nat. Rev. Neurosci. 2001, 2, 685–694. [Google Scholar] [CrossRef] [PubMed]
- Mason, M.F.; Norton, M.I.; Van Horn, J.D.; Wegner, D.M.; Grafton, S.T.; Macrae, C.N. Wandering Minds: The Default Network and Stimulus-Independent Thought. Science 2007, 315, 393–395. [Google Scholar] [CrossRef]
- Johnson, S.C.; Baxter, L.C.; Wilder, L.S.; Pipe, J.G.; Heiserman, J.E.; Prigatano, G.P. Neural correlates of self-reflection. Brain 2002, 125, 1808–1814. [Google Scholar] [CrossRef]
- Goldberg, I.I.; Harel, M.; Malach, R. When the Brain Loses Its Self: Prefrontal Inactivation during Sensorimotor Processing. Neuron 2006, 50, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Schneider, F.; Bermpohl, F.; Heinzel, A.; Rotte, M.; Walter, M.; Tempelmann, C.; Wiebking, C.; Dobrowolny, H.; Heinze, H.; Northoff, G. The resting brain and our self: Self-relatedness modulates resting state neural activity in cortical midline structures. Neuroscience 2008, 157, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Andrews-Hanna, J.R.; Reidler, J.S.; Sepulcre, J.; Poulin, R.; Buckner, R.L. Functional-Anatomic Fractionation of the Brain’s Default Network. Neuron 2010, 65, 550–562. [Google Scholar] [CrossRef] [PubMed]
- Mrazek, M.D.; Franklin, M.S.; Phillips, D.T.; Baird, B.; Schooler, J.W. Mindfulness Training Improves Working Memory Capacity and GRE Performance While Reducing Mind Wandering. Psychol. Sci. 2013, 24, 776–781. [Google Scholar] [CrossRef]
- Josipovic, Z.; Dinstein, I.; Weber, J.; Heeger, D.J. Influence of meditation on anti-correlated networks in the brain. Front. Hum. Neurosci. 2012, 5, 183. [Google Scholar] [CrossRef]
- Garrison, K.A.; Zeffiro, T.A.; Scheinost, D.; Constable, R.T.; Brewer, J.A. Meditation leads to reduced default mode network activity beyond an active task. Cogn. Affect. Behav. Neurosci. 2015, 15, 712–720. [Google Scholar] [CrossRef] [PubMed]
- Maccoon, N.G.; MacLean, K.A.; Davidson, R.J.; Saron, C.D.; Lutz, A. No Sustained Attention Differences in a Longitudinal Randomized Trial Comparing Mindfulness Based Stress Reduction versus Active Control. PLoS ONE 2014, 9, e97551. [Google Scholar] [CrossRef] [PubMed]
- Jovicich, J.; Peters, R.J.; Koch, C.; Braun, J.; Chang, L.; Ernst, T. Brain Areas Specific for Attentional Load in a Motion-Tracking Task. J. Cogn. Neurosci. 2001, 13, 1048–1058. [Google Scholar] [CrossRef]
- Drew, T.; Vogel, E.K. Neural Measures of Individual Differences in Selecting and Tracking Multiple Moving Objects. J. Neurosci. 2008, 28, 4183–4191. [Google Scholar] [CrossRef]
- Fougnie, D.; Marois, R. Distinct Capacity Limits for Attention and Working Memory. Psychol. Sci. 2006, 17, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Laukkonen, R.; Slagter, H.A. From many to one: Meditation and the plasticity of the predictive mind. PsyArXiv 2020. [Google Scholar] [CrossRef]
- Cahn, B.R.; Polich, J. Meditation (Vipassana) and the P3a event-related brain potential. Int. J. Psychophysiol. 2009, 72, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Bauer, C.C.C.; Whitfield-Gabrieli, S.; Díaz, J.L.; Pasaye, E.H.; Barrios, F.A. From State-to-Trait Meditation: Reconfiguration of Central Executive and Default Mode Networks. Eneuro 2019, 6. [Google Scholar] [CrossRef] [PubMed]
- Christoff, K.; Irving, Z.C.; Fox, K.C.; Spreng, R.N.; Andrews-Hanna, J.R. Mind-wandering as spontaneous thought: A dynamic framework. Nat. Rev. Neurosci. 2016, 17, 718–731. [Google Scholar] [CrossRef] [PubMed]

| MOT Behavioral Data | |||
|---|---|---|---|
| Group | Hit Rate | False Alarm | k Score |
| Meditation | 0.64 ± 0.04 | 0.33 ± 0.06 | 1.25 ± 0.32 |
| Control | 0.57 ± 0.05 | 0.39 ± 0.05 | 0.82 ± 0.22 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Devaney, K.J.; Levin, E.J.; Tripathi, V.; Higgins, J.P.; Lazar, S.W.; Somers, D.C. Attention and Default Mode Network Assessments of Meditation Experience during Active Cognition and Rest. Brain Sci. 2021, 11, 566. https://doi.org/10.3390/brainsci11050566
Devaney KJ, Levin EJ, Tripathi V, Higgins JP, Lazar SW, Somers DC. Attention and Default Mode Network Assessments of Meditation Experience during Active Cognition and Rest. Brain Sciences. 2021; 11(5):566. https://doi.org/10.3390/brainsci11050566
Chicago/Turabian StyleDevaney, Kathryn J., Emily J. Levin, Vaibhav Tripathi, James P. Higgins, Sara W. Lazar, and David C. Somers. 2021. "Attention and Default Mode Network Assessments of Meditation Experience during Active Cognition and Rest" Brain Sciences 11, no. 5: 566. https://doi.org/10.3390/brainsci11050566
APA StyleDevaney, K. J., Levin, E. J., Tripathi, V., Higgins, J. P., Lazar, S. W., & Somers, D. C. (2021). Attention and Default Mode Network Assessments of Meditation Experience during Active Cognition and Rest. Brain Sciences, 11(5), 566. https://doi.org/10.3390/brainsci11050566






