Tablet Use Affects Preschoolers’ Executive Function: fNIRS Evidence from the Dimensional Change Card Sort Task
Abstract
1. Introduction
1.1. Tablet Use and Early Childhood Development
1.2. Neuroimaging Studies on Early Executive Function
- Will the heavy users perform significantly differently from the non-users in the DCCS task?
- What are the significant differences in prefrontal activation as evidenced by fNIRS between the heavy-users and non-users in the DCCS task?
2. Materials and Methods
2.1. Participants
2.2. Measures
2.2.1. Home Learning Environment and Practice Survey (HLEP)
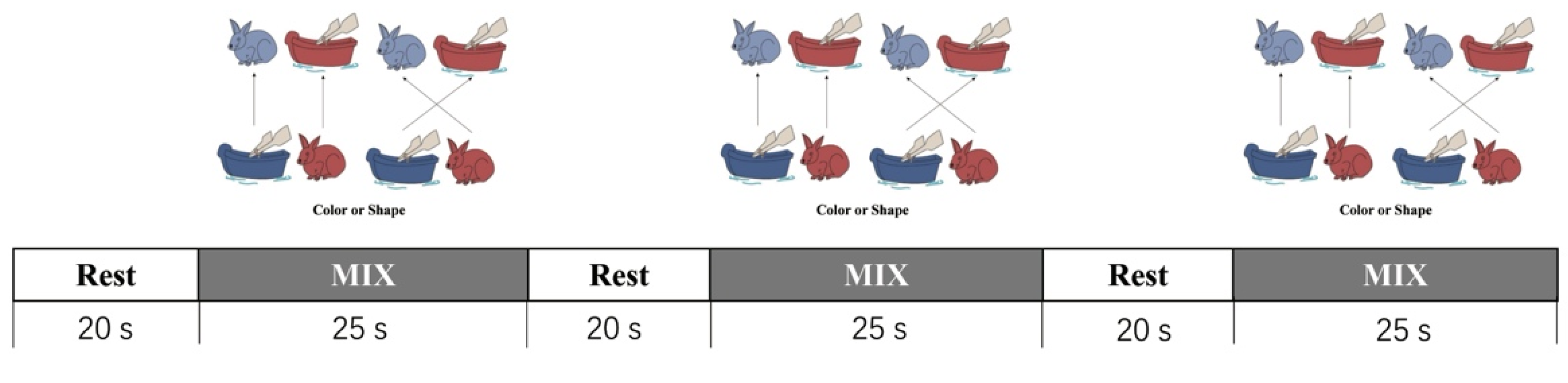
2.2.2. The Dimensional Change Card Sort (DCCS) Tasks
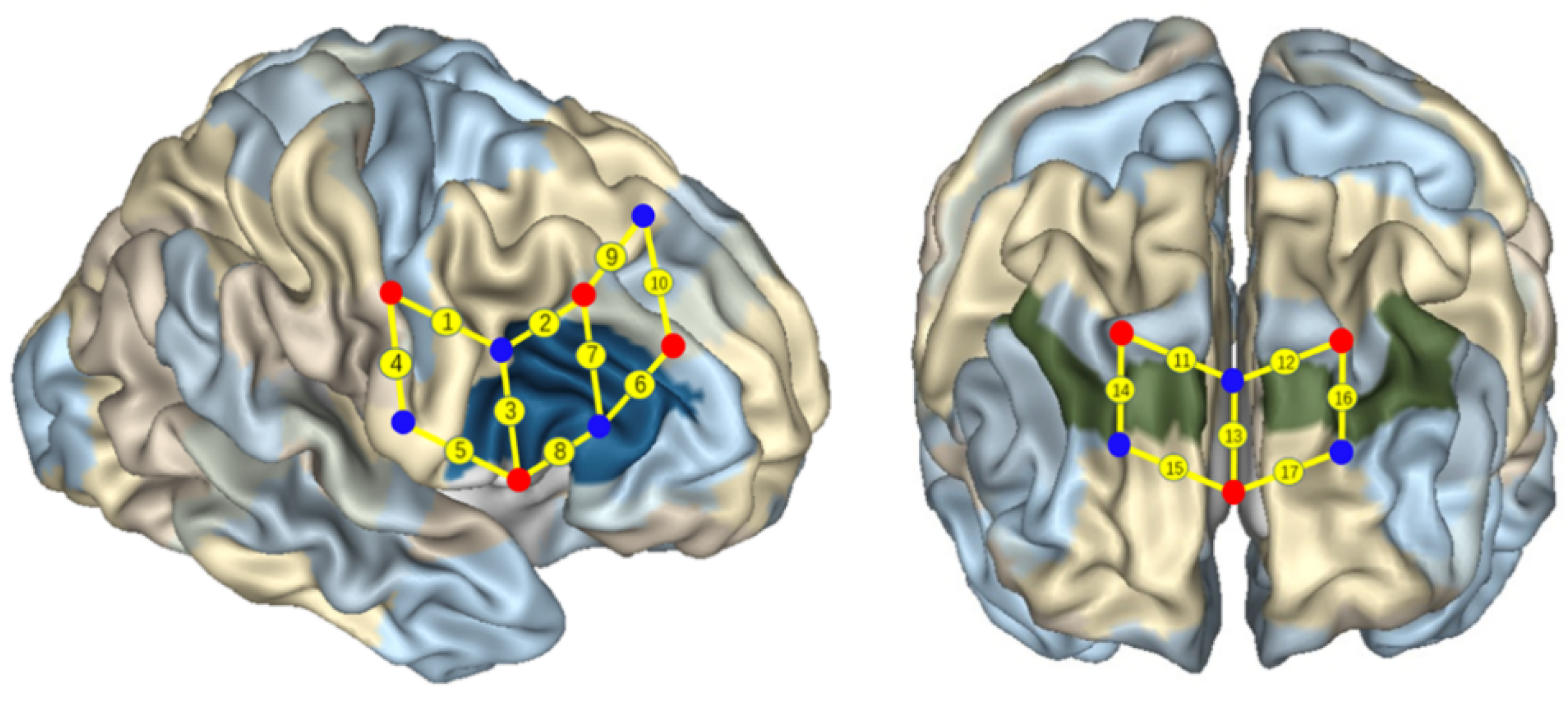
2.2.3. The fNIRS Examination
2.3. Procedure
2.3.1. Data Collecting
2.3.2. Data Processing
2.3.3. Data Analysis
3. Results
3.1. Behavioral Results
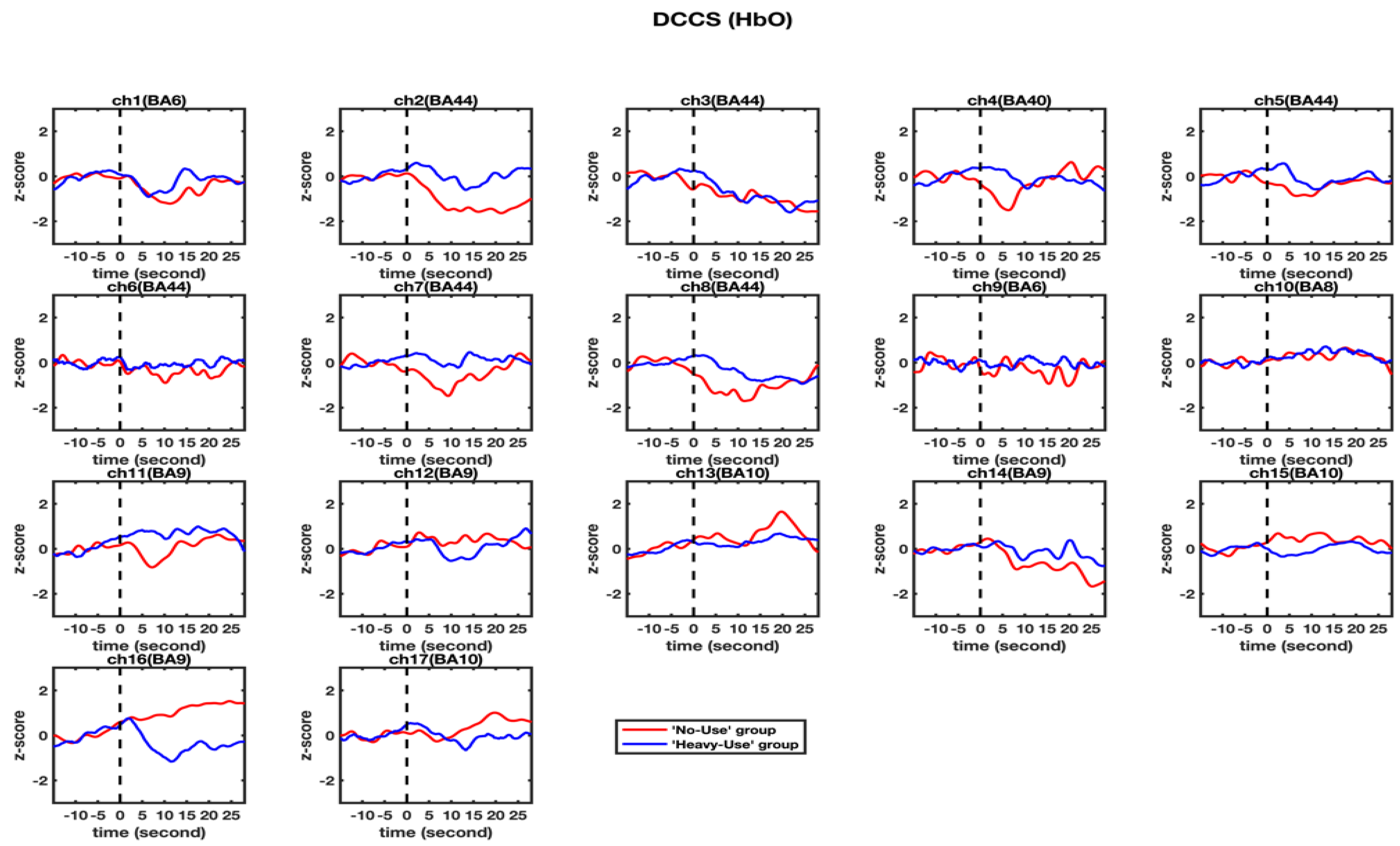
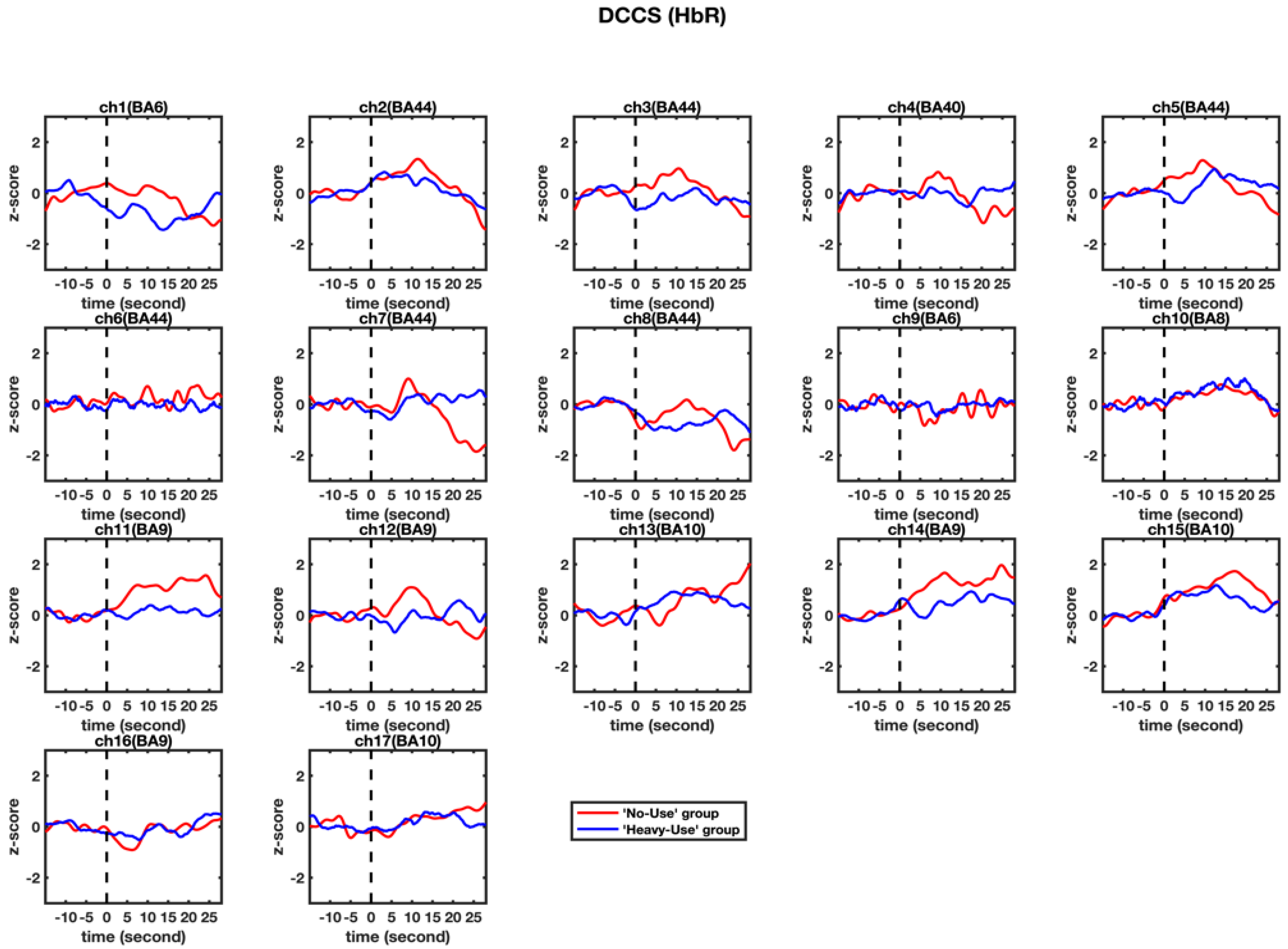
3.2. fNIRS Results
4. Discussion
4.1. ‘Non-User’ Outperformed ‘Heavy-User’ Group in the DCCS
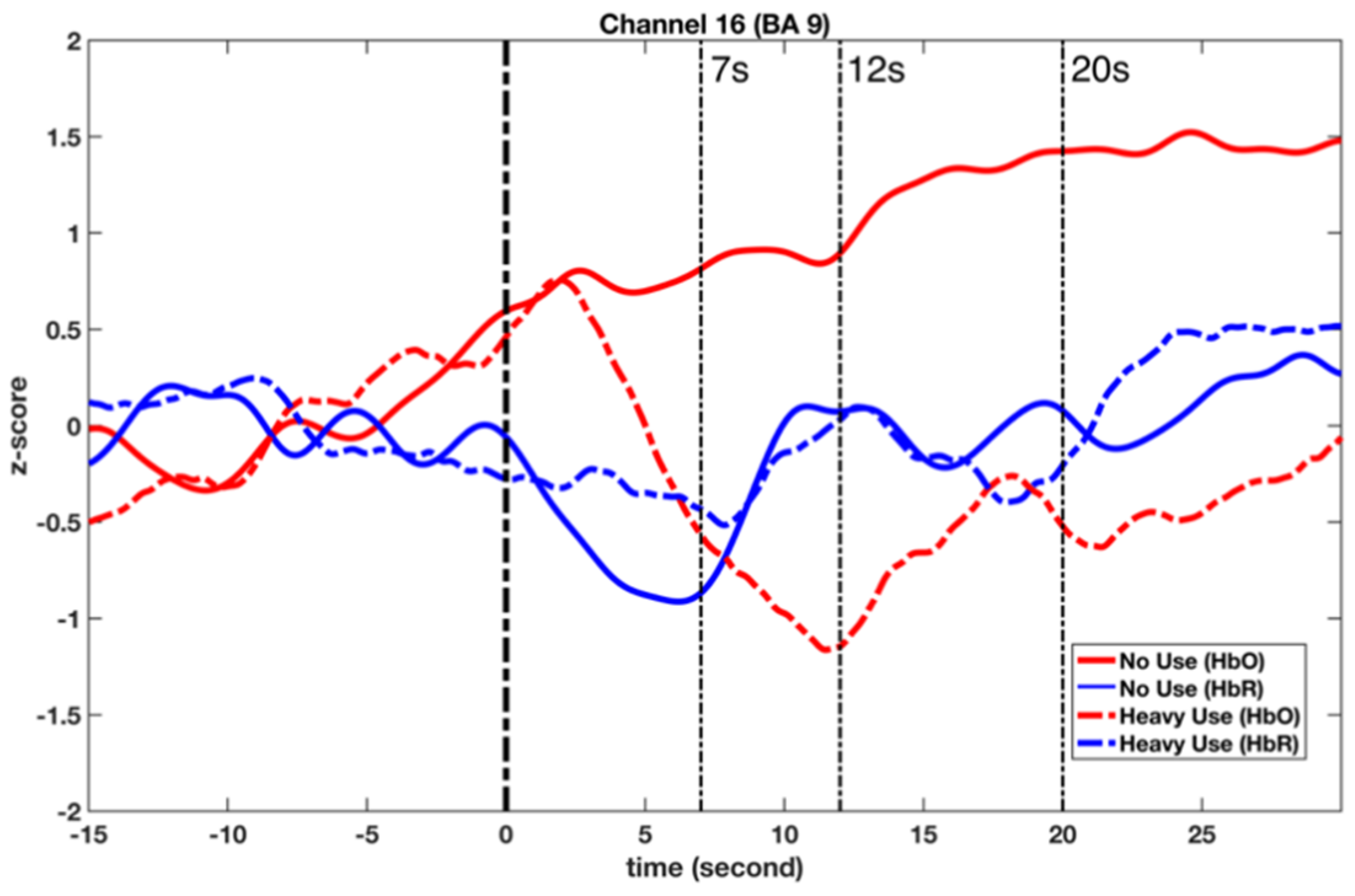
4.2. Two Activation Patterns in BA 9
5. Conclusions, Limitations, and Implications
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Savina, E.; Mills, J.L.; Atwood, K.; Cha, J. Digital Media and Youth: A Primer for School Psychologists. Contemp. Sch. Psychol. 2017, 21, 80–91. [Google Scholar] [CrossRef]
- Rocha, B.; Nunes, C. Benefits and damages of the use of touchscreen devices for the development and behavior of children under 5 years old—A systematic review. Psicologia Reflexão e Crítica 2020, 33, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Small, G.W.; Lee, J.; Kaufman, A.; Jalil, J.; Siddarth, P.; Gaddipati, H.; Moody, T.D.; Bookheimer, S.Y. Brain health consequences of digital technology use. Dialog Clin. Neurosci. 2020, 22, 179–187. [Google Scholar] [CrossRef]
- Beatty, C.; Egan, S.M. Screen time in early childhood: A review of prevalence, evidence and guidelines. An Leanhb Óg 2020, 13, 17–31. [Google Scholar]
- McEwen, R.; Dubé, A.K. Intuitive or idiomatic: An interdisciplinary study of child-tablet computer interaction. J. Assoc. Inf. Sci. Technol. 2015, 67, 1169–1181. [Google Scholar] [CrossRef]
- Neumann, M.M.; Neumann, D.L. Touch Screen Tablets and Emergent Literacy. J. Fam. Econ. Issues 2013, 42, 231–239. [Google Scholar] [CrossRef]
- Dong, C.; Cao, S.; Li, H. Young children’s online learning during COVID-19 pandemic: Chinese parents’ beliefs and attitudes. Child. Youth Serv. Rev. 2020, 118, 105440. [Google Scholar] [CrossRef]
- Sivrikova, N.V.; Ptashko, T.G.; Perebeynos, A.E.; Chernikova, E.G.; Gilyazeva, N.V.; Vasilyeva, V.S. Parental reports on digital devices use in infancy and early childhood. Educ. Inf. Technol. 2020, 25, 3957–3973. [Google Scholar] [CrossRef]
- Lawrence, S.M. Preschool Children and iPads: Observations of Social Interactions During Digital Play. Early Educ. Dev. 2017, 29, 207–228. [Google Scholar] [CrossRef]
- Xie, S.; Wu, D.; Yang, J.; Luo, J.; Chang, C.; Li, H. An fNIRS examination of executive function in bilingual young children. Int. J. Biling. 2020. [Google Scholar] [CrossRef]
- Li, H.; Wu, D.; Yang, J.; Xie, S.; Luo, J.; Chang, C. A Functional Near-Infrared Spectroscopy Examination of the Neural Correlates of Cognitive Shifting in Dimensional Change Card Sort Task. Front. Hum. Neurosci. 2021, 14, 561223. [Google Scholar] [CrossRef] [PubMed]
- Miyake, A.; Friedman, N.P.; Emerson, M.J.; Witzki, A.H.; Howerter, A.; Wager, T.D. The Unity and Diversity of Executive Functions and Their Contributions to Complex “Frontal Lobe” Tasks: A Latent Variable Analysis. Cogn. Psychol. 2000, 41, 49–100. [Google Scholar] [CrossRef] [PubMed]
- Buss, A.T.; Spencer, J.P. Changes in frontal and posterior cortical activity underlie the early emergence of executive function. Dev. Sci. 2017, 21, e12602. [Google Scholar] [CrossRef] [PubMed]
- Moriguchi, Y.; Hiraki, K. Neural origin of cognitive shifting in young children. Proc. Natl. Acad. Sci. USA 2009, 106, 6017–6021. [Google Scholar] [CrossRef] [PubMed]
- Moriguchi, Y. Neural mechanisms of executive function development during early childhood. In Executive Function; Wiebe, S.A., Karbach, J., Eds.; Routledge: New York, NY, USA, 2017; pp. 89–104. [Google Scholar]
- Blair, C. Developmental Science and Executive Function. Curr. Dir. Psychol. Sci. 2016, 25, 3–7. [Google Scholar] [CrossRef]
- NAEYC and Fred Rogers Center for Early Learning. In Technology and Interactive Media as Tools in Early Childhood Programs Serving Children from Birth through Age 8; Position Statement; NAEYC: Washington, DC, USA, 2012.
- Rideout, V. The Common Sense Census: Media Use by Kids Age Zero to Eight; Common Sense Media: San Francisco, CA, USA, 2017. [Google Scholar]
- Li, H.; Rao, N. Parental influences on Chinese literacy development: A comparison of preschoolers in Beijing, Hong Kong, and Singapore. Int. J. Behav. Dev. 2000, 24, 82–90. [Google Scholar] [CrossRef]
- LI, H. Teaching Chinese Literacy in the Early Years: A Comparison of L1 and L2 Preschool Classrooms in Shenzhen and Singapore. Asia Pac. J. Res. Early Child. Educ. 2013, 7, 3. [Google Scholar]
- Duncan, A.; Meek, J.H.; Clemence, M.; Elwell, C.E.; Fallon, P.; Tyszczuk, L.; Cope, M.; Delpy, D.T. Measurement of Cranial Optical Path Length as a Function of Age Using Phase Resolved Near Infrared Spectroscopy. Pediatr. Res. 1996, 39, 889–894. [Google Scholar] [CrossRef]
- Horowitz-Kraus, T.; Hutton, J.S. Brain connectivity in children is increased by the time they spend reading books and decreased by the length of exposure to screen-based media. Acta Paediatr. 2018, 107, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Lachert, P.; Janusek, D.; Pulawski, P.; Liebert, A.; Milej, D.; Blinowska, K.J. Coupling of Oxy- and Deoxyhemoglobin concentrations with EEG rhythms during motor task. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Scholkmann, F.; Metz, A.J.; Wolf, M. Measuring tissue hemodynamics and oxygenation by continuous-wave functional near-infrared spectroscopy—how robust are the different calculation methods against movement artifacts? Physiol. Meas. 2014, 35, 717–734. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.K.; Tremblay, J.; Pouliot, P.; Vannasing, P.; Florea, O.; Carmant, L.; Lepore, F.; Sawan, M.; Lesage, F.; Lassonde, M. Non-invasive continuous EEG-fNIRS recording of temporal lobe seizures. Epilepsy Res. 2012, 99, 112–126. [Google Scholar] [CrossRef] [PubMed]
Short Biography of Authors

| Non-Users Mean (SD) | Heavy Users Mean (SD) | t | p | |
|---|---|---|---|---|
| Age | 5.03 (0.41) | 4.80 (0.68) | 0.850 | 0.404 |
| DCCS Score | 1 (0) | 0.922 (0.14) | 2.256 | 0.039 * |
| Group | M | SD | t | p | |
|---|---|---|---|---|---|
| Channel 1 | Non-user | −0.692 | 2.045 | −0.511 | 0.615 |
| Heavy-user | −0.248 | 1.989 | |||
| Channel 2 | Non-user | −1.414 | 1.630 | −1.468 | 0.156 |
| Heavy-user | −0.105 | 2.230 | |||
| Channel 3 | Non-user | −1.024 | 1.454 | 0.011 | 0.991 |
| Heavy-user | −1.034 | 2.297 | |||
| Channel 4 | Non-user | −0.269 | 1.550 | −0.178 | 0.86 |
| Heavy-user | −0.146 | 1.628 | |||
| Channel 5 | Non-user | −0.439 | 2.180 | −0.318 | 0.753 |
| Heavy-user | −0.176 | 1.767 | |||
| Channel 6 | Non-user | −0.460 | 0.698 | −1.162 | 0.258 |
| Heavy-user | −0.045 | 0.877 | |||
| Channel 7 | Non-user | −0.660 | 1.699 | −0.803 | 0.431 |
| Heavy-user | 0.111 | 2.423 | |||
| Channel 8 | Non-user | −1.198 | 2.068 | −0.633 | 0.533 |
| Heavy-user | −0.603 | 2.222 | |||
| Channel 9 | Non-user | −0.356 | 0.603 | −1.534 | 0.139 |
| Heavy-user | −0.015 | 0.466 | |||
| Channel 10 | Non-user | 0.360 | 1.667 | −0.172 | 0.865 |
| Heavy-user | 0.465 | 1.256 | |||
| Channel 11 | Non-user | 0.052 | 1.322 | −1.287 | 0.211 |
| Heavy-user | 0.761 | 1.247 | |||
| Channel 12 | Non-user | 0.406 | 2.823 | 0.502 | 0.621 |
| Heavy-user | −0.044 | 1.606 | |||
| Channel 13 | Non-user | 0.790 | 3.030 | 0.457 | 0.652 |
| Heavy-user | 0.352 | 1.702 | |||
| Channel 14 | Non-user | −0.804 | 0.984 | −0.772 | 0.448 |
| Heavy-user | −0.142 | 2.304 | |||
| Channel 15 | Non-user | 0.444 | 2.123 | 0.48 | 0.636 |
| Heavy-user | 0.037 | 1.882 | |||
| Channel 16 | Non-user | 1.166 | 2.019 | 2.285 | 0.032 * |
| Heavy-user | −0.623 | 1.699 | |||
| Channel 17 | Non-user | 0.391 | 1.057 | 0.724 | 0.477 |
| Heavy-user | −0.140 | 1.920 |
| Group | M | SD | t | p | |
|---|---|---|---|---|---|
| Channel 1 | Non-user | −0.305 | 1.768 | 0.604 | 0.552 |
| Heavy-user | −0.895 | 2.452 | |||
| Channel 2 | Non-user | 0.583 | 2.760 | 0.307 | 0.762 |
| Heavy-user | 0.282 | 1.990 | |||
| Channel 3 | Non-user | 0.304 | 1.521 | 0.647 | 0.524 |
| Heavy-user | −0.139 | 1.609 | |||
| Channel 4 | Non-user | −0.083 | 2.041 | −0.041 | 0.967 |
| Heavy-user | −0.045 | 2.186 | |||
| Channel 5 | Non-user | 0.486 | 2.210 | 0.014 | 0.989 |
| Heavy-user | 0.472 | 2.306 | |||
| Channel 6 | Non-user | 0.277 | 0.734 | 1.339 | 0.194 |
| Heavy-user | −0.055 | 0.479 | |||
| Channel 7 | Non-user | −0.289 | 1.472 | −0.566 | 0.577 |
| Heavy-user | 0.178 | 2.076 | |||
| Channel 8 | Non-user | −0.482 | 2.949 | 0.162 | 0.872 |
| Heavy-user | −0.702 | 3.215 | |||
| Channel 9 | Non-user | −0.212 | 0.631 | −0.637 | 0.530 |
| Heavy-user | −0.062 | 0.497 | |||
| Channel 10 | Non-user | 0.470 | 1.988 | −0.253 | 0.802 |
| Heavy-user | 0.634 | 1.186 | |||
| Channel 11 | Non-user | 1.233 | 1.060 | 1.441 | 0.164 |
| Heavy-user | 0.178 | 1.914 | |||
| Channel 12 | Non-user | 0.203 | 1.020 | 0.329 | 0.745 |
| Heavy-user | 0.027 | 1.327 | |||
| Channel 13 | Non-user | 0.683 | 2.835 | −0.042 | 0.966 |
| Heavy-user | 0.724 | 1.848 | |||
| Channel 14 | Non-user | 1.378 | 1.535 | 1.107 | 0.280 |
| Heavy-user | 0.536 | 1.851 | |||
| Channel 15 | Non-user | 1.276 | 1.816 | 0.519 | 0.609 |
| Heavy-user | 0.699 | 2.846 | |||
| Channel 16 | Non-user | −0.167 | 2.602 | −0.072 | 0.943 |
| Heavy-user | −0.109 | 1.395 | |||
| Channel 17 | Non-user | 0.391 | 1.784 | 0.127 | 0.900 |
| Heavy-user | 0.298 | 1.651 |
| Group | β | ΔR2 | F | t |
|---|---|---|---|---|
| Non-user | 0.944 | 0.890 | 803.452 *** | 28.345 *** |
| ‘Heavy-user’ | −0.332 | 0.101 | 12.171 ** | −3.489 ** |
| Group | β | ΔR2 | F | t |
|---|---|---|---|---|
| Non-user | 0.761 | 0.575 | 134.987 *** | 11.618 *** |
| Heavy-user | 0.807 | 0.647 | 182.730 *** | 13.518 *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Wu, D.; Yang, J.; Luo, J.; Xie, S.; Chang, C. Tablet Use Affects Preschoolers’ Executive Function: fNIRS Evidence from the Dimensional Change Card Sort Task. Brain Sci. 2021, 11, 567. https://doi.org/10.3390/brainsci11050567
Li H, Wu D, Yang J, Luo J, Xie S, Chang C. Tablet Use Affects Preschoolers’ Executive Function: fNIRS Evidence from the Dimensional Change Card Sort Task. Brain Sciences. 2021; 11(5):567. https://doi.org/10.3390/brainsci11050567
Chicago/Turabian StyleLi, Hui, Dandan Wu, Jinfeng Yang, Jiutong Luo, Sha Xie, and Chunqi Chang. 2021. "Tablet Use Affects Preschoolers’ Executive Function: fNIRS Evidence from the Dimensional Change Card Sort Task" Brain Sciences 11, no. 5: 567. https://doi.org/10.3390/brainsci11050567
APA StyleLi, H., Wu, D., Yang, J., Luo, J., Xie, S., & Chang, C. (2021). Tablet Use Affects Preschoolers’ Executive Function: fNIRS Evidence from the Dimensional Change Card Sort Task. Brain Sciences, 11(5), 567. https://doi.org/10.3390/brainsci11050567







