Migraine Frequency Decrease Following Prolonged Medical Cannabis Treatment: A Cross-Sectional Study
Abstract
1. Background
2. Materials and Methods
2.1. Subject
2.2. Study Procedure
2.3. Study Questionnaires
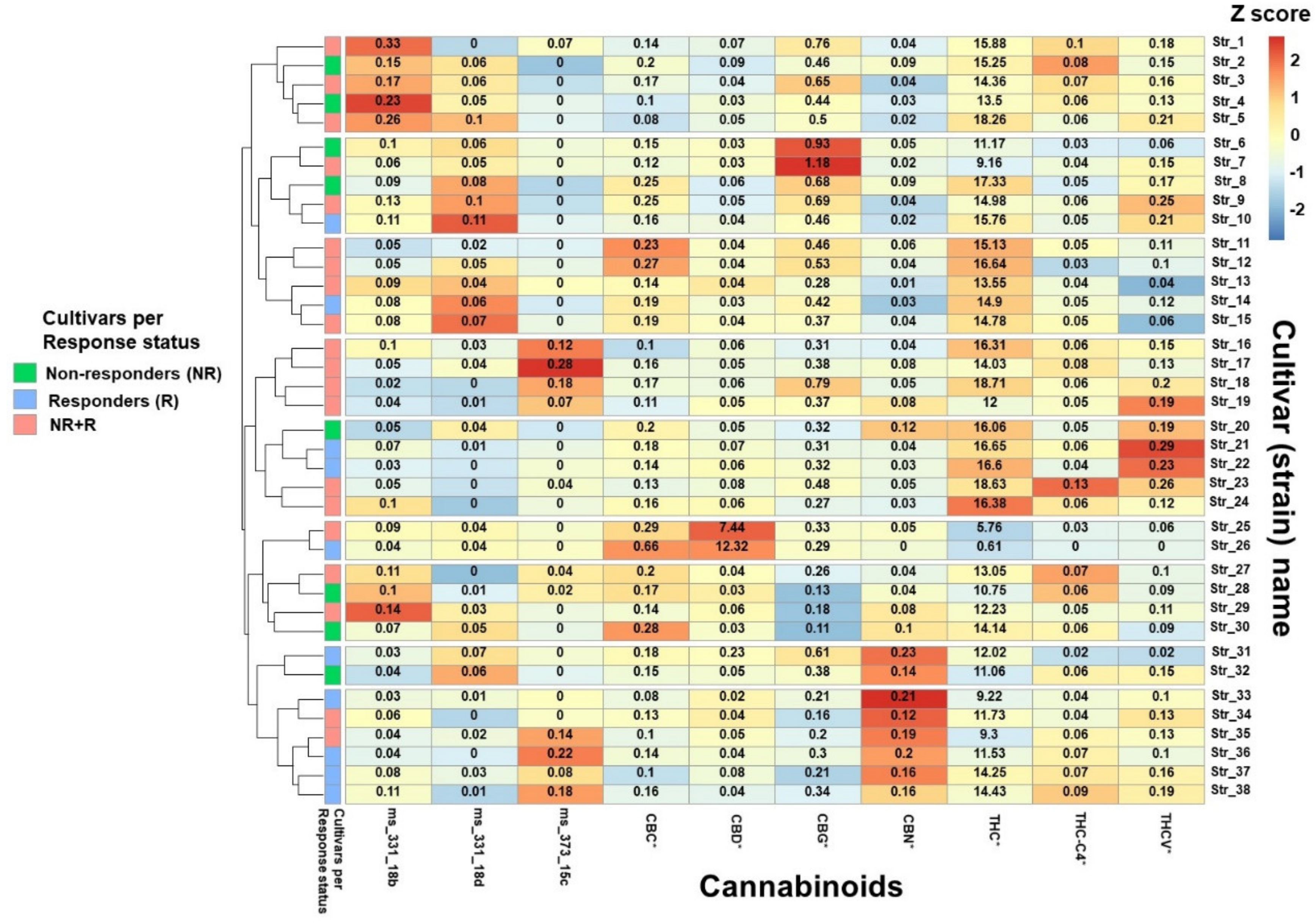
2.4. Phytocannabinoid Profiling of Cannabis Cultivars
2.5. Statistical Analysis
3. Results
3.1. Subjects
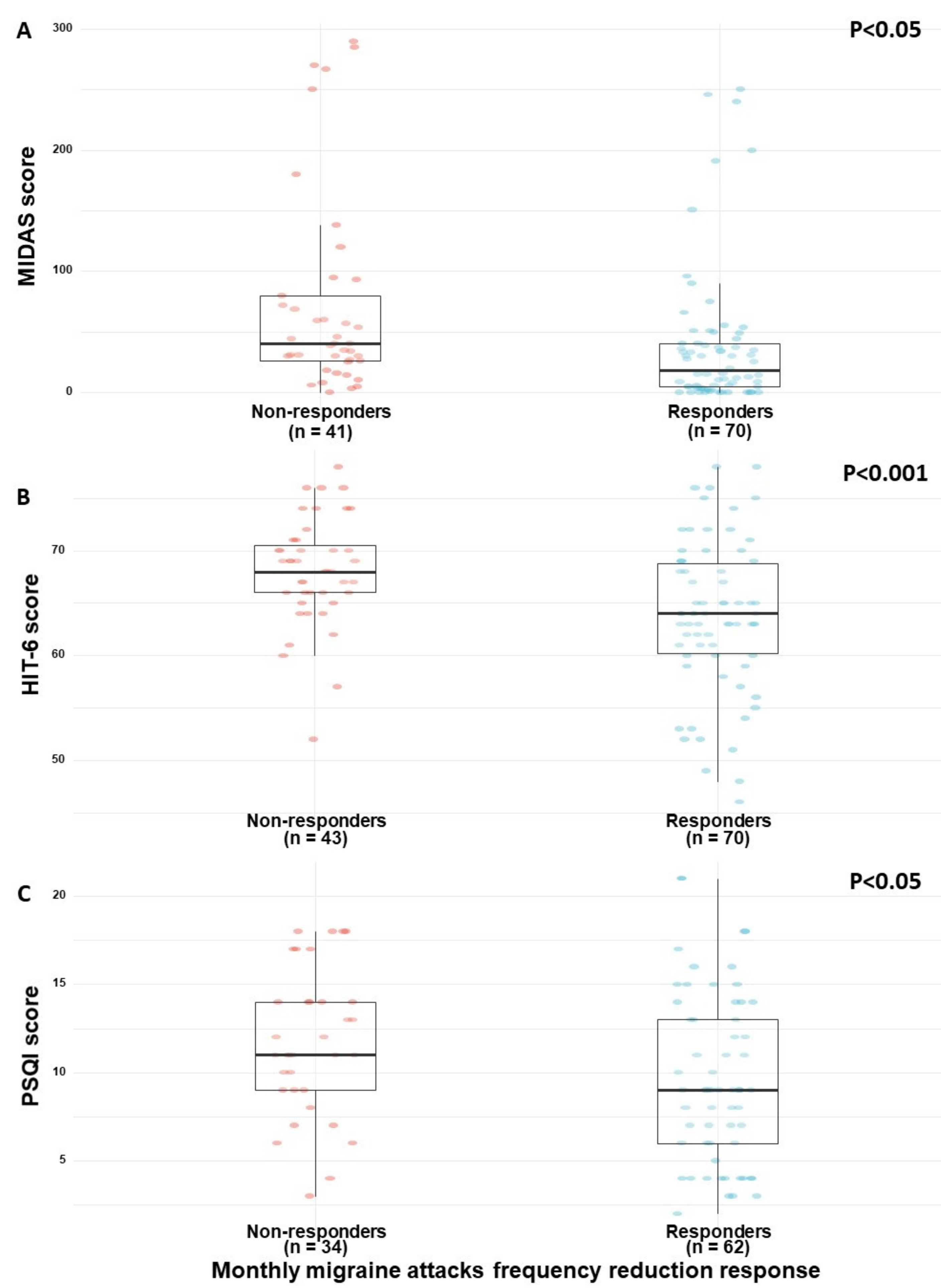
3.2. Migraine and Sleep Features
3.3. MC Treatment Safety
3.4. MC Treatment Complexity
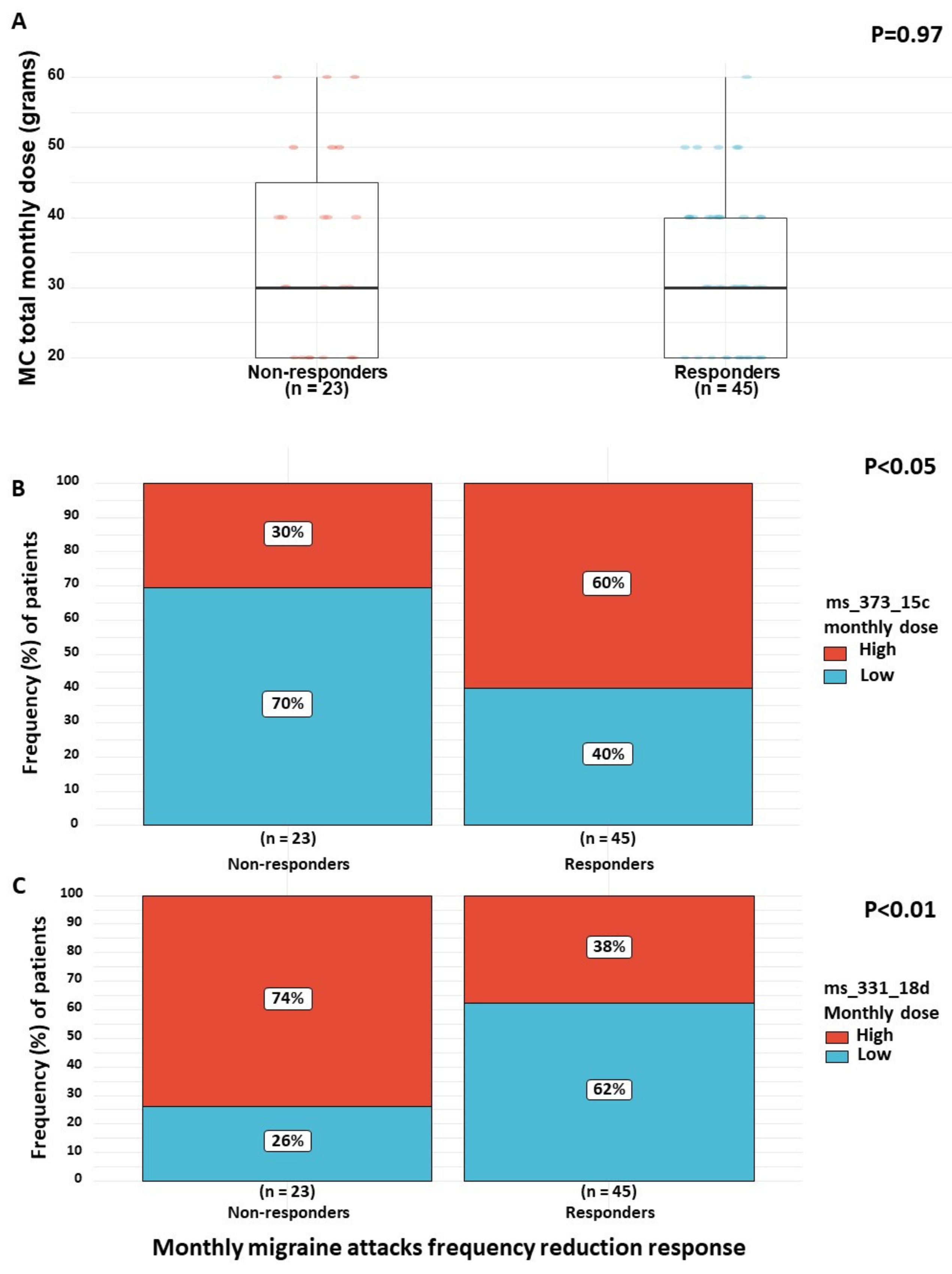
3.5. MC Treatment Characteristics
3.6. Migraine Treatment Characteristics
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Burch, R.C.; Buse, D.C.; Lipton, R.B. Migraine: Epidemiology, Burden, and Comorbidity. Neurol. Clin. 2019, 37, 631–649. [Google Scholar] [CrossRef] [PubMed]
- Cameron, C.; Kelly, S.; Hsieh, S.C.; Murphy, M.; Chen, L.; Kotb, A.; Peterson, J.; Coyle, D.; Skidmore, B.; Gomes, T.; et al. Triptans in the Acute Treatment of Migraine: A Systematic Review and Network Meta-Analysis. Headache 2015, 55, 221–235. [Google Scholar] [CrossRef] [PubMed]
- Pardutz, A.; Schoenen, J. NSAIDs in the acute treatment of migraine: A review of clinical and experimental data. Pharmaceuticals 2010, 3, 1966–1987. [Google Scholar] [CrossRef] [PubMed]
- Derry, S.; Moore, R.A. Paracetamol (acetaminophen) with or without an antiemetic for acute migraine headaches in adults. Cochrane Database Syst. Rev. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Tfelt-Hansen, P.; Saxena, P.R.; Dahlöf, C.; Pascual, J.; Láinez, M.; Henry, P.; Diener, H.-C.; Schoenen, J.; Ferrari, M.D.; Goadsby, P.J. Ergotamine in the acute treatment of migraine: A review and European consensus. Brain 2000, 123, 9–18. [Google Scholar] [CrossRef]
- Kelley, N.E.; Tepper, D.E. Rescue Therapy for Acute Migraine, Part 3: Opioids, NSAIDs, Steroids, and Post-Discharge Medications. Headache J. Head Face Pain 2012, 52, 467–482. [Google Scholar] [CrossRef]
- Becker, W.J. Acute Migraine Treatment in Adults. Headache J. Head Face Pain 2015, 55, 778–793. [Google Scholar] [CrossRef]
- Silberstein, S.D. Preventive migraine treatment. Contin. Lifelong Learn. Neurol. 2015, 21, 973–989. [Google Scholar] [CrossRef]
- Sexton, M.; Cuttler, C.; Finnell, J.S.; Mischley, L.K. A Cross-Sectional Survey of Medical Cannabis Users: Patterns of Use and Perceived Efficacy. Cannabis Cannabinoid Res. 2016, 1, 131–138. [Google Scholar] [CrossRef]
- Cuttler, C.; Spradlin, A.; Cleveland, M.J.; Craft, R.M. Short- and Long-Term Effects of Cannabis on Headache and Migraine. J. Pain 2019. [Google Scholar] [CrossRef]
- Sarchielli, P.; Pini, L.A.; Coppola, F.; Rossi, C.; Baldi, A.; Mancini, M.L.; Calabresi, P. Endocannabinoids in Chronic Migraine: CSF Findings Suggest a System Failure. Neuropsychopharmacology 2007, 32, 1384–1390. [Google Scholar] [CrossRef] [PubMed]
- Cupini, L.M.; Costa, C.; Sarchielli, P.; Bari, M.; Battista, N.; Eusebi, P.; Calabresi, P.; Maccarrone, M. Degradation of endocannabinoids in chronic migraine and medication overuse headache. Neurobiol. Dis. 2008, 30, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Greco, R.; Mangione, A.S.; Sandrini, G.; Maccarrone, M.; Nappi, G.; Tassorelli, C. Effects of anandamide in migraine: Data from an animal model. J. Headache Pain 2011, 12, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Kandasamy, R.; Dawson, C.T.; Craft, R.M.; Morgan, M.M. Anti-migraine effect of ∆9-tetrahydrocannabinol in the female rat. Eur. J. Pharmacol. 2018, 818, 271–277. [Google Scholar] [CrossRef]
- ElSohly, M.A.; Slade, D. Chemical constituents of marijuana: The complex mixture of natural cannabinoids. Life Sci. 2005, 78, 539–548. [Google Scholar] [CrossRef]
- Russo, E.B. Taming THC: Potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. Br. J. Pharmacol. 2011, 163, 1344–1364. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Fride, E.; Sheskin, T.; Tamiri, T.; Rhee, M.H.; Vogel, Z.; Bisogno, T.; De Petrocellis, L.; Di Marzo, V.; Mechoulam, R. An entourage effect: Inactive endogenous fatty acid glycerol esters enhance 2-arachidonoyl-glycerol cannabinoid activity. Eur. J. Pharmacol. 1998, 353, 23–31. [Google Scholar] [CrossRef]
- Baram, L.; Peled, E.; Berman, P.; Yellin, B.; Besser, E.; Benami, M.; Louria-Hayon, I.; Lewitus, G.M.; Meiri, D. The heterogeneity and complexity of Cannabis extracts as antitumor agents. Oncotarget 2019, 10, 4091–4106. [Google Scholar] [CrossRef]
- Berman, P.; Futoran, K.; Lewitus, G.M.; Mukha, D.; Benami, M.; Shlomi, T.; Meiri, D. A new ESI-LC/MS approach for comprehensive metabolic profiling of phytocannabinoids in Cannabis. Sci. Rep. 2018, 8, 1–15. [Google Scholar] [CrossRef]
- Landshaft, Y.; Albo, B.; Mechoulam, R.; Afek, A. The Updated Green Book (May 2019): The Official Guide to Clinical Care in Medical Cannabis. Available online: https://www.health.gov.il/hozer/mmk154_2016.pdf (accessed on 8 June 2020).
- Qualtrics, L.L.C. Qualtrics (Version 12018); Qualtrics Labs Inc.: Provo, UT, USA, 2015; Available online: http//www.qualtrics.com (accessed on 10 May 2020).
- Stewart, W.F.; Lipton, R.B.; Dowson, A.J.; Sawyer, J. Development and testing of the Migraine Disability Assessment (MIDAS) Questionnaire to assess headache-related disability. Neurology 2001, 56, S20–S28. [Google Scholar] [CrossRef]
- Yang, M.; Rendas-Baum, R.; Varon, S.F.; Kosinski, M. Validation of the Headache Impact Test (HIT-6TM) across episodic and chronic migraine. Cephalalgia 2011, 31, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Shochat, T.; Tzischinsky, O.; Oksenberg, A.; Peled, R. Validation of the Pittsburgh Sleep Quality Index Hebrew translation (PSQI-H) in a sleep clinic sample. Isr. Med. Assoc. J. 2007, 9, 853–856. [Google Scholar] [PubMed]
- Wickham, H. Tidyverse: Easily Install and Load ’Tidyverse" Packages; Version 1.3.0; Comprehensive R Archive Network (CRAN): Vienna, Austria, 2019. [Google Scholar]
- Kolde, R. R Package pheatmap: Pretty Heatmaps; Version 1.0.8; Comprehensive R Archive Network (CRAN): Vienna, Austria, 2015. [Google Scholar]
- Ströbel, A.; Haynes, A. R Package a Table: Create Tables for Reporting Clinical Trials; Version 0.1.5; Comprehensive R Archive Network (CRAN): Vienna, Austria, 2019. [Google Scholar]
- Christensen, C.E.; Younis, S.; Deen, M.; Khan, S.; Ghanizada, H.; Ashina, M. Migraine induction with calcitonin gene-related peptide in patients from erenumab trials. J. Headache Pain 2018, 19, 105. [Google Scholar] [CrossRef] [PubMed]
- Hergenrather, J.Y.; Aviram, J.; Vysotski, Y.; Campisi-pinto, S.; Lewitus, G.M.; Meiri, D. Cannabinoid and Terpenoid Doses are Associated with Adult ADHD Status of Medical Cannabis Patients. Rambam Maimonides Med. J. 2020, 11, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rhyne, D.N.; Anderson, S.L.; Gedde, M.; Borgelt, L.M. Effects of Medical Marijuana on Migraine Headache Frequency in an Adult Population. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2016, 36, 505–510. [Google Scholar] [CrossRef]
- Fan, P. Cannabinoid agonists inhibit the activation of 5-HT3receptors in rat nodose ganglion neurons. J. Neurophysiol. 1995, 73, 907–910. [Google Scholar] [CrossRef]
- Lichtman, A.H.; Cook, S.A.; Martin, B.R. Investigation of Brain Sites Mediating Cannabinoid-Induced Antinociception in Rats: Evidence Supporting Periaqueductal Gray Involvement. J. Pharmacol. Exp. Ther. 1996, 276, 585–593. [Google Scholar]
- Goadsby, P.J.; Gundlach, A.L. Localization of 3H-Dihydroergotamine-binding sites in the cat central nervous system: Relevance to migraine. Ann. Neurol. 1991, 29, 91–94. [Google Scholar] [CrossRef]
- Magnusson, J.E.; Becker, W.J. Migraine Frequency and Intensity: Relationship with Disability and Psychological Factors. Headache 2003, 43, 1049–1059. [Google Scholar] [CrossRef]
- Lin, Y.K.; Lin, G.Y.; Lee, J.T.; Lee, M.S.; Tsai, C.K.; Hsu, Y.W.; Lin, Y.Z.; Tsai, Y.C.; Yang, F.C. Associations between Sleep Quality and Migraine Frequency. Medicine USA 2016, 95, 1–7. [Google Scholar] [CrossRef]
- Bradford, A.C.; Bradford, W.D.; Abraham, A.; Adams, G.B. Association between US state medical cannabis laws and opioid prescribing in the Medicare Part D population. JAMA Intern. Med. 2018, 178, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Haroutounian, S.; Meidan, R.; Davidson, E. The Effect of Medicinal Cannabis on Pain and Quality of Life Outcomes in Chronic Pain: A Prospective Open-label Study. Clin. J. Pain 2016, 32, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- McCarty, D. Does Medical Cannabis Reduce Use of Prescription Opioids? Am. J. Psychiatry 2018, 1, 6–7. [Google Scholar] [CrossRef] [PubMed]
- Stith, S.S.; Vigil, J.M.; Adams, I.M.; Reeve, A.P. Effects of Legal Access to Cannabis on Scheduled II–V Drug Prescriptions. J. Am. Med. Dir. Assoc. 2017, 19, 59–64. [Google Scholar] [CrossRef]
- Yassin, M.; Garti, A.; Robinson, D. Effect of Medicinal Cannabis Therapy (MCT) on Severity of Chronic Low Back Pain, Sciatica and Lumbar Range of Motion. Int. J. Anesthesiol. Pain Med. 2016, 2, 1–6. [Google Scholar] [CrossRef]
- Loder, E. Triptan Therapy in Migraine. N. Engl. J. Med. 2010, 363, 63–70. [Google Scholar] [CrossRef]
- Russo, E. Cannabis for migraine treatment: The once and future prescription? An historical and scientific review. Pain 1998, 76, 3–8. [Google Scholar] [CrossRef]
- Baron, E.P.; Lucas, P.; Eades, J.; Hogue, O. Patterns of medicinal cannabis use, strain analysis, and substitution effect among patients with migraine, headache, arthritis, and chronic pain in a medicinal cannabis cohort. J. Headache Pain 2018, 19, 37. [Google Scholar] [CrossRef]
- Hazekamp, A.; Fischedick, J.T. Cannabis—From cultivar to chemovar. Drug Test. Anal. 2012, 4, 660–667. [Google Scholar] [CrossRef]
| Non-Responders N = 56 | Responders N = 89 | Total N = 145 | Statistic (p) | Effect Size (CI) | |
|---|---|---|---|---|---|
| Measure | Number of patients (%) | ||||
| Gender | |||||
| Female | 35 (62) | 62 (70) | 97 (67) | 0.51 (0.48) | 0.73 (0.34–1.6) |
| Male | 21 (38) | 27 (30) | 48 (33) | ||
| Missing N | 0 | 0 | 0 | ||
| Median (IQR) | |||||
| Age (years) | 46 (35–54) | 44 (34–54) | 45 (34–54) | 0.08 (0.96) | −0.02 (−0.37–0.32) |
| Missing N | 3 | 3 | 6 | ||
| BMI | 25 (22–27) | 25 (22–28) | 25 (22–27) | 0.13 (0.64) | 0.05 (−0.28–0.39) |
| Missing N | 0 | 1 | 1 | ||
| MC treatment duration (years) | 3.5 (2.8–5.2) | 3 (2–4) | 3 (2.4–4.6) | 0.22 (0.09) | 0.46 (0.10–0.81) |
| Missing N | 4 | 5 | 9 | ||
| Measure | Number of patients (%) | ||||
| MC administration route | |||||
| Inflorescence | 40 (71) | 72 (81) | 112 (77) | 2.4 (0.31) | 0.13 (0–0.29) |
| Oil extract | 7 (12) | 5 (6) | 12 (8) | ||
| Combination # | 7 (12) | 12 (13) | 19 (13) | ||
| Missing N | 2 | 0 | |||
| Inflorescence administration method * | |||||
| Pure MC cigarettes | 22 (39) | 33 (37) | 55 (38) | 0.04 (0.84) | 1.10 (0.54–2.40) |
| MC cigarettes mixed with tobacco | 17 (30) | 30 (34) | 47 (32) | 0.01 (0.89) | 0.89 (0.40–1.90) |
| Bhang | 3 (5) | 11 (12) | 14 (10) | 1.1 (0.29) | 0.41 (0.07–1.70) |
| Electronic vaporizer | 14 (25) | 15 (17) | 29 (20) | 1.1 (0.29) | 0.59 (0.24–1.50) |
| Manual vaporizer | 5 (9) | 20 (22) | 25 (17) | 3.3 (0.06) | 2.9 (0.96–10.00) |
| Missing N | 2 | 1 | |||
| Oil extract administration method * | |||||
| Sublingual | 13 (23) | 13 (15) | 26 (18) | 1.4 (0.24) | 0.55 (0.21–1.40) |
| Swallowing | 2 (4) | 1 (1) | 3 (2) | 0.19 (0.67) | 0.30 (0.005–5.90) |
| Missing N | 2 | 1 | |||
| Non-Responders N = 56 | Responders N = 89 | Statistic (p) | Effect Size (CI) | |
|---|---|---|---|---|
| Measure | Median (IQR) | |||
| Age of migraine diagnosis (years) | 20 (14–36) | 22 (14–32) | 0.07 (0.98) | 0.07 (−0.27–0.42) |
| Missing N | 1 | 4 | ||
| Average migraine duration (hours) | 20 (5.8–35) | 15 (5–48) | 0.12 (0.72) | 0.15 (−0.19–0.49) |
| Missing N | 1 | 2 | ||
| Number of patients (%) | ||||
| Activity induced aggravation of migraine | 32 (57) | 61 (69) | 1.20 (0.28) | 1.60 (0.73–3.3) |
| Missing N | 1 | 0 | ||
| Unilateral migraine | 40 (71) | 59 (66) | 0.39 (0.53) | 0.74 (0.33–1.60) |
| Missing N | 1 | 0 | ||
| Aura+ | 16 (29) | 31 (35) | 0.28 (0.60) | 1.30 (0.60–2.9) |
| Missing N | 1 | 0 | ||
| Nausea+ | 25 (45) | 51 (57) | 1.50 (0.23) | 1.60 (0.78–3.40) |
| Missing N | 1 | 0 | ||
| Phono/photo phobia+ | 38 (68) | 60 (67) | 0.00 (0.98) | 0.93 (0.42–2.00) |
| Missing N | 1 | 0 | ||
| Non-Responders N = 56 | Responders N = 89 | Statistic (p) | Effect Size (CI) | |
| Measure | Median (IQR) | |||
| Sleep quality global score (PSQI, 0–21) | 11 (9–14) | 9 (6–13) | 0.30 (0.04) | 0.46 (0.03–0.89) |
| Missing N | 22 | 27 | ||
| Sleep latency (minutes) | 32 (20–60) | 30 (15–60) | 0.09 (0.97) | −0.07 (−0.46–0.33) |
| Missing N | 16 | 21 | ||
| Sleep duration (hours) | 6.2 (5–7) | 6 (5–7) | 0.11 (0.92) | −0.09 (−0.49–0.30) |
| Missing N | 16 | 20 | ||
| Subjective sleep quality * | 3 (2–3) | 2.5 (1–3) | 0.18 (0.39) | 0.42 (0.02–0.81) |
| Missing N | 15 | 19 | ||
| Sleep latency * | 2 (1.8–3) | 2 (1–3) | 0.15 (0.65) | 0.2 (−0.20–0.59) |
| Missing N | 16 | 21 | ||
| Sleep duration * | 1 (0–2) | 1 (0–2) | 0.1 (0.95) | −0.02 (−0.41–0.37) |
| Missing N | 16 | 20 | ||
| Habitual sleep efficiency * | 1 (0–2) | 0 (0–2) | 0.09 (0.99) | 0.08 (−0.32–0.49) |
| Missing N | 18 | 22 | ||
| Sleep disturbances * | 2 (2–2) | 2 (1–2) | 0.19 (0.33) | 0.59 (0.19–0.98) |
| Missing N | 15 | 19 | ||
| Use of sleeping medication * | 1 (0–3) | 0 (0–1.2) | 0.19 (0.34) | 0.35 (−0.05–0.75) |
| Missing N | 17 | 21 | ||
| Daytime dysfunction * | 2 (1–2) | 1 (1–2) | 0.18 (0.40) | 0.34 (−0.06–0.74) |
| Missing N | 17 | 23 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aviram, J.; Vysotski, Y.; Berman, P.; Lewitus, G.M.; Eisenberg, E.; Meiri, D. Migraine Frequency Decrease Following Prolonged Medical Cannabis Treatment: A Cross-Sectional Study. Brain Sci. 2020, 10, 360. https://doi.org/10.3390/brainsci10060360
Aviram J, Vysotski Y, Berman P, Lewitus GM, Eisenberg E, Meiri D. Migraine Frequency Decrease Following Prolonged Medical Cannabis Treatment: A Cross-Sectional Study. Brain Sciences. 2020; 10(6):360. https://doi.org/10.3390/brainsci10060360
Chicago/Turabian StyleAviram, Joshua, Yelena Vysotski, Paula Berman, Gil M. Lewitus, Elon Eisenberg, and David Meiri. 2020. "Migraine Frequency Decrease Following Prolonged Medical Cannabis Treatment: A Cross-Sectional Study" Brain Sciences 10, no. 6: 360. https://doi.org/10.3390/brainsci10060360
APA StyleAviram, J., Vysotski, Y., Berman, P., Lewitus, G. M., Eisenberg, E., & Meiri, D. (2020). Migraine Frequency Decrease Following Prolonged Medical Cannabis Treatment: A Cross-Sectional Study. Brain Sciences, 10(6), 360. https://doi.org/10.3390/brainsci10060360





