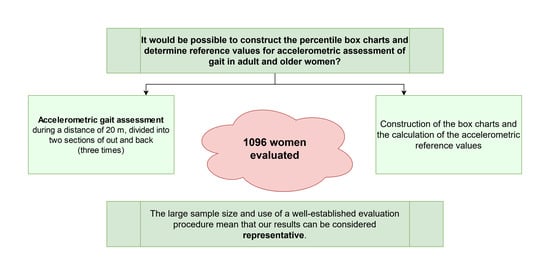
Percentiles and Reference Values for Accelerometric Gait Assessment in Women Aged 50–80 Years
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample
2.2. Procedure
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bischoff-Ferrari, H.; Orav, J.; Kanis, J.; Rizzoli, R.; Schlögl, M.; Staehelin, H.; Willet, W.C.; Dawson-Hughes, B. Comparative performance of current definitions of sarcopenia against the prospective incidence of falls among community-dwelling seniors age 65 and older. Osteoporosis Int. 2015, 26, 2793–2802. [Google Scholar] [CrossRef] [PubMed]
- Finlayson, M.L.; Peterson, E.W. Falls, aging, and disability. Phys. Med. Rehabil. Clin. N. Am. 2010, 21, 357–373. [Google Scholar] [CrossRef] [PubMed]
- Maki, B.E.; Sibley, K.M.; Jaglal, S.B.; Bayley, M.; Brooks, D.; Fernie, G.R.; Flint, A.J.; Gage, W.; Liu, B.A.; McIlroy, W.E.; et al. Reducing fall risk by improving balance control: Development, evaluation and knowledge-translation of new approaches. J. Saf. Res. 2011, 42, 473–485. [Google Scholar] [CrossRef] [PubMed]
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K. Frailty in elderly people. Lancet 2013, 381, 752–762. [Google Scholar] [CrossRef]
- Donath, L.; Faude, O.; Lichtenstein, E.; Nüesch, C.; Mündermann, A. Validity and reliability of a portable gait analysis system for measuring spatiotemporal gait characteristics: Comparison to an instrumented treadmill. J. Neuroeng. Rehabil. 2016, 13, 6. [Google Scholar] [CrossRef] [PubMed]
- McGinley, J.L.; Baker, R.; Wolfe, R.; Morris, M.E. The reliability of three-dimensional kinematic gait measurements: A systematic review. Gait Posture 2009, 29, 360–369. [Google Scholar] [CrossRef]
- Holliday, W.; Fisher, J.; Theo, R.; Swart, J. Static versus dynamic kinematics in cyclists: A comparison of goniometer, inclinometer and 3D motion capture. Eur. J. Sport Sci. 2017, 17, 1129–1142. [Google Scholar] [CrossRef]
- Bruijn, S.M.; Meijer, O.G.; Beek, P.J.; van Dieen, J.H. Assessing the stability of human locomotion: A review of current measures. J. R. Soc. Interface 2013, 10, 20120999. [Google Scholar] [CrossRef]
- Muro, A.; García-Zapiraín, B.; Méndez-Zorrilla, A. Gait analysis methods: An overview of wearable and non-wearable systems, highlighting clinical applications. Sensors 2014, 14, 3362–3394. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Lach, J.; Lo, B.; Yang, G. Toward pervasive gait analysis with wearable sensors: A systematic review. IEEE J. Biomed. Health Inform. 2016, 20, 1521–1537. [Google Scholar] [CrossRef]
- Mapelli, A.; Zago, M.; Fusini, L.; Galante, D.; Colombo, A.; Sforza, C. Validation of a protocol for the estimation of three-dimensional body center of mass kinematics in sport. Gait Posture 2014, 39, 460–465. [Google Scholar] [CrossRef]
- Hodges, P.; Gurfinkel, V.; Brumagne, S.; Smith, T.; Cordo, P. Coexistence of stability and mobility in postural control: Evidence from postural compensation for respiration. Exp. Brain Res. 2002, 144, 293–302. [Google Scholar] [CrossRef]
- Chen, T.; Chou, L. Altered center of mass control during sit-to-walk in elderly adults with and without history of falling. Gait Posture 2013, 38, 696–701. [Google Scholar] [CrossRef] [PubMed]
- Kressig, R.W.; Beauchet, O. Guidelines for clinical applications of spatio-temporal gait analysis in older adults. Aging Clin. Exp. Res. 2006, 18, 174–176. [Google Scholar] [CrossRef]
- Winter, D.A. Human balance and posture control during standing and walking. Gait Posture 1995, 3, 193–214. [Google Scholar] [CrossRef]
- Horak, F.B. Postural orientation and equilibrium: What do we need to know about neural control of balance to prevent falls? Age Ageing 2006, 35, ii7–ii11. [Google Scholar] [CrossRef]
- Leirós-Rodríguez, R.; García-Soidán, J.L.; Romo-Pérez, V. Analyzing the use of accelerometers as a method of early diagnosis of alterations in balance in elderly people: A systematic review. Sensors 2019, 19, 3883. [Google Scholar] [CrossRef]
- Leirós-Rodríguez, R.; Romo-Pérez, V.; García-Soidán, J.L.; García-Liñeira, J. Percentiles and reference values for the Accelerometric assessment of static balance in women aged 50–80 years. Sensors 2020, 20, 940. [Google Scholar] [CrossRef]
- Bouten, C.V.; Koekkoek, K.T.; Verduin, M.; Kodde, R.; Janssen, J.D. A triaxial accelerometer and portable data processing unit for the assessment of daily physical activity. IEEE Trans. Biomed. Eng. 1997, 44, 136–147. [Google Scholar] [CrossRef]
- Preece, S.J.; Goulermas, J.Y.; Kenney, L.P.; Howard, D. A comparison of feature extraction methods for the classification of dynamic activities from accelerometer data. IEEE Trans. Biomed. Eng. 2009, 56, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, A.; Luzi, S.; Murer, K.; de Bie, R.A.; de Bruin, E.D. Concurrent validity of a trunk tri-axial accelerometer system for gait analysis in older adults. Gait Posture 2009, 29, 444–448. [Google Scholar] [CrossRef]
- Buuren, S.; Fredriks, M. Worm plot: A simple diagnostic device for modelling growth reference curves. Stat. Med. 2001, 20, 1259–1277. [Google Scholar] [CrossRef]
- Rigby, R.A.; Stasinopoulos, D.M. The GAMLSS project: A flexible approach to statistical modelling. In New Trends in Statistical Modelling, Proceedings of the 16th International Workshop on Statistical Modelling; Statistical Modelling Society: Odense, Denmark, 2001; pp. 337–345. [Google Scholar]
- Rispens, S.M.; van Schooten, K.S.; Pijnappels, M.; Daffertshofer, A.; Beek, P.J.; van Dieen, J.H. Do extreme values of daily-life gait characteristics provide more information about fall risk than median values? JMIR Res. Protoc. 2015, 4, e4. [Google Scholar] [CrossRef]
- Moe-Nilssen, R.; Helbostad, J.L. Trunk accelerometry as a measure of balance control during quiet standing. Gait Posture 2002, 16, 60–68. [Google Scholar] [CrossRef]
- Thiede, R.; Toosizadeh, N.; Mills, J.L.; Zaky, M.; Mohler, J.; Najafi, B. Gait and balance assessments as early indicators of frailty in patients with known peripheral artery disease. Clin. Biomech. 2015, 32, 1–7. [Google Scholar] [CrossRef]
- Bautmans, I.; Jansen, B.; van Keymolen, B.; Mets, T. Reliability and clinical correlates of 3D-accelerometry based gait analysis outcomes according to age and fall-risk. Gait Posture 2011, 33, 366–372. [Google Scholar] [CrossRef]
- Tadano, S.; Takeda, R.; Sasaki, K.; Fujisawa, T.; Tohyama, H. Gait characterization for osteoarthritis patients using wearable gait sensors (H-Gait systems). J. Biomech. 2016, 49, 684–690. [Google Scholar] [CrossRef]
- Studenski, S.; Perera, S.; Patel, K.; Rosano, C.; Faulkner, K.; Inzitari, M.; Brach, J.; Chandler, J.; Cawthon, P.; Barrett, E.; et al. Gait speed and survival in older adults. JAMA 2011, 305, 50–58. [Google Scholar] [CrossRef]
- Butterworth, P.A.; Landorf, K.; Gilleard, W.; Urquhart, D.; Menz, H. The association between body composition and foot structure and function: A systematic review. Obes. Rev. 2014, 15, 348–357. [Google Scholar] [CrossRef]
- Senden, R.; Savelberg, H.; Grimm, B.; Heyligers, I.; Meijer, K. Accelerometry-based gait analysis, an additional objective approach to screen subjects at risk for falling. Gait Posture 2012, 36, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Senden, R.; Grimm, B.; Heyligers, I.; Savelberg, H.; Meijer, K. Acceleration-based gait test for healthy subjects: Reliability and reference data. Gait Posture 2009, 30, 192–196. [Google Scholar] [CrossRef]
- Rispens, S.M.; van Schooten, K.S.; Pijnappels, M.; Daffertshofer, A.; Beek, P.J.; van Dieen, J.H. Identification of fall risk predictors in daily life measurements: Gait characteristics’ reliability and association with self-reported fall history. Neurorehabil. Neural. Repair 2015, 29, 54–61. [Google Scholar] [CrossRef]
- Kavanagh, J.J. Lower trunk motion and speed-dependence during walking. J. Neuroeng. Rehabil. 2009, 6. [Google Scholar] [CrossRef] [PubMed]
- Camicioli, R.; Howieson, D.; Lehman, S.; Kaye, J. Talking while walking: The effect of a dual task in aging and Alzheimer’s disease. Neurology 1997, 48, 955–958. [Google Scholar] [CrossRef] [PubMed]
- Grimbergen, Y.A.; Schrag, A.; Mazibrada, G.; Borm, G.F.; Bloem, B.R. Impact of falls and fear of falling on health-related quality of life in patients with Parkinson’s disease. J. Parkinsons Dis. 2013, 3, 409–413. [Google Scholar] [CrossRef] [PubMed]

| Age Group | N | Age (Years) | Weight (kg) | Height (cm) | Body Mass Index (kg/m2) |
|---|---|---|---|---|---|
| All | 1096 | 68.8 ± 10.4 | 65.6 ±10.1 | 153.9 ± 5.4 | 27.6 ± 4.1 |
| G1 (51–55 years) | 187 | 53.4 ± 4.4 | 63 ± 7.6 | 155.6 ± 5 | 26 ± 3.3 |
| G2 (56–60 years) | 172 | 57.4 ± 4.3 | 64 ± 6.5 | 154.6 ± 6 | 26.8 ± 5.3 |
| G3 (61–65 years) | 185 | 64.2 ± 2.7 | 66.4 ± 11.1 | 154 ± 5.5 | 28 ± 4.7 |
| G4 (66–70 years) | 192 | 68.4 ± 3.8 | 63.8 ± 9.6 | 152.9 ± 6.1 | 29 ± 6.3 |
| G5 (71–75 years) | 187 | 74.2 ± 4.6 | 66.5 ± 10.1 | 151.8 ± 5.2 | 28.3 ± 3.2 |
| G6 (76–80 years) | 173 | 77.6 ± 2.2 | 68.1 ± 11.7 | 151.3 ± 4.2 | 29.2 ± 1.8 |
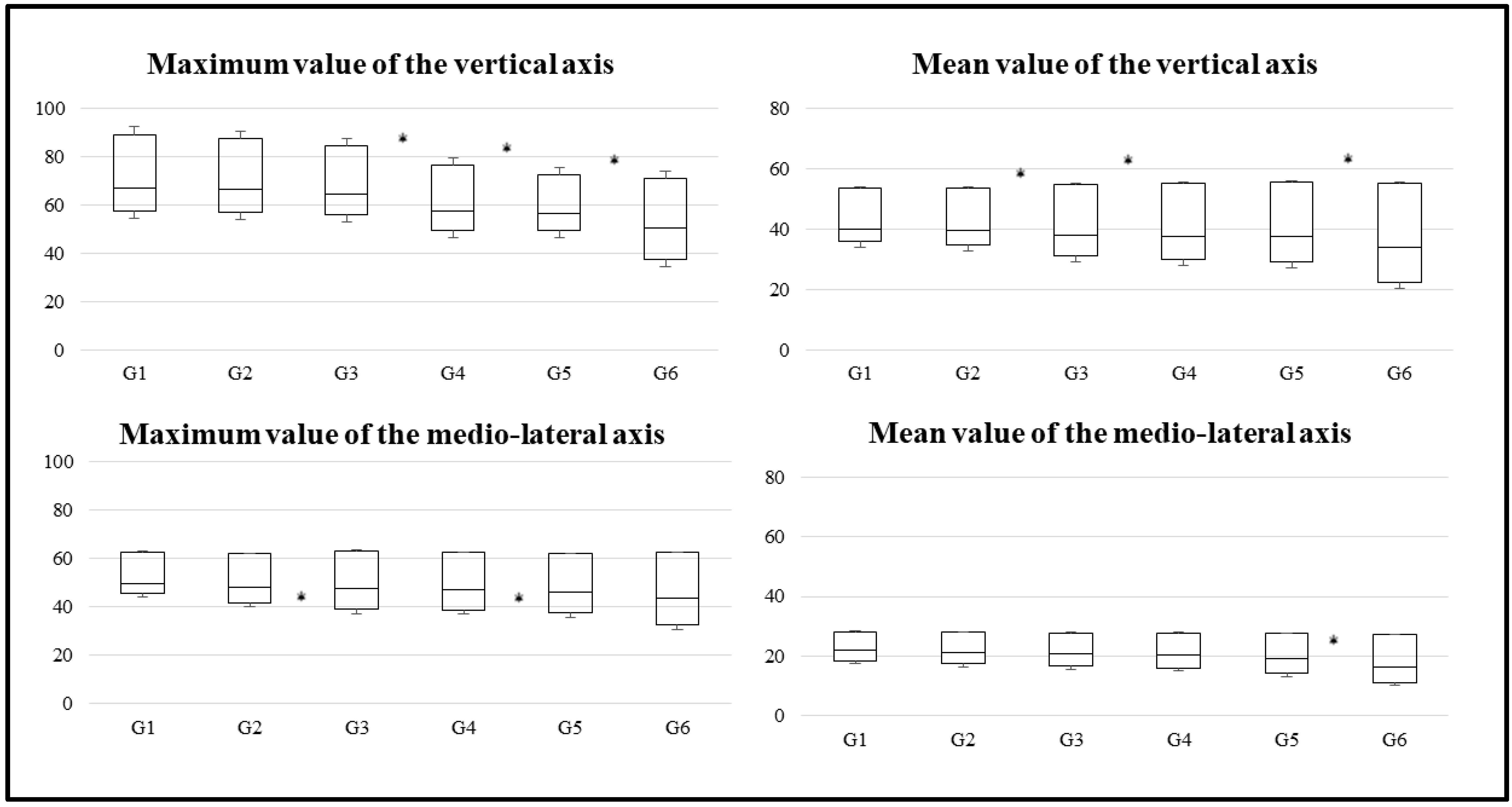
| Variable | G1 (n = 187) | G2 (n = 172) | G3 (n = 185) | G4 (n = 192) | G5 (n = 187) | G6 (n = 173) |
|---|---|---|---|---|---|---|
| Maximum value of vertical axis | ||||||
| Mean ± standard deviation | 67.7 ± 17.4 | 67.1 ± 19.8 | 63.7 ± 16.3 | 56.8 ± 13 | 57.3 ± 11.4 | 51.3 ± 14.1 |
| Kurtosis | 3.9 | 3 | 2.9 | 3.6 | 3.4 | 1.7 |
| Percentile 25 | 57.3 | 57 | 56 | 49.7 | 49.3 | 37.7 |
| Percentile 50 (median) | 67 | 66.3 | 64.3 | 57.3 | 56.3 | 50.7 |
| Percentile 75 | 79.3 | 78 | 76 | 68.7 | 65.3 | 57.7 |
| Interquartile range | 22 | 21 | 20 | 19 | 16 | 20 |
| Mean value of vertical axis | ||||||
| Mean ± standard deviation | 44.1 ± 13.7 | 41.9 ± 13.7 | 41.1 ± 12.6 | 35.9 ± 10 | 36 ± 9.6 | 32.7 ± 11.1 |
| Kurtosis | 3.7 | 2.5 | 3.1 | 3.2 | 3.2 | 1.5 |
| Percentile 25 | 36 | 34.9 | 31 | 29.9 | 29 | 22.6 |
| Percentile 50 (median) | 40 | 39.4 | 38 | 37.8 | 37.5 | 34 |
| Percentile 75 | 49.5 | 48.9 | 47.9 | 47.3 | 47.1 | 43.6 |
| Interquartile range | 13.5 | 14 | 16.9 | 17.5 | 18.1 | 21 |
| Maximum value of mediolateral axis | ||||||
| Mean ± standard deviation | 53.9 ± 16.6 | 52.7 ± 12.7 | 46.9 ± 11.3 | 48.9 ± 12.4 | 45.6 ± 12.1 | 41 ± 10 |
| Kurtosis | 2.8 | 4.6 | 3.8 | 3.3 | 4.5 | 2 |
| Percentile 25 | 45.7 | 41.7 | 39 | 38.7 | 37.3 | 32.3 |
| Percentile 50 (median) | 49.7 | 48 | 47.5 | 47.2 | 46 | 43.3 |
| Percentile 75 | 58.7 | 55.7 | 54.8 | 54.1 | 53.3 | 51.3 |
| Interquartile range | 13 | 14 | 15.8 | 15.4 | 16 | 19 |
| Mean value of mediolateral axis | ||||||
| Mean ± standard deviation | 22.8 ± 9 | 21.9 ± 6.3 | 20.7 ± 6 | 21 ± 6.9 | 19.5 ± 7 | 15.6 ± 4.2 |
| Kurtosis | 2 | 3.1 | 4.2 | 2.4 | 2.2 | 2.4 |
| Percentile 25 | 18.5 | 17.6 | 16.6 | 16.1 | 14.3 | 11.2 |
| Percentile 50 (median) | 22 | 21.3 | 20.8 | 20.5 | 19 | 16.4 |
| Percentile 75 | 24.6 | 24.3 | 23.4 | 23.4 | 22.8 | 21.9 |
| Interquartile range | 6.2 | 6.7 | 6.9 | 7.3 | 8.5 | 10.7 |
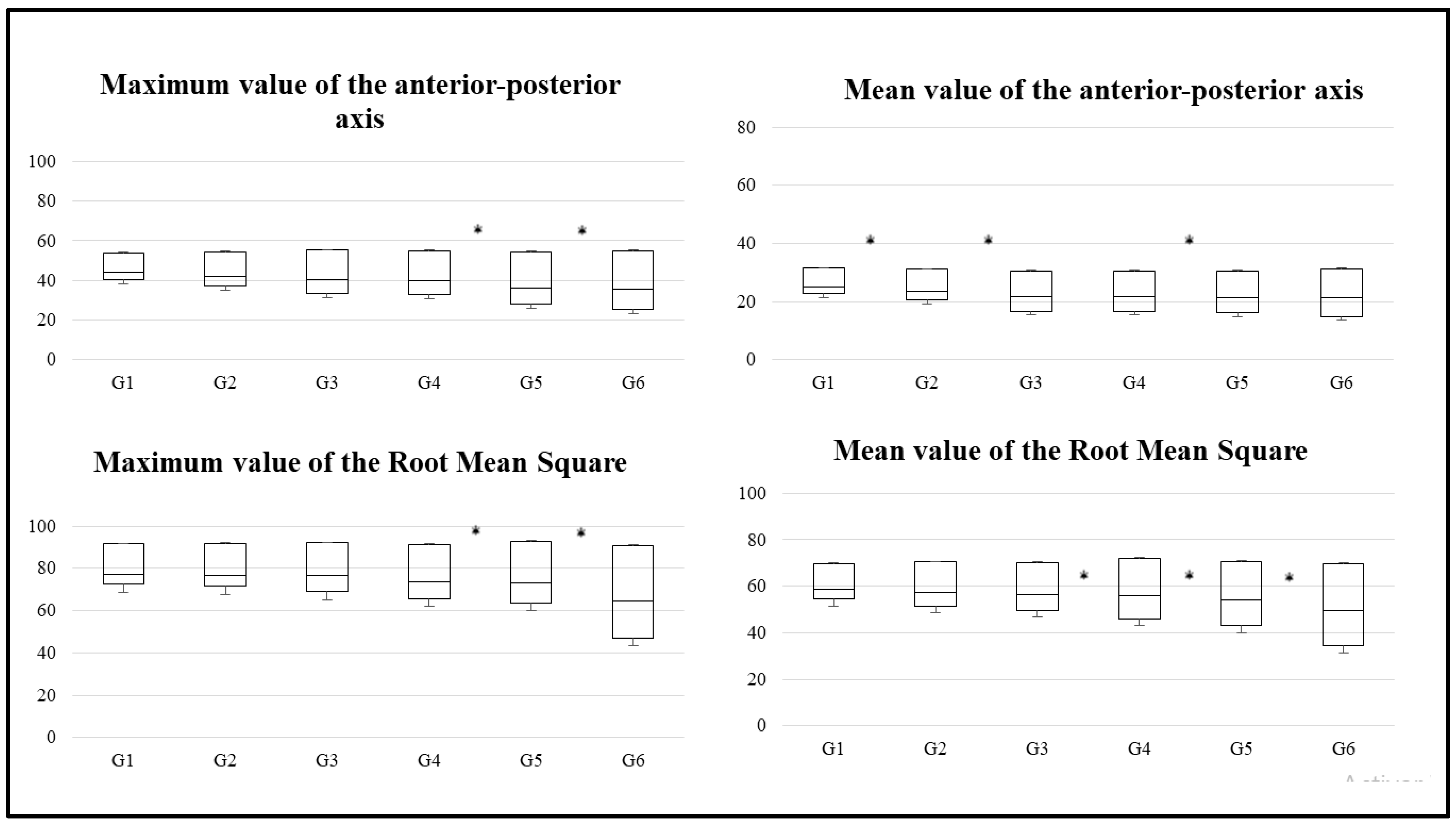
| Variable | G1 (n = 187) | G2 (n = 172) | G3 (n = 185) | G4 (n = 192) | G5 (n = 187) | G6 (n = 173) |
| Maximum value of anterior-posterior axis | ||||||
| Mean ± standard deviation | 48.6 ± 14 | 40.2 ± 10.9 | 42.8 ± 9.5 | 39.4 ± 12 | 40.9 ± 11 | 33.1 ± 8.6 |
| Kurtosis | 2.4 | 3.4 | 3.4 | 2.3 | 5.4 | 1.8 |
| Percentile 25 | 40.3 | 37.3 | 33.7 | 33 | 28 | 25.3 |
| Percentile 50 (median) | 44 | 42 | 40.4 | 40 | 36 | 35.7 |
| Percentile 75 | 50.3 | 49.9 | 48.6 | 48.1 | 46.5 | 44.8 |
| Interquartile range | 10 | 12.6 | 14.9 | 15.1 | 18.5 | 19.5 |
| Mean value of anterior-posterior axis | ||||||
| Mean ± standard deviation | 30.5 ± 9.8 | 23.5 ± 8 | 24.8 ± 7.8 | 22.6 ± 7.2 | 22.1 ± 6.9 | 19.3 ± 6 |
| Kurtosis | 1.8 | 3.5 | 3.5 | 1.8 | 1.4 | 2.3 |
| Percentile 25 | 22.6 | 20.5 | 16.7 | 16.6 | 16.1 | 14.9 |
| Percentile 50 (median) | 25.1 | 23.5 | 21.6 | 21.6 | 21.3 | 21.2 |
| Percentile 75 | 28.9 | 28.1 | 25.6 | 25.6 | 25.5 | 25.1 |
| Interquartile range | 6.3 | 7.6 | 9 | 9 | 9.4 | 10.2 |
| Maximum value of root mean square of accelerations | ||||||
| Mean ± standard deviation | 85.7 ± 18.2 | 81.3 ± 19.8 | 78.9 ± 13.5 | 72.4 ± 13.5 | 71.9 ± 9.9 | 61 ± 13.8 |
| Kurtosis | 2.4 | 3.8 | 3.4 | 2.2 | 2.6 | 1.4 |
| Percentile 25 | 72.7 | 71.7 | 69 | 65.8 | 63.7 | 47.1 |
| Percentile 50 (median) | 77.4 | 76.9 | 76.5 | 73.6 | 73.2 | 64.7 |
| Percentile 75 | 86.9 | 86.6 | 84.6 | 83.6 | 83.4 | 73.3 |
| Interquartile range | 14.3 | 15 | 15.6 | 17.8 | 19.6 | 26.2 |
| Mean value of root mean square of accelerations | ||||||
| Mean ± standard deviation | 62.6 ± 15 | 56.9 ± 14 | 56.5 ± 11.6 | 51.5 ± 10.1 | 50.7 ± 8.8 | 44.3 ± 11.1 |
| Kurtosis | 2.5 | 3.2 | 2.6 | 2.3 | 2.5 | 1.4 |
| Percentile 25 | 54.4 | 51.5 | 49.5 | 46 | 42.9 | 34.4 |
| Percentile 50 (median) | 58.5 | 57.4 | 56.6 | 56.2 | 54 | 49.5 |
| Percentile 75 | 65.7 | 64.5 | 63.1 | 61.7 | 59.7 | 54.5 |
| Interquartile range | 11.3 | 13 | 13.6 | 15.7 | 16.7 | 20.1 |
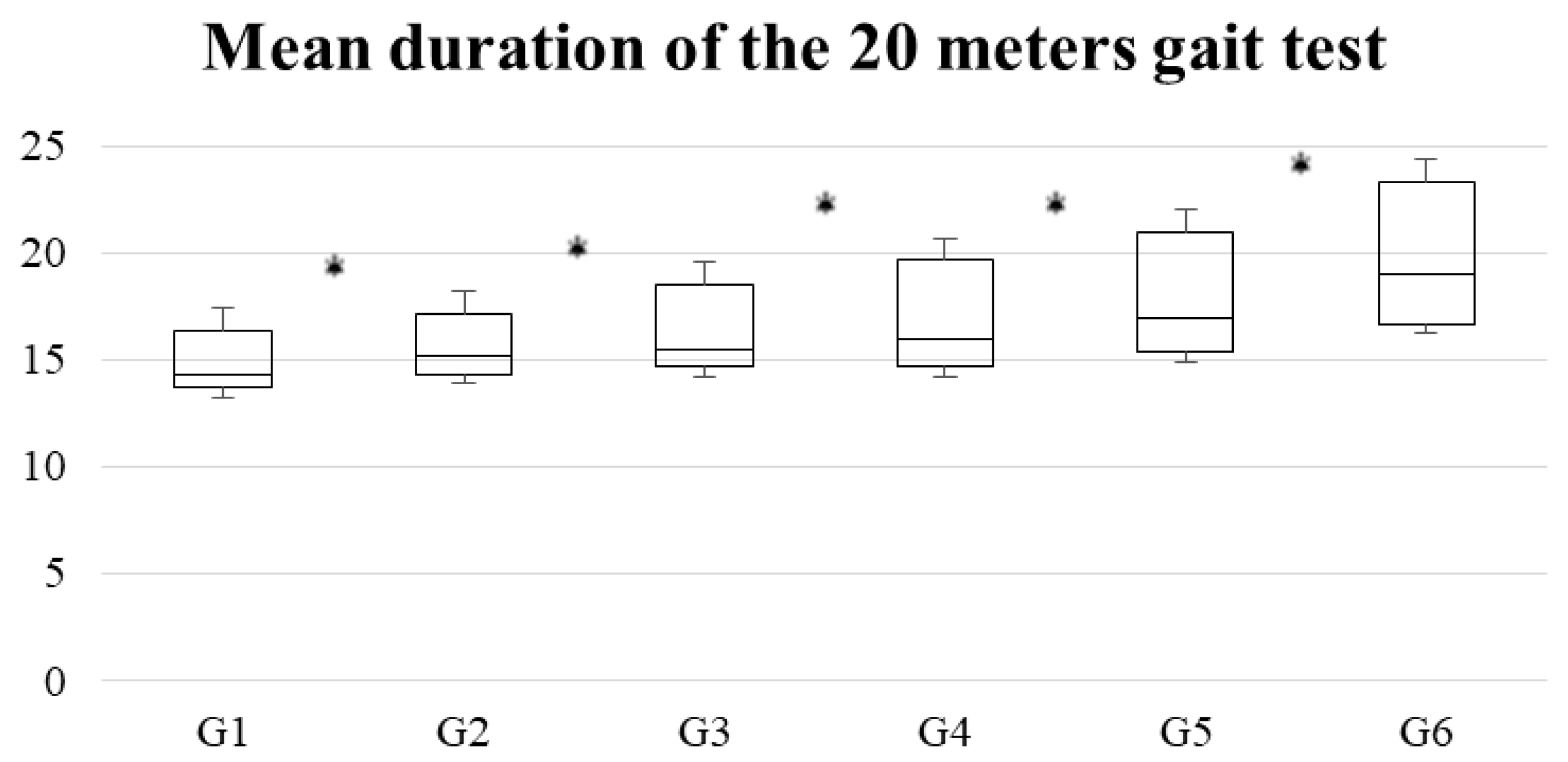
| Variable | G1 (n = 187) | G2 (n = 172) | G3 (n = 185) | G4 (n = 192) | G5 (n = 187) | G6 (n = 173) |
|---|---|---|---|---|---|---|
| Mean ± standard deviation | 15.9 ± 2.3 | 15.5 ± 2 | 15.9 ± 1.9 | 17.1 ± 2.9 | 16.6 ± 2.1 | 18.8 ± 3.2 |
| Kurtosis | 2 | 2.4 | 3.2 | 4.4 | 3.5 | 2.3 |
| Percentile 25 | 13.7 | 14.3 | 14.7 | 14.7 | 15.3 | 16.7 |
| Percentile 50 (median) | 14.3 | 15.2 | 15.5 | 16 | 17 | 19 |
| Percentile 75 | 15.7 | 16.3 | 17.7 | 18.3 | 19.3 | 21 |
| Interquartile range | 2 | 2 | 3 | 3.7 | 4 | 4.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leirós-Rodríguez, R.; García-Liñeira, J.; Soto-Rodríguez, A.; García-Soidán, J.L. Percentiles and Reference Values for Accelerometric Gait Assessment in Women Aged 50–80 Years. Brain Sci. 2020, 10, 832. https://doi.org/10.3390/brainsci10110832
Leirós-Rodríguez R, García-Liñeira J, Soto-Rodríguez A, García-Soidán JL. Percentiles and Reference Values for Accelerometric Gait Assessment in Women Aged 50–80 Years. Brain Sciences. 2020; 10(11):832. https://doi.org/10.3390/brainsci10110832
Chicago/Turabian StyleLeirós-Rodríguez, Raquel, Jesús García-Liñeira, Anxela Soto-Rodríguez, and Jose L. García-Soidán. 2020. "Percentiles and Reference Values for Accelerometric Gait Assessment in Women Aged 50–80 Years" Brain Sciences 10, no. 11: 832. https://doi.org/10.3390/brainsci10110832
APA StyleLeirós-Rodríguez, R., García-Liñeira, J., Soto-Rodríguez, A., & García-Soidán, J. L. (2020). Percentiles and Reference Values for Accelerometric Gait Assessment in Women Aged 50–80 Years. Brain Sciences, 10(11), 832. https://doi.org/10.3390/brainsci10110832