Recent Advances in the Treatment of Cerebellar Disorders
Abstract
1. Introduction
1.1. History
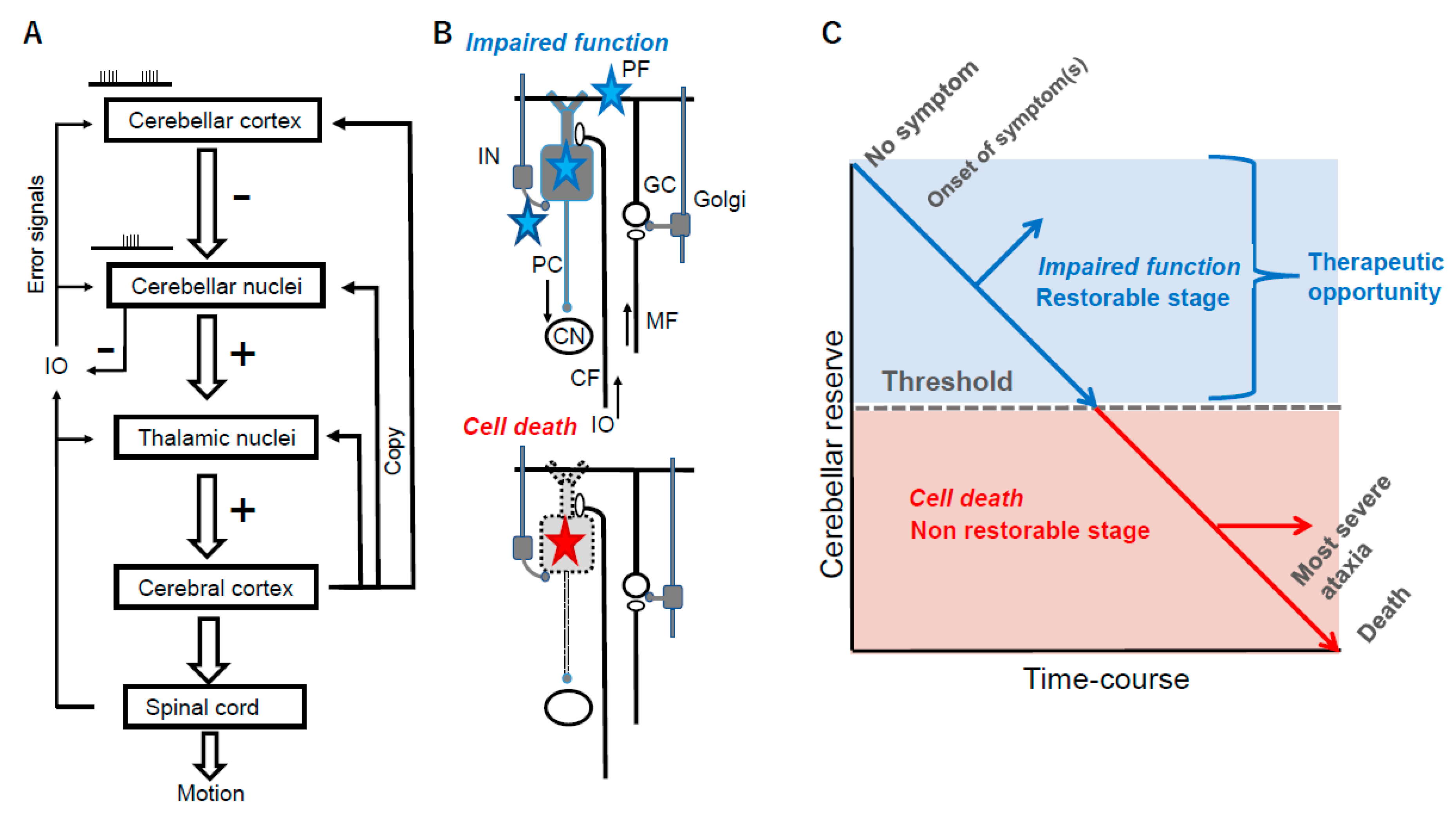
1.2. Therapeutic Strategies Based on Cerebellar Reserve
2. Cause-Cure Treatment; Treatments Designed to Prevent Disease Progression
2.1. Metabolic Cerebellar Ataxias
2.1.1. Alcohol-Related Cerebellar Ataxias
2.1.2. Vitamin B1 Deficiency (Wernicke’s Encephalopathy)
2.1.3. Iron Deposition: Superficial Siderosis
2.2. Immune-Mediated CAs
2.2.1. Gluten Ataxia (GA)
2.2.2. Paraneoplastic Cerebellar Degeneration (PCD)
2.2.3. Post-Infectious Cerebellitis (PIC)
2.2.4. Anti-GAD Ataxia
2.3. Autosomal Recessive Cerebellar Ataxias (ARCAs)
2.3.1. Friedreich’s Ataxia (FRDA)
2.3.2. Ataxia-Telangiectasia (AT)
2.3.3. Ataxia with Vitamin E Deficiency (AVED)
2.3.4. Abetalipoproteinemia
2.3.5. Wilson’s Disease (WD)
2.3.6. GLUT1 Deficiency
2.3.7. Refsum’s Disease (RD)
2.3.8. Cerebrotendinous Xantomatosis (CTX)
2.3.9. Niemann-Pick Disease Type C (NPC)
2.4. Episodic Ataxias (EAs)
3. Neuromodulation Therapies: Therapies Enhancing the Cerebellar Reserve
3.1. Motor Rehabilitation
3.2. Cognitive Rehabilitation
3.3. Non-Invasive Cerebellar Stimulation
- A single magnetic pulse inhibits the amplitude of motor evoked potentials, an index of excitability of the primary motor cortex, which is termed cerebellar brain inhibition (CBI) [94]. Thus, activation of Purkinje cells (PCs) inhibits the excitatory facilitation of the dentato-thalamo-cortical pathways [84,85]. On the other hand, tDCS modulates spontaneous neural activities at the target site through constant electrical current during a particular period, generally 20 min [84,85,95]. This sustained modulation of excitability in the cerebellar cortex changes CBI in a polarity-dependent manner [84,85]; i.e., anodal tDCS increases CBI, whereas cathodal tDCS decreases CBI [96,97]. It should be acknowledged that the long-term therapeutic effect corroborates plasticity changes in the cerebellar cortex.
- A previous physiological study [98] showed that activation of neurons of the dentate nucleus induced by diminished inhibition from PCs (i.e., disinhibition) facilitates the execution of a particular movement, while suppression of the dentate nucleus neurons by increased PC activity (i.e., inhibition) contributes to the stabilization of unnecessary movement. Thus, the cerebellum serves as a predictive modulator through disinhibition/inhibition of the dentato-thalamo-cortical pathway. Thus, damage of the cerebellar circuits could impair the formation of disinhibition/inhibition, thus leading to asthenia (impairment of initiation) and adventitious movements (impairment of stabilization), respectively [99].
- The tDCS-induced improvement was associated with facilitation of CBI [88].
4. Novel Therapies
4.1. Recent Advances in Treatments of Autosomal Dominant Cerebellar Ataxias (ADCAs)
4.1.1. Polyglutaminopathies
4.1.2. ADCAs Induced by Toxic RNAs
4.1.3. ADCAs Caused by Point Mutations
4.2. Recent Advances in Neurotransplantaion
4.2.1. Therapeutic Rationale
4.2.2. Possible Indications for Neurotransplantation
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Manto, M.; Bower, J.M.; Conforto, A.B.; Delgado-García, J.M.; da Guarda, S.N.; Gerwig, M.; Habas, C.; Hagura, N.; Ivry, R.B.; Mariën, P.; et al. Consensus Paper: Roles of the cerebellum in motor control—The diversity of ideas on cerebellar involvement in movement. Cerebellum 2012, 11, 457–487. [Google Scholar] [CrossRef]
- Schmahmann, J.D.; Caplan, D. Cognitive, emotion and the cerebellum. Brain 2006, 129, 290–292. [Google Scholar] [CrossRef]
- Holmes, G. The symptoms of acute cerebellar injuries due to gunshot injuries. Brain 1917, 40, 461–535. [Google Scholar] [CrossRef]
- Mitoma, H.; Manto, M. The physiological basis for therapies of cerebellar ataxias. Ther. Adv. Neurol. Disord. 2016, 9, 396–413. [Google Scholar] [CrossRef]
- Mitoma, H.; Manto, M. Advances in therapies of cerebellar disorders. CNS Neurol. Disord. Drug Targets 2018, 17, 157–160. [Google Scholar] [CrossRef]
- Mitoma, H.; Manto, M. The era of cerebellar therapy. Curr. Neuropharmacol. 2019, 17, 3–6. [Google Scholar] [CrossRef]
- Mitoma, H.; Manto, M.; Hampe, C.S. Time is Cerebellum. Cerebellum 2018, 17, 387–391. [Google Scholar] [CrossRef]
- Kawato, M.; Furukawa, K.; Suzuki, R. A hierarchical neural network model for control and learning of voluntary movement. Biol. Cybern. 1987, 57, 169–185. [Google Scholar] [CrossRef]
- Ito, M. Cerebellar circuitry as a neural machine. Prog. Neurobiol. 2006, 78, 272–303. [Google Scholar] [CrossRef]
- Tanaka, H.; Ishikawa, T.; Kakei, S. Neural evidence of the cerebellum as a state predictor. Cerebellum 2019, 18, 349–371. [Google Scholar] [CrossRef]
- Deuschl, G.; Toro, C.; Zeffiro, T.; Massaquoi, S.; Hallett, M. Adaptation motor learning of arm movements in patients with cerebellar disease. J. Neurol. Neurosurg. Psychiatry 1996, 60, 515–519. [Google Scholar] [CrossRef]
- Kakei, S.; Lee, J.; Mitoma, H.; Tanaka, H.; Manto, M.; Hampe, C.S. Contribution of the cerebellum to predictive motor controls and its evaluation in ataxic patients. Front. Hum. Neurosci. 2019, 13, 216. [Google Scholar] [CrossRef]
- Victor, M.; Adams, R.D.; Mancall, E.L. A restricted form of cerebellar cortical degeneration occurring in alcoholic patients: Cerebellar cortical degenerations. Arch. Neurol. 1959, 1, 579–688. [Google Scholar] [CrossRef]
- Laureno, R. Nutritional cerebellar degeneration, with comments on its relationship to Wernicke disease and alcoholism. In Handbook of Clinical Neurology; 3rd. ed; Subramony, S.H., Dürr, A., Eds.; Elsevier: Amsterdam, the Netherlands, 2012; Volume 103, pp. 175–187. [Google Scholar]
- Sechi, G.; Serra, A. Wernicke’s encephalopathy: New clinical settings and recent advances in diagnosis and management. Lancet Neurol. 2007, 6, 442–455. [Google Scholar] [CrossRef]
- Manzo, G.; Gennaro, A.D.; Cozzolino, A.; Serino, A.; Fenza, G.; Manto, A. MR imaging findings in alcoholic and nonalcoholic acute Wernicke’s encephalopathy: A review. BioMed Res. Int. 2014, 2014, 503596. [Google Scholar] [CrossRef]
- Kopelman, M.D.; Thomson, A.D.; Guerrini, I.; Marshall, E.J. The Korsakoff syndrome: Clinical aspects, psychology and treatment. Alcohol. Alcohol. 2009, 44, 148–154. [Google Scholar] [CrossRef]
- Zuccoli, G.; Gallucci, M.; Capellades, J.; Regnicolo, L.; Tumiati, B.; Giadás, T.C.; Bertolini, M. Wernicke encephalopathy: MR findings at clinical presentation in twenty-six alcoholic and nonalcoholic patients. Am. J. Neuroradiol. 2007, 28, 1328–1331. [Google Scholar] [CrossRef]
- Galvin, R.; Bråthen, G.; Ivashynka, A.; Hillbom, M.; Tanasescu, R.; Leone, M.A. EFNS guidelines for diagnosis, therapy and prevention of Wernicke encephalopathy. Eur. J. Neurol. 2010, 17, 1408–1418. [Google Scholar] [CrossRef]
- Ambrose, M.L.; Bowden, S.C.; Wehan, G. Thiamine treatment and working memory function of alcohol dependent people: Preliminary findings. Alcohol. Clin. Exp. Res. 2010, 25, 112–116. [Google Scholar] [CrossRef]
- Fearnley, J.M.; Stvens, J.M.; Rudge, P. Superficial siderosis of the central nervous system. Brain 1995, 118, 1051–1066. [Google Scholar] [CrossRef]
- Charidimou, A.; Linn, J.; Vernooij, M.W.; Opherk, C.; Akoudad, S.; Baron, J.C.; Greenberg, S.M.; Jäger, H.R.; Werring, D.J. Cortical superficial siderosis; detection and clinical significance in cerebral amyloid angiopathy and related conditions. Brain 2015, 138, 2126–2139. [Google Scholar] [CrossRef]
- Parnes, S.M.; Weaver, S.A. Superficial siderosis of the central nervous system: A neglected cause of sensorineural hearing loss. Otolaryngol. Head Neck Surg. 1992, 107, 69–77. [Google Scholar] [CrossRef]
- Levy, M.; Llinas, R.H. Update on a patient with superficial siderosis on deferiprone. AJNR Am. J. Neuroradiol. 2012, 33, E99–E100. [Google Scholar] [CrossRef]
- Kuo, P.H.; Kuo, S.H.; Lo, R.Y. Deferiprone reduces hemosiderin deposition in superficial siderosis. Can. J. Neurol. Sci. 2017, 44, 219–220. [Google Scholar] [CrossRef]
- Hadjivassiliou, M. Immune-mediated acquired ataxias. Handb. Clin. Neurol. 2012, 103, 189–199. [Google Scholar]
- Mitoma, H.; Adhikari, K.; Aeschlimann, D.; Chattopadhyay, P.; Hadjivassiliou, M.; Hampe, C.S.; Honnorat, J.; Joubert, B.; Kakei, S.; Lee, J. Consensus Paper: Neuroimmune mechanisms of cerebellar ataxias. Cerebellum 2016, 15, 2313–2332. [Google Scholar] [CrossRef]
- Mitoma, H.; Hadjivassiliou, M.; Honnorat, J. Guidelines for treatment of immune-mediated cerebellar ataxias. Cerebellum Ataxias 2015, 2, 14. [Google Scholar] [CrossRef]
- Mitoma, H.; Manto, M.; Hampe, C.S. Immune-mediated cerebellar ataxias: Practical guidelines and therapeutic challenges. Curr. Neuropharmacol. 2018. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Grünewald, R.A.; Chattopadhyay, A.K.; Davies-Jones, G.A.; Gibson, A.; Jarratt, J.A.; Kandler, R.H.; Lobo, A.; Powell, A.; Smith, C.M.L. Clinical, radiological, neurophysiological and neuropathological characteristics of gluten ataxia. Lancet 1998, 352, 1582–1585. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Davies-Jones, G.A.B.; Sandres, D.S.; Grünewald, R.A. Dietary treatment of gluten ataxia. J. Neurol. Neurosurg. Psychiatry 2003, 74, 1221–1224. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Grünewald, R.A.; Davies-Jones, G.A.B. Gluten sensitivity as a neurological illness. J. Neurol. Neurosurg. Psychiatry 2002, 72, 560–563. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Sanders, D.S.; Woodroofe, N.; Williamson, C.; Grünewald, R.A. Gluten ataxia. Cerebellum 2008, 7, 494–498. [Google Scholar] [CrossRef]
- Graus, F.; Delattre, J.Y.; Antoine, J.C.; Dalmau, J.; Giometto, B.; Grisold, W.; Honnorat, J.; Smitt, P.S.; Vedeler, C.; Verschuuren, J.J.; et al. Recommended diagnostic criteria for paraneoplastic neurological syndromes. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1135–1140. [Google Scholar] [CrossRef]
- Dalmau, J.; Rosenfeld, M.R. Paraneoplastic syndromes of the CNS. Lancet Neurol. 2008, 7, 327–340. [Google Scholar] [CrossRef]
- Ducray, F.; Demarquay, G.; Graus, F.; Decullier, E.; Antoine, J.-C.; Giometto, B.; Psimaras, D.; Delattre, J.Y.; Carpentier, A.F.; Honnorat, J. Seronegative paraneoplastic cerebellar degeneration: The PNS Euronetwork experience. Eur. J. Neurol. 2014, 21, 731–735. [Google Scholar] [CrossRef]
- Voltz, R. Paraneoplastic neurological syndromes: An update on diagnosis, pathogenesis, and therapy. Lancet Neurol. 2002, 1, 294–305. [Google Scholar] [CrossRef]
- Demarquay, G.; Honnorat, J. Clinical presentation of immune-mediated cerebellar ataxia. Rev. Neurol. Paris 2011, 167, 408–417. [Google Scholar] [CrossRef]
- Höftberger, R.; Rosenfeld, M.; Dalmau, J. Update on neurological paraneoplastic syndromes. Curr. Opin. Oncol. 2015, 27, 489–495. [Google Scholar] [CrossRef]
- Keime-Guibert, F.; Graus, F.; Fleury, A.; Renė, R.; Honnorat, J.; Broet, P.; Delattre, J.Y. Treatment of paraneoplastic neurological syndromes with antineuronal antibodies (Anti-Hu, Anti-Yo) with a combination of immunoglobulins, cyclophosphamide, and methylprednisolone. J. Neurol. Neurosurg. Psychiatry 2000, 68, 479–482. [Google Scholar] [CrossRef]
- Candler, P.M.; Hart, P.E.; Barnett, M.; Weil, R.; Ress, J.H. A follow up study of patients with paraneoplastic neurological disease in the United Kingdom. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1411–1415. [Google Scholar] [CrossRef]
- Shams’ili, S.; Grefkens, J.; de Leeuw, B.; van den Bent, M.; Hooijkaas, H.; van der Holt, B.; Vecht, C.; Sillevis Smitt, P. Paraneoplastic cerebellar degeneration associated with antineuronal antibodies: Analysis of 50 patients. Brain 2003, 126, 1409–1418. [Google Scholar] [CrossRef]
- Blumkin, L.; Pranzatelli, M.R. Aquired ataxias, infectious and para-infectious. Handb. Clin. Neurol. 2012, 103, 137–146. [Google Scholar]
- Sivaswamy, L. Approach to acute ataxia in childhood: Diagnosis and evaluation. Pediatr. Ann. 2014, 43, 153–159. [Google Scholar] [CrossRef]
- Connolly, A.M.; Dodson, W.E.; Prensky, A.L.; Rust, R.S. Course and outcome of acute cerebellar ataxia. Ann. Neurol. 1994, 35, 673–679. [Google Scholar] [CrossRef]
- Honnorat, J.; Saiz, A.; Giometto, B.; Vincent, A.; Brieva, L.; Andres, C.; Maestre, J.; Fabien, N.; Vighetto, A.; Casamitjana, R.; et al. Cerebellar ataxia with anti-glutamic acid decarboxylase antibodies. Study of 14 patients. Arch. Neurol. 2001, 58, 225–230. [Google Scholar] [CrossRef]
- Mitoma, M.; Manto, M.; Hampe, C.S. Pathogenic roles of glutamic acid decarboxylase 65 autoantibodies in cerebellar ataxias. J. Immunol. Res. 2017, 2913297. [Google Scholar] [CrossRef]
- Manto, M.; Mitoma, H.; Hampe, C.S. Anti-GAD antibodies and the cerebellum: Where do we stand? Cerebellum 2018, 18, 153–156. [Google Scholar] [CrossRef]
- Arińo, H.; Gresa–Arribas, N.; Blanco, Y.; Martínez-Hernández, E.; Sabater, L.; Petit-Pedrol, M.; Rouco, I.; Bataller, L.; Dalmau, J.O.; Saiz, A.; et al. Cerebellar ataxia and glutamic acid decarboxylase antibodies. Immunologic profile and long-term effect of immunotherapy. JAMA Neurol. 2014, 71, 1009–1016. [Google Scholar] [CrossRef]
- Pier-Martel, P.; Dupre, N. Current and promising therapies in autosomal recessive ataxias. CNS Neurol. Disord. Drug Targets 2018, 17, 161–171. [Google Scholar] [CrossRef]
- Beaudin, M.; Matilla-Dueñas, A.; Soong, B.W.; Pedroso, J.L.; Barsottini, O.G.; Mitoma, H.; Tsuji, S.; Schmahmann, J.D.; Manto, M.; Rouleau, G.A.; et al. The Classification of Autosomal Recessive Cerebellar Ataxias: A Consensus Statement from the Society for Research on the Cerebellum and Ataxias Task Force. Cerebellum 2019, 18, 1098–1125. [Google Scholar] [CrossRef]
- Artuch, R.; Aracil, A.; Mas, A.; Colome, C.; Rissech, M.; Monros, E.; Pineda, M. Friedreich’s ataxia: Idebenone treatment in early stage patients. Neuropediatrics 2002, 33, 190–193. [Google Scholar] [CrossRef]
- Pineda, M.; Arpa, J.; Montero, R.; Aracil, A.; Dominguez, F.; Galvan, M.; Mas, A.; Martorell, L.; Sierra, C.; Brandi, N.; et al. Idebenone treatment in paediatric and adult patients with Friedreich ataxia: Long-term follow-up. Eur. J. Paediatr. Neurol. 2008, 12, 470–475. [Google Scholar] [CrossRef]
- Pandolfo, M.; Hausmann, L. Deferiprone for the treatment of Friedreich’s ataxia. J. Neurochem. 2013, 126, 142–146. [Google Scholar] [CrossRef]
- Seyer, L.; Greeley, N.; Foerster, D.; Strawser, C.; Gelbard, S.; Dong, Y.; Schadt, K.; Cotticelli, M.G.; Brocht, A.; Farmer, J.; et al. Open-label pilot study of interferon gamma-1b in Friedreich ataxia. Acta. Neurol. Scand. 2015, 132, 7–15. [Google Scholar] [CrossRef]
- Gatti, R.A.; Berkel, I.; Boder, E.; Braedt, G.; Charmley, P.; Concannon, P.; Ersoy, F.; Foroud, T.; Jaspers, N.G.; Lange, K.; et al. Localization of an ataxia-telangiectasia gene to chromosome 11q22-23. Nature 1988, 336, 577–580. [Google Scholar] [CrossRef]
- Abrahao, A.; Pedroso, J.L.; Braga-Neto, P.; Bor-Seng-Shu, E.; de Carvalho Aguiar, P.; Barsottini, O.G. Milestones in Friedreich ataxia: More than a century and still learning. Neurogenetics 2015, 16, 151–160. [Google Scholar] [CrossRef]
- Cooper, J.M.; Korlipara, L.V.; Hart, P.E.; Bradley, J.L.; Schapira, A.H. Coenzyme Q10 and vitamin E deficiency in Friedreich’s ataxia: Predictor of efficacy of vitamin E and coenzyme Q10 therapy. Eur. J. Neurol. 2008, 15, 1371–1379. [Google Scholar] [CrossRef]
- Broccoletti, T.; Del Giudice, E.; Cirillo, E.; Vigliano, I.; Giardino, G.; Ginocchio, V.M.; Bruscoli, S.; Riccardi, C.; Pignata, C. Efficacy of very-low-dose betamethasone on neurological symptoms in ataxia-telangiectasia. Eur. J. Neurol. 2011, 18, 564–570. [Google Scholar] [CrossRef]
- Zannolli, R.; Buoni, S.; Betti, G.; Salvucci, S.; Plebani, A.; Soresina, A.; Pietrogrande, M.C.; Martino, S.; Leuzzi, V.; Finocchi, A.; et al. A randomized trial of oral betamethasone to reduce ataxia symptoms in ataxia telangiectasia. Mov. Disord. 2012, 27, 1312–1316. [Google Scholar] [CrossRef]
- Ouahchi, K.; Arita, M.; Kayden, H.; Hentati, F.; Ben Hamida, M.; Sokol, R.; Arai, H.; Inoue, K.; Mandel, J.L.; Koenig, M. Ataxia with isolated vitamin E deficiency is caused by mutations in the alpha-tocopherol transfer protein. Nat. Genet. 1995, 9, 141–145. [Google Scholar] [CrossRef]
- Mihalik, S.J.; Morrell, J.C.; Kim, D.; Sacksteder, K.A.; Watkins, P.A.; Gould, S.J. Identification of PAHX, a Refsum disease gene. Nat. Genet. 1997, 17, 185–189. [Google Scholar] [CrossRef]
- van den Brink, D.M.; Brites, P.; Haasjes, J.; Wierzbicki, A.S.; Mitchell, J.; Lambert-Hamill, M.; de Belleroche, J.; Jansen, G.A.; Waterham, H.R.; Wanders, R.J. Identification of PEX7 as the second gene involved in Refsum disease. Am. J. Hum. Genet. 2003, 72, 471–477. [Google Scholar] [CrossRef]
- Cali, J.J.; Hsieh, C.L.; Francke, U.; Russell, D.W. Mutations in the bile acid biosynthetic enzyme sterol 27-hydroxylase underlie cerebrotendinous xanthomatosis. J. Biol. Chem. 1991, 266, 7779–7783. [Google Scholar]
- Carstea, E.D.; Morris, J.A.; Coleman, K.G.; Loftus, S.K.; Zhang, D.; Cummings, C.; Gu, J.; Rosenfeld, M.A.; Pavan, W.J.; Krizman, D.B.; et al. Niemann-Pick C1 disease gene: Homology to mediators of cholesterol homeostasis. Science 1997, 277, 228–231. [Google Scholar] [CrossRef]
- Wraith, J.E.; Vecchio, D.; Jacklin, E.; Abel, L.; Chadha-Boreham, H.; Luzy, C.; Giorgino, R.; Patterson, M.C. Miglustat in adult and juvenile patients with Niemann-Pick disease type C: Long-term data from a clinical trial. Mol. Genet. Metab. 2010, 99, 351–357. [Google Scholar] [CrossRef]
- Kalla, R.; Strupp, M. Aminopyridines and acetyl-DL-leucine: New therapies in cerebellar disorders. Curr. Neuropharmacol. 2019, 17, 7–13. [Google Scholar] [CrossRef]
- Ophoff, R.A.; Terwindt, G.M.; Vergouwe, M.N.; van Eijk, R.; Oefner, P.J.; Hoffman, S.M.; Lamerdin, J.E.; Mohrenweiser, H.W.; Bulman, D.E.; Ferrari, M.; et al. Familial hemiplegic migraine and episodic ataxia type-2 are caused by mutations in the Ca2+ channel gene CACNL1A4. Cell 1996, 87, 543–552. [Google Scholar] [CrossRef]
- Strupp, M.; Kalla, R.; Dichgans, M.; Freilinger, T.; Glasauer, S.; Brandt, T. Treatment of episodic ataxia type 2 with the potassium channel blocker 4-aminopyridine. Neurology 2004, 62, 1623–1625. [Google Scholar] [CrossRef]
- Strupp, M.; Kalla, R.; Claassen, J.; Adrion, C.; Mansmann, U.; Klopstock, T.; Freilinger, T.; Neugebauer, H.; Spiegel, R.; Dichgans, M.; et al. A randomized trial of 4-aminopyridine in EA2 and related familial episodic ataxias. Neurology 2011, 77, 269–275. [Google Scholar] [CrossRef]
- Alvina, K.; Khodakhah, K. The therapeutic mode of action of 4-aminopyridine in cerebellar ataxia. J. Neurosci. 2010, 30, 7258–7268. [Google Scholar] [CrossRef]
- Strupp, M.; Schüler, O.; Krafczyk, S.; Jahn, K.; Schautzer, F.; Büttner, U.; Brandt, T. Treatment of downbeat nystagmus with 3,4-diaminopyridine. A placebo-controlled study. Neurology 2003, 61, 165–170. [Google Scholar] [CrossRef]
- Kalla, R.; Glasauer, S.; Büttner, U.; Brandt, T.; Strupp, M. 4-Aminopyridine restores vertical and horizontal neural integrator function in downbeat nystagmus. Brain 2007, 130, 2441–2445. [Google Scholar] [CrossRef]
- Ilg, W.; Brötz, D.; Burkard, S.; Giese, M.A.; Schöls, L.; Synofzik, M. Long-term effects of coordinative training in degenerative cerebellar disease. Mov. Disord. 2010, 25, 2239–2246. [Google Scholar] [CrossRef]
- Ilg, W.; Bastian, A.J.; Boesch, S.; Celnik, P.; Claaßen, J.; Feil, K.; Kalla, R.; Miyai, I.; Nachbauer, W.; Schöls, L.; et al. Consensus paper: Management of degenerative cerebellar disorders. Cerebellum 2014, 13, 248–268. [Google Scholar] [CrossRef]
- Miyai, I.; Ito, M.; Hattori, N.; Mihara, M.; Hatakenaka, M.; Yagura, H.; Sobue, G.; Nishizawa, M. Cerebellar ataxia rehabilitation trial in degenerative cerebellar diseases. Neurorehabil. Neural. Repair 2012, 26, 515–522. [Google Scholar] [CrossRef]
- Schmahmann, J.D.; Sherman, J.C. The cerebellar cognitive-affective syndrome. Brain 1998, 121, 561–579. [Google Scholar] [CrossRef]
- van Dun, K.; van Overwalle, F.; Manto, M.; Mariën, P. Cognitive impact of cerebellar damage: Is there a future for cognitive rehabilitation? CNS Neurol. Disord. Drug Targets 2018, 17, 199–206. [Google Scholar] [CrossRef]
- Maeshima, S.; Osawa, A. Stroke rehabilitation in a patient with cerebellar cognitive affective syndrome. Brain Inj. 2007, 21, 877–883. [Google Scholar] [CrossRef]
- Schweizer, T.A.; Levine, B.; Rewilak, D.; O’Connor, C.; Turner, G.; Alexander, M.P.; Cusimano, M.; Manly, T.; Robertson, I.H.; Stuss, D.T. Rehabilitation of executive functioning after focal damage to the cerebellum. Neurorehabil. Neural. Repair 2008, 22, 72–77. [Google Scholar] [CrossRef]
- Komuro, A.; Kurabayashi, H.; Aasaki, Y.; Sugiura, T.; Hashinuma, A.; majima, M. Clinical improvements in higher brain function and rapid functional recovery in a case of cerebellar hemorrhage treated by neurocognitive rehabilitation. Neurocase 2014, 20, 260–262. [Google Scholar] [CrossRef]
- Ruffieux, N.; Colombo, F.; Gentaz, E.; Annoni, J.M.; Chouiter, L.; Roulin Hefti, S.; Ruffieux, A.; Bihl, T. Successful neuropsychological rehabilitation in a patient with Cerebellar Cognitive Affective Syndrome. Appl. Neuropsychol. Child 2017, 6, 180–188. [Google Scholar] [CrossRef]
- Schmahmann, J.D. The role of the cerebellum in cognition and emotion: Personal reflections since 1982 on the dysmetria of thought hypothesis, and its historical evolution from theory to therapy. Neuropsychol. Rev. 2010, 20, 236–260. [Google Scholar] [CrossRef]
- Di Nuzzo, C.; Ruggiero, F.; Cortese, F.; Cova, I.; Priori, A.; Ferrucci, R. Non-invasive cerebellar stimulation in cerebellar disorders. CNS Neurol. Disord. Drug Targets 2018, 17, 193–198. [Google Scholar] [CrossRef]
- Ferrucci, R.; Bocci, T.; Cortese, F.; Ruggiero, F.; Priori, A. Noninvasive cerebellar stimulation as a complemental tool to Pharmacotherapy. Curr. Neuropharmacol. 2019, 17, 14–20. [Google Scholar] [CrossRef]
- Grimaldi, G.; Taib, N.; Manto, M. Marked reduction of cerebellar deficits in upper limbs following transcranial cerebello-cerebral DC stimulation: Tremor reduction and re-programming of the timing of antagonist commands. Front. Syst. Neurosci. 2014, 8, 9. [Google Scholar] [CrossRef]
- Benussi, A.; Koch, G.; Cotelli, M.; Padovani, A.; Borroni, B. Cerebellar transcranial direct current stimulation in patients with ataxia: A double-blind, randomized, sham-controlled study. Mov. Disord. 2015, 30, 1701–1705. [Google Scholar] [CrossRef]
- Benussi, A.; Dell’Era, V.; Cotelli, M.; Padovani, A.; Borroni, B. Long term clinical and neurophysiological effects of cerebellar transcranial direct current stimulation in patients with neurodegenerative ataxia. Brain Stimul. 2017, 10, 242–250. [Google Scholar] [CrossRef]
- Shimizu, H.; Tsuda, T.; Shiga, Y.; Miyazawa, K.; Onodera, Y.; Matsuzaki, M.; Nakashima, I.; Furukawa, K.; Aoki, M.; Kato, H.; et al. Therapeutic efficacy of transcranial magnetic stimulation for hereditary spinocerebellar degeneration. Tohoku J. Exp. Med. 1999, 189, 203–211. [Google Scholar] [CrossRef]
- Shiga, Y.; Tsuda, T.; Itoyama, Y.; Shimizu, H.; Miyazawa, K.; Jin, K.; Yamazaki, T. Transcranial magnetic stimulation alleviates truncal ataxia in spinocerebellar degeneration. J. Neurol. Neurosurg. Psychiatry 2002, 72, 124–126. [Google Scholar] [CrossRef]
- Kim, W.S.; Jun, S.H.; Oh, M.K.; Min, Y.S.; Lim, J.Y.; Paik, N.J. Effect of repetitive transcranial magnetic stimulation over the cerebellum on patients with ataxia after posterior circulation stroke: A pilot study. J. Rehabil. Med. 2014, 46, 418–423. [Google Scholar] [CrossRef]
- Bodranghien, F.C.A.A.; Langlois Mahe, M.; Clément, S.; Manto, M.U. A pilot study on the effects of transcranial direct current stimulation on brain rhythms and entropy during self-paced finger movement using epoc helmet. Front. Hum. Neurosci. 2017, 11, 201. [Google Scholar] [CrossRef]
- Grimaldi, G.; Manto, M. Anodal transcranial direct current stimulation (tDCS) decreases the amplitudes of long-latency stretch reflexes in cerebellar ataxia. Ann. Biomed. 2013, 41, 2437–2447. [Google Scholar] [CrossRef]
- Ugawa, Y.; Genba-Shimizu, K. Electrical stimulation of the human descending motor tracts at several levels. Can. J. Neurol. Sci. 1995, 22, 36–42. [Google Scholar] [CrossRef]
- Concetta, M.; Brignani, D.; Miniussi, C. NeuroImage Excitability modulation of the motor system induced by transcranial direct current stimulation: A multimodal approach. Neuroimage 2013, 83, 569–580. [Google Scholar]
- Galea, J.; Jayaram, G.; Ajagbe, L. Modulation of cerebellar excitability by polarity-specific noninvasive direct current stimulation. J. Neurosci. 2009, 29, 9115–9122. [Google Scholar] [CrossRef]
- Nitsche, M.A.; Paulus, W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J. Physiol. 2000, 527, 633–639. [Google Scholar] [CrossRef]
- Ishikawa, T.; Tomatsu, S.; Tsunoda, Y.; Lee, J.; Hoffman, D.S.; Kakei, S. Releasing dentate nucleus cells from Purkinje cell inhibition generates output from the cerebrocerebellum. PLoS ONE 2014, 9, e108774. [Google Scholar] [CrossRef]
- Ishikawa, T.; Kakei, S.; Mitoma, H. Overlooked Holmes’ clinical signs: Reevaluation by recent physiological findings. Cerebellum Ataxias 2015, 2, 13. [Google Scholar] [CrossRef]
- Sebastian, R.; Tippett, D.; Celnik, P.; Hillis, A.E. Cerebellar Transcranial Direct Stimulation to Augment Aphasia Therapy. Stroke 2017, 48, ATP142. [Google Scholar]
- Oulad Ben Taib, N.; Manto, M. The in vivo reduction of afferent facilitation induced by low frequency electrical stimulation of the motor cortex is antagonized by cathodal direct current stimulation of the cerebellum. Cerebellum Ataxias 2016, 3, 15. [Google Scholar] [CrossRef]
- Manto, M.; Oulad Ben Taib, N. The contributions of the cerebellum in sensorimotor control: What are the prevailing opinions which will guide forthcoming studies? Cerebellum 2013, 12, 313–315. [Google Scholar] [CrossRef] [PubMed]
- Ashizawa, T.; Öz, G.; Paulson, H.L. Spinocerebellar ataxias: Prospects and challenges for therapy development. Nat. Rev. Neurol. 2018, 14, 590–605. [Google Scholar] [CrossRef] [PubMed]
- Matilla-Dueñas, A.; Ashizawa, T.; Brice, A.; Magri, S.; McFarland, K.N.; Pandolfo, M.; Pulst, S.M.; Riess, O.; Rubinsztein, D.C.; Schmidt, J.; et al. Consensus paper; Pathological mechanisms underlying neurodegeneration in spinocerebellar ataxias. Cerebellum 2014, 13, 269–302. [Google Scholar] [CrossRef] [PubMed]
- Bence, N.F.; Sampat, R.M.; Kopito, R.R. Impairment of the ubiquitin-proteasome system by protein aggregation. Science 2001, 292, 1552–1555. [Google Scholar] [CrossRef]
- Sakahira, H.; Breuer, P.; Hayer-Hartl, M.K.; Hartl, F.U. Molecular chaperones as modulators of polyglutamine protein aggregation and toxicity. Proc. Natl. Acad. Sci. USA 2002, 99, 16412–16418. [Google Scholar] [CrossRef]
- Nascimento-Ferreira, I.; Nóbrega, C.; Vasconcelos-Ferreira, A.; Onofre, I.; Albuquerque, D.; Aveleira, C.; Hirai, H.; Déglon, N.; Pereira de Almeida, L. Beclin 1 mitigates motor and neuropathological deficits in genetic mouse models of Machado-Joseph disease. Brain 2013, 136, 2173–2188. [Google Scholar] [CrossRef]
- Shen, K.; Gamerdinger, M.; Chan, R.; Gense, K.; Martin, E.M.; Sachs, N.; Knight, P.D.; Schlömer, R.; Calabrese, A.N.; Stewart, K.L.; et al. Dual role of ribosome binding domain of NAC as a potent suppressor of protein aggregation and aging-related proteinopathies. Mol. Cell 2019, 74, 729–741. [Google Scholar]
- Kouzarides, T. Chromatin modifications and their function. Cell 2007, 128, 693–705. [Google Scholar] [CrossRef]
- Yi, J.; Zhang, L.; Tang, B.; Han, W.; Zhou, Y.; Chen, Z.; Jia, D.; Jiang, H. Sodium valproate alleviates neurodegeneration in SCA3/MJD via suppressing apoptosis and rescuing the hypoacetylation levels of histone H3 and H4. Gonzalez-Alegre P, editor. PLoS ONE 2013, 8, e54792. [Google Scholar] [CrossRef]
- Moore, L.R.; Rajpalm, G.; Dillingham, I.T.; Qutob, M.; Blumenstein, K.G.; Gattis, D.; Hung, G.; Kordasiewicz, H.B.; Paulson, H.L.; McLoughlin, H.S. Evaluation of antisense oligonucleotides targeting ATXN3 in SCA3 mouse models. Mol. Ther. Nucleic. Acids 2017, 7, 200–210. [Google Scholar] [CrossRef]
- Sullivan, R.; Yau, W.Y.; O’Connor, E.; Houlden, H. Spinocerebellar ataxia: An update. J. Neurol. 2019, 266, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Scoles, D.R.; Meera, P.; Schneider, M.D.; Paul, S.; Dansithong, W.; Figueroa, K.P.; Hung, G.; Rigo, F.; Bennett, C.F.; Otis, T.S.; et al. Antisense oligonucleotide therapy for spinocerebellar ataxia type 2. Nature 2017, 544, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Becker, L.A.; Huang, B.; Bieri, G.; Ma, R.; Knowles, D.A.; Jafar-Nejad, P.; Messing, J.; Kim, H.J.; Soriano, A.; Auburger, G.; et al. Therapeutic reduction of ataxin-2 extends lifespan and reduces pathology in TDP-43 mice. Nature 2017, 544, 367–371. [Google Scholar] [CrossRef] [PubMed]
- McLoughlin, H.S.; Moore, L.R.; Chopra, R.; Komlo, R.; McKenzie, M.; Blumenstein, K.G.; Zhao, H.; Kordasiewicz, H.B.; Shakkottai, V.G.; Paulson, H.L. Oligonucleotide therapy mitigates disease in spinocerebellar ataxia type 3 mice. Ann. Neurol. 2018, 84, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Toonen, L.J.A.; Rigo, F.; van Attikum, H.; van Roon-Mom, W.M.C. Antisense oligonucleotide-mediated removal of the polyglutamine repeat in spinocerebellar ataxia type 3 mice. Mol. Ther. Nucleic Acids 2017, 8, 232–242. [Google Scholar] [CrossRef]
- Kim, H.D.; Rossi, J.J. RNAi mechanisms and applications. Biotechniques 2008, 44, 613–616. [Google Scholar] [CrossRef]
- Almeida, R.; Allshire, R.C. RNA silencing and genome regulation. Trends Cell Biol. 2005, 15, 251–258. [Google Scholar] [CrossRef]
- Zhang, H.; Kolb, F.A.; Jaskiewicz, L.; Westhof, E.; Filipowicz, W. Single processing center models for human Dicer and bacterial RNase III. Cell 2004, 118, 57–68. [Google Scholar] [CrossRef]
- Ramachandran, P.S.; Boudreau, R.L.; Schaefer, K.A.; La Spada, A.R.; Davidson, B.L. Nonallele specific silencing of ataxin-7 improves disease phenotypes in a mouse model of SCA7. Mol. Ther. 2014, 22, 1635–1642. [Google Scholar] [CrossRef]
- Keiser, S.M.; Boudreau, R.L.; Davidson, B.L. Broad therapeutic benefit after RNAi expression vector delivery to deep cerebellar nuclei: Implications for spinocerebellar ataxia type 1 therapy. Moleculartherapy 2014, 22, 588–595. [Google Scholar] [CrossRef]
- Paulson, H.L.; Shakkottai, V.G.; Clark, H.B.; Orr, H.T. Polyglutamine spinocerebellar ataxias-from genes to potential treatments. Nat. Rev. Neurosci. 2017, 18, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Mao, Q.; Eliason, S.L.; Harper, S.Q.; Martins, I.H.; Orr, H.T.; Paulson, H.L.; Yang, L.; Kotin, R.M.; Davidson, B.L. RNAi suppresses polyglutamine-induced neurodegeneration in a model of spinocerebellar ataxia. Nat. Med. 2004, 10, 816–820. [Google Scholar] [CrossRef] [PubMed]
- Alves, S.; Nascimento-Ferreira, I.; Auregan, G.; Hassig, R.; Dufour, N.; Brouillet, E.; Pedroso de Lima, M.C.; Hantraye, P.; Pereira de Almeida, L.; Déglon, N. Allele-specific RNA silencing of mutant ataxin-3 mediates neuroprotection in a rat model of Machado-Joseph disease. PLoS ONE 2008, 3, e3341. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Lebron, E.; Costa Mdo, C.; Luna-Cancalon, K.; Peron, T.M.; Fischer, S.; Boudreau, R.L.; Davidson, B.L.; Paulson, H.L. Silencing mutant ATXN3 expression resolves molecular phenotypes in SCA3 transgenic mice. Mol. Ther. 2013, 21, 1909–1918. [Google Scholar] [CrossRef]
- Miyazaki, Y.; Du, X.; Muramatsu, S.; Gomez, C.M. An miRNA-mediated therapy for SCA6 blocks IRES-driven translation of the CACNA1A second cistron. Sci. Transl. Med. 2016, 8, 347ra94. [Google Scholar] [CrossRef]
- Ramachandran, P.S.; Bhattarai, S.; Singh, P.; Boudreau, R.L.; Thompson, S.; Laspada, A.R.; Drack, A.V.; Davidson, B.L. RNA interference-based therapy for spinocerebellar ataxia type 7 retinal degeneration. PLoS ONE 2014, 9, e95362. [Google Scholar] [CrossRef]
- Park, J.; Al-Ramahl, I.; Tan, Q.; Mollema, N.; Diaz-Garcia, J.R.; Gallego-Flores, T.; Lu, H.C.; Lagalwar, S.; Duvick, L.; Kang, H.; et al. RAS-MAPK-MSK1 pathway modulates ataxin 1 protein levels and toxicity in SCA1. Nature 2013, 498, 325–331. [Google Scholar] [CrossRef]
- Liu, J.; Tang, T.S.; Tu, H.; Nelson, O.; Herndon, E.; Huynh, D.P.; Pulst, S.M. Deranged calcium signaling and neurodegeneration in spinocerebellar ataxia type 2. J. Neurosci. 2009, 29, 9148–9162. [Google Scholar] [CrossRef]
- Chen, X.; Tang, T.S.; Tu, H.; Nelson, O.; Pook, M.; Hammer, R.; Nukina, N.; Bezprozvanny, I. Deranged calcium signaling and neurodegeneration in spinocerebellar ataxia type 3. J. Neurosci. 2008, 28, 12713–12724. [Google Scholar] [CrossRef]
- Teixeira-Castro, A.; Jalles, A.; Esteves, S.; Kang, S.; da Siliva Santos, L.; Silva-Fernandes, A.; Neto, M.F.; Brielmann, R.M.; Bessa, C.; Duarte-Silva, S.; et al. Serotonergic signalling suppresses ataxin 3 aggregation and neurotoxicity in animal models of Machado-Joseph disease. Brain 2015, 138, 3221–3237. [Google Scholar] [CrossRef]
- Costa, M.D.; Ashraf, N.S.; Fischer, S.; Yang, Y.; Schapka, E.; Joshi, G.; McQuade, T.J.; Dharia, R.M.; Dulchavsky, M.; Ouyang, M.; et al. Unbiased screen identifies aripiprazole as a modulator of abundance of the polyglutamine disease protein, ataxin-3. Brain 2016, 139, 2891–2908. [Google Scholar] [CrossRef] [PubMed]
- Egorova, P.A.; Zakharova, O.A.; Vlasova, O.L.; Bezprozvanny, I.B. In vivo analysis of cerebellar Purkinje cell activity in SCA2 transgenic mouse model. J. Neurophysiol. 2016, 115, 2840–2851. [Google Scholar] [CrossRef] [PubMed]
- Notartomaso, S.; Zappulla, C.; Biagioni, F.; Cannella, M.; Bucci, D.; Mascio, G.; Scarselli, P.; Fazio, F.; Weisz, F.; Lionetto, L.; et al. Pharmacological enhancement of mGlu1 metabotropic glutamate receptors causes a prolonged symptomatic benefit in a mouse model of spinocerebellar ataxia type 1. Mol. Brain 2013, 6, 48. [Google Scholar] [CrossRef] [PubMed]
- Rossi, F.; Cattaneo, E. Opinion: Neural stem cell therapy for neurological diseases: Dreams and reality. Nat. Rev. Neurosci. 2002, 3, 401–409. [Google Scholar] [CrossRef]
- Cendelin, J.; Mitoma, H. Neurotransplantation therapy. Hand. Clin. Neurol. 2018, 155, 379–391. [Google Scholar]
- Cendelin, J.; Mitoma, H.; Manto, M. Neurotransplantation therapy and cerebellar reserve. CNS Neurol. Disord. Drug Targets 2018, 17, 172–183. [Google Scholar] [CrossRef]
- Cendelin, J.; Buffo, A.; Hirai, H.; Magrassi, L.; Mitoma, H.; Sherrard, R.; Vozeh, F.; Manto, M. Task force paper on cerebellar transplantation: Are we ready to treat cerebellar disorders with cell therapy? Cerebellum 2019, 18, 575–592. [Google Scholar] [CrossRef]
- Carletti, B.; Piemonte, F.; Rossi, F. Neuroprotection: The emerging concept of restorative neural stem cell biology the treatment of neurodegenerative diseases. Curr. Neuropharmacol. 2011, 9, 313–317. [Google Scholar]
- Kumar, A.; Narayanan, K.; Chaudhary, R.K.; Mishra, S.; Kumar, S.; Vinoth, K.J.; Padmanabhan, P.; Gulyás, B. Current perspective of stem cell therapy in neurodegenerative and metabolic diseases. Mol. Neurobiol. 2016, 54, 7276–7296. [Google Scholar] [CrossRef]
- Jones, J.; Jaramillo-Merchan, J.; Bueno, C.; Pastor, D.; Viso-León, M.; Martinez, S. Mesenchymal stem cells rescue Purkinje cells and improve motor functions in a mouse model of cerebellar ataxia. Neurobiol. Dis. 2010, 40, 415–423. [Google Scholar] [CrossRef]
- Bae, J.S.; Furuya, S.; Ahn, S.J.; Hirabayashi, Y.; Jin, H.K. Neuroglial activation in Niemann-Pick Type C mice is suppressed by intracerebral transplantation of bone marrow-derived mesenchymal stem cells. Neurosci. Lett. 2005, 381, 234–236. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Lee, J.K.; Min, W.K.; Bae, J.H.; He, X.; Schuchman, E.H.; Bae, J.S.; Jin, H.K. Bone marrow-derived mesenchymal stem cells prevent the loss of Niemann-Pick type C mouse Purkinje neurons by correcting sphingolipid metabolism and increasing sphingosine-1-phosphate. Stem Cells 2010, 28, 821–831. [Google Scholar] [CrossRef] [PubMed]
- Jäderstad, J.; Jäderstad, L.M.; Li, J.; Chintawar, S.; Salto, C.; Pandolfo, M.; Ourednik, V.; Teng, Y.D.; Sidman, R.L.; Arenas, E.; et al. Communication via gap junctions underlies early functional and beneficial interactions between grafted neural stem cells and the host. Proc. Natl. Acad. Sci. USA 2010, 107, 5184–5189. [Google Scholar] [CrossRef] [PubMed]
- Weimann, J.M.; Johansson, C.B.; Trejo, A.; Blau, H.M. Stable reprogrammed heterokaryons form spontaneously in Purkinje neurons after bone marrow transplant. Nat. Cell Biol. 2003, 5, 959–966. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Imitola, J.; Snyder, E.Y.; Sidman, R.L. Neural stem cells rescue nervous purkinje neurons by restoring molecular homeostasis of tissue plasminogen activator and downstream targets. J. Neurosci. 2016, 26, 7839–7848. [Google Scholar] [CrossRef] [PubMed]
- Carter, A.R.; Chen, C.; Schwartz, P.M.; Segal, R.A. Brain-derived neurotrophic factor modulates cerebellar plasticity and synaptic ultrastructure. J. Neurosci. 2002, 22, 1316–1327. [Google Scholar] [CrossRef]
- Huang, Y.; Ko, H.; Cheung, Z.H.; Yung, K.K.; Yao, T.; Wang, J.J.; Morozov, A.; Ke, Y.; Ip, N.Y.; Yung, W.H. Dual actions of brain-derived neurotrophic factor on GABAergic transmission in cerebellar Purkinje neurons. Exp. Neurol. 2012, 233, 791–798. [Google Scholar] [CrossRef]
- Lindvall, O. Developing dopaminergic cell therapy for Parkinson’s disease—Give up or move forward? Mov. Disord. 2013, 28, 268–273. [Google Scholar] [CrossRef]
- Triarhou, L.C.; Zhang, W.; Lee, W.H. Amelioration of the behavioral phenotype in genetically ataxic mice through bilateral intracerebellar grafting of fetal Purkinje cells. Cell Transplant. 1996, 5, 269–277. [Google Scholar] [CrossRef]
- Carletti, B.; Williams, I.M.; Leto, K.; Nakajima, K.; Magrassi, L.; Rossi, F. Time constraints and positional cues in the developing cerebellum regulate Purkinje cell placement in the cortical architecture. Dev. Biol. 2008, 317, 147–160. [Google Scholar] [CrossRef]
- Triarhou, L.C.; Low, W.C.; Ghetti, B. Intraparenchymal grafting of cerebellar cell suspensions to the deep cerebellar nuclei of pcd mutant mice, with particular emphasis on re-establishment of a Purkinje cell cortico-nuclear projection. Anat. Embryol. Berl. 1992, 185, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Roy, N.S.; Cleren, C.; Singh, S.K.; Yang, L.; Beal, M.F.; Goldman, S.A. Functional engraftment of human ES cell-derived dopaminergic neurons enriched by coculture with telomerase-immortalized midbrain astrocytes. Nat. Med. 2006, 12, 1259–1268. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.; Ng, H.H.; Takahashi, R.; Tan, E.K. Induced pluripotent stem cells in Parkinson’s disease: Scientific and clinical challenges. J. Neurol. Neurosurg. Pshychiatry 2016, 87, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Morizane, A.; Li, J.Y.; Brundin, P. From bench to bed: The potential of stem cells for the treatment of Parkinson’s disease. Cell Tissue Res. 2008, 331, 323–336. [Google Scholar] [CrossRef]
| Disorder | Management |
|---|---|
| Metabolic cerebellar ataxias | |
| Alcohol-related cerebellar ataxias | Abstinence and correction of malnutrition, rehabilitation |
| Wernicke’s encephalopathy | Replenishment of vitamin B1 using: (1) thiamine at 100 mg/day (Galvin et al., 2010 [19]), (2) thiamine at a minimum dose of 500 mg three times a day for patients with clinical features (Sechi and Serra, 2007 [15]), (3) parenteral thiamine at 200 mg for patients without apparent clinical features (Ambrose et al., 2001 [20]). |
| Superficial siderosis | Administration of iron chelator: deferiprone; 15 mg/kg body weight/day (Kuo et al., 2017 [25]). |
| Immune-mediated cerebellar ataxias | |
| Gluten ataxia | Strict gluten-free diet. If no benefits are observed, check adherence or hypersensitivity |
| Paraneoplastic cerebellar degeneration | Surgical excision of the tumor followed by immunotherapy: mPSL, IVIg, immunosuppressants, or/and plasma exchange |
| Post-infectious cerebellitis | Often self-limiting. Antibiotics in selected cases. Surgical decompression in case of herniation |
| Anti-GAD ataxia | Induction therapy (mPSL, IVIg, immunosuppressants, plasma exchange, or/and rituximab) followed by maintenance therapy (long-term oral PSL, IVIg, immunosuppressants, or/and rituximab) |
| Proposed Mechanism | Treatment | Efficacy |
|---|---|---|
| Friedreich’s ataxia | ||
| Anti-oxidant | Combination of vitamin E and coenzyme Q10 | Potential therapies
|
| Idebenone | Potential therapies
| |
| Chelation of accumulated iron | Deferiprone | No evidence
|
| Increase in frataxin protein expression level | Interferon | Potential therapies
|
| Ataxia-telangiectasia | ||
| Anti-oxidants | Betamethasone | Potential therapies
|
| Ataxia with vitamin E deficiency | ||
| Replacement of vitamin E | Vitamin E | Approved and supportive therapy |
| Abetalipoproteinemia | ||
| Low fat diet, decreased long-chain fatty acids and oral essential fatty acids Replacement of fat-soluble vitamin | Vitamin A, E, D, K | Approved and supportive therapy |
| Wilson’s disease | ||
| Chelation of accumulated copper | D-penicillamine Trientine Zinc acetate/sulfate | Approved and supportive therapy |
| GLUT1 deficiency | ||
| Ketogenic diet | - | Approved and supportive therapy |
| Refsum’s diseases | ||
| Phytanic acid-free food | Dietary restriction | Approved and supportive therapy |
| Cerebrotendinous xantomatosis | ||
| Replacement of decreased bile acid | chenodeoxycholic acid, ursodeoxycholic acid, cholic acid, and taurocholic acid | Approved and supportive therapy |
| Niemann-Pick disease type C | ||
| Inhibition of glucosylceramide synthesis | Miglustat | Approved and supportive therapy |
| Studies | Protocols | Outcomes |
|---|---|---|
| Motor rehabilitation | ||
| Ilg et al. (2010) [74] |
|
|
| Miyai et al. (2012) [76] |
|
|
| Cognitive rehabilitation | ||
| Maeshima and Osawa (2007) [79] |
|
|
| Schweizer et al. (2008) [80] |
|
|
| Komuro et al. (2014) [81] |
|
|
| Ruffieux et al. (2017) [82] |
|
|
| Studies | Protocols | Outcomes |
|---|---|---|
| Shimizu et al. (1999) [89] |
|
|
| Shiga et al. (2002) [90] rTMS |
|
|
| Kim et al. (2014) [91] rTMS |
|
|
| Grimaldi et al. (2013) [93] anodal tDCS |
|
|
| Grimaldi et al. (2014) [86] anodal tDCS |
|
|
| Benussi et al. (2015) [87] anodal tDCS |
|
|
| Benussi et al. (2017) [88] anodal tDCS |
|
|
| SCA Type | Candidate Drug | Assumed Therapeutic Rationale |
|---|---|---|
| Disease-modifying drugs | ||
| Oligonucleotide therapeutics | ||
| SCA1, 2, 3 | ASO against ATXN1, ATXN2 or ATXN3 | |
| SCA1 | AAV-mediated delivery of short hairpin RNA | |
| SCA3 | Lentiviral-mediated delivery of short hairpin RNA |
|
| SCA3 | AAV-mediated delivery of micro RNA |
|
| SCA6 | AAV-mediated delivery of micro RNA (miR-3139-5q) |
|
| SCA7 | AAV-mediated delivery of micro RNA to retina |
|
| SCA7 | AAV-mediated delivery of micro RNA |
|
| Intervention on downstream pathways | ||
| SCA1 | MSK inhibitor |
|
| SCA2 | Dantrolene |
|
| SCA3 | Dantrolene |
|
| SCA3 | Citalopram |
|
| SCA3 | Aripiprazole |
|
| Neuromodulation therapies | ||
| SCA6 and other SCAs | 4-Aminopyridine |
|
| SCA2 and other SCAs | Chlorzoxazone |
|
| SCA44 | Nitazoxanide |
|
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mitoma, H.; Manto, M.; Gandini, J. Recent Advances in the Treatment of Cerebellar Disorders. Brain Sci. 2020, 10, 11. https://doi.org/10.3390/brainsci10010011
Mitoma H, Manto M, Gandini J. Recent Advances in the Treatment of Cerebellar Disorders. Brain Sciences. 2020; 10(1):11. https://doi.org/10.3390/brainsci10010011
Chicago/Turabian StyleMitoma, Hiroshi, Mario Manto, and Jordi Gandini. 2020. "Recent Advances in the Treatment of Cerebellar Disorders" Brain Sciences 10, no. 1: 11. https://doi.org/10.3390/brainsci10010011
APA StyleMitoma, H., Manto, M., & Gandini, J. (2020). Recent Advances in the Treatment of Cerebellar Disorders. Brain Sciences, 10(1), 11. https://doi.org/10.3390/brainsci10010011







