Separation of N–C5H12–C9H20 Paraffins Using Boehmite by Inverse Gas Chromatography
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Boehmite
2.2. Characterization Techniques
2.3. IGC Determinations
3. Results
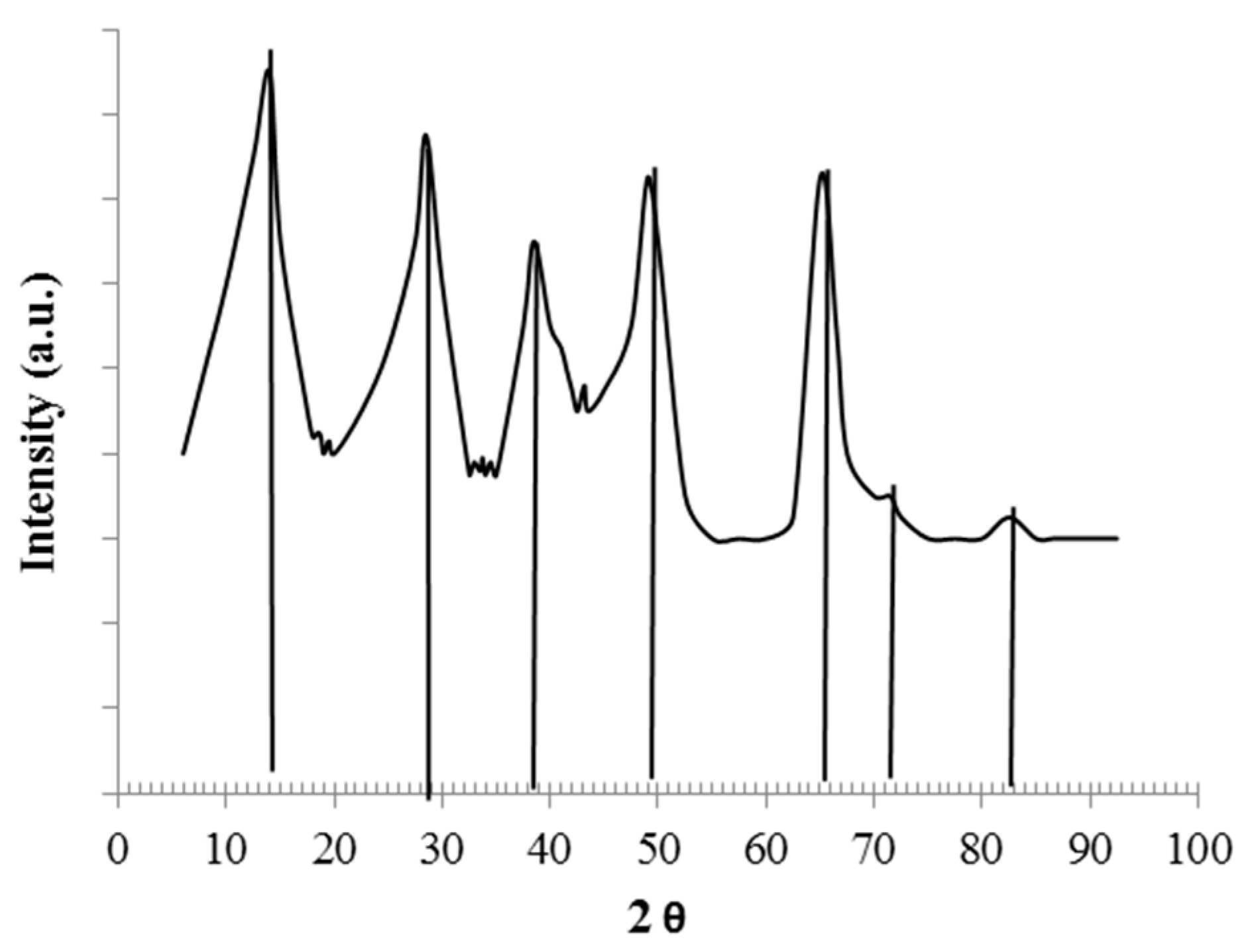
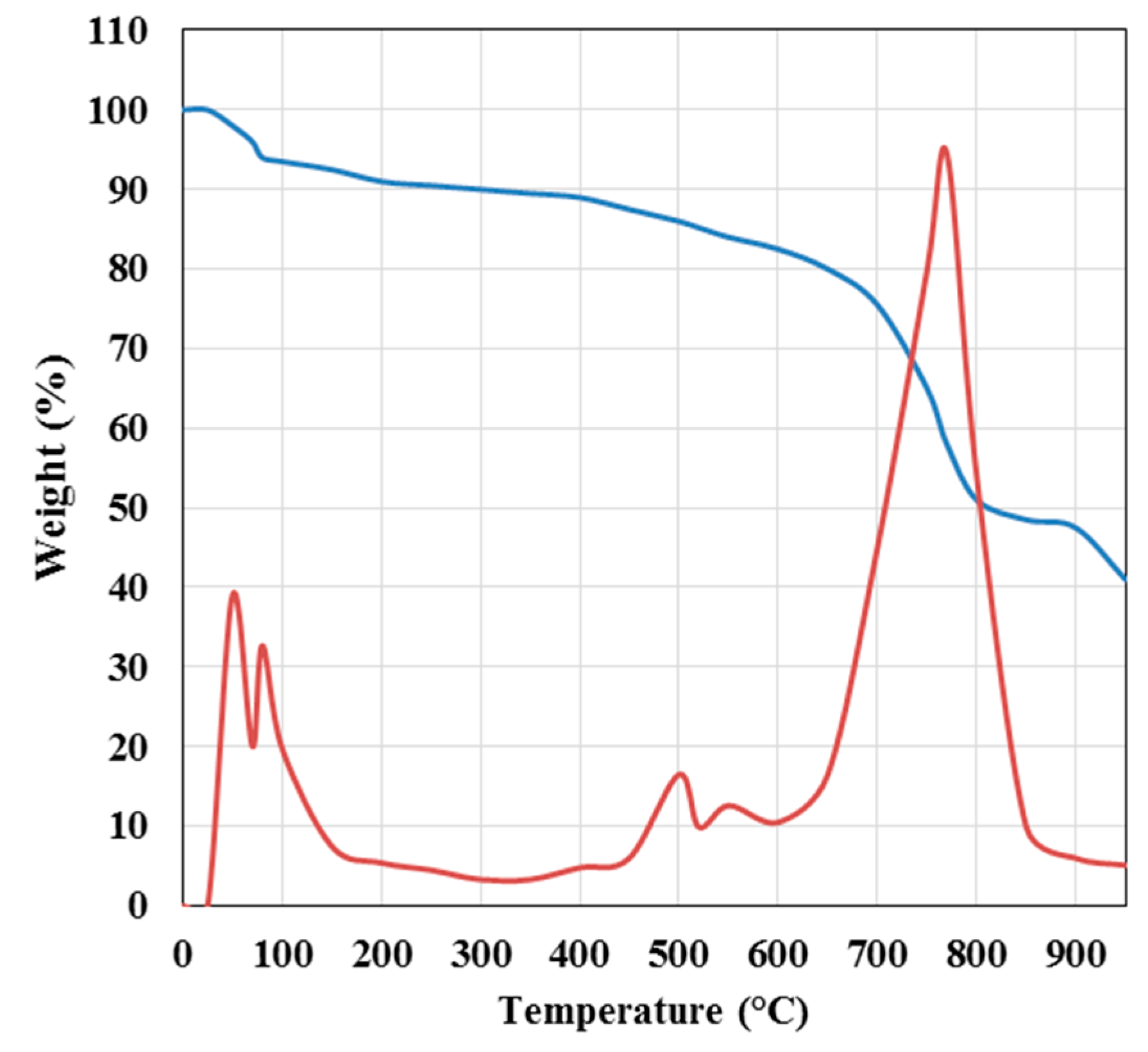
3.1. Samples Characterization
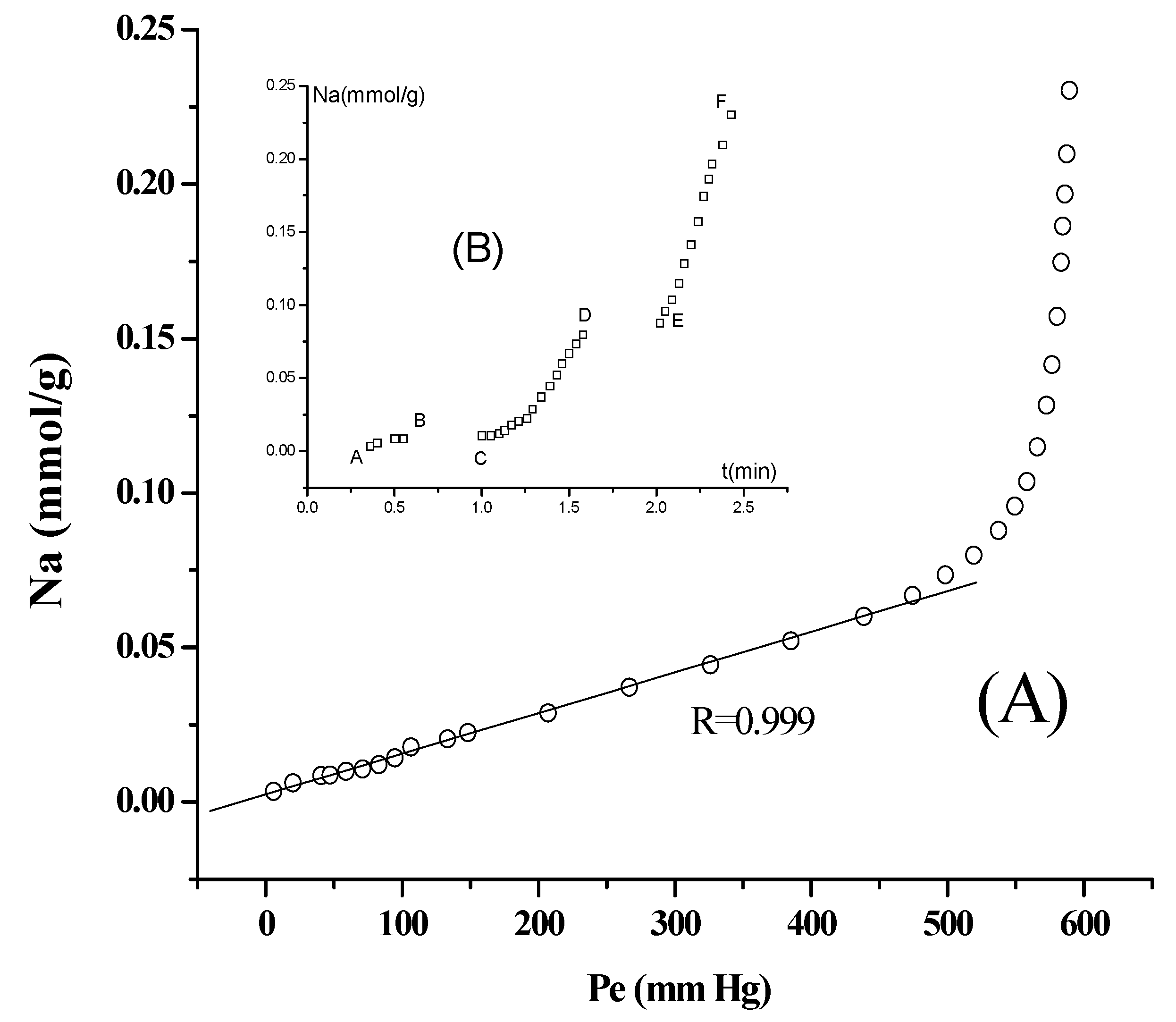
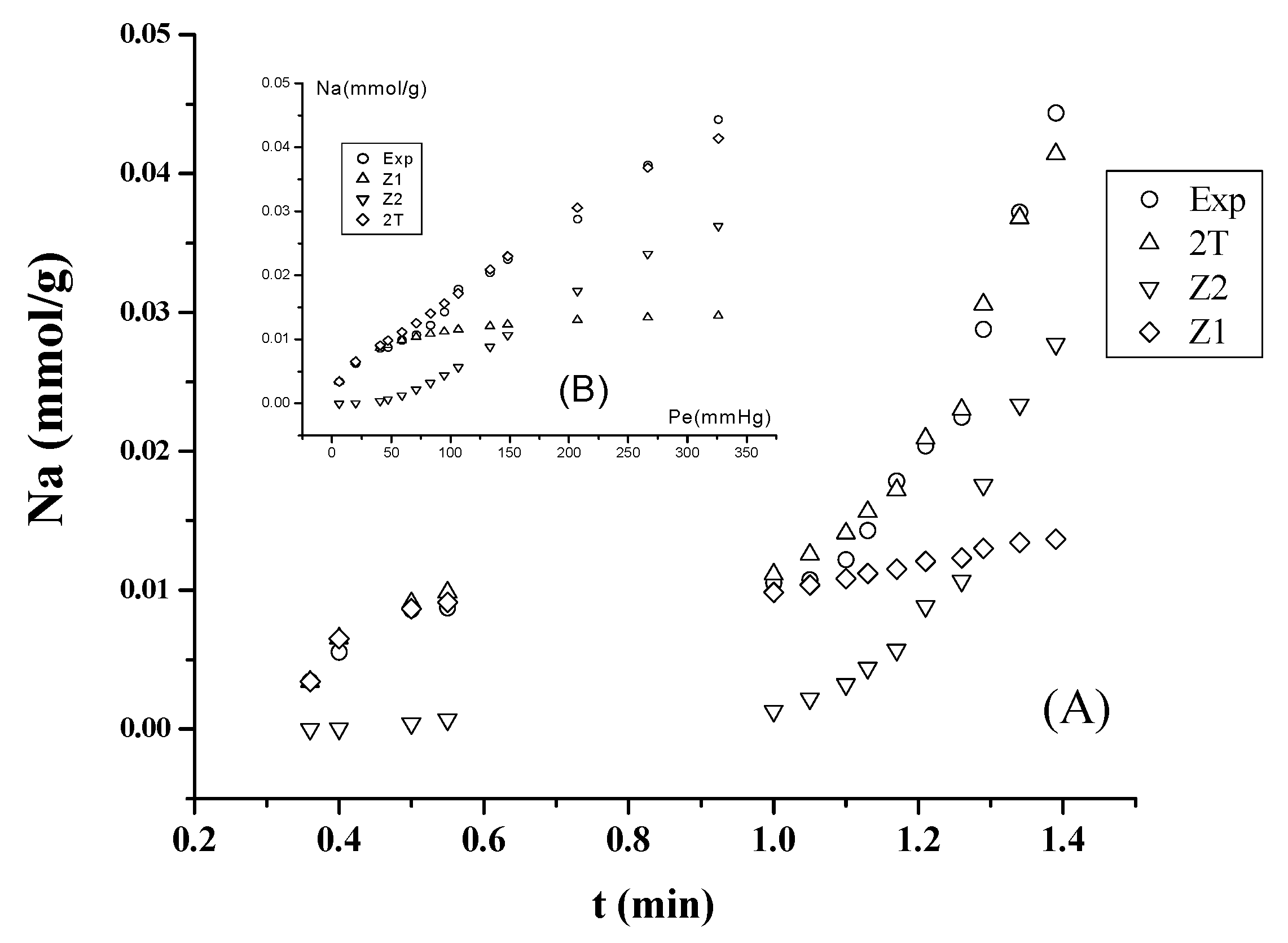
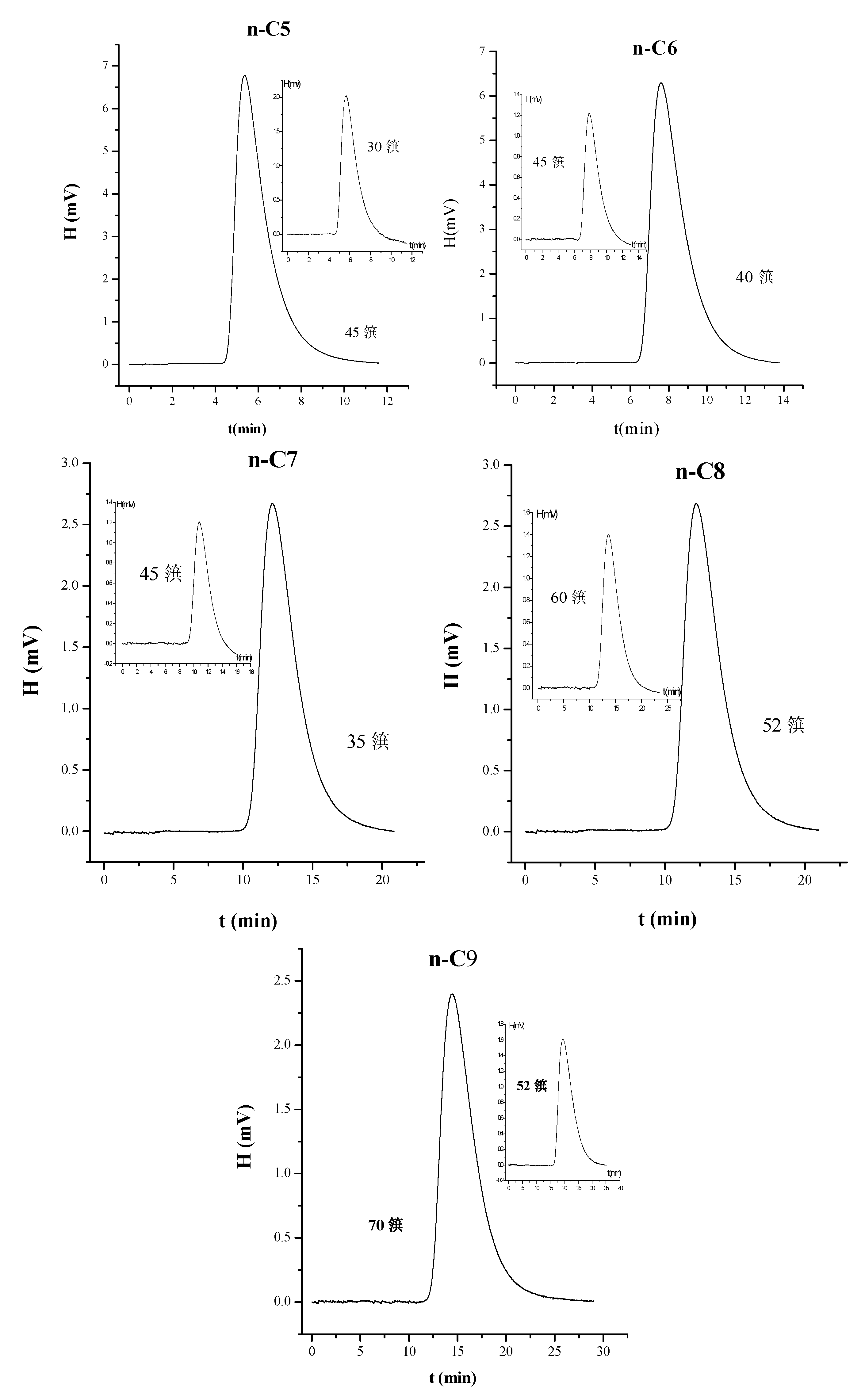
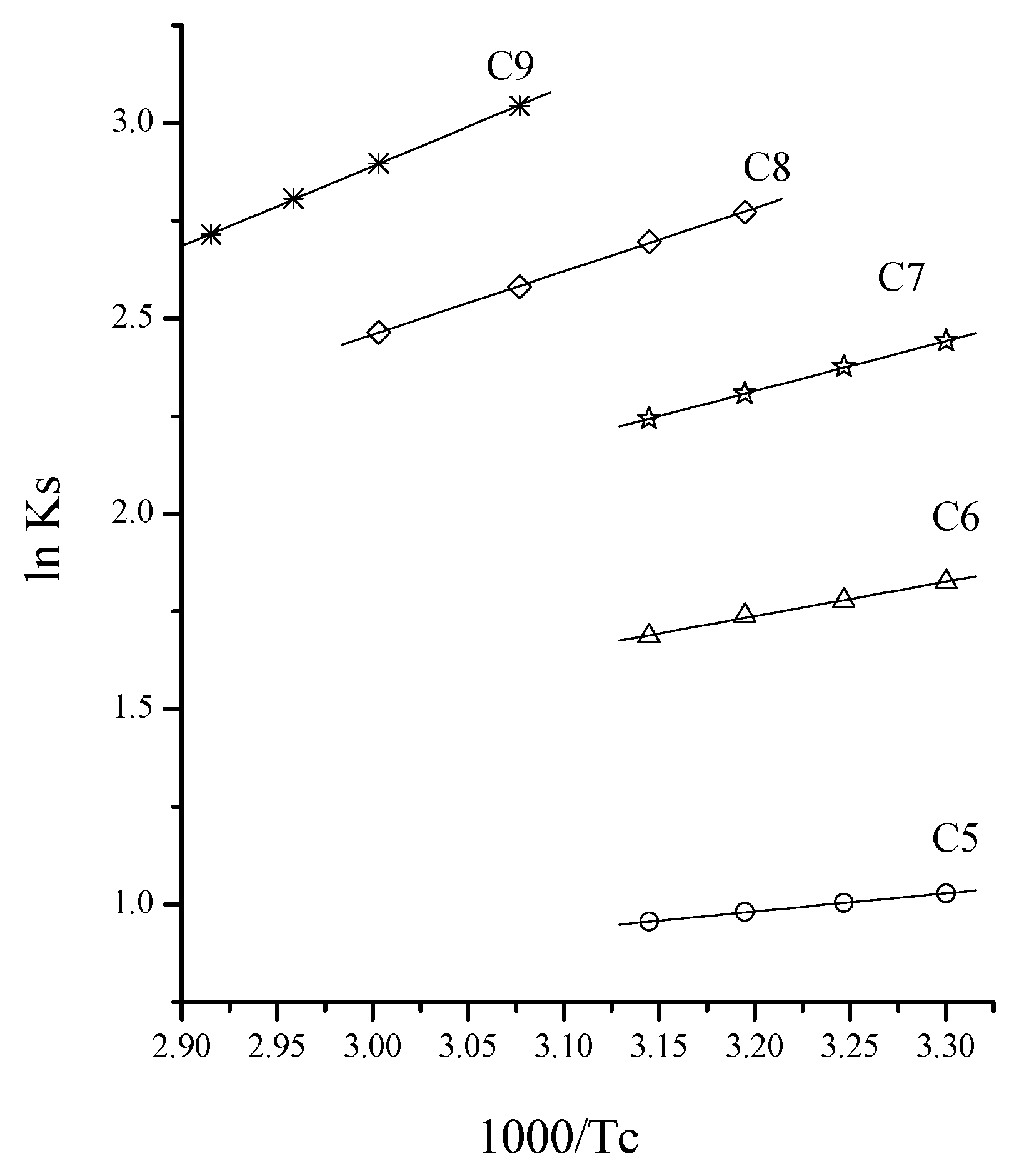
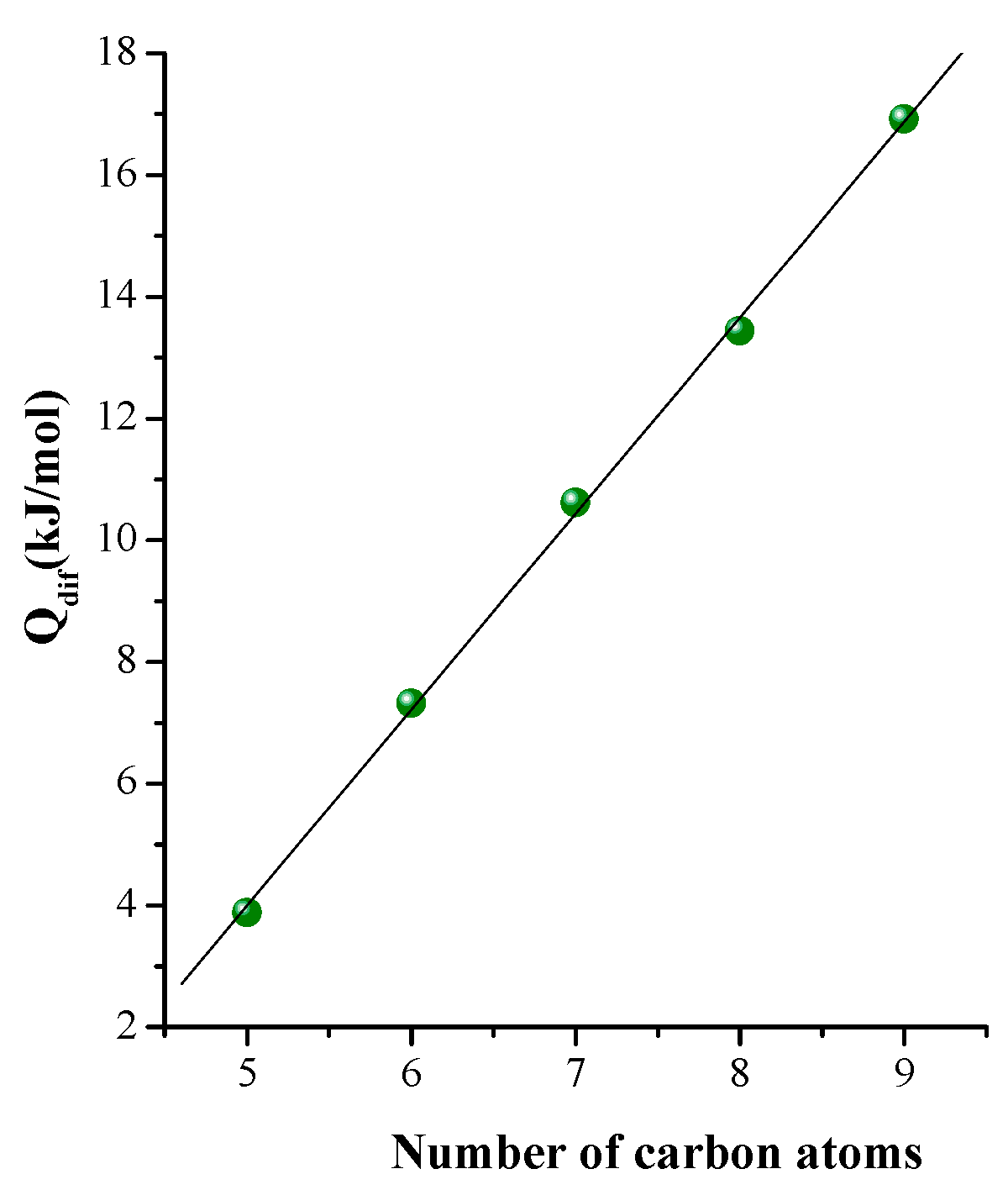
3.2. Separation of n-Paraffins C5–C9 by IGC
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, D.Y.; Lin, Y.S.; Li, Y.C.; Shieh, D.L.; Lin, J.L. Synthesis of mesoporous pseudoboehmite and alumina templated with 1-hexadecyl-2,3-dimethyl-imidazolium chloride. Microporous Mesoporous Mater. 2008, 108, 276–282. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, Y.; Han, B.; Xu, B.; Liu, X.; Yan, Z. Effects of synthetic conditions on the textural structure of pseudo-boehmite. J. Colloid Interface Sci. 2016, 469, 1–7. [Google Scholar] [CrossRef]
- Al-Khattaf†, S. The Influence of Alumina on the Performance of FCC Catalysts during Hydrotreated VGO Catalytic Cracking. Energy Fuels 2002, 17, 62–68. [Google Scholar] [CrossRef]
- Kwak, J.H.; Mei, D.; Peden, C.H.F.; Rousseau, R.; Szanyi, J. (100) facets of γ-Al2O3: The active surfaces for alcohol dehydration reactions. Catal. Lett. 2011, 141, 649–655. [Google Scholar] [CrossRef]
- Keyvanloo, K.; Mardkhe, M.K.; Alam, T.M.; Bartholomew, C.H.; Woodfield, B.F.; Hecker, W.C. Supported Iron Fischer–Tropsch Catalyst: Superior Activity and Stability Using a Thermally Stable Silica-Doped Alumina Support. ACS Catal. 2014, 4, 1071–1077. [Google Scholar] [CrossRef]
- Benítez Guerrero, M.; Pérez-Maqueda, L.A.; Castro, P.P.; Pascual Cosp, J. Alúminas porosas: El método de bio-réplica para la síntesis de alúminas estables de alta superficie específica. Bol. Soc. Esp. Ceram. Vidrio 2013, 52, 251–267. [Google Scholar] [CrossRef]
- Sanchez-Valente, J.; Bokhimi, X.; Toledo, J. Synthesis and catalytic properties of nanostructured aluminas obtained by sol–gel method. Appl. Catal. A Gen. 2004, 264, 175–181. [Google Scholar] [CrossRef]
- García, G.; Falco, M.; Crespo, P.; Cabrera, S.; Sedran, U. Characterization and catalytic evaluation of aluminum oxides obtained by the atrane route. Catal. Today 2011, 166, 60–66. [Google Scholar] [CrossRef]
- Chen, J.-F.; Shao, L.; Guo, F.; Wang, X.-M. Synthesis of nano-fibers of aluminum hydroxide in novel rotating packed bed reactor. Chem. Eng. Sci. 2003, 58, 569–575. [Google Scholar] [CrossRef]
- Yuan, Q.; Yin, A.-X.; Luo, C.; Sun, L.-D.; Zhang, Y.-W.; Duan, W.-T.; Liu, H.-C.; Yan, C.-H. Facile Synthesis for Ordered Mesoporous γ-Aluminas with High Thermal Stability. J. Am. Chem. Soc. 2008, 130, 3465–3472. [Google Scholar] [CrossRef]
- Voelkel, A.; Strzemiecka, B.; Adamska, K.; Milczewska, K. Inverse gas chromatography as a source of physiochemical data. J. Chromatogr. A 2009, 1216, 1551–1566. [Google Scholar] [CrossRef] [PubMed]
- Batko, K.; Voelkel, A. Inverse gas chromatography as a tool for investigation of nanomaterials. J. Colloid Interface Sci. 2007, 315, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Papirer, E.; Perrin, J.-M.; Siffert, B.; Philipponneau, G. Surface characteristics of aluminas in relation with polymer adsorption. J. Colloid Interface Sci. 1991, 144, 263–270. [Google Scholar] [CrossRef]
- Onjia, A.E.; Milonjić, S.K.; Rajaković, L.V. Inverse gas chromatography of chromia. Part I. Zero surface coverage. J. Serbian Chem. Soc. 2001, 66, 259–271. [Google Scholar] [CrossRef]
- Onjia, A.E.; Milonjić, S.K.; Rajaković, L.V. Inverse gas chromatography of chromia. Part II. Finite surface coverage. J. Serbian Chem. Soc. 2002, 67, 165–178. [Google Scholar] [CrossRef]
- Rials, T.G.; Simonsen, J. Investigating interphase development in woodpolymer composites by inverse gas chromatography. Compos. Interfaces 2000, 7, 81–92. [Google Scholar] [CrossRef][Green Version]
- Wang, B.; Sain, M. The effect of chemically coated nanofiber reinforcement on biopolymer based nanocomposites. BioResources 2007, 2, 371–388. [Google Scholar]
- Autie-Castro, G.; Autie, M.; Reguera, E.; Santamaría-González, J.; Moreno-Tost, R.; Rodríguez-Castellón, E.; Jiménez-López, A. Adsorption and separation of light alkane hydrocarbons by porous hexacyanocobaltates (III). Surf. Interface Anal. 2009, 41, 730–734. [Google Scholar] [CrossRef]
- Rivera, A.; Farías, T.; de Ménorval, L.C.; Autié-Castro, G.; Yee-Madeira, H.; Contreras, J.L.; Autié-Pérez, M. Acid natural clinoptilolite: Structural properties against adsorption/separation of n-paraffins. J. Colloid Interface Sci. 2011, 360, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Hechevarría, H.M.; Labadie Suarez, J.M.; Santamaría-González, J.; Infantes-Molina, A.; Autié-Castro, G.; Cavalcante, C.L.; Rodríguez-Castellón, E.; Autie-Pérez, M. Adsorption and separation of propane and propylene by Cuban natural volcanic glass. Mater. Chem. Phys. 2015, 168, 132–137. [Google Scholar] [CrossRef]
- Autie-Pérez, M.; Infantes-Molina, A.; Cecilia, J.A.; Labadie-Suárez, J.M.; Rodríguez-Castellón, E. Separation of light liquid paraffin C5-C9 with Cuban volcanic glass previously used in copper elimination from water solutions. Appl. Sci. 2018, 8, 295. [Google Scholar] [CrossRef]
- Fuertes, A.B.; Menendez, I. Separation of hydrocarbon gas mixtures using phenolic resin-based carbon membranes. Sep. Purif. Technol. 2002, 28, 29–41. [Google Scholar] [CrossRef]
- Kwon, H.T.; Jeong, H.-K. In Situ Synthesis of Thin Zeolitic–Imidazolate Framework ZIF-8 Membranes Exhibiting Exceptionally High Propylene/Propane Separation. J. Am. Chem. Soc. 2013, 135, 10763–10768. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.G.; Lin, Y.S. Granulation of Sol-Gel-Derived Nanostructured Alumina. AIChE J. 1997, 43, 505–514. [Google Scholar] [CrossRef]
- Yang, Z.; Lin, Y.S. Sol-Gel Synthesis of Silicalite/γ-Alumina Granules. Ind. Eng. Chem. Res. 2000, 39, 4944–4948. [Google Scholar] [CrossRef]
- Dubinin, M.; Stoeckli, H. Homogeneous and heterogeneous micropore structures in carbonaceous adsorbents. J. Colloid Interface Sci. 1980, 75, 34–42. [Google Scholar] [CrossRef]
- Dubinin, M.M. Generalization of the theory of volume filling of micropores to nonhomogeneous microporous structures. Carbon N. Y. 1985, 23, 373–380. [Google Scholar] [CrossRef]
- Rivera, A.; Farías, T.; Charles de Meorval, L.; AutiePeez, M.; Lam, A. Natural and Sodium Clinoptilolites Submitted to Acid Treatments: Experimental and Theoretical Studies. J. Phys. Chem. C 2013, 117, 4079–4088. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, M.K.; Rhee, C.K.; Kim, W.W. Control of hydrolytic reaction of aluminum particles for aluminum oxide nanofibers. Mater. Sci. Eng. A 2004, 375–377, 1263–1268. [Google Scholar] [CrossRef]
- Aguado, J.; Escola, J.M.; Castro, M.C.; Paredes, B. Sol–gel synthesis of mesostructured γ-alumina templated by cationic surfactants. Microporous Mesoporous Mater. 2005, 83, 181–192. [Google Scholar] [CrossRef]
- Frost, R.L.; Gobac, Ž.Ž.; López, A.; Xi, Y.; Scholz, R.; Lana, C.; Lima, R.M.F. Characterization of the sulphate mineral coquimbite, a secondary iron sulphate from Javier Ortega mine, Lucanas Province, Peru - Using infrared, Raman spectroscopy and thermogravimetry. J. Mol. Struct. 2014, 1063, 251–258. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, A.; Wang, X.; Zhang, T. Morphologically controlled synthesis of mesoporous alumina. Microporous Mesoporous Mater. 2007, 100, 35–44. [Google Scholar] [CrossRef]
- Park, Y.K.; Tadd, E.H.; Zubris, M.; Tannenbaum, R. Size-controlled synthesis of alumina nanoparticles from aluminum alkoxides. Mater. Res. Bull. 2005, 40, 1506–1512. [Google Scholar] [CrossRef]
- Busca, G. The surface of transitional aluminas: A critical review. Catal. Today 2014, 226, 2–13. [Google Scholar] [CrossRef]
- Ram, S. Infrared spectral study of molecular vibrations in amorphous, nanocrystalline and AlO(OH) · αH2O bulk crystals. Infrared Phys. Technol. 2001, 42, 547–560. [Google Scholar] [CrossRef]
- Kanti Naskar, M.; Chatterjee, M. Boehmite Nanoparticles by the Two-Reverse Emulsion Technique. J. Am. Ceramic Soc. 2005, 88, 3322–3326. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, S.; Lin, J.; Song, H.; Luo, J.; Elssfah, E.M.; Ammar, E.; Huang, Y.; Ding, X.; Gao, J.; Qi, S.; Tang, C. Self-Assembly of Flowerlike AlOOH (Boehmite) 3D Nanoarchitectures. J. Phys. Chem. 2006, 110, 14249–14252. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Shi, F.; Lin, J.; Wei, S.Y.; Chen, D.; Gao, J.M.; Huang, Z.; Ding, X.X.; Tang, C. Nanoparticles assembly of boehmite nanofibers without a surfactant. Mater. Res. Bull. 2008, 43, 1709–1715. [Google Scholar] [CrossRef]
- Liu, Y.; Ma, D.; Han, X.; Bao, X.; Frandsen, W.; Wang, D.; Su, D. Hydrothermal synthesis of microscale boehmite and gamma nanoleaves alumina. Mater. Lett. 2008, 62, 1297–1301. [Google Scholar] [CrossRef]
- Elizalde-González, M.P.; Pérez-Cruz, M.A. Interaction between organic vapors and clinoptilolite–mordenite rich tuffs in parent, decationized, and lead exchanged forms. J. Colloid Interface Sci. 2007, 312, 317–325. [Google Scholar] [CrossRef]
- Christoph, G.G.; Corbato, C.E.; Hofmann, D.A.; Tettenhorst, R.T. Crystal Structure of Boehmite. Clays Clay Miner. 1979, 27, 81–86. [Google Scholar] [CrossRef]
- Hill, R.J. Hydrogen Atoms in Boehmite. a Single Crystal X-Ray Diffraction and Molecular Orbital Study. Clays Clay Miner. 1981, 29, 435. [Google Scholar] [CrossRef]
- Tettenhorst, R.; Hofmann, D.A. Crystal Chemistry of Boehmite. Clays Clay Miner. 1980, 28, 373–380. [Google Scholar] [CrossRef]


| Zone | Nm [mmol/g] | Ec [J/mol] | Dp [nm]. |
|---|---|---|---|
| 1 | 0.041 | 2481 | 5.2 |
| 2 | 0.056 | 669 | 19.4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Contreras-Larios, J.L.; Infantes-Molina, A.; Negrete-Melo, L.A.; Labadie-Suárez, J.M.; Yee-Madeira, H.T.; Autie-Pérez, M.A.; Rodríguez-Castellón, E. Separation of N–C5H12–C9H20 Paraffins Using Boehmite by Inverse Gas Chromatography. Appl. Sci. 2019, 9, 1810. https://doi.org/10.3390/app9091810
Contreras-Larios JL, Infantes-Molina A, Negrete-Melo LA, Labadie-Suárez JM, Yee-Madeira HT, Autie-Pérez MA, Rodríguez-Castellón E. Separation of N–C5H12–C9H20 Paraffins Using Boehmite by Inverse Gas Chromatography. Applied Sciences. 2019; 9(9):1810. https://doi.org/10.3390/app9091810
Chicago/Turabian StyleContreras-Larios, José L., Antonia Infantes-Molina, Luís A. Negrete-Melo, Juan M. Labadie-Suárez, Hernani T. Yee-Madeira, Miguel A. Autie-Pérez, and Enrique Rodríguez-Castellón. 2019. "Separation of N–C5H12–C9H20 Paraffins Using Boehmite by Inverse Gas Chromatography" Applied Sciences 9, no. 9: 1810. https://doi.org/10.3390/app9091810
APA StyleContreras-Larios, J. L., Infantes-Molina, A., Negrete-Melo, L. A., Labadie-Suárez, J. M., Yee-Madeira, H. T., Autie-Pérez, M. A., & Rodríguez-Castellón, E. (2019). Separation of N–C5H12–C9H20 Paraffins Using Boehmite by Inverse Gas Chromatography. Applied Sciences, 9(9), 1810. https://doi.org/10.3390/app9091810







