Conversion of Stearic Acid into Bio-Gasoline over Pd/ZSM-5 Catalysts with Enhanced Accessibility
Abstract
1. Introduction
2. Experimental Procedure
2.1. Catalysts Preparation
2.2. Catalysts Characterization
2.3. Catalytic Tests of Stearic Acid Conversion
2.4. Analysis of the Reaction Products
3. Results and Discussion
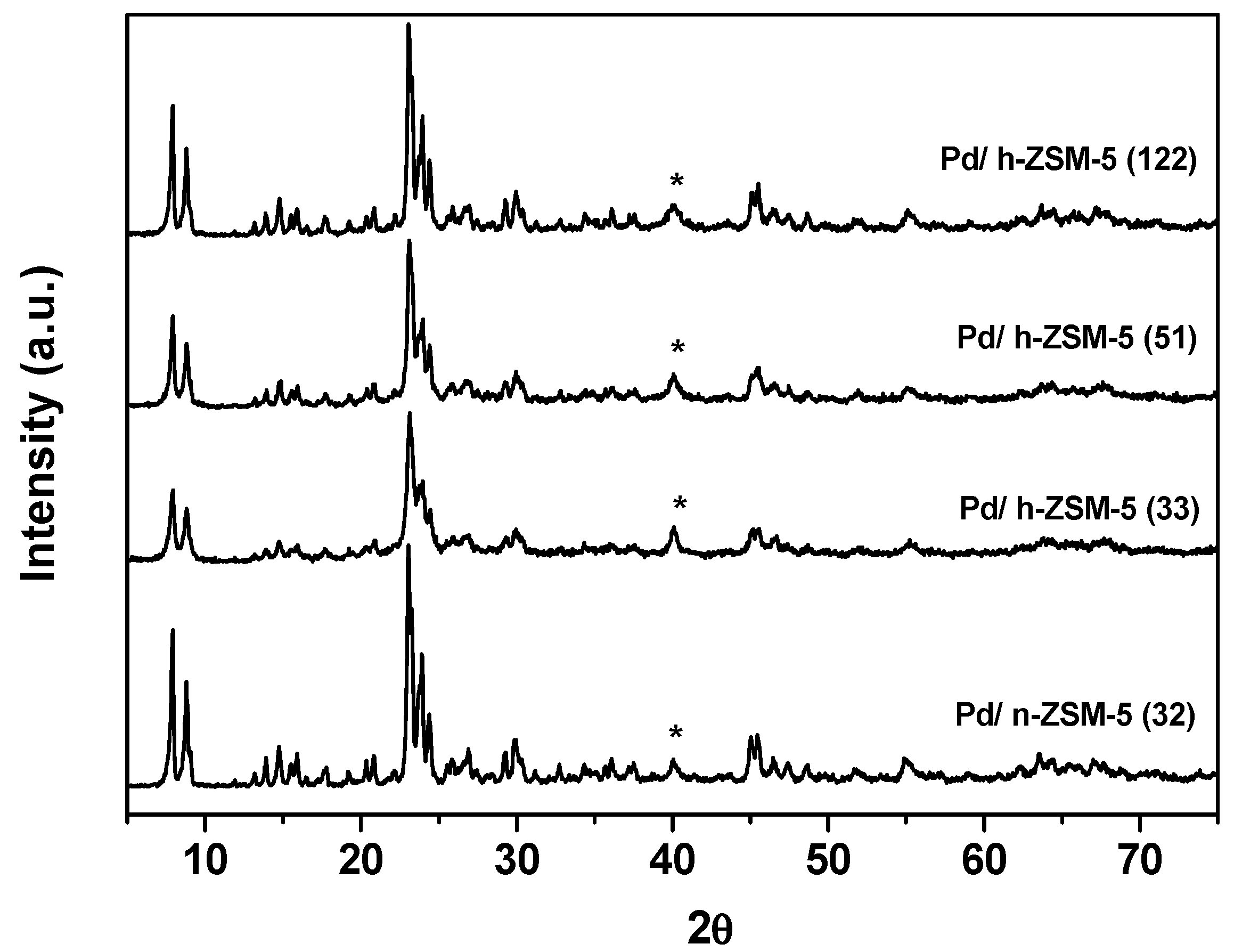
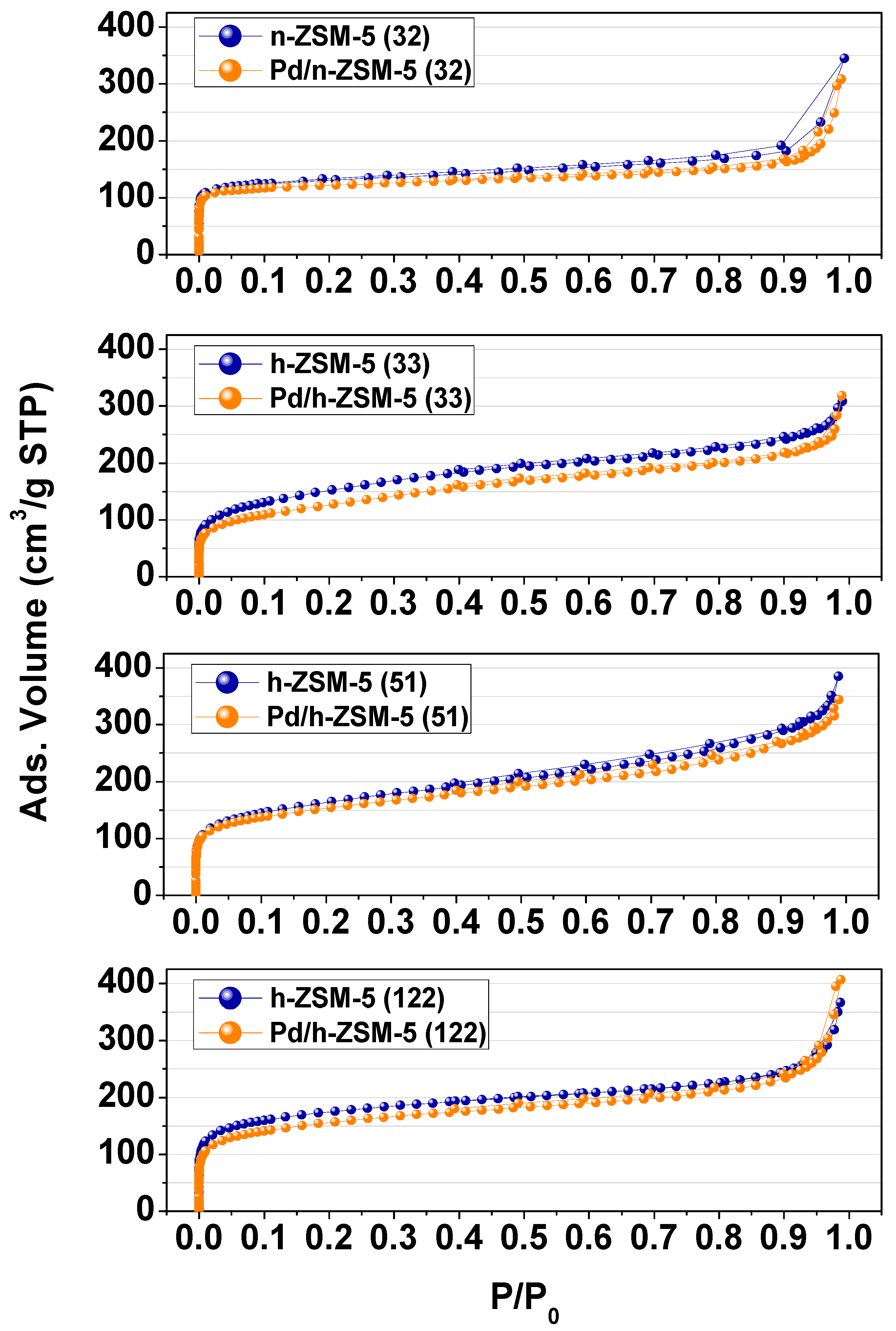
3.1. Catalysts Properties
3.2. Reactions of Stearic Acid
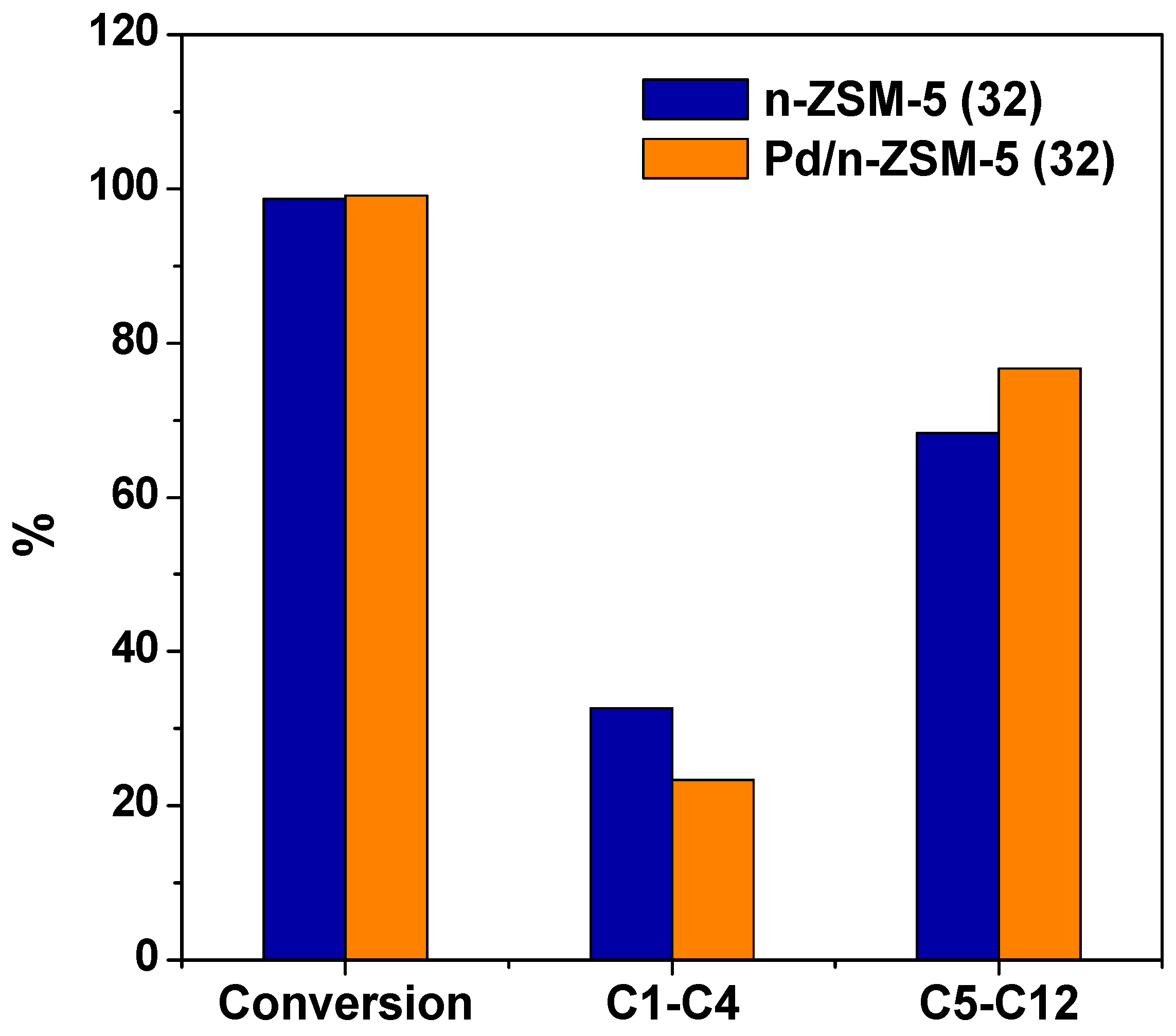
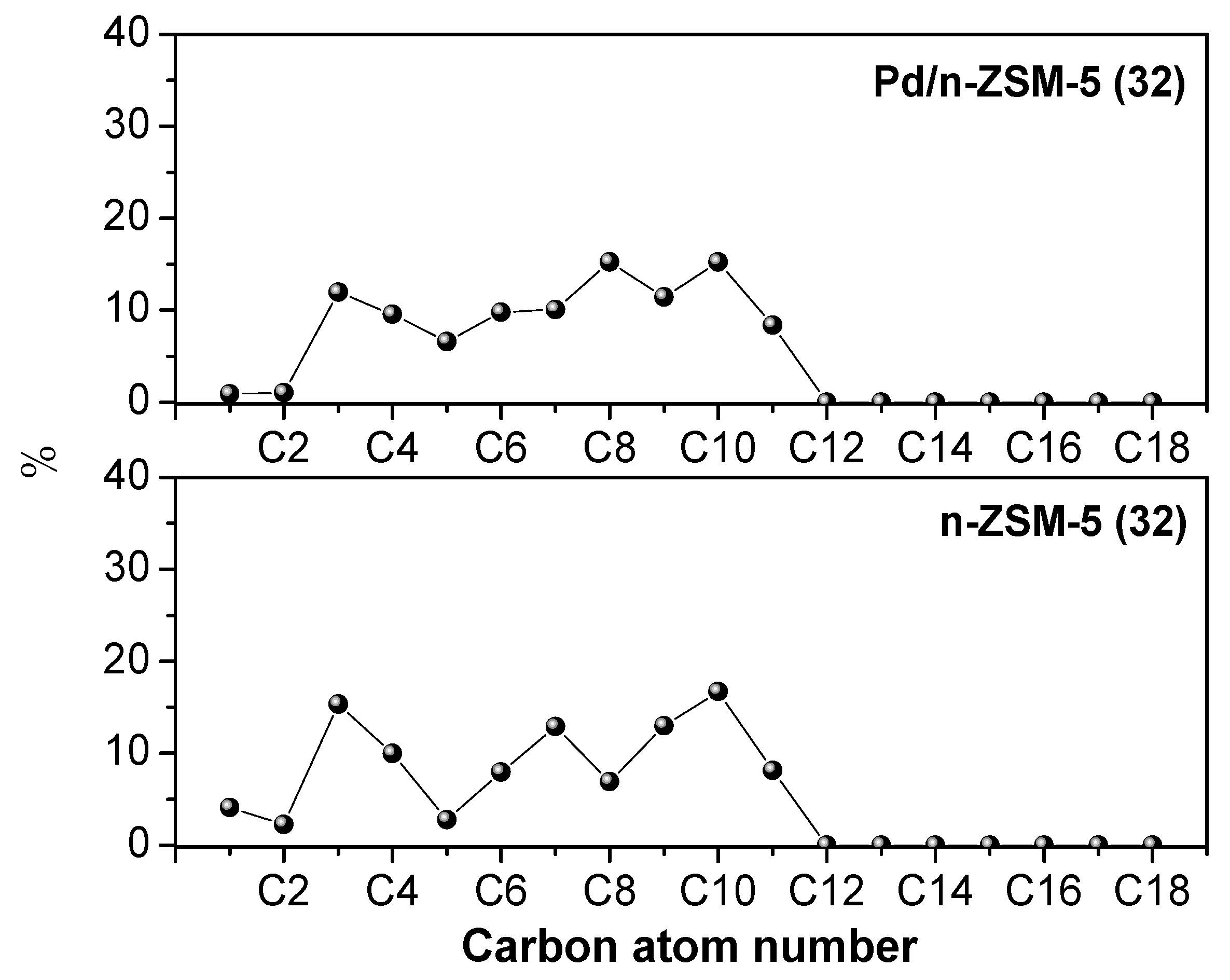
3.2.1. Effect of the Pd Incorporation
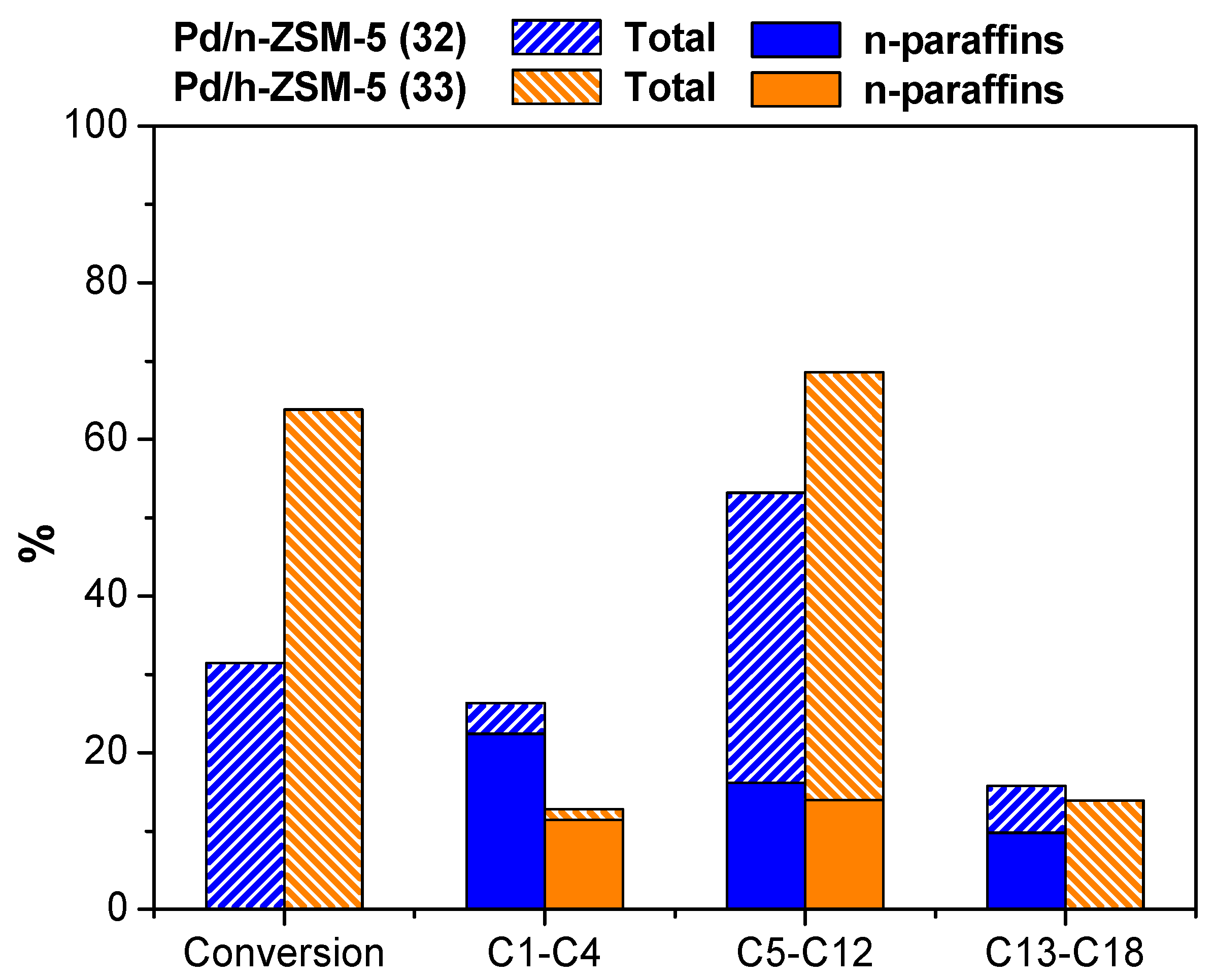
3.2.2. Influence of the Hierarchical Porosity
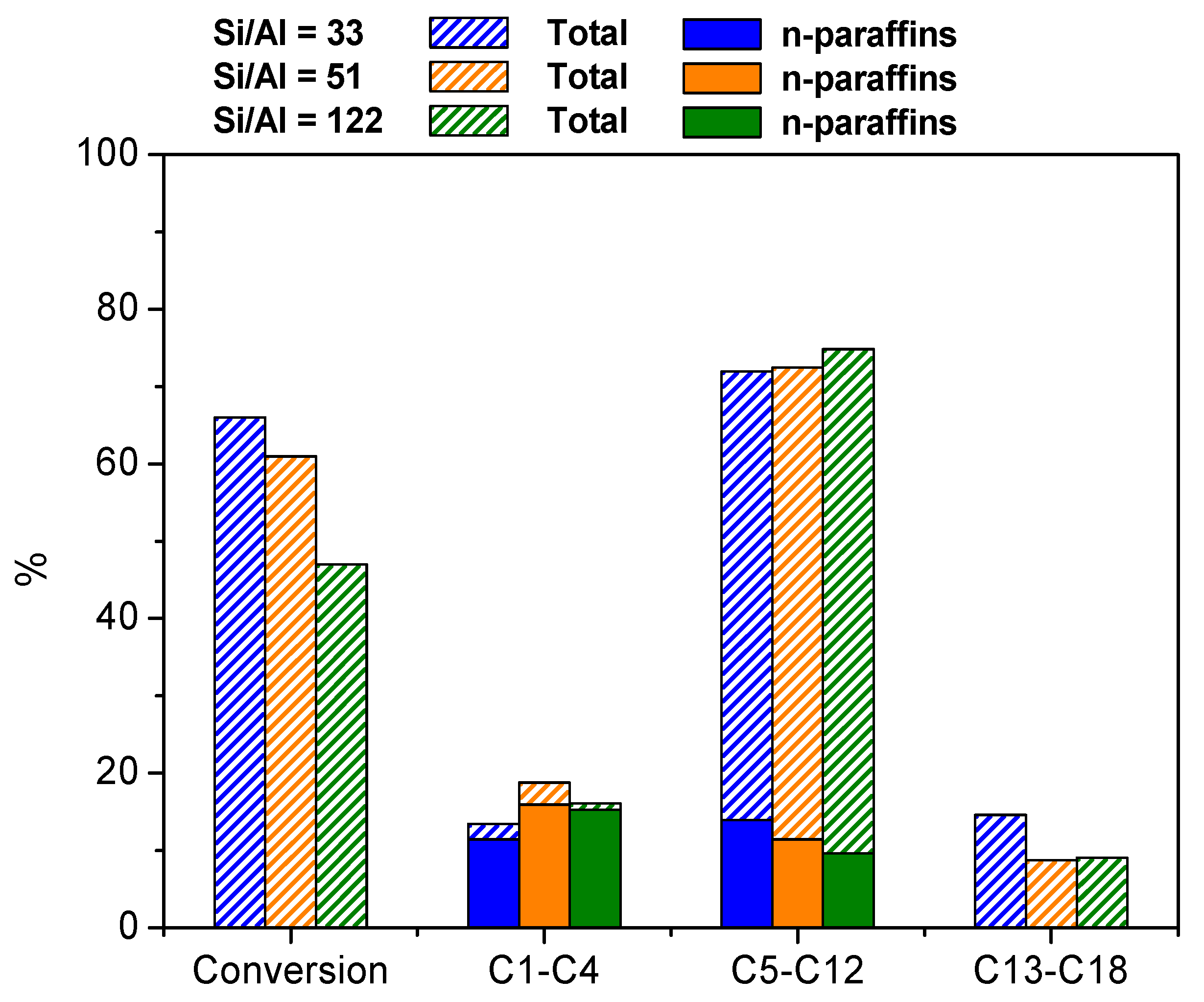
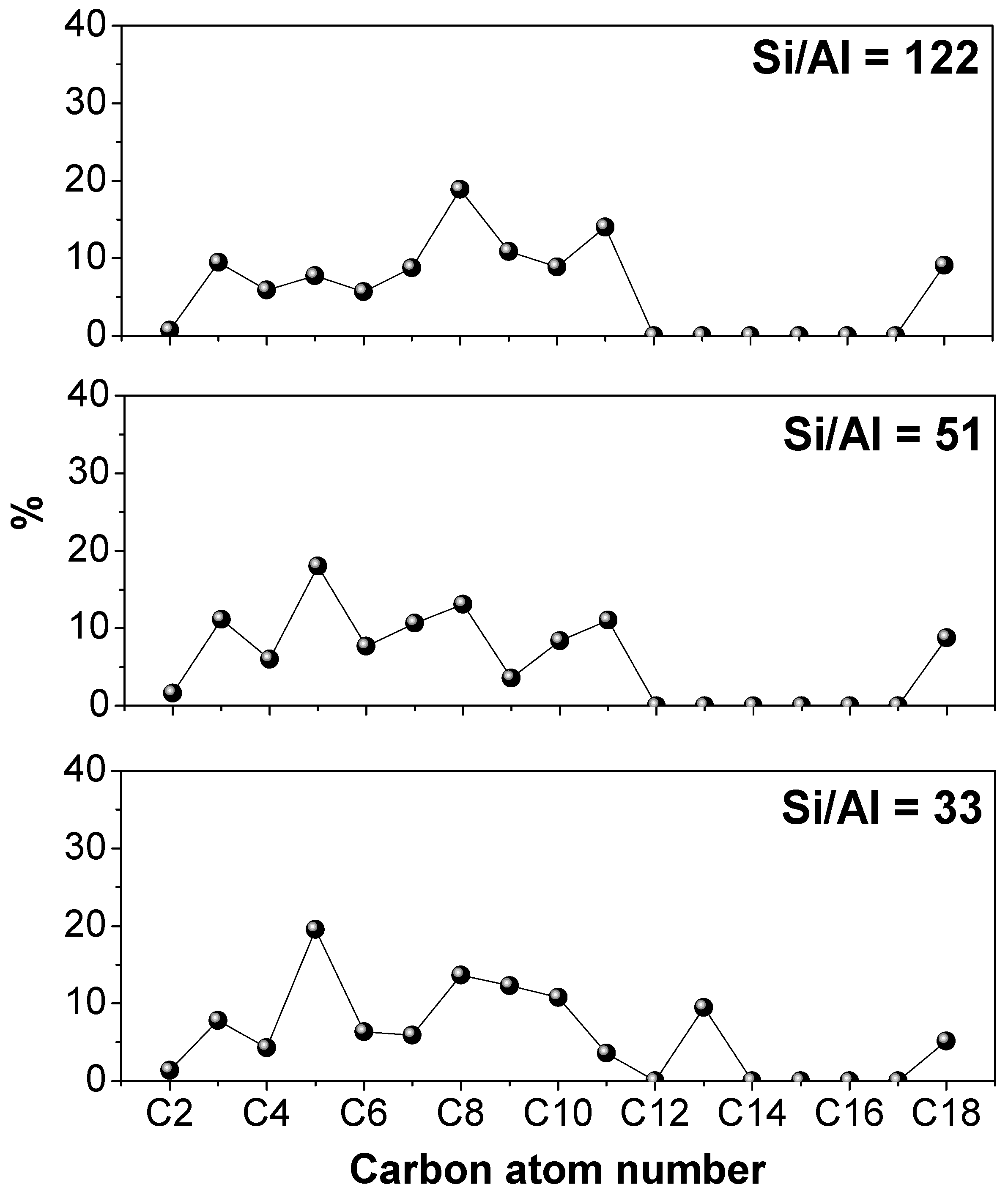
3.2.3. Effect of the Si/Al Atomic Ratio in Hierarchical ZSM-5
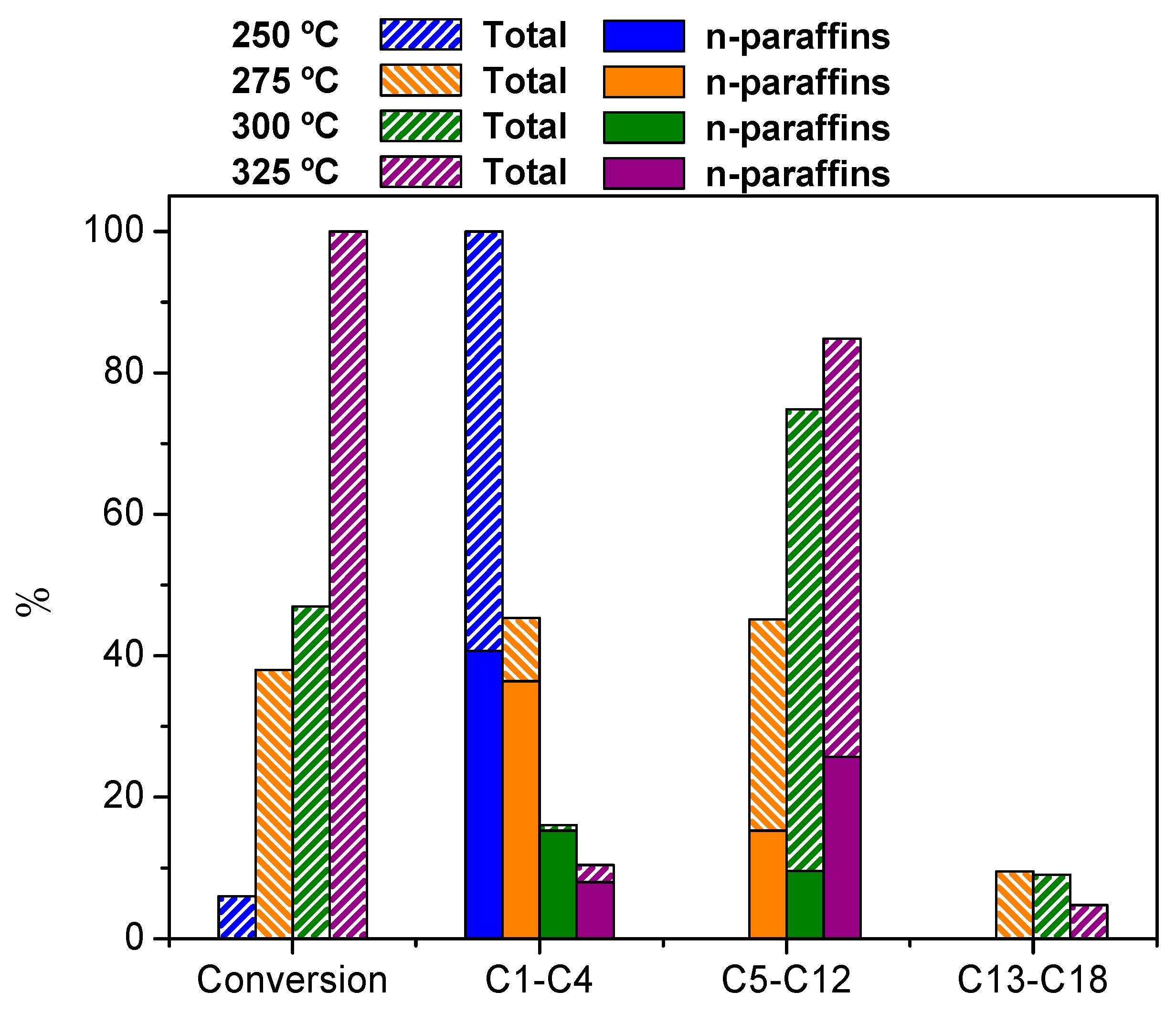
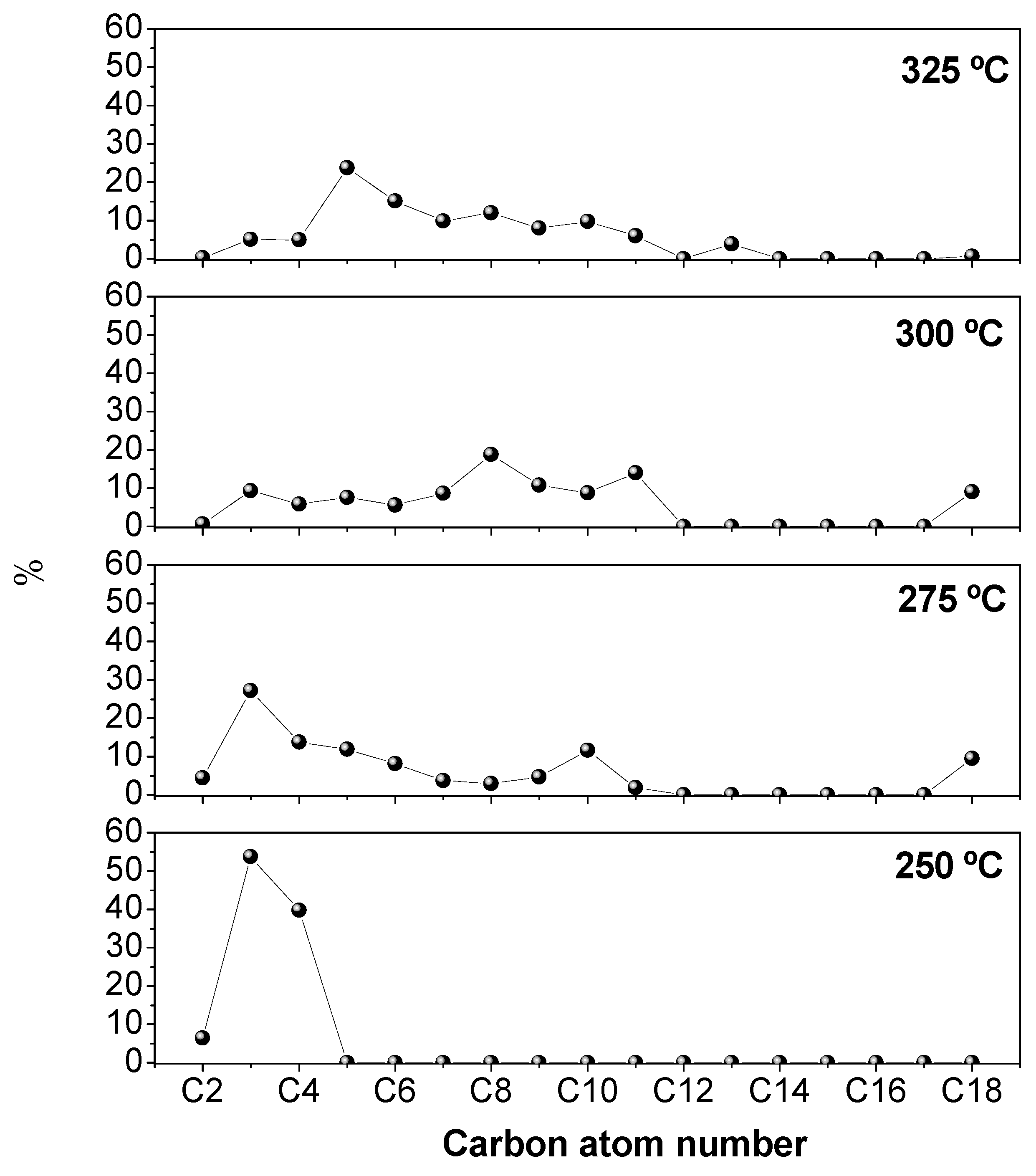
3.2.4. Effect of the Reaction Temperature over Hierarchical ZSM-5
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Meher, L.C.; Vidya Sagar, D.; Naik, S.N. Technical aspects of biodiesel production by transesterification-a review. Renew. Sustain. Energy Rev. 2006, 10, 248–268. [Google Scholar] [CrossRef]
- Leung, D.Y.C.; Wu, X.; Leung, M.K.H. A review on biodiesel production using catalyzed transesterification. Appl. Energy 2010, 87, 1083–1095. [Google Scholar] [CrossRef]
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef]
- Zhao, C.; Brück, T.; Lercher, J.A. Catalytic deoxygenation of microalgae oil to green hydrocarbons. Green Chem. 2013, 15, 1720–1739. [Google Scholar] [CrossRef]
- Benjumea, P.; Agudelo, J.; Agudelo, A. Basic properties of palm oil biodiesel—Diésel blends. Fuel 2008, 87, 2069–2075. [Google Scholar] [CrossRef]
- Corma, A.; Huber, G.W.; Sauvanaud, L.; O’Connor, P. Processing biomass derived oxygenates in the oil refinery: Catalytic cracking (FCC) reaction pathways and role of catalyst. J. Catal. 2007, 247, 307–327. [Google Scholar] [CrossRef]
- Huber, G.W.; Corma, A. Synergies between bio- and oil refineries for the production of fuel from biomass. Angew. Chem. Int. Ed. 2007, 46, 7184–7201. [Google Scholar] [CrossRef]
- Twaiq, F.A.; Mohamed, A.R.; Bhatia, S. Liquid hydrocarbon fuels from palm oil by catalytic cracking over aluminosilicate mesoporous catalysts with various Si/Al ratios. Microporous Mesoporous Mater. 2003, 64, 95–107. [Google Scholar] [CrossRef]
- Katikaneni, S.P.R.; Adjaye, J.D.; Bakhshi, N.N. Studies on the catalytic conversión of canola oil to hydrocarbons: Influence of the hybrid catalysts and steam. Energy Fuels 1995, 9, 599–609. [Google Scholar] [CrossRef]
- Ooi, Y.-S.; Zakaria, R.; Mohamed, A.R.; Bhatia, S. Catalytic conversión of fatty acids mixture to liquid fuel and chemicals over composite microporous/mesoporous catalysts. Energy Fuels 2005, 19, 736–743. [Google Scholar] [CrossRef]
- Danuthai, T.; Jongpatiwut, S.; Rirksomboon, T.; Osuwan, S.; Resaco, R.E. Conversion of methylesters to hydrocarbons over Zn-modified HZSM-5 zeolite catalyst. Catal. Lett. 2009, 132, 197–204. [Google Scholar] [CrossRef]
- Kubička, D.; Kaluža, L. Deoxygenation of vegetable oils over sulfided Ni, Mo and NiMo catalysts. Appl. Catal. A 2010, 372, 199–208. [Google Scholar] [CrossRef]
- Toba, M.; Abe, Y.; Kuramochi, H.; Osako, M.; Mochizuki, T.; Yoshimura, Y. Hydrodeoxygenation of waste vegetable oil over sulfide catalysts. Catal. Today 2011, 164, 533–537. [Google Scholar] [CrossRef]
- Ochoa-Hernández, C.; Yang, Y.; Pizarro, P.; de la Peña O’Shea, V.A.; Coronado, J.M.; Serrano, D.P. Hydrocarbons production through hydrotreating of methyl esters over Ni and Co supported on SBA-15 and Al-SBA-15. Catal. Today 2013, 210, 81–88. [Google Scholar] [CrossRef]
- Boda, L.; Onyestyák, G.; Solt, H.; Lóny, F.; Valyon, J.; Thernesz, A. Catalytic hydroconversion of tricaprylin and caprylic acid as model reaction for biofuel production from triglycerides. Appl. Catal. A 2010, 374, 158–169. [Google Scholar] [CrossRef]
- Berenguer, A.; Bennett, J.A.; Hunns, J.; Moreno, I.; Coronado, J.M.; Lee, A.F.; Pizarro, P.; Wilson, K.; Serrano, D.P. Catalytic hydrodeoxygenation of m-cresol over Ni2P/hierarchical ZSM-5. Catal. Today 2018, 304, 72–79. [Google Scholar] [CrossRef]
- Dierks, M.; Cao, Z.; Manayil, J.C.; Akilavasan, J.; Wilson, K.; Schüth, F.; Rinaldi, R. Impact of hydrophobic organohybrid silicas on the stability of Ni2P catalysts phase in the hydrodeoxygenation of biophenols. ChemCatChem 2018, 10, 2219–2231. [Google Scholar] [CrossRef]
- Snåre, M.; Kubickova, I.; Mäki-Arvela, P.; Murzin, D.Y. Heterogeneous catalytic deoxygenation of stearic acid for production of biodiesel. Ind. Eng. Chem. Res. 2006, 45, 5708–5715. [Google Scholar] [CrossRef]
- Ping, E.W.; Pierson, J.; Wallace, R.; Miller, J.T.; Fuller, T.F.; Jones, C.W. On the nature of the deactivation of supported palladium nanoparticle catalysts in the decarboxylation of fatty acids. Appl. Catal. A 2011, 396, 85–90. [Google Scholar] [CrossRef]
- Madsen, A.T.; Rozmysłowicz, B.; Simakova, I.L.; Kilpïo, T.; Leino, A.R.; Kordás, K.; Eränen, K.; Mäki-Arvela, P.; Murzin, D.Y. Step changes and deactivation behaviour in the continuous decarboxylation of stearic acid. Ind. Eng. Chem. Res. 2011, 50, 11049–11058. [Google Scholar] [CrossRef]
- Madsen, A.T.; Rozmysłowicz, B.B.; Mäki-Arvela, P.; Simakova, I.L.; Eränen, K.; Murzin, D.Y.; Fehrmann, R. Deactivation in continuous deoxygenation of C18 fatty feedstock over Pd/Subunit. Top. Catal. 2013, 56, 714–724. [Google Scholar] [CrossRef]
- Simakova, I.; Simakova, O.; Mäki-Arvela, P.; Simakov, A.; Estrada, M.; Murzin, D.Y. Deoxygenation of palmitic and stearic acid over supported Pd catalysts: Effect of metal dispersion. Appl. Catal. A 2009, 355, 100–108. [Google Scholar] [CrossRef]
- Mäki-Arvela, P.; Kubickova, I.; Snåre, M.; Eränen, K.; Murzin, D.Y. Deoxygenation of fatty acids and their derivatives. Energy Fuels 2007, 21, 30–41. [Google Scholar] [CrossRef]
- Lestari, S.; Mäki-Arvela, P.; Bernas, H.; Simakova, O.; Sjöholm, R.; Beltramini, J.; Max Lu, G.Q.; Myllyoja, J.; Simakova, I.; Murzin, D.Y. Catalytic deoxygenation of stearic acid in a continuous reactor over a mesoporous carbon supported Pd catalyst. Energy Fuels 2009, 23, 3842–3845. [Google Scholar] [CrossRef]
- Fu, J.; Lu, X.; Savage, P.E. Catalytic hydrothermal deoxygenation of palmitic acid. Energy Environ. Sci. 2010, 3, 311–317. [Google Scholar] [CrossRef]
- Ford, J.P.; Immer, J.G.; Lamb, H.H. Palladium catalysts for fatty acid deoxygenation: Influence of the support and fatty acid chain length on decarboxylation kinetics. Top. Catal. 2012, 55, 175–184. [Google Scholar] [CrossRef]
- Kubička, D.; Bejblová, M.; Vlk, J. Conversion of vegetable oils into hydrocarbons over CoMo/MCM-41 catalysts. Top. Catal. 2010, 53, 168–178. [Google Scholar] [CrossRef]
- Lestari, S.; Mäki-Arvela, P.; Eränen, K.; Beltramini, J.; Max Lu, C.Q.; Murzin, D.Y. Diesel-like hydrocarbons from catalytic deoxygenation of stearic acid over supported Pd nanoparticles on SBA-15 catalysts. Catal. Lett. 2010, 134, 250–257. [Google Scholar] [CrossRef]
- Duan, J.; Han, J.; Sun, H.; Chen, P.; Lou, H.; Zheng, X. Diesel-like hydrocarbons obtained by direct hydrodeoxygenation of sunflower oil over Pd/Al-SBA-15 catalysts. Catal. Commun. 2012, 17, 76–80. [Google Scholar] [CrossRef]
- Peng, B.; Yao, Y.; Zhao, C.; Lercher, J.A. Upgrading pyrolysis oil over Ni/HZSM-5 by cascade reactions. Angew. Chem. Int. Ed. 2012, 124, 2114–2117. [Google Scholar] [CrossRef]
- Murata, K.; Liu, Y.; Inaba, M.; Takahara, I. Production of synthetic diesel by hydrotreatment of jatropha oils using Pt-Re/HZSM-5 catalyst. Energy Fuels 2010, 24, 2404–2409. [Google Scholar] [CrossRef]
- Serrano, D.P.; Escola, J.M.; Briones, L.; Arroyo, M. Selective hydrodecarboxylation of fatty acids into long chain hydrocarbons catalyzed by Pd/Al-SBA-15. Microporous Mesoporous Mater. 2019, 280, 88–96. [Google Scholar] [CrossRef]
- Li, J.; Li, X.; Zhou, G.; Wang, W.; Wang, C.; Komarneni, S.; Wang, Y. Catalytic fast pyrolysis of biomass with mesoporous ZSM-5 zeolites prepared by desilication with NaOH solutions. Appl. Catal. A 2014, 470, 115–122. [Google Scholar] [CrossRef]
- Botas, J.A.; Serrano, D.P.; García, A.; Ramos, R. Catalytic conversion of rapeseed oil for the production of raw chemicals, fuels and carbon nanotubes over Ni-modified nanocrystalline and hierarchical ZSM-5. Appl. Catal. B 2014, 145, 205–215. [Google Scholar] [CrossRef]
- Serrano, D.P.; Aguado, J.; Escola, J.M.; Rodríguez, J.M.; Peral, A. Hierarchical zeolites with enhanced textural and catalytic properties synthesized from organofunctionalized seeds. Chem. Mater. 2006, 18, 2462–2464. [Google Scholar] [CrossRef]
- Serrano, D.P.; Escola, J.M.; Briones, L.; Arroyo, M. Hydroprocessing of the LDPE thermal cracking oil into transportation fuels over Pd supported on hierarchical ZSM-5 catalyst. Fuel 2017, 206, 190–198. [Google Scholar] [CrossRef]
- Serrano, D.P.; Pinnavaia, T.J.; Aguado, J.; Escola, J.M.; Peral, A.; Villalba, L. Hierarchical ZSM-5 zeolites synthesized by silanization of protozeolitic units: Mediating the mesoporosity contribution by changing the organosilane type. Catal. Today 2014, 227, 15–25. [Google Scholar] [CrossRef]
- Wang, C.; Xu, J.; Qi, G.; Gong, Y.; Wang, W.; Gao, P.; Wang, Q.; Feng, N.; Liu, X.; Deng, F. Methylbenzene hydrocarbon pool in methanol-to-olefins conversion over zeolite H-ZSM-5. J. Catal. 2015, 332, 127–137. [Google Scholar] [CrossRef]
- Chung, Y.-M.; Mores, D.; Weckhuysen, B.M. Spatial and temporal mapping of coke formation during paraffin and olefin aromatization in individual H-ZSM-5 crystals. Appl. Catal. A 2011, 404, 12–20. [Google Scholar] [CrossRef]
- Dai, S.; Zhou, S.; Zhang, W.; Chen, C. Systematic investigation of ligand steric effects on α-Diimine Palladium catalyzed olefin polymerization and copolymerization. Macromolecules 2016, 49, 8855–8862. [Google Scholar] [CrossRef]
- Netalkar, S.P.; Budagumpi, S.; Abdallah, H.H.; Netalkar, P.P.; Revankar, V.K. Sterically modulated binuclear bis α-diimine Pd(II) complexes: Synthesis, characterization, DFT studies and catalytic behaviour towards ethylene oligomerization. J. Mol. Struct. 2014, 1075, 559–565. [Google Scholar] [CrossRef]
- Karatun, O.N.; Dorogochinskii, A.Z. Oligomerization and aromatization of propylene. Pd-containing catalysts on high-silica zeolite. Chem. Technol. Fuels Oils 2000, 36, 416–420. [Google Scholar] [CrossRef]
- Wang, J.; Wang, L.; Yu, H.; Ullah, R.S.; Haroon, M.; Zain-ul-Abdin, X.; Khan, R.U. Recent progress in ethylene polymerization catalysed by Ni and Pd catalysts. Eur. J. Inorg. Chem. 2018, 1450–1468. [Google Scholar] [CrossRef]
- Puthiaraj, P.; Kim, K.; Ahn, W.S. Catalytic transfer hydrogenation of bio-based furfural by palladium supported on nitrogen-doped porous carbon. Catal. Today 2019, 49–58. [Google Scholar] [CrossRef]
- Alam, M.I.; Khan, T.S.; Haider, M.A. Alternate biobased route to produce I-decalactone: Elucidating the role of solvent and hydrogen evolution in catalytic transfer hydrogenation. ACS Sustain. Chem. Eng. 2019, 7, 2894–2898. [Google Scholar] [CrossRef]
- Shafaghat, H.; Rezaei, P.S.; Daud, W.M.A.W. Using decalin and tetralin as hydrogen source for transfer hydrogenation of renewable lignin-derived phenolics over activated carbon supported Pd and Pt catalysts. J. Taiwan Inst. Chem. Eng. 2016, 65, 91–100. [Google Scholar] [CrossRef]
- Muzart, J. Pd-catalyzed hydrogen transfer reactions from alcohols to C=C, C=O and C=N bonds. Eur. J. Org. Chem. 2015, 26, 5693–5707. [Google Scholar] [CrossRef]

| Sample | Pd/n-ZSM-5 (32) | Pd/h-ZSM-5 (33) | Pd/h-ZSM-5 (51) | Pd/h-ZSM-5 (122) |
|---|---|---|---|---|
| Si/Al Molar Ratio | 32 | 33 | 51 | 122 |
| Pd Content (wt %) | 0.84 | 0.96 | 0.91 | 0.94 |
| Pd Mean Size (nm) | 23 | 13 | 14 | 17 |
| BET Surface Area (m2 g−1) | 377 | 477 | 479 | 486 |
| Total Pores Volume (cm3 g−1) | 0.434 | 0.497 | 0.471 | 0.557 |
| Micropores Volume (cm3 g−1) | 0.171 | 0.130 | 0.157 | 0.165 |
| Micropore Surface Area (m2/g) | 274 | 209 | 252 | 265 |
| External/Mesopores Surface Area (m2/g) | 103 | 268 | 227 | 221 |
| Tmax NH3 Desorption (°C) | 330 | 340 | 340 | 332 |
| Acidity (meq NH3 g−1) | 0.345 | 0.305 | 0.242 | 0.122 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arroyo, M.; Briones, L.; Escola, J.M.; Serrano, D.P. Conversion of Stearic Acid into Bio-Gasoline over Pd/ZSM-5 Catalysts with Enhanced Accessibility. Appl. Sci. 2019, 9, 2386. https://doi.org/10.3390/app9112386
Arroyo M, Briones L, Escola JM, Serrano DP. Conversion of Stearic Acid into Bio-Gasoline over Pd/ZSM-5 Catalysts with Enhanced Accessibility. Applied Sciences. 2019; 9(11):2386. https://doi.org/10.3390/app9112386
Chicago/Turabian StyleArroyo, Marta, Laura Briones, José María Escola, and David P. Serrano. 2019. "Conversion of Stearic Acid into Bio-Gasoline over Pd/ZSM-5 Catalysts with Enhanced Accessibility" Applied Sciences 9, no. 11: 2386. https://doi.org/10.3390/app9112386
APA StyleArroyo, M., Briones, L., Escola, J. M., & Serrano, D. P. (2019). Conversion of Stearic Acid into Bio-Gasoline over Pd/ZSM-5 Catalysts with Enhanced Accessibility. Applied Sciences, 9(11), 2386. https://doi.org/10.3390/app9112386




