Fermentation Characteristics of Lactobacillus Plantarum and Pediococcus Species Isolated from Sweet Sorghum Silage and Their Application as Silage Inoculants
Abstract
:Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Procedures
2.1.1. LAB Strains Isolation and Identification
2.1.2. Morphological, Physiological and Biochemical Tests
2.1.3. Extraction of Bacterial Deoxyribonucleic acid
2.1.4. LAB Identification by 16S rRNA Sequencing Analysis
2.2. Silage Making
Chemical and Microbial Analysis of Fresh Grasses and Silage
2.3. Data Analysis
3. Results
3.1. Characteristics of LAB Isolates
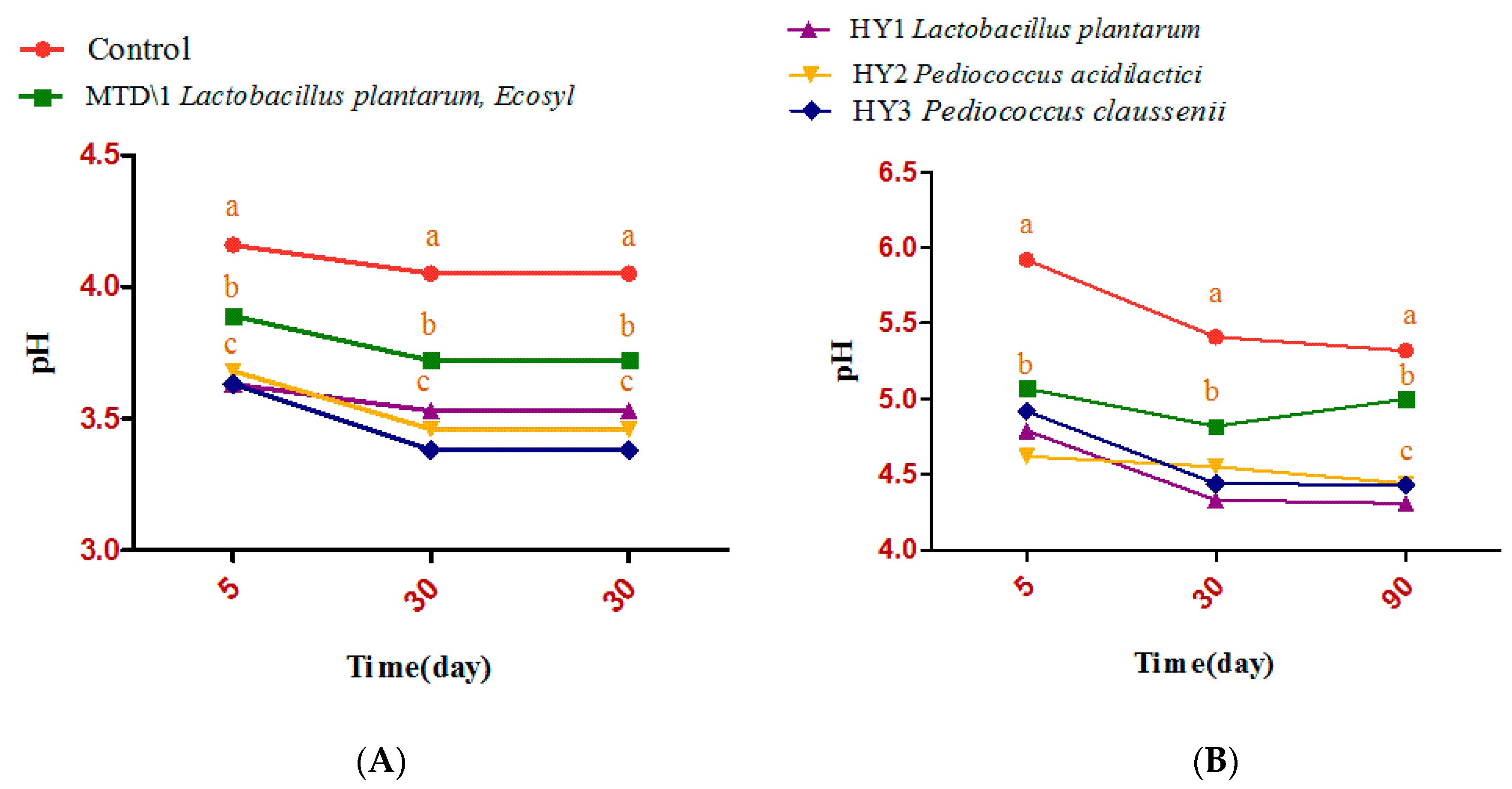
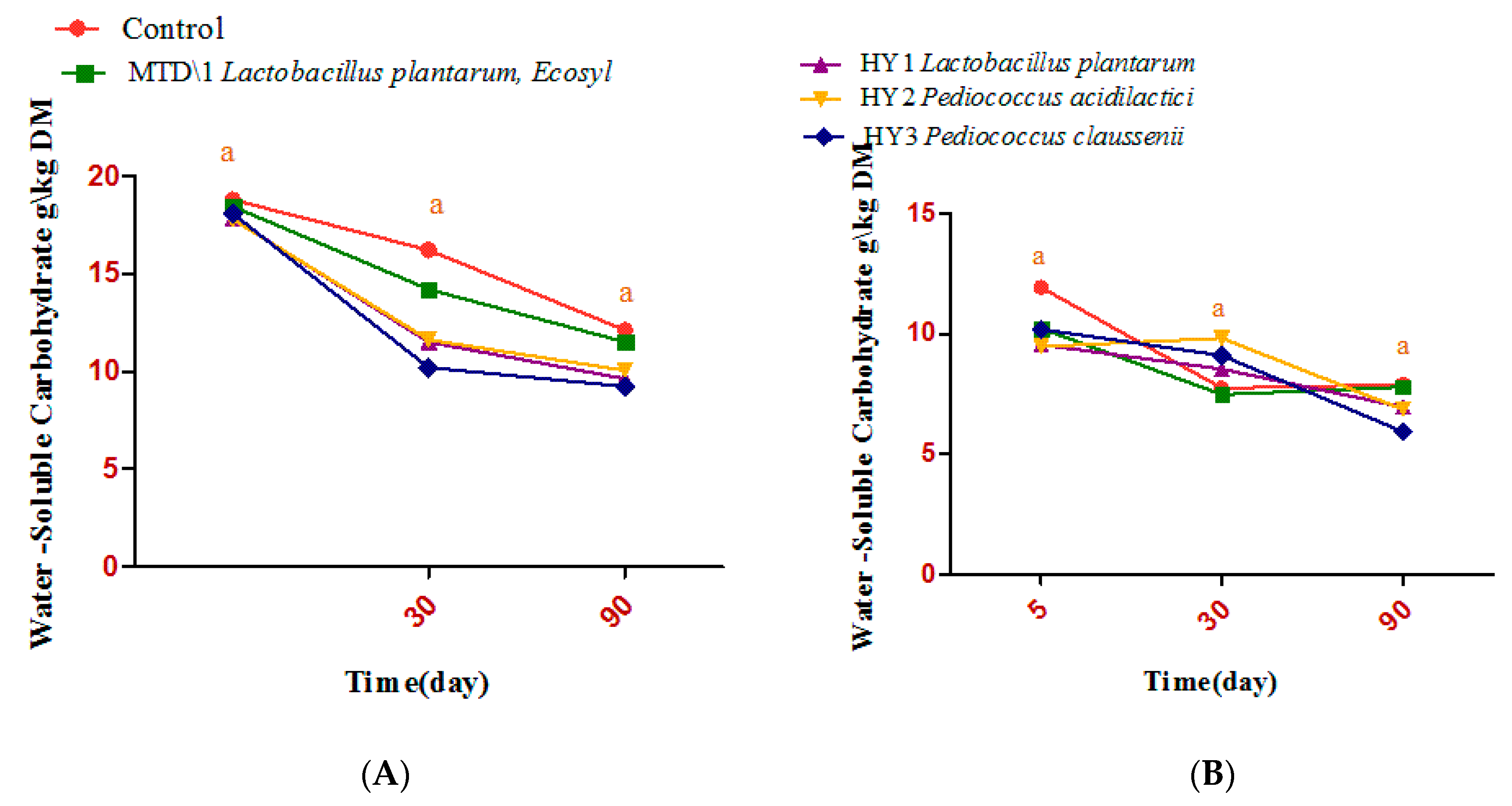
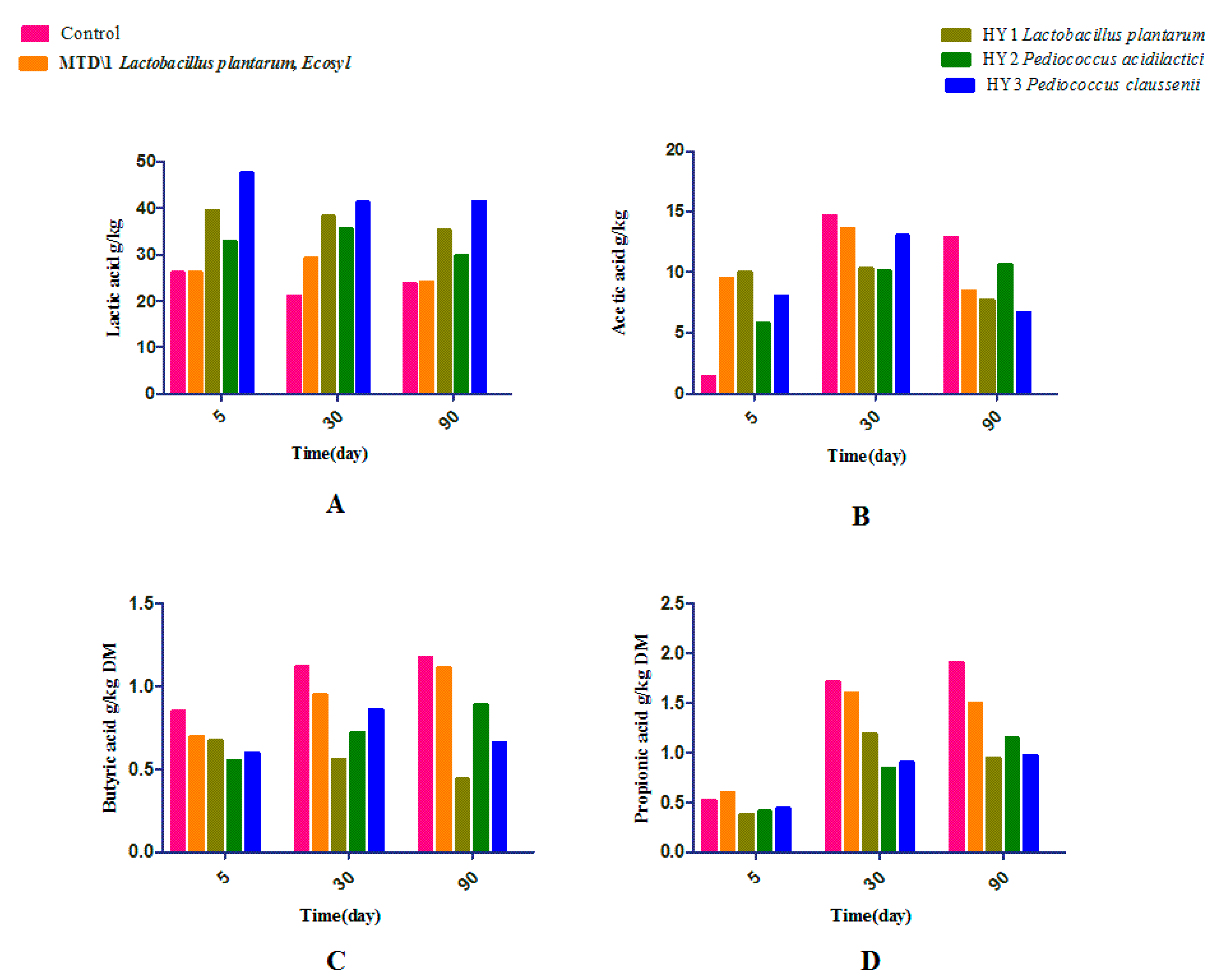
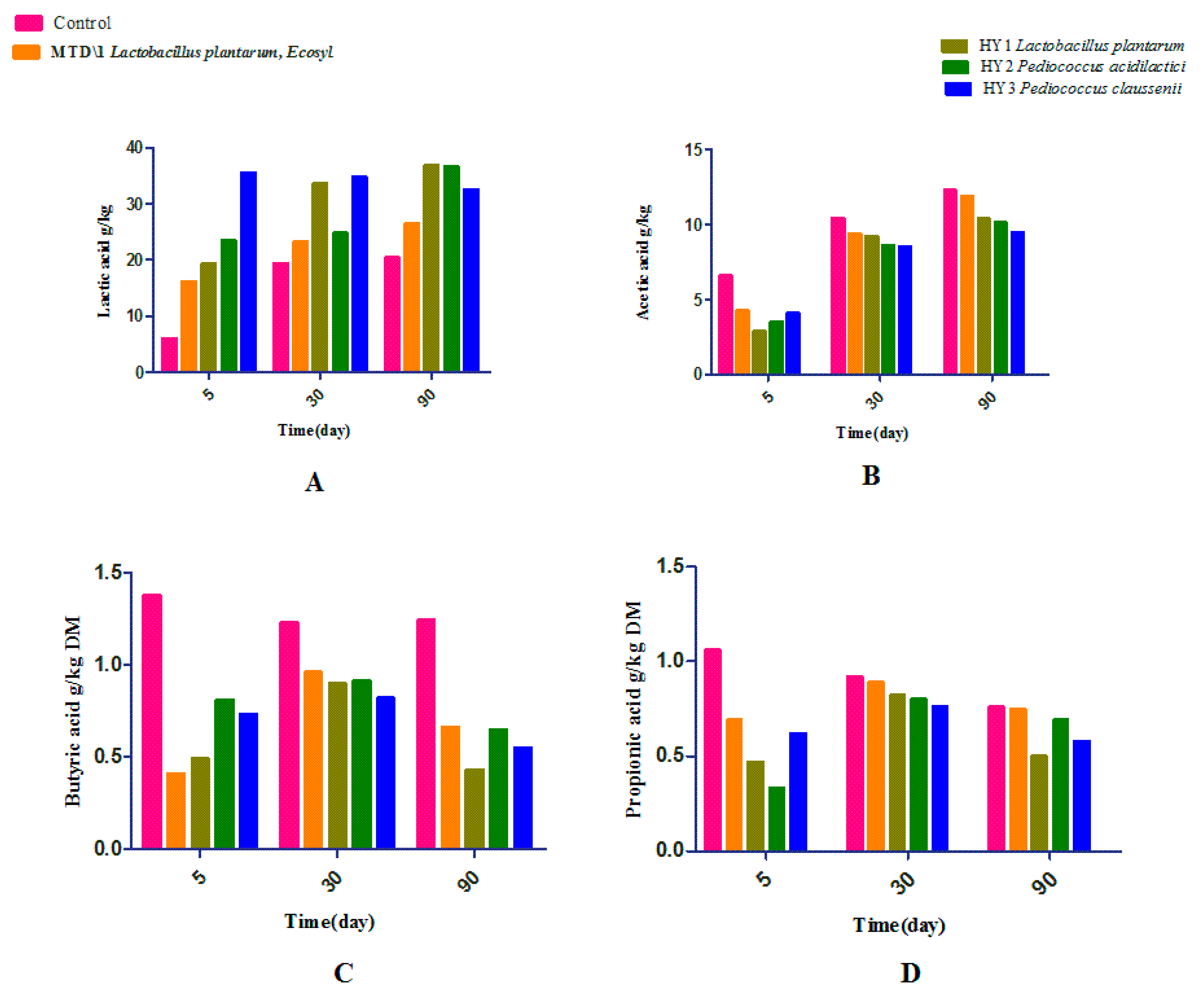
3.2. Fermentation Quality of Sweet Sorghum and Napier Grass
3.3. Microbial Profile of Sweet Sorghum and Napier Grass Silage
3.4. Statistical AnalysisFresh and End Product of Silage
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Titterton, M.; Bareeba, F. Grass and Legume Silages in the Tropics. In Proceedings of the FAO electronic conference on tropical silage; Silage making in the tropics with particular emphasis on smallholders, Rome, Italy, 1 September–15 December 1999. [Google Scholar]
- Cai, Y. Identification and characterization of enterococcus species isolated from forage crops and their influence on silage fermentation. J. Dairy Sci. 1999, 82, 2466–2471. [Google Scholar] [CrossRef]
- MacDonald, P.; Henderson, A.; Heron, S. Chapter 2: Crops for silage. In The Biochemistry of Silage; Chalcombe Publications: Southampton, UK, 1991. [Google Scholar]
- Mohd-Setapar, S.; Abd-Talib, N.; Aziz, R. Review on crucial parameters of silage quality. APCBEE Procedia 2012, 3, 99–103. [Google Scholar] [CrossRef]
- Li, L.; Sun, Y.; Yuan, Z.; Kong, X.; Wao, Y.; Yang, L.; Zhang, Y.; Li, D. Effect of microalgae supplementation on the silage quality and anaerobic digestion performance of many flower silvergrass. Bioresour. Technol. 2015, 189, 334–340. [Google Scholar] [CrossRef]
- Saarisalo, E.; Skyttä, E.; Haikara, A.; Jalava, T.; Jaakkola, S. Screening and selection of lactic acid bacteria strains suitable for ensiling grass. J. Appl. Microbiol. 2007, 102, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.; Pauly, T.; Udén, P. Storage of small bale silage and haylage–influence of storage period on fermentation variables and microbial composition. Grass Forage Sci. 2007, 62, 274–283. [Google Scholar] [CrossRef]
- Latorre, A.; Dagnac, T.; Lorenzo, B.F.; Llompart, M. Occurrence and stability of masked fumonisins in corn silage samples. Food Chem. 2015, 189, 38–44. [Google Scholar] [CrossRef]
- Alonso, V.A.; Pereyra, C.M.; Keller, L.A.M.; Dalcero, A.M.; Rosa, C.; Chiacchiera, S.M.; Cavaglieri, L.R. Fungi and mycotoxins in silage: An overview. J. Appl. Microbiol. 2013, 115, 637–643. [Google Scholar]
- Fijałkowska, M.; Przemieniecki, S.W.; Kurowski, T.; Lipiński, K.; Nogalski, Z.; Purwin, C. Ensiling suitability and microbiological quality of virginia fanpetals biomass. Can. J. Anim. Sci. 2017, 97, 541–544. [Google Scholar] [CrossRef]
- Przemieniecki, S.W.; Damszel, M.; Kurowski, T.P.; Mastalerz, J.; Kotlarz, K. Identification, ecological evaluation and phylogenetic analysis of non-symbiotic endophytic fungi colonizing timothy grass and perennial ryegrass grown in adjacent plots. Grass Forage Sci. 2019. [Google Scholar] [CrossRef]
- Kizilşimşek, M.; Küsek, M.; Gezginc, Y.; Erol, A. Isolation and identification of high lactic acid producer bacteria from forage and their silages grown in different ecologies. Kafkas Univ. Vet. Fak. Derg. 2016, 22, 291–296. [Google Scholar]
- Salvucci, E.; LeBlanc, J.G.; Pérez, G. Technological properties of lactic acid bacteria isolated from raw cereal material. LWT Food Sci. Technol. 2016, 70, 185–191. [Google Scholar] [CrossRef]
- Dalheim, M.O.; Arnfinnsdottir, N.B.; Widmalm, G.; Christensen, B.E. The size and shape of three water-soluble, non-ionic polysaccharides produced by lactic acid bacteria: A comparative study. Carbohydr. Polym. 2016, 142, 91–97. [Google Scholar] [PubMed]
- Anadón, A.; Martínez-Larrañaga, M.R.; Martínez, M.A. Probiotics for animal nutrition in the european union. Regulation and safety assessment. Regul. Toxicol. Pharmacol. 2006, 45, 91–95. [Google Scholar] [PubMed]
- Datta, R.; Henry, M. Lactic acid: Recent advances in products, processes and technologies—A review. J. Chem. Technol. Biotechnol. 2006, 81, 1119–1129. [Google Scholar] [CrossRef]
- Filya, I. The effect of lactobacillus buchneri and lactobacillus plantarum on the fermentation, aerobic stability, and ruminal degradability of low dry matter corn and sorghum silages. J. Dairy Sci. 2003, 86, 3575–3581. [Google Scholar] [CrossRef]
- Kasra-Kermanshahi, R.; Fooladi, J.; Peymanfar, S. Isolation and microencapsulation of lactobacillus spp. From corn silage for probiotic application. Iran. J. Microbiol. 2010, 2, 98. [Google Scholar]
- Marti, E.; Variatza, E.; Balcazar, J.L. The role of aquatic ecosystems as reservoirs of antibiotic resistance. Trends Microbiol. 2014, 22, 36–41. [Google Scholar] [CrossRef]
- Van Loveren, H.; Sanz, Y.; Salminen, S. Health claims in europe: Probiotics and prebiotics as case examples. Annu. Rev. Food Sci. Technol. 2012, 3, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Porto, M.C.W.; Kuniyoshi, T.M.; de Azevedo, P.O.D.S.; Vitolo, M.; de Souza Oliveira, R.P. Pediococcus spp.: An important genus of lactic acid bacteria and pediocin producers. Biotechnol. Adv. 2017, 35, 361–374. [Google Scholar] [CrossRef] [PubMed]
- Fitzsimons, A.; Duffner, F.; Curtin, D.; Brophy, G.; O’kiely, P.; O’connell, M. Assessment of pediococcus acidilactici as a potential silage inoculant. Appl. Environ. Microbiol. 1992, 58, 3047–3052. [Google Scholar]
- Moran, J.; O’kiely, P.; Wilson, R.; Crombie-Quilty, M. Enumeration of lactic acid bacteria on grass and the effects of added bacteria on silage fermentation. Ir. Grassl. Anim. Prod. Assoc. J. 1990, 24, 46–55. [Google Scholar]
- Pahlow, G.; Muck, R.E.; Driehuis, F.; Elferink, S.; Spoelstra, S.F. Microbiology of ensiling. Agronomy 2003, 42, 31–94. [Google Scholar]
- Thonart, P.; Dortu, C. Les bactériocines des bactéries lactiques: Caractéristiques et intérêts pour la bioconservation des produits alimentaires. Biotechnol. Agron. Soc. Environ. 2009, 13, 143–154. [Google Scholar]
- Kozaki, M.; Uchimura, T.; Okada, S. Experimental Manual of Lactic Acid Bacteria; Asakurasyoten: Tokyo, Japan, 1992; pp. 34–37. [Google Scholar]
- Cheng, H.-R.; Jiang, N. Extremely rapid extraction of DNA from bacteria and yeasts. Biotechnol. Lett. 2006, 28, 55–59. [Google Scholar] [CrossRef]
- Lane, D. 16s/23s rrna sequencing. In Nucleic Acid Techniques in Bacterial Systematics; John Wiley & Sons: New York, NY, USA, 1991; pp. 115–175. [Google Scholar]
- Shehata, M.; El Sohaimy, S.; El-Sahn, M.A.; Youssef, M. Screening of isolated potential probiotic lactic acid bacteria for cholesterol lowering property and bile salt hydrolase activity. Ann. Agric. Sci. 2016, 61, 65–75. [Google Scholar] [CrossRef]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped blast and psi-blast: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef]
- Liu, Q.; Shao, T.; Bai, Y. The effect of fibrolytic enzyme, lactobacillus plantarum and two food antioxidants on the fermentation quality, alpha-tocopherol and beta-carotene of high moisture napier grass silage ensiled at different temperatures. Anim. Feed Sci. Technol. 2016, 221, 1–11. [Google Scholar] [CrossRef]
- Murphy, R. A method for the extraction of plant samples and the determination of total soluble carbohydrates. J. Sci. Food Agric. 1958, 9, 714–717. [Google Scholar] [CrossRef]
- Playne, M.; McDonald, P. The buffering constituents of herbage and of silage. J. Sci. Food Agric. 1966, 17, 264–268. [Google Scholar] [CrossRef]
- Lima, R.; Díaz, R.; Castro, A.; Hoedtke, S.; Fievez, V. Multifactorial models to assess responses to sorghum proportion, molasses and bacterial inoculant on in vitro quality of sorghum–soybean silages. Anim. Feed Sci. Technol. 2011, 164, 161–173. [Google Scholar] [CrossRef]
- Pahlow, G.; Rammer, C.; Slottner, D.; Tuori, M. Ensiling of legumes. Landbauforsch. Voelkenrode 2002, 234, 27–31. [Google Scholar]
- Herrmann, C.; Heiermann, M.; Idler, C. Effects of ensiling, silage additives and storage period on methane formation of biogas crops. Bioresour. Technol. 2011, 102, 5153–5161. [Google Scholar] [CrossRef] [PubMed]
- Heinritz, S. Ensiling Suitability of High Protein Tropical Forages and Their Nutritional Value for Feeding Pigs. Thesis (Agribiology), University of Hohenheim, Stuttgart, Germany, University of Rostock, Rostock, Germany, 2011. [Google Scholar]
- Lemus, R. Understanding Silage Making Process and Utilization; Cooperative Extension Service, Mississippi State University: Starkville, MS, USA, 2010; Volume 3. [Google Scholar]
- Podkówka, Z.; Podkówka, L. Chemical composition and quality of sweet sorghum and maize silages. J. Cent. Eur. Agric. 2011, 12. [Google Scholar] [CrossRef]
- Joo, Y.H.; Kim, D.H.; Paradhipta, D.H.; Lee, H.J.; Amanullah, S.M.; Kim, S.B.; Chang, J.S.; Kim, S.C. Effect of microbial inoculants on fermentation quality and aerobic stability of sweet potato vine silage. Asian-Australas. J. Anim. Sci. 2018, 31, 1897. [Google Scholar] [CrossRef]
- Makoni, N.; Broderick, G.; Muck, R. Effect of modified atmospheres on proteolysis and fermentation of ensiled alfalfa1. J. Dairy Sci. 1997, 80, 912–920. [Google Scholar] [CrossRef]
- Reyes-Gutiérrez, J.A.; Montañez-Valdez, O.D.; Rodríguez-Macias, R.; Ruíz-López, M.; Salcedo-Pérez, E.; Guerra-Medina, C.E. Effect of a bacterial inoculum and additive on dry matter in situ degradability of sugarcane silage. J. Integr. Agric. 2015, 14, 497–502. [Google Scholar]
- Oladosu, Y.; Rafii, M.Y.; Abdullah, N.; Magaji, U.; Hussin, G.; Ramli, A.; Miah, G. Fermentation quality and additives: A case of rice straw silage. BioMed Res. Int. 2016, 2016, 7985167. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Guo, G.; Wen, A.; Desta, S.T.; Wang, J.; Wang, Y.; Shao, T. The effect of different additives on the fermentation quality, in vitro digestibility and aerobic stability of a total mixed ration silage. Anim. Feed Sci. Technol. 2015, 207, 41–50. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, X.; Zhao, M.; Yu, Z. Isolating and evaluating lactic acid bacteria strains for effectiveness of leymus chinensis silage fermentation. Lett. Appl. Microbiol. 2014, 59, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.J.; Chaudhry, A.S.; Osman, A.; Shi, C.Q.; Edwards, G.R.; Dewhurst, R.J.; Cheng, L. Associative effects of ensiling mixtures of sweet sorghum and alfalfa on nutritive value, fermentation and methane characteristics. Anim. Feed Sci. Technol. 2015, 206, 29–38. [Google Scholar] [CrossRef]
- Schmidt, J.; Sipocz, J.; Kaszas, I.; Szakacs, G.; Gyepes, A.; Tengerdy, R. Preservation of sugar content in ensiled sweet sorghum. Bioresour. Technol. 1997, 60, 9–13. [Google Scholar]
- Driehuis, F.; van Wikselaar, P.G. The occurrence and prevention of ethanol fermentation in high-dry-matter grass silage. J. Sci. Food Agric. 2000, 80, 711–718. [Google Scholar]
- Charmley, E. Towards improved silage quality—A review. Can. J. Anim. Sci. 2001, 81, 157–168. [Google Scholar]
- Pruckler, M.; Lorenz, C.; Endo, A.; Kraler, M.; Durrschmid, K.; Hendriks, K.; Soares da Silva, F.; Auterith, E.; Kneifel, W.; Michlmayr, H. Comparison of homo- and heterofermentative lactic acid bacteria for implementation of fermented wheat bran in bread. Food Microbiol. 2015, 49, 211–219. [Google Scholar] [PubMed]
- Knicky, M. Possibilities to Improve Silage Conservation. Doctoral Thesis, Swedish University of Agricultural Sciences, Uppsala, Sweden, 2005. [Google Scholar]
- Acosta, Y. The use of probiotic strains as silage inoculants. In Probiotic in Animals; InTech: Rio de Janeiro, Brazil, 2012. [Google Scholar]
- Cai, Y.; Benno, Y.; Ogawa, M.; Kumai, S. Effect of applying lactic acid bacteria isolated from forage crops on fermentation characteristics and aerobic deterioration of silage. J. Dairy Sci. 1999, 82, 520–526. [Google Scholar] [CrossRef]
- Jansson, S. Lactic Acid Bacteria in Silage: Growth, Antibacterial Activity and Antibiotic Resistance. Ph.D. Thesis, Sveriges Lantbruksuniversitet, Uppsala, Sweden, 2005. [Google Scholar]
- Nkosi, B.; Vadlani, P.; Brijwani, K.; Nanjunda, A.; Meeske, R. Effects of bacterial inoculants and an enzyme on the fermentation quality and aerobic stability of ensiled whole-crop sweet sorghum. S. Afr. J. Anim. Sci. 2012, 42, 232–240. [Google Scholar]
- Te Giffel, M.T.; Wagendorp, A.; Herrewegh, A.; Driehuis, F. Bacterial spores in silage and raw milk. Antonie Van Leeuwenhoek 2002, 81, 625–630. [Google Scholar] [CrossRef] [PubMed]

| Items | Mean ± Standard Deviation | |
|---|---|---|
| Sweet Sorghum | Napier Grass | |
| Dry matter (DM)g/kg | 225 ± 4.56 | 241 ± 0.56 |
| pH | 5.76 ± 0.02 | 5.97± 0.05 |
| water soluble carbohydrates (WSC) g/kg DM | 30.2 | 25 |
| buffering capacity(BC) mEq/kg DM | 260 | 94.4 |
| WSC/BC ratio | 0.11 | 0.26 |
| Neutral detergent fibre (g/kg DM) | 642 ± 0.63 | 697 ± 0.25 |
| Acid detergent fibre (g/kg DM) | 398 ± 0.61 | 475 ± 6.61 |
| Crude protein (g/kg DM) | 80.7 ± 0.07 | 62.5± 0.01 |
| Lactic acid bacteria (log10 cfu/g) | 6.94 ± 0.07 | 5.20 ± 0.20 |
| Aerobic bacteria (log10 cfu/g) | ND | 4.41± 0.05 |
| yeast (log10 cfu/g) | 2.11± 0.11 | 2.15± 1.11 |
| Characteristics | HY1 | HY2 | HY3 |
|---|---|---|---|
| Shape | Coccus | Rod | Rod |
| Gram stain | + | + | + |
| Gas from glucose | + | - | + |
| catalase | - | - | - |
| Fermentation type | Homo | Homo | Homo |
| Acidification | 3.82 | 3.95 | 3.91 |
| Growth at pH | |||
| 3 | + | + | + |
| 3.5 | + | + | + |
| 4.5 | + | + | + |
| 5.5 | + | + | + |
| 6.5 | + | + | + |
| 7.5 | + | + | + |
| Growth at temperatures(°C) | |||
| 5 | - | - | - |
| 10 | - | - | - |
| 15 | - | - | - |
| 40 | + | + | + |
| 45 | + | + | + |
| 50 | + | - | - |
| Growth in NaCl (w/v %) | |||
| 3.50% | + | + | + |
| 6.50% | + | + | + |
| 9.50% | - | - | - |
| 16Sr DNA similarity (%) | 95% Lactobacillus plantarum | 96% Pediococcus acidilactici | 99% Pediococcusclaussenii |
| D-Glucose | + | + | + |
| D-Fructose | + | + | + |
| D-Xylose | + | + | + |
| Melezitose | + | + | + |
| Amygdalin | + | + | + |
| Melibiose | - | w | + |
| Ribose | + | + | + |
| Sorbose | w | - | + |
| L-Arabinose | + | + | + |
| Galactose | + | + | + |
| Rhamnose | + | - | + |
| Lactose | + | + | + |
| Mannose | + | + | + |
| Saccharose | + | + | + |
| Sorbitol | + | + | + |
| Salicine | + | + | + |
| Mannitol | + | + | + |
| Maltose | - | - | + |
| Inositol | + | + | - |
| Esculine | + | + | + |
| Parameter | Treatments | Days | ESM | Significance | ||||
|---|---|---|---|---|---|---|---|---|
| 5 | 30 | 90 | Day | Treat | Day *Treat | |||
| DM g/kg | C | 231Aa | 231Aa | 210Aa | 1.63 | 0.178 | 0.028 | 0.379 |
| MTD/1 | 225Aa | 230Aa | 217Aa | |||||
| HY1 | 224Aa | 227Aa | 229Aa | |||||
| HY2 | 228Aa | 235Aa | 228Aa | |||||
| HY3 | 215Aa | 213Aa | 218Aa | |||||
| Ammonia-N g/kg DM | C | 45.5AB | 61.2Aba | 77.0Aa | 2.31 | <0.0001 | 0.0004 | 0.124 |
| MTD/1 | 40.4BAa | 54.3Aa | 69.7BAa | |||||
| HY1 | 35.2BAc | 49.7BAb | 59.5BAa | |||||
| HY2 | 37.5BAa | 47.4Aa | 54.9BAa | |||||
| HY3 | 26.8Ba | 38.3 Ba | 60.1Ba | |||||
| Lactic acid bacteria (log10 cfu/g) | C | 3.39Aa | 5.93ABa | ND | 0.46 | <0.0001 | 0.093 | <0.0001 |
| MTD/1 | 4.86Aa | ND | ND | |||||
| HY1 | 6.74Aa | 6.25Aa | 5.60Aa | |||||
| HY2 | 2.08Aa | 3.66ABa | 0.33Bb | |||||
| HY3 | 7.65Aa | 2.19ABb | 1.86BAb | |||||
| Aerobic bacteria (log10 cfu/g) | C | 3.39Aa | 5.36Aa | 3.49Aa | 0.37 | 0.041 | 0.549 | 0.007 |
| MTD/1 | 4.86Aa | ND | 2.66Aa | |||||
| HY1 | ND | 2.89ABa | 1.33Aa | |||||
| HY2 | 2.08Aa | 5.14Aa | 1.33Aa | |||||
| HY3 | 1.65Aa | 5.21Aa | 1.33Aa | |||||
| yeast (log10 cfu/g) | C | 3.70Aa | 1.00Aba | 0.33Ab | 0.56 | 0.3 | 0.235 | 0.056 |
| MTD/1 | 1.15ABa | ND | ND | |||||
| HY1 | ND | ND | ND | |||||
| HY2 | 1.10ABa | ND | ND | |||||
| HY3 | ND | ND | ND | |||||
| Parameter | Treatments | Days | ESM | Significance | ||||
|---|---|---|---|---|---|---|---|---|
| 5 | 30 | 90 | Day | Treat | Day *Treat | |||
| DM g/kg | C | 289Aa | 280Aa | 257Aa | 3.73 | 0.346 | 0.01 | 0.254 |
| MTD/1 | 288Aa | 263Aa | 269Aa | |||||
| HY1 | 264Aa | 281Aa | 252Aa | |||||
| HY2 | 286Aa | 299Aa | 316Aa | |||||
| HY3 | 305Aa | 293Aa | 281Aa | |||||
| Ammonia-N g/kg DM | C | 30.2ABb | 48.9Aba | 89.9Aa | 4.58 | <0.0001 | 0.0001 | 0.0041 |
| MTD/1 | 35.3Ab | 50.2BAa | 63.9Aa | |||||
| HY1 | 33.1ABb | 46.9BAb | 60.2Ba | |||||
| HY2 | 30.1Ab | 57.3Aa | 63.9Ba | |||||
| HY3 | 23.3Bb | 33.2Bba | 54.3Ba | |||||
| Lactic acid bacteria (log10 cfu/g) | C | 3.90Aa | 6.08Ab | 6.35Aba | 0.22 | <0.0001 | 0.128 | 0.12 |
| MTD/1 | 3.99Ab | 3.05Ab | 6.03Aa | |||||
| HY1 | 6.52Aa | 5.52Aa | 6.39Aa | |||||
| HY2 | 6.56Aa | 6.34Aa | 6.35Aa | |||||
| HY3 | 6.39Aa | 3.58Aa | 6.18Aa | |||||
| Aerobic bacteria (log10 cfu/g) | C | 3.27Aa | 5.31Aa | 4.88Aa | 0.28 | 0.153 | 0.01 | 0.175 |
| MTD/1 | 4.08Aa | 2.30Aa | 5.15Aa | |||||
| HY1 | 5.47Aa | 4.19Aa | 4.83Aa | |||||
| HY2 | 5.46Aa | 3.14Aa | 1.66Aa | |||||
| HY3 | 5.32Aa | 4.73Aa | 1.42Ab | |||||
| yeast (log10 cfu/g) | C | 4.72Aa | 4.66Aa | ND | 0.32 | <0.0001 | 0.007 | 0.004 |
| MTD/1 | 4.17BAa | 3.91ABa | ND | |||||
| HY1 | 4.07ABa | 2.97ABa | ND | |||||
| HY2 | 4.62Aa | 2.73ABb | ND | |||||
| HY3 | 3.90Ba | ND | ND | |||||
| Parameter | Treatments | Sweet Sorghum | Napier Grass | ||||
|---|---|---|---|---|---|---|---|
| Mean | p-Value | ESM | Mean | p-Value | ESM | ||
| Neutral detergent fibre g/kg DM | C | 61.1A | 3.24 | 0.961 | 62.7 A | 1.39 | 0.056 |
| MTD/1 | 60.4A | 62.8 A | |||||
| HY1 | 60.5 A | 63.2 A | |||||
| HY2 | 58.1 A | 62.6 A | |||||
| HY3 | 59.3 A | 62.0 A | |||||
| Acid detergent fibre g/kg DM | C | 35.9 A | 3.89 | 0.916 | 44.2 A | 1.41 | 0.628 |
| MTD/1 | 34.9 A | 41.8 A | |||||
| HY1 | 39.0 A | 41.4 A | |||||
| HY2 | 41.7 A | 41.7A | |||||
| HY3 | 41.6 A | 39.7 A | |||||
| Statistics | pH | WSC (g/kg DM) | LAB (log10 cfu/g) | Aerobic Bacteria (log10 cfu/g) | Yeas (log10 cfu/g)t and Mould | NDF (g/kg DM) | ADF (g/kg DM) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| p-values | SEM | p-values | SEM | p-values | SEM | p-values | SEM | p-values | SEM | p-values | SEM | p-values | SEM | |
| SS | <0.0001 | 0.38 | 0.0013 | 4.18 | 0.0453 | 1.3 | 0.0013 | 0.66 | 0.0013 | 0.08 | 0.0013 | 0.08 | 0.0013 | 0.08 |
| SS*MTD/1 | 0.0004 | 0.52 | <0.0001 | 4.75 | <0.0001 | 1.44 | <0.0001 | 0.98 | <0.0001 | 0.61 | <0.0001 | 0.61 | <0.0001 | 0.61 |
| SS*HY1 | 0.0004 | 0.52 | 0.0002 | 5.47 | <0.0001 | 1.44 | 0.0002 | 0.14 | 0.0002 | 0.61 | 0.0002 | 0.61 | 0.0002 | 0.61 |
| SS*HY2 | 0.0001 | 0.46 | 0.0003 | 5.03 | 0.05 | 1.31 | 0.0003 | 0.66 | 0.0003 | 0.57 | 0.0003 | 0.57 | 0.0003 | 0.57 |
| SS*HY3 | <0.0001 | 0.48 | 0.0005 | 4.94 | 0.117 | 1.36 | 0.0005 | 0.98 | 0.0005 | 0.24 | 0.0005 | 0.24 | 0.0005 | 0.24 |
| NS | 0.0005 | 0.13 | 0.0005 | 4.44 | 0.0463 | 0.21 | 0.0005 | 0.14 | 0.0005 | 0.68 | 0.0005 | 0.68 | 0.0005 | 0.68 |
| NS*MTD/1 | <0.0001 | 0.23 | 0.0005 | 4.05 | 0.900 | 0.15 | 0.0005 | 0.14 | 0.0005 | 0.70 | 0.0005 | 0.70 | 0.0005 | 0.70 |
| NS*HY1 | <0.0001 | 0.23 | 0.0004 | 4.50 | 0.900 | 0.15 | 0.0004 | 0.14 | 0.0004 | 0.70 | 0.0004 | 0.70 | 0.0004 | 0.70 |
| NS*HY2 | 0.0006 | 0.15 | 0.0003 | 4.80 | 0.016 | 0.27 | 0.0003 | 0.23 | 0.0003 | 0.22 | 0.0003 | 0.22 | 0.0003 | 0.22 |
| NS*HY3 | 0.0001 | 0.26 | 0.0004 | 4.55 | 0.010 | 0.25 | 0.0004 | 0.25 | 0.0004 | 0.20 | 0.0004 | 0.2 | 0.0004 | 0.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alhaag, H.; Yuan, X.; Mala, A.; Bai, J.; Shao, T. Fermentation Characteristics of Lactobacillus Plantarum and Pediococcus Species Isolated from Sweet Sorghum Silage and Their Application as Silage Inoculants. Appl. Sci. 2019, 9, 1247. https://doi.org/10.3390/app9061247
Alhaag H, Yuan X, Mala A, Bai J, Shao T. Fermentation Characteristics of Lactobacillus Plantarum and Pediococcus Species Isolated from Sweet Sorghum Silage and Their Application as Silage Inoculants. Applied Sciences. 2019; 9(6):1247. https://doi.org/10.3390/app9061247
Chicago/Turabian StyleAlhaag, Hager, Xianjun Yuan, Azizza Mala, Junfeng Bai, and Tao Shao. 2019. "Fermentation Characteristics of Lactobacillus Plantarum and Pediococcus Species Isolated from Sweet Sorghum Silage and Their Application as Silage Inoculants" Applied Sciences 9, no. 6: 1247. https://doi.org/10.3390/app9061247
APA StyleAlhaag, H., Yuan, X., Mala, A., Bai, J., & Shao, T. (2019). Fermentation Characteristics of Lactobacillus Plantarum and Pediococcus Species Isolated from Sweet Sorghum Silage and Their Application as Silage Inoculants. Applied Sciences, 9(6), 1247. https://doi.org/10.3390/app9061247




