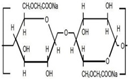
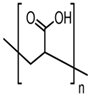
Abstract
Hydrogels are widely used materials which have many medical applications. Their ability to absorb aqueous solutions and biological fluids gives them innovative characterizations resulting in increased compatibility with biological activity. In this sense, they are used extensively for encapsulation of several targets such as biomolecules, viruses, bacteria, and mammalian cells. Indeed, many methods have been published which are used in hydrogel formulation and biomedical encapsulations involving several cross-linkers. This system is still rich with the potential of undiscovered features. The physicochemical properties of polymers, distinguished by their interactions with biological systems into mucoadhesive, gastro-adhesive, and stimuli responsive polymers. Hydrogel systems may be assembled as tablets, patches, gels, ointments, and films. Their potential to be co-formulated as nanoparticles extends the limits of their assembly and application. In this review, mucoadhesive nanoparticles and their importance for biomedical applications are highlighted with a focus on mechanisms of overcoming mucosal resistance.
1. Introduction
Hydrogels can be defined as network structures configured at two or three dimensions which have the ability to imbibe high amounts of aqueous solutions or biological fluids [1]. This affinity for water adsorption is mostly related to the presence of hydrophilic (e.g., amide, amino, carboxyl, and hydroxyl) groups on their molecular chains [2]. Due to the presence of these groups, several physicochemical properties have contributed to the classification of hydrogel systems such as porosity, structure softness, swelling capacity, and elasticity [3]. These properties are more similar to biological system than those of any other synthetic biomaterial [4]. To reinforce their structure and strengthen their mechanical force, hydrogels are cross-linked either by using physical mechanisms or chemical cross-linkers [5]. The cross-linkers in the polymer network are formulated by covalent bonds, hydrogen binding, van der Waals interactions, or by physical entanglements [6]. Furthermore, the hydrogel system becomes more solid and insoluble in any solvent, once its polymer chains are cross-linked [7]. Such hydrogels can be assembled with cryo-gelation [8], gas foaming [9], micro-emulsion formation [10], freeze-drying [11], and porogen leaching [12]. It is reported that the porous capacity and swelling behavior of ahydrogel mostly depend uponcross-linkers [13], chemical structure of the repeating unit or chemical composition [14], network structure [15], solvent concentration, surrounding medium, polymer molecular weight, quality of solvent, and the specific stimulation. Hydrophilic hydrogels show distinctive properties due to adsorption of high amounts of water in their structures compared to hydrophobic polymericnetworks. Hence, hydrophobic groups may collapse in the presence of water. The collapsed chains minimize their exposure to water molecules, ultimately resulting in lower swelling ratios [16].
Efficient delivery systems have been developed based upon on the understanding of their interactions with the biological environment, target cell population, target cell surface receptors, changes in cell receptors that occur with progression of disease, mechanism and site of drug action, drug retention, multiple drug administration, molecular mechanisms, and pathology [17]. For these reasons, many reports have referred to bio-adhesive polymers as promising materials that should be integrated into the moieties of nanoparticles [18]. This is in part due to their reactive potential and good penetration into stomach and mucosal layers. Bio-adhesive polymers are classified according to their interactions with biological systems into subgroups: mucoadhesive polymers which can penetrate mucus layers (e.g., ocular, nasal, vaginal, and small intestinal); gastro-adhesive polymers which can adhere onto stomach lining; and stimuli responsive polymers which can react to biological stimulants (e.g. glucose, enzymes, pH, and inflammation) (Figure 1) [19]. In addition to tablets, patches, gels, ointments, and film formulations; mucoadhesive nanoparticles have emerged recently to improve the controlled delivery of drugs. For nasal, vaginal, ocular, and oral administration, many nanoparticles have been optimized to possess mucoadhesive properties [20,21,22].
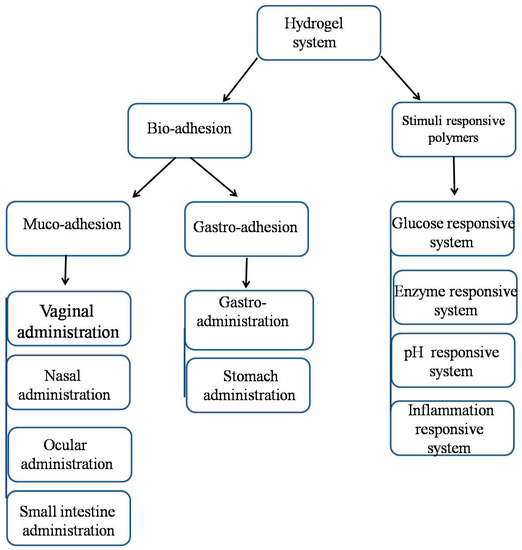
Figure 1.
Classification of hydrogel systems according to their response to biological systems.
2. Structure of Mucus Layers
Mucosal layers mainly composed of viscoelastic fluid are secreted by glandular columnar epithelial cells and contain around 99% mucin and water, with trace amounts of lipids, other proteins, and muco-polysaccharides [23]. They line the walls of various bodily cavities such as gastrointestinal and respiratory tracts, as well as vaginal and ocular surfaces. They consist of a connective tissue layer (the lamina propria) above that is an epithelial layer, the surface of which is made moist usually by the presence of a mucus layer. The epithelia may be composed of a single layer (e.g., in the bronchi, stomach, and small intestine) or be multilayered/stratified (e.g., in the esophagus, vagina and cornea). The former contain goblet cells that secrete mucus directly onto the epithelial surfaces and the latter contain (or are adjacent to tissues containing) specialized glands such as salivary glands that secrete mucus onto the epithelial surface. Mucus is a visco-elastic gel which contains mucin as the main dry weight component, typically at 2–5% (w/v), with smaller proportions of lipids and other proteins [24].
Mucins consist of high molecular weight glycoproteins encoded for by more than 20 different MUC genes. Mucins are either secreted or membrane-bound, and their expression pattern varies with their physiological site. In the gastrointestinal tract the membrane-bound MUC1, MUC3, MUC4, MUC12, MUC13, MUC16, and MUC17 proteins are expressed in addition to the secreted MUC2, MUC5AC, MUC5B, MUC6, and MUC7 [25]. MUC2 and MUC5A are mucins secreted in the small intestine and the stomach, respectively. The protein structure of mucin is rich in repeats of serine, threonine, and proline. These domains are heavily glycosylated with fucose, galactose, sialicacids, N-acetylglucosamine (GlcNAc), and N-acetylgalactosamine (GalNAc) [26]. The oligosaccharides account for 70–80% of the molecular weight of mucin and are believed to protect the protein core from degradation. The carbohydrate-bound ester sulphate residues and carboxyl groups of sialic acids provide mucus with a net negative charge [27]. Thus, the charge of the attached oligosaccharides causes intestinal mucus to bear a more negative charge, as compared to gastric mucus. The glycosylated regions of mucins are hydrophilic, whereas the non-glycosylated protein domains are hydrophobic. These naked domains are rich in cysteine, and disulphide bonds link the mucin monomers. On average, mucins are composed of four monomers with each monomer having four to five glycosylated domains [28]. Some of the lipids in mucus are covalently linked to the non-glycosylated domains of mucin, while most phospholipids adsorb and form a hydrophobic luminal lining of the mucus. The anatomic structures of mucosal layers found in different organs are shown in Table 1 [29].

Table 1.
Comparison of mucus membranes in different organs.
3. Mucus Barrier Resistance to Drugs and Delivery Systems
The viscous state of mucus structure allows poor penetration for drugs and drug delivery systems, in most cases causing high limitations for hydrophobic drug compounds [30,31]. Upon a drug’s interaction with mucosal layers, it may suffer from one or more obstructions such as dynamic, steric, and interactive barriers.
3.1. The Dynamic Barrier
Mucus secretion occurs mostly in a continuous state and can be increased or modified by disease. Additionally, peristalsis contributes to the horizontal flow along mucosal surfaces. Thus, drugs or drug delivery systems that adhere and bind to the outermost layer of mucus might be more successful in penetrating the mucosal surface.
3.2. The Steric Barrier
It is reported that mucin can form network properties exhibiting a size exclusion filter. Thus, larger compounds maybe blocked. The viscosity of the mucus and the unstirred water layer also contribute to the steric properties of the mucus barrier.
3.3. The Interactive Barrier
Mucus moieties can interact with drugs or drug delivery systems forming multiple low affinity interactions. The carbohydrates of mucin provide numerous hydrogen bond acceptors and donors as well as the potential for ionic interactions. Thus, hydrophobic interactions between the naked protein core of mucin or the lipids of mucus with diffusing compounds can be observed [32].
4. Mucoadhesive System
Mucoadhesion describes the ability of hydrogel nanoparticles to adhere to and penetrate the surface of mucosal layers [33]. Indeed, nanoparticles produced by mucoadhesive polymers have the advantage of being immobilized at the mucosal surface by an adhesion mechanism. The mucoadhesive action depends on the physical reaction of hydrogel moieties with the structure of mucosal content, and results in the swelling and expanding of the hydrogel. The swelling state perhaps facilitates and supports the adhesion of the hydrogel along mucosal layers. This stage is followed by a chemical interaction between hydrogels and mucosal contents allowing for cellular penetration and internalization of nanoparticles (Figure 2) [34]. Many factors can affect mucosal–hydrogel interactions, such as average molecular weight of polymers, chain flexibility, hydration, hydrogen bonding capacity, charge, and biological environmental factors (e.g., pH, ionic strength, and mucins) [35].
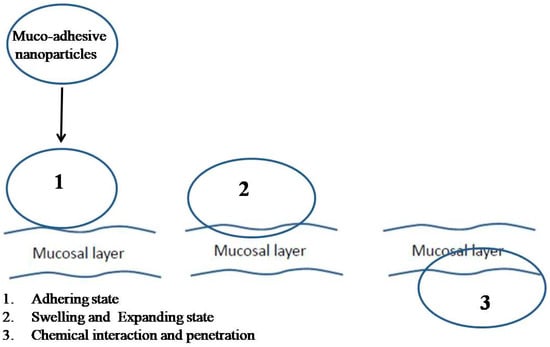
Figure 2.
Scheme illustrating a possible penetration mechanism of synthetic mucoadhesive nanoparticles on mucosal structure.
5. The Theories of Mucoadhesion
Mucoadhesion is thought to be caused by two mechanisms: the contact stage and the consolidation stage. Mucoadhesive nanoparticles which attach to a mucus membrane may result in swelling of the formation. This condition leads to the spread of the formation along the surface of the mucosal layer. This may facilitate penetration of nanoparticles inside mucosal layers [36]. In both ocular and vaginal administration, a delivery system is mechanically attached to the membrane, while in the nasal route an aerodynamic mechanism enables the delivery system to be attached. In the consolidation step, moieties of nanoparticles initiate their interaction with the moisture of mucus with weak van der Waals forces, followed by the formation of hydrogen bonds in most cases of penetration by nanoparticles into deeper layers of mucus. For instance, the glycoproteins of the mucus contribute to interpenetration of delivery systems by building secondary bonds [30]. Several theories describe physical, chemical, and mechanical attachment of a mucoadhesive system to mucous layers. These theories are shown in Table 2.

Table 2.
Theories of mucoadhesion.
6. Mucoadhesion and Its Relation with Certain Polymers
Several approaches have been designed to optimize hydrogel with bio-adhesive properties to overcome the issue of mucosal penetration and improve gastro-intestinal tract uptake [37]. Such properties can be obtained by using marine mussels incorporating the catechol moiety 3,4-dihydroxy-L-phenylalanine (DOPA) in hydrogels to promote bio-adhesion [38]. Bio-adhesion may also be improved by the interconnection of polymers having mucoadhesive properties such as chitosan, hyaluronic acid, polygalacturonic acid (pectin), alginate, and poly(acrylic acid) (Table 3) [39].

Table 3.
Polymers used to optimize mucoadhesive systems.
Additionally, lectin proteins canfurtherincrease the adherence of microparticles to the intestinal epithelium and enhance the penetration of drugs. Recently, lectins have been incorporated into nanoparticle moieties as a targetedtherapy that can bind to carbohydrate moieties attached to glycoproteins expressed on the surface of cancer cells [40]. Thiolated polymers have also emerged as a promising approach that can be used for mucoadhesion and muco-penetration in drug delivery [41].
The advantage of thiolated polymers is the ability of their free thiol groups to bind with cysteine domains in mucin [42]. The inter- and intramolecular disulfide bonds that form between mucin and thiol groups, can lead to relatively improved stability, prolonged residence and disintegration time in the mucus, and more sustained drug release [43]. Various thiolated polymers include chitosan–iminothiolane, poly(acrylic acid)–cysteine, poly(acrylic acid)–homocysteine, chitosan–thioglycolic acid, chitosan–thioethylamidine, alginate–cysteine, poly(methacrylic acid)–cysteine, and sodium carboxymethylcellulose–cysteine [44].
It is demonstrated that chitosan adheres to mucosal surfaces within the body [45] and canopen tight junctions between epithelial cells even through well organized epithelia [46]. The electrostatic interaction between the positively charged amines on chitosan polymer chains (pKa = 6.5) and the negative sialic acid residues on the glycoproteins in mucosal fluid plays a role in the mucoadhesive properties of chitosan [47]. Moreover, chitosan possesses NH2 groups that can react with mucin forming electrostatic interpenetration [48].
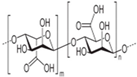
Polygalacturonic acid (PgA) is a natural polymer with a heterogeneous structure bonded via α (1,4) glycosidic linkage. Hanafy and his colleagues have demonstrated that PgA has limited dissolution in distilled water, Phosphate buffer saline (pH7.3), and acidic distilled water (pH3), resulting in white turbidity (Figure 3) [49]. This result is consistent with its hydrophobic properties [50]. PgA is not degraded in the upper gastrointestinal tract due to its insolubility in acidic conditions [51]. However, PgA can be dissolved in alkaline distilled water (pH 10), exhibiting a translucent yellow-green color. This means it can be hydrolyzed in the high pH environment of the colon. The cause of this dissolution is related to the pKa of its carboxyl group, i.e., at low pH it is protonated and the polymer is insoluble, while it is deprotonated at higher pH leading to polymer dissolution [52]. It is reported that the combination of PgA with a second polymer into a composite may alter the degree of swelling and can change its mechanical properties [53], improving both the stability of encapsulated drugs and the control of their release.
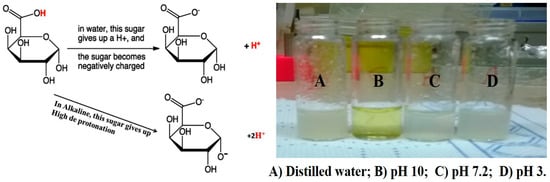
Figure 3.
Dissolution of PgA by alkaline solution (figure adapted from [49]).
It is reported that the addition of pectin to mucin in deionized water can form an aggregated compound, demonstrating an association between pectin and mucin [54]. Despite both polymers bearing a negative charge, this reaction probably induces a hydrogen–molecular interaction, where electrostatic repulsion may cause the uncoiling of polymer chains, facilitating chain entanglement and bond formation [55].
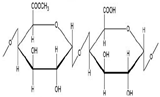
Poly(acrylic acid) (PAA) is a hydrophilic polymer [56] that adopts a random coil conformation in solution. In their recent work, Hanafy and his colleagues have further demonstrated that these coils have swelling properties under certain ionic strengths and salt concentrations, leading to extended chain conformations in alkaline solutions (Figure 4) [49,57,58,59]. This is due to the COOH side chain which is pH responsive, i.e., protonated (COOH) at pH ≤ 5 while de-protonated (COO–) at pH > 5 [60]. In aqueous media of appropriate pH and ionic strength, the carboxylic groups ionize and develop fixed charges on the polymer network, generating electrostatic repulsive forces responsible for pH-dependent behavior, causing swelling or shrinking of the hydrogel structure [61]. PAA is expected to form a hydrophilic corona. While in alkaline pH, the interaction of the swelling coils of PAA with water makes it more favorable for protection, as it adsorbs water at a weight many more times than that of its own. [62,63]. Additionally, PAA is poorly soluble in water at low pH, causing structure shrinkage. This mechanical elasticity is predicted to protect the micelles during oral administration and through the gastrointestinal tract.
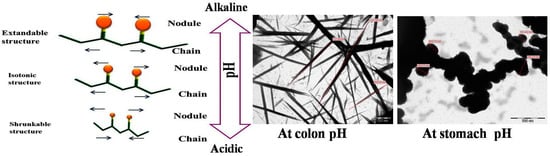
Figure 4.
Poly(acrylic acid)extended at alkaline condition and shrunk at acidic condition(figure adapted from [49] and [56]).
Poly(acrylic acid) doped with cysteine can increase mucus glycoprotein adhesion via thiol–disulfide reaction. This reaction can significantly improve the mucoadhesive properties of PAA [64].
Carboxymethyl cellulose (CMC) is a biodegradable polymer and a cellulose derivative. CMC is a good candidate for hydrogel formation because it has a good swelling capacity. Its carboxyl and hydroxyl groups are mainly responsible for hydrogel assembly due to their hydrogen intramolecular reaction [65]. Hanafy and his colleagues fabricated a hydrogel system upon a surface of fluoro-magnetic nanoparticles by using polyethylene glycol and carboxymethyl cellulose. This hydrogel was constructed using layer-by-layer assembled polymers. Further, their hydrogel properties were used to derive activin-like kinase proteins [66].
The adhesion of pectin [51], polyacrylic acid [60], hyaluronic acid [67], and alginates to mucin makes them stronger electron donors than water. Also, hydroxyl groups can act as electron acceptors. Superior mucoadhesion is further demonstrated for polymers with low molecular weights because they can more easily interpenetrate the mucin network, or display intermolecular interactions with mucin molecules [68,69,70,71].
7. Importance of Mucoadhesive Polymers for Optimizing Drug Delivery Systems
Mucoadhesive delivery has received great interest for optimizing drug delivery systems for many reasons. This system can enhance the bioavailability of therapeutic agents by avoiding drug degradation. Hydrophobic and hydrophilic molecules can be integrated inside acore–shell assembly while the surface can be further functionalized by ligands and other moleculesto increase its cellular attachment. This system is suitable for multidrug encapsulation. The physicochemical properties of mucoadhesive systems (e.g., swelling, expanding, and shrinking), are advantageous to reaction with mucosal layers, allowing nanoparticles to be internalized in tissues [49,56,58]. This system can be functionalized with several monomers that could represent other advantages for mucoadhesion either by simple fabrication or in case Layer by Layer assembly for inorganic [72] and metal nanoparticles [66,73,74]. Moreover, this system is suitable for drug delivery in oral, vaginal, nasal, and ocular administration. It may be summarized that mucoadhesive hydrogel nanoparticles represent a new and vital frontier in the optimization of drug delivery systems.
8. Conclusions
Hydrogel systems can be more easily functionalized during fabrication than any other drug delivery system, with the addition of several monomers conferring great physicochemical advantages. These advantages can increase biocompatibility, biodegradability, porosity, and mucoadhesion. Accordingly, any delivery system that is not developed with mucoadhesive moieties should not be recommended for tissues containing mucosal structure (such as ocular, vaginal, nasal, and small intestinal surfaces). Considering this limitation, many efforts have been made to improve mucoadhesion by adding thiol groups to polymer chains.
Author Contributions
N.A.N.H. designed the paper and wrote the manuscript; M.A.E.-K. guided and revised the manuscript; S.L. supervised and revised the manuscript. All authors read and approved the final manuscript version.
Funding
This research received no external funding.
Acknowledgments
Hanafy would like to thank president of Kafrelsheikh University and director of Nanoscience and Nanotechnology Institute, Maged El Kemary for his generous help, advice, and support. The highest appreciation and gratitude go to the wonderful advisor and mentor, Stefano Leporattifor his care, support and guidance.
Conflicts of Interest
Authors declare no conflict of interest.
References
- Ottenbrite, R.M.; Park, K.; Okano, T.; Peppas, N.A. Biomedical Applications of Hydrogels Handbook, 2010th ed.; Springer: New York, NY, USA, 2010; p. 432. [Google Scholar]
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2002, 43, 3–12. [Google Scholar] [CrossRef]
- Vashist, A.; Gupta, Y.K.; Ahmad, S. Recent advances in hydrogel based drug delivery systems for the human body. J. Mater. Chem. 2014, 2, 147–166. [Google Scholar] [CrossRef]
- Biondi, M.; Borzacchiello, A.; Mayol, L.; Luigi Ambrosio, L. Nanoparticle-Integrated Hydrogels as Multifunctional Composite Materials for Biomedical Applications. Gels 2015, 1, 162–178. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.H.; Patel, L.H.; Shah, H.Z.; Modasiya, K.M. Review on Hydrogel Nanoparticles in Drug Delivery. Am. J. Pharm. Technol. Res. 2011, 1, 19–38. [Google Scholar]
- Panyam, J.; Labhasetwar, V. Biodegradable nanoparticles for drug and gene delivery to cells and tissue. Adv. Drug Deliv. Rev. 2003, 55, 329–347. [Google Scholar] [CrossRef]
- Matanovi’c, M.R.; Kristl, J.; Grabnar, P.A. Thermoresponsive polymers: Insights into decisive hydrogel characteristics, mechanisms of gelation, and promising biomedical applications. Int. J. Pharm. 2014, 472, 262–275. [Google Scholar] [CrossRef] [PubMed]
- Plieva, F.M.; Galaev, I.Y.; Noppe, W.; Mattiasson, B. Cryogel applications in microbiology. Trends Microbiol. 2008, 16, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Sheridan, M.; Shea, L.; Peters, M.; Mooney, D. Bio-absorbable polymer scaffolds for tissue engineering capable of sustained growth factor delivery. J. Control Release 2000, 64, 91–102. [Google Scholar] [CrossRef]
- Zhou, S.; Bismarck, A.; Steinke, J.H. Ion-responsive alginate based macroporous injectable hydrogel scaffolds prepared by emulsion templating. J. Mater. Chem. B 2013, 1, 4736–4745. [Google Scholar] [CrossRef]
- Hassan, C.M.; Peppas, N.A. Structure and morphology of freeze/thawed PVA hydrogels. Macromolecules 2000, 33, 2472–2479. [Google Scholar] [CrossRef]
- Huebsch, N.; Lippens, E.; Lee, K.; Mehta, M.; Koshy, S.T.; Darnell, M.C.; Desai, R.M.; Madl, C.M.; Xu, M.; Zhao, X.; et al. Matrix elasticity of void-forming hydrogels controls transplanted-stem-cell-mediated bone formation. Nat. Mater. 2015, 14, 1269–1277. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-C.; Metters, A.T. Hydrogels in controlled release formulations: Network design and mathematical modeling. Adv. Drug Deliv. Rev. 2006, 58, 1379–1408. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, M.R.; Reis, A.V.; Takahashi, S.H.; Rubira, A.F.; Feitosa, J.P.A.; Muniz, E.C. Synthesis of a novel superabsorbent hydrogel by copolymerization of acrylamide and cashew gum modified with glycidyl methacrylate. Carbohydr. Polym. 2005, 61, 464–471. [Google Scholar] [CrossRef]
- Patachia, S.; Valente, A.J.M.; Baciu, C. Effect of non-associated electrolyte solutions on the behaviour of poly(vinyl alcohol)-based hydrogels. Eur. Polym. J. 2007, 43, 460–467. [Google Scholar] [CrossRef]
- Chai, Q.; Jiao, Y.; Yu, X. Hydrogels for Biomedical Applications: Their Characteristics and the Mechanisms behind Them. Gels 2017, 3, 6. [Google Scholar] [CrossRef]
- Khanna, K.V. Targeted Delivery of Nanomedicines. ISRN Pharmacol. 2012, 2012, 571394. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.; Saini, M.; Neeta. Mucoadhesive drug delivery system: An overview. Pharm. Biol. Eval. 2017, 4, 183–187. [Google Scholar] [CrossRef]
- Kharia, A.A.; Singhai, A.A. Stomach specific mucoadhesive nanoparticles as a controlled release drug delivery system. Mintage J. Pharm. Med Sci. 2014, 1, 12–24. [Google Scholar]
- Senyiğit, Z.A.; Karavana, S.Y.; Eraç, B.; Gürsel, O.; Limoncu, M.H.; Baloğlu, E. Evaluation of chitosan based vaginal bioadhesive gel formulations for antifungal drugs. Acta Pharm. 2014, 64, 139–156. [Google Scholar] [CrossRef] [PubMed]
- Kraisit, P.; Limmatvapirat, S.; Luangtana-Anan, M.; Sriamornsak, P. Buccal administration of mucoadhesive blend films saturated with propranolol loaded nanoparticles. Asian J. Pharm. Sci. 2017, 13, 34–43. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, H.; Wu, Z.; Wang, Z.; Niu, H.; Li, C. Nasal absorption enhancement of insulin using PEG-grafted chitosan nanoparticles. Eur. J. Pharm. Biopharm. 2008, 68, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Marriot, C.; Gregory, N. Bioadhesive Drug Delivery Systems; CRC Press: Boca Raton, FL, USA, 1990. [Google Scholar]
- Boegh, M.; Nielsen, H.M. Mucus as a barrier to drug delivery—Understanding and mimicking the barrier properties. BasicClin. Pharmacol. Toxicol. 2015, 116, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.E.; Sjovall, H.; Hansson, G.C. The gastrointestinal mucus system in health and disease. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Bennett, E.P.; Mandel, U.; Clausen, H.; Gerken, T.A.; Fritz, T.A.; Tabak, L.A. Control of mucin-type O-glycosylation: A classification of the polypeptide GalNAc-transferase gene family. Glycobiology 2012, 22, 736–756. [Google Scholar] [CrossRef] [PubMed]
- Cone, R.A. Barrier properties of mucus. Adv. Drug Deliv. Rev. 2009, 61, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Svensson, O.; Arnebrant, T. Mucin layers and multilayers—Physicochemical properties and applications. Curr. Opin. Colloid Interface Sci. 2010, 15, 395–405. [Google Scholar] [CrossRef]
- Shaikh, R.; RajSingh, T.R.; Garland, M.J.; Woolfson, A.D.; Donnelly, R.F. Mucoadhesive drug delivery systems. J. Pharm. Bioallied. Sci. 2011, 3, 89–100. [Google Scholar] [PubMed]
- Fröhlich, E.; Roblegg, E. Mucus as barrier for drug delivery by nanoparticles. J. Nanosci. Nanotechnol. 2014, 14, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Ways, T.M.M.; Lau, W.M.; Khutoryanskiy, V.V. Chitosan and its derivatives for application in mucoadhesive drug delivery systems. Polymers 2018, 10, 267. [Google Scholar] [CrossRef]
- Boddupalli, B.M.; Mohammed, Z.N.; Nath, R.A.; Banji, D. Mucoadhesive drug delivery system: An overview. J. Adv. Pharm. Technol. Res. 2010, 1, 381–387. [Google Scholar] [CrossRef] [PubMed]
- European Pharmacopoeia, 8th ed.; Council of Europe: Strasbourg, France, 2014; Volume 1.
- Andrews, G.P.; Laverty, T.P.; Jones, D.S. Mucoadhesive polymeric platforms for controlled drug delivery. Eur. J. Pharm. Biopharm. 2009, 71, 505–518. [Google Scholar] [CrossRef] [PubMed]
- Bansil, R.; Turner, B. Mucin structure, aggregation, physiological functions and biomedical applications. Curr. Opin. Colloid Interface Sci. 2006, 11, 164–170. [Google Scholar] [CrossRef]
- Hägerström, H.; Edsman, K.; Strømme, M. Low-frequency dielectric spectroscopy as a tool for studying the compatibility between pharmaceutical gels and mucus tissue. J. Pharm. Sci. 2003, 92, 1869–1881. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, M.; Kumar, M.; Pathak, K. A review on mucoadhesive polymer used in nasal drug delivery system. J. Adv. Pharm. Technol. Res. 2011, 2, 215. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.P.; Messersmith, P.B.; Israelachvili, J.N.; Waite, J.H. Mussel-inspired adhesives and coatings. Ann. Rev. Mater. Res. 2011, 41, 99. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Strandman, S.; Zhu, J.X.; Barralet, J.; Cerruti, M. Genipin-crosslinked catechol-chitosan mucoadhesive hydrogels for buccal drug delivery. Biomaterials 2015, 37, 395–404. [Google Scholar] [PubMed]
- Martínez-Carmona, M.; Lozano, D.; Colilla, M.; Vallet-Regí, M. Lectin-conjugated pH-responsive mesoporous silica nanoparticles for targeted bone cancer treatment. Acta Biomater. 2018, 65, 393–404. [Google Scholar]
- Cho, I.S.; Oh, H.M.; Cho, M.O.; Jang, B.S.; Cho, J.K.; Park, K.H.; Kang, S.W.; Huh, K.M. Synthesis and characterization ofthiolatedhexanoyl glycol chitosan as a mucoadhesivethermogellingpolymer. Biomater. Res. 2018, 22, 30. [Google Scholar] [CrossRef] [PubMed]
- Sosnik, A.; DasNeves, J.; Sarmento, B. Mucoadhesive polymers in the design of nano-drug delivery systems for administration by non-parenteral routes: A review. Prog. Polym. Sci. 2014, 39, 2030–2075. [Google Scholar] [CrossRef]
- Bernkop-Schnürch, A.; Greimel, A. Thiomers: The next generation of mucoadhesivepolymers. Am. J. Drug Deliv. 2005, 3, 141–154. [Google Scholar] [CrossRef]
- Sreenivas, S.A.; Pai, K.V. Thiolated chitosan: Noval polymer and mucoadhesive drug delivery—A review. Trop. J. Pharm. Res. 2008, 7, 1077–1088. [Google Scholar] [CrossRef]
- Sharma, S.; Kulkarni, J.; Pawar, A.P. Permeation enhancers in the transmucosal delivery of macromolecules. Pharmazie 2006, 61, 495–504. [Google Scholar] [PubMed]
- Schipper, N.G.M.; Varum, K.M.; Stenberg, P.; Ockind, G.; Lennernäis, H.; Artursson, P. Chitosan as absorption enhancers for poorly absorbable drugs 3: Influence of mucus on absorption enhancement. Eur. J. Pharm. Sci. 1999, 8, 335–343. [Google Scholar] [CrossRef]
- Menchicchi, B.; Fuenzalida, J.P.; BobbiliK, B.A.; Hensel, A.; Swamy, M.J.; Goycoolea, F.M. Structure of chitosan determines its interactions with mucin. Biomacromolecules 2014, 15, 3550–3558. [Google Scholar] [CrossRef] [PubMed]
- Salamat-Miller, N.; Chittchang, M.; Johnston, T.P. The use of mucoadhesive polymers in buccal drug delivery. Adv. Drug Deliv. Rev. 2005, 57, 1666–1691. [Google Scholar] [CrossRef] [PubMed]
- Hanafy, N.A.N.; Quarta, A.; Ferraro, M.M.; Dini, L.; Nobile, C.; De Giorgi, M.L.; Carallo, S.; Citti, C.; Gaballo, A.; Cannazza, G.; et al. Polymeric Nano-MicellesasNovel Cargo-Carriers for LY2157299 LiverCancerCells Delivery. Int. J. Mol. Sci. 2018, 6, 19. [Google Scholar]
- Ralet, M.C.; Dronnet, V.; Buchholt, H.C.; Thibault, J.F. Enzymatically and chemically de-esterified lime pectins: Characterisation, polyelectrolyte behaviour and calcium binding properties. Carbohydr. Res. 2001, 336, 117–125. [Google Scholar] [CrossRef]
- Chivite, A.T. Ionic Complexes of Biodegradable Polyelectrolytes. Ph.D. Thesis, Universitat Politècnica de Catalunya, Barcelona, Spain, 2014. [Google Scholar]
- Liu, L.; Fishman, M.L.; Kost, J.; Hicks, K.B. Pectin-based systems for colon-specific drug delivery via oral route. Biomaterials 2003, 24, 3333–3343. [Google Scholar] [CrossRef]
- Sriamornsak, P.; Wattanakorn, N.; Takeuchi, H. Study on the mucoadhesion mechanism of pectin by atomic force microscopy and mucin-particle method. Carbohydr. Polym. 2010, 79, 54–59. [Google Scholar] [CrossRef]
- Huang, Y.; Peppas, N.A. Nanoscale analysis of mucus–carrier interactions for improved drug absorption. In Nanotechnology in Therapeutics; Thomas, J.B., Ed.; HorizonBioscience: Wymondham, UK, 2007; pp. 109–129. [Google Scholar]
- Fang, X.; Somasundaran, P. Swelling of Poly(acrylic acid) in Concentrated Sodium Carbonate Solution. J. Chem. Eng. Data 2010, 55, 3555–3559. [Google Scholar] [CrossRef]
- Hanafy, N.A.N.; El-Kemary, M.; Leporatti, S. Reduction diameter of CaCO3 crystals by using poly acrylic acid might improve cellular uptake of encapsulated curcumin in breast cancer. J. Nanomed. Res. 2018, 7, 235–239. [Google Scholar]
- Hanafy, N.A.N.; El-Kemary, M.; Leporatti, S. Micelles structure development as a strategy to improve drug delivery system. Cancers 2018, 10, 238. [Google Scholar] [CrossRef] [PubMed]
- Hanafy, N.A.; DeGiorgi, M.L.D.; Nobile, C.; Rinaldi, R.; Leporatti, S. Control of Colloidal CaCO3 suspension by using biodegradable polymers during fabrication. Beni-Suef Univ. J. Basic Appl. Sci. 2015, 4, 60–70. [Google Scholar] [CrossRef]
- Peppas, N.A.; Wood, K.M.; Blanchette, J.O. Hydrogels for oral delivery of therapeutic proteins. Expert Opin. Biol. Ther. 2004, 4, 881–887. [Google Scholar] [CrossRef] [PubMed]
- Bastakotin, B.P.; Liao, S.H.; Inoue, M.; Yusa, S.I.; Imura, M.; Nakashima, K.; Wu, K.C.; Yamauchi, Y. pH-responsive polymeric micelles with Core-shell corona architectures as intracellular anti-cancer drug carriers. Sci. Technol. Adv. Mater. 2013, 14, 044402. [Google Scholar] [CrossRef] [PubMed]
- Iatridi, Z.; Tsitsilianis, C. Water-Soluble Stimuli Responsive Star-Shape Segmented Macromolecules. Polymers 2011, 3, 1911–1933. [Google Scholar] [CrossRef]
- Bhattarai, N.; Gunn, J.; Zhang, M. Chitosan-based hydrogels for controlled, localized drug delivery. Adv. Drug Deliv. Rev. 2010, 62, 83–99. [Google Scholar] [CrossRef] [PubMed]
- Hanafy, N.A.N. Development and production of multifunctional bio nanoengineered drug delivery systems loaded by TGFβ1 inhibitors for delivering into hepatocellular carcinoma cells. Ph.D. Thesis, Salento University, Lecce, Italy, 2017. [Google Scholar]
- Dünnhaupt, S.; Barthelmes, J.; Kollner, S.; Sakloetsakun, D.; Shahnaz, G.; Düregger, A.; Bernkop-Schnürch, A. Thiolatednanocarriers for oral delivery of hydrophilic macromolecular drugs. Carbohydr. Polym. 2015, 117, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Parodi, B.; Russo, E.; Gatti, P.; Cafaggi, S.; Bignardi, G. Development and in vitro evaluation of buccoadhesive tablets using a new model substrate for bioadhesionmeasures: The eggshellmembrane. Drug Dev. Ind. Pharm. 1999, 25, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Hanafy, N.A.; Ferraro, M.M.; Gaballo, A.; Dini, L.; Tasco, V.; Nobile, C.; DeGiorgi, M.L.; Carallo, S.; Rinaldi, R.; Leporatti, S. Fabrication and Characterization of ALK1fc-Loaded Fluoro-Magnetic Nanoparticles Rods for Inhibiting TGF β1 in HCC. RSC Adv. 2016, 6, 48834–48842. [Google Scholar] [CrossRef]
- Liao, Y.H.; Jones, S.A.; Forbes, B.; Martin, G.P.; Brown, M.B. Hyaluronan: Pharmaceutical characterization and drug delivery. Drug Deliv. 2005, 12, 327–342. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, L.; Klösgen, B.; Simonsen, A.C.; Borch, J.; Hagesaether, E. New insights into the mucoadhesion of pectins by AFM roughness parameters in combination with SPR. Int. J. Pharm. 2011, 411, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Malik, A.; Gupta, M.; Gupta, V.; Gogoi, H.; Bhatnagar, R. Novel application of trimethyl chitosan as an adjuvant in vaccine delivery. Int. J. Nanomed. 2018, 13, 7959–7970. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Fu, D.; Kang, H.; Rong, G.; Jin, Z.; Wang, X.; Zhao, K. Advances and Potential Applications of Chitosan Nanoparticles as a Delivery Carrier for the Mucosal Immunity of Vaccine. Curr. Drug Deliv. 2017, 14, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Anil, A.; Sudheer, P. Design and Evaluation of Mucoadhesive Buccal Patch of Ramipril. Int. J. Pharm. Sci. Rev. Res. 2018, 50, 121–129. [Google Scholar]
- Hanafy, N.A.N.; El-Kemary, M.; Leporatti, S. Optimizing CaCO3 matrix might allow to raise their potential use in biomedical application. J. Nanosci. Curr. Res. 2018, 3, 124. [Google Scholar] [CrossRef]
- El-banna, F.S.; Mafouz, M.; Elkemary, M.; Hanafy, N.A.N. Gold Nanoparticles: Fabrication, Modification and Biomedical Applications. JSM Nanotechnol. Nanomed. 2018, 6, 1071. [Google Scholar]
- Magdy, A.; Sadaka, E.; Elkemary, M.; Hanafy, N.A.N. Silver Nanoparticles and Cytotoxicity. JSM Nanotechnol. Nanomed. 2018, 6, 1072. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).