Effects of Graphene Nanoplatelets and Multiwall Carbon Nanotubes on the Structure and Mechanical Properties of Poly(lactic acid) Composites: A Comparative Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Nanocomposites
2.3. Preparation of Test Samples
2.4. Experimental Methods
3. Results and Discussion
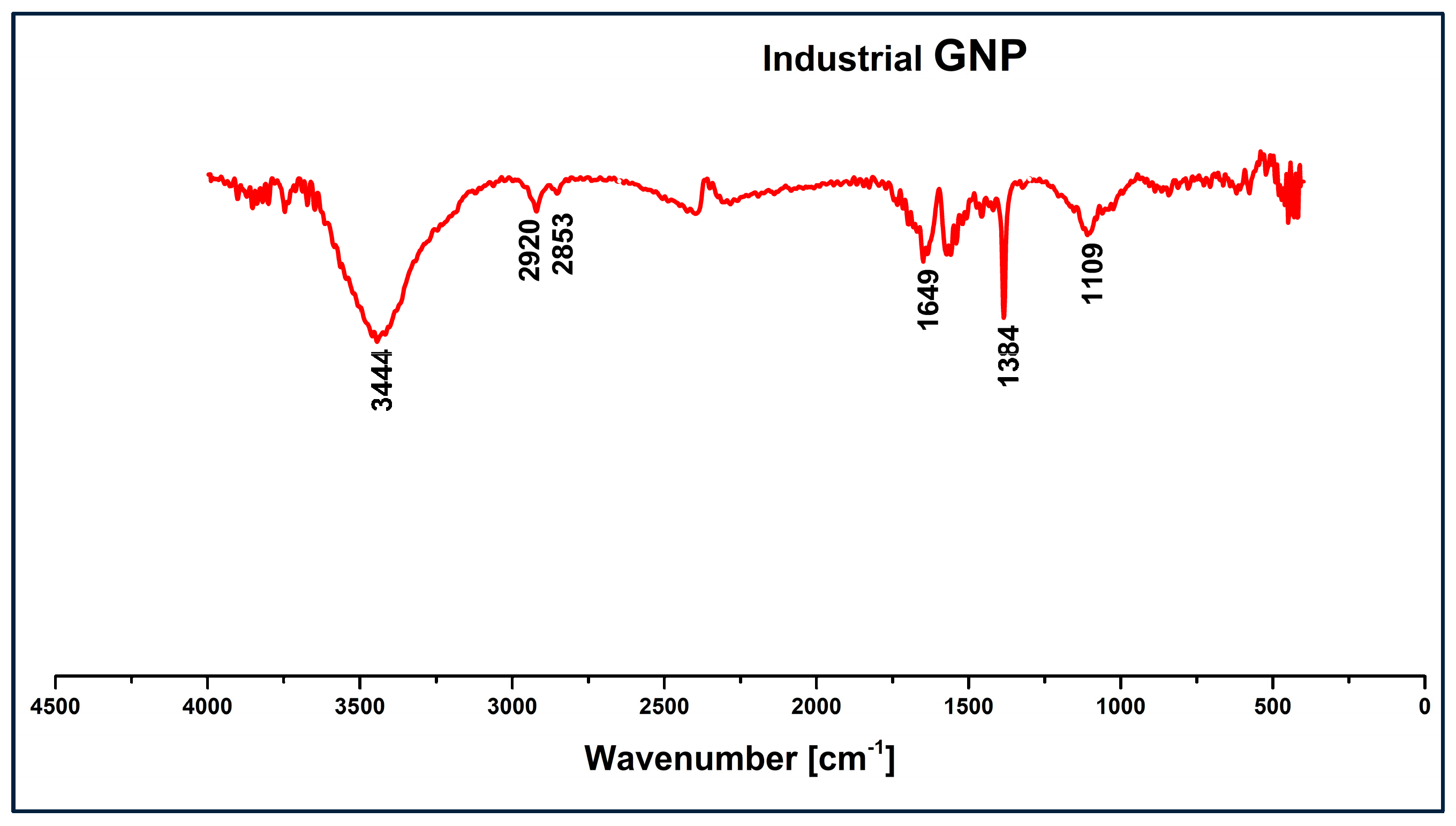
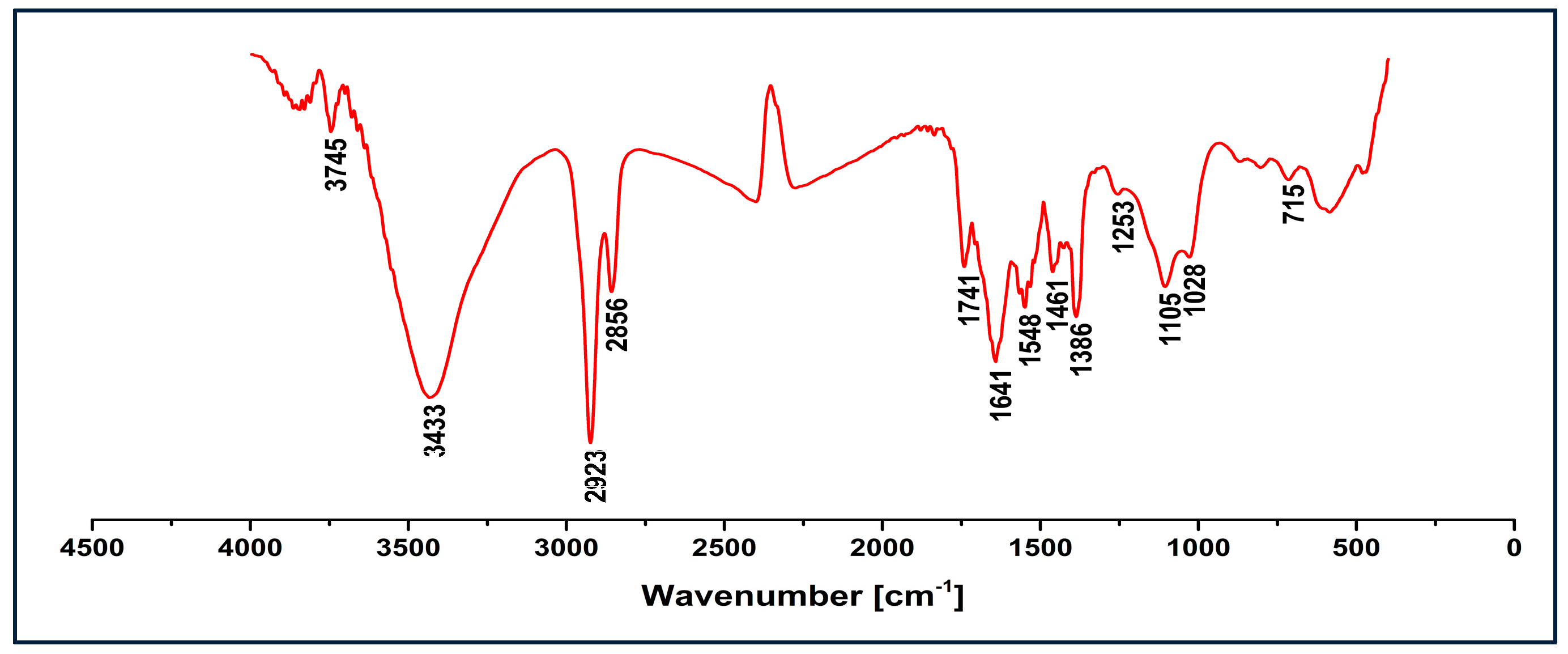
3.1. Infrared Spectroscopy Analysis
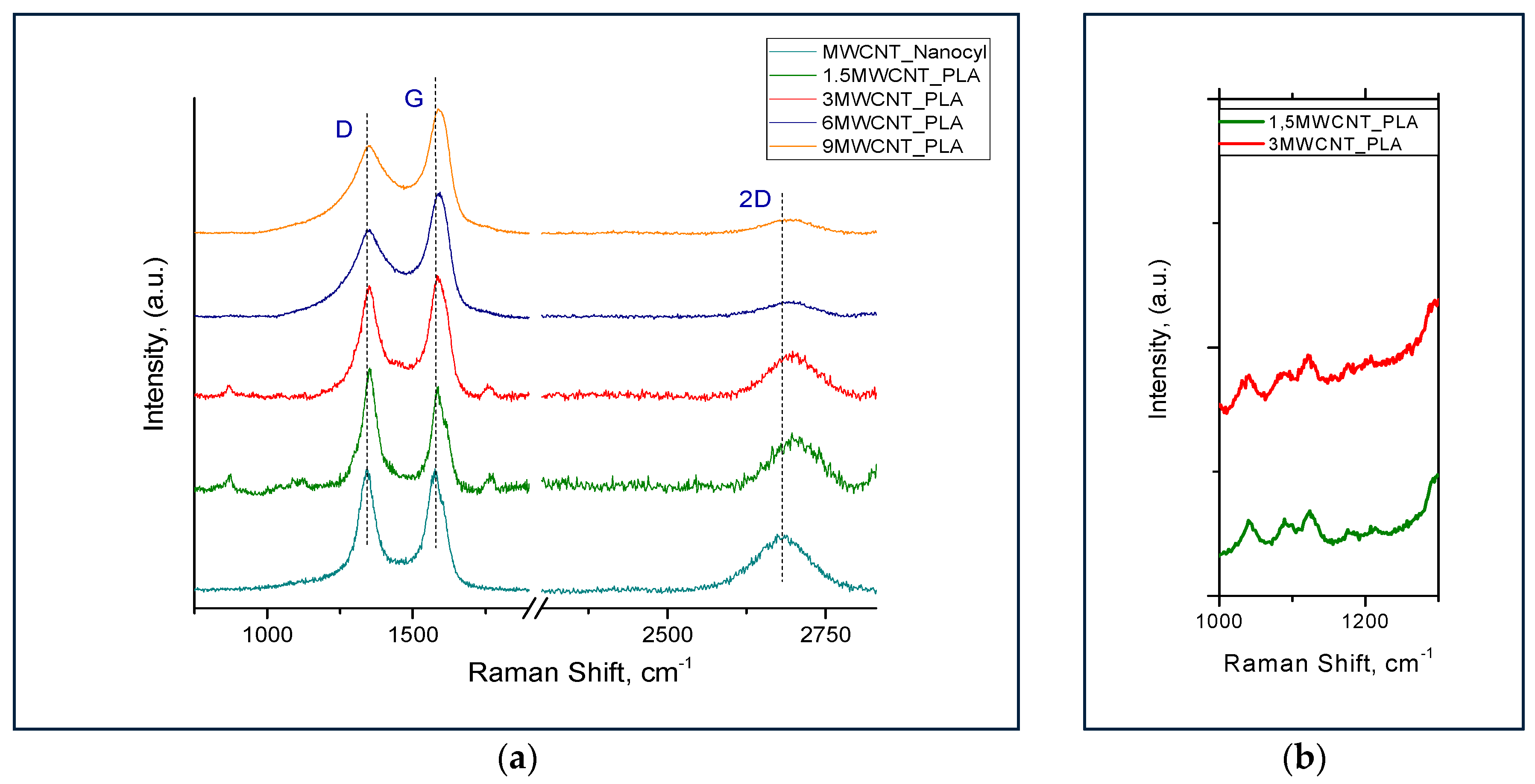
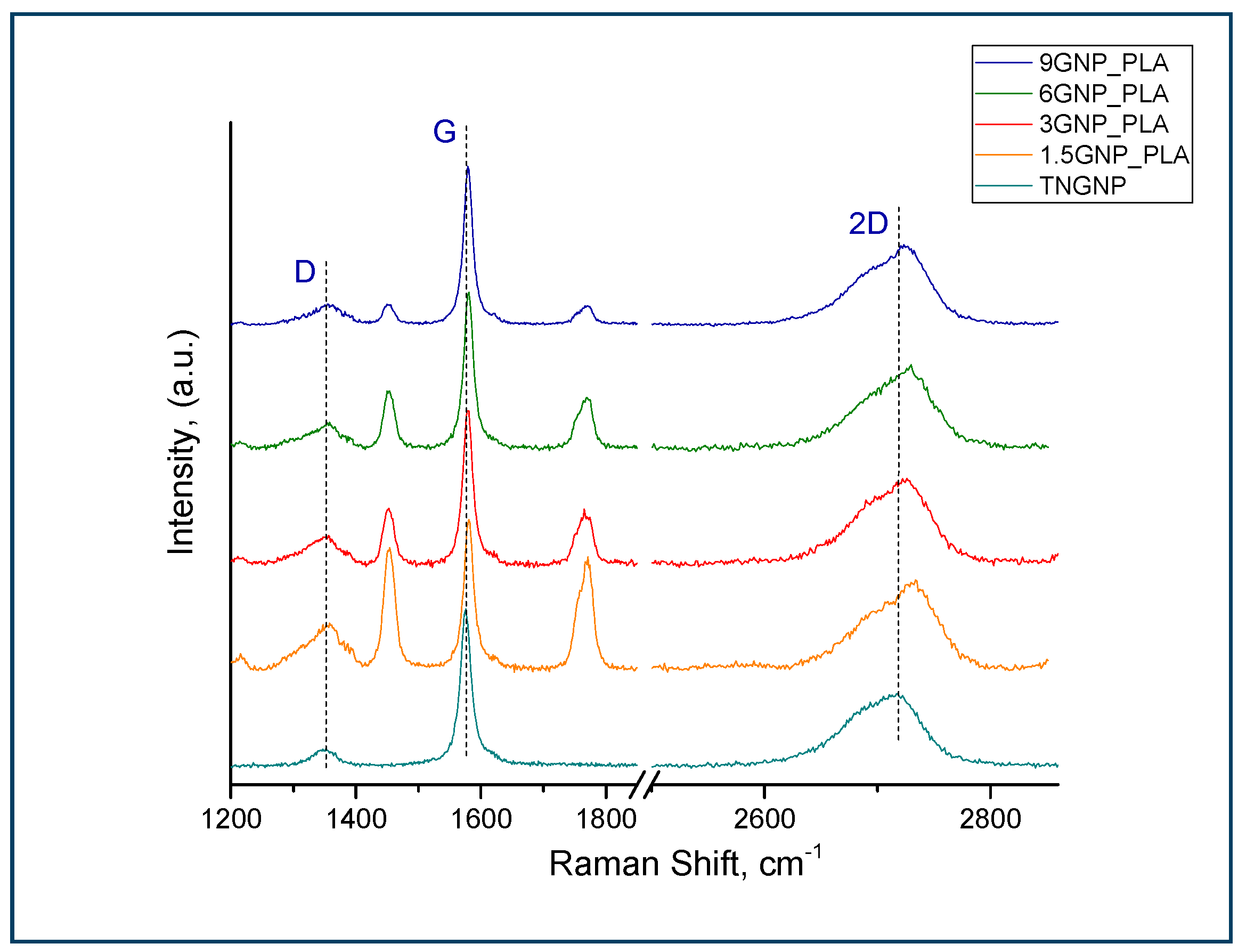
3.2. Raman Analysis
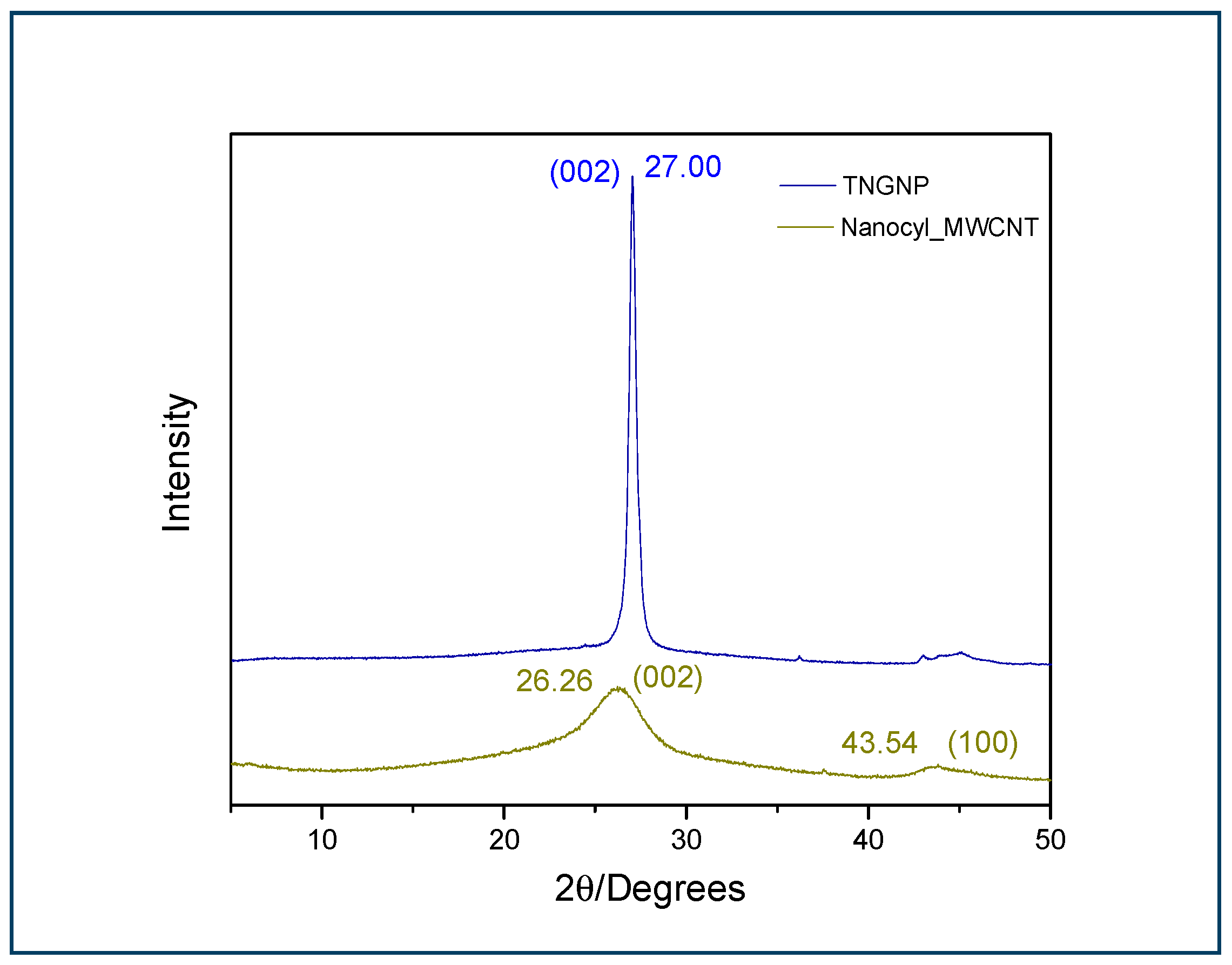
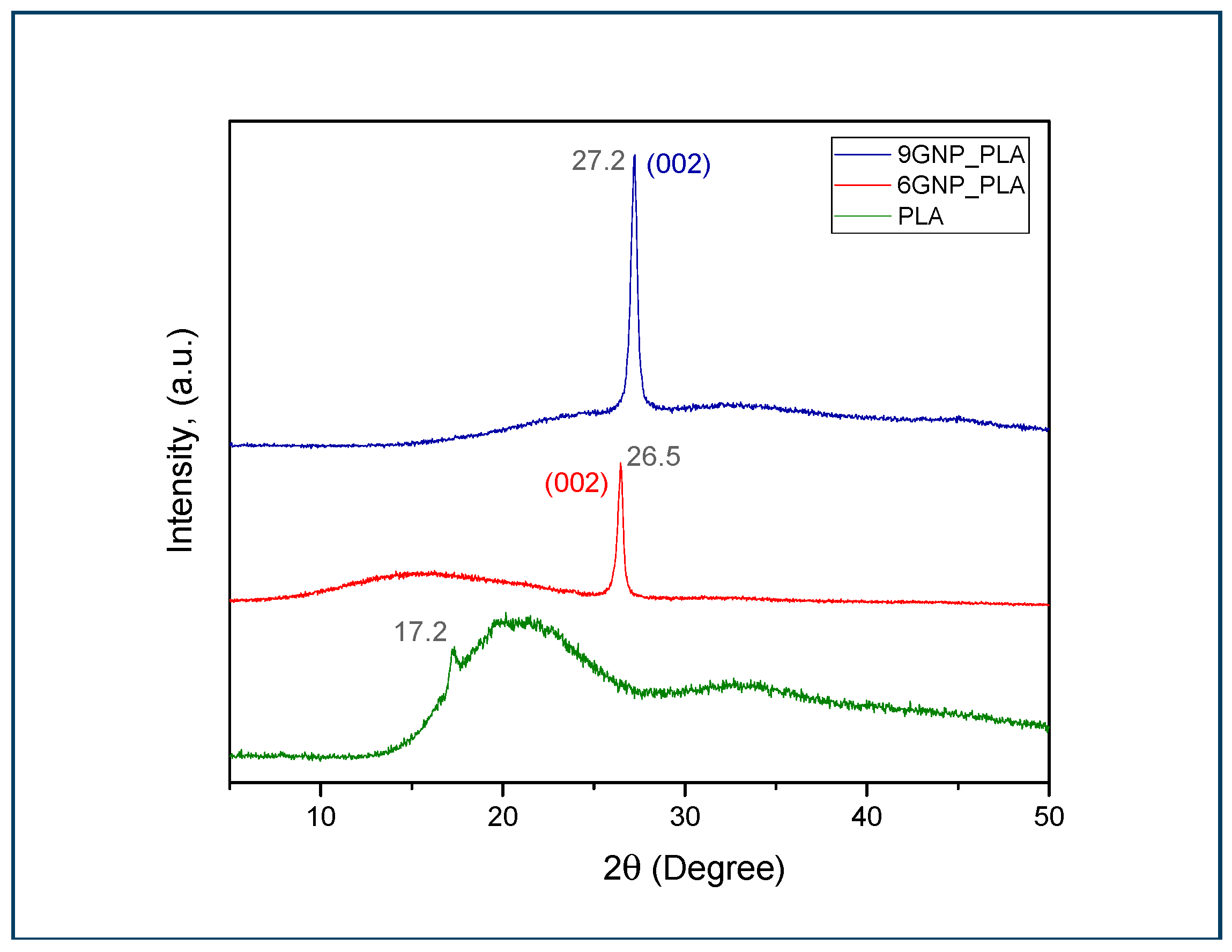
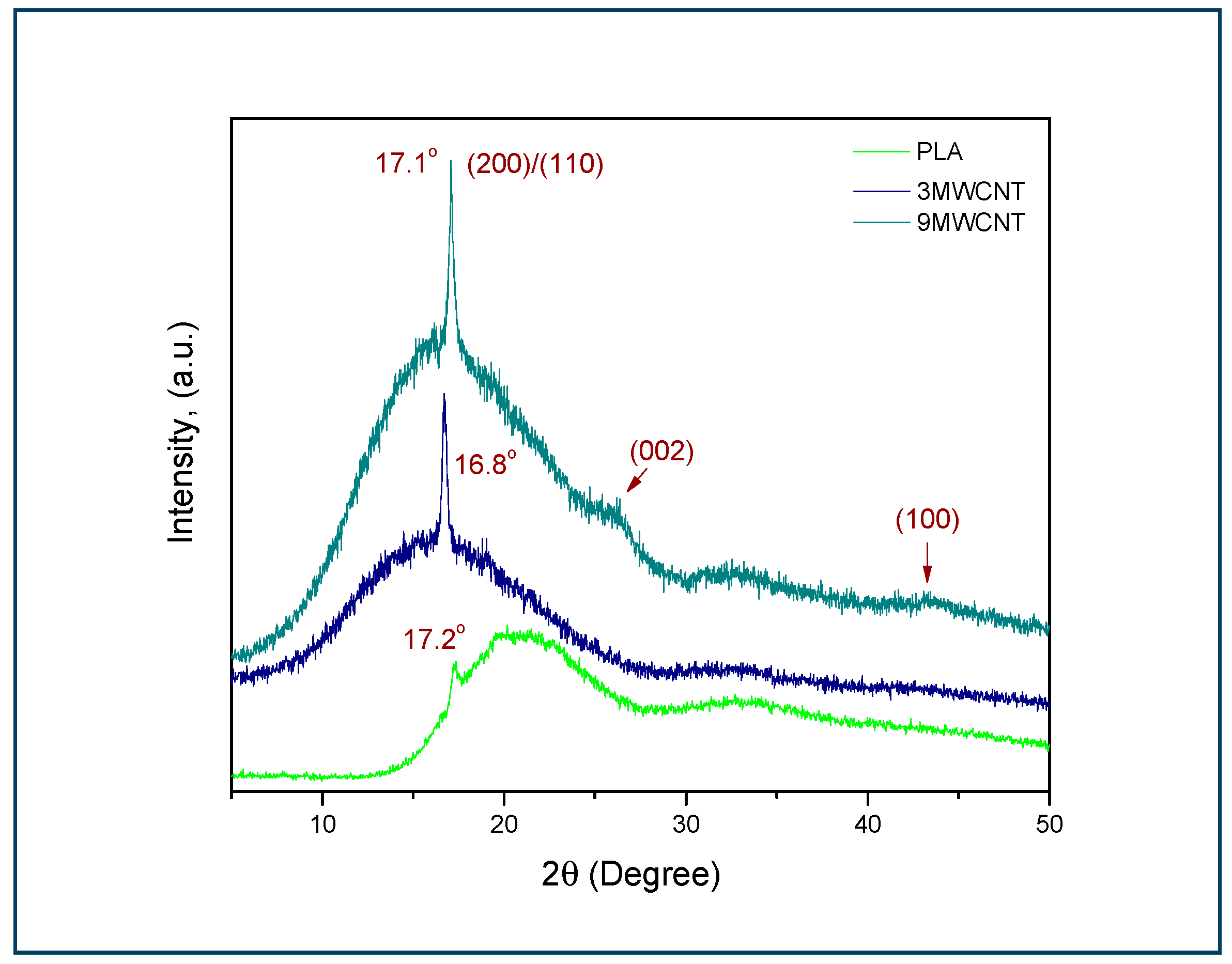
3.3. XRD Analysis
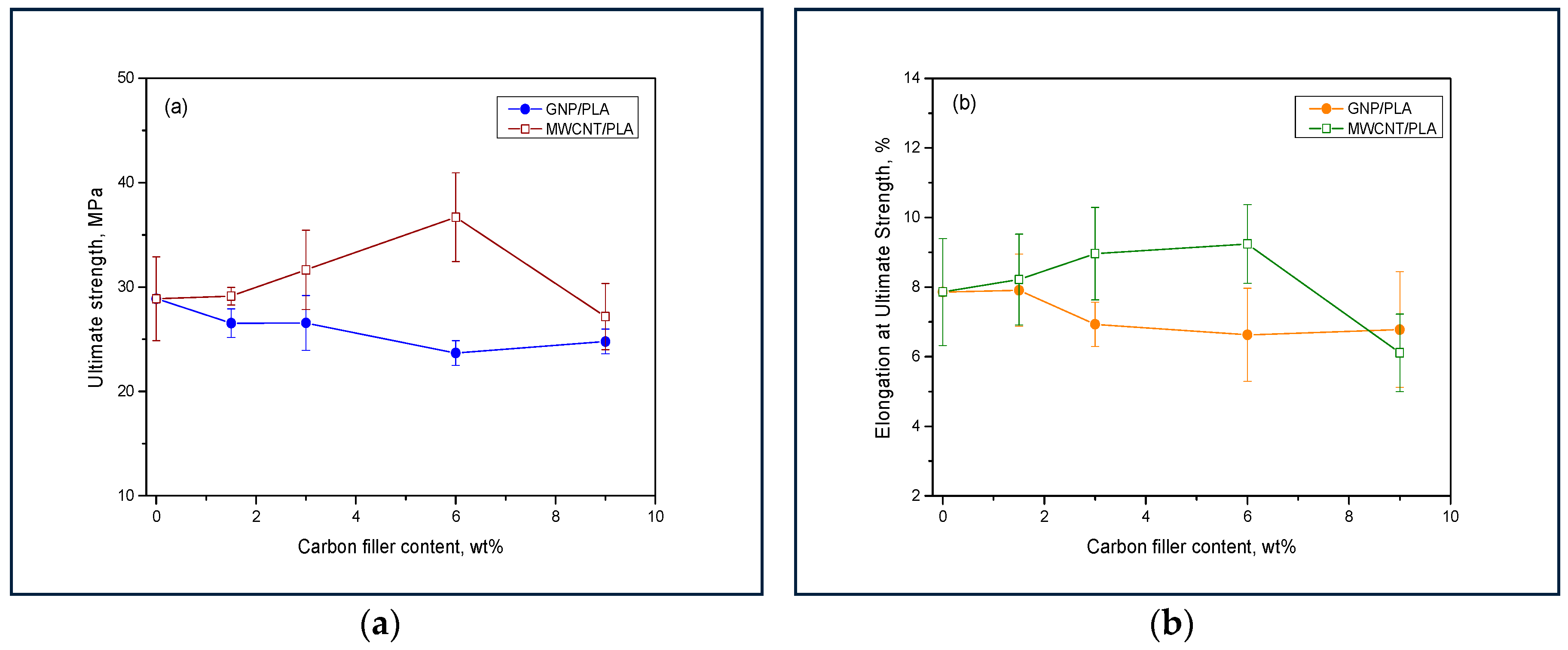
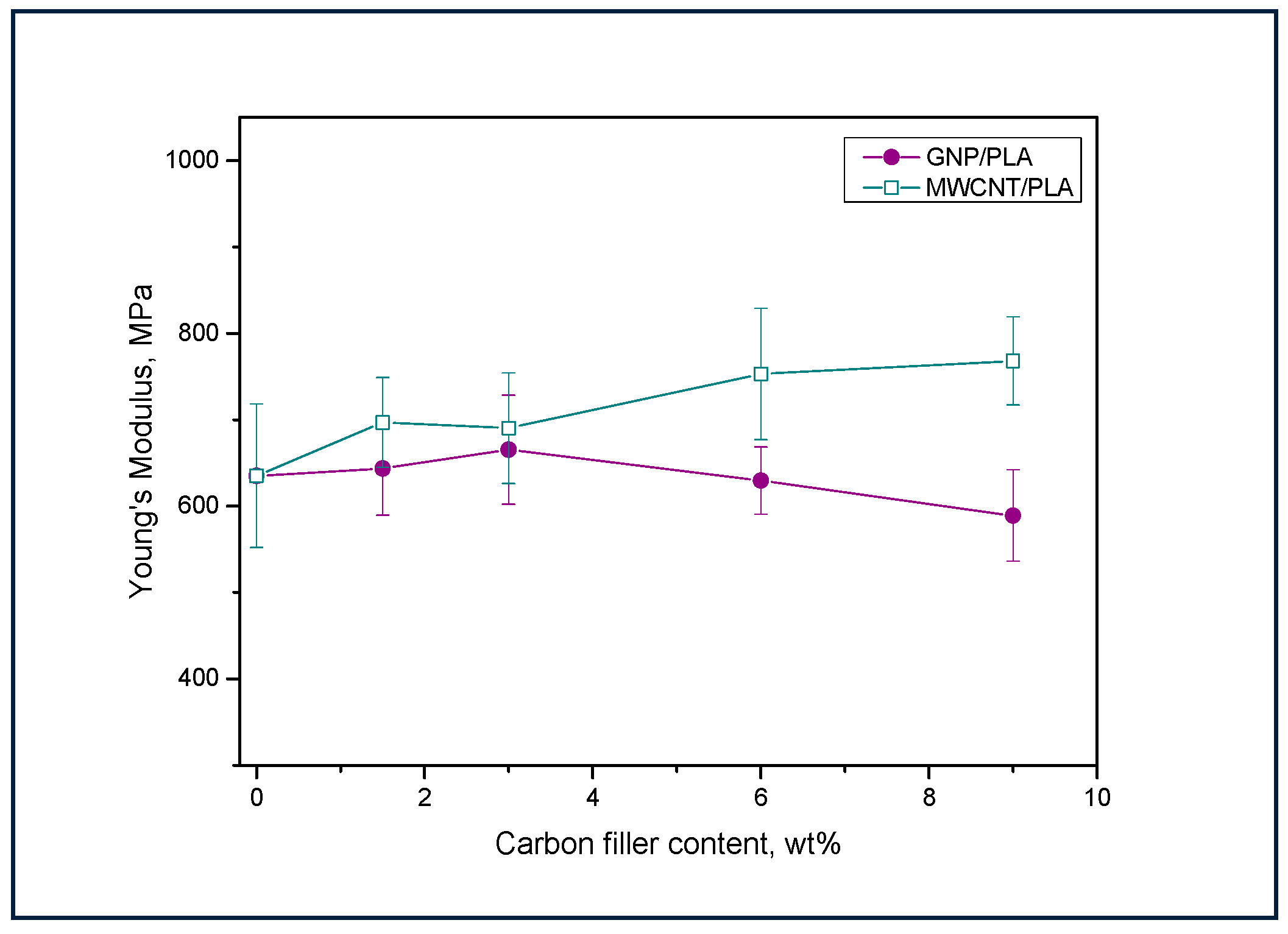
3.4. Tensile Test Analysis
3.5. SEM Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Byrne, M.T.; Gun’ko, Y.K. Recent advances in research on carbon nanotube–polymer composites. Adv. Mater. 2010, 22, 1672–1688. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Winey, K.I. Polymer nanocomposites containing carbon nanotubes. Macromolecules 2006, 39, 5194–5205. [Google Scholar] [CrossRef]
- Tasis, D.; Tagmatarchis, N.; Bianco, A.; Prato, M. Chemistry of carbon nanotubes. Chem. Rev. 2006, 106, 1105–1136. [Google Scholar] [CrossRef]
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.B.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Graphene-based composite materials. Nature 2006, 442, 282–286. [Google Scholar] [CrossRef]
- Pinto, A.M.; Susana, M.; Goncalves, I.C.; Gama, F.M.; Mendes, A.M.; Magalhães, F.D. Biocompatibility of poly(lactic acid) with incorporated graphene-based materials. Colloids Surf. B 2013, 104, 229–238. [Google Scholar] [CrossRef]
- Via, M.D.; King, J.A.; Keith, J.M.; Miskioglu, I.; Cieslinski, M.J.; Anderson, J.J.; Bogucki, G.R. Tensile modulus modeling of carbon black/polycarbonate, carbon nanotube/polycarbonate, and exfoliated graphite nanoplatelet/polycarbonate composites. J. Appl. Polym. Sci. 2012, 124, 2269–2277. [Google Scholar] [CrossRef]
- Xu, Z.; Gao, C. In situ polymerization approach to graphene–reinforced nylon–6 composites. Macromolecules 2010, 43, 6716–6723. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, X.; Jin, X.; Wang, T. Study on the PMMA/GO nanocomposites with good thermal stability prepared by in situ Pickering emulsion polymerization. J. Therm. Anal. Calorim. 2014, 117, 755–763. [Google Scholar] [CrossRef]
- Chandrasekaran, S.; Seidel, C.; Schulte, K. Preparation and characterization of graphite nano–platelet (GNP)/epoxy nano–composite: Mechanical, electrical and thermal properties. Eur. Polym. J. 2013, 49, 3878–3888. [Google Scholar] [CrossRef]
- Wang, J.; Wang, X.; Xu, C.; Zhang, M.; Shang, X. Preparation of graphene/poly(vinyl alcohol) nanocomposites with enhanced mechanical properties and water resistance. Polym. Int. 2011, 60, 816–822. [Google Scholar] [CrossRef]
- Oxfall, H.; Rondin, J.; Bouquey, M.; Muller, R.; Rigdahl, M.; Rychwalski, R.W. Elongational flow mixing for manufacturing of graphite nanoplatelet/polystyrene composites. J. Appl. Polym. Sci. 2013, 128, 2679–2686. [Google Scholar] [CrossRef]
- Murray, E.; Thompson, B.C.; Sayyar, S.; Wallace, G.G. Enzymatic degradation of graphene/polycaprolactone materials for tissue engineering. Polym. Degrad. Stab. 2015, 111, 71–77. [Google Scholar] [CrossRef]
- Quiles-Díaz, S.; Enrique-Jimenez, P.; Papageorgiou, D.G.; Ania, F.; Flores, A.; Kinloch, I.A.; Gómez-Fatou, M.A.; Young, R.J.; Salavagione, H.J. Influence of the chemical functionalization of graphene on the properties of polypropylene-based nanocomposites. Compos. Part A 2017, 100, 31–39. [Google Scholar] [CrossRef]
- Bao, C.; Song, L.; Xing, W.; Yuan, B.; Wilkie, C.A.; Huang, J.; Guo, Y.; Hu, Y. Preparation of graphene by pressurized oxidation and multiplex reduction and its polymer nanocomposites by masterbatch-based melt blending. J. Mater. Chem. 2012, 22, 6088–6096. [Google Scholar] [CrossRef]
- Raquez, J.M.; Habibi, Y.; Murariu, M.; Dubois, P. Polylactide (PLA)-based nanocomposites. Prog. Polym. Sci. 2013, 38, 1504–1542. [Google Scholar] [CrossRef]
- Huang, Y.Y.; Terentjev, E.M. Dispersion of carbon nanotubes: Mixing, sonication, stabilization, and composite properties. Polymers 2012, 4, 275–295. [Google Scholar] [CrossRef]
- Pinto, A.M.; Martins, J.; Moreira, J.A.; Mendes, A.M.; Magalhães, F.D. Dispersion of graphene nanoplatelets in poly(vinyl acetate) latex and effect on adhesive bond strength. Polym. Int. 2013, 62, 928–935. [Google Scholar] [CrossRef]
- Chieng, B.W.; Ibrahim, N.A.; Wan Yunus, W.M.Z.; Hussein, M.Z. The Mechanical, Thermal and Morphological Properties of Graphene Nanoplatelets Filled Poly(lactic acid)/Epoxidized Palm Oil Blends. Sci. Res. J. 2014, 11, 1–13. [Google Scholar]
- Valapa, R.B.; Pugazhenthi, G.; Katiyar, V. Effect of graphene content on the properties of poly(lactic acid) nanocomposites. RSC Adv. 2015, 5, 28410–28423. [Google Scholar] [CrossRef]
- Chieng, B.W.; Ibrahim, N.A.; Wan Yunus, W.M.Z.; Hussein, M.Z.; Then, Y.Y.; Loo, Y.Y. Effects of Graphene Nanoplatelets and Reduced Graphene Oxide on Poly(lactic acid) and Plasticized Poly(lactic acid): A Comparative Study. Polymers 2014, 6, 2232–2246. [Google Scholar] [CrossRef]
- Kashi, S.; Gupta, R.K.; Kao, N.; Bhattacharya, S.N. Preparation and Characterization of Poly Lactide and Poly (Butylene Adipate-co-Terephthalate) Nanocomposites Reinforced with Graphene Nanoplatelet. In Proceedings of the 40th Annual Condensed Matter and Materials Meeting, Wagga Wagga, Australia, 2–5 February 2016. [Google Scholar]
- Abdal-Hay, A.; Park, S.G.; Abdel-Jaber, G.T.; Lim, J.K. Biodegradable Poly(Lactic acid)/Multiwalled Carbon Nanotube Nanocomposites Fabrication Using Casting and Hot Press Techniques. Arch. Metall. Mater. 2015, 60, 1557–1559. [Google Scholar]
- Bounos, G.; Andrikopoulos, K.S.; Karachalios, T.K.; Voyiatzis, G.A. Evaluation of multi-walled carbon nanotube concentrations in polymer nanocomposites by Raman spectroscopy. Carbon 2014, 76, 301–309. [Google Scholar] [CrossRef]
- Román-Doval, R.; Morales-Corona, J.; Olayo, R.; Escamilla-Rivera, V.; Uribe-Ramírez, M.; Ortega-López, M. Evaluation of nanoparticles of hydroxyapatite and MWCNT’s in scaffolds of poly lactic acid. Mater. Res. Express 2016, 3, 125402. [Google Scholar] [CrossRef]
- Hsu, Y.W.; Wu, C.C.; Wu, S.M.; Su, C.C. Synthesis and Properties of Carbon Nanotube-Grafted Silica Nanoarchitecture-Reinforced Poly(Lactic Acid). Materials 2017, 10, 829. [Google Scholar] [CrossRef]
- Gao, Y.; Picot, O.T.; Bilotti, E.; Peijs, T. Influence of filler size on the properties of poly(lactic acid) (PLA)/graphene nanoplatelet (GNP) nanocomposites. Eur. Polym. J. 2017, 86, 117–131. [Google Scholar] [CrossRef]
- Li, X.; Xiao, Y.; Bergeret, A.; Longerey, M.; Che, J. Preparation of Polylactide/Graphene Composites from Liquid-Phase Exfoliated Graphite Sheets. Polym. Comp. 2014, 35, 396–403. [Google Scholar] [CrossRef]
- Singh, D.; Iyer, P.K.; Giri, P.K. Diameter dependence of interwall separation and strain in multiwalled carbon nanotubes probed by X-ray diffraction and Raman scattering studies. Diam. Relat. Mater. 2010, 19, 1281–1288. [Google Scholar] [CrossRef]
- Pinto, V.C.; Ramos, T.; Alves, S.; Xavier, J.; Tavares, P.; Moreira, P.M.; Guedes, R.M. Comparative failure analysis of PLA, PLA/GNP and PLA/CNT-COOH biodegradable nanocomposites thin films. Procedia Eng. 2015, 114, 635–642. [Google Scholar] [CrossRef]
- Botta, L.; Scaffaro, R.; Sutera, F.; Mistretta, M.C. Reprocessing of PLA/Graphene Nanoplatelets Nanocomposites. Polymers 2018, 10, 18. [Google Scholar] [CrossRef]
- Mat Desa, M.S.Z.; Hassan, A.; Arsad, A.; Mohammad, N.N.B. Mechanical properties of poly(lactic acid)/multiwalled carbon nanotubes nanocomposites. Mater. Res. Innov. 2014, 18, 14–17. [Google Scholar] [CrossRef]
- Batakliev, T.; Georgiev, V.; Ivanov, E.; Kotsilkova, R.; Di Maio, R.; Silvestre, C.; Cimmino, S. Nanoindentation analysis of 3D printed poly(lactic acid)-based composites reinforced with graphene and multiwall carbon nanotubes. J. Appl. Polym. Sci. 2018, 136, 47260. [Google Scholar] [CrossRef]
- Kuan, C.F.; Kuan, H.C.; Ma, C.C.M.; Chen, C.H. Mechanical and electrical properties of multi-wall carbon nanotube/poly(lactic acid) composites. J. Phys. Chem. Solids 2008, 69, 1395–1398. [Google Scholar] [CrossRef]
- Estévez-Martínez, Y.; Velasco-Santos, C.; Martínez- Hernández, A.; Delgado, G.; Cuevas-Yáñez, E.; Alaníz-Lumbreras, D.; Duron-Torres, S.; Castaño, V.M. Grafting of Multiwalled Carbon Nanotubes with Chicken Feather Keratin. J. Nanomater. 2013, 2013, 702157. [Google Scholar] [CrossRef]
- Yousefi, A.T.; Bagheri, S.; Shinji, K.; Rouhi, J.; Mahmood, M.R.; Ikeda, S. Fast Synthesis of Multilayer Carbon Nanotubes from Camphor Oil as an Energy Storage Material. BioMed Res. Inter. 2014, 2014, 691537. [Google Scholar]
- Dresselhaus, M.S.; Dresselhaus, G.; Saito, R.; Jorio, A. Raman spectroscopy of carbon nanotubes. Phys. Rep. 2005, 409, 47–99. [Google Scholar] [CrossRef]
- Osswald, S.; Flahaut, E.; Gogotsi, Y. In Situ Raman Spectroscopy Study of Oxidation of Double- and Single-Wall Carbon Nanotubes. Chem. Mater. 2006, 18, 1525–1533. [Google Scholar] [CrossRef]
- Osswald, S.; Havel, M.; Gogotsi, Y. Monitoring oxidation of multiwalled carbon nanotubes by Raman spectroscopy. J. Raman Spectrosc. 2007, 38, 728–736. [Google Scholar] [CrossRef]
- Jorio, A.; Saito, R.; Dresselhaus, G.; Dresselhaus, M.S. Determination of nanotubes properties by Raman spectroscopy. Philos. Trans. A Math. Phys. Eng. Sci. 2004, 362, 2311–2336. [Google Scholar] [CrossRef] [PubMed]
- Yano, T.A.; Inouye, Y.; Kawata, S. Nanoscale uniaxial pressure effect of a carbon nanotube bundle on tip-enhanced near-field Raman spectra. Nano Lett. 2006, 6, 1269–1273. [Google Scholar] [CrossRef] [PubMed]
- Cronin, S.B.; Swan, A.K.; Unlu, M.S.; Goldberg, B.B.; Dresselhaus, M.S.; Tinkham, M. Measuring the Uniaxial Strain of Individual Single-Wall Carbon Nanotubes: Resonance Raman Spectra of Atomic-Force-Microscope Modified Single-Wall Nanotubes. Phys. Rev. Lett. 2004, 93, 167401. [Google Scholar] [CrossRef]
- Kumar, R.; Cronin, S.B. Raman scattering of carbon nanotube bundles under axial strain and strain-induced debundling. Phys. Rev. B 2007, 75, 155421. [Google Scholar] [CrossRef]
- Schadler, L.S.; Giannaris, S.C.; Ajayan, P.M. Load transfer in carbon nanotube epoxy composites. Appl. Phys. Lett. 1998, 73, 3842. [Google Scholar] [CrossRef]
- Chang, T.E.; Jensen, L.R.; Kisliuk, A.; Pipes, R.B.; Pyrz, R.; Sokolov, A. Microscopic mechanism of reinforcement in single-wall carbon nanotube/polypropylene nanocomposite. Polymer 2005, 46, 439–444. [Google Scholar] [CrossRef]
- Bokobza, L.; Rahmani, M.; Belin, C.; Bruneel, J.-L.; El Bounia, N.-E. Blends of carbon blacks and multiwall carbon nanotubes as reinforcing fillers for hydrocarbon rubbers. J. Polym. Sci. Part B Polym. Phys. 2008, 46, 1939–1951. [Google Scholar] [CrossRef]
- Zhang, D.; Kandadai, M.A.; Cech, J.; Roth, S.; Curran, S. Poly(l-lactide) (PLLA)/Multiwalled Carbon Nanotube (MWCNT) Composite: Characterization and Biocompatibility Evaluation. J. Phys. Chem. B 2006, 110, 12910–12915. [Google Scholar] [CrossRef] [PubMed]
- Vano-Herrera, K.; Misuin, A.; Vogt, K. Preparation and characterization of poly(lactic acid)/poly(methyl methacrylate) blend tablets for application in quantitative analysis by micro Raman spectroscopy. J. Raman Spectrosc. 2015, 46, 273–279. [Google Scholar] [CrossRef]
- Casiraghi, C.; Pisana, S.; Novoselov, K.S.; Geim, A.K.; Ferrari, A.C. Raman fingerprint of charged impurities in graphene. Appl. Phys. Lett. 2007, 91, 233108. [Google Scholar] [CrossRef]
- Bhattacharyya, A.R.; Sreekumar, T.V.; Liu, T.; Kumar, S.; Ericson, L.M.; Hauge, R.H.; Smalley, R.E. Crystallization and orientation studies in polypropylene/single wall carbon nanotube composite. Polymer 2003, 44, 2373–2377. [Google Scholar] [CrossRef]
- Ryan, K.P.; Cadek, M.; Nicolosi, V.; Blond, D.; Ruether, M.; Armstrong, G.; Swan, H.; Fonseca, A.; Nagy, J.B.; Maser, W.K.; et al. Carbon nanotubes for reinforcement of plastics? A case study with poly(vinyl alcohol). Comp. Sci. Technol. 2007, 67, 1640–1649. [Google Scholar] [CrossRef]
- Li, Y.; Wang, Y.; Liu, L.; Han, L.; Xiang, F.; Zhou, Z. Crystallization improvement of poly(L-lactide) induced by functionalized multiwalled carbon nanotubes. J. Polym. Sci. Part B Polym. Phys. 2009, 47, 326–339. [Google Scholar] [CrossRef]
- Xu, J.; Chen, T.; Yang, C.; Li, Z.; Mao, Y.; Zeng, B.; Hsiao, B. Isothermal Crystallization of Poly(l-lactide) Induced by Graphene Nanosheets and Carbon Nanotubes: A Comparative Study. Macromolecules 2010, 43, 5000–5008. [Google Scholar] [CrossRef]
- Mirkhani, S.A.; Arjmand, M.; Sadeghi, S.; Krause, B.; Pötschke, P.; Sundararaj, U. Impact of synthesis temperature on morphology, rheology and electromagnetic interference shielding of CVD-grown carbon nanotube/polyvinylidene fluoride nanocomposites. Synth. Met. 2017, 230, 39–50. [Google Scholar] [CrossRef]
- Keteklahijani, Y.Z.; Arjmand, M.; Sundararaj, U. Cobalt Catalyst Grown Carbon Nanotube/Poly(Vinylidene Fluoride) Nanocomposites: Effect of Synthesis Temperature on Morphology, Electrical Conductivity and Electromagnetic Interference Shielding. ChemistrySelect 2017, 2, 10271–10284. [Google Scholar] [CrossRef]

| No. | Sample Index | PLA Content (wt%) | MWCNT Content (wt%) | GNP Content (wt%) |
|---|---|---|---|---|
| 1 | 1.5GNP/PLA | 98.5 | - | 1.5 |
| 2 | 3GNP/PLA | 97 | - | 3 |
| 3 | 6GNP/PLA | 94 | - | 6 |
| 4 | 9GNP/PLA | 91 | - | 9 |
| 5 | 1.5MWCNT/PLA | 98.5 | 1.5 | - |
| 6 | 3MWCNT/PLA | 97 | 3 | - |
| 7 | 6MWCNT/PLA | 94 | 6 | - |
| 8 | 9MWCNT/PLA | 91 | 9 | - |
| 9 | PLA | 100 | - | - |
| Sample | ID/IG | I2D/IG |
|---|---|---|
| TNGNP | 0.10 ± 0.01 | 0.45 ± 0.01 |
| MWCNT_Nanocyl | 1.00 ± 0.01 | 0.55 ± 0.02 |
| 1.5MWCNT/PLA | 1.14 ± 0.03 | 0.50 ± 0.01 |
| 3MWCNT/PLA | 0.85 ± 0.03 | 0.33 ± 0.02 |
| 6MWCNT/PLA | 0.71 ± 0.002 | 0.14 ± 0.01 |
| 9MWCNT/PLA | 0.72 ± 0.005 | 0.12 ± 0.01 |
| 1.5GNP/PLA | 0.30 ± 0.06 | 0.63 ± 0.03 |
| 3GNP/PLA | 0.18 ± 0.04 | 0.50 ± 0.04 |
| 6GNP/PLA | 0.15 ± 0.002 | 0.49 ± 0.03 |
| 9GNP/PLA | 0.11±0.02 | 0.48±0.04 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batakliev, T.; Petrova-Doycheva, I.; Angelov, V.; Georgiev, V.; Ivanov, E.; Kotsilkova, R.; Casa, M.; Cirillo, C.; Adami, R.; Sarno, M.; et al. Effects of Graphene Nanoplatelets and Multiwall Carbon Nanotubes on the Structure and Mechanical Properties of Poly(lactic acid) Composites: A Comparative Study. Appl. Sci. 2019, 9, 469. https://doi.org/10.3390/app9030469
Batakliev T, Petrova-Doycheva I, Angelov V, Georgiev V, Ivanov E, Kotsilkova R, Casa M, Cirillo C, Adami R, Sarno M, et al. Effects of Graphene Nanoplatelets and Multiwall Carbon Nanotubes on the Structure and Mechanical Properties of Poly(lactic acid) Composites: A Comparative Study. Applied Sciences. 2019; 9(3):469. https://doi.org/10.3390/app9030469
Chicago/Turabian StyleBatakliev, Todor, Ivanka Petrova-Doycheva, Verislav Angelov, Vladimir Georgiev, Evgeni Ivanov, Rumiana Kotsilkova, Marcello Casa, Claudia Cirillo, Renata Adami, Maria Sarno, and et al. 2019. "Effects of Graphene Nanoplatelets and Multiwall Carbon Nanotubes on the Structure and Mechanical Properties of Poly(lactic acid) Composites: A Comparative Study" Applied Sciences 9, no. 3: 469. https://doi.org/10.3390/app9030469
APA StyleBatakliev, T., Petrova-Doycheva, I., Angelov, V., Georgiev, V., Ivanov, E., Kotsilkova, R., Casa, M., Cirillo, C., Adami, R., Sarno, M., & Ciambelli, P. (2019). Effects of Graphene Nanoplatelets and Multiwall Carbon Nanotubes on the Structure and Mechanical Properties of Poly(lactic acid) Composites: A Comparative Study. Applied Sciences, 9(3), 469. https://doi.org/10.3390/app9030469









