Chemical and Enzymatic Treatment of Hemp Biomass for Bioethanol Production
Abstract
1. Introduction
2. Materials and Methods
2.1. Strain
2.2. Biomass Preparation
2.3. Chemical (Alkaline) Pretreatment of Hemp Biomass
2.4. Enzymatic Hydrolysis Process
2.5. Ethanol Fermentation
2.6. Analytical Methods
2.7. Statistical Analysis
3. Results and Discussion
3.1. Chemical Pretreatment of Hemp Biomass
3.2. Enzymatic Hydrolysis Process
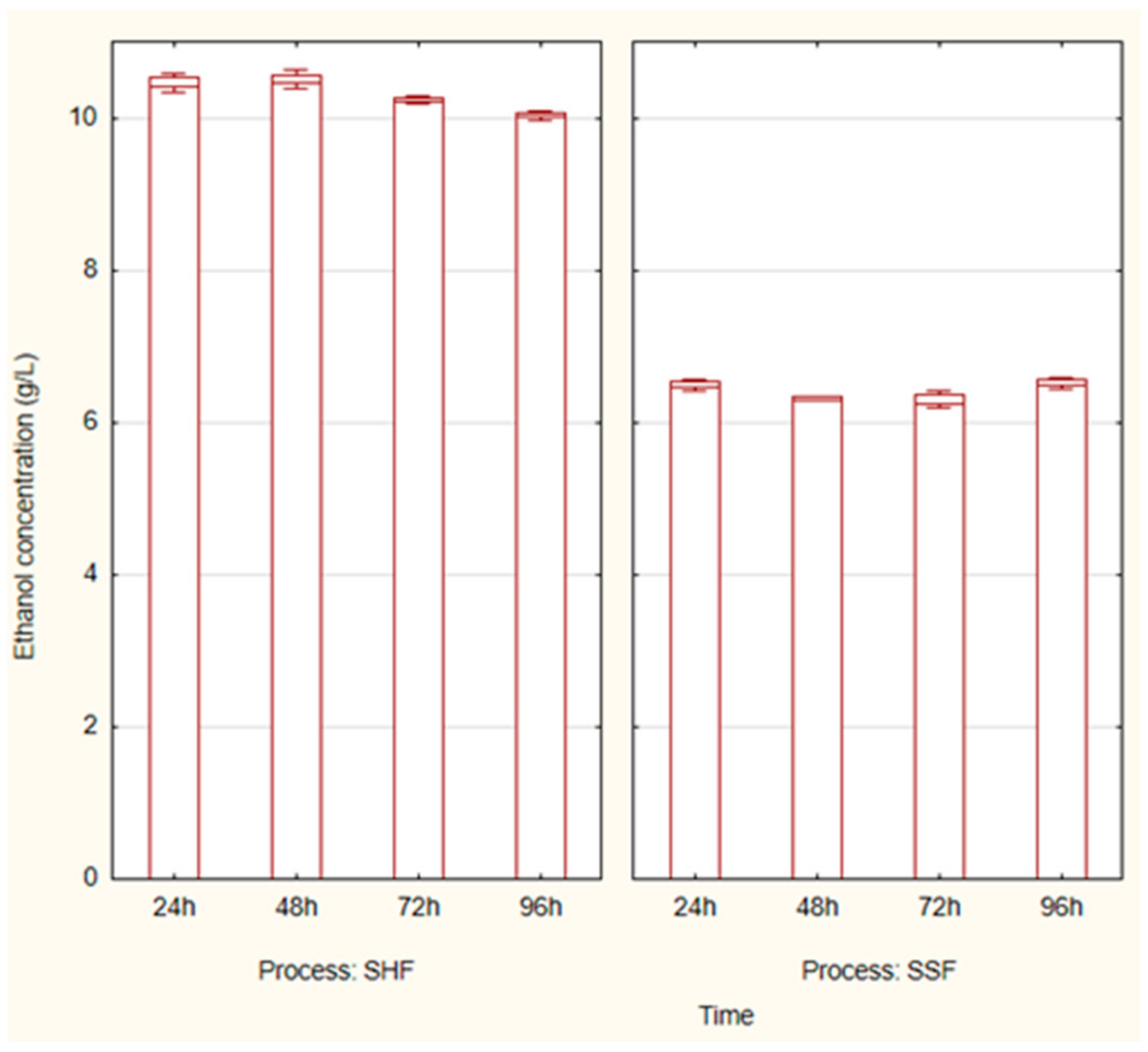
3.3. Ethanol Fermentation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Burczyk, H.; Grabowska, L.; Kołodziej, J.; Strybe, M. Industrial hemp as a raw material for energy production. J. Ind. Hemp. 2008, 37–48. [Google Scholar] [CrossRef]
- Żuk-Gołaszewska, K.; Gołaszewski, J. Cannabis sativa L.-cultivation and quality of raw material. J. Elem. 2018, 23, 971–984. [Google Scholar] [CrossRef]
- Pareek, N.; Gillgren, T.; Jönsson, L.J. Adsorption of proteins involved in hydrolysis of lignocellulose on lignins and hemicelluloses. Bioresour. Technol. 2013, 148, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Rahikainen, J.L.; Martin-Sampedro, R.; Heikkinen, H.; Rovio, S.; Marjamaa, K.; Tamminen, T.; Rojas, O.J.; Kruus, K. Inhibitory effect of lignin during cellulose bioconversion: The effect of lignin chemistry on non-productive enzyme adsorption. Bioresour. Technol. 2013, 9, 270–278. [Google Scholar] [CrossRef]
- Kim, D. Physico-chemical conversion of lignocellulose: Inhibitor effects and detoxification strategies. Molecules 2018, 23, 309. [Google Scholar] [CrossRef]
- Kumar, A.K.; Sharma, S. Recent updates on different methods of pretreatment of lignocellulosic feedstocks: A review. Bioresour. Bioprocess 2017, 4, 7. [Google Scholar] [CrossRef]
- Jędrzejczyk, M.; Soszka, E.; Czapnik, M.; Ruppert, A.M.; Grams, J. Physical and chemical pretreatment of lignocellulosic biomass. In Second and Third Generation of Feedstocks. The Evolution of Biofuels; Basile, A., Dalena, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 143–196. [Google Scholar] [CrossRef]
- Asgher, M.; Ahmad, Z.; Iqbal, H.M.N. Alkali and enzymatic delignification of sugarcane bagasse to expose cellulose polymers for saccharification and bioethanol production. Ind. Crop. Prod. 2013, 44, 488–495. [Google Scholar] [CrossRef]
- Parawira, W.; Tekere, M. Biotechnological strategies to overcome inhibitors in lignocellulose hydrolysates for ethanol production: Review. Crit. Rev. Biotechnol. 2011, 31, 20–31. [Google Scholar] [CrossRef]
- Jaisamut, K.; Paulová, L.; Patáková, P.; Rychtera, M.; Melzoch, K. Optimization of alkali pretreatment of wheat straw to be used as substrate for biofuels production. Plant Soil Environ. 2013, 59, 537–542. [Google Scholar] [CrossRef]
- Sharma, V.; Sharma, S.; Kuila, A. A review on current technological advancement of lignocellulosic bioethanol production. J. Appl. Biotechnol. Bioeng. 2016, 1, 61–66. [Google Scholar] [CrossRef]
- Maurya, D.P.; Singla, A.; Negi, S. An overview of key pretreatment processes for biological conversion of lignocellulosic biomass to bioethanol. 3 Biotech. 2015, 5, 597–609. [Google Scholar] [CrossRef]
- Lee, C.-R.; Sung, B.H.; Lim, K.-M.; Kim, M.-J.; Sohn, M.J.; Bae, J.-H.; Sohn, J.-H. Co-fermentation using recombinant Saccharomyces cerevisiae yeast strains hyper-secreting different cellulases for the production of cellulosic bioethanol. Sci. Rep. 2017, 7, 4428. [Google Scholar] [CrossRef]
- Dong, J.-J.; Ding, J.-C.; Zhang, Y.; Ma, L.; Xu, G.-C.; Han, R.-Z.; Ni, Y. Simultaneous saccharification and fermentation of dilute alkaline-pretreated corn stover for enhanced butanol production by Clostridium saccharobutylicum DSM 13864. FEMS Microbiol. Lett. 2016, 363, 1–6. [Google Scholar] [CrossRef]
- El-Naggar, N.E.; Deraz, S.; Khalil, A. Bioethanol production from lignocellulosic feedstocks based on enzymatic hydrolysis: Current status and recent developments. Biotechnology 2014, 13, 1–21. [Google Scholar] [CrossRef]
- Suttikul, S.; Srinorakutara, T.; Butivate, E.; Orasoon, K. Comparison of SHF and SSF processes for ethanol production from alkali-acid pretreated sugarcane trash. KKU Res. J. 2016, 21, 229–235. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugars. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- TAPPI 17 m-55. In Cellulose in Wood; TAPPI Press: Atlanta, GA, USA, 1955.
- TAPPI T9 m-54. In Holocellulose in Wood; TAPPI Press: Atlanta, GA, USA, 1998.
- Bagby, M.O.; Nelson, G.H.; Helman, E.G.; Clark, T.F. Determination of lignin non-wood plant fiber sources. TAPPI J. 1971, 54, 11. [Google Scholar]
- Stevulova, N.; Cigasova, J.; Estokova, A.; Terpakova, E.; Geffert, A.; Kacik, F.; Singovszka, E.; Holub, M. Properties characterization of chemically modified hemp hurds. Materials 2014, 7, 8131–8150. [Google Scholar] [CrossRef]
- Łukajtis, R.; Rybarczyk, P.; Kucharska, K.; Konopacka-Łyskawa, D.; Słupek, E.; Wychodnik, K.; Kamiński, M. Optimization of saccharification conditions of lignocellulosic biomass under alkaline pretreatment and enzymatic hydrolysis. Energies 2018, 11, 886. [Google Scholar] [CrossRef]
- Kumar, P.; Barrett, D.M.; Delwiche, M.J.; Stroeve, P. Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production. Ind. Eng. Chem. Res. 2009, 488, 3713–3729. [Google Scholar] [CrossRef]
- Gupta, A.D.; Pandey, S.; Kumar Jaiswal, V.; Bhadauria, V.; Singh, H. Simultaneous oxidation and esterification of cellulose for use in treatment of water containing Cu (II) ions. Carbohydr. Polym. 2019, 222, 114964. [Google Scholar] [CrossRef] [PubMed]
- Bregado, J.L.; Secchi, A.R.; Tavares, F.W.; de Sousa Rodrigues, D.; Gambetta, R. Amorphous paracrystalline structures from native crystalline cellulose: A molecular dynamics protocol. Fluid Phase Equilibr. 2019, 491, 56. [Google Scholar] [CrossRef]
- Šutka, A.; Kukle, S.; Grāvītis, J. STEX treated and untreated hemp fiber comparative structural analysis. Material Science. Text. Cloth. Technol. 2011, 6, 36–42. [Google Scholar]
- Plácido, J.; Capareda, S. Analysis of alkali ultrasonication pretreatment in bioethanol production from cotton gin trash using FT-IR spectroscopy and principal component analysis. Bioresour. Bioprocess. 2014, 1, 23. [Google Scholar] [CrossRef]
- Fang, G.; Chen, H.; Chen, A.; Mao, K.; Wang, Q. An efficient method of bio-chemical combined treatment for obtaining high-quality hemp fiber. BioResources 2017, 12, 1566–1578. [Google Scholar] [CrossRef]
- Abraham, R.E.; Barrow, C.J.; Puri, M. Relationship to reducing sugar production and scanning electron microscope structure to pretreated hemp hurd biomass (Cannabis sativa). Biomass Bioenerg. 2013, 58, 180–187. [Google Scholar] [CrossRef]
- Izmirlioglu, G.; Demirci, A. Ethanol production from waste potato mash by using Saccharomyces cerevisiae. Appl. Sci. 2012, 2, 738–753. [Google Scholar] [CrossRef]
- Abraham, R.E. Bioprocessing of Hemp Hurd (Cannabis sativa) for Biofuel Production. Ph.D. Thesis, Deakin University, Deakin, Australia, 30 October 2014. [Google Scholar]
- Salimi, M.N.; Lim, S.E.; Yusoff, A.H.M.; Jamlos, M.F. Conversion of rice husk into fermentable sugar by two stage hydrolysis. J. Phys. Conf. Ser. 2017, 908, 012056. [Google Scholar] [CrossRef]
- Fojas, J.J.R.; Rosario, E.J.D. Optimization of pretreatment and enzymatic saccharification of cogon grass prior ethanol production. Chem. Mol. Eng. 2013, 7, 296–299. [Google Scholar]
- Maryanto, D.; Sonifa, R.; Kurniawan, S.A.; Hadiyanto, H. Pretreatment of Starch-Free Sugar Palm Trunk (Arenga pinnata) to Enhance Saccharification in Bioethanol Production. MATEC Web Conf. 2018, 156, 6. [Google Scholar] [CrossRef]
- Moreno, A.D.; Alvira, P.; Ibarra, D.; Tomas-Pejo, E. Production of ethanol from lignocellulosic biomass. In Production of Platform Chemicals from Sustainable Resources; Biofuels and Biorefineries; Feng, Z., Smith, R.L., Jr., Qi, X., Eds.; Springer: Singapore, 2017; pp. 375–410. [Google Scholar] [CrossRef]
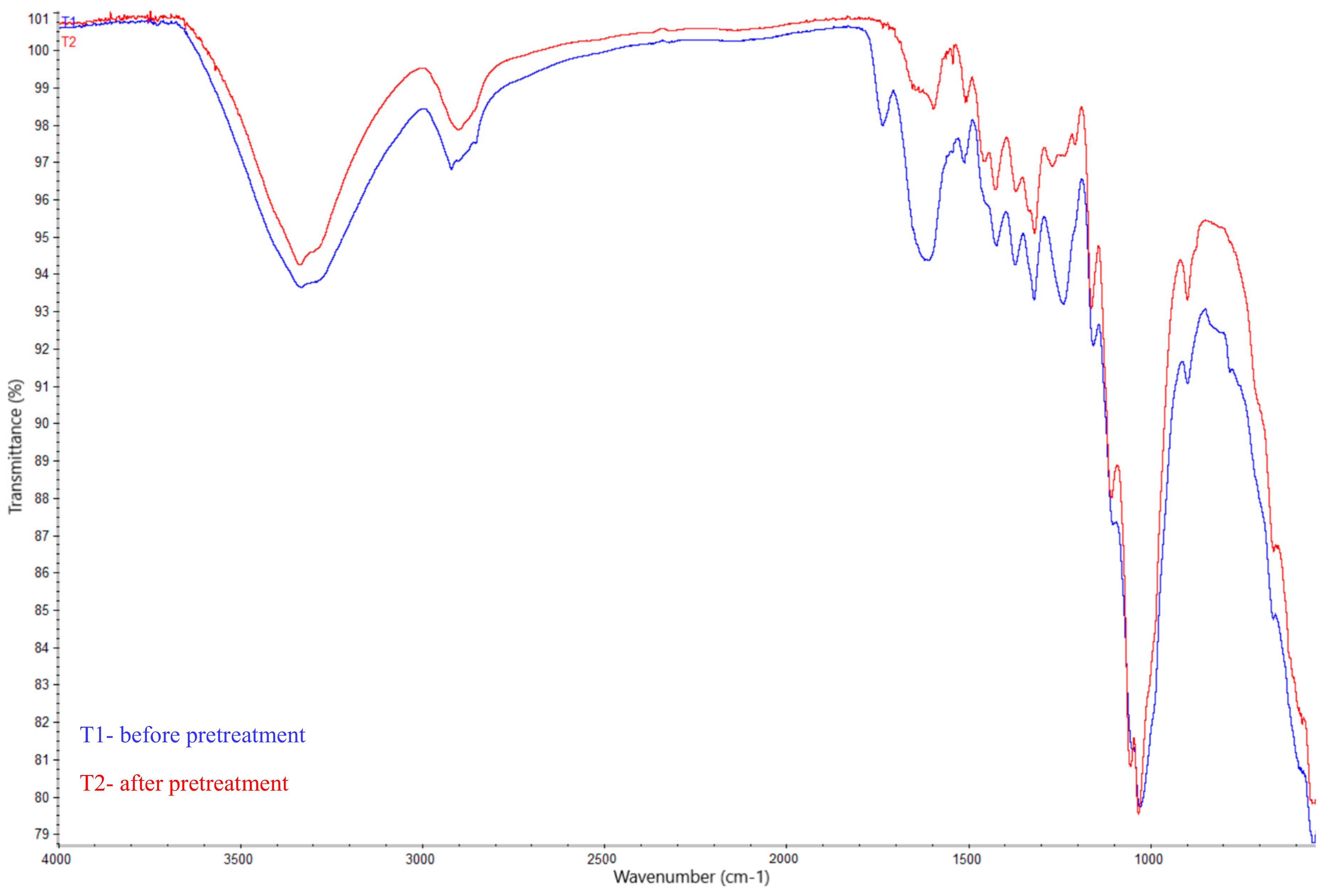


| Samples | Cellulose (%) | Hemicellulose (%) | Lignin (%) |
|---|---|---|---|
| BP | 50.82 ± 0.12 | 27.79 ± 0.33 | 14.68 ± 0.46 |
| AP | 62.70 ± 0.09 | 20.16 ± 0.16 | 15.12 ± 0.22 |
| Vibration of Function Group | Source | Wavenumber (cm−1) |
|---|---|---|
| OH stretching | polysaccharides | 3340 |
| C-H symmetrical stretching | polysaccharides | 2897 |
| C = O unconjugated stretching | hemicellulose | 1733 |
| OH (water) | cellulose | 1636 |
| C = C symmetrical stretching of the aromatic ring | lignin | 1507 |
| CH2 bending | cellulose | 1422 |
| glycosidic bonds symmetric ring-stretching mode | polysaccharides | 896 |
| Enzymes | Reducing Sugars (mg/g) | |
|---|---|---|
| 55 °C (SHF) | 38 °C (SSF) | |
| Flashzyme Plus 200 | 1100 | 1214 |
| Celluclast 1.5 L | 875 | 909 |
| Flashzyme/Celluclast 1.5 L (30/70) | 1197 | 1306 |
| Flashzyme/Celluclast 1.5 L (50/50) | 1265 | 1295 |
| Flashzyme/Celluclast 1.5 L (70/30) | 1258 | 1315 |
| Flashzyme/glucosidase | 1547 | 1905 |
| Flashzyme/xylanase | 1518 | 1914 |
| Flashzyme/glucosidase/xylanase | 1653 | 2005 |
| Flashzyme/Celluclast 1.5 L (70/30)/glucosidase | 1488 | 2035 |
| Flashzyme/Celluclast 1.5 L (70/30)/xylanase | 1507 | 2311 |
| Flashzyme/Celluclast 1.5 L (70/30)/glucosidase/xylanase | 1431 | 2289 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wawro, A.; Batog, J.; Gieparda, W. Chemical and Enzymatic Treatment of Hemp Biomass for Bioethanol Production. Appl. Sci. 2019, 9, 5348. https://doi.org/10.3390/app9245348
Wawro A, Batog J, Gieparda W. Chemical and Enzymatic Treatment of Hemp Biomass for Bioethanol Production. Applied Sciences. 2019; 9(24):5348. https://doi.org/10.3390/app9245348
Chicago/Turabian StyleWawro, Aleksandra, Jolanta Batog, and Weronika Gieparda. 2019. "Chemical and Enzymatic Treatment of Hemp Biomass for Bioethanol Production" Applied Sciences 9, no. 24: 5348. https://doi.org/10.3390/app9245348
APA StyleWawro, A., Batog, J., & Gieparda, W. (2019). Chemical and Enzymatic Treatment of Hemp Biomass for Bioethanol Production. Applied Sciences, 9(24), 5348. https://doi.org/10.3390/app9245348





