Effects of Whole-Body Vibration Exercises on Parameters Related to the Sleep Quality in Metabolic Syndrome Individuals: A Clinical Trial Study
Abstract
1. Introduction
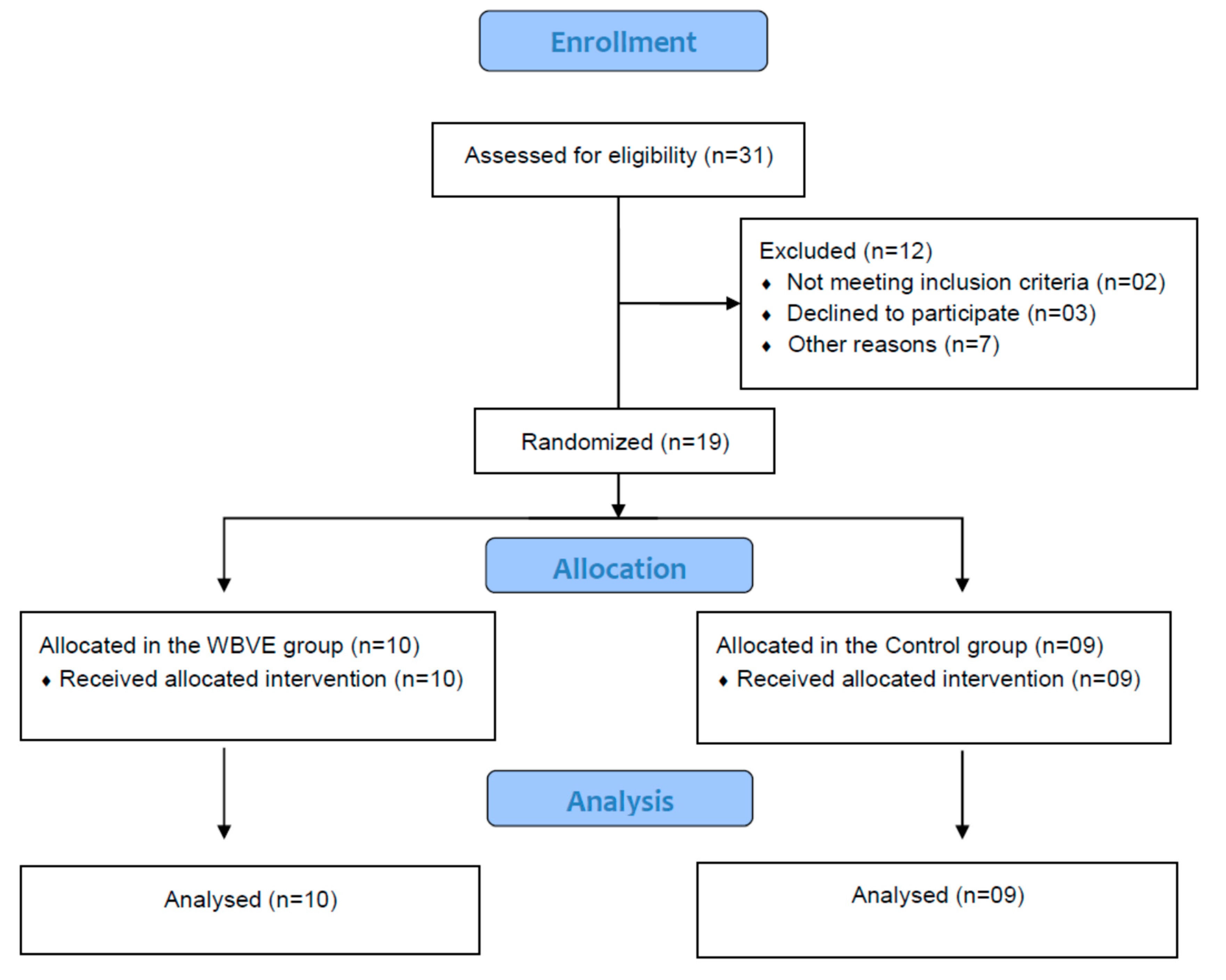
2. Materials and Methods
2.1. Individuals
2.2. Interventions
2.2.1. Fixed Frequency Group (Control Group)
2.2.2. Variable Frequency Group (WBVE Group)
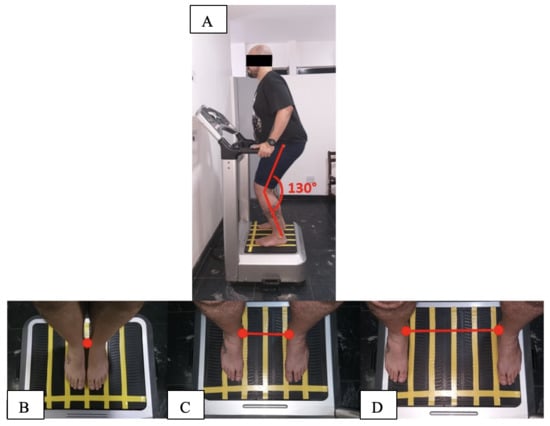
2.3. Anthropometric Measurements and Physiological Parameters
2.4. Questionnaires
2.5. Statistical Analyses
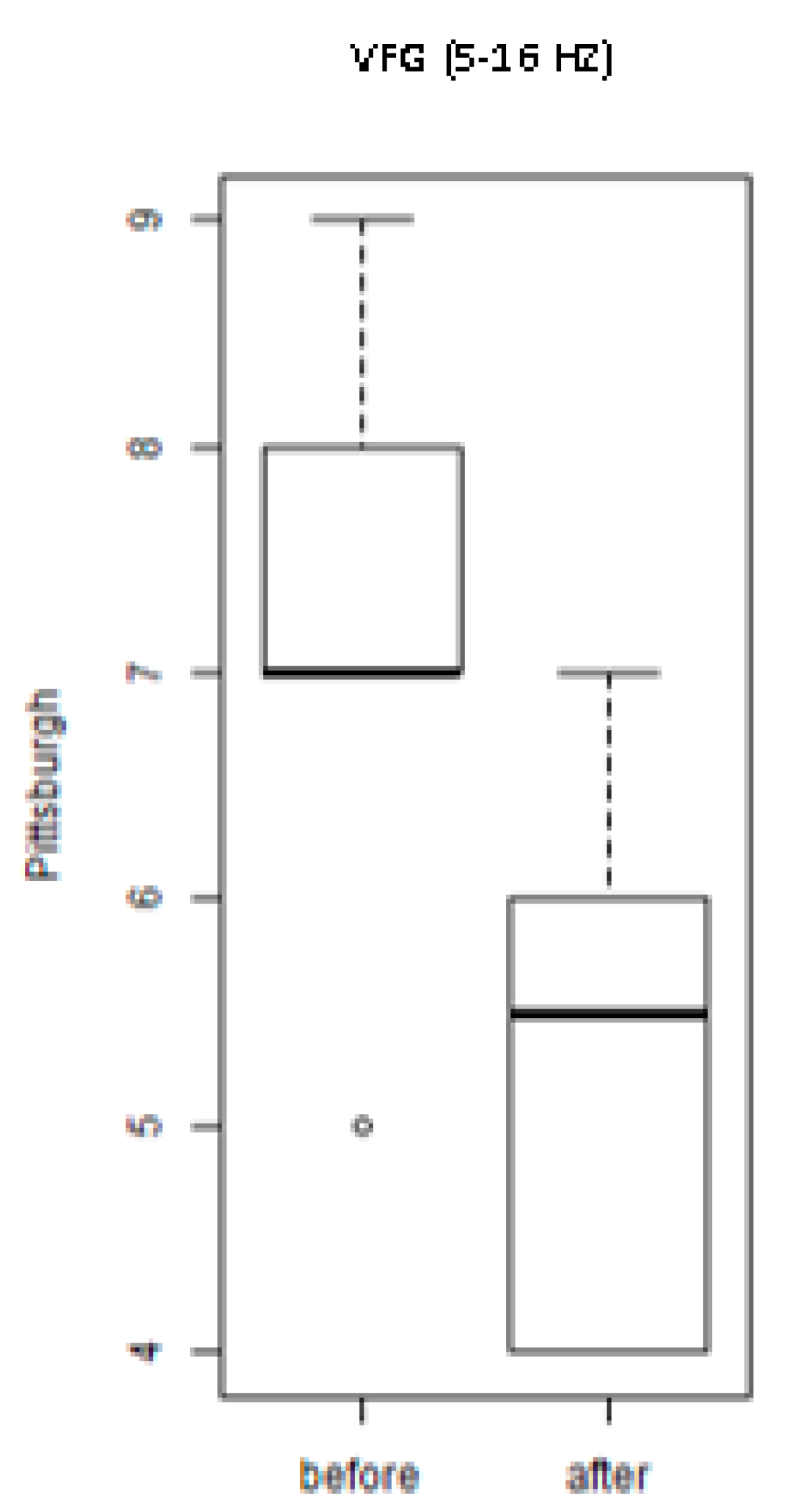
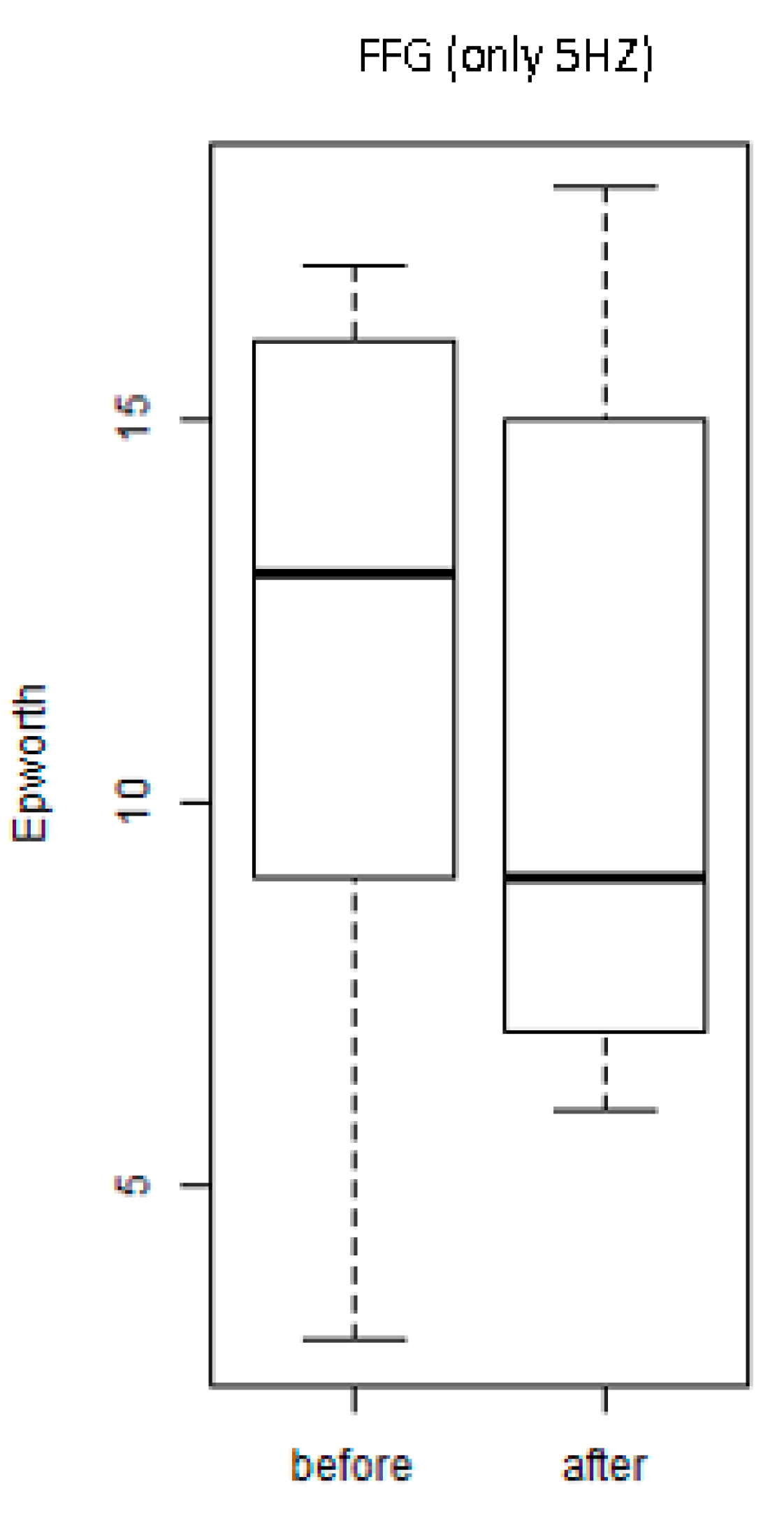
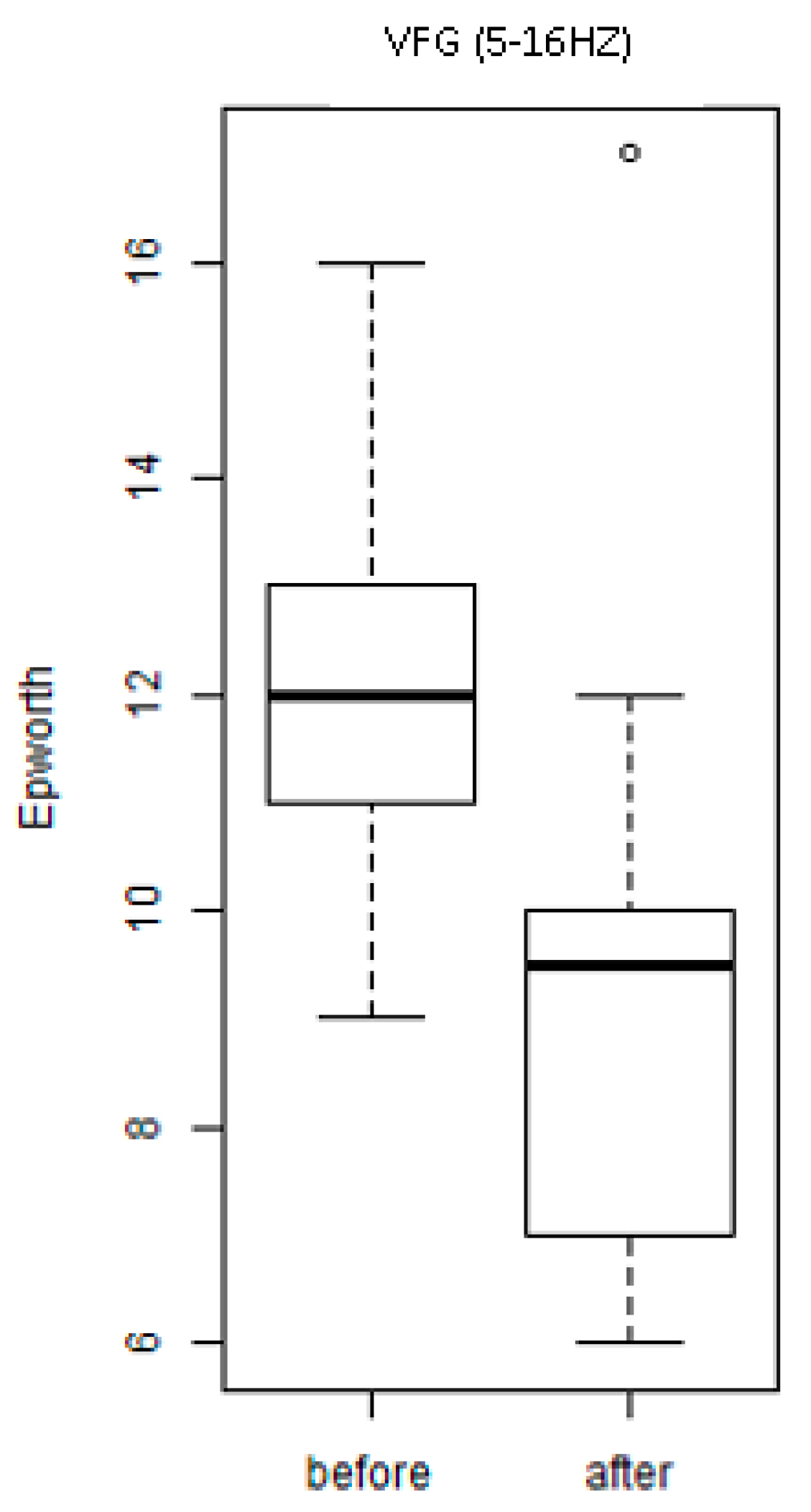
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kaur, J. Retracted: A Comprehensive Review on Metabolic Syndrome. Cardiol. Res. Pract. 2019, 2019, 4301528. [Google Scholar]
- Seaman, D.R.; Palombo, A.D. An Overview of the Identification and Management of the Metabolic Syndrome in Chiropractic Practice. J. Chiropr. Med. 2014, 13, 210–219. [Google Scholar] [CrossRef]
- Dominguez, L.J.; Barbagallo, M. The Biology of the Metabolic Syndrome and Aging:2006. Curr. Opin. Clin. Nutr. & Metab. Care 2016, 9, 5–11. [Google Scholar]
- Alberti, K.G.M.M.; Zimmet, P.; Shaw, J. Metabolic syndrome-a new world-wide definition. A Consensus Statement from the International Diabetes Federation. Diabet. Med. 2006, 23, 469–480. [Google Scholar] [CrossRef]
- Lian, Y.; Yuan, Q.; Wang, G.; Tang, F. Association between sleep quality and metabolic syndrome: A systematic review and meta-analysis. Psychiatry Res. 2019, 274, 66–74. [Google Scholar] [CrossRef]
- Massar, S.A.A.; Liu, J.C.J.; Mohammad, N.B.; Chee, M.W.L. Poor habitual sleep efficiency is associated with increased cardiovascular and cortisol stress reactivity in men. Psychoneuroendocrinology 2017, 81, 151–156. [Google Scholar] [CrossRef]
- Tsai, H.-J.; Kuo, T.B.J.; Lin, Y.-C.; Yang, C.C.H. The association between prolonged sleep onset latency and heart rate dynamics among young sleep-onset insomniacs and good sleepers. Psychiatry Res. 2015, 230, 892–898. [Google Scholar] [CrossRef]
- Salo, P.; Sivertsen, B.; Oksanen, T.; Sjösten, N.; Pentti, J.; Virtanen, M.; Kivimäki, M.; Vahtera, J. Insomnia symptoms as a predictor of incident treatment for depression: Prospective cohort study of 40,791 men and women. Sleep Med. 2012, 13, 278–284. [Google Scholar] [CrossRef]
- Wang, S.; Wu, Y.; Ungvari, G.S.; Ng, C.H.; Forester, B.P.; Gatchel, J.R.; Chiu, H.F.K.; Kou, C.; Fu, Y.; Qi, Y.; et al. Sleep duration and its association with demographics, lifestyle factors, poor mental health and chronic diseases in older Chinese adults. Psychiatry Res. 2017, 257, 212–218. [Google Scholar] [CrossRef]
- Iftikhar, I.H.; Donley, M.A.; Mindel, J.; Pleister, A.; Soriano, S.; Magalang, U.J. Sleep Duration and Metabolic Syndrome. An Updated Dose–Risk Metaanalysis. Ann. Am. Thorac. Soc. 2015, 12, 1364–1372. [Google Scholar] [CrossRef]
- Stamatakis, K.A.; Punjabi, N.M. Effects of Sleep Fragmentation on Glucose Metabolism in Normal Subjects. Chest 2010, 137, 95–101. [Google Scholar] [CrossRef]
- Mesas, A.E.; Guallar-Castillón, P.; López-García, E.; León-Muñoz, L.M.; Graciani, A.; Banegas, J.R.; Rodríguez-Artalejo, F. Sleep quality and the metabolic syndrome: The role of sleep duration and lifestyle. Diabetes. Metab. Res. Rev. 2014, 30, 222–231. [Google Scholar] [CrossRef]
- Resta, O.; Foschino-Barbaro, M.P.; Legari, G.; Talamo, S.; Bonfitto, P.; Palumbo, A.; Minenna, A.; Giorgino, R.; De Pergola, G. Sleep-related breathing disorders, loud snoring and excessive daytime sleepiness in obese subjects. Int. J. Obes. 2001, 25, 669–675. [Google Scholar] [CrossRef]
- Drager, L.F.; Lopes, H.F.; Maki-Nunes, C.; Trombetta, I.C.; Toschi-Dias, E.; Alves, M.J.N.N.; Fraga, R.F.; Jun, J.C.; Negrão, C.E.; Krieger, E.M.; et al. The Impact of Obstructive Sleep Apnea on Metabolic and Inflammatory Markers in Consecutive Patients with Metabolic Syndrome. PLoS ONE 2010, 5, e12065. [Google Scholar] [CrossRef]
- Kline, C.E.; Crowley, E.P.; Ewing, G.B.; Burch, J.B.; Blair, S.N.; Durstine, J.L.; Davis, J.M.; Youngstedt, S.D. The Effect of Exercise Training on Obstructive Sleep Apnea and Sleep Quality: A Randomized Controlled Trial. Sleep 2011, 34, 1631–1640. [Google Scholar] [CrossRef]
- Soler, X.; Liao, S.-Y.; Marin, J.M.; Lorenzi-Filho, G.; Jen, R.; DeYoung, P.; Owens, R.L.; Ries, A.L.; Malhotra, A. Age, gender, neck circumference, and Epworth sleepiness scale do not predict obstructive sleep apnea (OSA) in moderate to severe chronic obstructive pulmonary disease (COPD): The challenge to predict OSA in advanced COPD. PLoS ONE 2017, 12, e0177289. [Google Scholar] [CrossRef]
- Qu, X.-X.; Esangbedo, I.C.; Zhang, X.-J.; Liu, S.-J.; Li, L.-X.; Gao, S.; Li, M. Obstructive Sleep Apnea Syndrome is Associated with Metabolic Syndrome among Adolescents and Youth in Beijing. Chin. Med. J. 2015, 128, 2278–2283. [Google Scholar] [CrossRef]
- Hoffstein, V.; Mateika, S. Differences in abdominal and neck circumferences in patients with and without obstructive sleep apnoea. Eur. Respir. J. 1992, 5, 377–381. [Google Scholar]
- Horner, R.L.; Shea, S.A.; McIvor, J.; Guz, A. Pharyngeal size and shape during wakefulness and sleep in patients with obstructive sleep apnoea. QJM 1989, 72, 719–735. [Google Scholar]
- Paley, C.A.; Johnson, M.I. Abdominal obesity and metabolic syndrome: Exercise as medicine? BMC Sports Sci. Med. Rehabil. 2018, 10, 7. [Google Scholar] [CrossRef]
- Figueroa, A.; Kalfon, R.; Madzima, T.A.; Wong, A. Whole-body vibration exercise training reduces arterial stiffness in postmenopausal women with prehypertension and hypertension. Menopause 2014, 21, 131–136. [Google Scholar] [CrossRef]
- Wong, A.; Alvarez-Alvarado, S.; Kinsey, A.W.; Figueroa, A. Whole-Body Vibration Exercise Therapy Improves Cardiac Autonomic Function and Blood Pressure in Obese Pre- and Stage 1 Hypertensive Postmenopausal Women. J. Altern. Complement. Med. 2016, 22, 970–976. [Google Scholar] [CrossRef]
- Anwer, S.; Alghadir, A.; Zafar, H.; Al-Eisa, E. Effect of whole body vibration training on quadriceps muscle strength in individuals with knee osteoarthritis: A systematic review and meta-analysis. Physiotherapy 2016, 102, 145–151. [Google Scholar] [CrossRef]
- Alev, A.; Mihriban, A.; Bilge, E.; Ayça, E.; Merve, K.; Şeyma, C.; Uğur, E.; Adnan, B.; Zeynel, K.; Mahmut, G.S. Effects of whole body vibration therapy in pain, function and depression of the patients with fibromyalgia. Complement. Ther. Clin. Pract. 2017, 28, 200–203. [Google Scholar] [CrossRef]
- Zhou, J.; Pang, L.; Chen, N.; Wang, Z.; Wang, C.; Hai, Y.; Lyu, M.; Lai, H.; Lin, F. Whole-body vibration training-better care for COPD patients: A systematic review and meta-analysis. Int. J. COPD 2018, 13, 3243–3254. [Google Scholar] [CrossRef]
- Sá-Caputo, D.; Paineiras-Domingos, L.L.; Francisca-Santos, A.; dos Anjos, E.M.; Reis, A.S.; Neves, M.F.T.; Oigman, W.; Oliveira, R.; Brandão, A.; Machado, C.B.; et al. Whole-body vibration improves the functional parameters of individuals with metabolic syndrome: An exploratory study. BMC Endocr. Disord. 2019, 19, 6. [Google Scholar] [CrossRef]
- Braz, D.S.; de Andrade, A.D.; Teixeira, A.S.; Cavalcanti, C.A.; Morais, A.B.; Marinho, P.E.M. Whole-body vibration improves functional capacity and quality of life in patients with severe chronic obstructive pulmonary disease (COPD): A pilot study. Int. J. COPD 2015, 10, 125–132. [Google Scholar]
- Rønnestad, B.R. Comparing the Performance-Enhancing Effects of Squats on a Vibration Platform With Conventional Squats in Recreationally Resistance-Trained Men. J. Strength Cond. Res. 2004, 18, 839–845. [Google Scholar]
- Merkert, J.; Butz, S.; Nieczaj, R.; Steinhagen-Thiessen, E.; Eckardt, R. Combined whole body vibration and balance training using Vibrosphere®. Z. für Gerontol. und Geriatr. 2011, 44, 256–261. [Google Scholar] [CrossRef]
- Cardim, A.B.; Marinho, P.E.; Nascimento, J.F.; Fuzari, H.K.; de Andrade, A.D. Does Whole-Body Vibration Improve the Functional Exercise Capacity of Subjects With COPD? A Meta-Analysis. Respir. Care 2016, 61, 1552–1559. [Google Scholar] [CrossRef]
- Sá-Caputo, D.C.; Paineiras-Domingos, L.L.; Oliveira, R.; Neves, M.F.T.; Brandão, A.; Marin, P.J.; Sañudo, B.; Furness, T.; Taiar, R.; Bernardo-Filho, M. Acute Effects of Whole-Body Vibration on the Pain Level, Flexibility, and Cardiovascular Responses in Individuals With Metabolic Syndrome. Dose-Response 2018, 16, 155932581880213. [Google Scholar] [CrossRef]
- Paineiras-Domingos, L.L.; da Cunha Sá-Caputo, D.; Reis, A.S.; Francisca Santos, A.; Sousa-Gonçalves, C.R.; dos Anjos, E.M.; dos Santos Pereira, M.J.; Sartorio, A.; Bernardo-Filho, M. Assessment Through the Short Physical Performance Battery of the Functionality in Individuals With Metabolic Syndrome Exposed to Whole-Body Vibration Exercises. Dose-Response 2018, 16, 155932581879453. [Google Scholar] [CrossRef]
- Moher, D.; Hopewell, S.; Schulz, K.F.; Montori, V.; Gøtzsche, P.C.; Devereaux, P.J.; Elbourne, D.; Egger, M.; Altman, D.G. CONSORT 2010 explanation and elaboration: Updated guidelines for reporting parallel group randomised trials. Int. J. Surg. 2012, 10, 28–55. [Google Scholar] [CrossRef]
- Casadei, K.; Kiel., J. Anthropometric Measurement; StatPearls Publishing LLC: Treasure Island, FL, USA, 2019. Available online: https://www.ncbi.nlm.nih.gov/books/NBK537315/ (accessed on 24 October 2019).
- Gasa, M.; López-Padrós, C.; Monasterio, C.; Salord, N.; Mayos, M.; Vilarrasa, N.; Fernandez-Aranda, F.; Montserrat, J.M.; Dorca, J.; López-Padrós, C.; et al. Anthropometrical phenotypes are important when explaining obstructive sleep apnea in female bariatric cohorts. J. Sleep Res. 2019, 28, e12830. [Google Scholar] [CrossRef]
- De Cardiologia, S.B. III Diretrizes Brasileiras Sobre Dislipidemias e Diretriz de Prevenção da Aterosclerose do Departamento de Aterosclerose da Sociedade Brasileira de Cardiologia. Arq. Bras. Cardiol. 2001, 77, 48. [Google Scholar]
- Kim, T.Y.; You, S.E.; Ko, Y.S. Association between Sasang constitutional types with obesity factors and sleep quality. Integr. Med. Res. 2018, 7, 341–350. [Google Scholar] [CrossRef]
- Schwab, R.J.; Pasirstein, M.; Pierson, R.; Mackley, A.; Hachadoorian, R.; Arens, R.; Maislin, G.; Pack, A.I. Identification of Upper Airway Anatomic Risk Factors for Obstructive Sleep Apnea with Volumetric Magnetic Resonance Imaging. Am. J. Respir. Crit. Care Med. 2003, 168, 522–530. [Google Scholar] [CrossRef]
- Duarte, R.L.; Mello, F.C.; Magalhães-da-Silveira, F.J.; Oliveira-e-Sá, T.S.; Rabahi, M.F.; Gozal, D. Comparative performance of screening instruments for obstructive sleep apnea in morbidly obese patients referred to a sleep laboratory: A prospective cross-sectional study. Sleep Breath. 2019, 23, 1123–1132. [Google Scholar] [CrossRef]
- Netzer, N.C.; Stoohs, R.A.; Netzer, C.M.; Clark, K.; Strohl, K.P. Using the Berlin Questionnaire to Identify Patients at Risk for the Sleep Apnea Syndrome. Ann. Intern. Med. 1999, 131, 485. [Google Scholar] [CrossRef]
- Ng, S.S.; Tam, W.; Chan, T.O.; To, K.W.; Ngai, J.; Chan, K.K.P.; Yip, W.H.; Lo, R.L.; Yiu, K.; Ko, F.W.; et al. Use of Berlin questionnaire in comparison to polysomnography and home sleep study in patients with obstructive sleep apnea. Respir. Res. 2019, 20, 40. [Google Scholar] [CrossRef]
- Cizza, G.; de Jonge, L.; Piaggi, P.; Mattingly, M.; Zhao, X.; Lucassen, E.; Rother, K.I.; Sumner, A.E.; Csako, G. Neck Circumference Is a Predictor of Metabolic Syndrome and Obstructive Sleep Apnea in Short-Sleeping Obese Men and Women. Metab. Syndr. Relat. Disord. 2014, 12, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Team, R. R Development Core Team. R A Lang. Environ. Stat. Comput. 2013, 3, 201. [Google Scholar]
- Yoshida, K.; Bohn, J.; Yoshida, M.K. Package ‘Tableone’; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Rao, D.P.; Orpana, H.; Krewski, D. Physical activity and non-movement behaviours: Their independent and combined associations with metabolic syndrome. Int. J. Behav. Nutr. Phys. Act. 2016, 13, 26. [Google Scholar] [CrossRef] [PubMed]
- Feairheller, D.L.; Diaz, K.M.; Kashem, M.A.; Thakkar, S.R.; Veerabhadrappa, P.; Sturgeon, K.M.; Ling, C.; Williamson, S.T.; Kretzschmar, J.; Lee, H.; et al. Effects of Moderate Aerobic Exercise Training on Vascular Health and Blood Pressure in African Americans. J. Clin. Hypertens. 2014, 16, 504–510. [Google Scholar] [CrossRef]
- Mahajan, A.; Mishra, T.; Singh, N.; Jain, A. Heart rate and blood pressure response to exercise and recovery in subclinical hypothyroid patients. Int. J. Appl. Basic Med. Res. 2013, 3, 106. [Google Scholar] [CrossRef]
- Kang, S.J.; Ha, G.C.; Ko, K.J. Association between resting heart rate, metabolic syndrome and cardiorespiratory fitness in Korean male adults. J. Exerc. Sci. Fit. 2017, 15, 27–31. [Google Scholar] [CrossRef]
- Sîrbu, E.; Buzaș, R.; Mihăescu, R.; Suceava, I.; Lighezan, D. Influence of exercise training and eating behavior on arterial stiffness in young healthy students. Wien. Klin. Wochenschr. 2015, 127, 555–560. [Google Scholar] [CrossRef]
- King, N.A.; Hopkins, M.; Caudwell, P.; Stubbs, R.J.; Blundell, J.E. Beneficial effects of exercise: Shifting the focus from body weight to other markers of health. Br. J. Sports Med. 2009, 43, 924–927. [Google Scholar] [CrossRef]
- Souza, A.K.F.; de Andrade, A.D.; de Medeiros, A.I.C.; de Aguiar, M.I.R.; de Souza Rocha, T.D.; Pedrosa, R.P.; de Lima, A.M.J. Effectiveness of inspiratory muscle training on sleep and functional capacity to exercise in obstructive sleep apnea: A randomized controlled trial. Sleep Breath. 2018, 22, 631–639. [Google Scholar] [CrossRef]
- Reid, K.J.; Baron, K.G.; Lu, B.; Naylor, E.; Wolfe, L.; Zee, P.C. Aerobic exercise improves self-reported sleep and quality of life in older adults with insomnia. Sleep Med. 2010, 11, 934–940. [Google Scholar] [CrossRef]
- Brandão, G.S.; Gomes, G.S.B.F.; Brandão, G.S.; Callou Sampaio, A.A.; Donner, C.F.; Oliveira, L.V.F.; Camelier, A.A. Home exercise improves the quality of sleep and daytime sleepiness of elderlies: A randomized controlled trial. Multidiscip. Respir. Med. 2018, 13, 2. [Google Scholar] [CrossRef] [PubMed]
- Kline, C.E.; Ewing, G.B.; Burch, J.B.; Blair, S.N.; Durstine, J.L.; Davis, J.M.; Youngstedt, S.D. Exercise Training Improves Selected Aspects of Daytime Functioning in Adults with Obstructive Sleep Apnea. J. Clin. Sleep Med. 2012, 8, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz Gokmen, G.; Akkoyunlu, M.E.; Kilic, L.; Algun, C. The Effect of T’ai Chi and Qigong Training on Patients with Obstructive Sleep Apnea: A Randomized Controlled Study. J. Altern. Complement. Med. 2019, 25, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Itoh, H.; Yokoyama, K.; Matsukawa, T.; Kitamura, F. Association between physical activity and sleep-disordered breathing in male Japanese workers: A cross-sectional study. BMC Res. Notes 2017, 10, 37. [Google Scholar] [CrossRef]
- Dixit, S.; Maiya, A.; Shastry, B. Effect of aerobic exercise on quality of life in population with diabetic peripheral neuropathy in type 2 diabetes: A single blind, randomized controlled trial. Qual. Life Res. 2014, 23, 1629–1640. [Google Scholar] [CrossRef]
- Farias, D.L.; Tibana, R.A.; Teixeira, T.G.; Vieira, D.C.L.; Tarja, V.; Nascimento, D.D.C.; Silva, A.D.O.; Funghetto, S.S.; Coura, M.A.D.S.; Valduga, R.; et al. Elderly women with metabolic syndrome present higher cardiovascular risk and lower relative muscle strength. Einstein 2013, 11, 174–179. [Google Scholar] [CrossRef][Green Version]
- World Health Organization. GLOBAL STATUS REPORT on Noncommunicable Diseases 2014—“Attaining the Nine Global Noncommunicable Diseases Targets; A Shared Responsibility”; WHO Libr. Cat. Data: Geneva, Switzerland, 2014; p. 298. [Google Scholar]
- Callaghan, B.C.; Xia, R.; Reynolds, E.; Banerjee, M.; Rothberg, A.E.; Burant, C.F.; Villegas-Umana, E.; Pop-Busui, R.; Feldman, E.L. Association Between Metabolic Syndrome Components and Polyneuropathy in an Obese Population. JAMA Neurol. 2016, 73, 1468. [Google Scholar] [CrossRef]
- Sossa, C.; Delisle, H.; Agueh, V.; Sodjinou, R.; Ntandou, G.; Makoutodé, M. Lifestyle and dietary factors associated with the evolution of cardiometabolic risk over four years in West-African adults: The Benin study. J. Obes. 2013, 2013, 298024. [Google Scholar] [CrossRef]


| FFG before (Median [IQR]) | FFG after (Median [IQR]) | p | VFG before (Median [IQR]) | VFG after (Median [IQR]) | p | |
|---|---|---|---|---|---|---|
| BMI (kg/m2) | 29.50 [29.0, 34.20] | 29.60 [28.90, 34.10] | 0.54 | 35.40 [32.40, 36.60] | 34.70 [32.88, 36.08] | 0.23 |
| WC (cm) | 105.00 [101.20, 106.30] | 101.00 [100.00, 105.00] | 0.23 | 108.90 [103.75, 116.10] | 107.00 [102.17, 112.85] | 0.02 |
| NC (cm) | 38.00 [36.40, 38.50] | 36.60 [35.60, 39.50] | 0.29 | 38.00 [37.58, 38.88] | 38.30 [37.17, 39.00] | 0.25 |
| SBP (mm Hg) | 129.00 [128.00, 130.00] | 127.00 [121.00, 128.00] | 0.12 | 130.00 [122.50, 132.50] | 130.00 [120.00, 132.00] | 0.76 |
| DBP (mm Hg) | 78.00 [75.00, 83.00] | 75.00 [68.00, 87.00] | 1.00 | 74.00 [72.25, 75.75] | 70.00 [60.75, 74.75] | 0.18 |
| HR (bpm) | 57.00 [56.00, 62.00] | 62.00 [60.00, 66.00] | 0.05 | 63.50 [59.75, 66.00] | 72.00 [61.25, 83.00] | 0.05 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Figueiredo Azeredo, C.; de Castro de Paiva, P.; Azeredo, L.; Reis da Silva, A.; Francisca-Santos, A.; Liane Paineiras-Domingos, L.; Lírio Pereira da Silva, A.; Leite Bernardes-Oliveira, C.; Pessanha-Freitas, J.; Moura-Fernandes, M.C.; et al. Effects of Whole-Body Vibration Exercises on Parameters Related to the Sleep Quality in Metabolic Syndrome Individuals: A Clinical Trial Study. Appl. Sci. 2019, 9, 5183. https://doi.org/10.3390/app9235183
Figueiredo Azeredo C, de Castro de Paiva P, Azeredo L, Reis da Silva A, Francisca-Santos A, Liane Paineiras-Domingos L, Lírio Pereira da Silva A, Leite Bernardes-Oliveira C, Pessanha-Freitas J, Moura-Fernandes MC, et al. Effects of Whole-Body Vibration Exercises on Parameters Related to the Sleep Quality in Metabolic Syndrome Individuals: A Clinical Trial Study. Applied Sciences. 2019; 9(23):5183. https://doi.org/10.3390/app9235183
Chicago/Turabian StyleFigueiredo Azeredo, Claudia, Patrícia de Castro de Paiva, Leandro Azeredo, Aline Reis da Silva, Arlete Francisca-Santos, Laisa Liane Paineiras-Domingos, Adriana Lírio Pereira da Silva, Camila Leite Bernardes-Oliveira, Juliana Pessanha-Freitas, Márcia Cristina Moura-Fernandes, and et al. 2019. "Effects of Whole-Body Vibration Exercises on Parameters Related to the Sleep Quality in Metabolic Syndrome Individuals: A Clinical Trial Study" Applied Sciences 9, no. 23: 5183. https://doi.org/10.3390/app9235183
APA StyleFigueiredo Azeredo, C., de Castro de Paiva, P., Azeredo, L., Reis da Silva, A., Francisca-Santos, A., Liane Paineiras-Domingos, L., Lírio Pereira da Silva, A., Leite Bernardes-Oliveira, C., Pessanha-Freitas, J., Moura-Fernandes, M. C., Guimarães Mendonça, R., Alexandre Bachur, J., Teixeira-Silva, Y., Moreira-Marconi, E., de Oliveira Guedes-Aguiar, E., Bessa Monteiro de Oliveira, B., Fritsch Neves, M., Felipe Ferreira-Souza, L., Layter Xavier, V., ... da Cunha de Sá-Caputo, D. (2019). Effects of Whole-Body Vibration Exercises on Parameters Related to the Sleep Quality in Metabolic Syndrome Individuals: A Clinical Trial Study. Applied Sciences, 9(23), 5183. https://doi.org/10.3390/app9235183