Colorimetric Technique for Antimony Detection Based on the Use of Gold Nanoparticles Conjugated with Poly-A Oligonucleotide
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Instruments and Measurements
2.3. Preparation of Gold Nanoparticles
2.4. Preparation of poly-A Oligonucleotide Immobilized GNPs
2.5. Characterization of Native Gold Nanoparticles and Poly-A Oligonucleotide Immobilized GNPs
2.6. Colorimetric Assay of Antimony
3. Results and Discussion
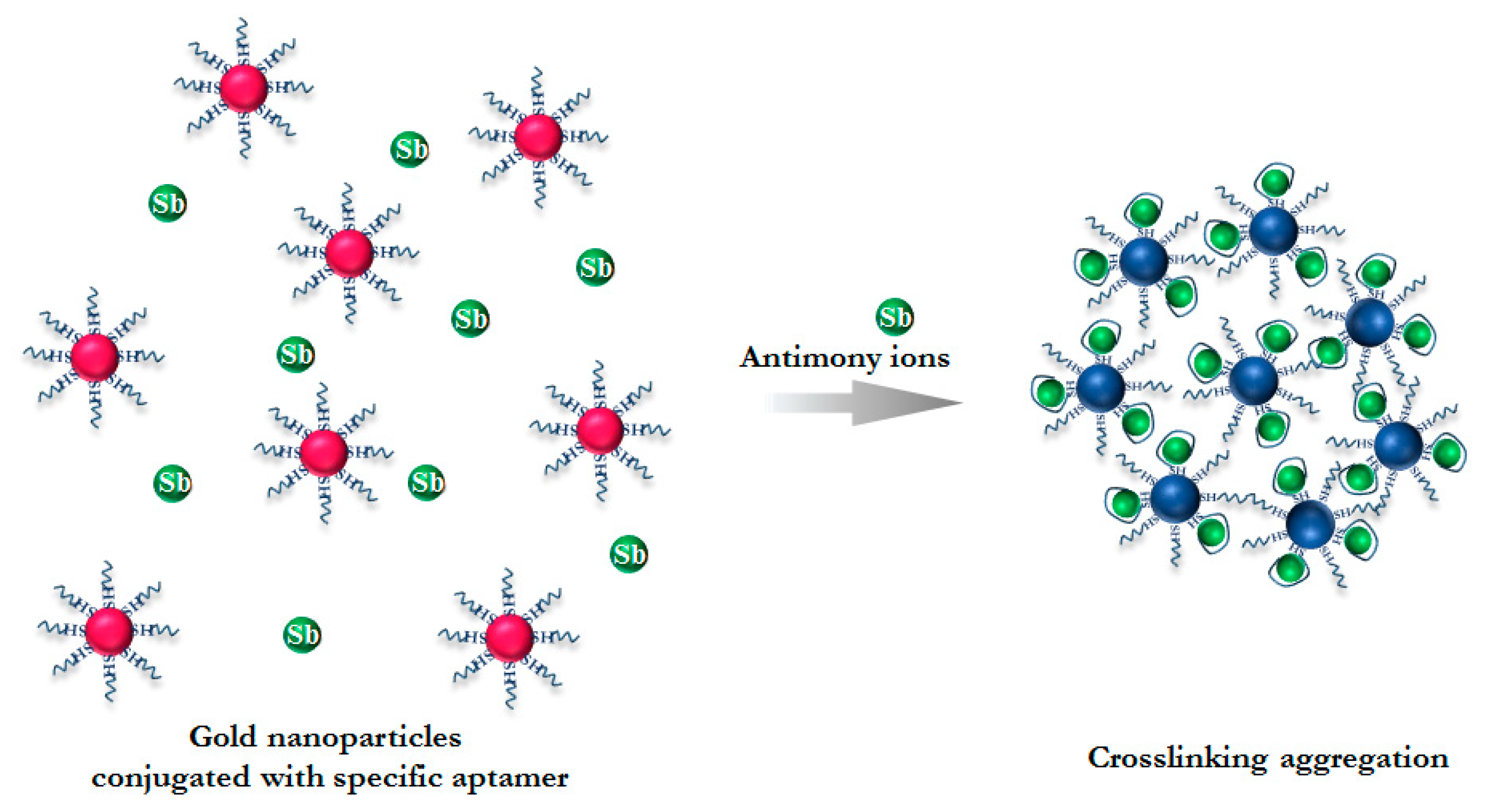
3.1. Principle of Antimony Detection
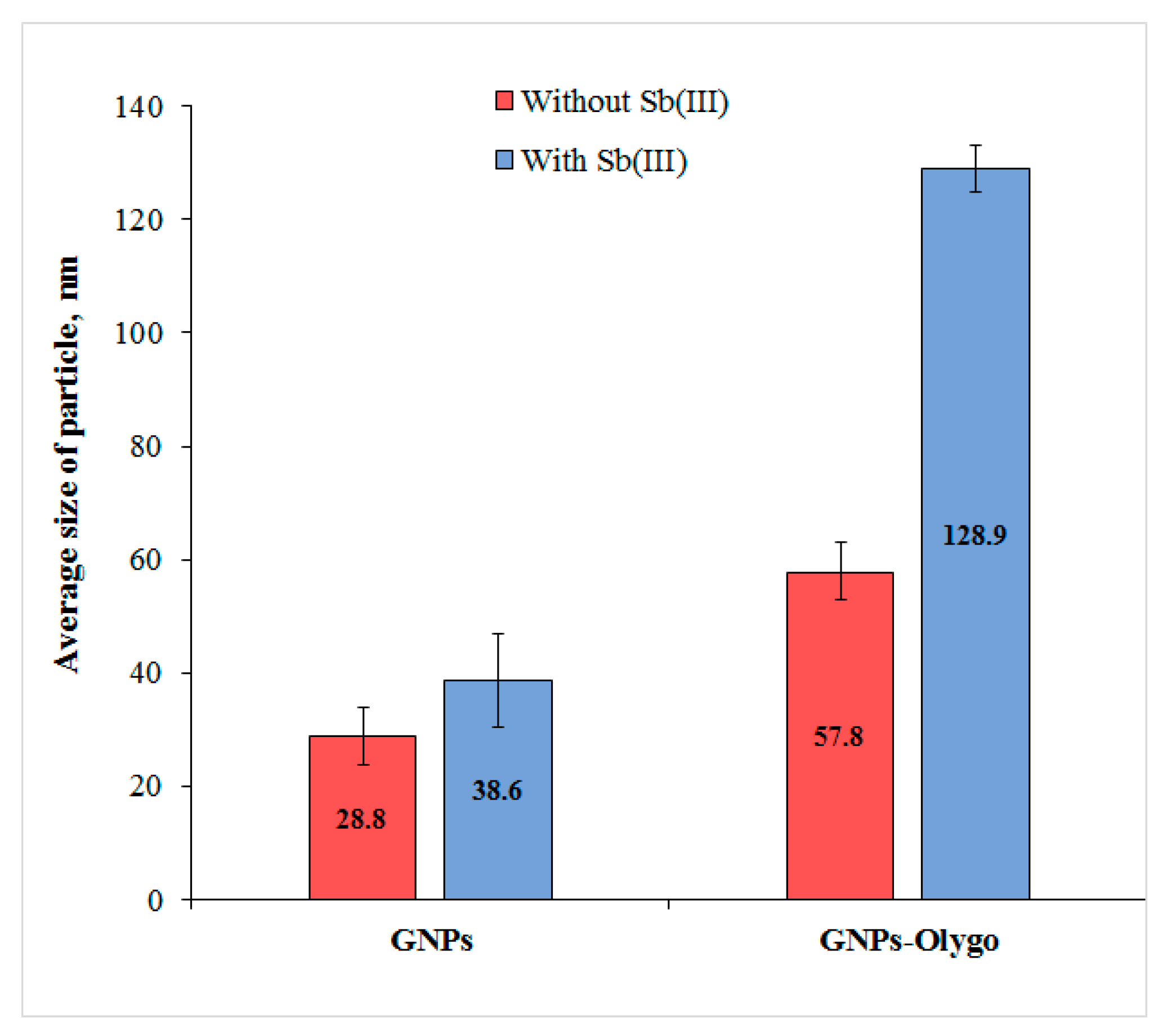
3.2. Characterization of PAO–GNPs Conjugate
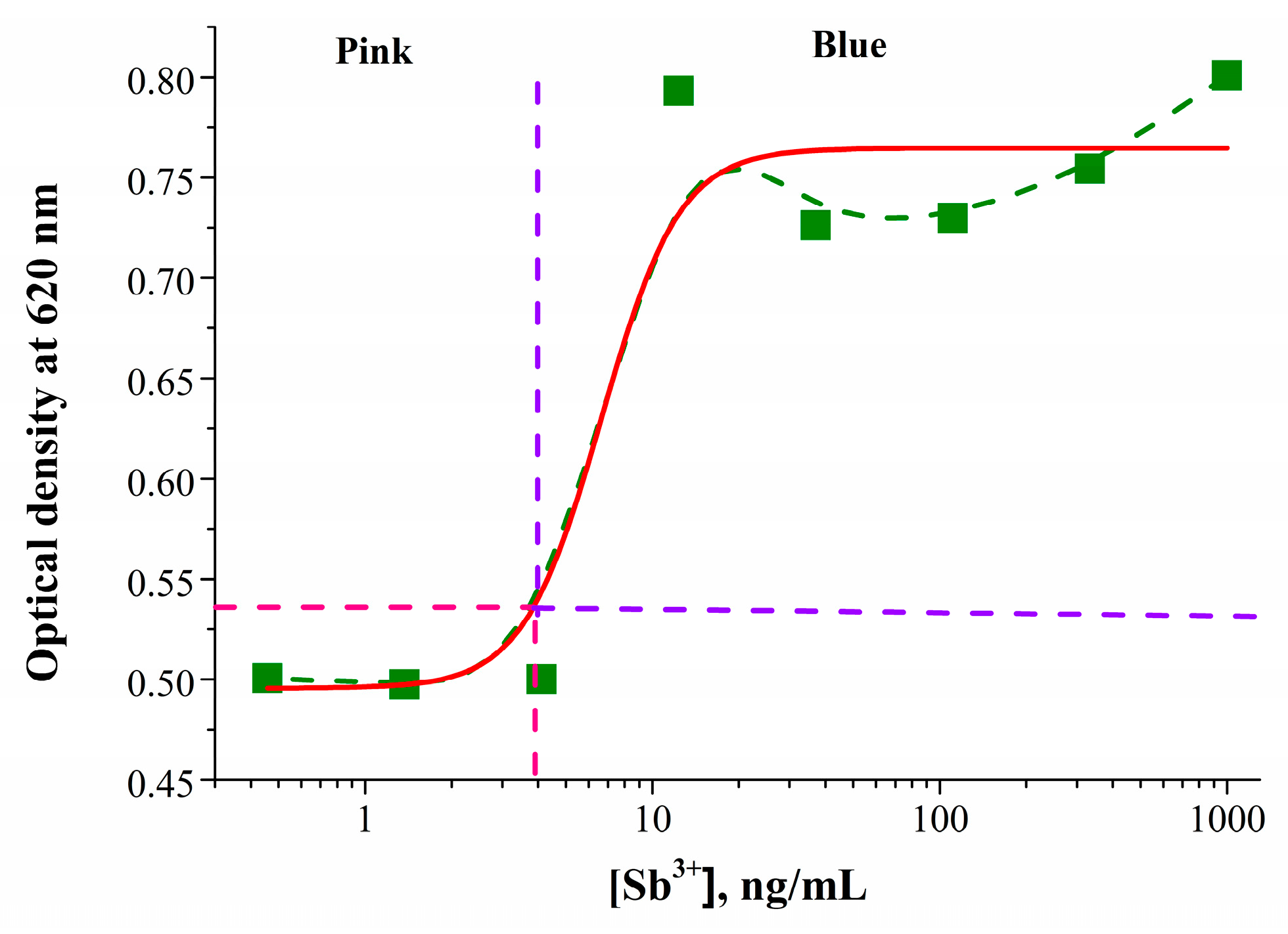
3.3. Colorimetric Detection of Antimony (III)
3.4. Selectivity Test
3.5. Real Sample Analysis
3.6. Estimation of Analytical Characteristics
3.7. The Discussion of the Interaction Mechanism of Antimony (III) with poly-A Modified Gold Nanoparticles
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sundar, S.; Chakravarty, J. Antimony toxicity. Int. J. Environ. Res. Public Health 2010, 7, 4267–4277. [Google Scholar] [CrossRef] [PubMed]
- Herath, I.; Vithanage, M.; Bundschuh, J. Antimony as a global dilemma: Geochemistry, mobility, fate and transport. Environ. Pollut. 2017, 223, 545–559. [Google Scholar] [CrossRef] [PubMed]
- Filella, M. Antimony interactions with heterogeneous complexants in waters, sediments and soils: A review of data obtained in bulk samples. Earth-Sci. Rev. 2011, 107, 325–341. [Google Scholar] [CrossRef]
- He, M.; Wang, N.; Long, X.; Zhang, C.; Ma, C.; Zhong, Q.; Wang, A.; Wang, Y.; Pervaiz, A.; Shan, J. Antimony speciation in the environment: Recent advances in understanding the biogeochemical processes and ecological effects. J. Environ. Sci. 2019, 75, 14–39. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.C.; Lockwood, P.V.; Ashley, P.M.; Tighe, M. The chemistry and behavior of antimony in the soil environment with comparisons to arsenic: A critical review. Environ. Pollut. 2010, 158, 1169–1181. [Google Scholar] [CrossRef] [PubMed]
- Council of the European Communities. Council Directive Relating to the Quality of Water Intended for Human Consumption (80/778/EEC). Off. J. L 1980, 229, 11–29. [Google Scholar]
- Sanz, J.; Gallarta, F.; Galban, J.; Castillo, J.R. Antimony determination by hydride generation—UV-visible molecular absorption spectrophotometry with diode-array detection. Fresenius’ Z. Anal. Chem. 1988, 330, 510–515. [Google Scholar] [CrossRef]
- De Gregori, I.; Pinochet, H.; Fuentes, E.; Potin-Gautier, M. Determination of antimony in soils and vegetables by hydride generation atomic fluorescence spectrometry and electrothermal atomic absorption spectrometry. Optimization and comparison of both analytical techniques. J. Anal. Atomic Spectrom. 2001, 16, 172–178. [Google Scholar] [CrossRef]
- Gunduz, S.; Akman, S.; Baysal, A.; Kahraman, M. The use of silver nanoparticles as an effective modifier for the determination of arsenic and antimony by electrothermal atomic absorption spectrometry. Spectrochim. Acta B 2010, 65, 297–300. [Google Scholar] [CrossRef]
- Gunduz, S.; Akman, S.; Baysal, A.; Culha, M. The use of gold nanoparticles as an effective modifier for the determination of arsenic and antimony by electrothermal atomic absorption spectrometry. Microchim. Acta 2011, 172, 403–407. [Google Scholar] [CrossRef]
- Serrano, N.; Díaz-Cruz, J.M.; Ariño, C.; Esteban, M. Antimony-based electrodes for analytical determinations. TrAC Trends Anal. Chem. 2016, 77, 203–213. [Google Scholar] [CrossRef]
- Renedo, O.D.; González, M.J.G.; Martínez, M.J.A. Determination of antimony (III) in real samples by anodic stripping voltammetry using a mercury film screen-printed electrode. Sensors 2009, 9, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Semenova, N.V.; Leal, L.O.; Forteza, R.; Cerdà, V. Antimony determination and speciation by multisyringe flow injection analysis with hydride generation-atomic fluorescence detection. Anal. Chim. Acta 2005, 530, 113–120. [Google Scholar] [CrossRef]
- Abbaspour, A.; Baramakeh, L. Simultaneous determination of antimony and bismuth by beta-correction spectrophotometry and an artificial neural network algorithm. Talanta 2005, 65, 692–699. [Google Scholar] [CrossRef] [PubMed]
- González, M.J.G.; Renedo, O.D.; Martínez, M.J.A. Simultaneous determination of antimony (III) and antimony (V) by UV–vis spectroscopy and partial least squares method (PLS). Talanta 2005, 68, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Rath, S.; Jardim, W.F.; Dórea, J.G. A simple spectrophotometric procedure for the determination of antimony (III) and (V) in antileishmanial drugs. Fresenius’ J. Anal. Chem. 1997, 358, 548–550. [Google Scholar] [CrossRef]
- Tighe, M.; Edwards, M.M.; Cluley, G.; Lisle, L.; Wilson, S.C. Colorimetrically determining total antimony in contaminated waters and screening for antimony speciation. J. Hydrol. 2018, 563, 84–91. [Google Scholar] [CrossRef]
- Lintschinger, J.; Koch, I.; Serves, S.; Feldmann, J.; Cullen, W.R. Determination of antimony species with high-performance liquid chromatography using element specific detection. Fresenius’ J. Anal. Chem. 1997, 359, 484–491. [Google Scholar] [CrossRef]
- Fogg, A.G.; Burgess, C.; Burns, D.T. Use of basic dyes in the determination of anions, particularly as a means of determining antimony, thallium, and gallium. Talanta 1971, 18, 1175–1196. [Google Scholar] [CrossRef]
- Hu, M.; Chen, J.; Li, Z.Y.; Au, L.; Hartland, G.V.; Li, X.; Marquez, M.; Xia, Y. Gold nanostructures: Engineering their plasmonic properties for biomedical applications. Chem. Soc. Rev. 2006, 35, 1084–1094. [Google Scholar] [CrossRef] [PubMed]
- Berlina, A.N.; Zherdev, A.V.; Pridvorova, S.M.; Gaur, M.S.; Dzantiev, B.B. Rapid visual detection of lead and mercury via enhanced crosslinking aggregation of aptamer-labeled gold nanoparticles. J. Nanosci. Nanotechnol. 2019, 19, 5489–5495. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Zhu, B.; Chen, X. Urine for plasmonic nanoparticle-based colorimetric detection of mercury Ion. Small 2013, 9, 4104–4111. [Google Scholar] [CrossRef] [PubMed]
- Ullah, N.; Mansha, M.; Khan, I.; Qurashi, A. Nanomaterial-based optical chemical sensors for the detection of heavy metals in water: Recent advances and challenges. TrAC Trends Anal. Chem. 2018, 100, 155–166. [Google Scholar] [CrossRef]
- Zhou, W.; Saran, R.; Liu, J. Metal sensing by DNA. Chem. Rev. 2017, 117, 8272–8325. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wang, X.; Cheng, X.; Yang, W.; Wu, Y.; Fu, F. Specifically and visually detect methyl-mercury and ethyl-mercury in fish sample based on DNA-templated alloy Ag–Au nanoparticles. Anal. Chem. 2018, 90, 5489–5495. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Liu, X.; Zhou, Y.; Luo, L.; Zhang, J.; Huang, A.; Mao, Q.; Chen, X.; Tang, L. Aptamer-based biosensors for detection of lead (ii) ion: A review. Anal. Meth. 2017, 9, 1976–1990. [Google Scholar] [CrossRef]
- Gan, Y.; Liang, T.; Hu, Q.; Zhong, L.; Wang, X.; Wan, H.; Wang, P. In-situ detection of cadmium with aptamer functionalized gold nanoparticles based on smartphone-based colorimetric system. Talanta 2020, 208, 120231. [Google Scholar] [CrossRef]
- Zhou, M.; Lin, T.; Gan, X. Colorimetric aggregation assay for silver(I) based on the use of aptamer modified gold nanoparticles and C-Ag(I)-C interaction. Microchim. Acta 2017, 184, 4671–4677. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, X.R.; Wen, S.H.; Liang, R.P.; Qiu, J.D. Optical sensors for inorganic arsenic detection. TrAC Trends Anal. Chem. 2019, 118, 869–879. [Google Scholar] [CrossRef]
- Huang, J.; Su, X.; Li, Z. Metal ion detection using functional nucleic acids and nanomaterials. Biosens. Bioelectron. 2017, 96, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Frens, G. Controlled nucleation for the regulation of the particle size in monodisperse gold suspensions. Nat. Phys. Sci 1973, 241, 20–22. [Google Scholar] [CrossRef]
- Tan, D.; He, Y.; Xing, X.; Zhao, Y.; Tang, H.; Pang, D. Aptamer functionalized gold nanoparticles based fluorescent probe for the detection of mercury (II) ion in aqueous solution. Talanta 2013, 113, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Liu, G.; Kai, M. DNA aptamers in the diagnosis and treatment of human diseases. Molecules (Basel) 2015, 20, 20979–20997. [Google Scholar] [CrossRef] [PubMed]
- Kreibig, U.; Genzel, L. Optical absorption of small metallic particles. Surf. Sci. 1985, 156, 678–700. [Google Scholar] [CrossRef]
- Michen, B.; Geers, C.; Vanhecke, D.; Endes, C.; Rothen-Rutishauser, B.; Balog, S.; Petri-Fink, A. Avoiding drying-artifacts in transmission electron microscopy: Characterizing the size and colloidal state of nanoparticles. Sci. Rep. 2015, 5, 9793. [Google Scholar] [CrossRef] [PubMed]
- Mirkin, C.A.; Letsinger, R.L.; Mucic, R.C.; Storhoff, J.J. A DNA-based method for rationally assembling nanoparticles into macroscopic materials. Nature 1996, 382, 607–609. [Google Scholar] [CrossRef] [PubMed]
- Gordon, C.K.L.; Wu, D.; Feagin, T.A.; Pusuluri, A.; Csordas, A.T.; Eisenstein, M.; Hawker, C.J.; Niu, J.; Soh, H.T. Click-PD: A quantitative method for base-modified aptamer discovery. ACS Chem. Biol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Hounslow, A. Water Quality Data: Analysis and Interpretation, 1st ed.; CRC Press: Boca Raton, FL, USA, 2018; p. 416. [Google Scholar]
- Hem, J.D. Study and Interpretation of the Chemical Characteristics of Natural Water, 3rd ed.; US Geological Survey: Charlottesville, VA, USA, 1985; p. 264.
- Berlina, A.N.; Zherdev, A.V.; Dzantiev, B.B. Progress in rapid optical assays for heavy metal ions based on the use of nanoparticles and receptor molecules. Microchim. Acta 2019, 186, 172. [Google Scholar] [CrossRef] [PubMed]
- WHO World Health Organization (WHO). Guidelines for Drinking-Water Quality, 4th ed.; WHO Library Cataloguing-in-Publication Data: Geneva, Switzerland, 2011. [Google Scholar]
- Hamilton, T.W.; Ellis, J.; Florence, T.M. Determination of arsenic and antimony in electrolytic copper by anodic stripping voltammetry at a gold film electrode. Anal. Chim. Acta 1980, 119, 225–233. [Google Scholar] [CrossRef]
- Renedo, O.D.; Martínez, M.J.A. A novel method for the anodic stripping voltammetry determination of Sb(III) using silver nanoparticle-modified screen-printed electrodes. Electrochem. Commun. 2007, 9, 820–826. [Google Scholar] [CrossRef]
- Renedo, O.D.; Martínez, M.J.A. Anodic stripping voltammetry of antimony using gold nanoparticle-modified carbon screen-printed electrodes. Anal. Chim. Acta 2007, 589, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Rojas, C.; Arancibia, V.; Gómez, M.; Nagles, E. High sensitivity adsorptive stripping voltammetric method for antimony(III) determination in the presence of quercetin-5′-sulfonic acid. Substituent effect on sensitivity. Sens. Actuators B 2013, 185, 560–567. [Google Scholar] [CrossRef]
- Unutkan, T.; Koyuncu, İ.; Diker, C.; Fırat, M.; Büyükpınar, Ç.; Bakırdere, S. Accurate and sensitive analytical strategy for the determination of antimony: Hydrogen assisted T-shaped slotted quartz tube-atom trap-flame atomic absorption spectrometry. Bull. Environ. Contam. Toxicol. 2019, 102, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Ge, S.; Zhang, C.; Zhu, Y.; Yu, J.; Zhang, S. BSA activated CdTe quantum dot nanosensor for antimony ion detection. Analyst 2010, 135, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Yang, C.; Yu, P.; Zheng, H.; Liu, Z.; Xing, Z.; Hu, S.H. Determination of antimony in water samples by hydride generation coupled with atmospheric pressure glow discharge atomic emission spectrometry. J. Anal. Atomic Spectrom. 2019, 34, 331–337. [Google Scholar] [CrossRef]
- Ong, Y.C.; Roy, S.; Andrews, P.C.; Gasser, G. Metal compounds against neglected tropical diseases. Chem. Rev. 2019, 119, 730–796. [Google Scholar] [CrossRef] [PubMed]
- Eddaikra, N.; Ait-Oudhia, K.; Kherrachi, I.; Oury, B.; Moulti-Mati, F.; Benikhlef, R.; Harrat, Z.; Sereno, D. Antimony susceptibility of Leishmania isolates collected over a 30-year period in Algeria. PLoS Negl. Trop. Dis. 2018, 12, e0006310. [Google Scholar] [CrossRef] [PubMed]
- Baiocco, P.; Colotti, G.; Franceschini, S.; Ilari, A. Molecular basis of antimony treatment in leishmaniasis. J. Med. Chem. 2009, 52, 2603–2612. [Google Scholar] [CrossRef] [PubMed]
- Krauth-Siegel, R.L.; Comini, M.A. Redox control in trypanosomatids, parasitic protozoa with trypanothione-based thiol metabolism. Biochim. Et Biophys. Acta Gen. Subj. 2008, 1780, 1236–1248. [Google Scholar] [CrossRef] [PubMed]
- Colotti, G.; Fiorillo, A.; Ilari, A. Metal-and metalloid-containing drugs for the treatment of trypanosomatid diseases. Front. Biosci. 2018, 23, 954–966. [Google Scholar]
- Demicheli, C.; Frézard, F.; Lecouvey, M.; Garnier-Suillerot, A. Antimony(V) complex formation with adenine nucleosides in aqueous solution. Biochim. Et Biophys. Acta Gen. Subj. 2002, 1570, 192–198. [Google Scholar] [CrossRef]
- Li, Y.; Liu, J.M.; Han, F.; Jiang, Y.; Yan, X.P. Probing interactions of antimony species with DNA by short column capillary electrophoresis coupled with inductively coupled plasma mass spectrometry. J. Anal. Atomic Spectrom. 2011, 26, 94–99. [Google Scholar] [CrossRef]

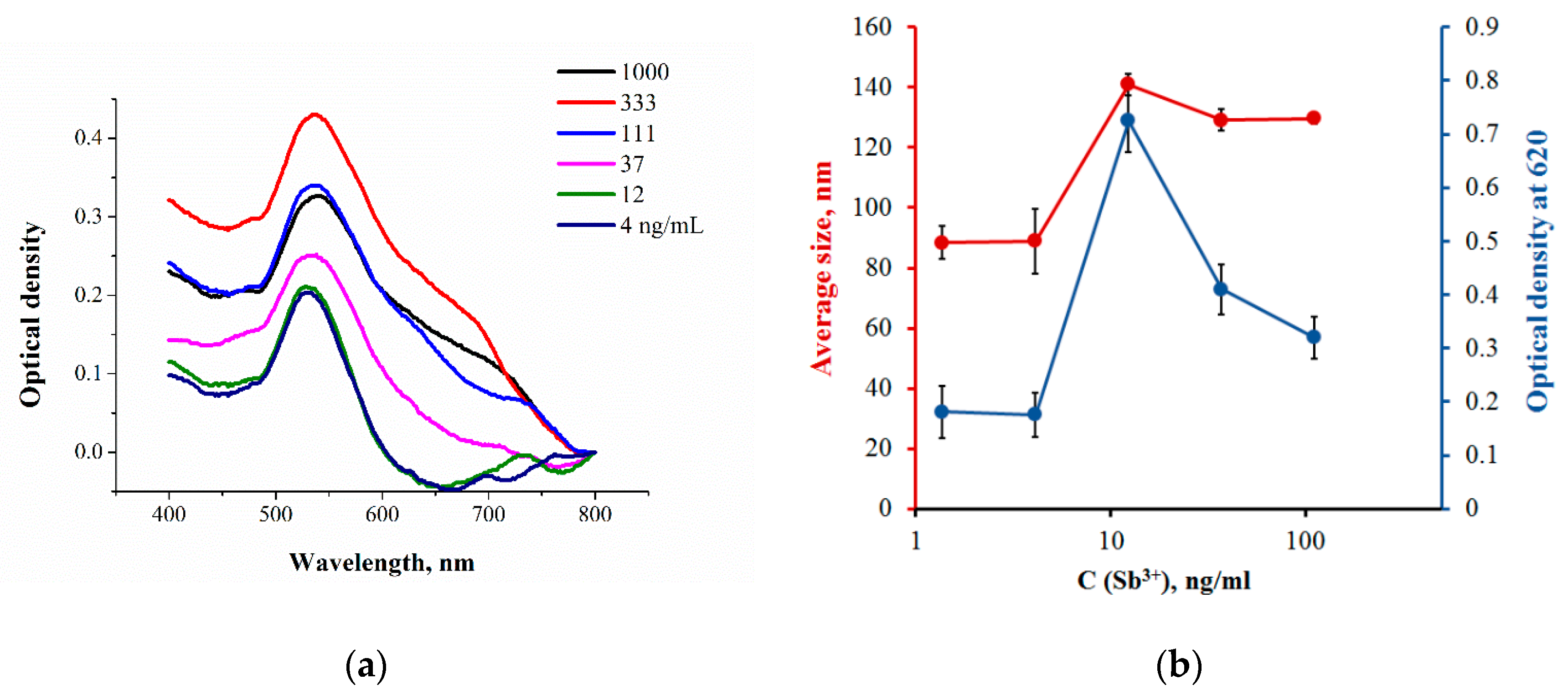

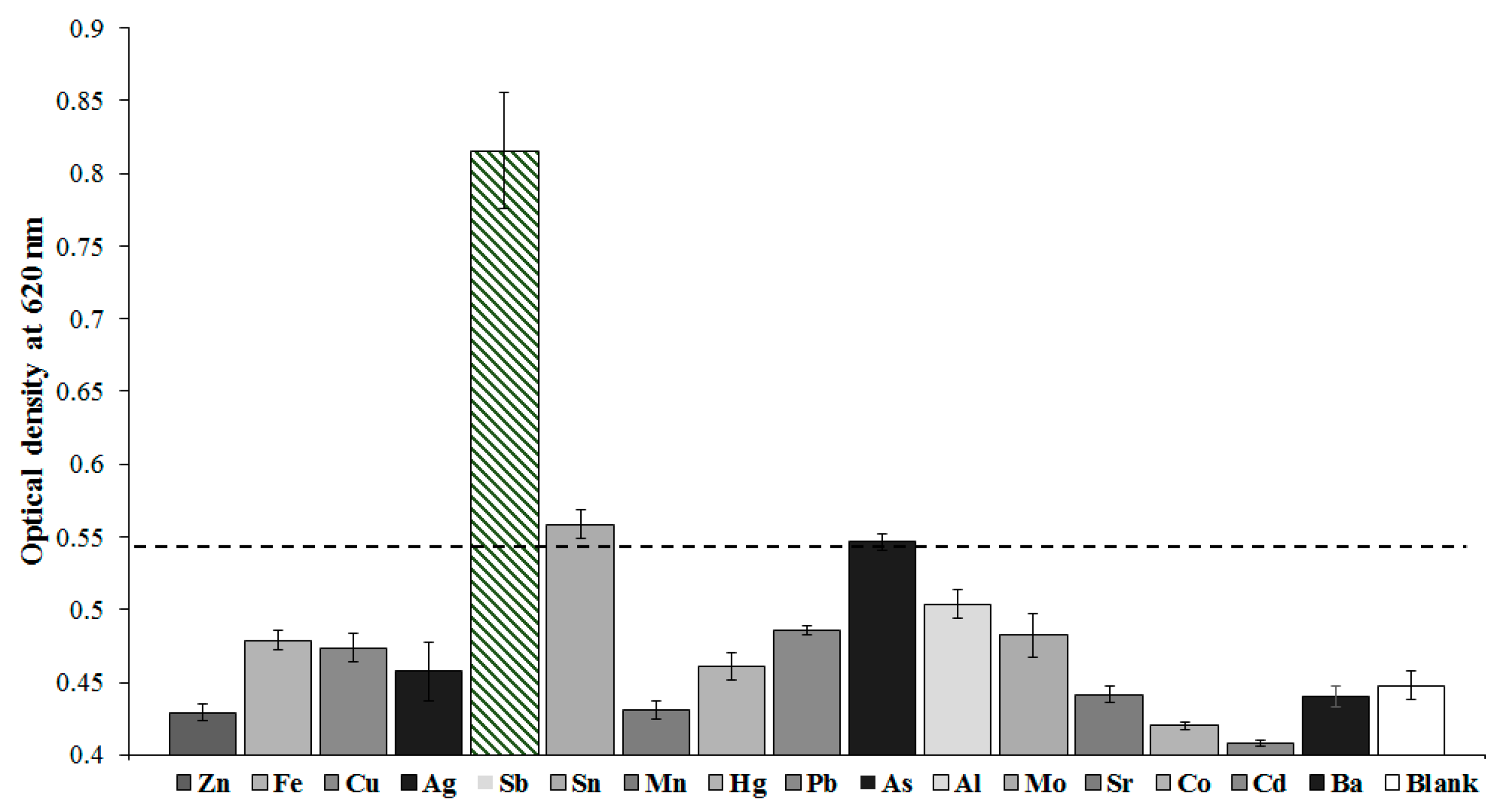
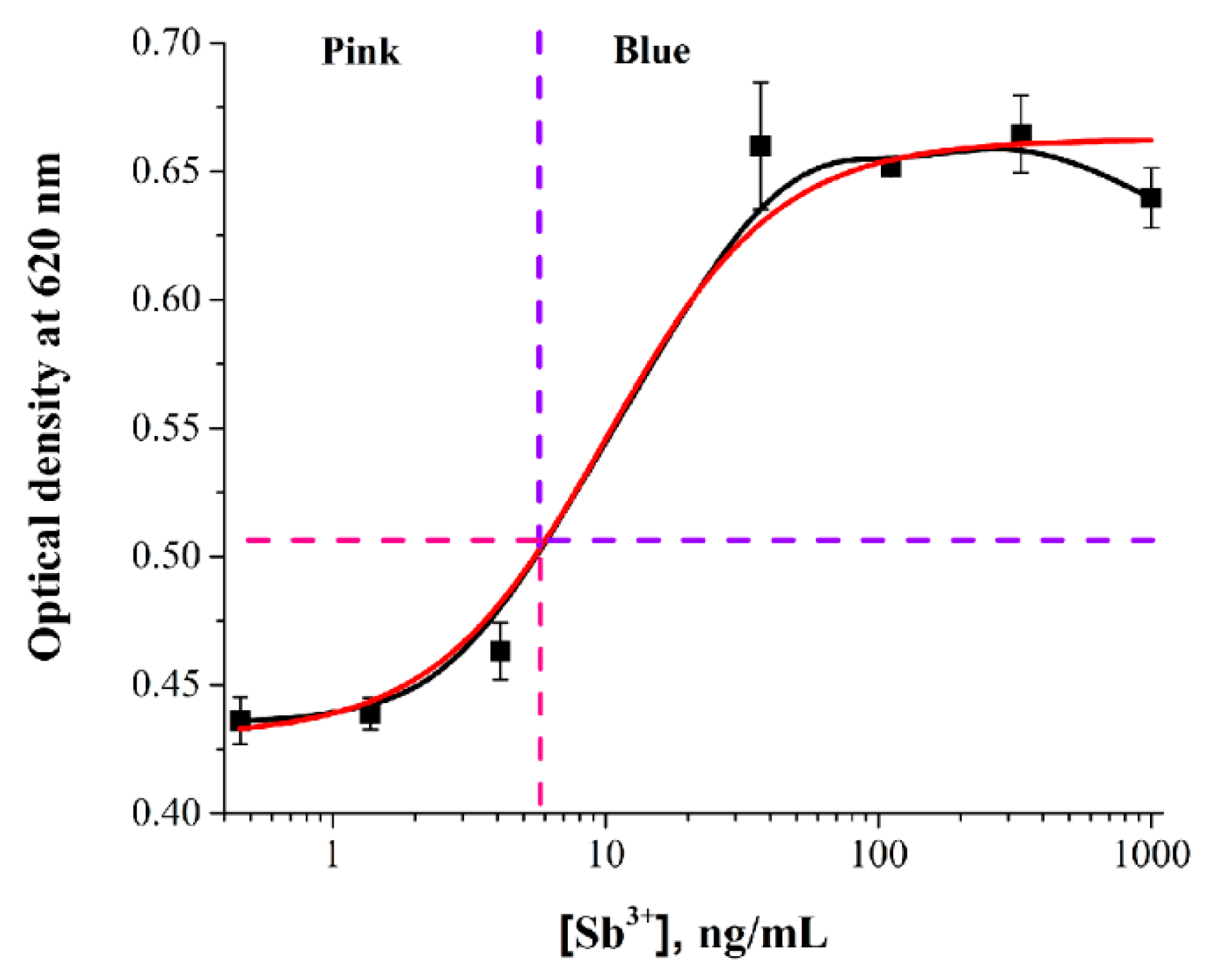
| № | Heavy Metal | Sensing Unit | Detection Method | LOD, Dynamic Range | Probe | Ref. |
|---|---|---|---|---|---|---|
| Sophisticated Instrumental Methods | ||||||
| 1 | As3+, Sb3+ | Gold film electrode | Anodic stripping voltammetry | As3+: 1.12 ng/mL 5.5–0.5 μg/mL | Copper | [42] |
| Sb3+: 0.56 ng/mL 2.6–0.6 μg/mL | ||||||
| 2 | Sb3+ | Silver nanoparticle-modified screen-printed electrodes | 0.083 ng/mL | Seawater samples and pharmaceutical preparations | [43] | |
| 0.012–0.11 μg/mL | ||||||
| 3 | Sb3+ | Gold nanoparticle-modified carbon screen-printed electrodes | 0.115 ng/mL | Seawater samples, drug samples | [44] | |
| 0.012–0.11 μg/mL | ||||||
| 4 | Sb3+ | Mercury film screen-printed electrode | 1.54 ng/mL | Pharmaceutical preparations and seawater | [12] | |
| 1.2–10.1 ng/mL | ||||||
| 5 | Sb3+ | Quercetin-5’-sulfonic acid | Adsorptive stripping voltammetric method | 3.6 pg/mL (60s) and 1.6 pg/mL (180s) | Tap water and a commercial mineral water | [45] |
| 1.0–5.0 ng/mL | ||||||
| 6 | As3+, Sb3+ | AgNPs as a modifier | Electrothermal atomic absorption spectrometry | As3+: 0.022 ng/mL | Seawater and tap water | [9] |
| Sb3+: 0.046 ng/mL | ||||||
| 7 | As3+, Sb3+ | GNPs | As3+: 2.3 ng/mL | Seawater and tap water | [10] | |
| Sb3+: 3.0 ng/mL | ||||||
| 8 | Sb3+ | - | Trap-flame atomic absorption spectrometry | 0.75 ng/mL | Tap and mineral water | [46] |
| 5.0–10.0 ng/mL | ||||||
| 9 | Sb3+, Sb5+ | EDTA and pyridine-2,6-dicarboxylic acid for chelating reaction with Sb | Anion exchange high-performance liquid chromatography | Sb5+: 0.5 μg/mL | Hot spring samples from New Zealand | [18] |
| Sb3+: 0.8 μg/mL | ||||||
| 5–200 μg/mL | ||||||
| 10 | Sb3+ | - | Electrothermal atomic absorption spectrometry | 0.45 μg/mL | Soils and alfalfa samples | [8] |
| 0–50 μg/mL | ||||||
| 11 | Sb3+ | - | Hydride generation coupled to atomic fluorescence spectroscopy | 0.08 μg/mL | Soils and alfalfa samples | [8] |
| 0.25–250 μg/mL | ||||||
| 12 | Sb3+ | - | Multi-syringe flow injection analysis with hydride generation-atomic fluorescence detection | 0.08 ng/mL | Hard drinking water, filtered and diluted lake water | [13] |
| 0.2–5.6 ng/mL | ||||||
| 13 | Sb3+ | BSA activated (TGA)-capped CdTe quantum dot nano-sensor | Fluorescence | 0.0294 ng/mL | Artificial pond water, spring water, sewage | [47] |
| 0.10–22.0 μg/mL | ||||||
| 14 | Sb3+ | - | Atmospheric pressure glow discharge atomic emission spectrometry | 0.14 ng/mL | Groundwater samples | [48] |
| 0.5–100 ng/mL | ||||||
| 15 | Sb3+ | Absorbance at 198 nm after hydride generation | UV-visible molecular absorption spectrophotometry with diode-array detection | 3–440 μg/mL | PVC sample | [7] |
| 16 | Sb3+, Bi3+ | Pyrogallol red | β-correction spectrophotometry | 0.05–5.0 μg/mL | River, tap, and industrial wastewater | [14] |
| 0.2–3.2 μg/mL | ||||||
| 16 | Sb3+, Sb5+ | Pyrogallol (complexing agent) | UV–vis spectroscopy | Sb5+: 1.26 μg/mL | Commercial sample of Glucantime® | [15] |
| Sb3+: 4.84 μg/mL | ||||||
| 17 | Sb3+ | Bromopyrogallol red | spectrophotometry | 0.2 μg/mL | Samples of Glucantime | [16] |
| 5.19 ± 0.16–10.52 ± 0.15 mg/mL | ||||||
| Non-Instrumental Methods | ||||||
| 18 | Sb3+ | Yellow potassium iodoantimonite KSbI4 | Colorimetry | 0.6 μg/mL | Contaminated waters | [17] |
| 0–5 μg/mL | ||||||
| 19 | Sb3+ | GNPs modified with PAO | Colorimetry | 2.0 ng/mL (instrumental LOD) | Drinking water | This study |
| 10 ng/mL (visual LOD) | ||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berlina, A.N.; Komova, N.S.; Zherdev, A.V.; Gaur, M.S.; Dzantiev, B.B. Colorimetric Technique for Antimony Detection Based on the Use of Gold Nanoparticles Conjugated with Poly-A Oligonucleotide. Appl. Sci. 2019, 9, 4782. https://doi.org/10.3390/app9224782
Berlina AN, Komova NS, Zherdev AV, Gaur MS, Dzantiev BB. Colorimetric Technique for Antimony Detection Based on the Use of Gold Nanoparticles Conjugated with Poly-A Oligonucleotide. Applied Sciences. 2019; 9(22):4782. https://doi.org/10.3390/app9224782
Chicago/Turabian StyleBerlina, Anna N., Nadezhda S. Komova, Anatoly V. Zherdev, Mulayam S. Gaur, and Boris B. Dzantiev. 2019. "Colorimetric Technique for Antimony Detection Based on the Use of Gold Nanoparticles Conjugated with Poly-A Oligonucleotide" Applied Sciences 9, no. 22: 4782. https://doi.org/10.3390/app9224782
APA StyleBerlina, A. N., Komova, N. S., Zherdev, A. V., Gaur, M. S., & Dzantiev, B. B. (2019). Colorimetric Technique for Antimony Detection Based on the Use of Gold Nanoparticles Conjugated with Poly-A Oligonucleotide. Applied Sciences, 9(22), 4782. https://doi.org/10.3390/app9224782