Investigations into the Potential Abrasive Release of Nanomaterials due to Material Stress Conditions—Part B: Silver, Titanium Nitride, and Laponite Nanoparticles in Plastic Composites
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Ag-NP Nanocomposites
2.1.2. TiN Nanocomposites
2.1.3. LAP Nanocomposites
2.2. Nanorelease Study
2.2.1. Basic Material Stressing—Abrasion Test
2.2.2. Additional Material Stressing—“Prestress Conditions”
- •
- Thermal stress—heating:
- •
- Thermal stress—freezing:
- •
- Solvent based stress—swelling:
- •
- Mechanical stress—stretching:
2.3. Analytical Set-Up for the Detection of Released Nanomaterials in the Abrasion
- •
- Silver nanocomposites
- •
- TiN nanocomposites
- •
- Laponite nanocomposites
Instrumentation
3. Results
3.1. Silver Nanocomposites
3.2. Titanium Nitride Nanocomposites
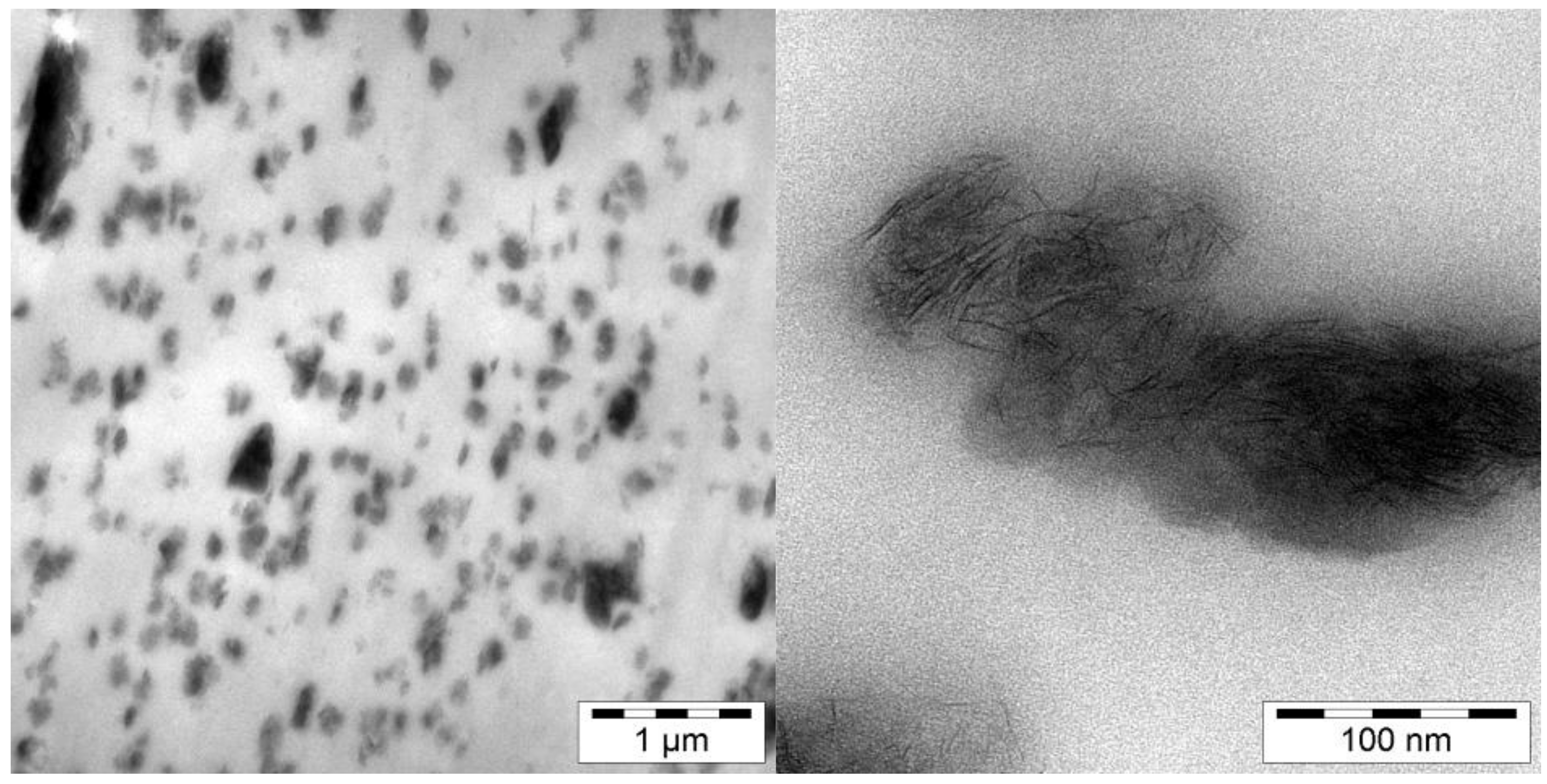
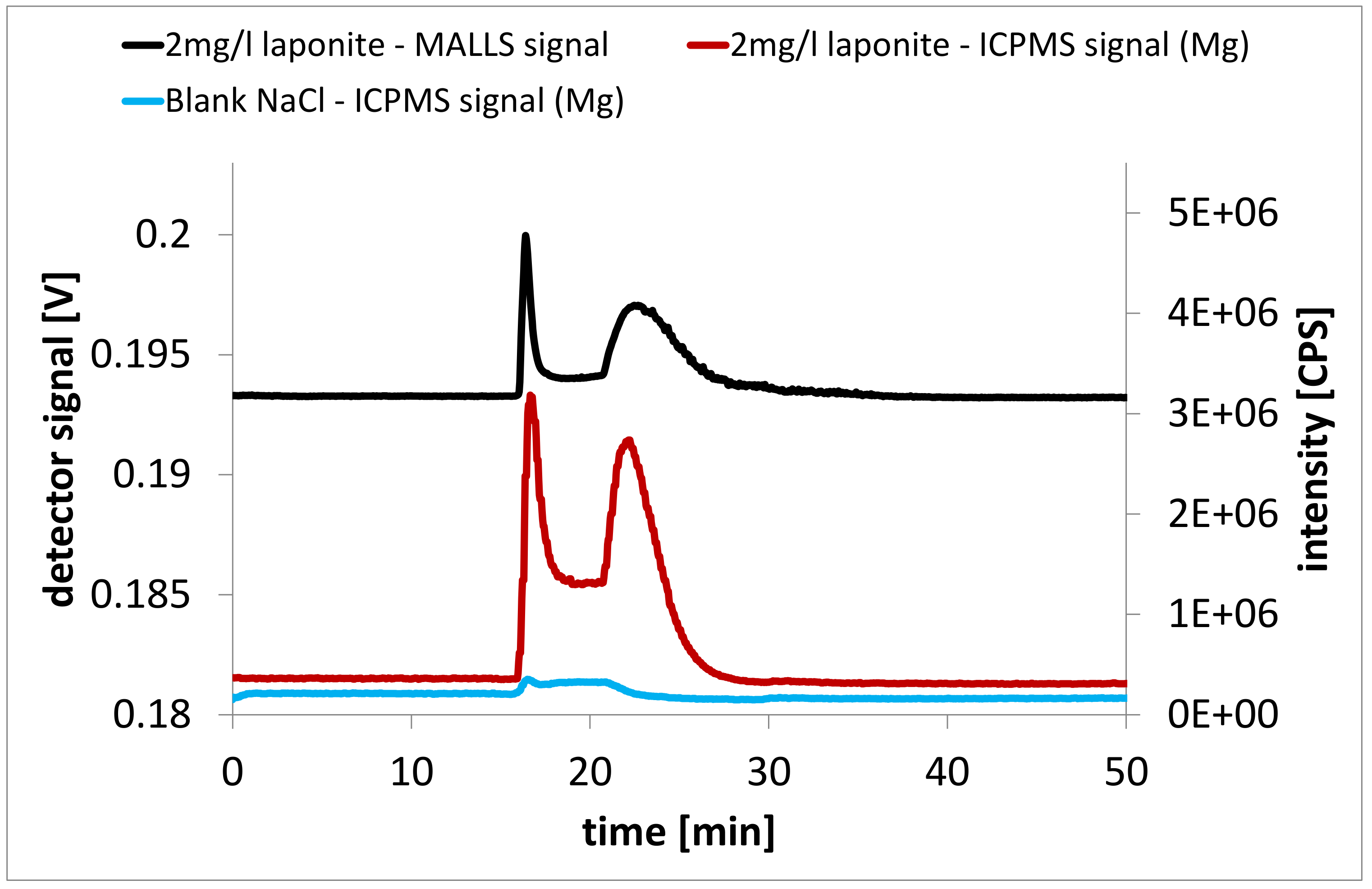
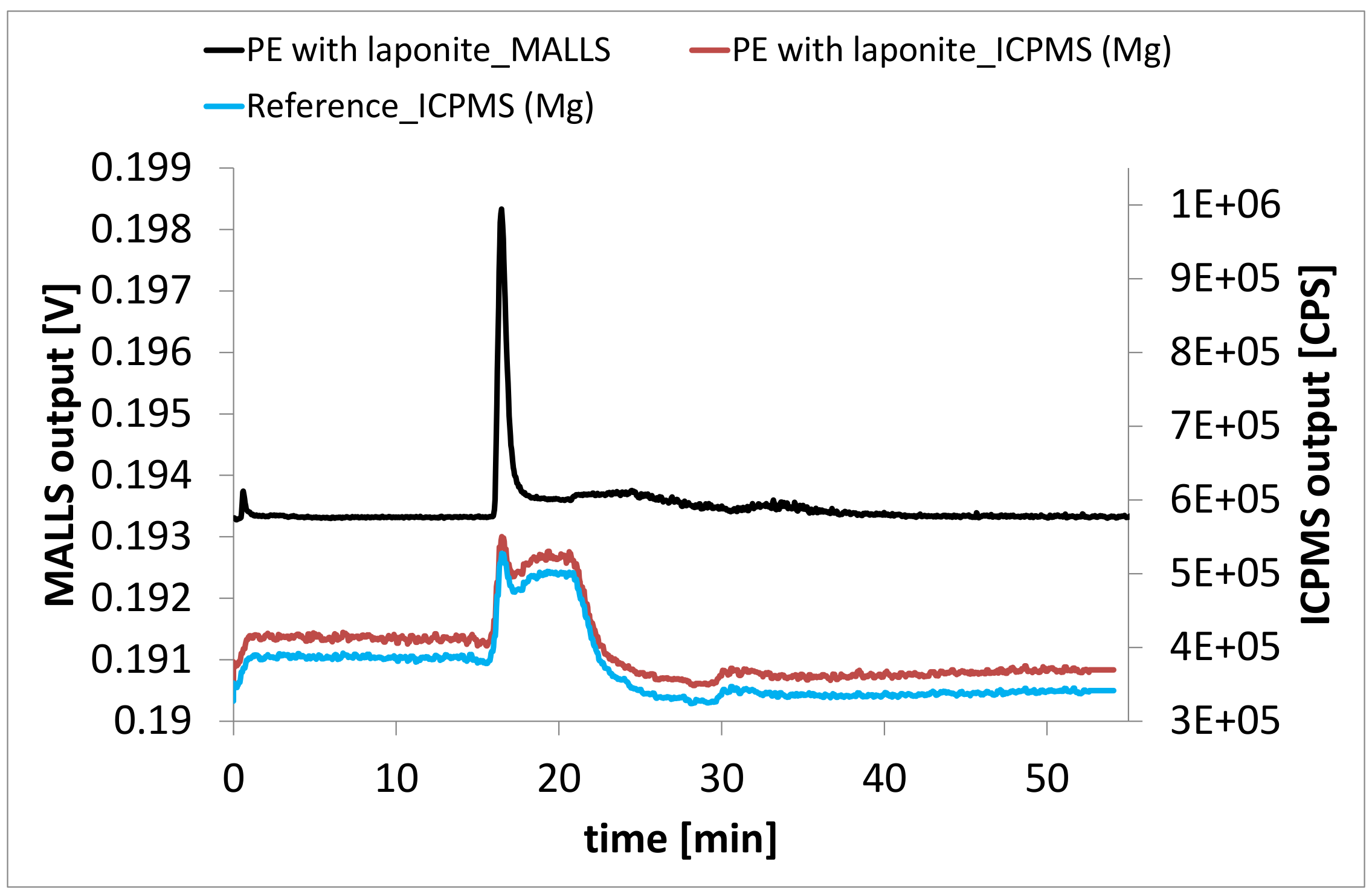
3.3. Laponite Nanocomposites
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Bott, J.; Störmer, A.; Franz, R. A Comprehensive Study into the Migration Potential of Nano Silver Particles from Food Contact Polyolefins, in Chemistry of Food, Food Supplements, and Food Contact Materials: From Production to Plate; Benvenuto, M.A., Ed.; American Chemical Society: Washington, DC, USA, 2014; pp. 51–70. [Google Scholar]
- Bott, J.; Störmer, A.; Franz, R. A model study into the migration potential of nanoparticles from plastics nanocomposites for food contact. Food Packag. Shelf Life 2014, 2, 73–80. [Google Scholar] [CrossRef]
- Bott, J.; Franz, R. Investigation into the Potential Migration of Nanoparticles from Laponite-Polymer Nanocomposites. Nanomaterials 2018, 8, 723. [Google Scholar] [CrossRef] [PubMed]
- Franz, R.; Welle, F. Mathematic modelling of migration of nanoparticles from food contact polymers. In The Use of Nanomaterials in Food Contact Materials—Design, Application, Safety; Veraart, R., Ed.; DEStech Publications Inc.: Lancaster, PA, USA, 2017. [Google Scholar]
- Störmer, A.; Bott, J.; Kemmer, D.; Franz, R. Critical review of the migration potential of nanoparticles in food contact plastics. Trends Food Sci. Technol. 2017, 63, 39–50. [Google Scholar] [CrossRef]
- Cavallaro, G.; Lazzara, G.; Milioto, S. Dispersions of Nanoclays of Different Shapes into Aqueous and Solid Biopolymeric Matrices. Extended Physicochemical Study. Langmuir 2011, 27, 1158–1167. [Google Scholar] [PubMed]
- Cavallaro, G.; Lazzara, G.; Milioto, S. Aqueous phase/nanoparticles interface: Hydroxypropyl cellulose adsorption and desorption triggered by temperature and inorganic salts. Soft Matter 2012, 8, 3627–3633. [Google Scholar] [CrossRef]
- Artiaga, G.; Ramos, K.; Ramos, L.; Cámara, C.; Gómez-Gómez, M. Migration and characterisation of nanosilver from food containers by AF(4)-ICP-MS. Food Chem. 2015, 166, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Chen, S.; Bing, X.; Gao, C.; Wang, T.; Yuan, B. Nanosilver Migrated into Food-Simulating Solutions from Commercially Available Food Fresh Containers. Packag. Technol. Sci. 2011, 24, 291–297. [Google Scholar] [CrossRef]
- Song, H.; Li, B.; Lin, Q.B.; Wu, H.J.; Chen, Y. Migration of silver from nanosilver-polyethylene composite packaging into food simulants. Food Addit. Contam. Part A 2011, 28, 1758–1762. [Google Scholar] [CrossRef] [PubMed]
- Von Goetz, N.; Fabricius, L.; Glaus, R.; Weitbrecht, V.; Gunther, D.; Hungerbuhler, K. Migration of silver from commercial plastic food containers and implications for consumer exposure assessment. Food Addit. Contam. Part A 2013, 30, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Echegoyen, Y.; Rodriguez, S.; Nerin, C. Nanoclay migration from food packaging materials. Food Addit. Contam. Part A 2016, 33, 530–539. [Google Scholar] [CrossRef] [PubMed]
- Farhoodi, M.; Mousavi, S.M.; Sotudeh-Gharebagh, R.; Emam-Djomeh, Z.; Oromiehie, A. Migration of Aluminum and Silicon from PET/Clay Nanocomposite Bottles into Acidic Food Simulant. Packag. Technol. Sci. 2013, 27, 161–168. [Google Scholar] [CrossRef]
- Schmidt, B.; Katiyar, V.; Plackett, D.; Larsen, E.H.; Gerds, N.; Koch, C.B.; Petersen, J.H. Migration of nanosized layered double hydroxide platelets from polylactide nanocomposite films. Food Addit. Contam. Part A 2011, 28, 956–966. [Google Scholar] [CrossRef] [PubMed]
- Lin, Q.B.; Li, H.; Zhong, H.N.; Zhao, Q.; Xiao, D.H.; Wang, Z.W. Migration of Ti from nano-TiO-polyethylene composite packaging into food simulants. Food Addit. Contam. Part A 2014, 31, 1284–1290. [Google Scholar]
- Duncan, T.V. Release of engineered nanomaterials from polymer nanocomposites: The effect of matrix degradation. ACS Appl. Mater. Interfaces 2015, 7, 20–39. [Google Scholar] [CrossRef] [PubMed]
- Duncan, T.V.; Pillai, K. Release of Engineered Nanomaterials from Polymer Nanocomposites: Diffusion, Dissolution, and Desorption. ACS Appl. Mater. Interfaces 2015, 7, 2–19. [Google Scholar] [CrossRef] [PubMed]
- Addo Ntim, S.; Norris, S.; Scott, K.; Thomas, T.A.; Noonan, G.O. Consumer use effects on nanoparticle release from commercially available ceramic cookware. Food Control 2018, 87, 31–39. [Google Scholar] [CrossRef]
- Göhler, D.; Nogowski, A.; Fiala, P.; Stintz, M. Nanoparticle release from nanocomposites due to mechanical treatment at two stages of the life-cycle. J. Phys. Conf. Ser. 2013, 429, 012045. [Google Scholar] [CrossRef]
- Golja, V.; Dražić, G.; Lorenzetti, M.; Vidmar, J.; Ščančar, J.; Zalaznik, M.; Kalin, M.; Novak, S. Characterisation of food contact non-stick coatings containing TiO2 nanoparticles and study of their possible release into food. Food Addit. Contam. Part A 2017, 34, 421–433. [Google Scholar] [CrossRef] [PubMed]






| Nanomaterial | Polymer Matrix | Nanomaterial Loading |
|---|---|---|
| Silver Nanoparticles (Ag-NP) | LD-PE | 250 mg/kg |
| Titaniumnitride (TiN) | LD-PE | 1000 mg/kg |
| Laponite (LAP) | LD-PE | 6% |
| Abrasion Test | Prestress Condition | Ag in Abrasion [ng/dm2] |
|---|---|---|
| 60 min at 275 rpm | none | <16.0 |
| 30 min at 275 rpm | “heating ”24 h at 100 °C | <16.0 |
| 30 min at 275 rpm | “freezing ”24 h at −50 °C | <16.0 |
| 30 min at 275 rpm | “swelling” 24 h at 40 °C in isooctane | <16.0 |
| 30 min at 275 rpm | “stretching” irreversible transformation | <16.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bott, J.; Franz, R. Investigations into the Potential Abrasive Release of Nanomaterials due to Material Stress Conditions—Part B: Silver, Titanium Nitride, and Laponite Nanoparticles in Plastic Composites. Appl. Sci. 2019, 9, 221. https://doi.org/10.3390/app9020221
Bott J, Franz R. Investigations into the Potential Abrasive Release of Nanomaterials due to Material Stress Conditions—Part B: Silver, Titanium Nitride, and Laponite Nanoparticles in Plastic Composites. Applied Sciences. 2019; 9(2):221. https://doi.org/10.3390/app9020221
Chicago/Turabian StyleBott, Johannes, and Roland Franz. 2019. "Investigations into the Potential Abrasive Release of Nanomaterials due to Material Stress Conditions—Part B: Silver, Titanium Nitride, and Laponite Nanoparticles in Plastic Composites" Applied Sciences 9, no. 2: 221. https://doi.org/10.3390/app9020221
APA StyleBott, J., & Franz, R. (2019). Investigations into the Potential Abrasive Release of Nanomaterials due to Material Stress Conditions—Part B: Silver, Titanium Nitride, and Laponite Nanoparticles in Plastic Composites. Applied Sciences, 9(2), 221. https://doi.org/10.3390/app9020221






