A Synthetic Approach for Biosynthesis of Miquelianin and Scutellarin A in Escherichia coli
(This article belongs to the Section Applied Biosciences and Bioengineering)
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Culture Conditions
2.2. Cloning and Expression of Recombinant Proteins
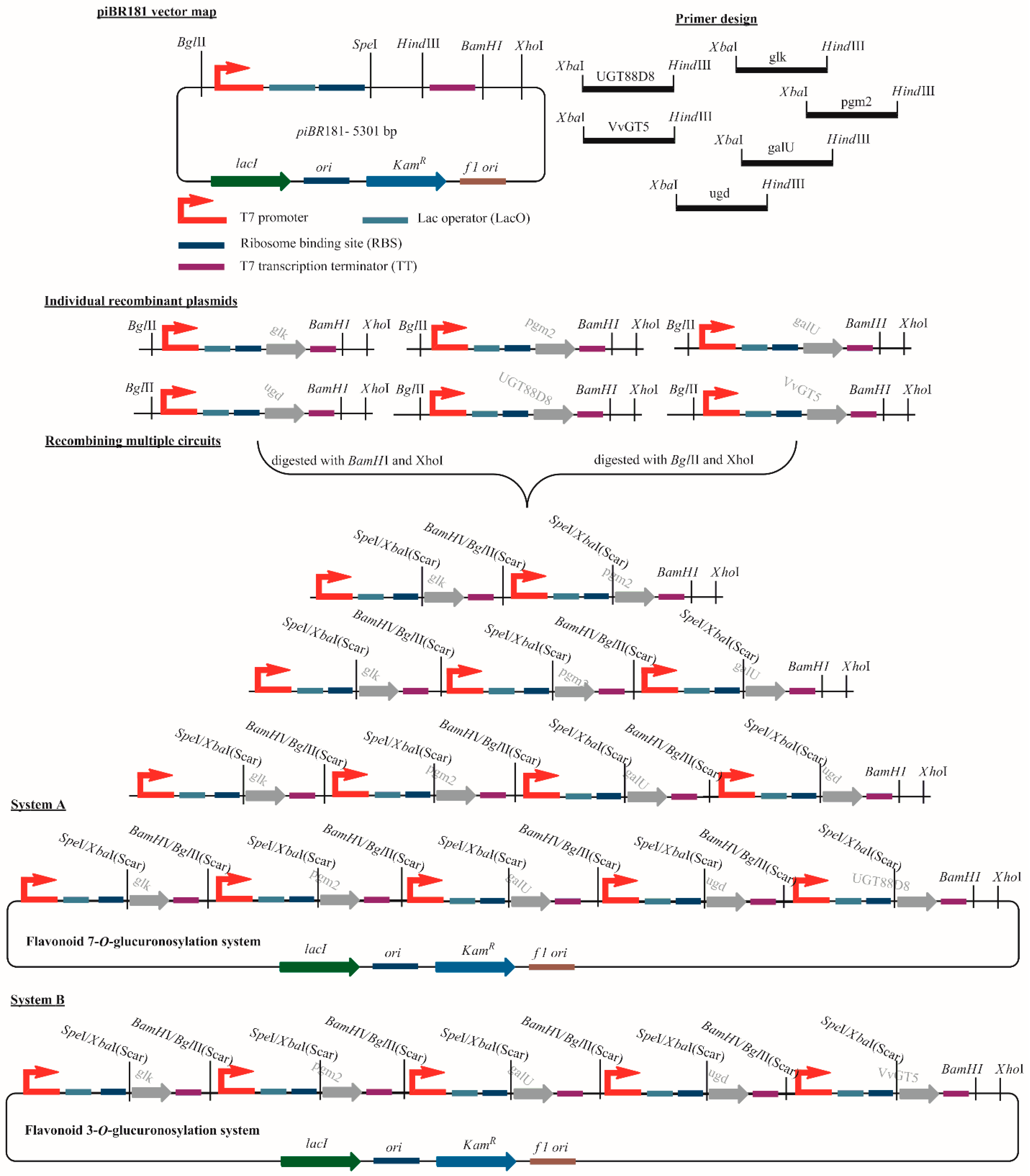
2.3. Construction of Sugar Cassettes
2.4. Whole-Cell Biotransformation and Flavonoid Extraction
2.5. Product Analysis and Quantification
3. Results and Discussion
3.1. Selection of Plant UGTs and Their Activities
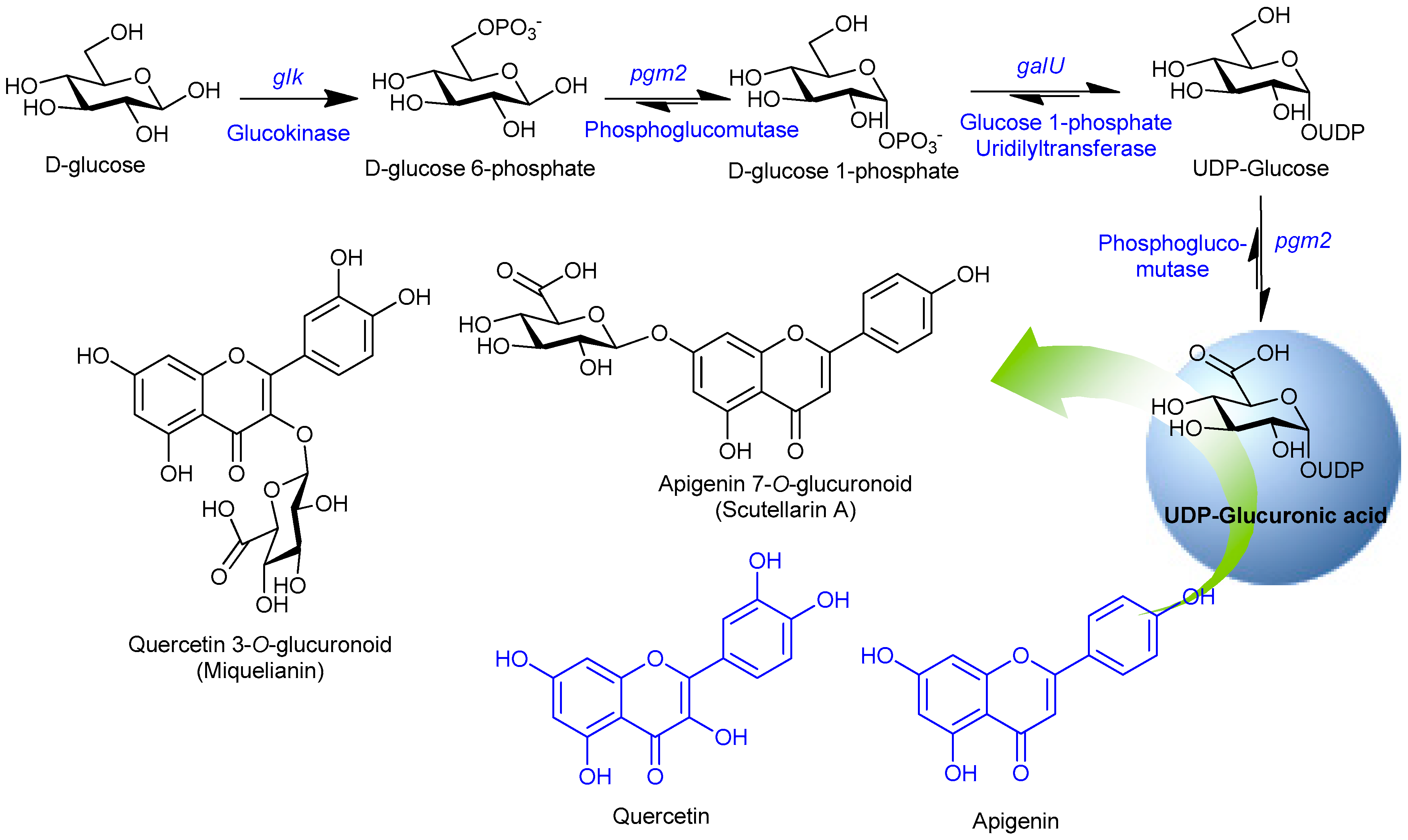
3.2. Cloning and Expression of NDP-Sugar Pathway Genes and Assembly in a Single Vector
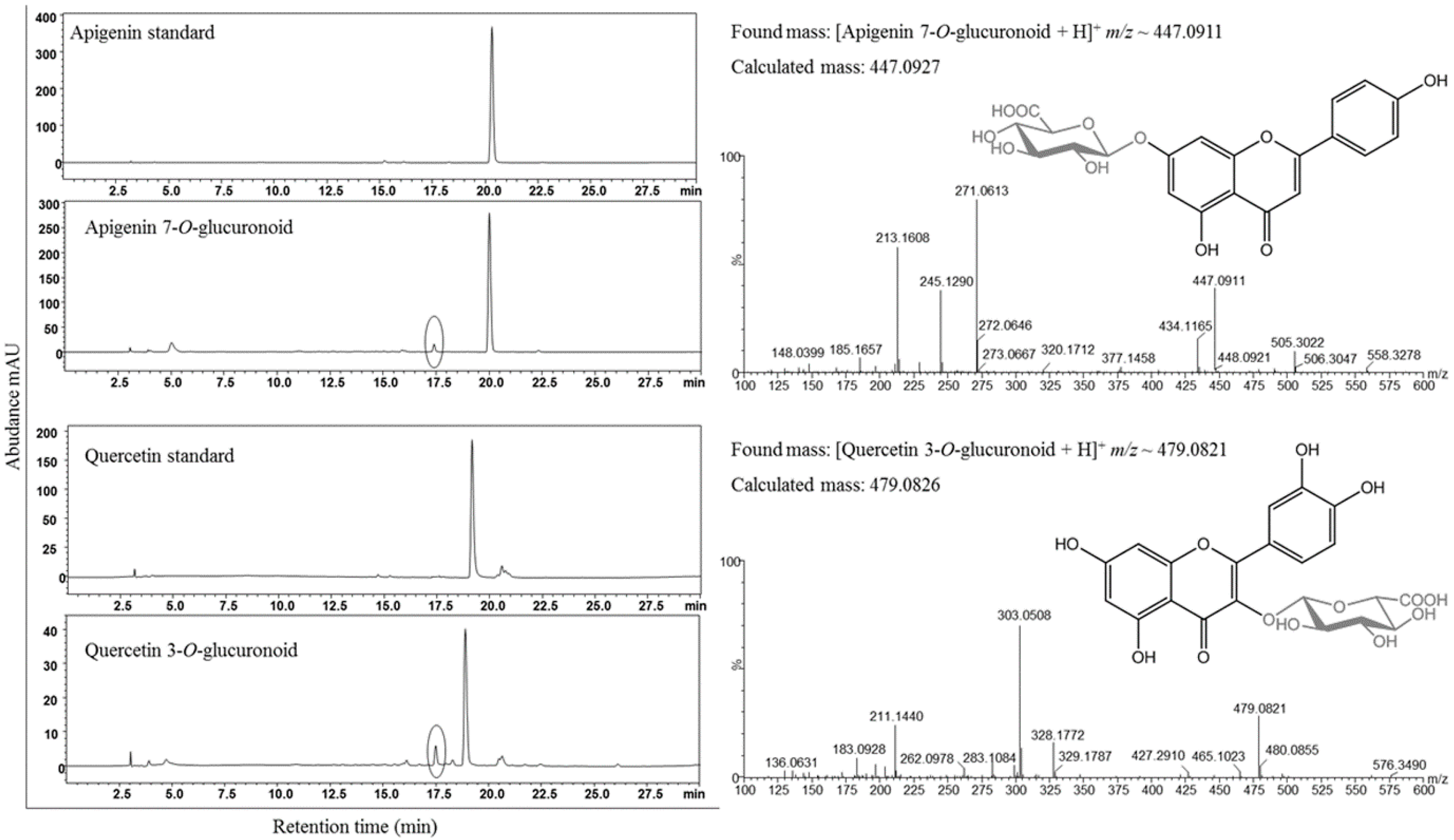
3.3. Synthesis of Glucuronoids in Recombinant Strains
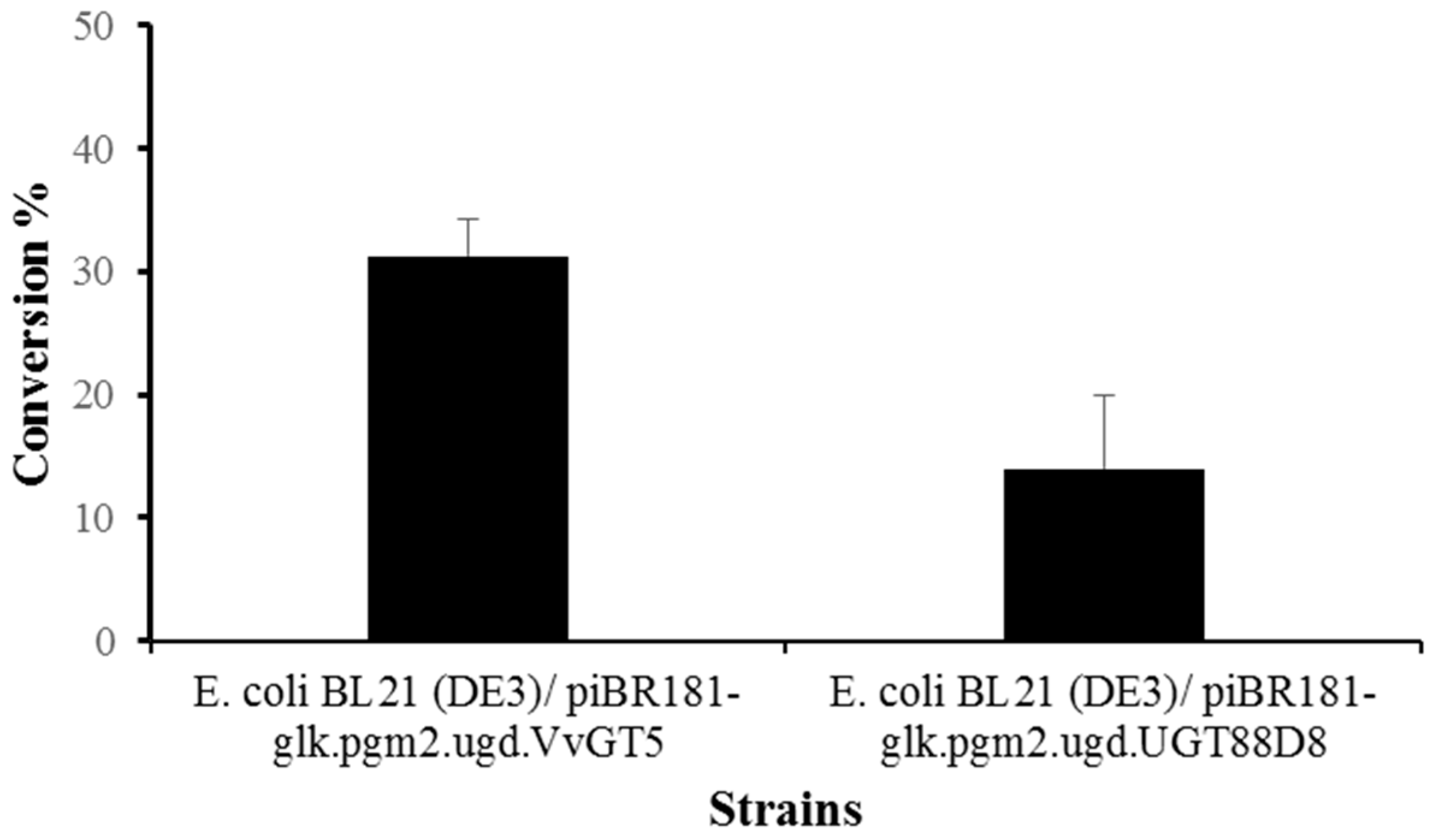
3.4. Enhanced Synthesis of Miquelianin and Scutellarin A
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Falcone Ferreyra, M.L.; Rius, S.P.; Casati, P. Flavonoids: Biosynthesis, biological functions, and biotechnological applications. Front. Plant Sci. 2012, 3, 222. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed]
- Airoldi, C.; La Ferla, B.; D Orazio, G.; Ciaramelli, C.; Palmioli, A. Flavonoids in the treatment of Alzheimer’s and other neurodegenerative diseases. Curr. Med. Chem. 2018, 25, 3228–3246. [Google Scholar] [CrossRef] [PubMed]
- Winkel-Shirley, B. Flavonoid biosynthesis, A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol. 2001, 126, 485–493. [Google Scholar] [CrossRef]
- Song, M.C.; Kim, E.J.; Kim, E.; Rathwell, K.; Nam, S.J.; Yoon, Y.J. Microbial biosynthesis of medicinally important plant secondary metabolites. Nat. Prod. Rep. 2014, 31, 1497–1509. [Google Scholar] [CrossRef]
- Veitch, N.C.; Grayer, R.J. Flavonoids and their glycosides, including anthocyanins. Nat. Prod. Rep. 2008, 25, 555–611. [Google Scholar] [CrossRef] [PubMed]
- Min, Y.S.; Yim, S.H.; Bai, K.L.; Choi, H.J.; Jeong, J.H.; Song, H.J.; Park, S.H.; Ham, I.; Whang, W.K.; Sohn, U.D. The effects of apigenin-7-O-β-d-glucuronopyranoside on reflux oesophagitis and fastritis in rats. Auton. Autacoid Pharmacol. 2005, 25, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Gupta, S. Apigenin: A promising molecule for cancer prevention. Pharm. Res. 2010, 27, 962–978. [Google Scholar] [CrossRef]
- Gurung, R.B.; Pandey, R.P.; Sohng, J.K. Role of apigenin in cancer prevention. In Apigenin and Naringenin: Natural Sources, Pharmacology and Role in Cancer Prevention; Stacks, N.M., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2016. [Google Scholar]
- Cermak, R.; Wolffiram, S. The potential of flavonoids to influence drug metabolism and pharmacokinetics by local gastrointestinal mechanisms. Curr. Drug Metab. 2006, 7, 729–744. [Google Scholar] [CrossRef] [PubMed]
- Theodosiou, E.; Loutrari, H.; Stamatis, H.; Roussos, C.; Kolisis, F.N. Biocatalytic synthesis and antitumor activities of novel silybin acylated derivatives with dicarboxylic acids. New Biotechnol. 2011, 28, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Desmet, T.; Soetaert, W.; Bojarova, P.; Kren, V.; Dijkhuizen, L.; Eastwick-Field, V.; Schiller, A. Enzymatic glycosylation of small molecules: Challenging substrates required tailored catalysts. Chemistry 2012, 18, 10786–10801. [Google Scholar] [CrossRef] [PubMed]
- Makino, T.; Shimizu, R.; Kanemaru, M.; Suzuki, Y.; Moriwaki, M.; Mizukami, H. Enzymatically modified isoquercitrin, alpha-oligoglucosyl quercetin 3-O-glucoside, is absorbed more easily than other quercetin glycosides or aglycone after oral administration in rats. Biol. Pharm. Bull. 2009, 32, 2034–2040. [Google Scholar] [CrossRef] [PubMed]
- Pandey, R.P.; Parajuli, P.; Koffas, M.A.G.; Sohng, J.K. Microbial production of natural and non-natural flavonoids: Pathway engineering, directed evolution and systems/synthetic biology. Biotechnol. Adv. 2016, 34, 634–662. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Lv, M.; Hu, J.; Huang, K.; Xu, H. Glycosylation and activities of natural products. Mini-Rev. Med. Chem. 2016, 16, 1013–1016. [Google Scholar] [CrossRef]
- King, C.D.; Rios, G.R.; Green, M.D.; Tephyl, T.R. UDP-glucuronosyltransferases. Curr. Drug Metab. 2000, 1, 143–161. [Google Scholar] [CrossRef]
- Sarma, P.S.; Ssastry, K.S. Glucuronic acid, a precursor of ascorbic acid in Aspergillus niger. Nature 1957, 179, 44–45. [Google Scholar]
- Yang, T.; Bar-Peled, Y.; Smith, J.A.; Glushka, J.; Bar-Peled, M. In-microbe formation of nucleotide sugars in engineered Escherichia coli. Anal. Biochem. 2012, 421, 691–698. [Google Scholar] [CrossRef]
- Wang, S. Structure, mechanism and engineering of plant natural product glycosyltransferases. FEBS Lett. 2009, 583, 3303–3309. [Google Scholar] [CrossRef]
- Sambrook, J.; Fritsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual, 2nd ed.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1989. [Google Scholar]
- Chaudhary, A.K.; Park, J.W.; Yoon, Y.J.; Kim, B.G.; Sohng, J.K. Re-engineering genetic circuit for 2-deoxystreptamine (2-DOS) biosynthesis in Escherichia coli BL21 (DE3). Biotechnol. Lett. 2013, 35, 285–293. [Google Scholar] [CrossRef]
- Parajuli, P.; Pandey, R.P.; Pokhrel, A.R.; Ghimire, G.P.; Sohng, J.K. Enzymatic glycosylation of the topical antibiotic mupirocin. Glycoconj. J. 2014, 31, 563–572. [Google Scholar] [CrossRef]
- Siedler, S.; Bringer, S.; Blank, L.M.; Bott, M. Engineering yield and rate of reductive biotransformation in Escherichia coli by partial cyclization of the pentose phosphate pathway and PTS-independent glucose transport. Appl. Microbiol. Biotechnol. 2012, 93, 1459–1467. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Muzashvili, T.S.; Georgiev, M.I. Advancees in the biotechnological glycosylation of valuable flavonoids. Biotechnol. Adv. 2014, 32, 1145–1156. [Google Scholar] [CrossRef] [PubMed]
- Ono, E.; Ruike, M.; Iwashita, T.; Nomoto, K.; Fukui, Y. Co-pigmentation and flavonoid glycosyltransferases in blue veronica a persica flowers. Phytochemistry 2010, 71, 726–735. [Google Scholar] [CrossRef]
- Mizohata, E.; Okuda, T.; Hatanaka, S.; Nakayama, T.; Horikawa, M.; Nakayama, T.; Ono, E.; Inoue, T. Crystallization and preliminary X-ray chrystallographic analysis of UDP-glucuronic acid: Flavonol-3-O-glucuronosyltransferase (VvGT5) from the grapevine Vitis vinifera. Acta Crystallogr. Sect. F 2013, 69, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Simkhada, D.; Lee, H.C.; Sohng, J.K. Genetic engineering approach for the production of rhamnosyl and allosyl flavonoids from Escherichia coli. Biotechnol. Bioeng. 2010, 107, 154–162. [Google Scholar] [CrossRef]
- Malla, S.; Pandey, R.P.; Kim, B.G.; Sohng, J.K. Regiospecific modifications of naringenin for astragalin production in Escherichia coli. Biotechnol. Bioeng. 2013, 110, 2525–2535. [Google Scholar] [CrossRef]
- Kim, B.G.; Jung, N.R.; Joe, E.J.; Hur, H.G.; Lim, Y.; Chong, Y.; Ahn, J.H. Bacterial synthesis of a flavonoid deoxyaminosugar conjugate in Escherichia coli expressing a glycosyltransferase of Arabidopsis thaliana. ChemBioChem 2010, 11, 2389–2392. [Google Scholar] [CrossRef]
- Kim, B.G.; Sung, S.H.; Ahn, J.H. Biological synthesis of quercetin 3-O-N-acetylglucosamine conjugate using engineered Escherichia coli expressing UGT78D2. Appl. Microbiol. Biotechnol. 2012, 93, 2447–2453. [Google Scholar] [CrossRef]
- Pandey, R.P.; Malla, S.; Simkhada, D.; Kim, B.G.; Sohng, J.K. Production of 3-O-xylosyl quercetin in Escherichia coli. Appl. Microbiol. Biotechnol. 2013, 97, 1889–1901. [Google Scholar] [CrossRef]
- Han, S.H.; Kim, B.G.; Yoon, J.A.; Chong, Y.; Ahn, J.H. Synthesis of flavonoid O-pentosides by Escherichia coli through engineering of nucleotide sugar pathways and glycosyltransferase. Appl. Environ. Microbiol. 2014, 80, 2754–2762. [Google Scholar] [CrossRef]
- Thuan, N.H.; Pandey, R.P.; Thuy, T.T.; Park, J.W.; Sohng, J.K. Improvement of regio-specific production of myricetin-3-O-α-L-rhamnoside in engineered Escherichia coli. Appl. Biochem. Biotechnol. 2013, 171, 1956–1967. [Google Scholar] [CrossRef]
- Parajuli, P.; Pandey, R.P.; Trang, N.T.; Chaudhary, A.K.; Sohng, J.K. Synthetic sugar cassettes for the efficient production of flavonol glycosides in Escherichia coli. Microb. Cell Factories 2015, 14, 76. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, A.; Pandey, R.P.; Dhakal, D.; Parajuli, P.; Sohng, J.K. Biosynthesis of flavone C-glucosides in engineered Escherichia coli. Appl. Microbiol. Biotechnol. 2018, 102, 1251–1267. [Google Scholar] [CrossRef] [PubMed]
- Pandey, R.P.; Parajuli, P.; Sohng, J.K. Metabolic engineering of glycosylated polyketides biosynthesis. Emerg. Top. Life Sci. 2018. [Google Scholar] [CrossRef]
- Kim, S.Y.; Lee, H.R.; Park, K.S.; Kim, B.G.; Ahn, J.H. Metabolic engineering of Escherichia coli for the biosynthesis of flavonoid-O-glucuronides and flavonoid-O-galactoside. Appl. Microbiol. Biotechnol. 2015, 99, 2233–2242. [Google Scholar] [CrossRef] [PubMed]
| Primers | Oligonucleotide Sequences (5′-3′) | Total PCR Size | Restriction Sites |
|---|---|---|---|
| glk-F | TCTAGAATGGAAATTGTTGCGATTGACATCGGT | 984 bp | XbaI |
| glk-R | AAGCTTTTATTCAACTTCAGAATATTTGTTGGC | 984 bp | HindIII |
| pgm2-F | TCTAGAATGAGCTGGAGAACGAGCTATGAACGC | 1749 bp | XbaI |
| pgm2-R | AAGCTTTTACGAATTTGAGGTCGCTTTTACAAT | 1749 bp | HindIII |
| galU-F | TCTAGAATGGCTGCCATTAATACGAAAGTCAAA | 908 bp | XbaI |
| galU-R | AAGCTTTTACTTCTTAATGCCCATCTCTTCTTC | 908 bp | HindIII |
| ugd-F | TCTAGAATGAAAATCACCATTTCCGGTACTGGC | 1167 bp | XbaI |
| ugd-R | AAGCTTTTAGTCGCTGCCAAAGAGATCGCGGGT | 1167 bp | HindIII |
| UGT88D8-F | TCTAGAATGGAAGACACGATTATTCTGTATGCC | XbaI | |
| UGT88D8-R | AAGCTTTGATGAACTTATCCAGATCCACCACGC | HindIII | |
| VvGT5-F | TCTAGAATGACCACGACCGCCTCCTCAATGGAT | XbaI | |
| VvGT5-R | AAGCTTCGTGTCCAGCGGCAGTTTAGAGGTGGT | HindIII |
| Strains/Plasmids | Description | Source/Reference |
|---|---|---|
| Escherichia coli XL-1 Blue (MRF′) | General cloning host | Stratagene, CA USA |
| E. coli BL21 (DE3) | ompT hsdT hsdS (rB-mB-) gal (DE3) | Novagen, Madison, WI, USA |
| E. coli BL21 (DE3)/piBR181-UGT88D8 | E. coli BL21 (DE3) carrying piBR181-UGT88D | This study |
| E. coli BL21 (DE3)/piBR181-VvGT5 | E. coli BL21 (DE3) carrying piBR181-VvGT5 | This study |
| E. coli BL21 (DE3)/piBR181-glk.pgm2.ugd.UGT88D8 | E. coli BL21 (DE3) carrying piBR181.glk, pgm2, galU, ugd and UGT88D8 | This study |
| E. coli BL21 (DE3)/piBR181-glk.pgm2.ugd.VvGT5 | E. coli BL21 (DE3) carrying piBR181.glk, pgm2, galU, ugd, and VvGT5 | This study |
| Plasmid and Vectors | ||
| pGEM-T® easy vector | General cloning vector, T7, and SP6 promoters, f1 ori, Ampr | Promega, Madison, WI, USA |
| pIBR181 | Mono-cistronic vector | [21] |
| piBR181 piBR181-UGT88D8 | The piBR181 vector carrying UGT88D8, codon optimized flavonoid 3-O-glucuronosyltransferase from blue flowers of Veronica persica | This study |
| piBR181 piBR181-VvGT5 | The piBR181 vector carrying VvGT5, codon optimized flavonoid 7-O-glucuronosyltransferase from grapevine (Vitis vinifera) | This study |
| piBR181-glk.pgm2.galU.ugd. UGT88D8 | The piBR181 vector carrying glk from Zymomonas mobilis, pgm2 from Bacillus licheniformis, galU and ugd from E. coli K12 and UGT88D8 from blue flowers of Veronica persica | This study |
| piBR181-glk.pgm2.galU.ugd. VvGT5 | The piBR181 vector carrying glk from Zymomonas mobilis, pgm2 Bacillus licheniformis, galU and ugd from E. coli K12 and VvGT5 from grapevine (Vitis vinifera) | This study |
| pVWEx1-glf-glk | pVWEx1 carrying glk | [23] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pandey, R.P.; Jung, H.Y.; Parajuli, P.; Nguyen, T.H.T.; Bashyal, P.; Sohng, J.K. A Synthetic Approach for Biosynthesis of Miquelianin and Scutellarin A in Escherichia coli. Appl. Sci. 2019, 9, 215. https://doi.org/10.3390/app9020215
Pandey RP, Jung HY, Parajuli P, Nguyen THT, Bashyal P, Sohng JK. A Synthetic Approach for Biosynthesis of Miquelianin and Scutellarin A in Escherichia coli. Applied Sciences. 2019; 9(2):215. https://doi.org/10.3390/app9020215
Chicago/Turabian StylePandey, Ramesh Prasad, Ha Young Jung, Prakash Parajuli, Thi Huyen Trang Nguyen, Puspalata Bashyal, and Jae Kyung Sohng. 2019. "A Synthetic Approach for Biosynthesis of Miquelianin and Scutellarin A in Escherichia coli" Applied Sciences 9, no. 2: 215. https://doi.org/10.3390/app9020215
APA StylePandey, R. P., Jung, H. Y., Parajuli, P., Nguyen, T. H. T., Bashyal, P., & Sohng, J. K. (2019). A Synthetic Approach for Biosynthesis of Miquelianin and Scutellarin A in Escherichia coli. Applied Sciences, 9(2), 215. https://doi.org/10.3390/app9020215






