Evaluation of the Potential Toxicity of Effluents from the Textile Industry before and after Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. General Characterisation of the Textile Effluent
Gas Chromatography Coupled to Mass Spectrometry (GC-MS) Methodology
2.2. Microtox® Test with Aliivibrio fischeri
2.3. Growth Test with Raphidocelis subcapitata
2.4. Toxicity Test with Lemna minor
2.5. Toxicity Test with Daphnia magna
2.5.1. Acute Immobilisation Test
2.5.2. Feeding Inhibition Assay
2.6. Statistical Analysis
3. Results and Discussion
3.1. Physico-Chemical Characterisation of Textile Effluent Samples
3.2. Toxicity Test with Aliivibrio fischeri (Microtox®)
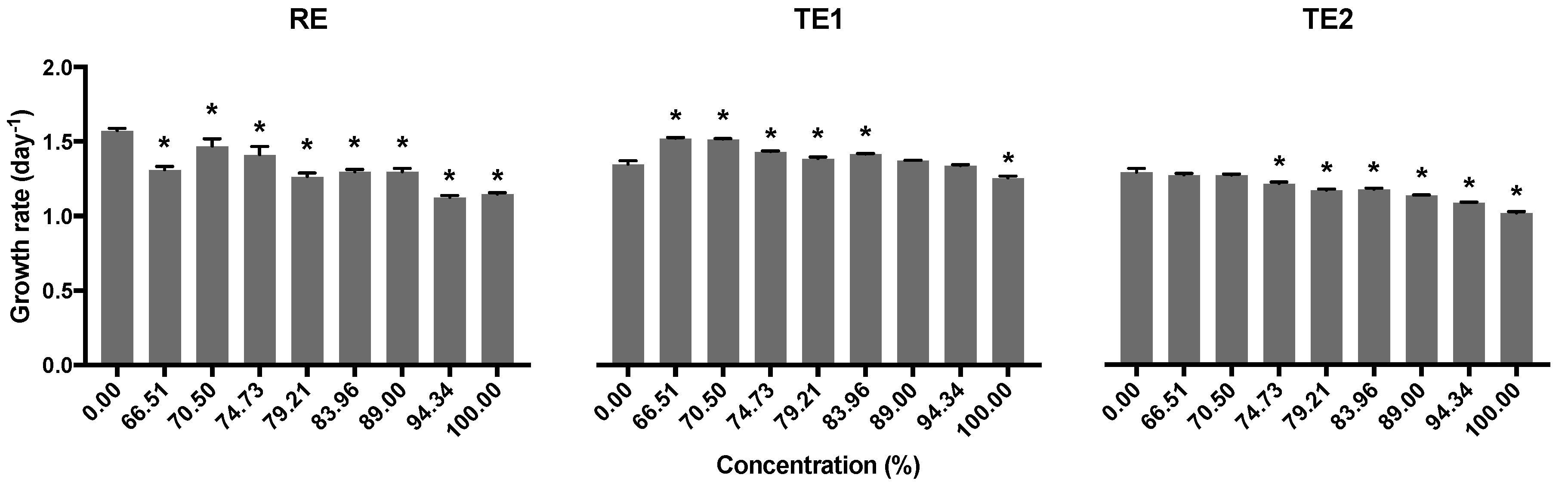
3.3. Growth Inhibition Assay with Raphidocelis subcapitata
3.4. Growth Inhibition Assay with Lemna minor
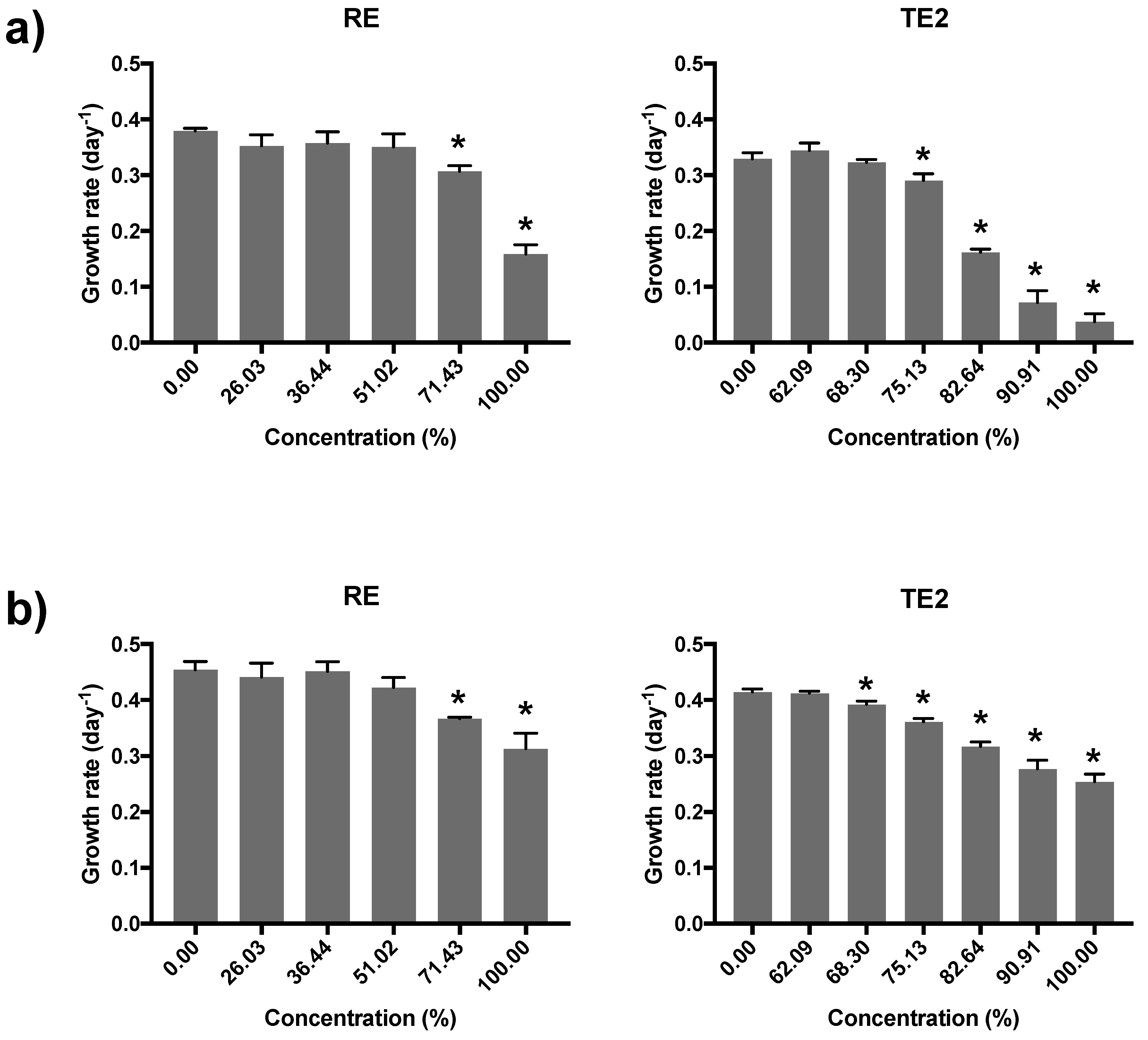
3.5. Acute Immobilisation Test with Daphnia magna
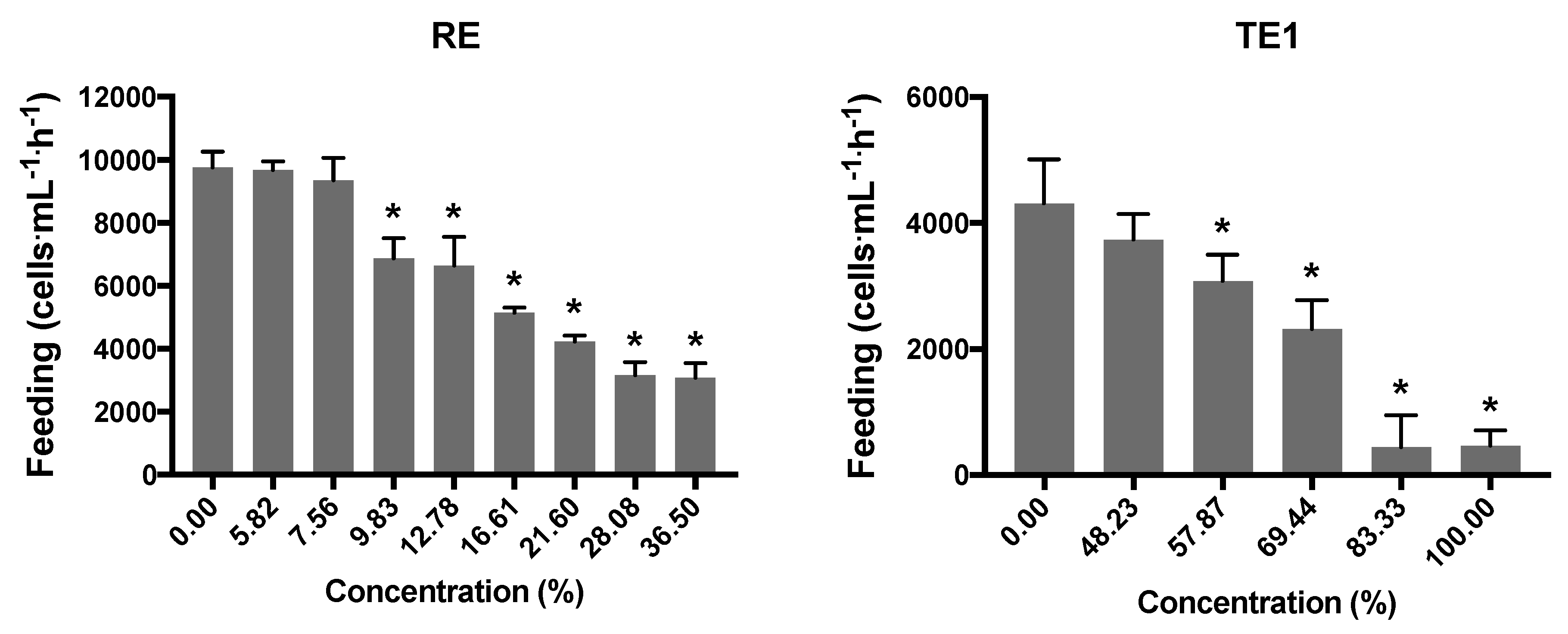
3.6. Feeding Inhibition Assay with D. magna
3.7. Comparison between the Ecotoxicological Assays
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ellouze, E.; Tahri, N.; Amar, R. Ben Enhancement of textile wastewater treatment process using Nanofiltration. Desalination 2012, 286, 16–23. [Google Scholar] [CrossRef]
- Eremektar, G.; Selcuk, H.; Meric, S. Investigation of the relation between COD fractions and the toxicity in a textile finishing industry wastewater: Effect of preozonation. Desalination 2007, 211, 314–320. [Google Scholar] [CrossRef]
- Sharma, K.P.; Sharma, S.; Sharma, S.; Singh, P.K.; Kumar, S.; Grover, R.; Sharma, P.K. A comparative study on characterization of textile wastewaters (untreated and treated) toxicity by chemical and biological tests. Chemosphere 2007, 69, 48–54. [Google Scholar] [CrossRef]
- Verma, Y. Acute toxicity assessment of textile dyes and textile and dye industrial effluents using Daphnia magna bioassay. Toxicol. Ind. Health 2008, 24, 491–500. [Google Scholar] [CrossRef]
- Tigini, V.; Giansanti, P.; Mangiavillano, A.; Pannocchia, A.; Varese, G.C. Evaluation of toxicity, genotoxicity and environmental risk of simulated textile and tannery wastewaters with a battery of biotests. Ecotoxicol. Environ. Saf. 2011, 74, 866–873. [Google Scholar] [CrossRef]
- Chhabra, M.; Mishra, S.; Sreekrishnan, T.R. Combination of chemical and enzymatic treatment for efficient decolorization/degradation of textile effluent: High operational stability of the continuous process. Biochem. Eng. J. 2015, 93, 17–24. [Google Scholar] [CrossRef]
- Mountassir, Y.; Benyaich, A.; Rezrazi, M.; Berçot, P.; Gebrati, L. Wastewater effluent characteristics from Moroccan textile industry. Water Sci. Technol. 2013, 67, 2791–2799. [Google Scholar] [CrossRef]
- Hussein, F.H. Photochemical treatments of textile industries wastewater. Asian J. Chem. 2012, 24, 5427–5434. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, W.; Zhang, J.; Zhao, H.; Zhang, Y.; Quan, X.; Jin, Y. Characterisation of acute toxicity, genotoxicity and oxidative stress posed by textile effluent on zebrafish. J. Environ. Sci. China 2012, 24, 2019–2027. [Google Scholar] [CrossRef]
- Yoo, J.; Ahn, B.; Oh, J.J.; Han, T.; Kim, W.K.; Kim, S.; Jung, J. Identification of toxicity variations in a stream affected by industrial effluents using Daphnia magna and Ulva pertusa. J. Hazard. Mater. 2013, 260, 1042–1049. [Google Scholar] [CrossRef]
- Tehrani-Bagha, A.R.; Mahmoodi, N.M.; Menger, F.M. Degradation of a persistent organic dye from colored textile wastewater by ozonation. Desalination 2010, 260, 34–38. [Google Scholar] [CrossRef]
- Ghaly, A.E.; Ananthashankar, R.; Alhattab, M.; Ramakrishnan, V. V Production, Characterization and Treatment of Textile Effluents: A Critical Review. J. Chem. Eng. Process Technol. 2014, 5, 1–18. [Google Scholar] [CrossRef]
- Sponza, D.T. Toxicity studies in a chemical dye production industry in Turkey. J. Hazard. Mater. 2006, 138, 438–447. [Google Scholar] [CrossRef]
- EC Directive 2000/60/EC of the European Parliament and of the Council establishing a framework for Community action in the field of water policy. Off. J. Eur. Communities 2000, L327, 1–73.
- EC Decision no 2455/2001/EC of the European Parliament and of the Council of 20 November of 2001 establishing the list of priority substances in the field of water policy and amending Directive 2000/60/EC. Off. J. Eur. Communities 2001, L331, 1–5.
- EC Directive 2008/105/EC of the European Parliament and of the Council of 16 December of 2008 on environmental quality standards in the field of water policy, amending and subsequently repealing Council Directives 82/176/EEC, 83/513/EEC, 84/156/EEC, 84/491/E. Off. J. Eur. Communities 2008, L348, 84–97.
- Diamond, J.; Altenburger, R.; Coors, A.; Dyer, S.D.; Focazio, M.; Kidd, K.; Koelmans, A.A.; Leung, K.M.Y.; Servos, M.R.; Snape, J.; et al. Use of prospective and retrospective risk assessment methods that simplify chemical mixtures associated with treated domestic wastewater discharges. Environ. Toxicol. Chem. 2018, 37, 690–702. [Google Scholar] [CrossRef]
- Maselli, B.D.S.; Luna, L.A.V.; Palmeira, J.D.O.; Tavares, K.P.; Barbosa, S.; Beijo, L.A.; Umbuzeiro, G.A.; Kummrow, F. Ecotoxicity of raw and treated effluents generated by a veterinary pharmaceutical company: A comparison of the sensitivities of different standardized tests. Ecotoxicology 2015, 24, 795–804. [Google Scholar] [CrossRef]
- Azur Environmental. Microtox Omnio Manual; Azur Environmental: Carlsbad, CA, USA, 1998. [Google Scholar]
- OECD. No. 201, Freshwater alga and cyanobacteria, growth inhibition test. In OECD Guidelines for the Testing of Chemicals; Organization for Economic Cooperation and Development: Paris, France, 2006. [Google Scholar]
- Geis, S.; Fleming, K.; Korthals, E.; Searle, G.; Reynolds, L.; Karner, D. Modifications to the algal growth inhibition test for use as a regulatory assay. Environ. Toxicol. Chem. 2000, 19, 36–41. [Google Scholar] [CrossRef]
- OECD. No. 221, Lemna sp. Growth inhibition test. In OECD Guidelines for the Testing of Chemicals; Organization for Economic Cooperation and Development: Paris, France, 2006. [Google Scholar]
- ASTM. Standard Pratice for Conducting Acute Toxicity Tests with Fishes, Macroinvertebrates and Amphibians; ASTM (American Society for Testing and Materials): Philadelphia, PA, USA, 1980. [Google Scholar]
- Baird, D.; Soares, A.; Girling, A.; Barber, I. The long-term maintenance of Daphnia magna Straus for use in ecotoxicity tests: Problems and prospects. In Proceedings of the first European Conference on Ecotoxicology, Lyngby, Denmark, 17–19 October 1988; pp. 144–148. [Google Scholar]
- OECD. No. 202, Daphnia sp., acute immobilization test. In OECD Guidelines for the Testing of Chemicals; Organization for Economic Cooperation and Development: Paris, France, 2004. [Google Scholar]
- McWilliam, R.; Baird, D.J. Postexposure feeding depression: A new toxicity endpoint for use in laboratory studies with Daphnia magna. Environ. Toxicol. Chem. 2002, 21, 1198–1205. [Google Scholar] [CrossRef]
- Finney, D.J. Probit Analysis, 3rd ed.; Cambridge University Press: Cambridge, UK, 1971. [Google Scholar]
- INETI. Guia Técnico do Sector téXtil; INETI: Lisboa, Portugal, 2000. [Google Scholar]
- WHO. Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2011; pp. 1–554. [Google Scholar]
- ME Decree-Law 236/98 Portuguese Legislation on Water Quality from the MInistry of Environment. D. Repúb. 1998, 176, 3676–3722.
- Pérez, M.; Torrades, F.; Domènech, X.; Peral, J. Fenton and photo-Fenton oxidation of textile effluents. Water Res. 2002, 36, 2703–2710. [Google Scholar] [CrossRef]
- Perez, P.; Pulgar, R.; Olea-Serrano, F.; Villalobos, M.; Rivas, A.; Metzler, M.; Pedraza, V.; Olea, N. The estrogenicity of bisphenol A-related diphenylalkanes with various substituents at the central carbon and the hydroxy groups. Environ. Health Perspect. 1998, 106, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Castillo, M.; Barceló, D. Analysis of industrial effluents to determine endocrine-disrupting chemicals. TrAC Trends Anal. Chem. 1997, 16, 574–583. [Google Scholar] [CrossRef]
- Castillo, M.; Barceló, D. Characterisation of organic pollutants in textile wastewaters and landfill leachate by using toxicity-based fractionation methods followed by liquid and gas chromatography coupled to mass spectrometric detection. Anal. Chim. Acta 2001, 426, 253–264. [Google Scholar] [CrossRef]
- Sharma, H.A.; Barber, J.T.; Ensley, H.E.; Polito, M.A. A comparison of the toxicity and metabolism of phenol and chlorinated phenols by Lemna gibba, with special reference to 2,4,5-trichlorophenol. Environ. Toxicol. Chem. 1997, 16, 346–350. [Google Scholar] [CrossRef]
- Tišler, T.; Zagorc-Končan, J. Comparative assessment of toxicity of phenol, formaldehyde, and industrial wastewater to aquatic organisms. Water. Air Soil Pollut. 1997, 97, 315–322. [Google Scholar] [CrossRef]
- Orvos, D.R.; Versteeg, D.J.; Inauen, J.; Capdevielle, M.; Rothenstein, A.; Cunningham, V. Aquatic toxicity of triclosan. Environ. Toxicol. Chem. 2002, 21, 1338–1349. [Google Scholar] [CrossRef]
- Calabrese, E.J. Hormesis: Why it is important to toxicology and toxicologists. Environ. Toxicol. Chem. 2008, 27, 1451–1474. [Google Scholar] [CrossRef]
- Galassi, S.; Benfenati, E. Fractionation and toxicity evaluation of waste waters. J. Chromatogr. A 2000, 889, 149–154. [Google Scholar] [CrossRef]
- Villegas-Navarro, A.; Romero González, M.C.; Rosas López, E.; Domínguez Aguilar, R.; Sachetin Marçal, W. Evaluation of Daphnia magna as an indicator of toxicity and treatment efficacy of textile wastewaters. Environ. Int. 1999, 25, 619–624. [Google Scholar] [CrossRef]
- Barata, C.; Alañon, P.; Gutierrez-Alonso, S.; Riva, M.C.; Fernández, C.; Tarazona, J.V. A Daphnia magna feeding bioassay as a cost effective and ecological relevant sublethal toxicity test for Environmental Risk Assessment of toxic effluents. Sci. Total Environ. 2008, 405, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Cleuvers, M.; Ratte, H.T. The importance of light intensity in algal tests with coloured substances. Water Res. 2002, 36, 2173–2178. [Google Scholar] [CrossRef]
- Blinova, I. Use of freshwater algae and duckweeds for phytotoxicity testing. Environ. Toxicol. 2004, 19, 425–428. [Google Scholar] [CrossRef] [PubMed]
| Raw Effluent (RE) | Treated Effluent 1 (TE1) | Treated Effluent 2 (TE2) | |
|---|---|---|---|
| Apparent Colour | Grey | Green | Green |
| Colour (PtCo) | 560 | 156 | 198 |
| pH | 3.33 | 7.36 | 6.76 |
| TDS (ppm) | 1215 | 1539 | 2322 |
| Conductivity (μS cm−1) | 2343 | 3085 | 4270 |
| Compound | Raw Effluent (RE) (μg L−1) | Treated Effluent 1 (TE1) (μg L−1) | Removal Percentage (%) |
|---|---|---|---|
| Phenol | 0.03 | 0.03 | 23.08 |
| Hexamine | 0.04 | 0.03 | 24.49 |
| Ethyl-tetramethyl-heptane | 0.02 | 0.01 | 29.29 |
| 2,6-Tert-butyl-quinone | 1.62 | 1.20 | 25.64 |
| Tetramethyl-dodecane | 0.02 | 0.01 | 29.27 |
| Ionol (2,6-di-tert-butyl-4 methyl-phenol) | 0.02 | 0.01 | 28.21 |
| Diethylphtalate | 0.09 | 0.07 | 21.67 |
| Nonylphenol | 0.36 | 0.27 | 26.95 |
| 3,5-Di-tert-butyl-4-hydroxi-benzoic acid | 0.03 | 0.02 | 30.65 |
| 2,6-Di-tert-butyl-4-ethyl-phenol | 1.69 | 1.23 | 26.89 |
| 2,6-Di-tert-butyl-4-methoxymethyl-phenol | 0.02 | 0.01 | 25.64 |
| Dimethyl-carboxymethylexyl-phtalate | 0.26 | 0.20 | 24.35 |
| Octadecane | 0.11 | 0.08 | 29.40 |
| Butyl-2-ethylhexyl-phtalate | 0.16 | 0.11 | 28.29 |
| Benzoic acid phenyl ester | 0.31 | 0.25 | 21.62 |
| 2,2-dimethyl-2-phenyl-acetophenone | 0.02 | 0.01 | 27.04 |
| Butyl-octyl-phtalate | 0.21 | 0.15 | 30.79 |
| 4,4′-Methylenebis-phenol | 0.02 | 0.02 | 26.83 |
| Benzil-quinoline | 0.02 | 0.01 | 25.64 |
| Docosane | 0.02 | 0.01 | 26.70 |
| Tributyl-O-acetylcitrate | 0.08 | 0.06 | 24.43 |
| Tricosane | 0.03 | 0.02 | 27.98 |
| 7-(Diethylamino)-4-methyl-2H-1-benzopyran-2-one | 0.13 | 0.09 | 31.92 |
| Benzyl-butyl-phtalate | 0.03 | 0.02 | 23.08 |
| Triphenyl-phosphonic acid | 0.25 | 0.19 | 24.31 |
| Diisooctylphtalate | 0.21 | 0.15 | 29.58 |
| Hexacosane | 0.26 | 0.19 | 25.85 |
| Triacontane | 0.66 | 0.47 | 29.41 |
| Dotriacontane | 0.39 | 0.28 | 28.31 |
| EC50 | EC20 | Reference | |
|---|---|---|---|
| RE | 7.18% (0.42–100) | 2.92% (0.152–56) | Present study |
| TE1 | HE < 20% (NT) | HE < 20% (NT) | |
| W1 | 36.40% (29.4–45.5) | - | [5] |
| W2 | HE < 50% (NT) | - |
| RE | TE1 | TE2 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| EC50 (%) | EC20 (%) | TU | EC50 (%) | EC20 (%) | TU | EC50 (%) | EC20 (%) | TU | |
| A. fischeri | 7.2 (0.4–100.0) | 2.92 (0.152–56) | 13.9 | NT | NT | <1 | - | - | - |
| R. subcapitata | NT | 79.5 (71.5–87.5) | <1 | NT | NT | <1 | NT | 98.5 (96.8–100.3) | <1 |
| L. minor (n° of fronds) | 95.7 (91.8–99.6) | 74.9 (69.2–80.7) | 1.04 | - | - | - | 82.9 (81.7–84.1) | 75.5 (73.6–77.4) | 1.22 |
| L. minor (dry biomass) | NT | 76.8 (65.8–87.8) | <1 | - | - | - | NT | 80.4 (76.3–84.6) | <1 |
| D. magna (immobilisation) | 24.2 (22.2–26.4) | - | 4.14 | NT | - | <1 | - | - | - |
| D. magna (feeding) | 18.2 (16.0–20.5) | 8.3 (6.4–10.2) | 5.48 | 68.4 (63.8–73.0) | 56.1 (49.6–62.6) | 1.46 | - | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castro, A.M.; Nogueira, V.; Lopes, I.; Rocha-Santos, T.; Pereira, R. Evaluation of the Potential Toxicity of Effluents from the Textile Industry before and after Treatment. Appl. Sci. 2019, 9, 3804. https://doi.org/10.3390/app9183804
Castro AM, Nogueira V, Lopes I, Rocha-Santos T, Pereira R. Evaluation of the Potential Toxicity of Effluents from the Textile Industry before and after Treatment. Applied Sciences. 2019; 9(18):3804. https://doi.org/10.3390/app9183804
Chicago/Turabian StyleCastro, Ana Margarida, Verónica Nogueira, Isabel Lopes, Teresa Rocha-Santos, and Ruth Pereira. 2019. "Evaluation of the Potential Toxicity of Effluents from the Textile Industry before and after Treatment" Applied Sciences 9, no. 18: 3804. https://doi.org/10.3390/app9183804
APA StyleCastro, A. M., Nogueira, V., Lopes, I., Rocha-Santos, T., & Pereira, R. (2019). Evaluation of the Potential Toxicity of Effluents from the Textile Industry before and after Treatment. Applied Sciences, 9(18), 3804. https://doi.org/10.3390/app9183804








