Application of Nitrogen and Oxygen Isotopes for Source and Fate Identification of Nitrate Pollution in Surface Water: A Review
Abstract
1. Introduction
2. Fundamental Principle of Using Nitrogen and Oxygen Stable Isotopes to Identify the Pollution Source and Fate of Nitrate of Surface Water
2.1. Compositions of Nitrogen and Oxygen Stable Isotopes
2.2. Isotopic Fractionation
3. Research Progress in Analytical Techniques of Nitrogen and Oxygen Isotopes
4. Application of Nitrogen and Oxygen Isotopes in Nitrate Source Tracing of Surface Water under Different Land-Use Types
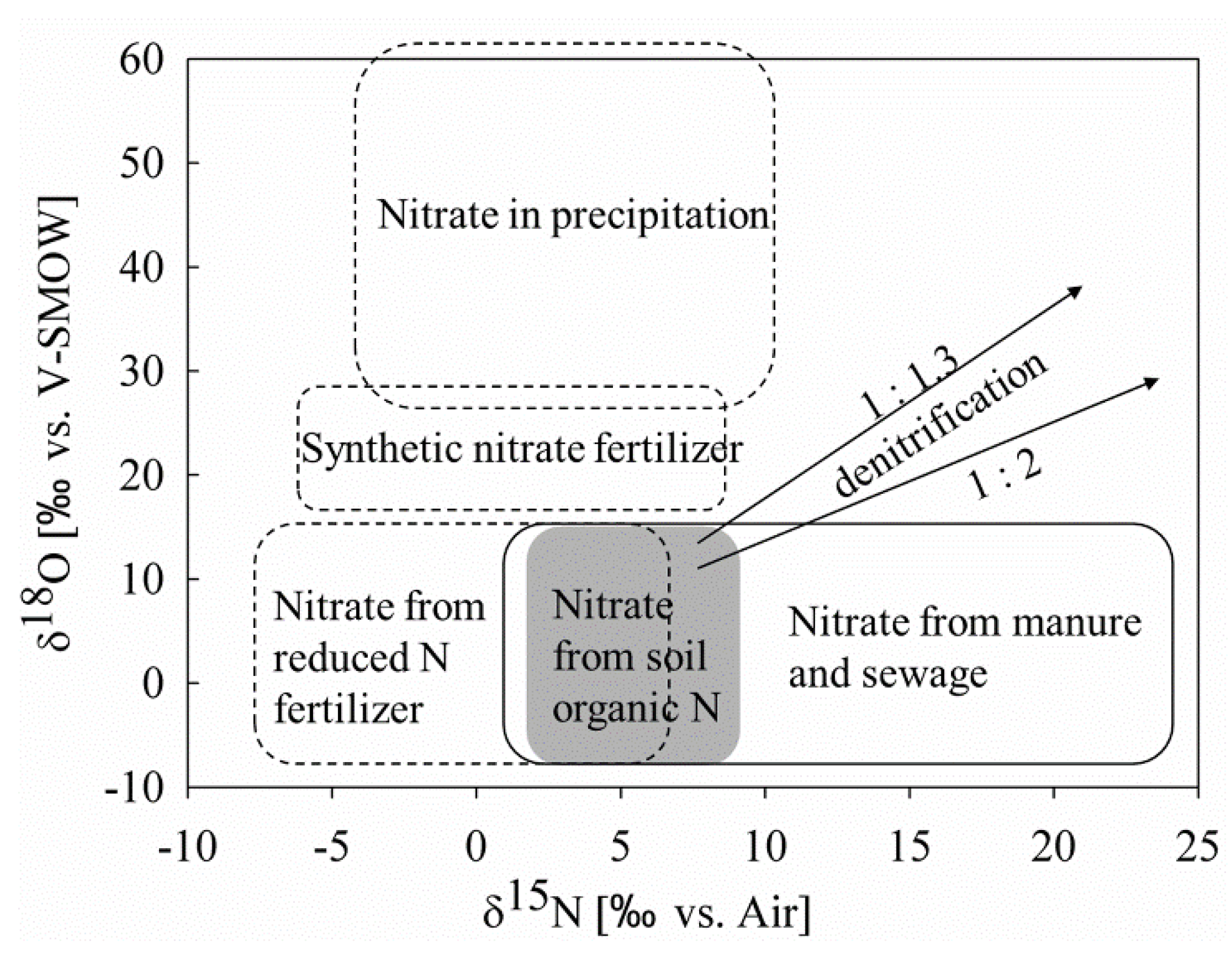
5. Source Identification of Nitrate in Surface Water by Nitrogen and Oxygen Isotope-Based Methods
6. Applications of Mathematical Models
6.1. Mass-Balance Mixing Model
6.2. SIAR Model
6.3. IsoSource Model
7. Conclusions and Suggestions
Funding
Conflicts of Interest
References
- Erisman, J.W.; Galloway, J.N.; Seitzinger, S.; Bleeker, A.; Dise, N.B.; Petrescu, A.M.R.; Leach, A.M.; de Vries, W. Consequences of human modification of the global nitrogen cycle. Philos. Trans. R. Soc. B-Boil. Sci. 2013, 368. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Kanae, S.; Oki, T.; Hirabayashi, Y.; Yamashiki, Y.; Takara, K. Assessment of global nitrogen pollution in rivers using an integrated biogeochemical modeling framework. Water Res. 2011, 45, 2573–2586. [Google Scholar] [CrossRef] [PubMed]
- Haas, M.B.; Guse, B.; Fohrer, N. Assessing the impacts of Best Management Practices on nitrate pollution in an agricultural dominated lowland catchment considering environmental protection versus economic development. J. Environ. Manag. 2017, 196, 347–364. [Google Scholar] [CrossRef]
- Zhang, Y.; Shi, P.; Li, F.; Wei, A.; Song, J.; Ma, J. Quantification of nitrate sources and fates in rivers in an irrigated agricultural area using environmental isotopes and a Bayesian isotope mixing model. Chemosphere 2018, 208, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, F.; Zhang, Q.; Li, J.; Liu, Q. Tracing nitrate pollution sources and transformation in surface- and ground-waters using environmental isotopes. Sci. Total Environ. 2014, 490, 213–222. [Google Scholar] [CrossRef]
- Bu, H.; Zhang, Y.; Meng, W.; Song, X. Effects of land-use patterns on in-stream nitrogen in a highly-polluted river basin in Northeast China. Sci. Total Environ. 2016, 553, 232–242. [Google Scholar] [CrossRef]
- Kohl, D.H.; Shearer, G.B.; Commoner, B. Fertilizer nitrogen: Contribution to nitrate in surface water in a corn belt watershed. Science 1971, 174, 1331–1334. [Google Scholar] [CrossRef]
- Stogbauer, A.; Strauss, H.; Arndt, J.; Marek, V.; Einsiedl, F.; van Geldern, R. Rivers of North-Rhine Westphalia revisited: Tracing changes in river chemistry. Appl. Geochem. 2008, 23, 3290–3304. [Google Scholar] [CrossRef]
- Townsend-Small, A.; McCarthy, M.J.; Brandes, J.A.; Yang, L.; Zhang, L.; Gardner, W.S. Stable isotopic composition of nitrate in Lake Taihu, China, and major inflow rivers. Hydrobiologia 2007, 581, 135–140. [Google Scholar] [CrossRef]
- Savard, M.M.; Somers, G.; Smirnoff, A.; Paradis, D.; van Bochove, E.; Liao, S. Nitrate isotopes unveil distinct seasonal N-sources and the critical role of crop residues in groundwater contamination. J. Hydrol. 2010, 381, 134–141. [Google Scholar] [CrossRef]
- Kendall, C. Tracing Nitrogen Sources and Cycling in Catchments. In Isotope Tracers in Catchment Hydrology, 1st ed.; Kendall, C., McDonnell, J.J., Eds.; Elsevier Science: Amsterdam, The Netherlands, 1998; pp. 519–576. [Google Scholar]
- Xue, D.; Botte, J.; De Baets, B.; Accoe, F.; Nestler, A.; Taylor, P.; Van Cleemput, O.; Berglund, M.; Boeckx, P. Present limitations and future prospects of stable isotope methods for nitrate source identification in surface- and groundwater. Water Res. 2009, 43, 1159–1170. [Google Scholar] [CrossRef] [PubMed]
- Finlay, J.C.; Sterner, R.W.; Kumar, S. Isotopic evidence for inlake production of accumulating nitrate in Lake Superior. Ecol. Appl. 2009, 17, 2323–2332. [Google Scholar] [CrossRef]
- Jia, G.D.; Chen, F.J. Monthly variations in nitrogen isotopes of ammonium and nitrate in wet deposition at Guangzhou, south China. Atmos. Environ. 2010, 44, 2309–2315. [Google Scholar] [CrossRef]
- Jiang, Y.J.; Wu, Y.X.; Yuan, D.X. Human impacts on karst groundwater contamination deduced by coupled nitrogen with strontium isotopes in the Nandong underground river system in Yunan, China. Environ. Sci. Technol. 2009, 43, 7676–7683. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.T.; Koba, K.; Wang, X.M.; Wen, D.Z.; Li, J.; Takebayashi, Y.; Liu, X.Y.; Yoh, M. Anthropogenic imprints on nitrogen and oxygen isotopic composition of precipitation nitrate in a nitrogen-polluted city in southern China. Atmos. Chem. Phys. 2011, 11, 1313–1325. [Google Scholar] [CrossRef]
- Felix, J.D.; Elliott, E.M.; Avery, G.B.; Kieber, R.J.; Mead, R.N.; Willey, J.D.; Mullaugh, K.M. Isotopic composition of nitrate in sequential Hurricane Irene precipitation samples: Implications for changing NOx sources. Atmos. Environ. 2015, 106, 191–195. [Google Scholar] [CrossRef]
- Xing, M.; Liu, W. Using dual isotopes to identify sources and transformations of nitrogen in water catchments with different land uses, Loess Plateau of China. Environ. Sci. Pollut. Res. 2016, 23, 388–401. [Google Scholar] [CrossRef] [PubMed]
- Li, S.L.; Liu, C.Q.; Li, J.; Liu, X.; Chetelat, B.; Wang, B.; Wang, F. Assessment of the sources of nitrate in the Changjiang River, China using a nitrogen and oxygen isotopic approach. Environ. Sci. Technol. 2010, 44, 1573–1578. [Google Scholar] [CrossRef]
- Michalski, G.; Kolanowski, M.; Riha, K.M. Oxygen and nitrogen isotopic composition of nitrate in commercial fertilizers, nitric acid, and reagent salts. Isot. Environ. Health Stud. 2015, 51, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Granger, J.; Wankel, S.D. Isotopic overprinting of nitrification on denitrification as a ubiquitous and unifying feature of environmental nitrogen cycling. Proc. Natl. Acad. Sci. USA 2016, 113, E6391–E6400. [Google Scholar] [CrossRef] [PubMed]
- Kellman, L.M. A study of tile drain nitrate-δ15N values as a tool for assessing nitrate sources in an agricultural region. Nutr. Cycl. Agroecosyst. 2005, 71, 131–137. [Google Scholar] [CrossRef]
- Kendall, C.; Elliott, E.M.; Wankel, S.D. Tracing anthropogenic inputs of nitrogen to ecosystems. Stable Isot. Ecol. Environ. Sci. 2007, 2, 375–449. [Google Scholar] [CrossRef]
- Rivett, M.O.; Buss, S.R.; Morgan, P.; Smith, J.W.N.; Bemment, C.D. Nitrate attenuation in groundwater: A review of biogeochemical controlling processes. Water Res. 2008, 42, 4215–4232. [Google Scholar] [CrossRef] [PubMed]
- Sebilo, M.; Billen, G.; Grably, M.; Mariotti, A. Isotopic composition of nitrate-nitrogen as a marker of riparian and benthic denitrification at the scale of the whole Seine River system. Biogeochemistry 2003, 63, 35–51. [Google Scholar] [CrossRef]
- Wenk, C.B.; Zopfi, J.; Blees, J.; Veronesi, M.; Niemann, H.; Lehmann, M.F. Community N and O isotope fractionation by sulfide-dependent denitrification and anammox in a stratified lacustrine water column. Geochim. Cosmochim. Acta 2014, 125, 551–563. [Google Scholar] [CrossRef]
- Kaushal, S.S.; Groffman, P.M.; Band, L.E.; Elliott, E.M.; Shields, C.A.; Kendall, C. Tracking Nonpoint Source Nitrogen Pollution in Human-Impacted Watersheds. Environ. Sci. Technol. 2011, 45, 8225–8232. [Google Scholar] [CrossRef]
- Burns, D.A.; Boyer, E.W.; Elliott, E.M.; Kendall, C. Sources and Transformations of Nitrate from Streams Draining Varying Land Uses: Evidence from Dual Isotope Analysis. J. Environ. Qual. 2009, 38, 1149–1159. [Google Scholar] [CrossRef]
- Osaka, K.i.; Ohte, N.; Koba, K.; Yoshimizu, C.; Katsuyama, M.; Tani, M.; Tayasu, I.; Nagata, T. Hydrological influences on spatiotemporal variations of δ15N and δ18O of nitrate in a forested headwater catchment in central Japan: Denitrification plays a critical role in groundwater. J. Geophys. Res. Biogeosci. 2010, 115. [Google Scholar] [CrossRef]
- Robinson, D. δ15N as an integrator of the nitrogen cycle. Trends Ecol. Evol. 2001, 16, 153–162. [Google Scholar] [CrossRef]
- Granger, J.; Sigman, D.M.; Needoba, J.A.; Harrison, P.J. Coupled nitrogen and oxygen isotope fractionation of nitrate during assimilation by cultures of marine phytoplankton. Limnol. Oceanogr. 2004, 49, 1763–1773. [Google Scholar] [CrossRef]
- Nestler, A.; Berglund, M.; Accoe, F.; Duta, S.; Xue, D.; Boeckx, P.; Taylor, P. Isotopes for improved management of nitrate pollution in aqueous resources: Review of surface water field studies. Environ. Sci. Pollut. Res. 2011, 18, 519–533. [Google Scholar] [CrossRef]
- Seitzinger, P.S.; Styles, R.V.; Boyer, E.; Boyer, E.W.; Alexander, R.B.; Billen, G.; Howarth, R.W.; Mayer, B.; Breemen, N.V. Nitrogen retention in rivers: Model development and application to watersheds in the northeastern USA. Biogeochemistry 2002, 57–58, 199–237. [Google Scholar] [CrossRef]
- Deutsch, B.; Voss, M.; Fischer, H. Nitrogen transformation processes in the Elbe River: Distinguishing between assimilation and denitrification by means of stable isotope ratios in nitrate. Aquat. Sci. 2009, 71, 228–237. [Google Scholar] [CrossRef]
- Silva, S.R.; Kendall, C.; Wilkison, D.H.; Ziegler, A.C.; Chang, C.C.Y.; Avanzino, R.J. A new method for collection of nitrate from fresh water and the analysis of nitrogen and oxygen isotope ratios. J. Hydrol. 2000, 228, 22–36. [Google Scholar] [CrossRef]
- Garten, C.T. Nitrogen isotope composition of ammonium and nitrate in bulk precipitation and forest throughfall. Int. J. Environ. Anal. Chem. 1992, 47, 32–45. [Google Scholar] [CrossRef]
- Tu, Y.; Fang, Y.; Liu, D.; Pan, Y. Modifications to the azide method for nitrate isotope analysis. Rapid Commun. Mass Spectrom. 2016, 30, 1213–1222. [Google Scholar] [CrossRef] [PubMed]
- Li, W.B.; Song, Y.B.; Xu, H.K.; Chen, L.Y.; Dai, W.H.; Dong, M. Ion-exchange method in the collection of nitrate from freshwater ecosystems for nitrogen and oxygen isotope analysis: A review. Environ. Sci. Pollut. Res. 2015, 22, 9575–9588. [Google Scholar] [CrossRef] [PubMed]
- Sigman, D.; Casciotti, K.; Andreani, M.; Barford, C.; Galanter, M.; Böhlke, J. A bacterial method for the nitrogen isotopic analysis of nitrate in seawater and freshwater. Anal. Chem. 2001, 73, 4145–4153. [Google Scholar] [CrossRef]
- Casciotti, K.L.; Sigman, D.M.; Hastings, M.G.; Bohlke, J.K.; Hilkert, A. Measurement of the oxygen isotopic composition of nitrate in seawater and freshwater using the denitrifier method. Anal. Chem. 2002, 74, 4905–4912. [Google Scholar] [CrossRef]
- Coplen, T.B.; Qi, H.P.; Revesz, K.; Casciotti, K.; Hannon, J.E. Determination of the δ15N and δ18O of Nitrate in Water; RSIL Lab Code 2900; U.S. Geological Survey: Reston, VA, USA, 2007. [CrossRef]
- Granger, J.; Sigman, D.M.; Prokopenko, M.G.; Lehmann, M.F.; Tortell, P.D. A method for nitrite removal in nitrate N and O isotope analyses. Limnol. Oceanogr. Methods 2006, 4, 205–212. [Google Scholar] [CrossRef]
- Casciotti, K.L.; Boehlke, J.K.; McIlvin, M.R.; Mroczkowski, S.J.; Hannon, J.E. Oxygen isotopes in nitrite: Analysis, calibration, and equilibration. Anal. Chem. 2007, 79, 2427–2436. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Yu, L.; Bakken, L.R.; Morkved, P.T.; Mulder, J.; Dorsch, P. Controlled induction of denitrification in Pseudomonas aureofaciens: A simplified denitrifier method for dual isotope analysis in NO3. Sci. Total Environ. 2018, 633, 1370–1378. [Google Scholar] [CrossRef] [PubMed]
- McIlvin, M.R.; Altabet, M.A. Chemical conversion of nitrate and nitrite to nitrous oxide for nitrogen and oxygen isotopic analysis in freshwater and seawater. Anal. Chem. 2005, 77, 5589–5595. [Google Scholar] [CrossRef] [PubMed]
- Wassenaar, L.I.; Douence, C.; Altabet, M.A.; Aggarwal, P.K. N and O isotope (δ15Nα, δ15Nβ, δ18O, δ17O) analyses of dissolved NO3− and NO2− by the Cd-azide reduction method and N2O laser spectrometry. Rapid Commun. Mass Spectrom. 2018, 32, 184–194. [Google Scholar] [CrossRef]
- Deutsch, B.; Mewes, M.; Liskow, I.; Voss, M. Quantification of diffuse nitrate inputs into a small river system using stable isotopes of oxygen and nitrogen in nitrate. Org. Geochem. 2006, 37, 1333–1342. [Google Scholar] [CrossRef]
- Ding, J.; Xi, B.; Gao, R.; He, L.; Liu, H.; Dai, X.; Yu, Y. Identifying diffused nitrate sources in a stream in an agricultural field using a dual isotopic approach. Sci. Total Environ. 2014, 484, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Peña-Haro, S.; Llopis-Albert, C.; Pulido-Velazquez, M.; Pulido-Velazquez, D. Fertilizer standards for controlling groundwater nitrate pollution from agriculture: El Salobral-Los Llanos case study, Spain. J. Hydrol. 2010, 392, 174–187. [Google Scholar] [CrossRef]
- Parnell, A.C.; Inger, R.; Bearhop, S.; Jackson, A.L. Source partitioning using stable isotopes: Coping with too much variation. PLoS ONE 2010, 5, e9672. [Google Scholar] [CrossRef]
- Barnes, R.T.; Raymond, P.A.; Casciotti, K.L. Dual isotope analyses indicate efficient processing of atmospheric nitrate by forested watersheds in the northeastern US. Biogeochemistry 2008, 90, 15–27. [Google Scholar] [CrossRef]
- Hales, H.C.; Ross, D.S.; Lini, A. Isotopic signature of nitrate in two contrasting watersheds of Brush Brook, Vermont, USA. Biogeochemistry 2007, 84, 51–66. [Google Scholar] [CrossRef]
- Buda, A.R.; DeWalle, D.R. Dynamics of stream nitrate sources and flow pathways during stormflows on urban, forest and agricultural watersheds in central Pennsylvania, USA. Hydrol. Process. 2009, 23, 3292–3305. [Google Scholar] [CrossRef]
- Chen, Z.X.; Liu, G.; Liu, W.G.; Lam, M.H.W.; Liu, G.J.; Yin, X.B. Identification of nitrate sources in Taihu Lake and its major inflow rivers in China, using 15N-NO3− and δ18O-NO3− values. Water Sci. Technol. 2012, 66, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.S.; Bong, Y.S.; Lee, D.; Kim, Y.; Kim, K. Tracing the sources of nitrate in the Han River watershed in Korea, using δ15N-NO3− and δ18O-NO3− values. Sci. Total Environ. 2008, 395, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Xu, Z.; Zhang, X.; Wang, X.; Sun, X.; Ballantine, D.J.; Wang, S. Nitrogen pollution and source identification of urban ecosystem surface water in Beijing. Front. Environ. Sci. Eng. 2014, 8, 106–116. [Google Scholar] [CrossRef]
- Popescu, R.; Mimmo, T.; Dinca, O.R.; Capici, C.; Costinel, D.; Sandru, C.; Ionete, R.E.; Stefanescu, I.; Axente, D. Using stable isotopes in tracing contaminant sources in an industrial area: A case study on the hydrological basin of the Olt River, Romania. Sci. Total Environ. 2015, 533, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Archana, A.; Thibodeau, B.; Geeraert, N.; Xu, M.N.; Kao, S.-J.; Baker, D.M. Nitrogen sources and cycling revealed by dual isotopes of nitrate in a complex urbanized environment. Water Res. 2018, 142, 459–470. [Google Scholar] [CrossRef] [PubMed]
- Battaglin, W.A.; Kendall, C.; Chang, C.C.Y.; Silva, S.R.; Campbell, D.H. Chemical and isotopic evidence of nitrogen transformation in the Mississippi River, 1997–1998. Hydrol. Process. 2001, 15, 1285–1300. [Google Scholar] [CrossRef]
- Liu, T.; Wang, F.; Michalski, G.; Xia, X.; Liu, S. Using 15N, 17O, and 18O to determine nitrate sources in the Yellow River, China. Environ. Sci. Technol. 2013, 47, 13412–13421. [Google Scholar] [CrossRef] [PubMed]
- Yue, F.J.; Liu, C.Q.; Li, S.L.; Zhao, Z.Q.; Liu, X.L.; Ding, H.; Liu, B.J.; Zhong, J. Analysis of δ15N and δ18O to identify nitrate sources and transformations in Songhua River, Northeast China. J. Hydrol. 2014, 519, 329–339. [Google Scholar] [CrossRef]
- Panno, S.V.; Kelly, W.R.; Hackley, K.C.; Hwang, H.H.; Martinsek, A.T. Sources and fate of nitrate in the Illinois River Basin, Illinois. J. Hydrol. 2008, 359, 174–188. [Google Scholar] [CrossRef]
- Rock, L.; Mayer, B. Isotopic assessment of sources of surface water nitrate within the Oldman River basin, Southern Alberta, Canada. In Biogeochemical Investigations of Terrestrial, Freshwater, and Wetland Ecosystems across the Globe, 2nd ed.; Wieder, R.K., Novák, M., Vile, M.A., Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 545–562. [Google Scholar]
- Soto, D.X.; Koehler, G.; Wassenaar, L.I.; Hobson, K.A. Spatio-temporal variation of nitrate sources to Lake Winnipeg using N and O isotope (δ15N, δ18O) analyses. Sci. Total Environ. 2019, 647, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Urresti-Estala, B.; Vadillo-Perez, I.; Jimenez-Gavilan, P.; Soler, A.; Sanchez-Garcia, D.; Carrasco-Cantos, F. Application of stable isotopes (δ34S-SO4, δ18O-SO4, δ15N-NO3, δ18O-NO3) to determine natural background and contamination sources in the Guadalhorce River Basin (southern Spain). Sci. Total Environ. 2015, 506, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Xue, D.; De Baets, B.; Van Cleemput, O.; Hennessy, C.; Berglund, M.; Boeckx, P. Classification of nitrate polluting activities through clustering of isotope mixing model outputs. J. Environ. Qual. 2013, 42, 1486–1497. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Jia, G.; Chen, J. Nitrate sources and watershed denitrification inferred from nitrate dual isotopes in the Beijiang River, south China. Biogeochemistry 2009, 94, 163–174. [Google Scholar] [CrossRef]
- Mayer, B.; Boyer, E.W.; Goodale, C.; Jaworski, N.A.; Van Breemen, N.; Howarth, R.W.; Seitzinger, S.; Billen, G.; Lajtha, L.J.; Nosal, M.; et al. Sources of nitrate in rivers draining sixteen watersheds in the northeastern US: Isotopic constraints. Biogeochemistry 2002, 57, 171–197. [Google Scholar] [CrossRef]
- Showers, W.J.; Genna, B.; McDade, T.; Bolich, R.; Fountain, J.C. Nitrate contamination in groundwater on an urbanized dairy farm. Environ. Sci. Technol. 2008, 42, 4683–4688. [Google Scholar] [CrossRef] [PubMed]
- Marfia, A.M.; Krishnamurthy, R.V.; Atekwana, E.A.; Panton, W.F. Isotopic and geochemical evolution of ground and surface waters in a karst dominated geological setting: A case study from Belize, Central America. Appl. Geochem. 2004, 19, 937–946. [Google Scholar] [CrossRef]
- Li, C.; Jiang, Y.; Guo, X.; Cao, Y.; Ji, H. Multi-isotope (15N, 18O and 13C) indicators of sources and fate of nitrate in the upper stream of Chaobai River, Beijing, China. Environ. Sci. Process. Impacts 2014, 16, 2644–2655. [Google Scholar] [CrossRef]
- Widory, D.; Petelet-Giraud, E.; Brenot, A.; Bronders, J.; Tirez, K.; Boeckx, P. Improving the management of nitrate pollution in water by the use of isotope monitoring: The δ15N, δ18O and δ11B triptych. Isot. Environ. Health Stud. 2013, 49, 29–47. [Google Scholar] [CrossRef]
- Michalski, G.; Scott, Z.; Kabiling, M.; Thiemens, M.H. First measurements and modeling of Δ17O in atmospheric nitrate. Geophys. Res. Lett. 2003, 30, 1870. [Google Scholar] [CrossRef]
- Tsunogai, U.; Daita, S.; Komatsu, D.D.; Nakagawa, F.; Tanaka, A. Quantifying nitrate dynamics in an oligotrophic lake using Δ17O. Biogeosciences 2011, 8, 687–702. [Google Scholar] [CrossRef]
- Saccon, P.; Leis, A.; Marca, A.; Kaiser, J.; Campisi, L.; Böttcher, M.E.; Savarino, J.; Escher, P.; Eisenhauer, A.; Erbland, J. Multi-isotope approach for the identification and characterisation of nitrate pollution sources in the Marano lagoon (Italy) and parts of its catchment area. Appl. Geochem. 2013, 34, 75–89. [Google Scholar] [CrossRef]
- Wollheim, W.M.; Mulukutla, G.K.; Cook, C.; Carey, R.O. Aquatic nitrate retention at river network scales across flow conditions determined using nested in situ sensors. Water Resour. Res. 2017, 53, 9740–9756. [Google Scholar] [CrossRef]
- Xue, D.M.; Pang, F.M.; Meng, F.Q.; Wang, Z.L.; Wu, W.L. Decision-tree-model identification of nitrate pollution activities in groundwater: A combination of a dual isotope approach and chemical ions. J. Contam. Hydrol. 2015, 180, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Phillips, D.L.; Gregg, J.W. Uncertainty in source partitioning using stable isotopes. Oecologia 2001, 127, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Serio, F.; Miglietta, P.P.; Lamastra, L.; Ficocelli, S.; Intini, F.; De Leo, F.; De Donno, A. Groundwater nitrate contamination and agricultural land use: A grey water footprint perspective in Southern Apulia Region (Italy). Sci. Total Environ. 2018, 645, 1425–1431. [Google Scholar] [CrossRef] [PubMed]
- Phillips, D.L.; Koch, P.L. Incorporating concentration dependence in stable isotope mixing models. Oecologia 2002, 130, 114–125. [Google Scholar] [CrossRef]
- Moore, J.W.; Semmens, B.X. Incorporating uncertainty and prior information into stable isotope mixing models. Ecol. Lett. 2008, 11, 470–480. [Google Scholar] [CrossRef]
- Voss, M.; Deutsch, B.; Elmgren, R.; Humborg, C.; Kuuppo, P.; Pastuszak, M.; Rolff, C.; Schulte, U. Source identification of nitrate by means of isotopic tracers in the Baltic Sea catchments. Biogeosciences 2006, 3, 663–676. [Google Scholar] [CrossRef]
- Xue, D.; De Baets, B.; Van Cleemput, O.; Hennessy, C.; Berglund, M.; Boeckx, P. Use of a Bayesian isotope mixing model to estimate proportional contributions of multiple nitrate sources in surface water. Environ. Pollut. 2012, 161, 43–49. [Google Scholar] [CrossRef]
- Zeng, Y.; Tian, S.; Pan, Y. Revealing the Sources of Atmospheric Ammonia: A Review. Curr. Pollut. Rep. 2018, 4, 189–197. [Google Scholar] [CrossRef]
- Derrien, M.; Kim, M.S.; Ock, G.; Hong, S.; Cho, J.; Shin, K.H.; Hur, J. Estimation of different source contributions to sediment organic matter in an agricultural-forested watershed using end member mixing analyses based on stable isotope ratios and fluorescence spectroscopy. Sci. Total Environ. 2018, 618, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Herwig, B.R.; Wahl, D.H.; Dettmers, J.M.; Soluk, D.A. Spatial and temporal patterns in the food web structure of a large floodplain river assessed using stable isotopes. Can. J. Fish. Aquat. Sci. 2007, 64, 495–508. [Google Scholar] [CrossRef]
- Lu, L.; Li, W.L.; Pei, J.G.; Wang, Z. A quantitative study of the sources of nitrate of Zhaidi Underground River in Guilin based on IsoSource. Acta Geosci. Sin. 2014, 35, 248–254. [Google Scholar] [CrossRef]
- Sheng, T.; Yang, P.H.; Xie, G.W.; Hong, A.H.; Cao, C.; Xie, S.Y.; Shi, W.Y. Nitrate-nitrogen pollution sources of and underground river in Karst agricultural area using 15N and 18O isotope technique. Environ. Sci. 2018, 39, 4547–4555. [Google Scholar] [CrossRef]
| Watersheds | Area (km2) | Main Sources | References |
|---|---|---|---|
| Mississippi River Basin, United States | 2.9 × 106 | Stream nitrogen assimilation | [59] |
| Changjiang River Basin, China | 1.8 × 106 | Nitrification (including “modified fertilizer”) and urban sewage effluent | [19] |
| Yellow River Basin, China | 7.5 × 105 | Sewage/manure effluents in the upstream; manure/sewage effluents and ammonium/urea-containing fertilizer in the middle and lower reaches | [60] |
| Songhua River Basin, China | 5.6 × 105 | Soil organic nitrogen, nitrogenous fertilizers, and sewage in the high flow season; soil organic nitrogen and sewage in the low flow season. | [61] |
| Illinois River Basin, United States | 7.8 × 104 | A mixture of relatively undenitrified tile drain waters, highly denitrified groundwater, and treated wastewater | [62] |
| Oldman River basin, Canada | 3.0 × 104 | Soil nitrification in the western tributaries; manure and/or sewage in the eastern tributaries | [63] |
| Lake Winnipeg, Canada | 2.45 × 104 | Manure and/or wastewater discharge and inorganic agricultural fertilizers | [64] |
| Guadalhorce River Basin, Spain | 3.2 × 103 | Fertilizers and organic sources (manure and sewage) | [65] |
| Taihu Lake, China | 2.3 × 103 | Sewage/manure, and soil organic N in winter; atmospheric precipitation and sewage/manure inputs in summer | [54] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Shi, P.; Song, J.; Li, Q. Application of Nitrogen and Oxygen Isotopes for Source and Fate Identification of Nitrate Pollution in Surface Water: A Review. Appl. Sci. 2019, 9, 18. https://doi.org/10.3390/app9010018
Zhang Y, Shi P, Song J, Li Q. Application of Nitrogen and Oxygen Isotopes for Source and Fate Identification of Nitrate Pollution in Surface Water: A Review. Applied Sciences. 2019; 9(1):18. https://doi.org/10.3390/app9010018
Chicago/Turabian StyleZhang, Yan, Peng Shi, Jinxi Song, and Qi Li. 2019. "Application of Nitrogen and Oxygen Isotopes for Source and Fate Identification of Nitrate Pollution in Surface Water: A Review" Applied Sciences 9, no. 1: 18. https://doi.org/10.3390/app9010018
APA StyleZhang, Y., Shi, P., Song, J., & Li, Q. (2019). Application of Nitrogen and Oxygen Isotopes for Source and Fate Identification of Nitrate Pollution in Surface Water: A Review. Applied Sciences, 9(1), 18. https://doi.org/10.3390/app9010018





