Effect of Functionally-Graded Calcium Titanate Film, Prepared by Metal-Organic Chemical Vapor Deposition, on Titanium Implant
Abstract
1. Introduction
2. Materials and Methods
2.1. Specimen Design
2.2. Surface Characterization
2.3. Cell Culture on the Substrates
2.3.1. Cell Proliferation Assay
2.3.2. ALP Activity Measurement
2.4. Animal Experiments
Push-in Test
2.5. Statistical Analysis
3. Results
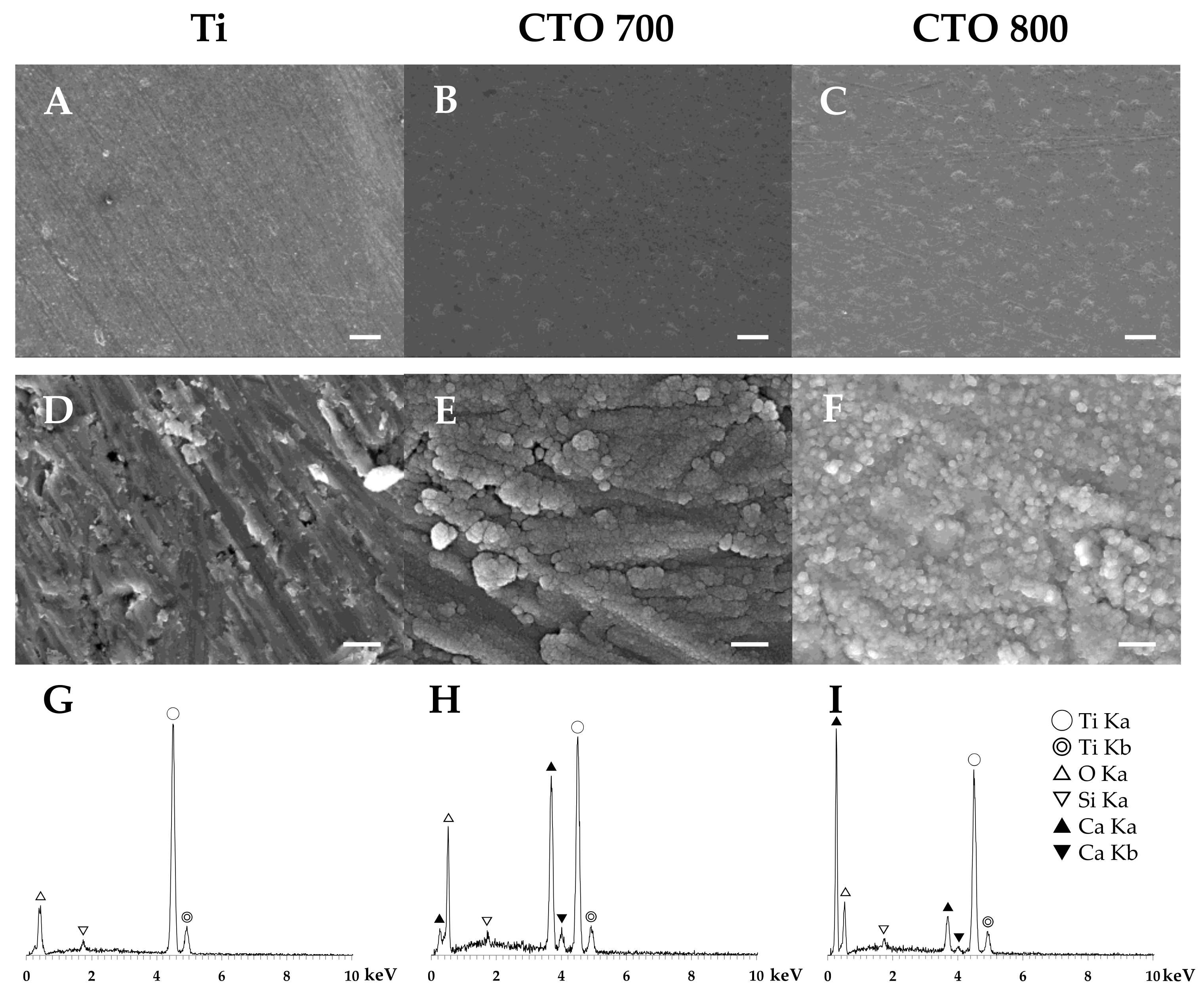
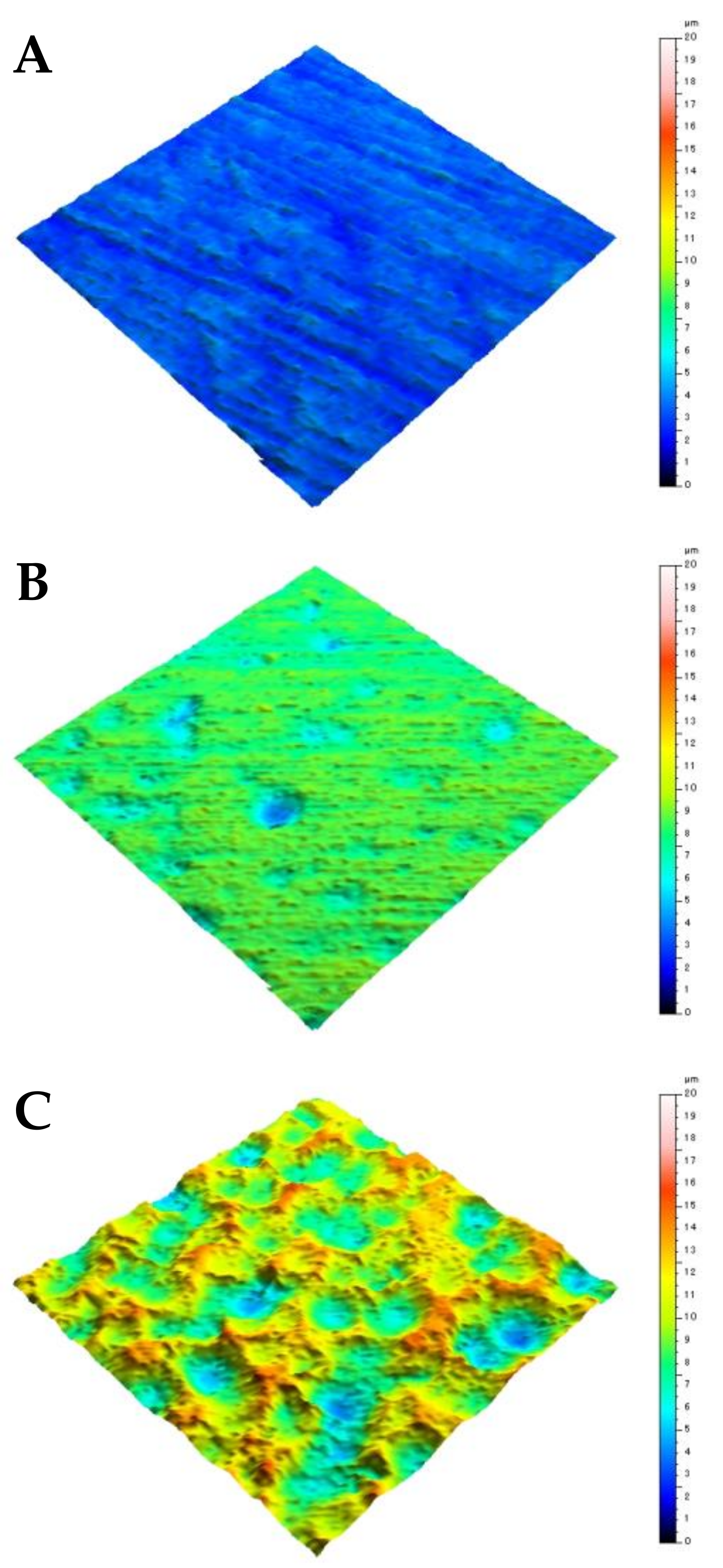
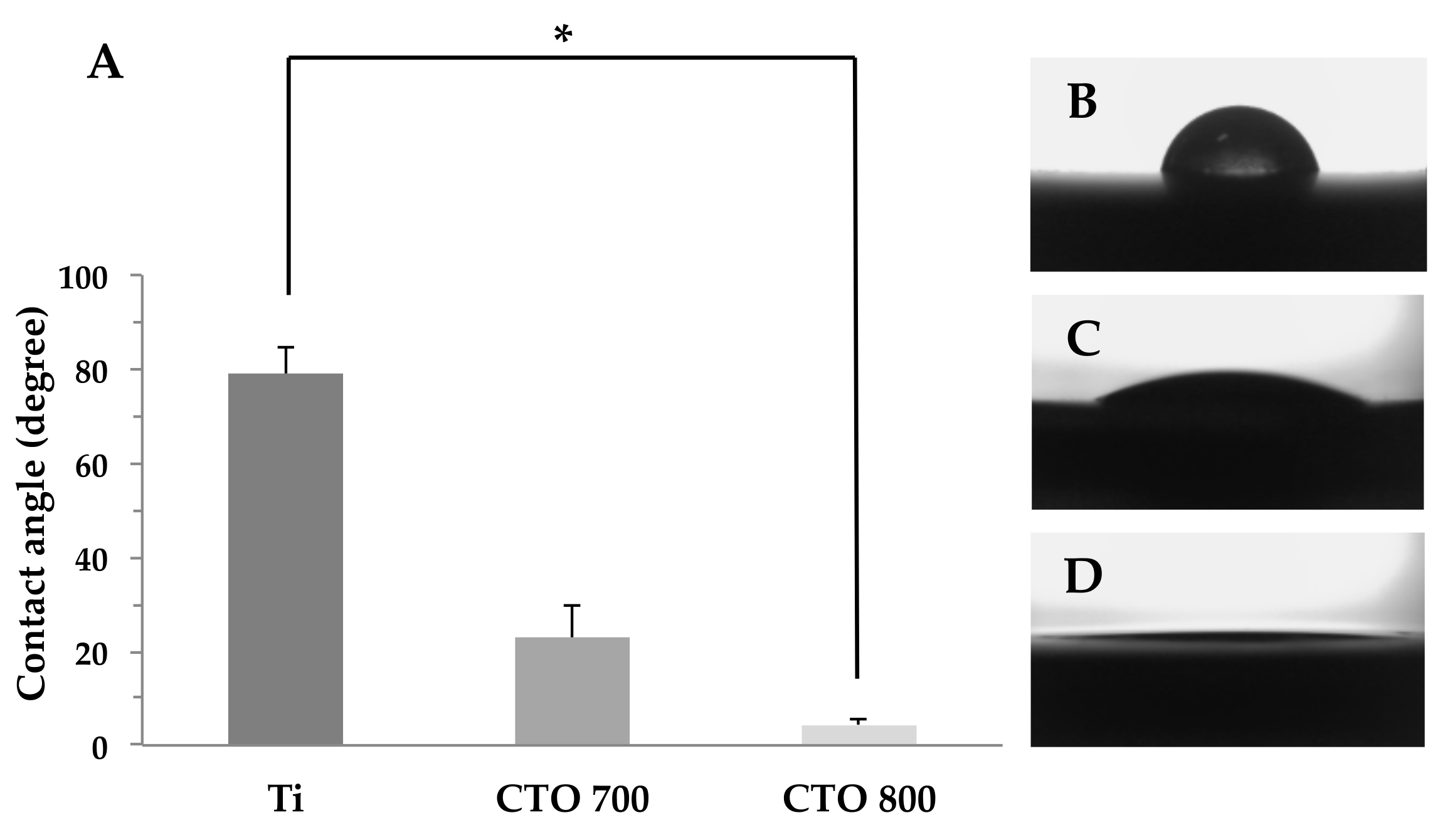
3.1. Surface Characteristics
3.1.1. Surface Morphology
3.1.2. Surface Roughness Measurement
3.1.3. Hydrophilicity Measurement
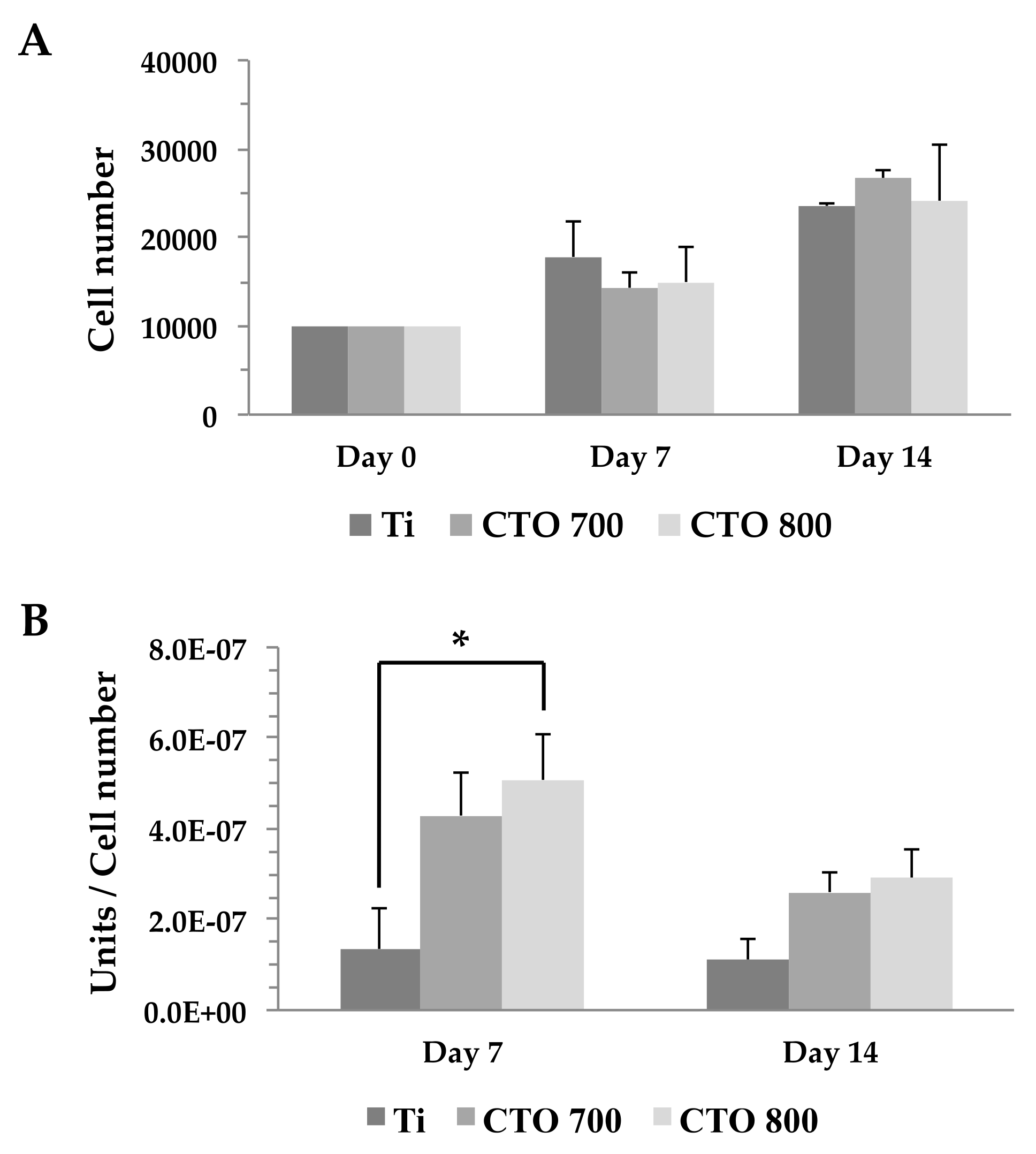
3.2. Cell Activities
3.2.1. Cell Proliferation Assay
3.2.2. Alkaline Phosphatase (ALP) Activity Measurement
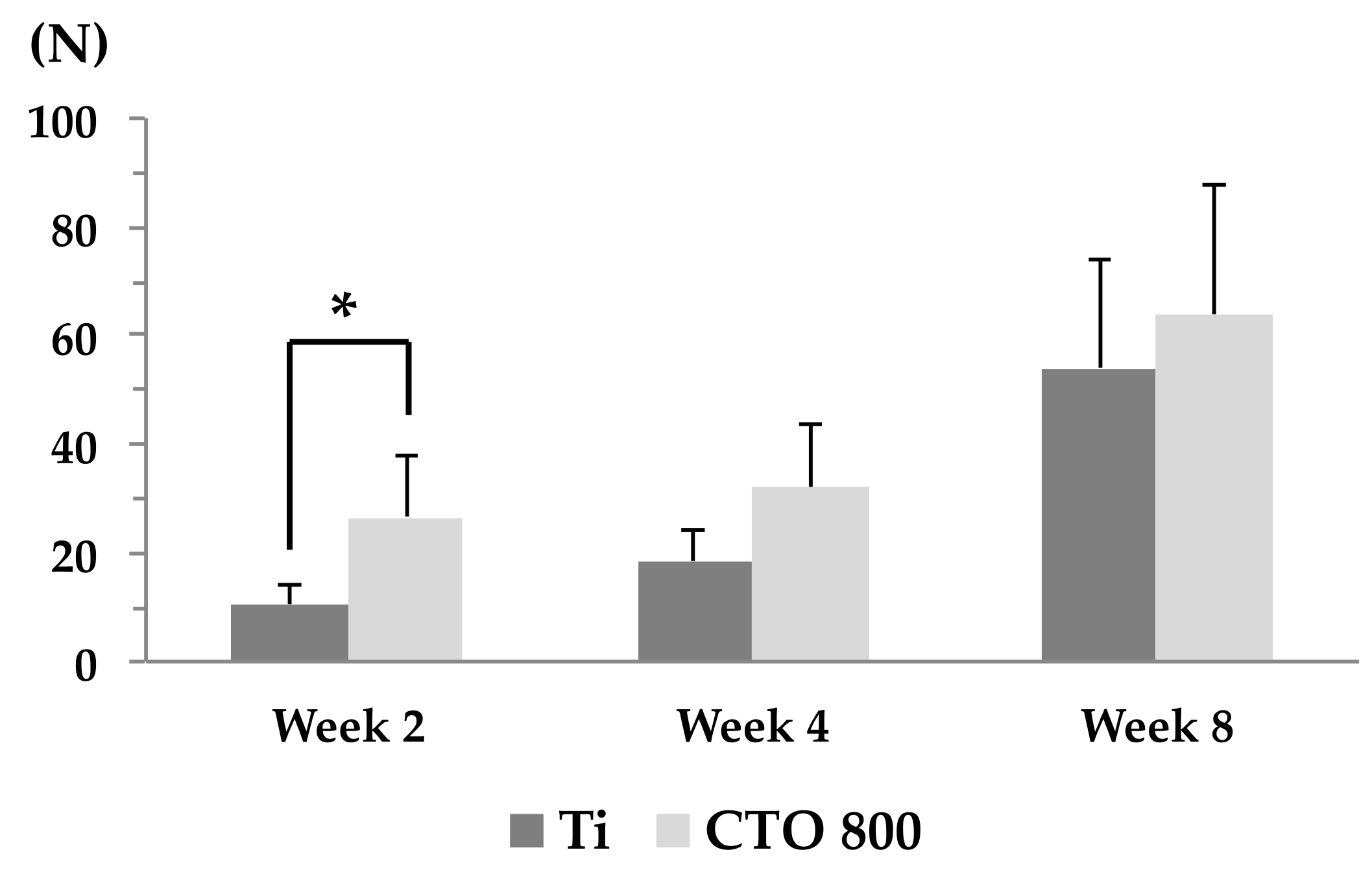
3.3. Push-in Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dumbleton, J.; Manley, M.T. Hydroxyapatite-coated prostheses in total hip and knee arthroplasty. J. Bone. Jt. Surq. A 2004, 86, 2526–2540. [Google Scholar] [CrossRef]
- Surmenev, R.A.; Surmeneva, M.A.; Ivanova, A.A. Significance of calcium phosphate coatings for the enhancement of new bone osteogenesis—A review. Acta Biomater. 2014, 10, 557–579. [Google Scholar] [CrossRef]
- Rahbek, O.; Overgaard, S.; Jensen, T.B.; Bendix, K.; Soballe, K. Sealing effect of hydroxyapatite coating: A 12-month study in canines. Acta Orthop. Scand. 2000, 71, 563–573. [Google Scholar] [CrossRef]
- Cunningham, B.W.; Hu, N.; Zorn, C.M.; McAfee, P.C. Bioactive titanium calcium phosphate coating for disc arthroplasty: Analysis of 58 vertebral end plates after 6 to 12-month implantation. Spine. J. 2009, 9, 836–845. [Google Scholar] [CrossRef]
- Massaro, C.; Baker, M.A.; Cosentino, F.; Ramires, P.A.; Klose, S.; Milella, E. Surface and biological evaluation of hydroxyapatite-based coatings on titanium deposited by different techniques. J. Biomed. Mater. Res. 2001, 58, 651–657. [Google Scholar] [CrossRef]
- Lee, I.S.; Kim, D.H.; Kim, H.E.; Jung, Y.C.; Han, C.H. Biological performance of calcium phosphate films formed on commercially pure Ti by electron-beam evaporation. Biomaterials 2002, 23, 609–615. [Google Scholar] [CrossRef]
- Yu, L.G.; Khor, K.A.; Li, H.; Cheang, P. Effect of spark plasma sintering on the microstructure and in vitro behavior of plasma sprayed HA coatings. Biomaterials 2003, 24, 2695–2705. [Google Scholar] [CrossRef]
- Ohtsu, N.; Sato, K.; Saito, K.; Asami, K.; Hanawa, T. Calcium phosphates formation on CaTiO3 coated titanium. J. Mater. Sci. Mater. Med. 2007, 18, 1009–1016. [Google Scholar] [CrossRef]
- Holliday, S.; Stanishevsky, A. Crystallization of CaTiO3 by sol-gel synthesis and rapid thermal processing. Surf. Coat. Technol. 2004, 188–189, 741–744. [Google Scholar] [CrossRef]
- Webster, T.J.; Ergun, C.; Doremus, R.H.; Lanford, W.A. Increased osteoblast adhesion on titanium-coated hydroxylapatite that forms CaTiO3. J. Biomed. Mater. Res. Part A 2003, 67, 975–980. [Google Scholar] [CrossRef]
- Coreño, J.; Coreño, O. Evaluation of calcium titanate as apatite growth promoter. J. Biomed. Mater. Res. Part A 2005, 75, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Ohtsu, N.; Sato, K.; Saito, K.; Hanawa, T.; Asami, K. Evaluation of degradability of CaTiO3 thin films in simulated body fluids. Mater. Trans. 2004, 45, 1778–1781. [Google Scholar] [CrossRef]
- Sato, M.; Tu, R.; Goto, T.; Ueda, K.; Narushima, T. Hydroxyapatite formation on CaTiO3 film prepared by metal-organic chemical vapor deposition. Mater. Trans. 2007, 48, 1505–1510. [Google Scholar] [CrossRef]
- Sato, M.; Tu, R.; Goto, T. Preparation conditions of CaTiO3 film by metal-organic chemical vapor deposition. Mater. Trans. 2006, 47, 1386–1390. [Google Scholar] [CrossRef]
- Ogawa, T.; Ozawa, S.; Shih, J.H.; Ryu, K.H.; Sukotjo, C.; Yang, J.M.; Nishimura, I. Biomechanical evaluation of osseous implants having different surface topographies in rats. J. Dent. Res. 2000, 79, 1857–1863. [Google Scholar] [CrossRef]
- Rupp, F.; Gittens, R.A.; Scheideler, L.; Scheideler, L.; Marmur, A.; Boyan, B.D.; Schwartz, Z.; Geis-Gerstorfer, J. A review on the wettability of dental implant surfaces I: Theoretical and experimental aspects. Acta Biomater. 2014, 10, 2894–2906. [Google Scholar] [CrossRef] [PubMed]
- Wennerberg, A.; Albrektsson, T. Effects of titanium surface topography on bone integration: A systematic review. Clin. Oral. Implants. Res. 2009, 20, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Andrukhov, O.; Huber, R.; Shi, B.; Berner, S.; Rausch-Fan, X.; Moritz, A.; Spencer, N.D.; Schedle, A. Proliferation, behavior, and differentiation of osteoblasts on surfaces of different microroughness. Dent. Mater. 2016, 32, 1374–1384. [Google Scholar] [CrossRef]
- Cheng, B.; Niu, Q.; Cui, Y.; Jiang, W.; Zhao, Y.; Kong, L. Effects of different hierarchical hybrid micro/nanostructure surfaces on implant osseointegration. Clin. Implant. Dent. Relat. Res. 2017, 19, 539–548. [Google Scholar] [CrossRef]
- Kim, B.S.; Shin, S.; Shin, S.J.; Kim, K.M.; Cho, H.H. Micro-nano hybrid structures with manipulated wettability using a two-step silicon etching on a large area. Nanoscale. Res. Lett. 2011, 6, 1–10. [Google Scholar] [CrossRef]
- Kim, D.S.; Lee, B.K.; Yeo, J.; Choi, M.J.; Yang, W.; Kwon, T.H. Fabrication of PDMS micro/nano hybrid surface for increasing hydrophobicity. Microelectron. Eng. 2009, 86, 1375–1378. [Google Scholar] [CrossRef]
- Eriksson, C.; Nygren, H.; Ohlson, K. Implantation of hydrophilic and hydrophobic titanium discs in rat tibia: Cellular reactions on the surfaces during the first 3 weeks in bone. Biomaterals 2004, 25, 4759–4766. [Google Scholar] [CrossRef] [PubMed]
- Aita, H.; Att, W.; Ueno, T.; Yamada, M.; Hori, N.; Iwasa, F.; Tsukimura, N.; Ogawa, T. Ultraviolet light-mediated photofunctionalization of titanium to promote human mesenchymal stem cell migration, attachment, proliferation and differentiation. Acta Biomater. 2009, 5, 3247–3257. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, S.B.; Washburn, N.R.; Simon, C.G., Jr.; Amis, E.J. Combinatorial screen of the effect of surface energy on fibronectin-mediated osteoblast adhesion, spreading and proliferation. Biomaterials 2006, 27, 3817–3824. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Q.; Sayer, M.; Kawaja, M.; Shen, X.; Davies, J.E. Attachment, morphology, and protein expression of rat marrow stromal cells cultured on charged substrate surfaces. J. Biomed. Mater. Res. 1998, 42, 117–127. [Google Scholar] [CrossRef]
- Zhao, G.; Schwartz, Z.; Wieland, M.; Rupp, F.; Geis-Gerstorfer, J.; Cochran, D.L.; Boyan, B.D. High surface energy enhances cell response to titanium substrate microstructure. J. Biomed. Mater. Res. 2005, 74, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Dworetzky, S.I.; Fey, E.G.; Penman, S.; Lian, J.B.; Stein, J.L.; Stein, G.S. Progressive changes in the protein composition of the nuclear matrix during rat osteoblast differentiation. Proc. Natl. Acad. Sci. USA 1990, 87, 4605–4609. [Google Scholar] [CrossRef]
- Bellows, C.G.; Heersche, J.N.; Aubin, J.E. Inorganic phosphate added exogenously or released from beta-glycerophosphate initiates mineralization of osteoid nodules in vitro. Bone. Miner. 1992, 17, 15–29. [Google Scholar] [CrossRef]
- Lian, J.B.; Stein, G.S. Concepts of osteoblast growth and differentiation: Basis for modulation of bone cell development and tissue formation. Crit. Rev. Oral. Biol. Med. 1992, 3, 269–305. [Google Scholar] [CrossRef]
- Brown, W.E.; Smith, J.P.; Lehr, J.R.; Frazier, A.W. Crystallographic and chemical relations between octacalcium phosphate and hydroxyapatite. Nature 1962, 196, 1050–1055. [Google Scholar] [CrossRef]
- Suzuki, O.; Nakamura, M.; Miyasaka, Y.; Kagayama, M.; Sakurai, M. Bone formation on synthetic precursors of hydroxyapatite. Tohoku J. Exp. Med. 1991, 164, 37–50. [Google Scholar] [CrossRef] [PubMed]
- Sai, Y.; Shiwaku, Y.; Anada, T.; Tsuchiya, K.; Takahashi, T.; Suzuki, O. Capacity of octacalcium phosphate to promote osteoblastic differentiation toward osteocytes in vitro. Acta Biomater. 2018, 69, 362–371. [Google Scholar] [CrossRef] [PubMed]

| (μm) | ||||
|---|---|---|---|---|
| Substrates | Sa | Sp | Sv | Sz |
| Ti | 0.49 ± 0.01 | 3.55 ± 0.71 | 3.73 ± 1.21 | 7.19 ± 1.24 |
| CTO 700 | 0.62 ± 0.08 | 4.14 ± 0.40 | 5.58 ± 1.13 | 9.71 ± 0.98 |
| CTO 800 | 0.87 ± 0.15 *,† | 5.39 ± 1.35 * | 6.25 ± 1.23 | 11.64 ± 0.98 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shiraishi, N.; Ishiko-Uzuka, R.; Takahashi, K.; Ogawa, T.; Anada, T.; Suzuki, O.; Goto, T.; Sasaki, K. Effect of Functionally-Graded Calcium Titanate Film, Prepared by Metal-Organic Chemical Vapor Deposition, on Titanium Implant. Appl. Sci. 2019, 9, 172. https://doi.org/10.3390/app9010172
Shiraishi N, Ishiko-Uzuka R, Takahashi K, Ogawa T, Anada T, Suzuki O, Goto T, Sasaki K. Effect of Functionally-Graded Calcium Titanate Film, Prepared by Metal-Organic Chemical Vapor Deposition, on Titanium Implant. Applied Sciences. 2019; 9(1):172. https://doi.org/10.3390/app9010172
Chicago/Turabian StyleShiraishi, Naru, Risa Ishiko-Uzuka, Kenta Takahashi, Toru Ogawa, Takahisa Anada, Osamu Suzuki, Takashi Goto, and Keiichi Sasaki. 2019. "Effect of Functionally-Graded Calcium Titanate Film, Prepared by Metal-Organic Chemical Vapor Deposition, on Titanium Implant" Applied Sciences 9, no. 1: 172. https://doi.org/10.3390/app9010172
APA StyleShiraishi, N., Ishiko-Uzuka, R., Takahashi, K., Ogawa, T., Anada, T., Suzuki, O., Goto, T., & Sasaki, K. (2019). Effect of Functionally-Graded Calcium Titanate Film, Prepared by Metal-Organic Chemical Vapor Deposition, on Titanium Implant. Applied Sciences, 9(1), 172. https://doi.org/10.3390/app9010172




