Influence of Preheating Temperature on Cold Metal Transfer (CMT) Welding–Brazing of Aluminium Alloy/Galvanized Steel
Abstract
1. Introduction
2. Experimental Methods
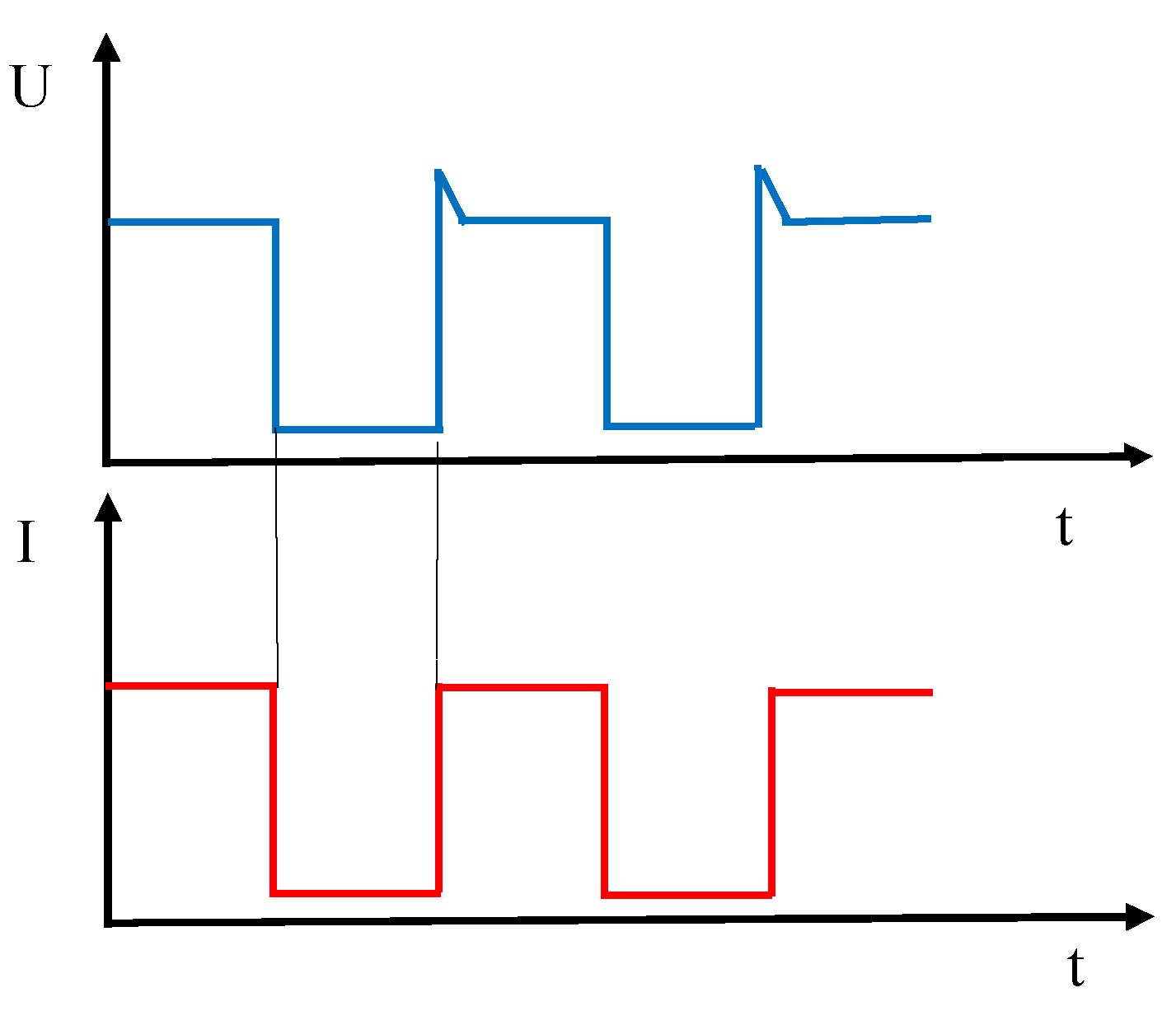
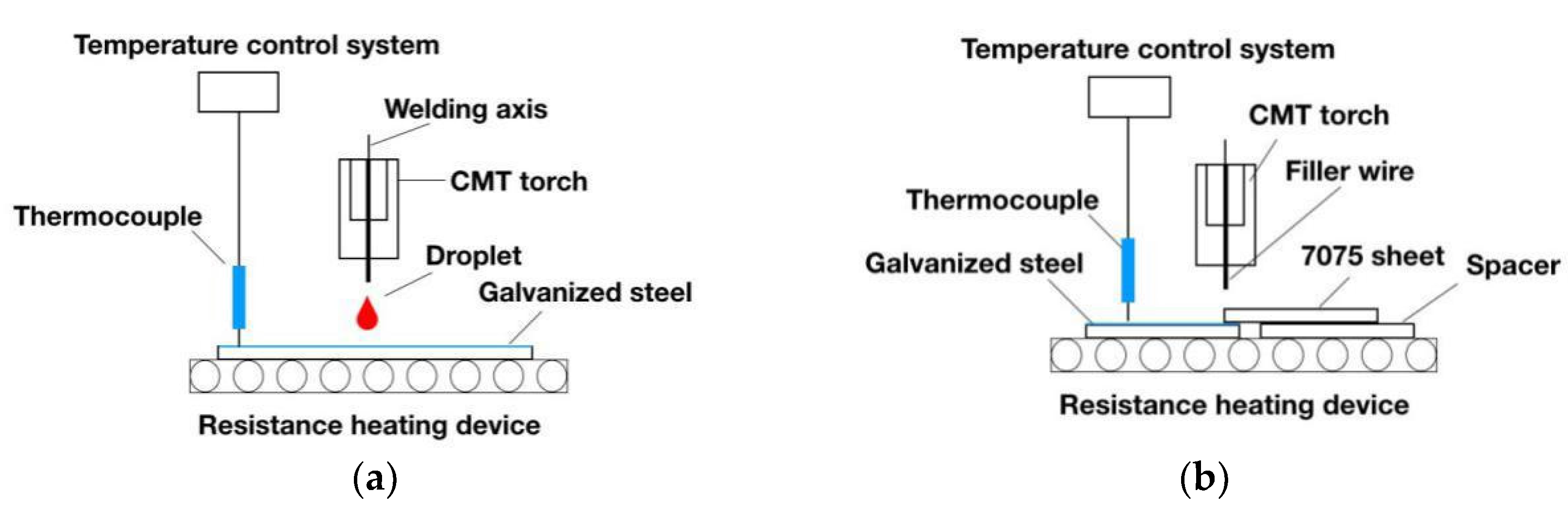
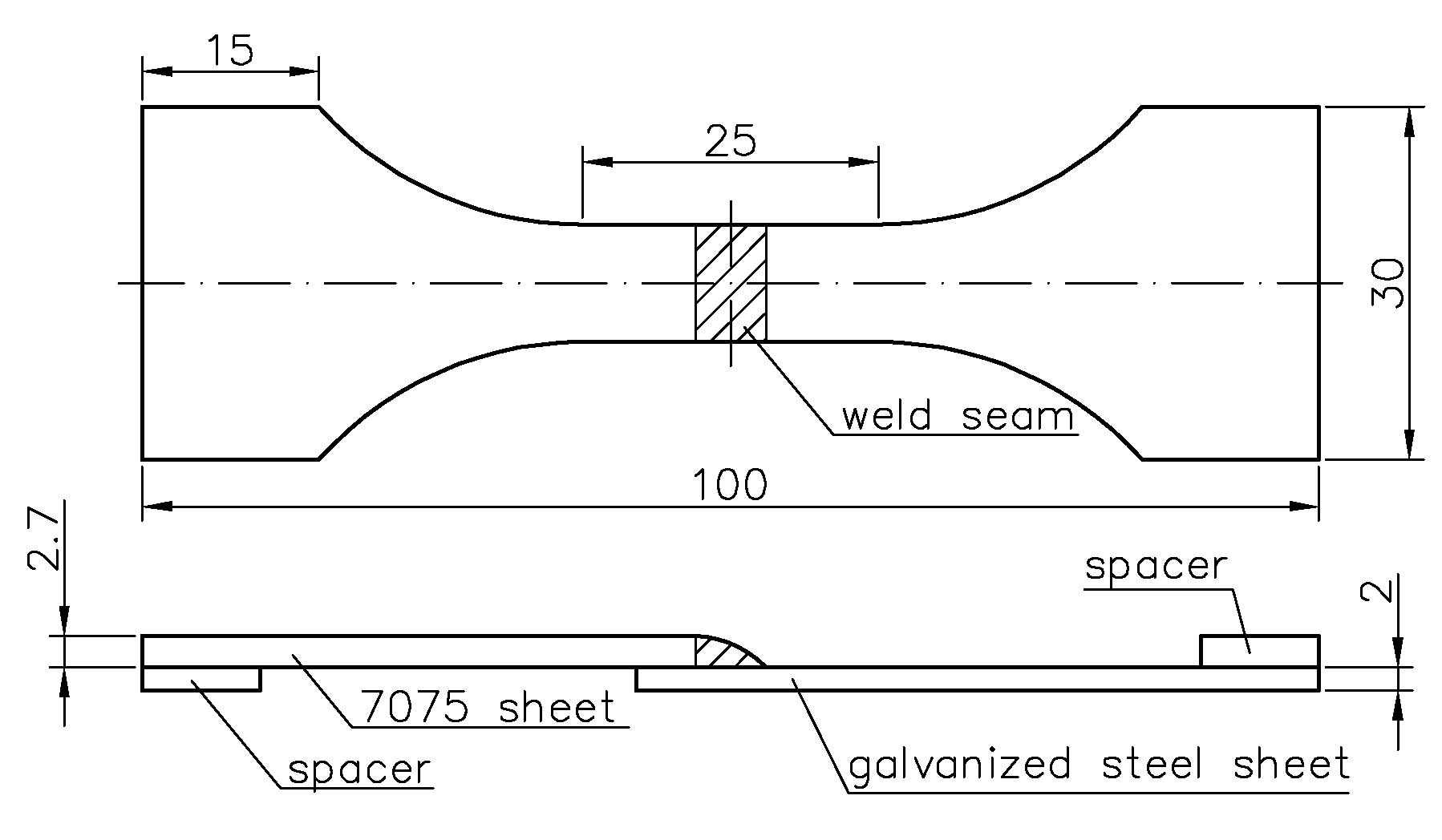
2.1. Materials and Procedure
2.2. Analysis Methods
3. Results and Discussion
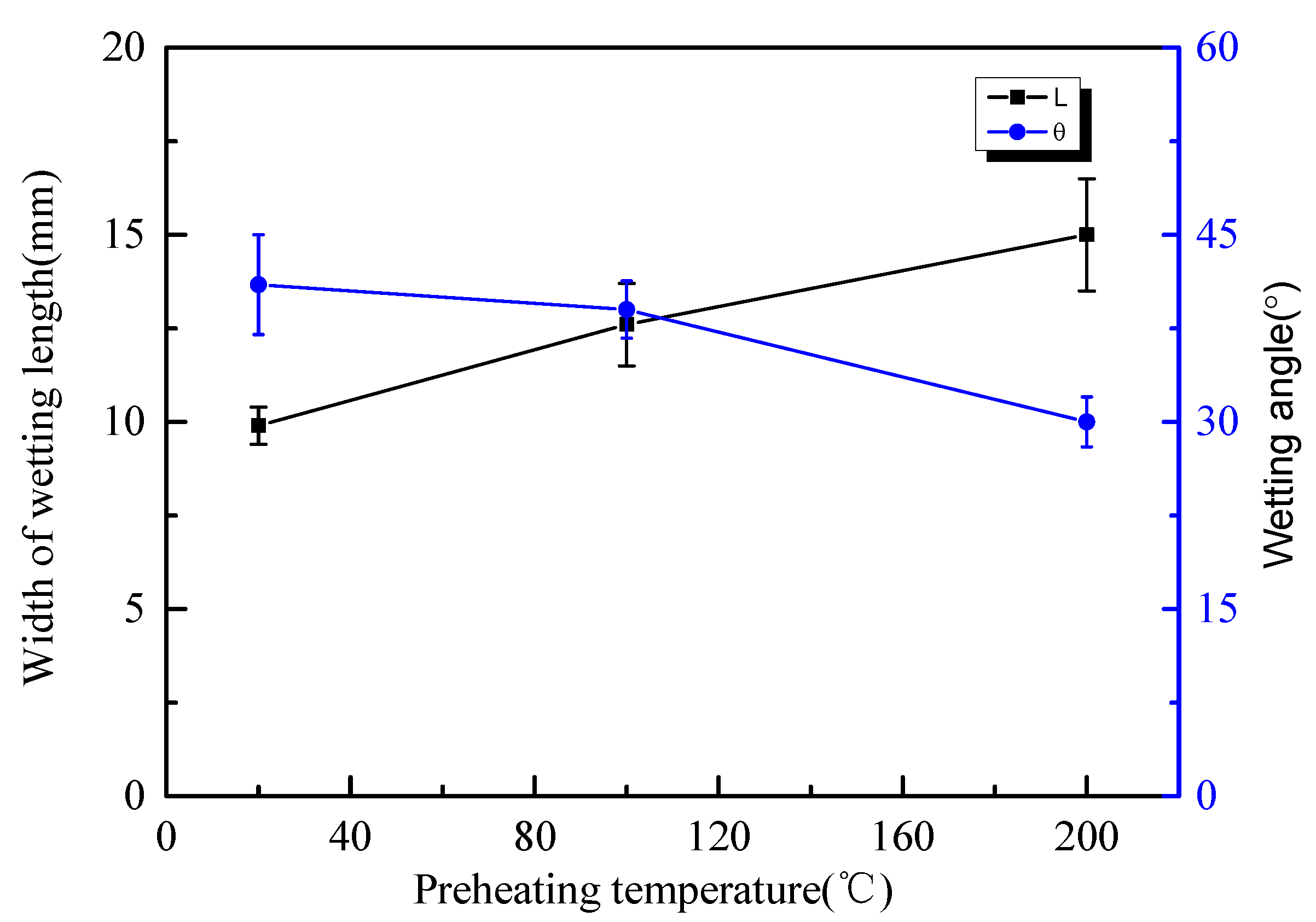
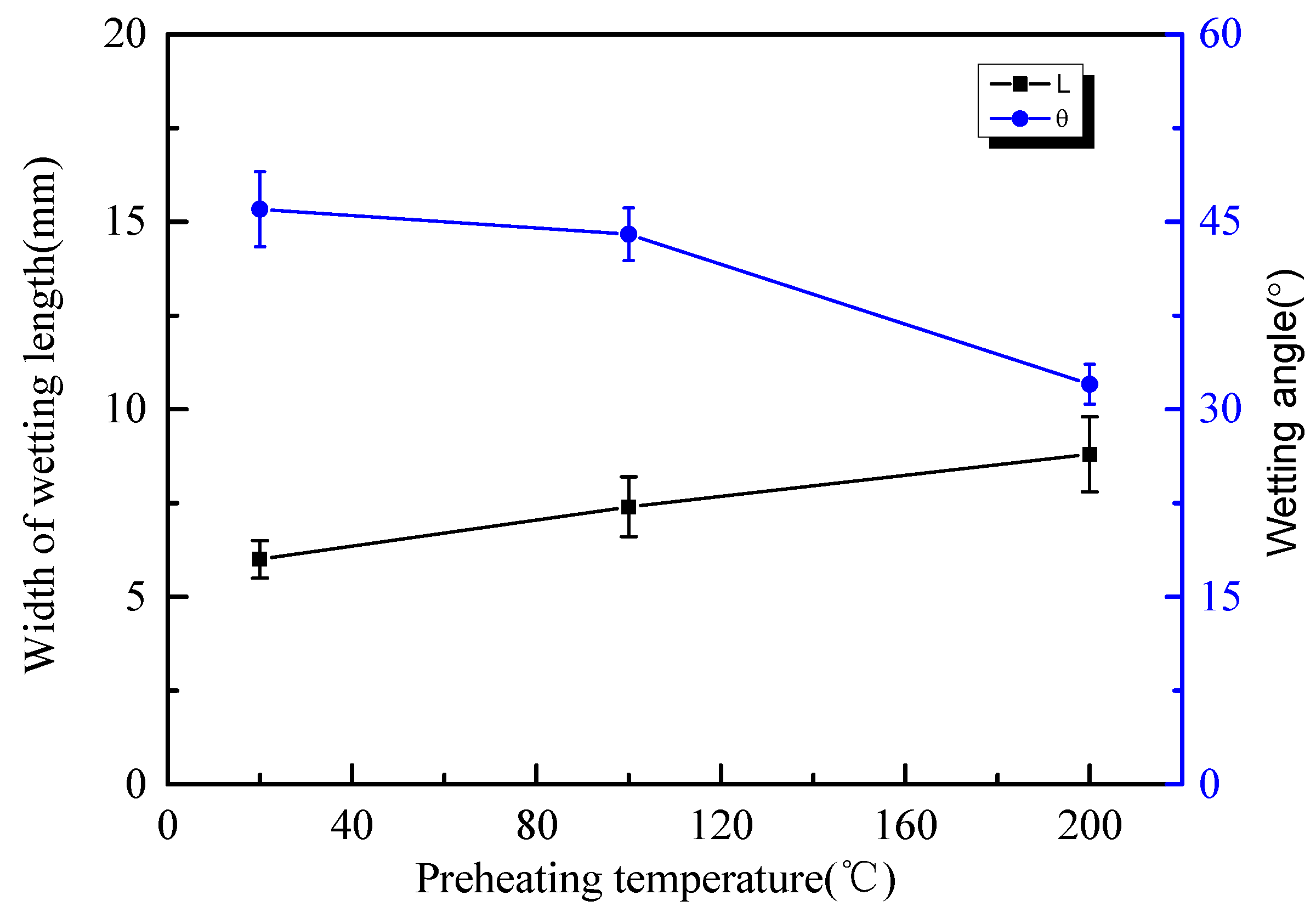
3.1. Wettability Characteristics of Bead-on-Plate Brazing
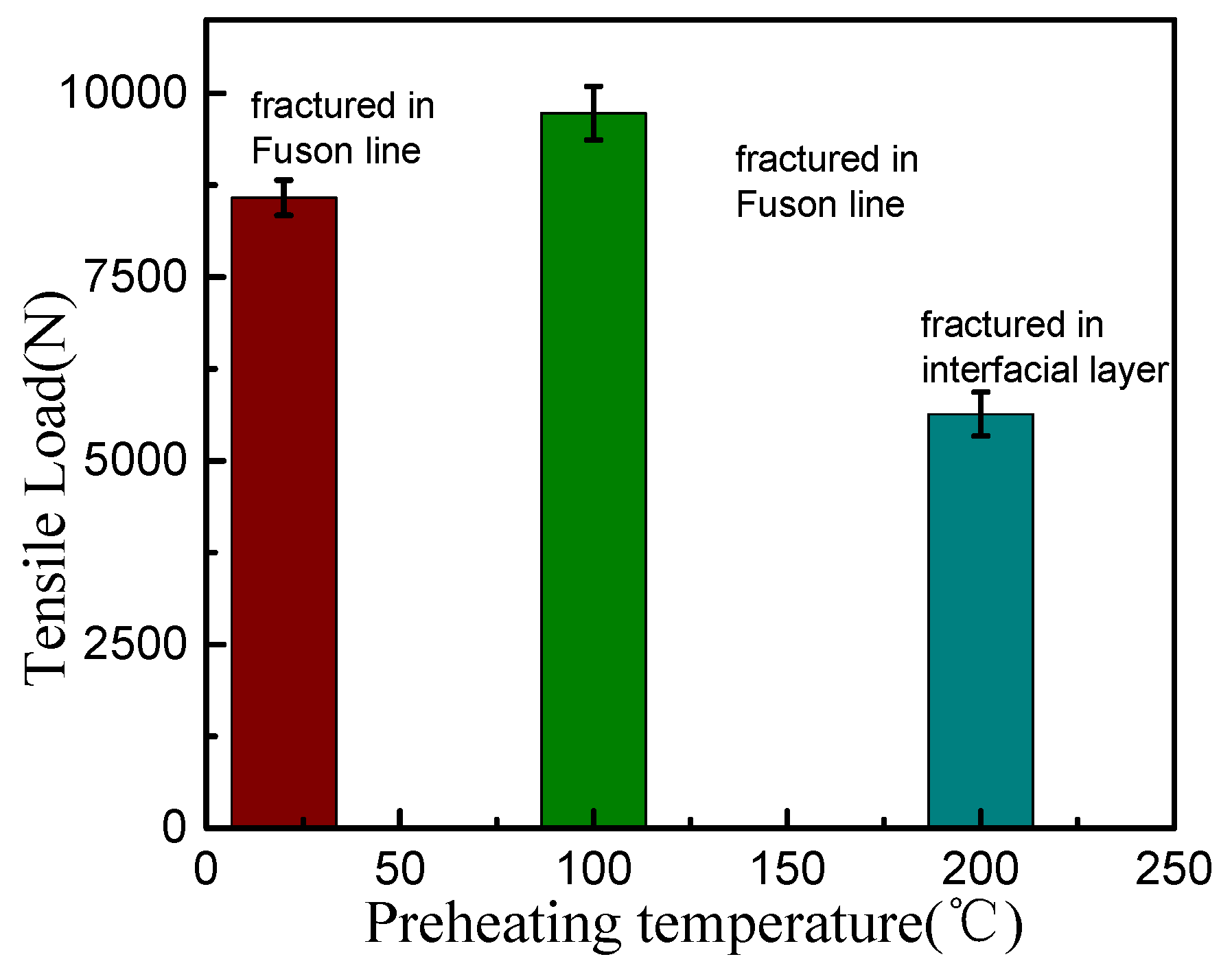
3.2. Influence of Preheating Temperature on the Overlap Welding–Brazing of Aluminium Alloy and Galvanized Steel
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, X.; Scherf, A.; Heilmaier, M.; Stein, F. The Al-Rich Part of the Fe-Al Phase Diagram. J. Phase Equilib. Diffus. 2016, 37, 162–173. [Google Scholar] [CrossRef]
- Pouranvari, M.; Abbasi, M. Dissimilar gas tungsten arc weld-brazing of Al/steel using Al-Si filler metal: Microstructure and strengthening mechanisms. J. Alloys Compd. 2018, 749, 121–127. [Google Scholar] [CrossRef]
- Pouranvari, M. Critical assessment: Dissimilar resistance spot welding of aluminium/steel: Challenges and opportunities. Mater. Sci. Technol. 2017, 33, 1705–1712. [Google Scholar] [CrossRef]
- Agudo, L.; Weber, S.; Wagner, J.; Eyidi, D.; Schmaranzer, C.H.; Arenholz, E.; Jank, N.; Bruckner, J.; Pyzalla, A.R. Intermetallic FexAly-phases in a steel/Al-alloy fusion weld. J. Mater. Sci. 2007, 42, 4205–4214. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, J.; Lian, J.; Lei, Y. Welding of aluminum alloy to zinc-coated steel by cold metal transfer. Mater. Des. 2013, 49, 602–612. [Google Scholar] [CrossRef]
- Springer, H.; Kostka, A.; Payton, E.J.; Raabe, D.; Kaysser-Pyzalla, A.; Eggeler, G. On the formation and growth of intermetallic phases during interdiffusion between low-carbon steel and aluminum alloys. Acta Mater. 2011, 59, 1586–1600. [Google Scholar] [CrossRef]
- Gatzen, M.; Radel, T.; Thomy, C.; Vollertsen, F. Wetting and solidification characteristics of aluminium on zinc coated steel in laser welding and brazing. J. Mater. Process. Technol. 2016, 238, 352–360. [Google Scholar] [CrossRef]
- Gatzen, M.; Radel, T.; Thomy, C.; Vollertsen, F. The role of zinc layer during wetting of aluminium on zinc-coated steel in laser brazing and welding. Phys. Procedia 2014, 56, 730–739. [Google Scholar] [CrossRef]
- Liu, J.; Jiang, S.; Shi, Y.; Cong, N.; Chen, J.; Huang, G. Effects of zinc on the laser welding of an aluminum alloy and galvanized steel. J. Mater. Process. Technol. 2015, 224, 49–59. [Google Scholar]
- Ma, H.; Qin, G.; Wang, L.; Meng, X.; Chen, L. Effects of preheat treatment on microstructure evolution and properties of brazed-fusion welded joint of aluminum alloy to steel. Mater. Des. 2016, 90, 330–339. [Google Scholar] [CrossRef]
- Qin, G.; Lei, Z.; Su, Y.; Fu, B.; Meng, X.; Lin, S. Large spot laser assisted GMA brazing-fusion welding of aluminum alloy to galvanized steel. J. Mater. Process. Technol. 2014, 214, 2684–2692. [Google Scholar] [CrossRef]
- Cao, R.; Yu, G.; Chen, J.H.; Wang, P.C. Cold metal transfer joining aluminum alloys-to-galvanized mild steel. J. Mater. Process. Technol. 2013, 213, 1753–1763. [Google Scholar] [CrossRef]
- Zhou, Y.L.; Lin, Q.L. Wetting of galvanized steel by Al 4043 alloys in the first cycle of CMT process. J. Alloys Compd. 2014, 589, 307–313. [Google Scholar] [CrossRef]
- Baker, H.; Okamoto, H. ASM Handbook Volume 3: Alloy Phase Diagrams; ASM International: Materials Park, OH, USA, 1992. [Google Scholar]





| Materials | C | Si | Fe | Cu | Mn | Mg | Zn | Al |
|---|---|---|---|---|---|---|---|---|
| galvanized steel | 0.041 | 0.018 | Bal. | - | 0.187 | - | - | - |
| 7075 | - | 0.40 | 0.50 | 1.20–2.0 | 0.30 | 2.10–2.90 | 5.10–6.10 | Bal. |
| ER4043 | - | 4.5–6.0 | 0.80 | 0.30 | 0.05 | 0.05 | 0.10 | Bal. |
| Wire Feeding Speed (m/min) | Welding Speed (m/min) | Preheating Temperature (°C) | |
|---|---|---|---|
| Bead-on-plate brazing | 5 | 0.5 | Room temperature, 100, and 200 |
| Overlap welding–brazing | 5 | 0.4 | Room temperature, 100, and 200 |
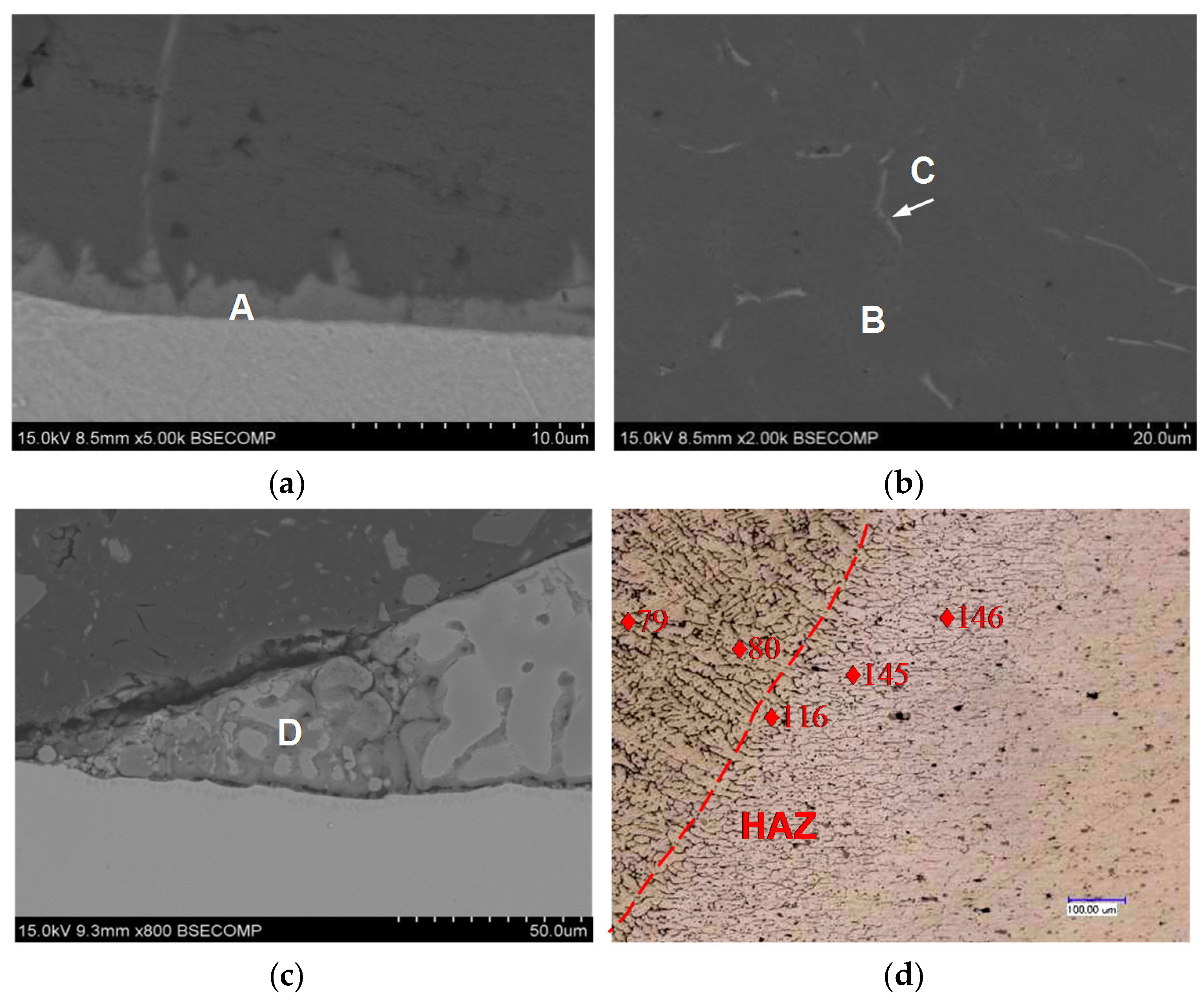
| Location | Fe | Al | Zn | Si | Possible Phase |
|---|---|---|---|---|---|
| A | 18.56 | 71.34 | 0.5 | 9.60 | Fe–Al intermetallic |
| B | 0.78 | 96.68 | 0.64 | 1.9 | α-Al |
| C | 0.88 | 74.66 | 0.76 | 23.7 | Al–Si eutectic phase |
| D | 0.93 | 71.39 | 19.39 | 09.29 | Al–Zn solid solution |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qin, Y.; He, X.; Jiang, W. Influence of Preheating Temperature on Cold Metal Transfer (CMT) Welding–Brazing of Aluminium Alloy/Galvanized Steel. Appl. Sci. 2018, 8, 1659. https://doi.org/10.3390/app8091659
Qin Y, He X, Jiang W. Influence of Preheating Temperature on Cold Metal Transfer (CMT) Welding–Brazing of Aluminium Alloy/Galvanized Steel. Applied Sciences. 2018; 8(9):1659. https://doi.org/10.3390/app8091659
Chicago/Turabian StyleQin, Youqiong, Xi He, and Wenxiang Jiang. 2018. "Influence of Preheating Temperature on Cold Metal Transfer (CMT) Welding–Brazing of Aluminium Alloy/Galvanized Steel" Applied Sciences 8, no. 9: 1659. https://doi.org/10.3390/app8091659
APA StyleQin, Y., He, X., & Jiang, W. (2018). Influence of Preheating Temperature on Cold Metal Transfer (CMT) Welding–Brazing of Aluminium Alloy/Galvanized Steel. Applied Sciences, 8(9), 1659. https://doi.org/10.3390/app8091659




