Quantitative Analysis of Patellar Tendon Abnormality in Asymptomatic Professional “Pallapugno” Players: A Texture-Based Ultrasound Approach
Abstract
:Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Subject Database
2.2. Ultrasound Image Acquisition and Protocol
2.3. Texture Feature Extraction
2.3.1. First-Order Statistical Descriptors
2.3.2. Haralick Features
2.3.3. Higher-Order Spectra, Entropy Features, and Hu’s Moments
2.4. Statistical Analysis
3. Results
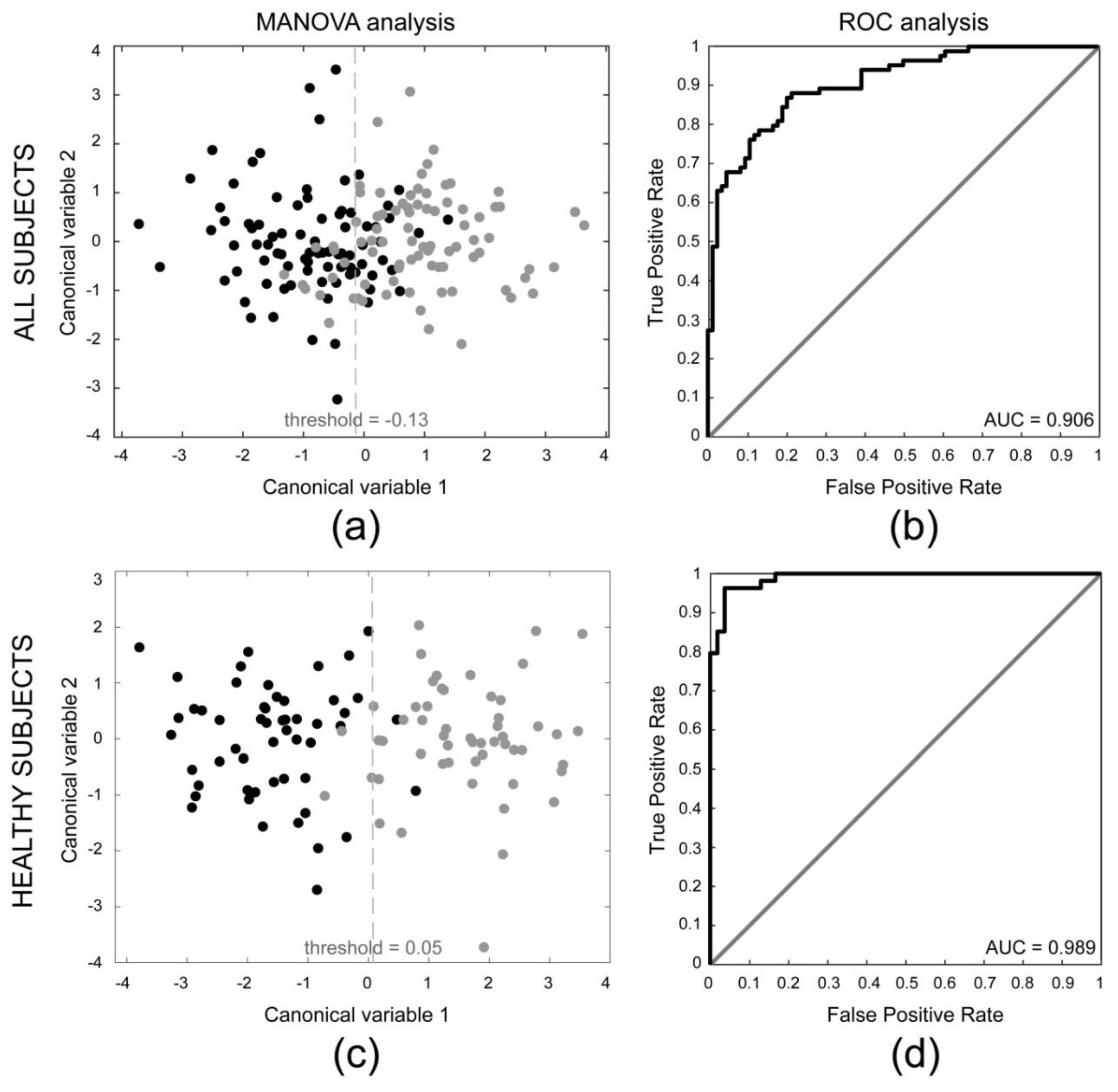
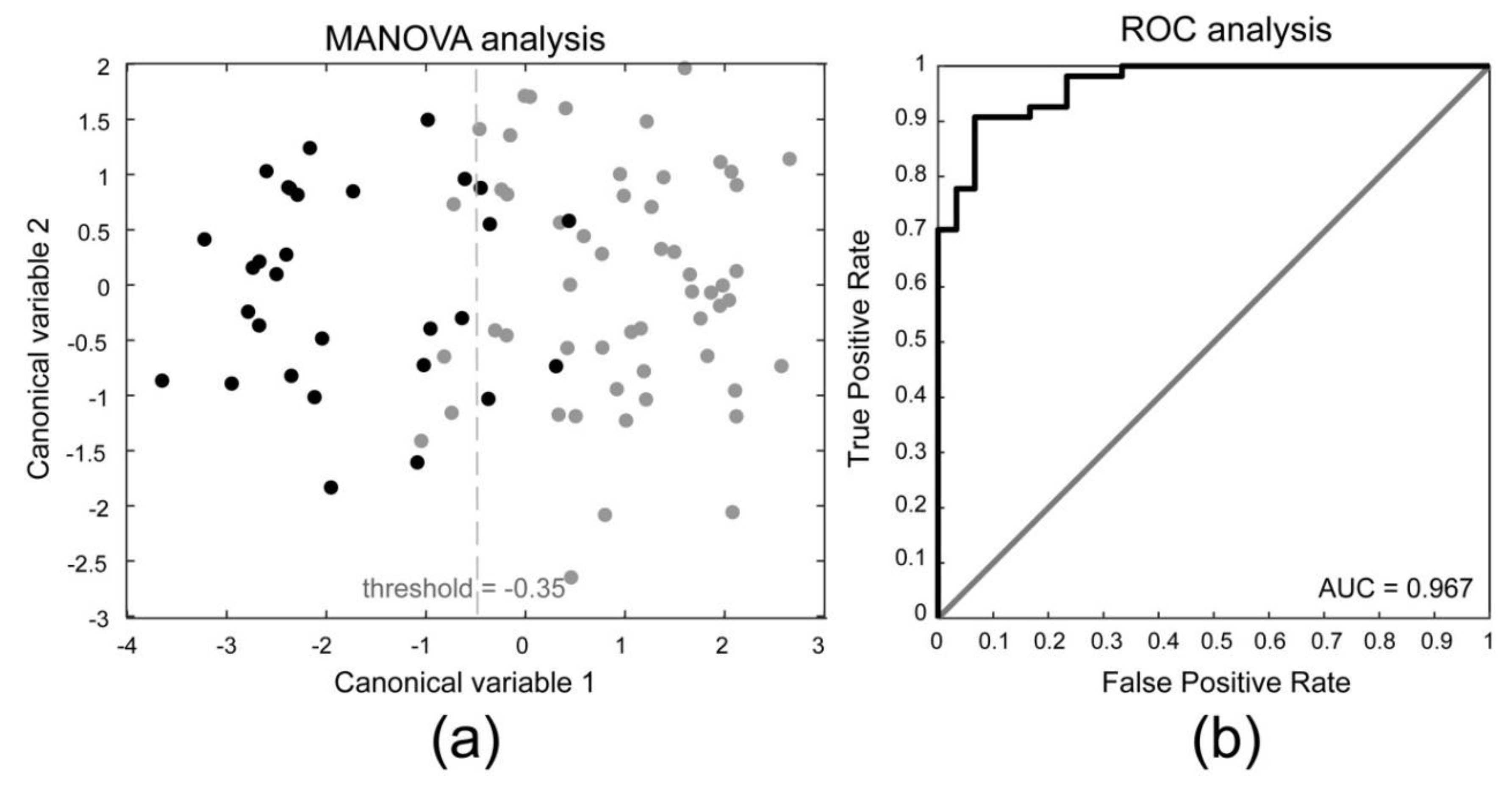
3.1. Comparison between Dominant and Non-Dominant Side
3.2. Comparison between Subclinical Tendinopathy and Non-Subclinical Tendinopathy
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Pillen, S.; Arts, I.M.P.; Zwarts, M.J. Muscle ultrasound in neuromuscular disorders. Muscle Nerve 2008, 37, 679–693. [Google Scholar] [CrossRef] [PubMed]
- Reeves, N.D.; Maganaris, C.N.; Narici, M.V. Ultrasonographic assessment of human skeletal muscle size. Eur. J. Appl. Physiol. 2004, 91, 116–118. [Google Scholar] [CrossRef] [PubMed]
- Meiburger, K.M.; Acharya, U.R.; Molinari, F. Automated localization and segmentation techniques for B-mode ultrasound images: A review. Comput. Biol. Med. 2018, 92, 210–235. [Google Scholar] [CrossRef] [PubMed]
- Caresio, C.; Salvi, M.; Molinari, F.; Meiburger, K.M.; Minetto, M.A. Fully Automated Muscle Ultrasound Analysis (MUSA): Robust and Accurate Muscle Thickness Measurement. Ultrasound Med. Biol. 2017, 43, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Martinoli, C.; Derchi, L.E.; Pastorino, C.; Bertolotto, M.; Silvestri, E. Analysis of echotexture of tendons with US. Radiology 1993, 186, 839–843. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, O.S. Sonography of tendons. Scand. J. Med. Sci. Sports 2000, 10, 360–364. [Google Scholar] [CrossRef] [PubMed]
- Fredberg, U.; Bolvig, L.; Andersen, N.; Stengaard-Pedersen, K. Ultrasonography in Evaluation of Achilles and Patella Tendon Thickness. Eur. J. Ultrasound 2007, 29, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Gellhorn, A.C.; Morgenroth, D.C.; Goldstein, B. A Novel Sonographic Method of Measuring Patellar Tendon Length. Ultrasound Med. Biol. 2012, 38, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Sunding, K.; Fahlström, M.; Werner, S.; Forssblad, M.; Willberg, L. Evaluation of Achilles and patellar tendinopathy with greyscale ultrasound and colour Doppler: Using a four-grade scale. Knee Surg. Sport Traumatol. Arthrosc. 2016, 24, 1988–1996. [Google Scholar] [CrossRef] [PubMed]
- Kulig, K.; Landel, R.; Chang, Y.-J.; Hannanvash, N.; Reischl, S.F.; Song, P.; Bashford, G.R. Patellar tendon morphology in volleyball athletes with and without patellar tendinopathy. Scand. J. Med. Sci. Sports 2013, 23, e81–e88. [Google Scholar] [CrossRef] [PubMed]
- Giombini, A.; Dragoni, S.; Di Cesare, A.; Di Cesare, M.; Del Buono, A.; Maffulli, N. Asymptomatic Achilles, patellar, and quadriceps tendinopathy: A longitudinal clinical and ultrasonographic study in elite fencers. Scand. J. Med. Sci. Sports 2013, 23, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Comin, J.; Cook, J.L.; Malliaras, P.; McCormack, M.; Calleja, M.; Clarke, A.; Connell, D. The prevalence and clinical significance of sonographic tendon abnormalities in asymptomatic ballet dancers: A 24-month longitudinal study. Br. J. Sports Med. 2013, 47, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Visnes, H.; Tegnander, A.; Bahr, R. Ultrasound characteristics of the patellar and quadriceps tendons among young elite athletes. Scand. J. Med. Sci. Sports 2015, 25, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Van der Worp, H.; de Poel, H.; Diercks, R.; van den Akker-Scheek, I.; Zwerver, J. Jumper’s Knee or Lander’s Knee? A Systematic Review of the Relation between Jump Biomechanics and Patellar Tendinopathy. Int. J. Sports Med. 2014, 35, 714–722. [Google Scholar] [CrossRef] [PubMed]
- Reinking, M.F. Current concepts in the treatment of patellar tendinopathy. Int. J. Sports Phys. Ther. 2016, 11, 854–866. [Google Scholar] [PubMed]
- Drakonaki, E.E.; Allen, G.M.; Wilson, D.J. Ultrasound elastography for musculoskeletal applications. Br. J. Radiol. 2012, 85, 1435–1445. [Google Scholar] [CrossRef] [PubMed]
- Arda, K.; Ciledag, N.; Aktas, E.; Arıbas, B.K.; Köse, K. Quantitative Assessment of Normal Soft-Tissue Elasticity Using Shear-Wave Ultrasound Elastography. Am. J. Roentgenol. 2011, 197, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.J.; Ng, G.Y.; Lee, W.C.; Fu, S.N. Changes in Morphological and Elastic Properties of Patellar Tendon in Athletes with Unilateral Patellar Tendinopathy and Their Relationships with Pain and Functional Disability. PLoS ONE 2014, 9, e108337. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.J.; Fu, S.N. Shear Elastic Modulus on Patellar Tendon Captured from Supersonic Shear Imaging: Correlation with Tangent Traction Modulus Computed from Material Testing System and Test-Retest Reliability. PLoS ONE 2013, 8, e68216. [Google Scholar] [CrossRef] [PubMed]
- Porta, F.; Damjanov, N.; Galluccio, F.; Iagnocco, A.; Matucci-Cerinic, M. Ultrasound elastography is a reproducible and feasible tool for the evaluation of the patellar tendon in healthy subjects. Int. J. Rheum. Dis. 2014, 17, 762–766. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, M.-Y.; Chen, Y.-C.; Lin, C.-Y.; Chen, W.-S.; Wang, T.-G. Reduced Patellar Tendon Elasticity with Aging: In Vivo Assessment by Shear Wave Elastography. Ultrasound Med. Biol. 2015, 41, 2899–2905. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.L.; Khan, K.M.; Kiss, Z.S.; Coleman, B.D.; Griffiths, L. Asymptomatic hypoechoic regions on patellar tendon ultrasound: A 4-year clinical and ultrasound followup of 46 tendons. Scand. J. Med. Sci. Sports 2001, 11, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Giacchino, M.; Caresio, C.; Gorji, N.E.; Molinari, F.; Massazza, G.; Minetto, M.A. Quantitative analysis of patellar tendon size and structure in asymptomatic professional players: Sonographic study. MLTJ 2017, 7, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Pillen, S.; Tak, R.O.; Zwarts, M.J.; Lammens, M.M.Y.; Verrijp, K.N.; Arts, I.M.P.; van der Laak, J.A.; Hoogerbrugge, P.M.; van Engelen, B.G.M.; Verrips, A. Skeletal muscle ultrasound: Correlation between fibrous tissue and echo intensity. Ultrasound Med. Biol. 2009, 35, 443–446. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.P.; Singh, B. Texture Features Extraction in Mammograms Using Non-Shannon Entropies; Springer: Dordrecht, The Netherlands, 2010; pp. 341–351. [Google Scholar]
- Acharya, U.R.; Krishnan, M.M.R.; Saba, L.; Molinari, F.; Guerriero, S.; Suri, J.S. Ovarian Tumor Characterization Using 3D Ultrasound. In Ovarian Neoplasm Imaging; Springer: Boston, MA, USA, 2013; pp. 399–412. [Google Scholar]
- Acharya, U.R.; Faust, O.; Sree, S.V.; Molinari, F.; Suri, J.S. ThyroScreen system: High resolution ultrasound thyroid image characterization into benign and malignant classes using novel combination of texture and discrete wavelet transform. Comput. Methods Programs Biomed. 2012, 107, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Peng, Y.; Guo, H.; Liu, W.; Gao, T.; Xu, Y.; Tang, X. Texture analysis and classification of ultrasound liver images. Biomed. Mater. Eng. 2014, 24, 1209–1216. [Google Scholar] [CrossRef] [PubMed]
- Molinari, F.; Caresio, C.; Acharya, U.R.; Mookiah, M.R.K.; Minetto, M.A. Advances in Quantitative Muscle Ultrasonography Using Texture Analysis of Ultrasound Images. Ultrasound Med. Biol. 2015, 41, 2520–2532. [Google Scholar] [CrossRef] [PubMed]
- Elias, L.J.; Bryden, M.P.; Bulman-Fleming, M.B. Footedness is a better predictor than is handedness of emotional lateralization. Neuropsychologia 1998, 36, 37–43. [Google Scholar] [CrossRef]
- Haralick, R.M.; Shanmugam, K.; Dinstein, I. Textural Features for Image Classification. IEEE Trans. Syst. Man Cybern. 1973, 6, 610–621. [Google Scholar] [CrossRef]
- Shao, Y.; Celenk, M. Higher-order spectra (HOS) invariants for shape recognition. Pattern Recognit. 2001, 34, 2097–2113. [Google Scholar] [CrossRef]
- Shannon, C.E. A Mathematical Theory of Communication. Bell Syst. Tech. J. 1948, 27, 379–423. [Google Scholar] [CrossRef]
- Rényi, A. On Measures of Entropy and Information. In Proceedings of the Fourth Berkeley Symposium on Mathematical Statistics and Probability, Berkeley, CA, USA, 14 April 1961. [Google Scholar]
- Chen, W.; Wang, Z.; Xie, H.; Yu, W. Characterization of Surface EMG Signal Based on Fuzzy Entropy. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Kapur, J.N. Information of orderα and typeβ. Proc. Indian Acad. Sci. 1968, 68, 65–75. [Google Scholar] [CrossRef]
- Ghosh, M.; Chakraborty, C.; Ray, A.K. Yager’s measure based fuzzy divergence for microscopic color image segmentation. In Proceedings of the 2013 Indian Conference on Medical Informatics and Telemedicine (ICMIT), Kharagpur, India, 28–30 March 2013; pp. 13–16. [Google Scholar]
- Vajda, I. Theory of Statistical Inference and Information; Kluwer Academic: Dordrecht, The Netherlands, 1989; ISBN 9027727813. [Google Scholar]
- Yin, P.-Y. Maximum entropy-based optimal threshold selection using deterministic reinforcement learning with controlled randomization. Signal Process. 2002, 82, 993–1006. [Google Scholar] [CrossRef]
- Hu, M.-K. Visual pattern recognition by moment invariants. IEEE Trans. Inf. Theory 1962, 8, 179–187. [Google Scholar] [CrossRef]
- Belsley, D.A.; Kuh, E.; Welsch, R.E. Regression Diagnostics: Identifying Influential Data and Sources of Collinearity; Wiley: Hoboken, NJ, USA, 2004; ISBN 9780471725145. [Google Scholar]
- Hanley, J.A.; McNeil, B.J. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 1982, 143, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Maffulli, N. Biology of tendon injury: Healing, modeling and remodeling. J. Musculoskelet. Neuronal Interact. 2006, 6, 181–190. [Google Scholar] [PubMed]
- Khan, K.M.; Cook, J.L.; Bonar, F.; Harcourt, P.; Astrom, M. Histopathology of common tendinopathies. Update and implications for clinical management. Sports Med. 1999, 27, 393–408. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.M.; Maffulli, N.; Coleman, B.D.; Cook, J.L.; Taunton, J.E. Patellar tendinopathy: Some aspects of basic science and clinical management. Br. J. Sports Med. 1998, 32, 346–355. [Google Scholar] [CrossRef] [PubMed]
- Scott, A.; Backman, L.J.; Speed, C. Tendinopathy: Update on Pathophysiology. J. Orthop. Sports Phys. Ther. 2015, 45, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Peltz, C.D.; Haladik, J.A.; Divine, G.; Siegal, D.; Van Holsbeeck, M.; Bey, M.J. ShearWave elastography: Repeatability for measurement of tendon stiffness. Skelet. Radiol. 2013, 42, 1151–1156. [Google Scholar] [CrossRef] [PubMed]
- Aubry, S.; Nueffer, J.-P.; Tanter, M.; Becce, F.; Vidal, C.; Michel, F. Viscoelasticity in Achilles Tendonopathy: Quantitative Assessment by Using Real-time Shear-Wave Elastography. Radiology 2015, 274, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Domenichini, R.; Pialat, J.-B.; Podda, A.; Aubry, S. Ultrasound elastography in tendon pathology: State of the art. Skelet. Radiol. 2017, 46, 1643–1655. [Google Scholar] [CrossRef] [PubMed]
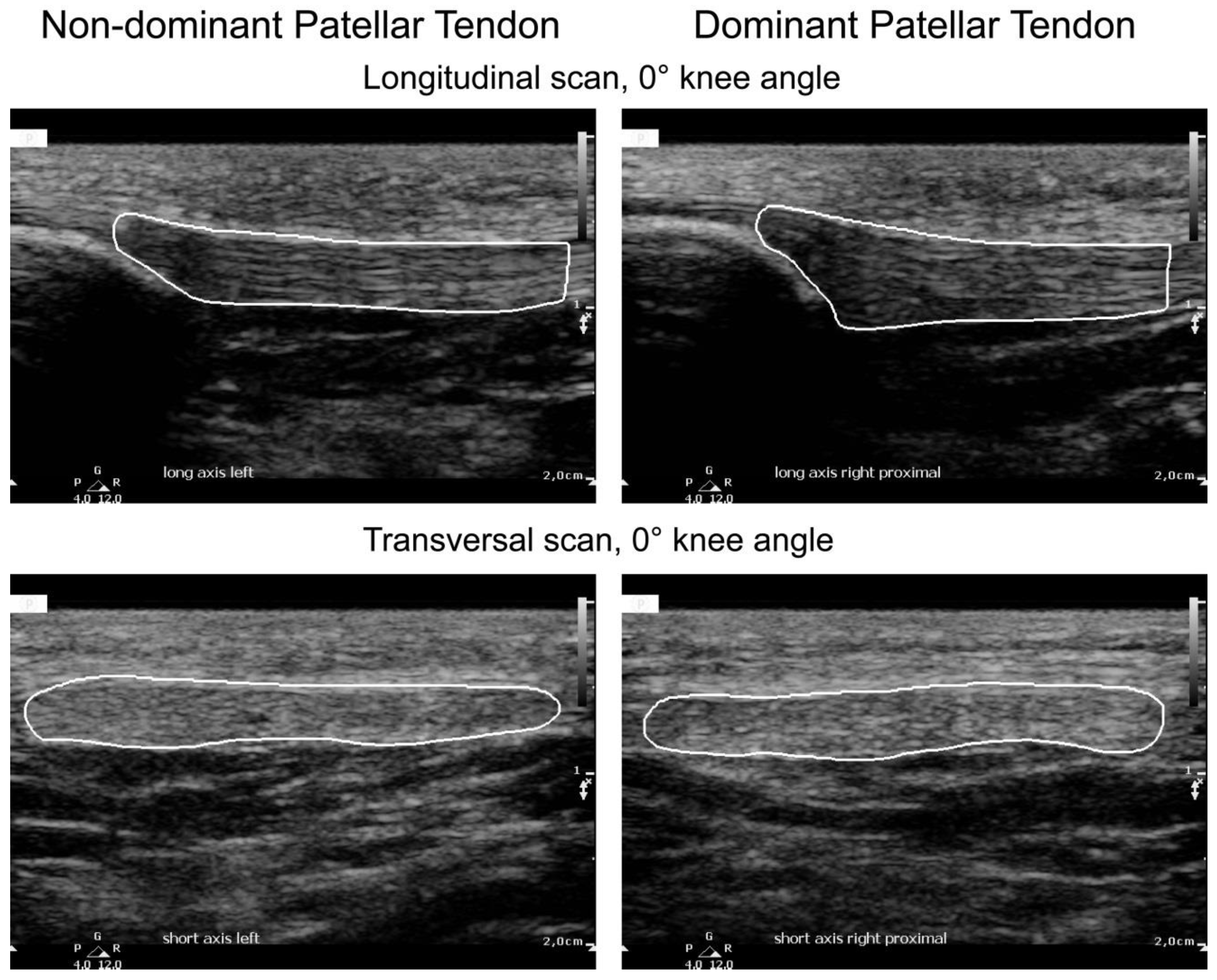
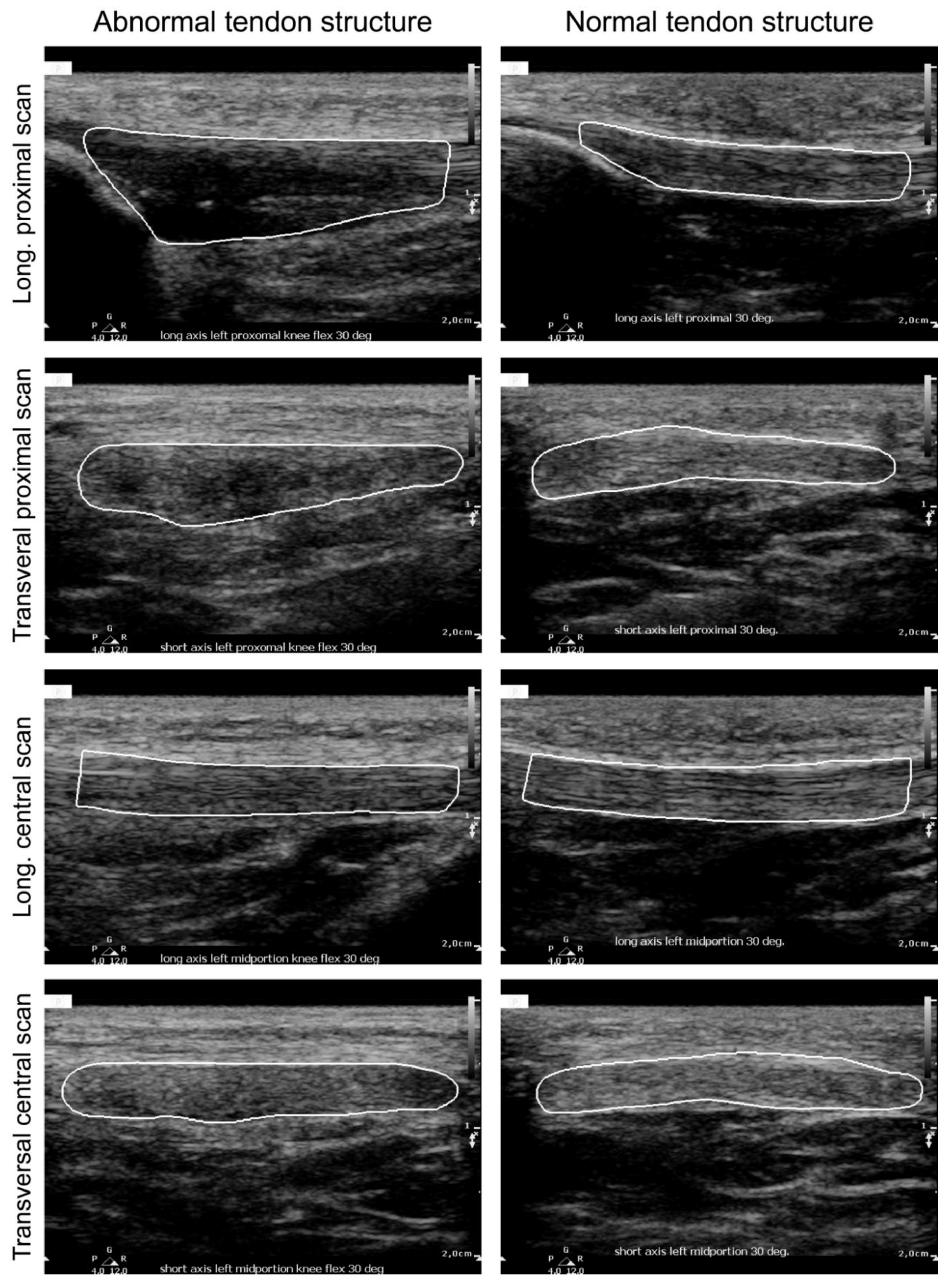
| Plane | Probe Position | Knee Angle |
|---|---|---|
| Transversal | Proximal | 0° |
| Longitudinal | Proximal | 0° |
| Transversal | Proximal | 30° |
| Longitudinal | Proximal | 30° |
| Transversal | Central | 30° |
| Longitudinal | Central | 30° |
| Feature Name | Mathematical Description |
|---|---|
| Mean (m) | |
| Standard deviation () | |
| Variance () | |
| Skewness (Sk) | |
| Kurtosis (Kt) | |
| Energy1 (E1) |
| Feature Name | Mathematical Description |
|---|---|
| Symmetry (Isym) | |
| Contrast (Icon) | |
| Homogeneity (Ihmg) | |
| Entropy (IEntr) | |
| Energy (IEnrg) | |
| Correlation (Icor) |
| Most Discriminant Features for Side Determination (Weight) | Most Discriminant Features for Side Determination (Only Healthy Subjects) (Weight) | Most Discriminant Features for Subclinical Tendinopathy Determination (Weight) | |||
|---|---|---|---|---|---|
| Kurtosis | (−65.0) | H. Homogeneity (45°) | (172.0) | H. Symmetry (0°) | (−24.5) |
| H. Correlation (0°) | (58.9) | H. Contrast (45°) | (−155.8) | H. Contrast (45°) | (−14.3) |
| H. Contrast (0°) | (12.9) | H. Symmetry (45°) | (−48.7) | H. Entropy (0°) | (14.2) |
| Skewness | (−12.3) | H. Symmetry (0°) | (−29.7) | H. Correlation (45°) | (13.4) |
| H. Entropy (0°) | (9.6) | H. Energy (0°) | (27.2) | H. Homogeneity (0°) | (5.5) |
| H. Correlation (45°) | (7.0) | H. Correlation (0°) | (27.1) | Mean Intensity | (4.8) |
| H. Energy (45°) | (−6.3) | H. Correlation (135°) | (6.1) | First−order Entropy | (4.3) |
| H. Homogeneity (0°) | (3.5) | H. Energy (45°) | (−5.3) | Kurtosis | (−4.2) |
| H. Entropy (45°) | (2.3) | Mean Intensity | (5.1) | Variance | (3.0) |
| H. Symmetry (45°) | (−2.2) | H. Entropy (45°) | (4.8) | H. Correlation (0°) | (2.9) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meiburger, K.M.; Salvi, M.; Giacchino, M.; Acharya, U.R.; Minetto, M.A.; Caresio, C.; Molinari, F. Quantitative Analysis of Patellar Tendon Abnormality in Asymptomatic Professional “Pallapugno” Players: A Texture-Based Ultrasound Approach. Appl. Sci. 2018, 8, 660. https://doi.org/10.3390/app8050660
Meiburger KM, Salvi M, Giacchino M, Acharya UR, Minetto MA, Caresio C, Molinari F. Quantitative Analysis of Patellar Tendon Abnormality in Asymptomatic Professional “Pallapugno” Players: A Texture-Based Ultrasound Approach. Applied Sciences. 2018; 8(5):660. https://doi.org/10.3390/app8050660
Chicago/Turabian StyleMeiburger, Kristen M., Massimo Salvi, Maurizio Giacchino, U. Rajendra Acharya, Marco A. Minetto, Cristina Caresio, and Filippo Molinari. 2018. "Quantitative Analysis of Patellar Tendon Abnormality in Asymptomatic Professional “Pallapugno” Players: A Texture-Based Ultrasound Approach" Applied Sciences 8, no. 5: 660. https://doi.org/10.3390/app8050660
APA StyleMeiburger, K. M., Salvi, M., Giacchino, M., Acharya, U. R., Minetto, M. A., Caresio, C., & Molinari, F. (2018). Quantitative Analysis of Patellar Tendon Abnormality in Asymptomatic Professional “Pallapugno” Players: A Texture-Based Ultrasound Approach. Applied Sciences, 8(5), 660. https://doi.org/10.3390/app8050660









