Plenty of Room at the Bottom: Nanotechnology as Solution to an Old Issue in Enhanced Oil Recovery
Abstract
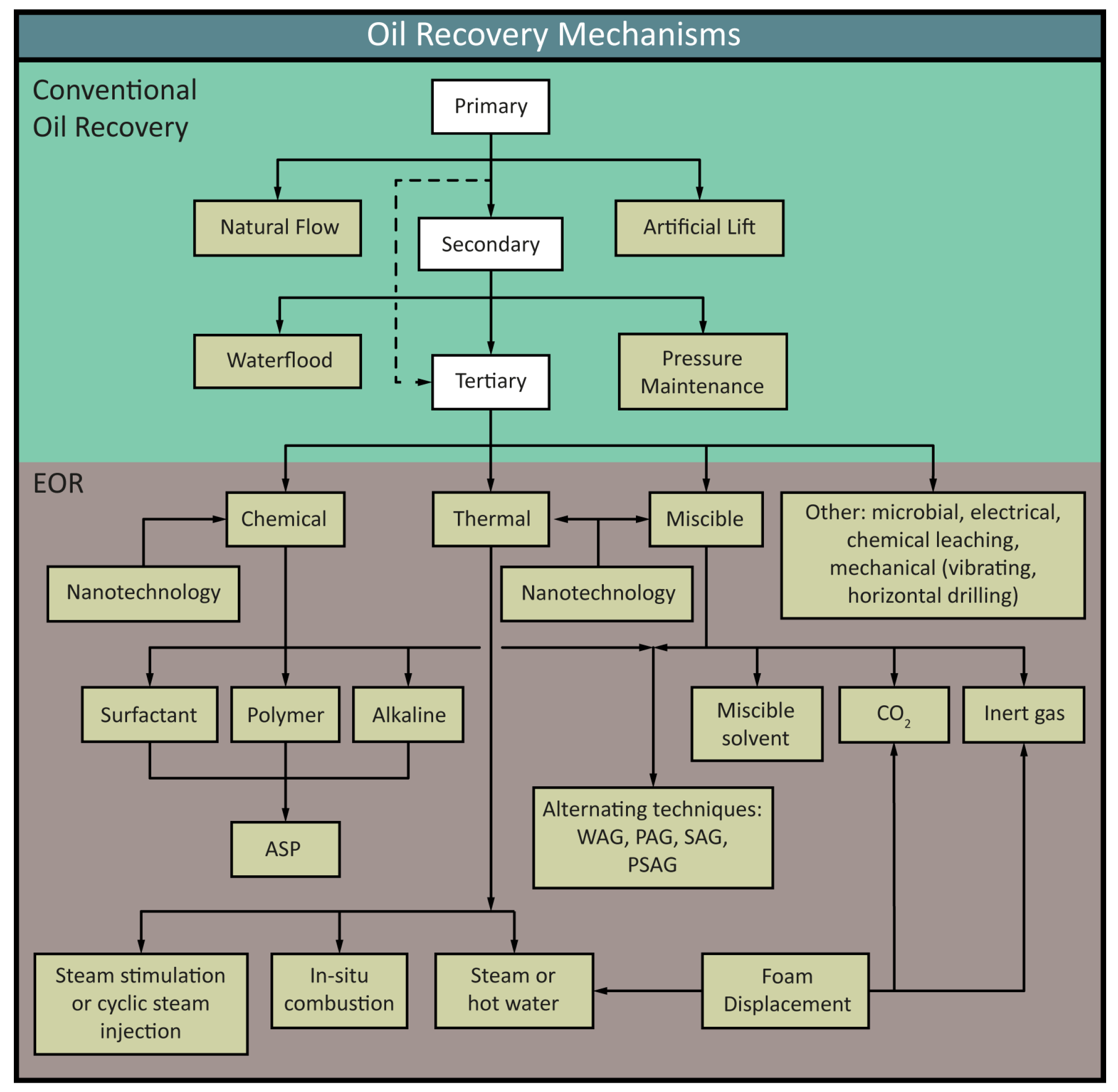
1. Introduction
Aim of This Review
2. Nanofluids
2.1. Nanofluids and Enhanced Oil Recovery
2.2. Effect of Nanoparticles on Reservoir and Fluid Properties
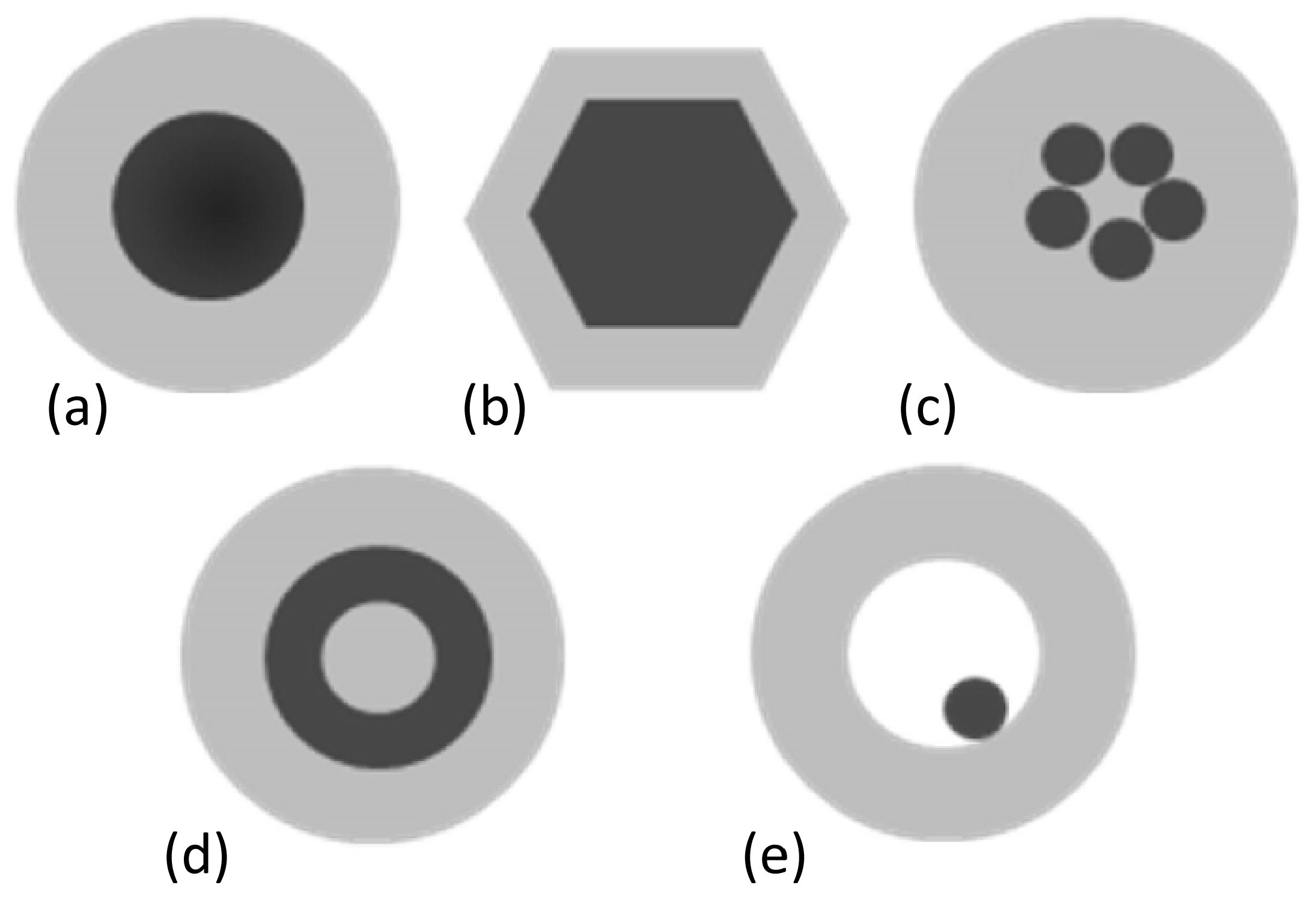
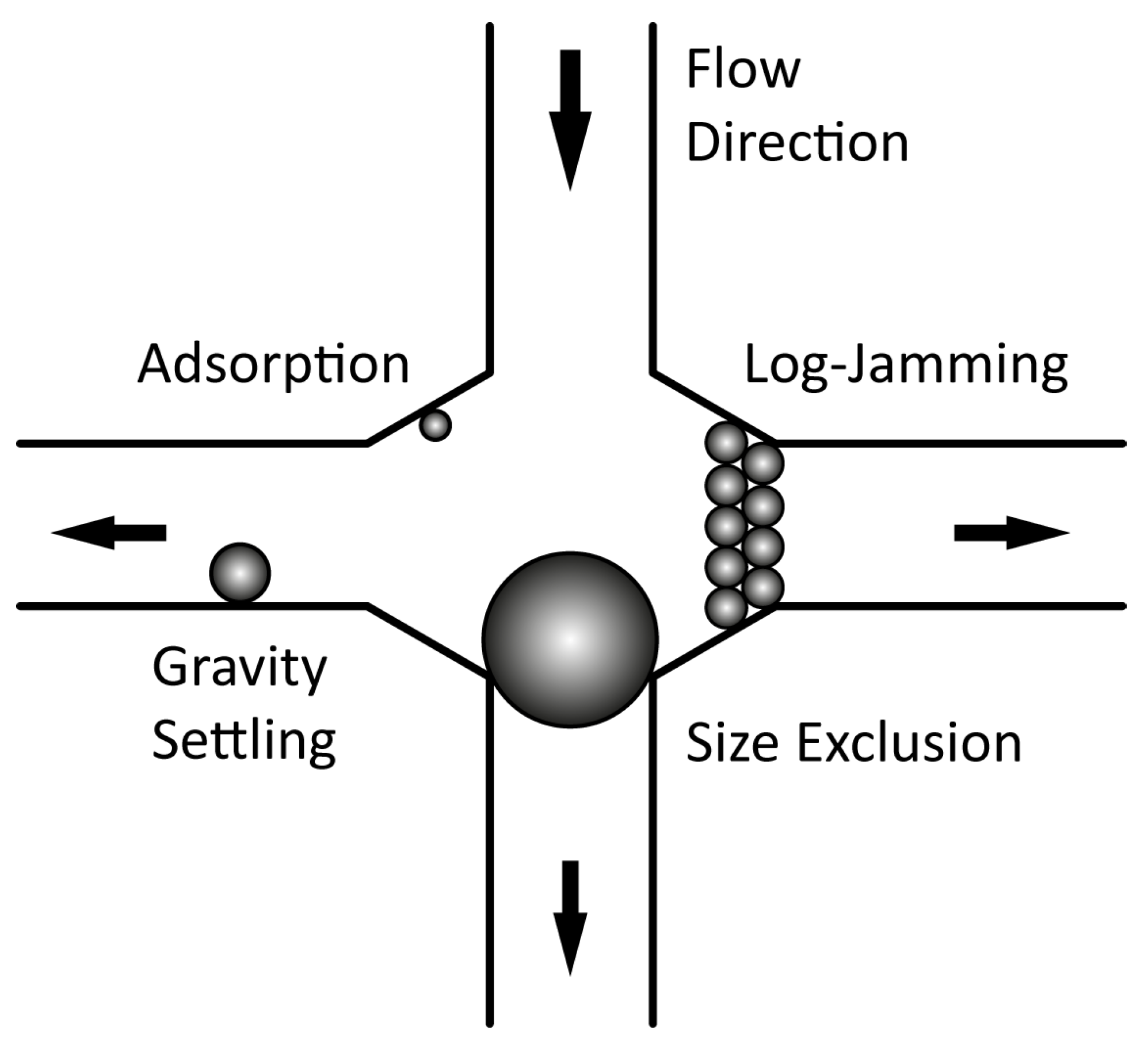
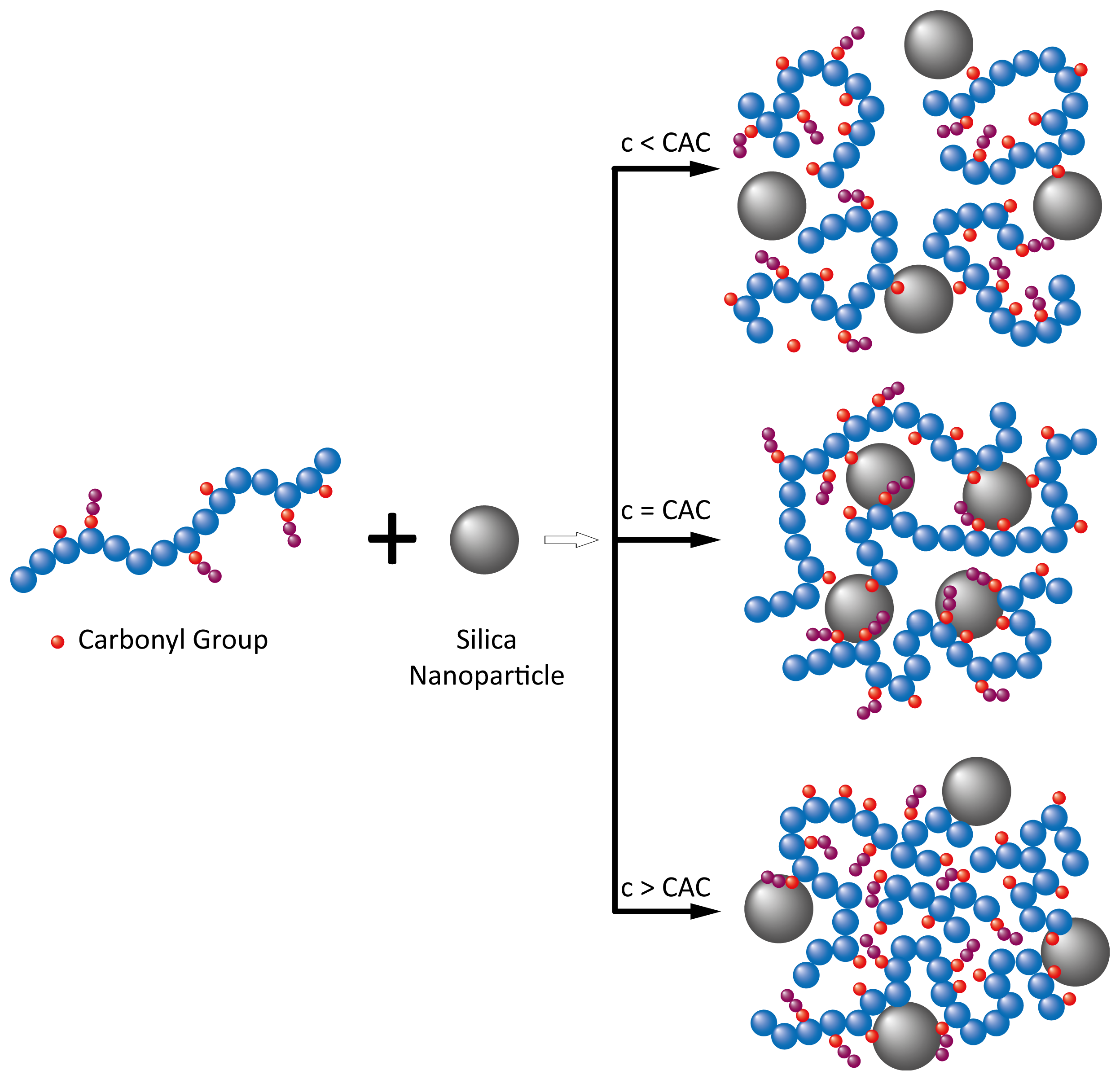
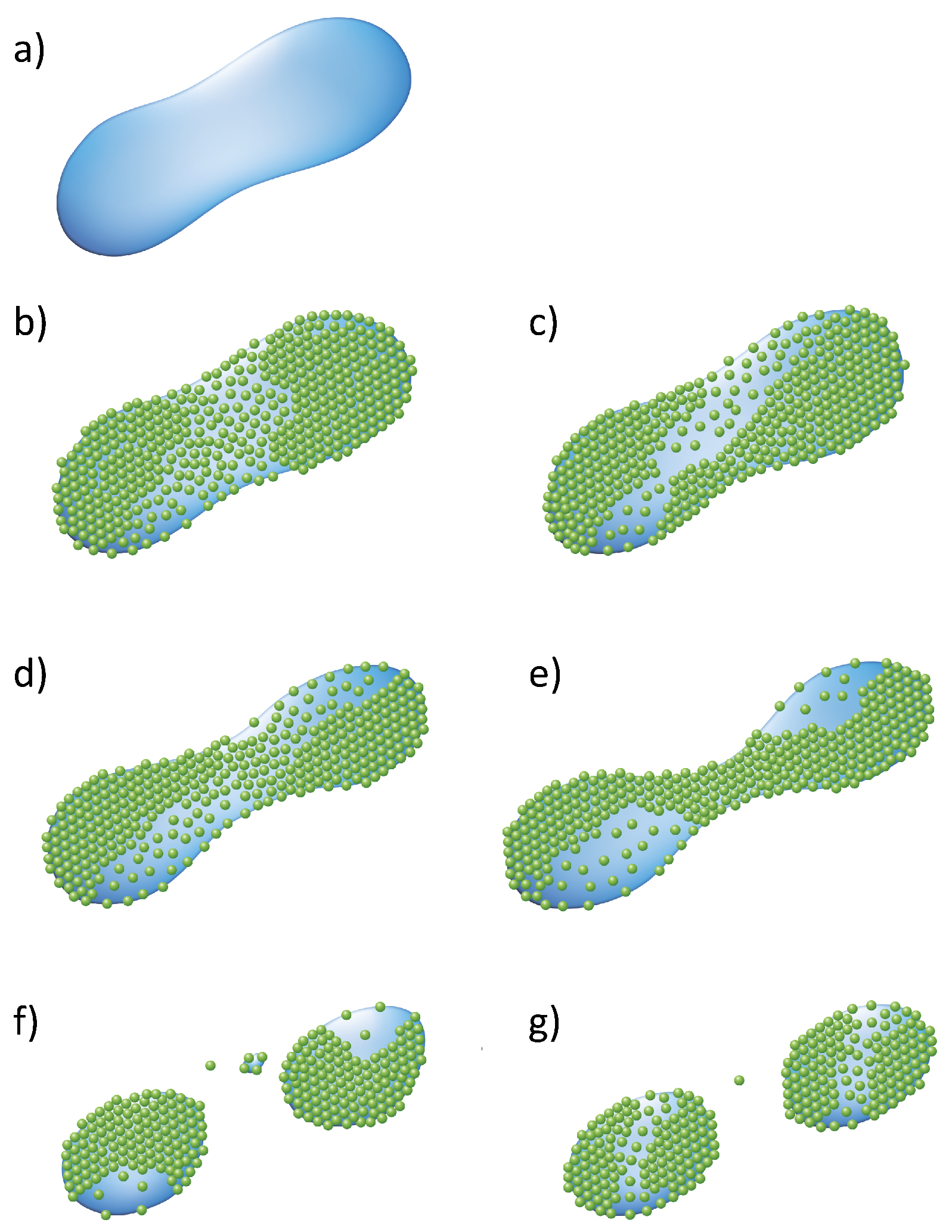
2.2.1. Aggregation and Retention in Porous Media
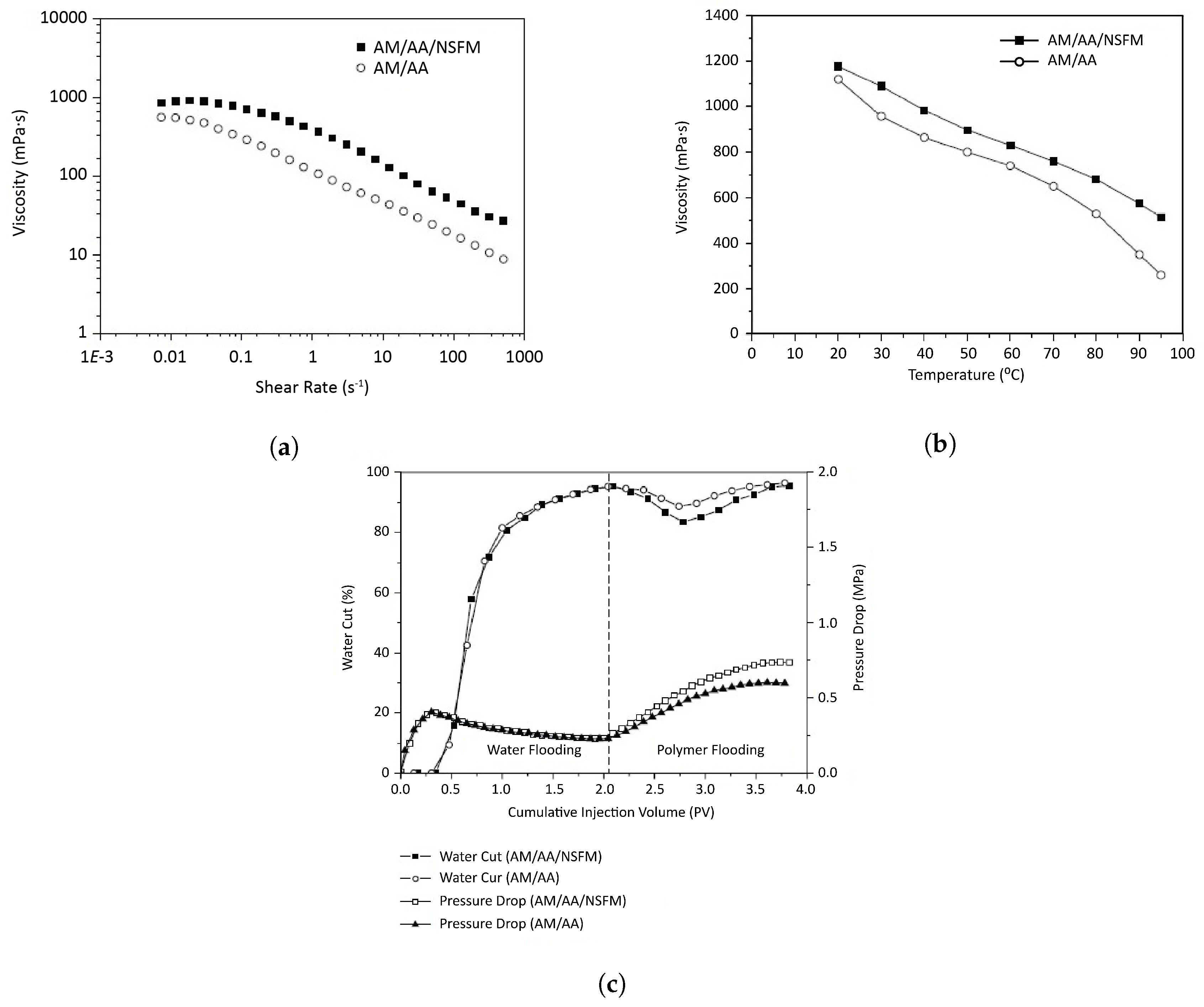
2.2.2. Effect on Rheology and Viscoelasticity
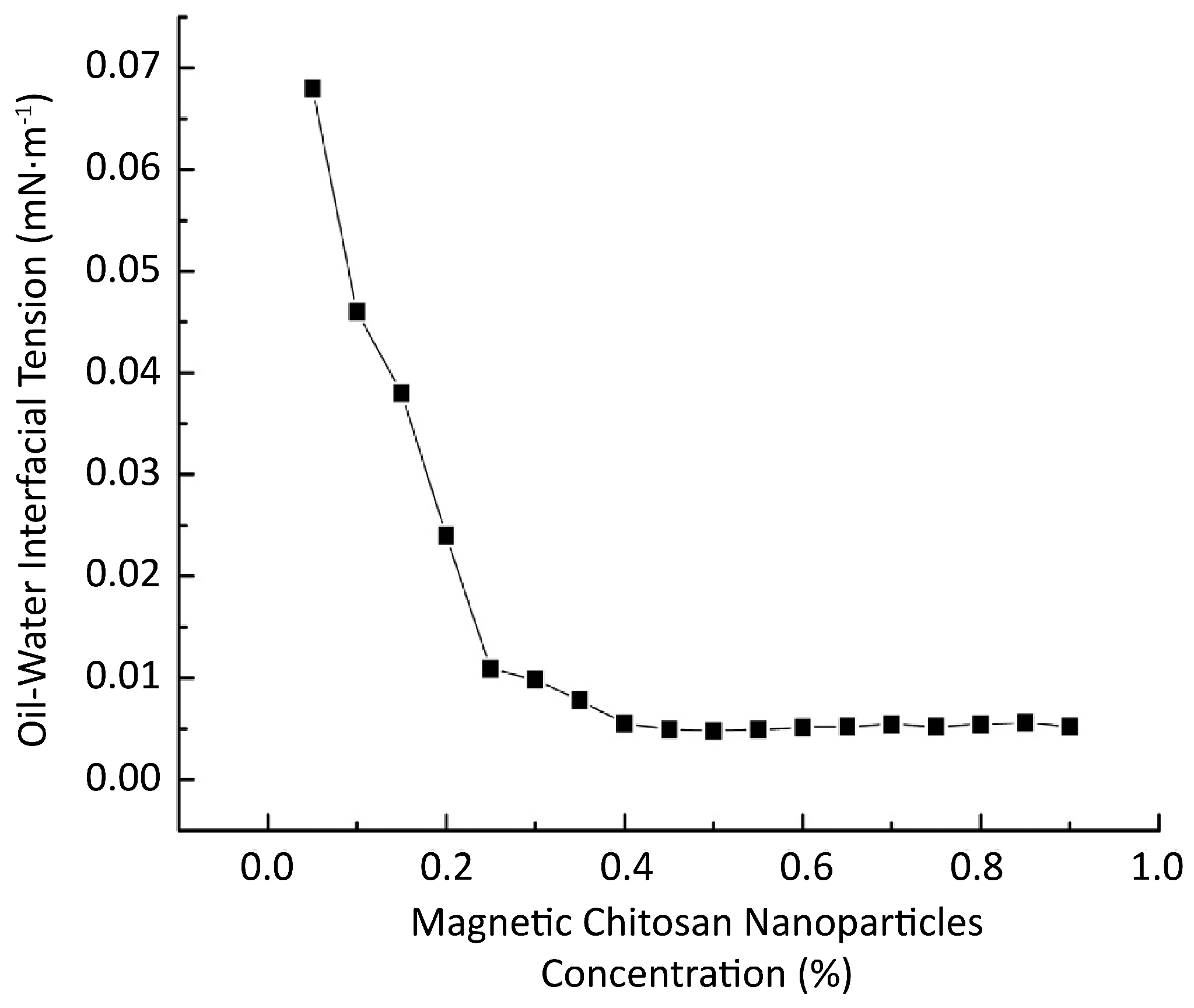
2.2.3. Effect on Interfacial Tension
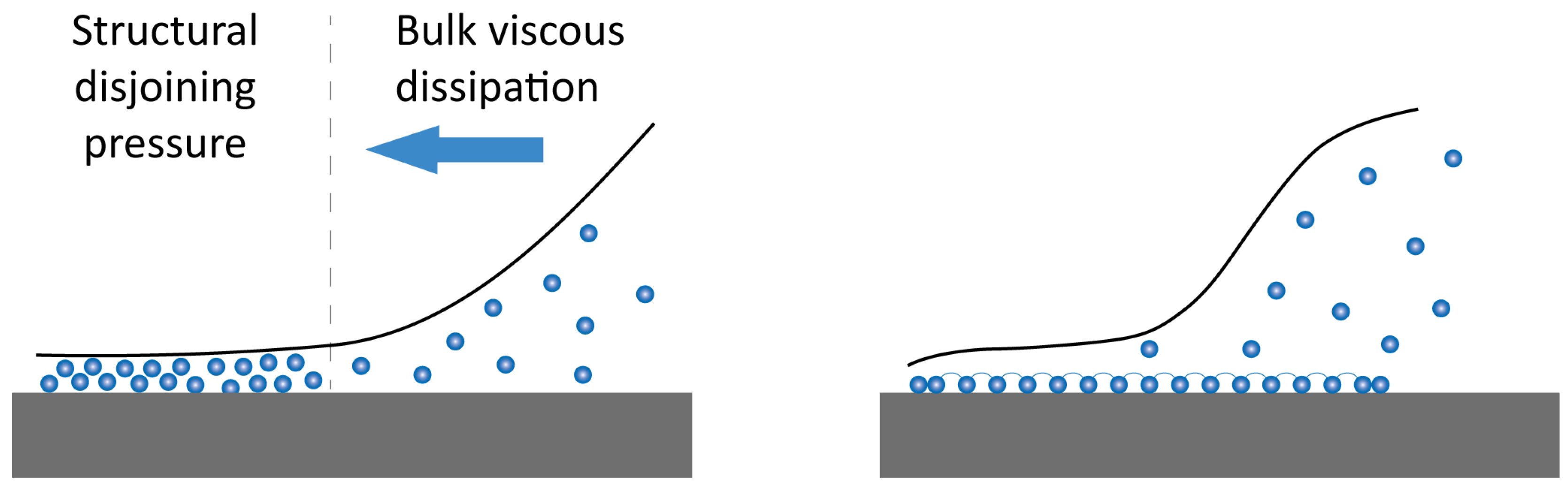
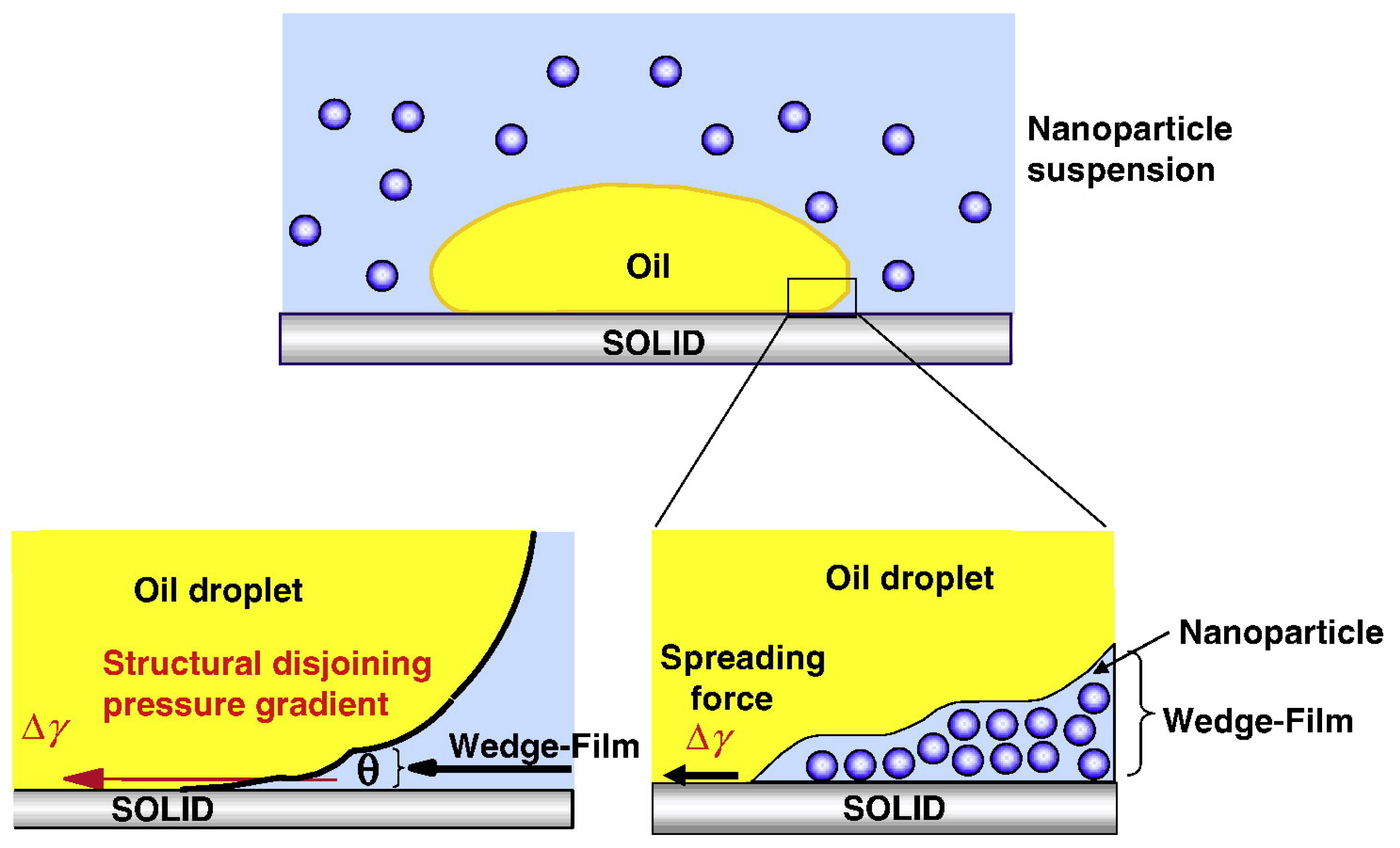
2.2.4. Effect on the Rock Wettability and Surface Wetting
3. Nanoemulsions
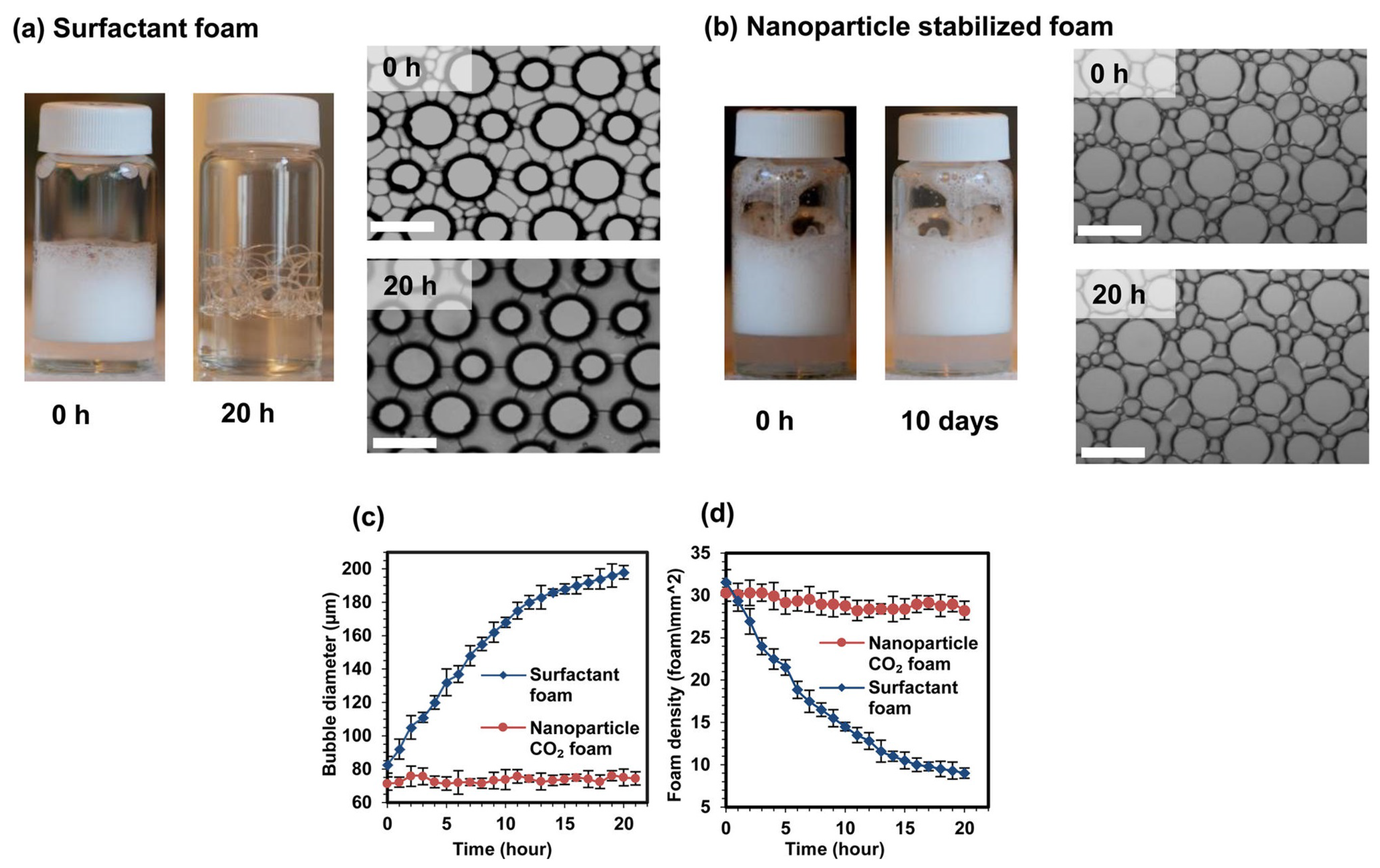
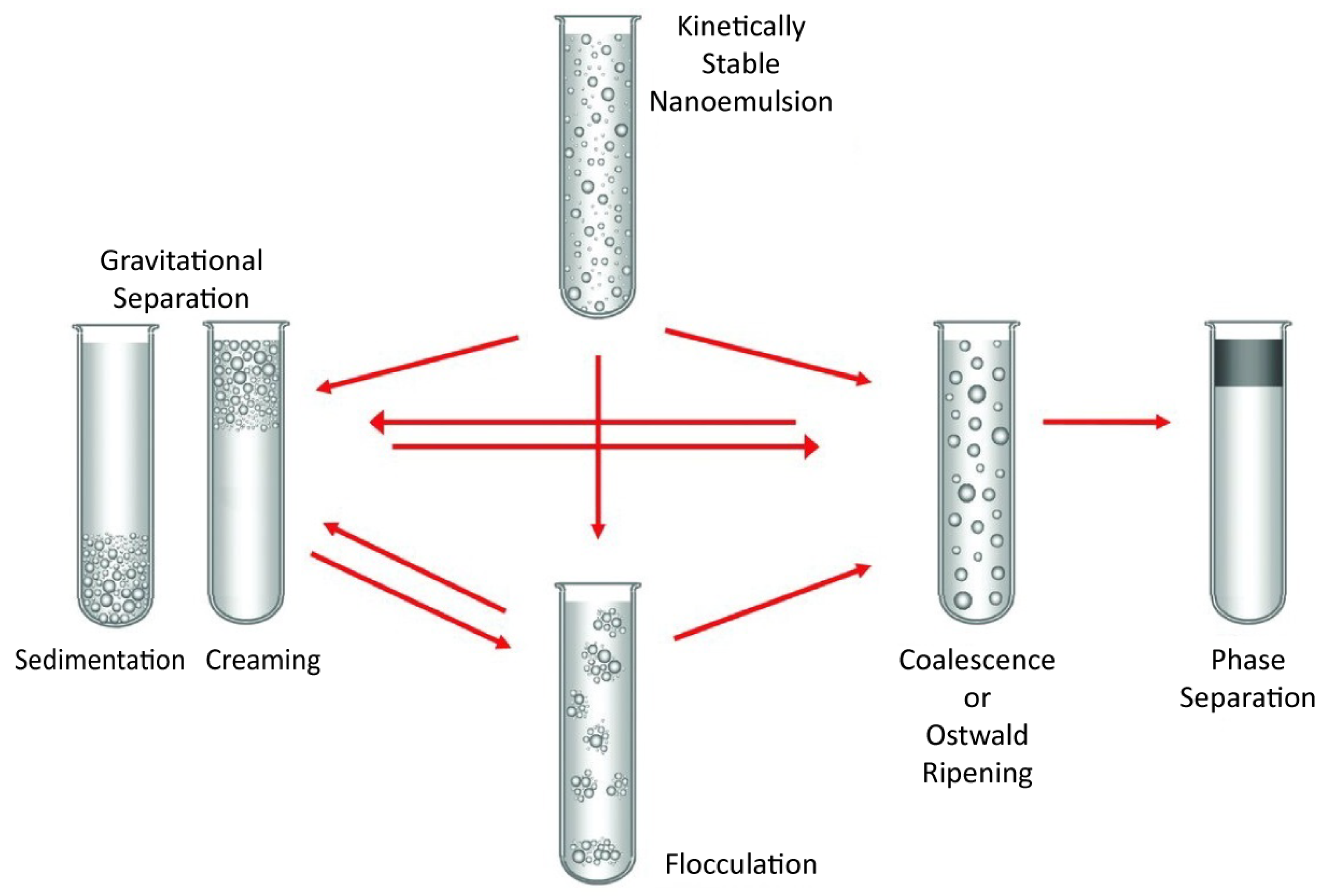
3.1. Nanoemulsion Stability
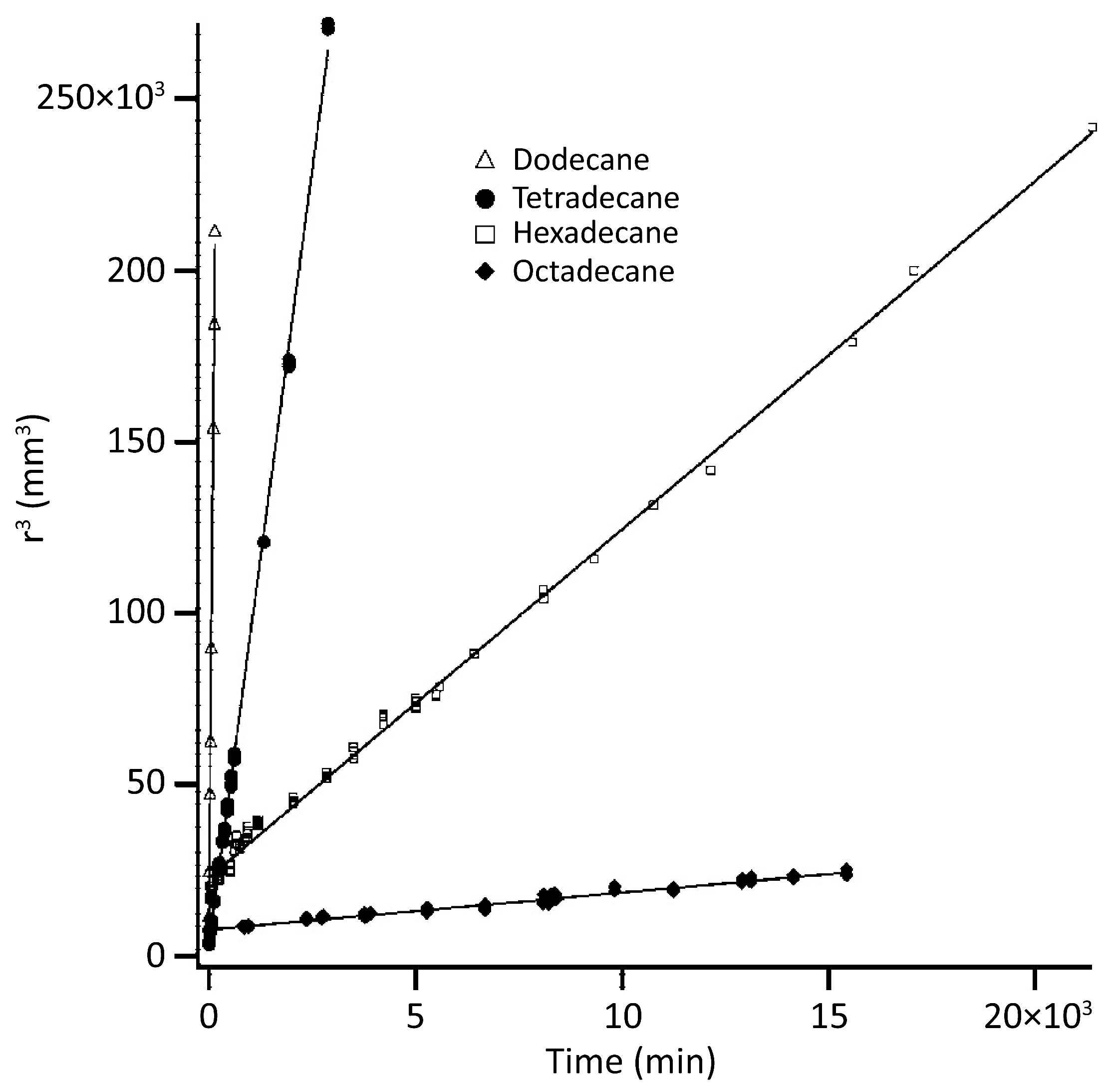
Ostwald Ripening
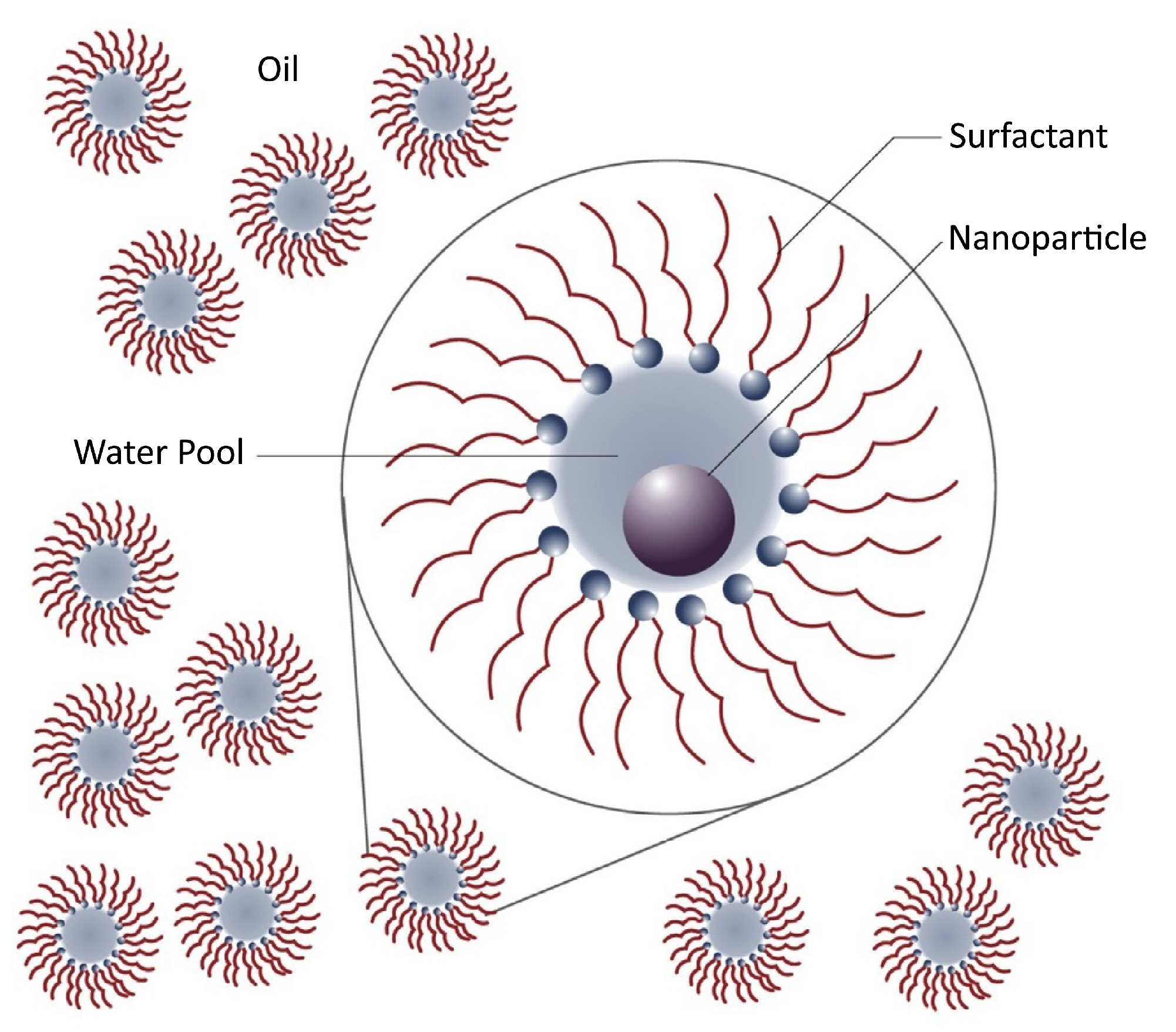
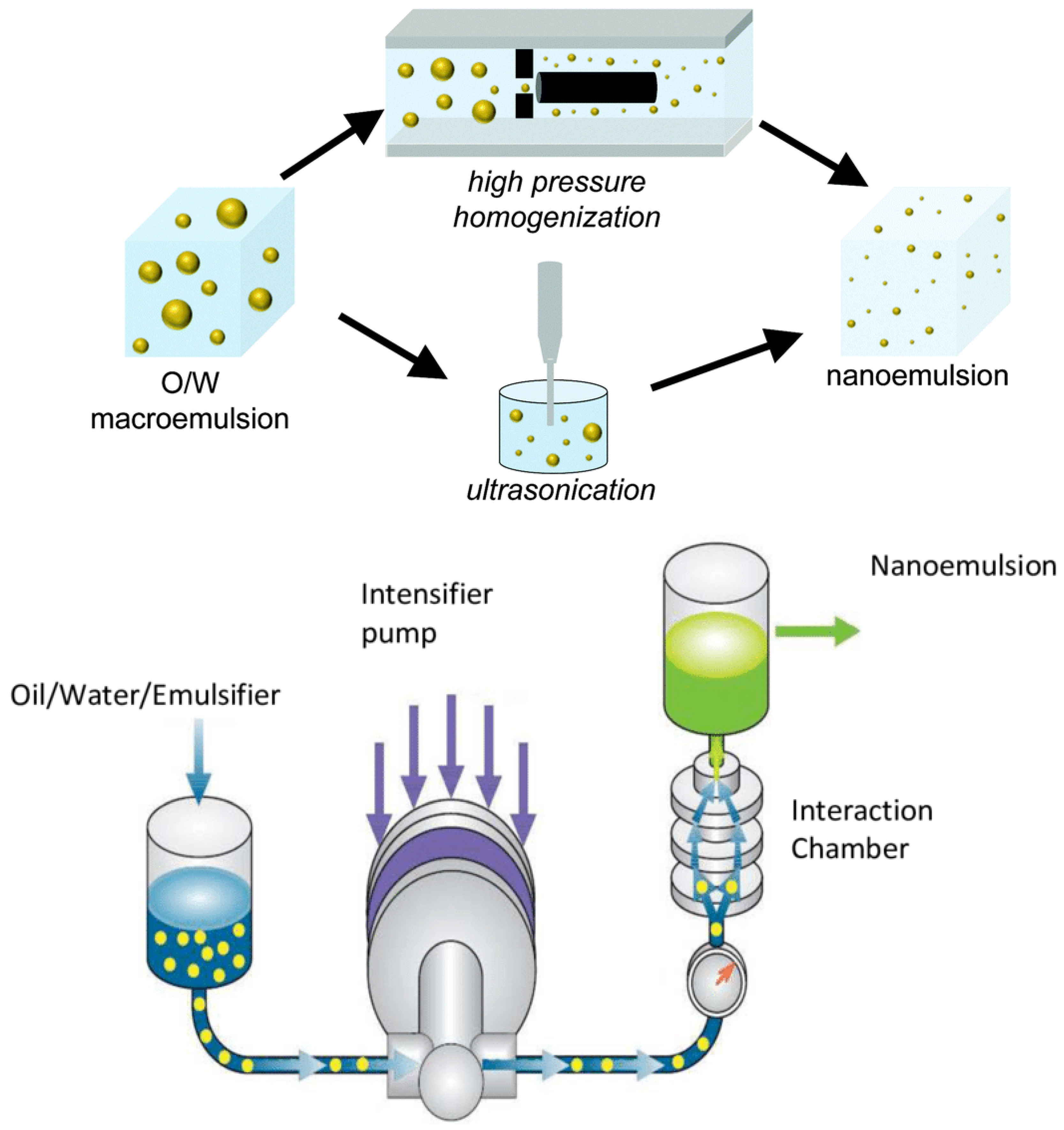
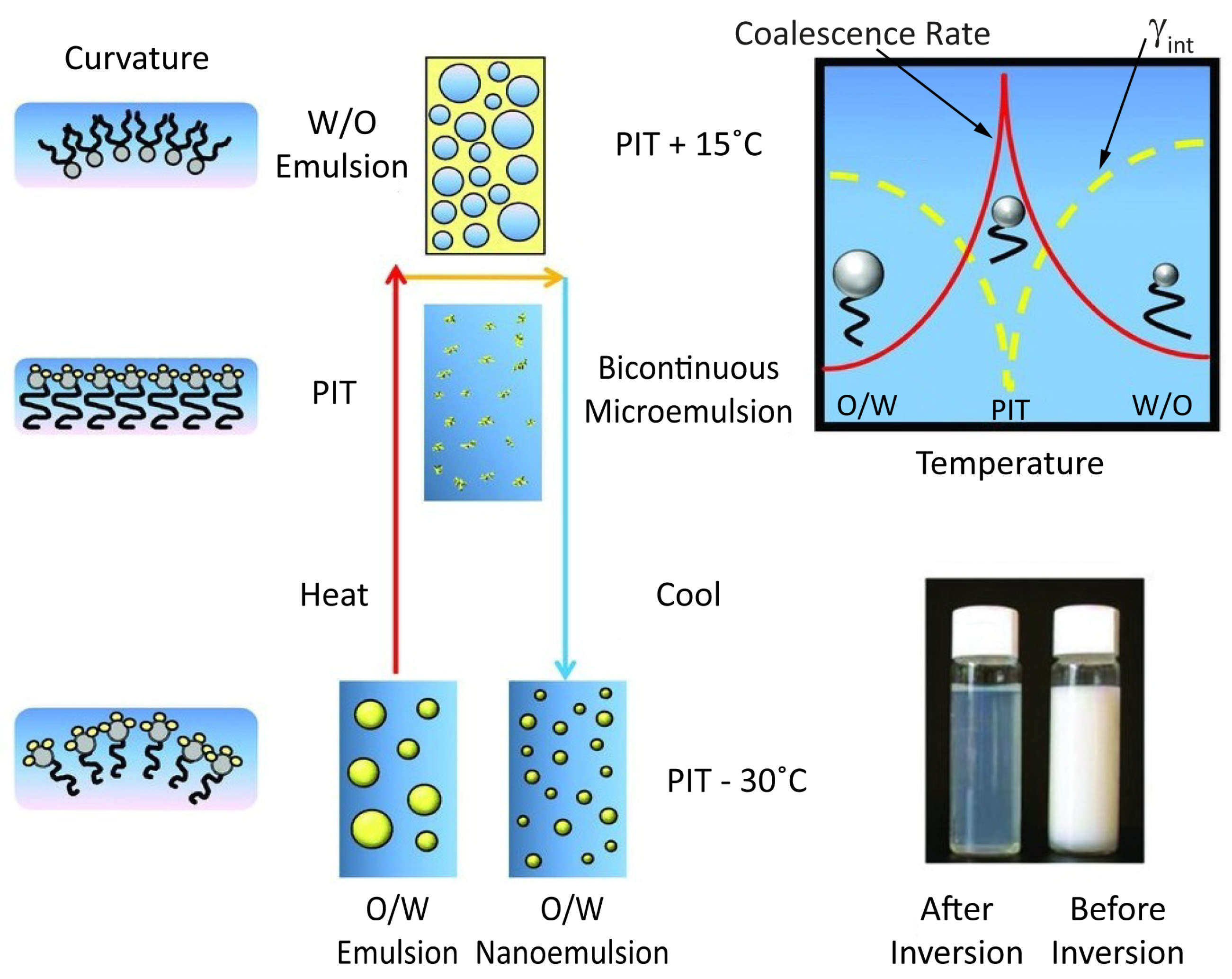
3.2. Preparation of Nanoemulsions
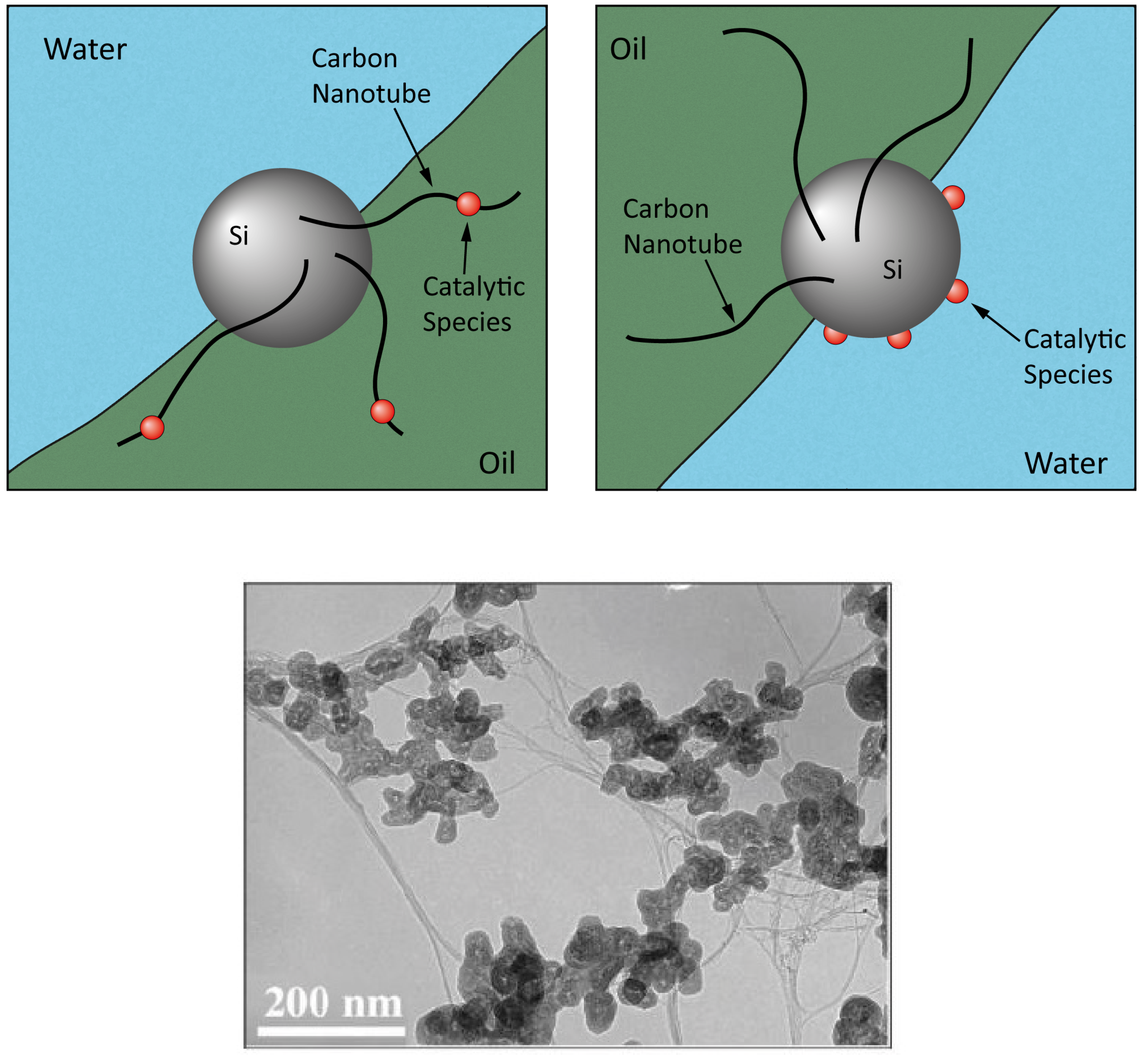
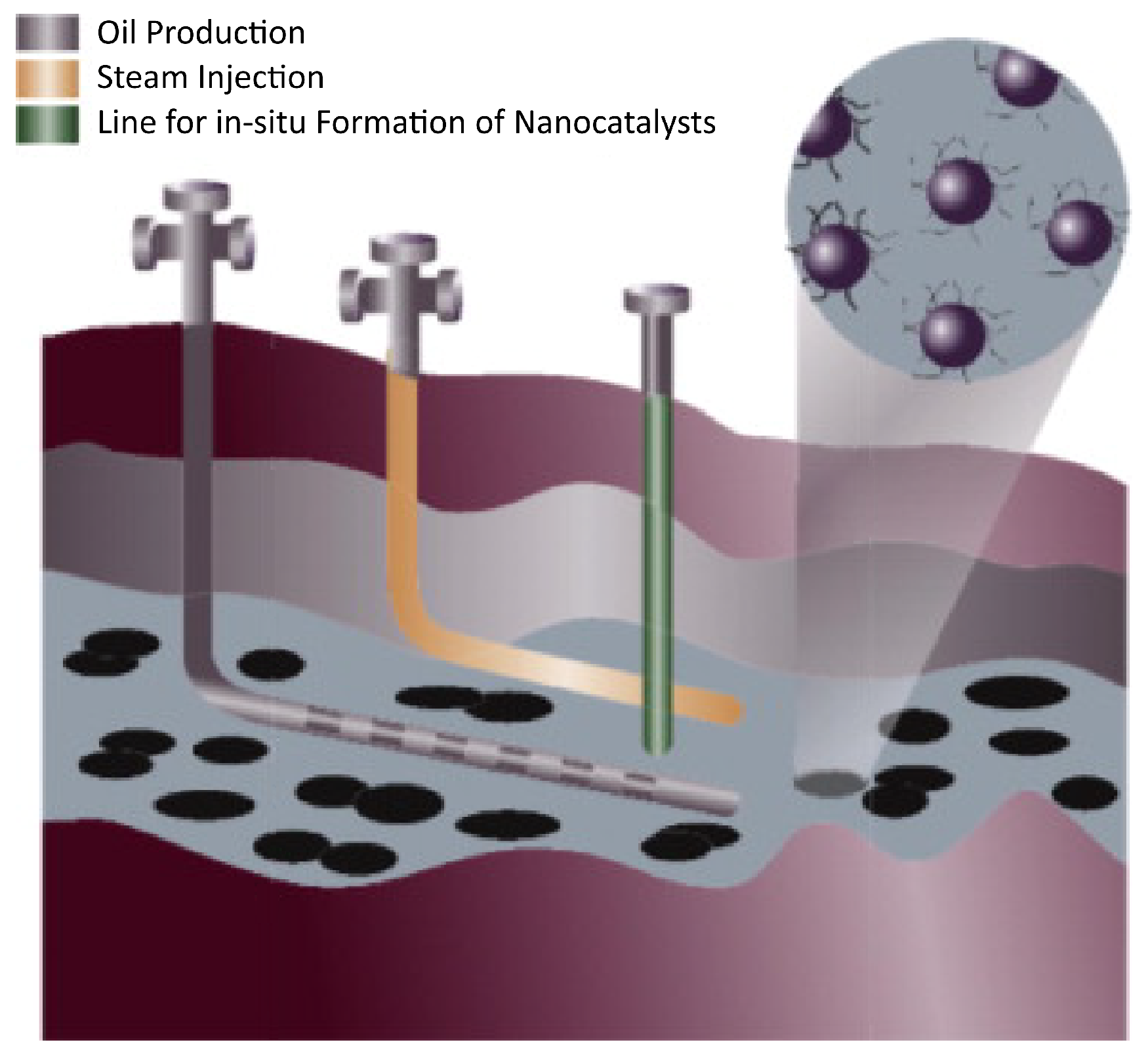
4. Nanocatalysts
4.1. Chemistry and Kinetics of the Process
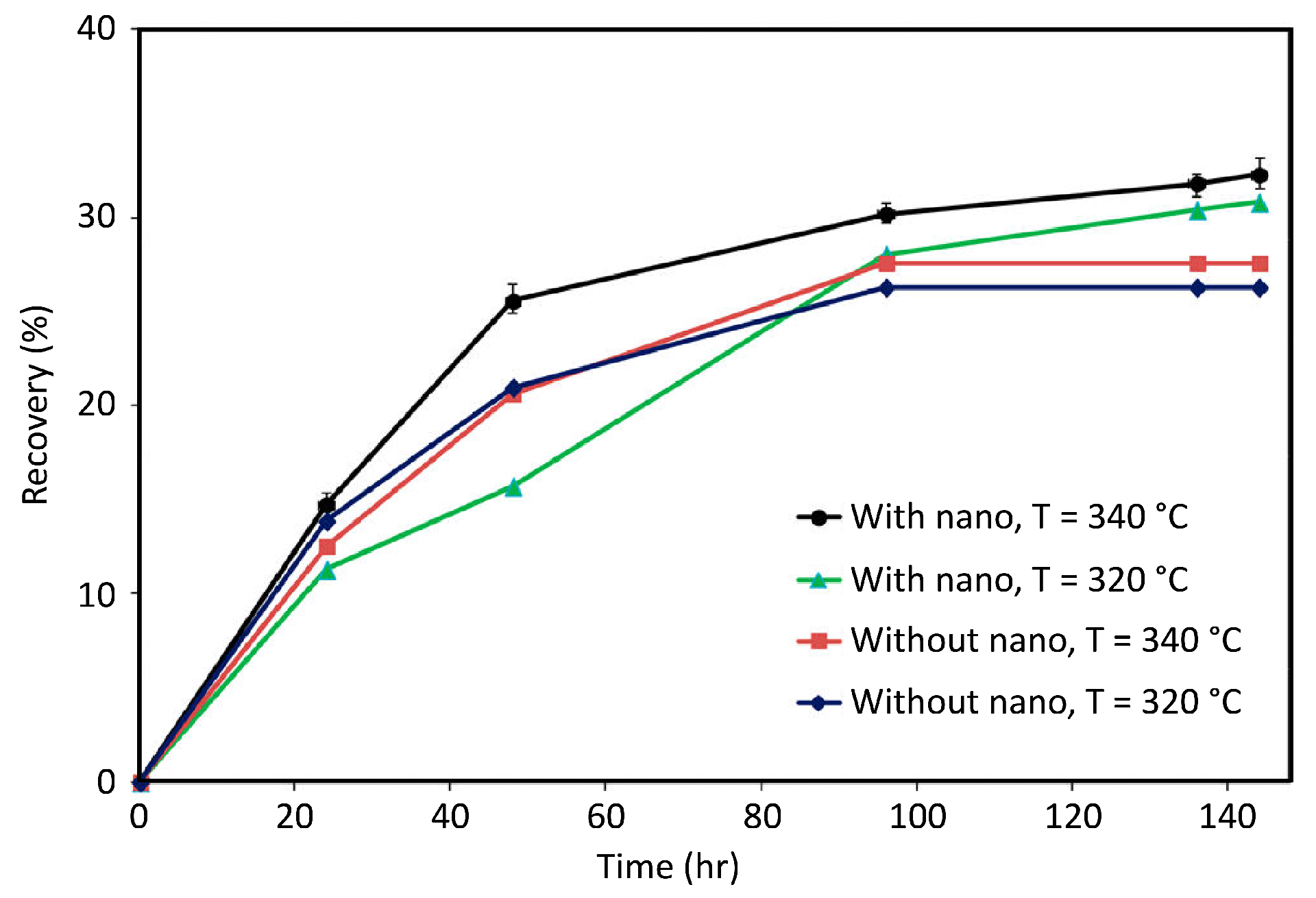
4.2. Factors Affecting Nanocatalysts in EOR Applications
4.2.1. Type, Size, and Concentration of Nanoparticles
4.2.2. Heat Transfer
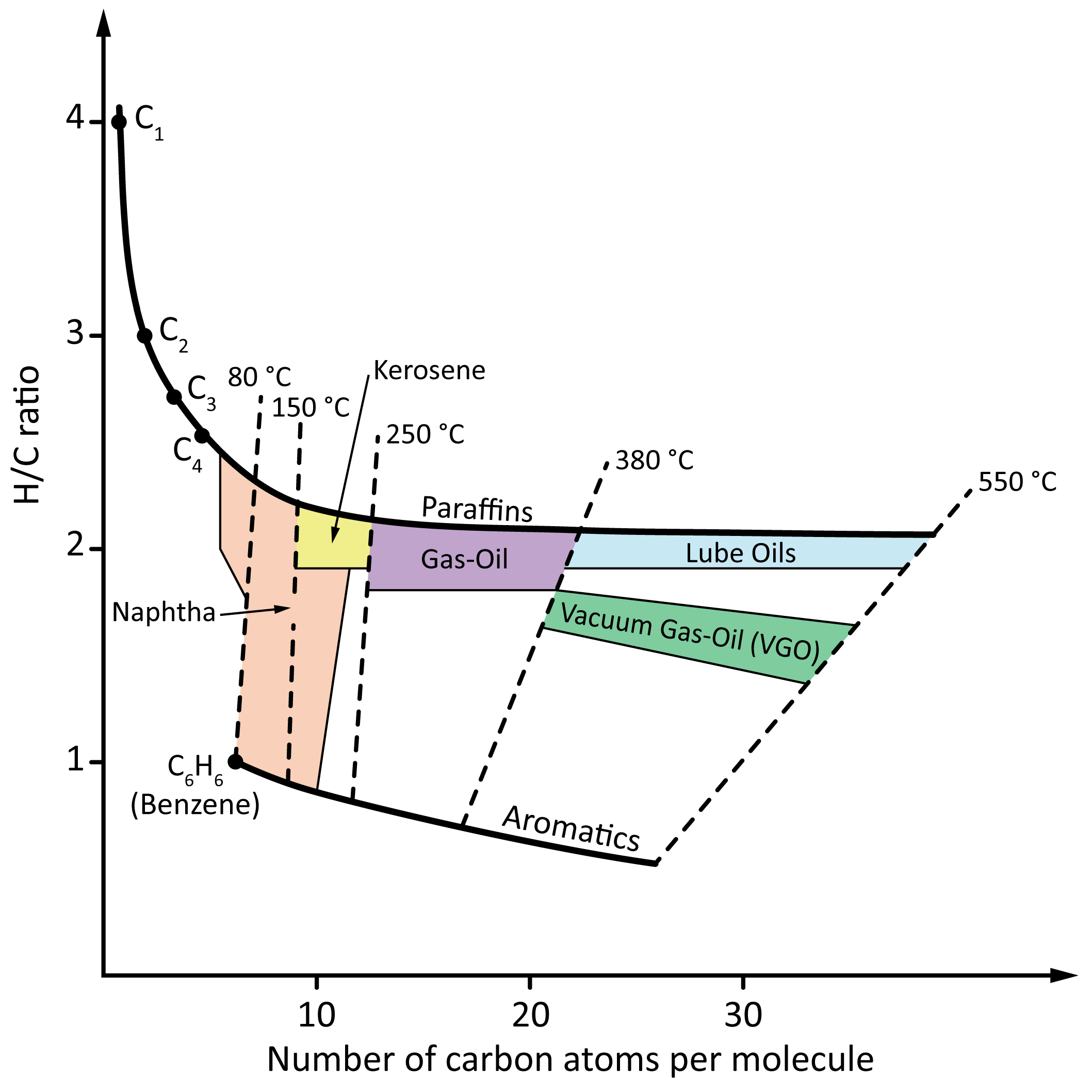
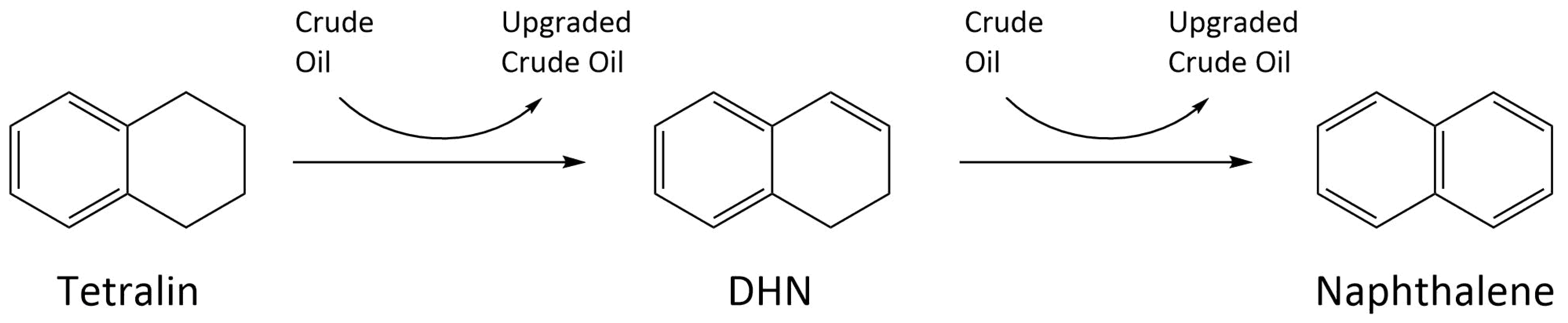
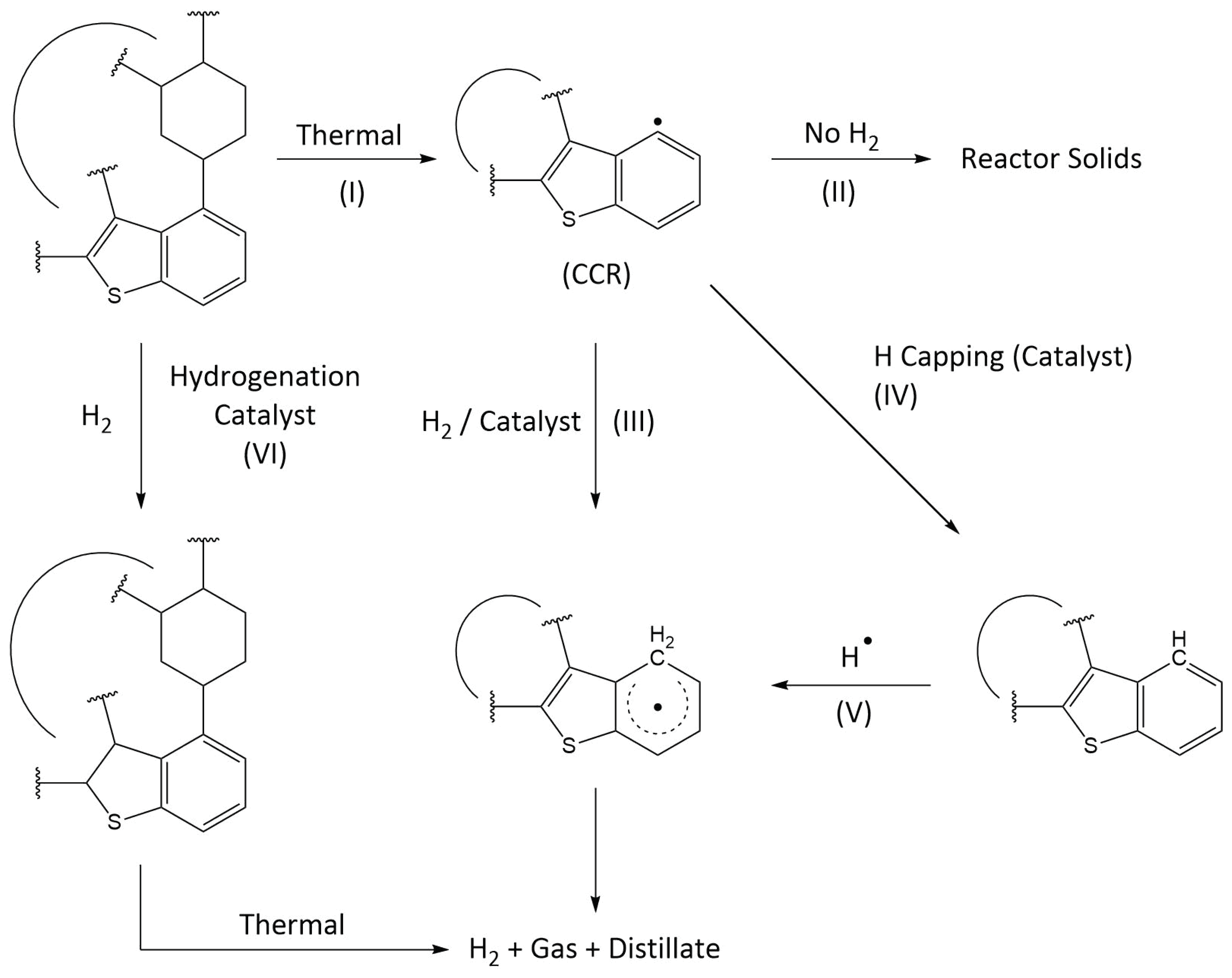
4.2.3. Crude Oil Composition
4.2.4. Porous Media
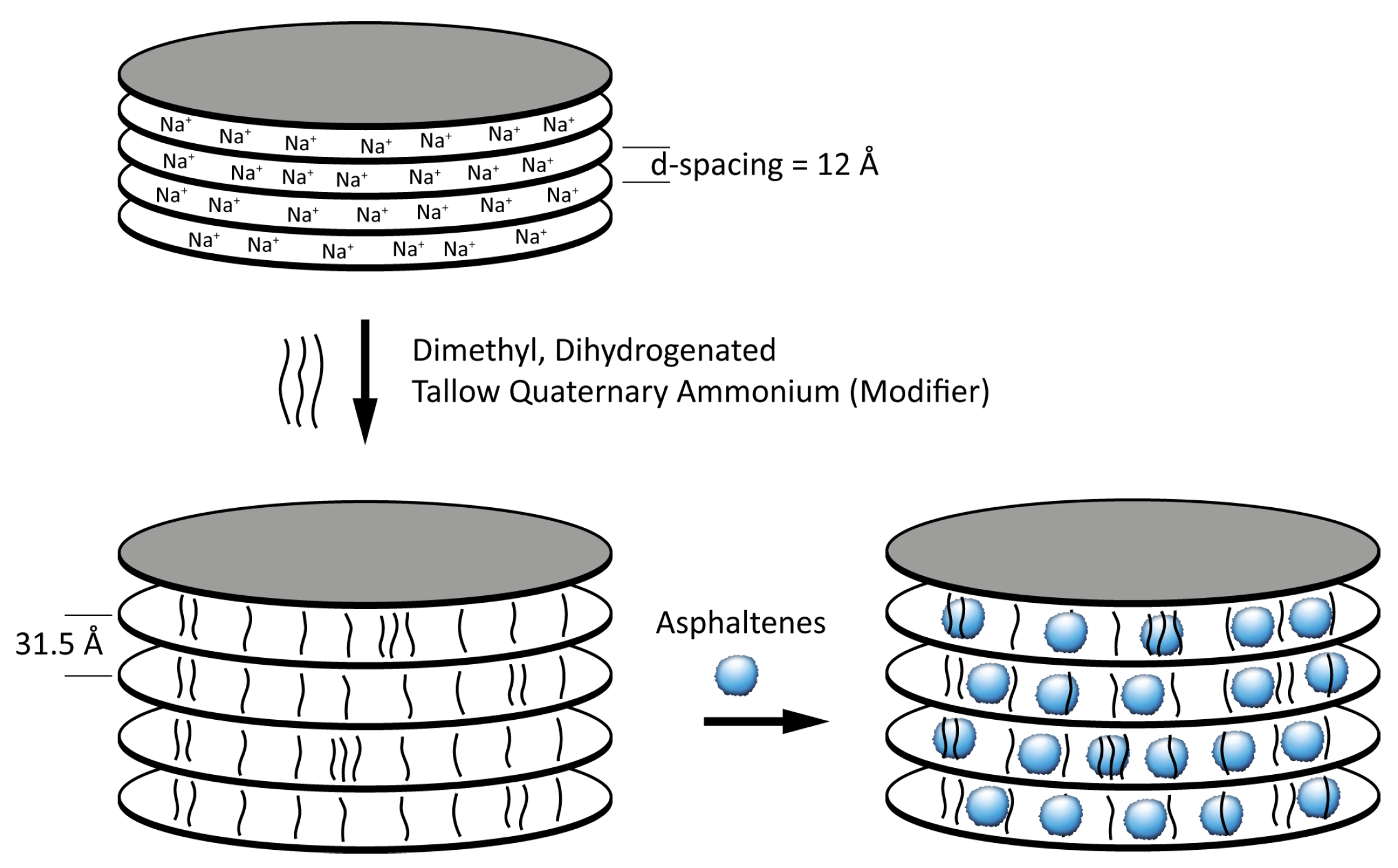
4.3. Inhibition of Formation Damage
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| EOR | Enhanced Oil Recovery |
| IOIP | Initial Oil in Place |
| PNP | Polymer-Coated Nanoparticle |
| IFT | Interfacial Tension |
| PSNP | Polysilicon Nanoparticle |
| HLPN | Hydrophobic and Lipophilic Nanoparticle |
| LHPN | Lipophobic and Hydrophilic Nanoparticle |
| NWPN | Neutral Wet Nanoparticle |
| DLVO | Derjaguin, Landau, Verwey, and Overbeek theory |
| HAPAM | Hydrophobically Associating Polyacrylamide |
| HPAM | Hydrolyzed Polyacrylamide |
| SPAM | Sulfonated Polyacrylamide |
| AMPS | 2-Acrylamido-2-methylpropane Sulfonic Acid |
| SDS | Sodium Dodecyl Sulfate |
| TDS | Total Dissolved Solids |
| AM | Acrylamide |
| AA | Acrylic Acid |
| NSFM | Nano-SiO2 Functional Monomer |
| CAC | Critical Aggregation Concentration |
| CNT | Carbon Nanotube |
| SEM | Scanning Electron Microscope |
| AFM | Atomic Force Microscope |
| PIT | Phase Inversion Temperature |
| EIP | Emulsion Inversion Point |
| VGO | Vacuum Gas Oil |
| VAPEX | Vapor Extraction Process |
| DHN | Decahydronaphthalene |
References
- Chapman, I. The end of Peak Oil? Why this topic is still relevant despite recent denials. Energy Policy 2014, 64, 93–101. [Google Scholar] [CrossRef]
- Mercer, J.W.; Cohen, R.M. A Review of Immiscible Fluids in the Subsurface: Properties, Models, Characterization and Remediation. J. Contam. Hydrol. 1990, 6, 107–163. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Doulabi, M. Improvement in Oil Absorbency by Using Modified Carbon Nanotubes in Preparation of Oil Sorbents. Adv. Polym. Technol. 2013, 32. [Google Scholar] [CrossRef]
- ShamsiJazeyi, H.; Miller, C.A.; Wong, M.S.; Tour, J.M.; Verduzco, R. Polymer-Coated Nanoparticles for Enhanced Oil Recovery. J. Appl. Polym. Sci. 2014, 131, 40576. [Google Scholar] [CrossRef]
- Zhang, H.; Nikolov, A.; Wasan, D. Enhanced Oil Recovery (EOR) Using Nanoparticle Dispersions: Underlying Mechanism and Imbibition Experiments. Energy Fuels 2014, 28, 3002–3009. [Google Scholar] [CrossRef]
- Suleimanov, B.A.; Ismailov, F.S.; Veliyev, E.F. Nanofluid for enhanced oil recovery. J. Pet. Sci. Eng. 2011, 78, 431–437. [Google Scholar] [CrossRef]
- Maghzi, A.; Mohebbi, A.; Kharrat, R.; Ghazanfari, M.H. Pore-Scale Monitoring of Wettability Alteration by Silica Nanoparticles During Polymer Flooding to Heavy Oil in a Five-Spot Glass Micromodel. Transp. Porous Media 2011, 87, 653–664. [Google Scholar] [CrossRef]
- Zhu, D.; Han, Y.; Zhang, J.; Li, X.; Feng, Y. Enhancing Rheological Properties of Hydrophobically Associative Polyacrylamide Aqueous Solutions by Hybriding with Silica Nanoparticles. J. Appl. Polym. Sci. 2014, 131, 40876. [Google Scholar] [CrossRef]
- Khezrnejad, A.; James, L.A.; Johansen, T.E. Water Enhancement Using Nanoparticles in Water Alternating Gas (WAG) Micromodel Experiments. In SPE Annual Technical Conference and Exhibition; Society of Petroleum Engineers: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Alvarado, V.; Manrique, E. Enhanced Oil Recovery: An Update Review. Energies 2010, 3, 1529–1575. [Google Scholar] [CrossRef]
- Schmidt, R.L.; Venuto, P.B.; Lake, L.W. A Niche for Enhanced Oil Recovery in the 1990’s. Oilfield Rev. 1992, 4, 55–61. [Google Scholar]
- Homsy, G.M. Viscous Fingering in Porous-Media. Ann. Rev. Fluid Mech. 1987, 19, 271–311. [Google Scholar] [CrossRef]
- Ratner, M.A.; Ratner, D. Nanotechnology: A Gentle Introduction to the Next Big Idea; Prentice Hall: Upper Saddle River, NJ, USA, 2003; ISBN 978-0-13101-400-8. [Google Scholar]
- Evdokimov, I.N.; Eliseev, N.Y.; Losev, A.P.; Novikov, M.A. Emerging Petroleum-Oriented Nanotechnologies for Reservoir Engineering. In SPE Russian Oil and Gas Technical Conference and Exhibition; Society of Petroleum Engineers: Moscow, Russia, 2006. [Google Scholar]
- Khalil, M.; Jan, B.M.; Tong, C.W.; Berawi, M.A. Advanced nanomaterials in oil and gas industry: Design, application and challenges. Appl. Energy 2017, 191, 287–310. [Google Scholar] [CrossRef]
- Cocuzza, M.; Pirri, C.; Rocca, V.; Verga, F. Current and Future Nanotech Applications in the Oil Industry. Am. J. Appl. Sci. 2012, 9, 784–793. [Google Scholar] [CrossRef]
- Terranova, M.; Piccirillo, S.; Sessa, V.; Rossi, M.; Botti, S. Microstructure and properties of nanocomposite diamond films obtained by a new CVD-based technique. J. Phys. IV 1999, 9, 365–371. [Google Scholar] [CrossRef]
- Jain, M.; Sadangi, R.; Cannon, W.; Kear, B. Processing of functionally graded WC/Co/diamond nanocomposites. Scr. Mater. 2001, 44, 2099–21031. [Google Scholar] [CrossRef]
- Terranova, M.; Piccirillo, S.; Sessa, V.; Rossi, M.; Cappuccio, G. A new CVD route for the synthesis of composite diamond-based materials. Chem. Vap. Depos. 1999, 5, 101–105. [Google Scholar] [CrossRef]
- Ryoo, S.; Rahmani, A.; Yoon, K.; Prodanovic, M.; Kotsmar, C.; Milner, T.; Johnston, K.; Bryant, S.; Huh, C. Theoretical and experimental investigation of the motion of multiphase fluids containing paramagnetic nanoparticles in porous media. In SPE Annual Technical Conference and Exhibition; Society of Petroleum Engineers: Florence, Italy, 2010. [Google Scholar]
- Dubrovinskaia, N.; Solozhenko, V.L.; Miyajima, N.; Dmitriev, V.; Kurakevych, O.O.; Dubrovinsky, L. Superhard nanocomposite of dense polymorphs of boron nitride: Noncarbon material has reached diamond hardness. Appl. Phys. Lett. 2007, 90, 101912. [Google Scholar] [CrossRef]
- Bera, A.; Belhaj, H. Application of nanotechnology by means of nanoparticles and nanodispersions in oil recovery—A comprehensive review. J. Nat. Gas Sci. Eng. 2016, 34, 1284–1309. [Google Scholar] [CrossRef]
- Yu, J.; Rahmani, A.; Yoon, K.; Prodanovic, M.; Kotsmar, C.; Milner, T.; Johnston, K.; Bryant, S.; Huh, C. Transport study of nanoparticles for oilfield application. In SPE International Conference on Oilfield Scale; Society of Petroleum Engineers: Aberdeen, UK, 2010. [Google Scholar]
- Fakoya, M.; Shah, S. Emergence of nanotechnology in the oil and gas industry: Emphasis on the application of silica nanoparticles. Petroleum 2017, 3, 391–405. [Google Scholar] [CrossRef]
- Negin, C.; Ali, S.; Xie, Q. Application of nanotechnology for enhancing oil recovery—A review. Petroleum 2016, 2, 324–333. [Google Scholar] [CrossRef]
- Fletcher, A.; Davis, J. How EOR Can be Transformed by Nanotechnology. In SPE Improved Oil Recovery Symposium; Society of Petroleum Engineers: Tulsa, USA, 2010. [Google Scholar]
- Alomair, O.A.; Matar, K.M.; Alsaeed, Y.H. Nanofluids Application for Heavy Oil Recovery. In SPE Asia Pacific Oil & Gas Conference and Exhibition; Society of Petroleum Engineers: Adelaide, Australia, 2014. [Google Scholar]
- Singh, R.; Mohanty, K. Nanoparticle-Stabilized Foams for High-Temperature, High-Salinity Oil Reservoirs. In SPE Annual Technical Conference and Exhibition; Society of Petroleum Engineers: San Antonio, OK, USA, 2017. [Google Scholar]
- Al-Hamad, M.; Sultan, A.; Khan, S.; Abdallah, W. Challenges for Extending the Application of Nanoparticles in High Salinity Reservoirs. In SPE Kingdom of Saudi Arabia Annual Technical Symposium and Exhibition; Society of Petroleum Engineers: Dammam, Saudi Arabia, 2016. [Google Scholar]
- Al-Anssari, S.; Barifcani, A.; Keshavarz, A.; Iglauer, S. Impact of nanoparticles on the CO2-brine interfacial tension at high pressure and temperature. J. Colloid Interface Sci. 2018, 532, 136–142. [Google Scholar] [CrossRef]
- Mcelfresh, P.; Wood, M.; Ector, D. Stabilizing Nano Particle Dispersions in High Salinity, High Temperature Downhole Environments. In SPE International Oilfield Nanotechnology Conference and Exhibition; Society of Petroleum Engineers: Noordwijk, The Netherlands, 2012. [Google Scholar]
- Engeset, B. The Potential of Hydrophilic Silica Nanoparticles for EOR Purposes. Master’s Thesis, Norwegian University of Science and Technology, Trondheim, Norway, 2012. [Google Scholar]
- Chaudhury, M.K. Complex fluids: Spread the word about nanofluids. Nature 2003, 423, 131–132. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, A. Applications of Nanotechnology in Oil and Gas Industry; American Institute of Physics Conference Series; Patel, R.B., Singh, B.P., Eds.; American Institute of Physics: College Park, MA, USA, 2011; Volume 1414, pp. 133–136. [Google Scholar]
- Hashemi, R.; Nassar, N.N.; Almao, P.P. Nanoparticle technology for heavy oil in-situ upgrading and recovery enhancement: Opportunities and challenges. Appl. Energy 2014, 133, 374–387. [Google Scholar] [CrossRef]
- Hashemi, R.; Nassar, N.N.; Almao, P.P. Enhanced Heavy Oil Recovery by in Situ Prepared Ultradispersed Multimetallic Nanoparticles: A Study of Hot Fluid Flooding for Athabasca Bitumen Recovery. Energy Fuels 2013, 27, 2194–2201. [Google Scholar] [CrossRef]
- Hashemi, R.; Nassar, N.N.; Pereira-Almao, P. Transport Behavior of Multimetallic Ultradispersed Nanoparticles in an Oil-Sands-Packed Bed Column at a High Temperature and Pressure. Energy Fuels 2012, 26, 1645–1655. [Google Scholar] [CrossRef]
- Peng, B.; Tang, J.; Luo, J.; Wang, P.; Ding, B.; Tam, K.C. Applications of nanotechnology in oil and gas industry: Progress and perspective. Can. J. Chem. Eng. 2018, 96, 91–100. [Google Scholar] [CrossRef]
- Agista, M.N.; Guo, K.; Yu, Z. A State-of-the-Art Review of Nanoparticles Application in Petroleum with a Focus on Enhanced Oil Recovery. Appl. Sci.-Basel 2018, 8, 871. [Google Scholar] [CrossRef]
- Ko, S.; Huh, C. Use of nanoparticles for oil production applications. J. Pet. Sci. Eng. 2019, 172, 97–114. [Google Scholar] [CrossRef]
- Agi, A.; Junin, R.; Gbadamosi, A. Mechanism governing nanoparticle flow behaviour in porous media: Insight for enhanced oil recovery applications. Int. Nano Lett. 2018, 8, 49–77. [Google Scholar] [CrossRef]
- Sergis, A.; Hardalupas, Y. Anomalous heat transfer modes of nanofluids: A review based on statistical analysis. Nanoscale Res. Lett. 2011, 6, 391. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.U.S.; Eastman, J.A. Enhancing thermal conductivity of fluids with nanoparticles. ASME-Publications-Fed 1995, 231, 99–106. [Google Scholar]
- Wong, K.V.; Leon, O.D. Applications of Nanofluids: Current and Future. Adv. Mech. Eng. 2010, 2, 1–11. [Google Scholar] [CrossRef]
- Taylor, R.; Coulombe, S.; Otanicar, T.; Phelan, P.; Gunawan, A.; Lv, W.; Rosengarten, G.; Prasher, R.; Tyagi, H. Small particles, big impacts: A review of the diverse applications of nanofluids. J. Appl. Phys. 2013, 113, 011301. [Google Scholar] [CrossRef]
- Chaudhuri, R.G.; Paria, S. Core/Shell Nanoparticles: Classes, Properties, Synthesis Mechanisms, Characterization, and Applications. Chem. Rev. 2012, 112, 2373–2433. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Xie, T.; Li, Y. Nanocrystals: Solution-Based Synthesis and Applications as Nanocatalysts. Nano Res. 2009, 2, 30–46. [Google Scholar] [CrossRef]
- Husein, M.M.; Nassar, N.N. Nanoparticle Preparation Using the Single Microemulsions Scheme. Curr. Nanosci. 2008, 4, 370–380. [Google Scholar] [CrossRef]
- Bock, C.; Paquet, C.; Couillard, M.; Botton, G.; MacDougall, B. Size-selected synthesis of PtRu nano-catalysts: Reaction and size control mechanism. J. Am. Chem. Soc. 2004, 126, 8028–8037. [Google Scholar] [CrossRef] [PubMed]
- Alivisatos, A. Semiconductor clusters, nanocrystals, and quantum dots. Science 1996, 271, 933–937. [Google Scholar] [CrossRef]
- Shen, S.C.; Hidajat, K.; Yu, L.E.; Kawi, S. Simple Hydrothermal Synthesis of Nanostructured and Nanorod Zn-Al Complex Oxides as Novel Nanocatalysts. Adv. Mater. 2004, 16, 541–545. [Google Scholar] [CrossRef]
- The Royal Society; The Royal Academy of Engineering. Nanoscience and Nanotechnologies: Opportunities and Uncertainties; The Royal Society: London, UK, 2004; ISBN 978-0-85403-604-0. [Google Scholar]
- Holsapple, M.; Farland, W.; Landry, T.; Monteiro-Riviere, N.; Carter, J.; Walker, N.; Thomas, K. Research strategies for safety evaluation of nanomaterials, part II: Toxicological and safety evaluation of nanomaterials, current challenges and data needs. Toxicol. Sci. 2005, 88, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Zhang, Y.; Chen, G.; Gai, Z. Application of Nanoparticles in Enhanced Oil Recovery: A Critical Review of Recent Progress. Energies 2017, 10, 345. [Google Scholar] [CrossRef]
- Cheraghian, G.; Hendraningrat, L. A review on applications of nanotechnology in the enhanced oil recovery part B: Effects of nanoparticles on flooding. Int. Nano Lett. 2016, 6, 1–10. [Google Scholar] [CrossRef]
- Ahmadi, M.A.; Shadizadeh, S.R. Nanofluid in Hydrophilic State for EOR Implication Through Carbonate Reservoir. J. Dispers. Sci. Technol. 2014, 35, 1537–1542. [Google Scholar] [CrossRef]
- Civan, F. Reservoir Formation Damage, 3rd ed.; Elsevier Science: Boston, MA, USA, 2016; ISBN 978-0-75067-738-7. [Google Scholar]
- Gao, C. Factors affecting particle retention in porous media. Emirates J. Eng. Res. 2007, 12, 1–7. [Google Scholar]
- Ju, B.; Fan, T. Experimental study and mathematical model of nanoparticle transport in porous media. Powder Technol. 2009, 192, 195–202. [Google Scholar] [CrossRef]
- Markus, A.A.; Parsons, J.R.; Roex, E.W.M.; de Voogt, P.; Laane, R.W.P.M. Modeling aggregation and sedimentation of nanoparticles in the aquatic environment. Sci. Total Environ. 2015, 506, 323–329. [Google Scholar] [CrossRef]
- Hu, Z.; Haruna, M.; Gao, H.; Nourafkan, E.; Wen, D. Rheological Properties of Partially Hydrolyzed Polyacrylamide Seeded by Nanoparticles. Ind. Eng. Chem. Res. 2017, 56, 3456–3463. [Google Scholar] [CrossRef]
- Maurya, N.K.; Mandal, A. Studies on behavior of suspension of silica nanoparticle in aqueous polyacrylamide solution for application in enhanced oil recovery. Pet. Sci. Technol. 2016, 34, 429–436. [Google Scholar] [CrossRef]
- Ye, Z.; Qin, X.; Lai, N.; Peng, Q.; Li, X.; Li, C. Synthesis and Performance of an Acrylamide Copolymer Containing Nano-SiO2 as Enhanced Oil Recovery Chemical. J. Chem. 2013, 2013, 1–10. [Google Scholar] [CrossRef]
- Khandavalli, S.; Rothstein, J.P. Extensional rheology of shear-thickening fumed silica nanoparticles dispersed in an aqueous polyethylene oxide solution. J. Rheol. 2014, 58, 411–431. [Google Scholar] [CrossRef]
- Balasubramanian, G.; Sen, S.; Puri, I.K. Shear viscosity enhancement in water/nanoparticle suspensions. Phys. Lett. A 2012, 376, 860–863. [Google Scholar] [CrossRef]
- Meyer, J.P.; Adio, S.A.; Sharifpur, M.; Nwosu, P.N. The Viscosity of Nanofluids: A Review of the Theoretical, Empirical, and Numerical Models. Heat Transf. Eng. 2016, 37, 387–421. [Google Scholar] [CrossRef]
- Mishra, P.; Mukherjee, S.; Nayak, S.; Panda, A. A brief review on viscosity of nanofluids. Int. Nano Lett. 2014, 4, 109–120. [Google Scholar] [CrossRef]
- Rudyak, V.Y. Viscosity of nanofluids. Why it is not described by the classical theories. Adv. Nanopart. 2013, 2, 266–279. [Google Scholar] [CrossRef]
- Yang-Chuan, K.; Guang-Yao, W.; Yi, W. Preparation, morphology and properties of nanocomposites of polyacrylamide copolymers with monodisperse silica. Eur. Polym. J. 2008, 44, 2448–2457. [Google Scholar] [CrossRef]
- Cheraghian, G.; Khalilinezhad, S.S. Effect of Nanoclay on Heavy Oil Recovery During Polymer Flooding. Pet. Sci. Technol. 2015, 33, 999–1007. [Google Scholar] [CrossRef]
- Cheraghian, G. An Experimental Study of Surfactant Polymer for Enhanced Heavy Oil Recovery Using a Glass Micromodel by Adding Nanoclay. Pet. Sci. Technol. 2015, 33, 1410–1417. [Google Scholar] [CrossRef]
- Moradi, B.; Pourafshary, P.; Farahani, F.J.; Mohammadi, M.; Emadi, M.A. Application of SiO2 Nano Particles to Improve the Performance of Water Alternating Gas EOR Process. In SPE Oil & Gas India Conference and Exhibition; Society of Petroleum Engineers: Mumbai, India, 2015. [Google Scholar]
- Matroushi, M.A.; Pourafshary, P.; Wahaibi, Y.A. Possibility of Nanofluid/Gas Alternating Injection as an EOR Method in an Oil Field. In Abu Dhabi International Petroleum Exhibition and Conference; Society of Petroleum Engineers: Abu Dhabi, UAE, 2015. [Google Scholar]
- Ponmani, S.; William, J.K.M.; Samuel, R.; Nagarajan, R.; Sangwai, J.S. Formation and characterization of thermal and electrical properties of CuO and ZnO nanofluids in xanthan gum. Colloids Surf. A Physicochem. Eng. Asp. 2014, 443, 37–43. [Google Scholar] [CrossRef]
- Fan, H.; Striolo, A. Nanoparticle effects on the water-oil interfacial tension. Phys. Rev. E 2012, 86, 051610. [Google Scholar] [CrossRef]
- Frijters, S.; Gunther, F.; Harting, J. Effects of nanoparticles and surfactant on droplets in shear flow. Soft Matter 2012, 8, 6542–6556. [Google Scholar] [CrossRef]
- Ragab, A.M. Investigating the Potential of Nanomaterials for Enhanced Oil Recovery: State of Art. J. Sci. Technol. 2014, 6, 25–40. [Google Scholar]
- Hendraningrat, L.; Torsaeter, O. Metal oxide-based nanoparticles: revealing their potential to enhance oil recovery in different wettability systems. Appl. Nanosci. 2015, 5, 181–199. [Google Scholar] [CrossRef]
- Baez, J.; Ruiz, M.P.; Faria, J.; Harwell, J.H.; Shiau, B.; Resasco, D.E. Stabilization of Interfacially-Active-Nanohybrids/Polymer Suspensions and Transport through Porous Media. In SPE Improved Oil Recovery Symposium; Society of Petroleum Engineers: Tulsa, OK, USA, 2012. [Google Scholar]
- Zheng, X.F.; Lian, Q. Synthesis and Evaluation of CoFe2O4-Chitosan Nanoparticles in Enhanced Oil Recovery. J. Dispers. Sci. Technol. 2015, 36, 245–251. [Google Scholar] [CrossRef]
- Sefiane, K.; Skilling, J.; MacGillivray, J. Contact line motion and dynamic wetting of nanofluid solutions. Adv. Colloid Interface Sci. 2008, 138, 101–120. [Google Scholar] [CrossRef] [PubMed]
- Moghaddam, R.N.; Bahramian, A.; Fakhroueian, Z.; Karimi, A.; Arya, S. Comparative Study of Using Nanoparticles for Enhanced Oil Recovery: Wettability Alteration of Carbonate Rocks. Energy Fuels 2015, 29, 2111–2119. [Google Scholar] [CrossRef]
- Wasan, D.; Nikolov, A. Spreading of nanofluids on solids. Nature 2003, 423, 156–159. [Google Scholar] [CrossRef] [PubMed]
- Wasan, D.; Nikolov, A.; Kondiparty, K. The wetting and spreading of nanofluids on solids: Role of the structural disjoining pressure. Curr. Opin. Colloid Interface Sci. 2011, 16, 344–349. [Google Scholar] [CrossRef]
- Kondiparty, K.; Nikolov, A.; Wu, S.; Wasan, D. Wetting and Spreading of Nanofluids on Solid Surfaces Driven by the Structural Disjoining Pressure: Statics Analysis and Experiments. Langmuir 2011, 27, 3324–3335. [Google Scholar] [CrossRef]
- Kondiparty, K.; Nikolov, A.D.; Wasan, D.; Liu, K.L. Dynamic Spreading of Nanofluids on Solids. Part I: Experimental. Langmuir 2012, 28, 14618–14623. [Google Scholar] [CrossRef]
- Nikolov, A.; Kondiparty, K.; Wasan, D. Nanoparticle Self-Structuring in a Nanofluid Film Spreading on a Solid Surface. Langmuir 2010, 26, 7665–7670. [Google Scholar] [CrossRef]
- Wang, F.C.; Wu, H.A. Enhanced oil droplet detachment from solid surfaces in charged nanoparticle suspensions. Soft Matter 2013, 9, 7974–7980. [Google Scholar] [CrossRef]
- Liu, K.L.; Kondiparty, K.; Nikolov, A.D.; Wasan, D. Dynamic Spreading of Nanofluids on Solids Part II: Modeling. Langmuir 2012, 28, 16274–16284. [Google Scholar] [CrossRef] [PubMed]
- Al-Anssari, S.; Barifcani, A.; Wang, S.; Maxim, L.; Iglauer, S. Wettability alteration of oil-wet carbonate by silica nanofluid. J. Colloid Interface Sci. 2016, 461, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Safari, M.; Jamialahmadi, M. Effect of polysilicon nanoparticles on enhanced oil recovery in Iranian oil reservoir. Int. J. Nano Dimens. 2013, 3, 199–205. [Google Scholar]
- Bayat, A.E.; Junin, R.; Samsuri, A.; Piroozian, A.; Hokmabadi, M. Impact of Metal Oxide Nanoparticles on Enhanced Oil Recovery from Limestone Media at Several Temperatures. Energy Fuels 2014, 28, 6255–6266. [Google Scholar] [CrossRef]
- McClements, D.J. Edible nanoemulsions: Fabrication, properties, and functional performance. Soft Matter 2011, 7, 2297–2316. [Google Scholar] [CrossRef]
- Friberg, S.; Larsson, K.; Sjoblom, J. Food Emulsions; CRC Press: New York, NY, USA, 2003; ISBN 978-0-82474-696-4. [Google Scholar]
- Tadros, T.; Izquierdo, R.; Esquena, J.; Solans, C. Formation and stability of nano-emulsions. Adv. Colloid Interface Sci. 2004, 108, 303–318. [Google Scholar] [CrossRef] [PubMed]
- Wooster, T.J.; Golding, M.; Sanguansri, P. Impact of Oil Type on Nanoemulsion Formation and Ostwald Ripening Stability. Langmuir 2008, 24, 12758–12765. [Google Scholar] [CrossRef]
- Mason, T.G.; Wilking, J.N.; Meleson, K.; Chang, C.B.; Graves, S.M. Nanoemulsions: formation, structure, and physical properties. J. Phys.-Condens. Matter 2006, 18, R635–R666. [Google Scholar] [CrossRef]
- Tadros, T.F. Applied Surfactants: Principles and Applications; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006; ISBN 978-3-52730-629-9. [Google Scholar]
- Shen, M.; Resasco, D.E. Emulsions Stabilized by Carbon Nanotube-Silica Nanohybrids. Langmuir 2009, 25, 10843–10851. [Google Scholar] [CrossRef]
- Binks, B.; Lumsdon, S. Stability of oil-in-water emulsions stabilised by silica particles. Phys. Chem. Chem. Phys. 1999, 1, 3007–3016. [Google Scholar] [CrossRef]
- Stocco, A.; Rio, E.; Binks, B.P.; Langevin, D. Aqueous foams stabilized solely by particles. Soft Matter 2011, 7, 1260–1267. [Google Scholar] [CrossRef]
- Dinsmore, A.; Hsu, M.; Nikolaides, M.; Marquez, M.; Bausch, A.; Weitz, D. Colloidosomes: Selectively permeable capsules composed of colloidal particles. Science 2002, 298, 1006–1009. [Google Scholar] [CrossRef] [PubMed]
- Nassar, N.N.; Husein, M.M.; Pereira-Almao, P. In-situ prepared nanoparticles in support of oilsands industry meeting future environmental challenges. Explor. Prod. Oil Gas Rev. 2011, 9, 46–48. [Google Scholar]
- Nguyen, P.; Fadaei, H.; Sinton, D. Pore-Scale Assessment of Nanoparticle-Stabilized CO2 Foam for Enhanced Oil Recovery. Energy Fuels 2014, 28, 6221–6227. [Google Scholar] [CrossRef]
- Zargartalebi, M.; Kharrat, R.; Barati, N. Enhancement of surfactant flooding performance by the use of silica nanoparticles. Fuel 2015, 143, 21–27. [Google Scholar] [CrossRef]
- Forgiarini, A.; Esquena, J.; Gonzalez, C.; Solans, C. Formation of nano-emulsions by low-energy emulsification methods at constant temperature. Langmuir 2001, 17, 2076–2083. [Google Scholar] [CrossRef]
- Villamizar, L.C.; Lohateeraparp, P.; Harwell, J.H.; Resasco, D.E.; Shiau, B.J. Dispersion Stability and Transport of Nanohybrids through Porous Media. Transp. Porous Media 2013, 96, 63–81. [Google Scholar] [CrossRef]
- Bornaee, A.H.; Manteghian, M.; Rashidi, A.; Alaei, M.; Ershadi, M. Oil-in-water Pickering emulsions stabilized with functionalized multi-walled carbon nanotube/silica nanohybrids in the presence of high concentrations of cations in water. J. Ind. Eng. Chem. 2014, 20, 1720–1726. [Google Scholar] [CrossRef]
- Sun, Q.; Li, Z.; Li, S.; Jiang, L.; Wang, J.; Wang, P. Utilization of Surfactant-Stabilized Foam for Enhanced Oil Recovery by Adding Nanoparticles. Energy Fuels 2014, 28, 2384–2394. [Google Scholar] [CrossRef]
- Avila, J.N.L.D.; Grecco Cavalcanti De Araujo, L.L.; Drexler, S.; de Almeida Rodrigues, J.; Nascimento, R.S.V. Polystyrene nanoparticles as surfactant carriers for enhanced oil recovery. J. Appl. Polym. Sci. 2016, 133, 43789. [Google Scholar] [CrossRef]
- Zaid, H.M.; Yahya, N.; Latiff, N.R.A. The Effect of Nanoparticles Crystallite Size on the Recovery Efficiency in Dielectric Nanofluid Flooding. J. Nano Res. 2013, 21, 103–108. [Google Scholar] [CrossRef]
- Guzman, E.; Liggieri, L.; Santini, E.; Ferrari, M.; Ravera, F. Influence of silica nanoparticles on dilational rheology of DPPC-palmitic acid Langmuir monolayers. Soft Matter 2012, 8, 3938–3948. [Google Scholar] [CrossRef]
- Worthen, A.; Taghavy, A.; Aroonsri, A.; Kim, I.; Johnston, K.; Huh, C.; Bryant, S.; DiCarlo, D. Multi-Scale Evaluation of Nanoparticle-Stabilized CO2-in-Water Foams: From the Benchtop to the Field. In SPE Annual Technical Conference and Exhibition; Society of Petroleum Engineers: Houston, TX, USA, 2015. [Google Scholar]
- Huang, Q. Nanotechnology in the Food, Beverage and Nutraceutical Industries, Elsevier Science ed.; Woodhead Publishing: Cambridge, UK, 2012; ISBN 978-1-84569-739-6. [Google Scholar]
- McClements, D.J.; Rao, J. Food-grade nanoemulsions: formulation, fabrication, properties, performance, biological fate, and potential toxicity. Crit. Rev. Food Sci. Nutr. 2011, 51, 285–330. [Google Scholar] [CrossRef] [PubMed]
- Kabalnov, A.; Shchukin, E. Ostwald Ripening Theory—Applications to Fluorocarbon Emulsion Stability. Adv. Colloid Interface Sci. 1992, 38, 69–97. [Google Scholar] [CrossRef]
- Lifshitz, I.M.; Slyozov, V.V. The kinetics of precipitation from supersaturated solid solutions. J. Phys. Chem. Solids 1961, 19, 35–50. [Google Scholar] [CrossRef]
- Walstra, P. Principles of Emulsion Formation. Chem. Eng. Sci. 1993, 48, 333–349. [Google Scholar] [CrossRef]
- Maali, A.; Mosavian, M.T.H. Preparation and Application of Nanoemulsions in the Last Decade (2000–2010). J. Dispers. Sci. Technol. 2013, 34, 92–105. [Google Scholar] [CrossRef]
- Meleson, K.; Graves, S.; Mason, T. Formation of concentrated nanoemulsions by extreme shear. Soft Mater. 2004, 2, 109–123. [Google Scholar] [CrossRef]
- Squires, T.; Quake, S. Microfluidics: Fluid physics at the nanoliter scale. Rev. Mod. Phys. 2005, 77, 977–1026. [Google Scholar] [CrossRef]
- Sutradhar, K.B.; Lutful, A.M. Nanoemulsions: increasing possibilities in drug delivery. Eur. J. Nanomed. 2013, 5, 97–110. [Google Scholar] [CrossRef]
- Shah, P.; Bhalodia, D.; Shelat, P. Nanoemulsion: a pharmaceutical review. Syst. Rev. Pharm. 2010, 1, 24–32. [Google Scholar] [CrossRef]
- Strydom, S.J.; Rose, W.E.; Otto, D.P.; Liebenberg, W.; de Villiers, M.M. Poly(amidoamine) dendrimer-mediated synthesis and stabilization of silver sulfonamide nanoparticles with increased antibacterial activity. Nanomed.-Nanotechnol. Biol. Med. 2013, 9, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Eral, H.; Hatton, T.; Doyle, P. Nanoemulsions: Formation, properties and applications. Soft Matter 2016, 12, 2826. [Google Scholar] [CrossRef]
- Panagiotou, T.; Fisher, R. Improving product quality with entrapped stable emulsions: From theory to industrial application. Challenges 2012, 3, 84–113. [Google Scholar] [CrossRef]
- Sole, I.; Pey, C.M.; Maestro, A.; Gonzalez, C.; Porras, M.; Solans, C.; Gutierrez, J.M. Nano-emulsions prepared by the phase inversion composition method: Preparation variables and scale up. J. Colloid Interface Sci. 2010, 344, 417–423. [Google Scholar] [CrossRef]
- Wang, L.; Mutch, K.J.; Eastoe, J.; Heenan, R.K.; Dong, J. Nanoemulsions prepared by a two-step low-energy process. Langmuir 2008, 24, 6092–6099. [Google Scholar] [CrossRef]
- Izquierdo, P.; Feng, J.; Esquena, J.; Tadros, T.; Dederen, J.; Garcia, M.; Azemar, N.; Solans, C. The influence of surfactant mixing ratio on nano-emulsion formation by the pit method. J. Colloid Interface Sci. 2005, 285, 388–394. [Google Scholar] [CrossRef]
- Chime, S.; Kenechukwu, F.; Attama, A. Nanoemulsions—Advances in Formulation, Characterization and Applications in Drug Delivery. In Application of Nanotechnology in Drug Delivery; InTech: Rijeka, Croatia, 2014; Chapter 3; pp. 77–126. [Google Scholar]
- Miller, C. Spontaneous Emulsification Produced by Diffusion—A Review. Colloids Surf. 1988, 29, 89–102. [Google Scholar] [CrossRef]
- Bell, A. The impact of nanoscience on heterogeneous catalysis. Science 2003, 299, 1688–1691. [Google Scholar] [CrossRef]
- Gharibi, M.; Zangeneh, F.T.; Yaripour, F.; Sahebdelfar, S. Nanocatalysts for conversion of natural gas to liquid fuels and petrochemical feedstocks. Appl. Catal. A-Gen. 2012, 443, 8–26. [Google Scholar] [CrossRef]
- Maity, S.K.; Ancheyta, J.; Marroquin, G. Catalytic Aquathermolysis Used for Viscosity Reduction of Heavy Crude Oils: A Review. Energy Fuels 2010, 24, 2809–2816. [Google Scholar] [CrossRef]
- Nassar, N.N.; Hassan, A.; Pereira-Almao, P. Application of Nanotechnology for Heavy Oil Upgrading: Catalytic Steam Gasification/Cracking of Asphaltenes. Energy Fuels 2011, 25, 1566–1570. [Google Scholar] [CrossRef]
- Billon, A.; Bigeard, P. Hydrocracking. Pet. Refin. 2001, 3, 333–364. [Google Scholar]
- Hascakir, B.; Babadagli, T.; Akin, S. Experimental and Numerical Simulation of Oil Recovery from Oil Shales by Electrical Heating. Energy Fuels 2008, 22, 3976–3985. [Google Scholar] [CrossRef]
- Shokrlu, Y.H.; Babadagli, T. Viscosity reduction of heavy oil/bitumen using micro- and nano-metal particles during aqueous and non-aqueous thermal applications. J. Pet. Sci. Eng. 2014, 119, 210–220. [Google Scholar] [CrossRef]
- Nassar, N.N.; Husein, M.M.; Pereira-Almao, P. Ultradispersed particles in heavy oil: Part II, sorption of H2S(g). Fuel Process. Technol. 2010, 91, 169–174. [Google Scholar] [CrossRef]
- Nassar, N.N.; Husein, M.M. Ultradispersed particles in heavy oil: Part I, preparation and stabilization of iron oxide/hydroxide. Fuel Process. Technol. 2010, 91, 164–168. [Google Scholar] [CrossRef]
- Clark, P.D.; Clarke, R.A.; Hyne, J.B.; Lesage, K.L. Studies on the effect of metal species on oil sands undergoing steam treatments. Aostra J. Res. 1990, 6, 53–64. [Google Scholar]
- Fan, H.; Zhang, Y.; Lin, Y. The catalytic effects of minerals on aquathermolysis of heavy oils. Fuel 2004, 83, 2035–2039. [Google Scholar] [CrossRef]
- Shokrlu, Y.H.; Babadagli, T. In-Situ Upgrading of Heavy Oil/Bitumen During Steam Injection by Use of Metal Nanoparticles: A Study on In-Situ Catalysis and Catalyst Transportation. SPE Reserv. Eval. Eng. 2013, 16, 333–344. [Google Scholar] [CrossRef]
- Olvera, J.N.R.; Gutierrez, G.J.; Serrano, J.A.R.; Ovando, A.M.; Febles, V.G.; Arceo, L.D.B. Use of unsupported, mechanically alloyed NiWMoC nanocatalyst to reduce the viscosity of aquathermolysis reaction of heavy oil. Catal. Commun. 2014, 43, 131–135. [Google Scholar] [CrossRef]
- Greff, J.; Babadagli, T. Use of nano-metal particles as catalyst under electromagnetic heating for in-situ heavy oil recovery. J. Pet. Sci. Eng. 2013, 112, 258–265. [Google Scholar] [CrossRef]
- Shokrlu, Y.H.; Maham, Y.; Tan, X.; Babadagli, T.; Gray, M. Enhancement of the efficiency of in situ combustion technique for heavy-oil recovery by application of nickel ions. Fuel 2013, 105, 397–407. [Google Scholar] [CrossRef]
- Drexler, S.; Faria, J.; Ruiz, M.P.; Harwell, J.H.; Resasco, D.E. Amphiphilic Nanohybrid Catalysts for Reactions at the Water/Oil Interface in Subsurface Reservoirs. Energy Fuels 2012, 26, 2231–2241. [Google Scholar] [CrossRef]
- Wang, H.; Wu, Y.; He, L.; Liu, Z. Supporting Tungsten Oxide on Zirconia by Hydrothermal and Impregnation Methods and Its Use as a Catalyst to Reduce the Viscosity of Heavy Crude Oil. Energy Fuels 2012, 26, 6518–6527. [Google Scholar] [CrossRef]
- Li, K.; Hou, B.; Wang, L.; Cui, Y. Application of Carbon Nanocatalysts in Upgrading Heavy Crude Oil Assisted with Microwave Heating. Nano Lett. 2014, 14, 3002–3008. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, M.; Schaffie, M.; Ranjbar, M. Thermocatalytic in situ combustion: Influence of nanoparticles on crude oil pyrolysis and oxidation. Fuel 2013, 113, 516–521. [Google Scholar] [CrossRef]
- Yang, Z.; Liu, X.; Li, X.; Zhao, M.; Zhang, Z.; Su, C. Preparation of silica supported nanoscale zero valence iron and its feasibility in viscosity reduction of heavy oil. Micro Nano Lett. 2014, 9, 355–358. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, Y.; Lu, J.; Wu, C. The viscosity reduction of nano-keggin-K3PMo12O40 in catalytic aquathermolysis of heavy oil. Fuel 2009, 88, 1426–1434. [Google Scholar] [CrossRef]
- Pourabdollah, K.; Moghaddam, A.Z.; Kharrat, R.; Mokhtari, B. An Experimental Feasibility Study of In-situ Nano-particles in Enhanced Oil Recovery and Heavy Oil Production. Energy Sources Part A-Recovery Util. Environ. Eff. 2013, 35, 2198–2208. [Google Scholar] [CrossRef]
- Clark, P.; Hyne, J. Steam-oil chemical reactions: Mechanisms for the aquathermolysis of heavy oils. AOSTRA J. Res. 1984, 1, 15–20. [Google Scholar]
- Hyne, J.B.A. A synopsis work on the chemical reaction between water (steam) and heavy oil sands during simulated stimulation. Fuel 1986, 62, 959–962. [Google Scholar]
- Hyne, J.; Clark, P.; Clarke, R.; Koo, J.; Greidanus, J.; Tyrer, J.; Verona, D. Aquathermolysis of Heavy Oils. Rev. Tec. Intevep 1982, 2, 87–94. [Google Scholar]
- Sanchez, S.; Rodriguez, M.; Ancheyta, J. Kinetic model for moderate hydrocracking of heavy oils. Ind. Eng. Chem. Res. 2005, 44, 9409–9413. [Google Scholar] [CrossRef]
- Ancheyta, J.; Sanchez, S.; Rodriguez, M. Kinetic modeling of hydrocracking of heavy oil fractions: A review. Catal. Today 2005, 109, 76–92. [Google Scholar] [CrossRef]
- Belgrave, J.; Moore, R.; Ursenbach, M. Comprehensive kinetic models for the aquathermolysis of heavy oils. J. Can. Pet. Technol. 1997, 36, 38–44. [Google Scholar] [CrossRef]
- Rezaei, M.; Schaffie, M.; Ranjbar, M. Kinetic Study of Catalytic In-situ Combustion Processes in the Presence of Nanoparticles. Energy Sources Part A-Recovery Util. Environ. Eff. 2014, 36, 605–612. [Google Scholar] [CrossRef]
- Nassar, N.N.; Hassan, A.; Vitale, G. Comparing kinetics and mechanism of adsorption and thermo-oxidative decomposition of Athabasca asphaltenes onto TiO2, ZrO2, and CeO2 nanoparticles. Appl. Catal. A-Gen. 2014, 484, 161–171. [Google Scholar] [CrossRef]
- Clark, P.D.; Hyne, J.B.; Tyrer, J.D. Chemistry of organosulphur compound types occurring in heavy oil sands: 1. High temperature hydrolysis and thermolysis of tetrahydrothiophene in relation to steam stimulation processes. Fuel 1983, 62, 959–962. [Google Scholar] [CrossRef]
- Clark, P.D.; Hyne, J.B.; Tyrer, J.D. Some chemistry of organosulphur compound types occurring in heavy oil sands: 2. Influence of pH on the high temperature hydrolysis of tetrahydrothiophene and thiophene. Fuel 1984, 63, 125–128. [Google Scholar] [CrossRef]
- Clark, P.D.; Hyne, J.B. Chemistry of organosulphur compound types occurring in heavy oil sands: 3. Reaction of thiophene and tetrahydrothiophene with vanadyl and nickel salts. Fuel 1984, 63, 1649–1654. [Google Scholar] [CrossRef]
- Carbognani, L. Upgrading of a Visbroken Vacuum Residue by Adsorption and Catalytic Steam Gasification of the Adsorbed Components. Master’s Thesis, University of Calgary, Calgary, AB, Canada, 2014. [Google Scholar]
- Siewe, C.; Ng, F. Hydrodesulfurization of cold lake diesel fraction using dispersed catalysts: Influence of hydroprocessing medium and sources of H-2. Energy Fuels 1998, 12, 598–606. [Google Scholar] [CrossRef]
- Kennepohl, D.; Sanford, E. Conversion of Athabasca bitumen with dispersed and supported Mo-based catalysts as a function of dispersed catalyst concentration. Energy Fuels 1996, 10, 229–234. [Google Scholar] [CrossRef]
- Ovalles, C.; Vallejos, C.; Vasquez, T.; Rojas, I.; Ehrman, U.; Benitez, J.; Martinez, R. Downhole upgrading of extra-heavy crude oil using hydrogen donors and methane under steam injection conditions. Pet. Sci. Technol. 2003, 21, 255–274. [Google Scholar] [CrossRef]
- Zhong, L.G.; Liu, Y.J.; Fan, H.F.; Jiang, S.J. Liaohe Extra-Heavy Crude Oil Underground Aquathermolytic Treatments Using Catalyst and Hydrogen Donors under Steam Injection Conditions. In SPE International Improved Oil Recovery Conference in Asia; Society of Petroleum Engineers: Kuala Lumpur, Malaysia, 2003. [Google Scholar]
- Shokrlu, Y.H. Enhancement of Heavy Oil/Bitumen Thermal Recovery Using Nano Metal Particles. Ph.D. Thesis, University of Alberta, Edmonton, AB, Canada, 2013. [Google Scholar]
- Xuan, Y.; Li, Q. Heat transfer enhancement of nanofluids. Int. J. Heat Fluid Flow 2000, 21, 58–64. [Google Scholar] [CrossRef]
- Choi, S.U.S.; Zhang, Z.G.; Yu, W.; Lockwood, F.E.; Grulke, E.A. Anomalous thermal conductivity enhancement in nanotube suspensions. Appl. Phys. Lett. 2001, 79, 2252–2254. [Google Scholar] [CrossRef]
- Kleinstreuer, C.; Feng, Y. Experimental and theoretical studies of nanofluid thermal conductivity enhancement: A review. Nanoscale Res. Lett. 2011, 6, 229. [Google Scholar] [CrossRef]
- Franco, C.A.; Nassar, N.N.; Ruiz, M.A.; Pereira-Almao, P.; Cortes, F.B. Nanoparticles for Inhibition of Asphaltenes Damage: Adsorption Study and Displacement Test on Porous Media. Energy Fuels 2013, 27, 2899–2907. [Google Scholar] [CrossRef]
- Adams, J.J. Asphaltene Adsorption, a Literature Review. Energy Fuels 2014, 28, 2831–2856. [Google Scholar] [CrossRef]
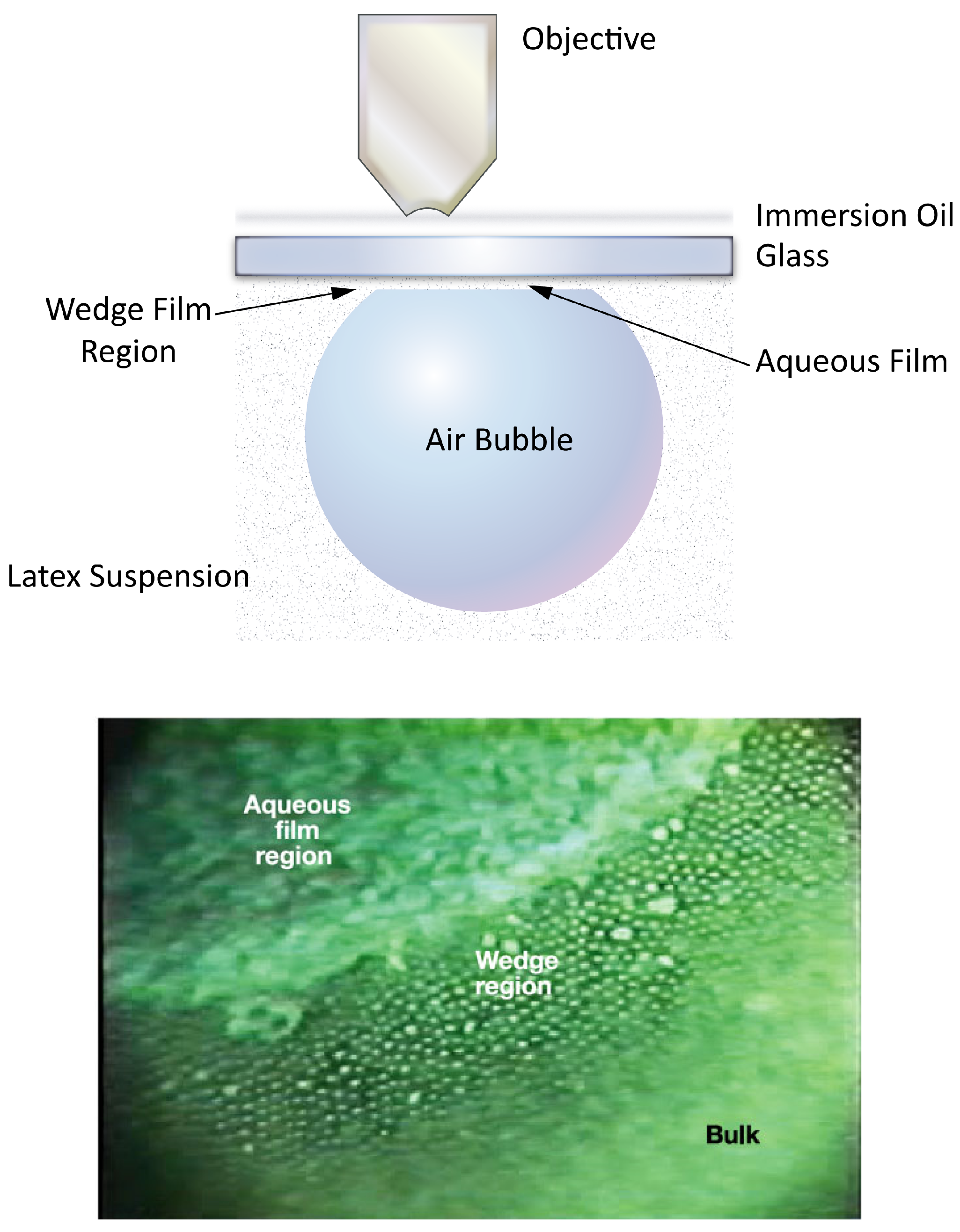
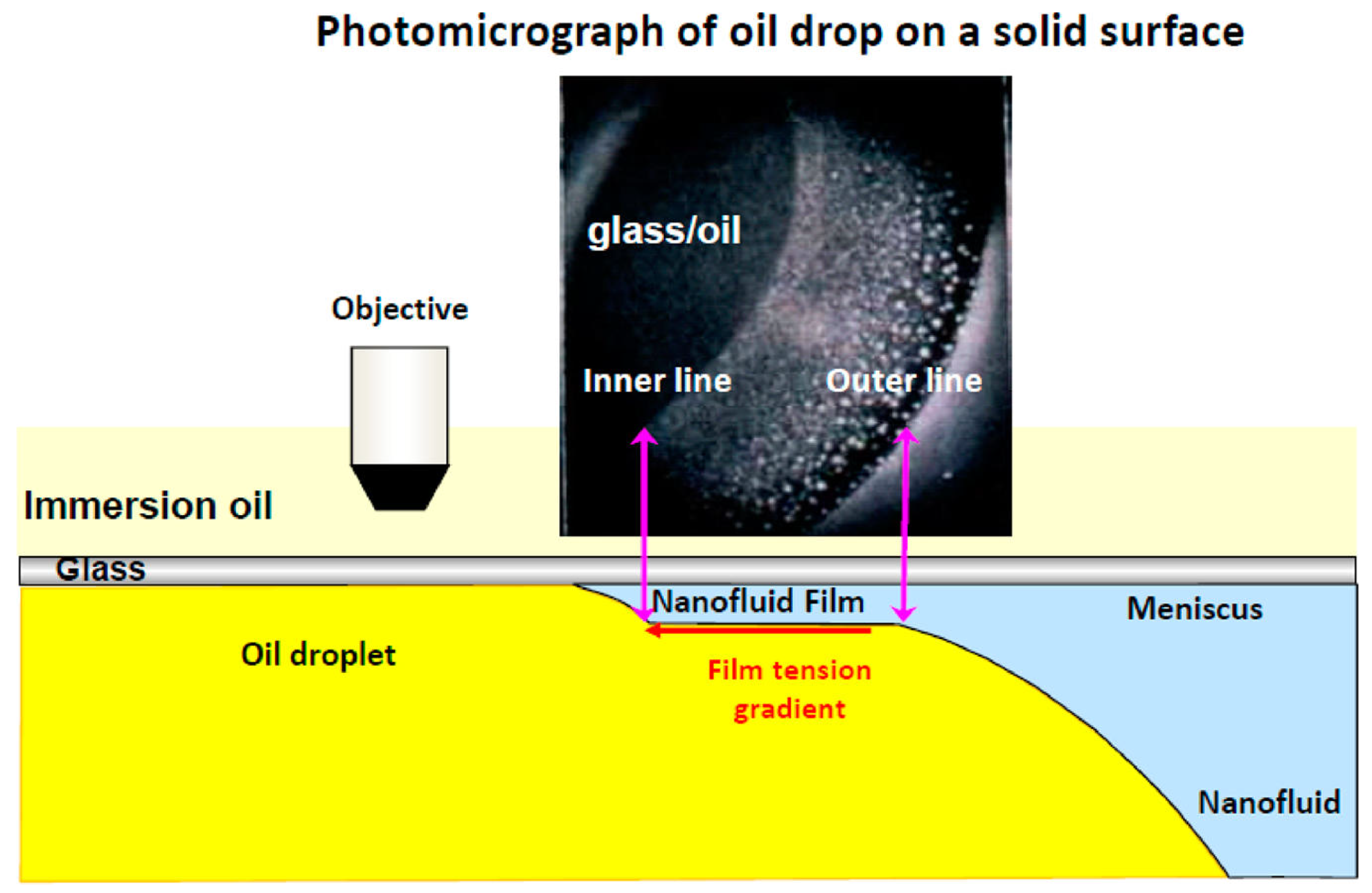
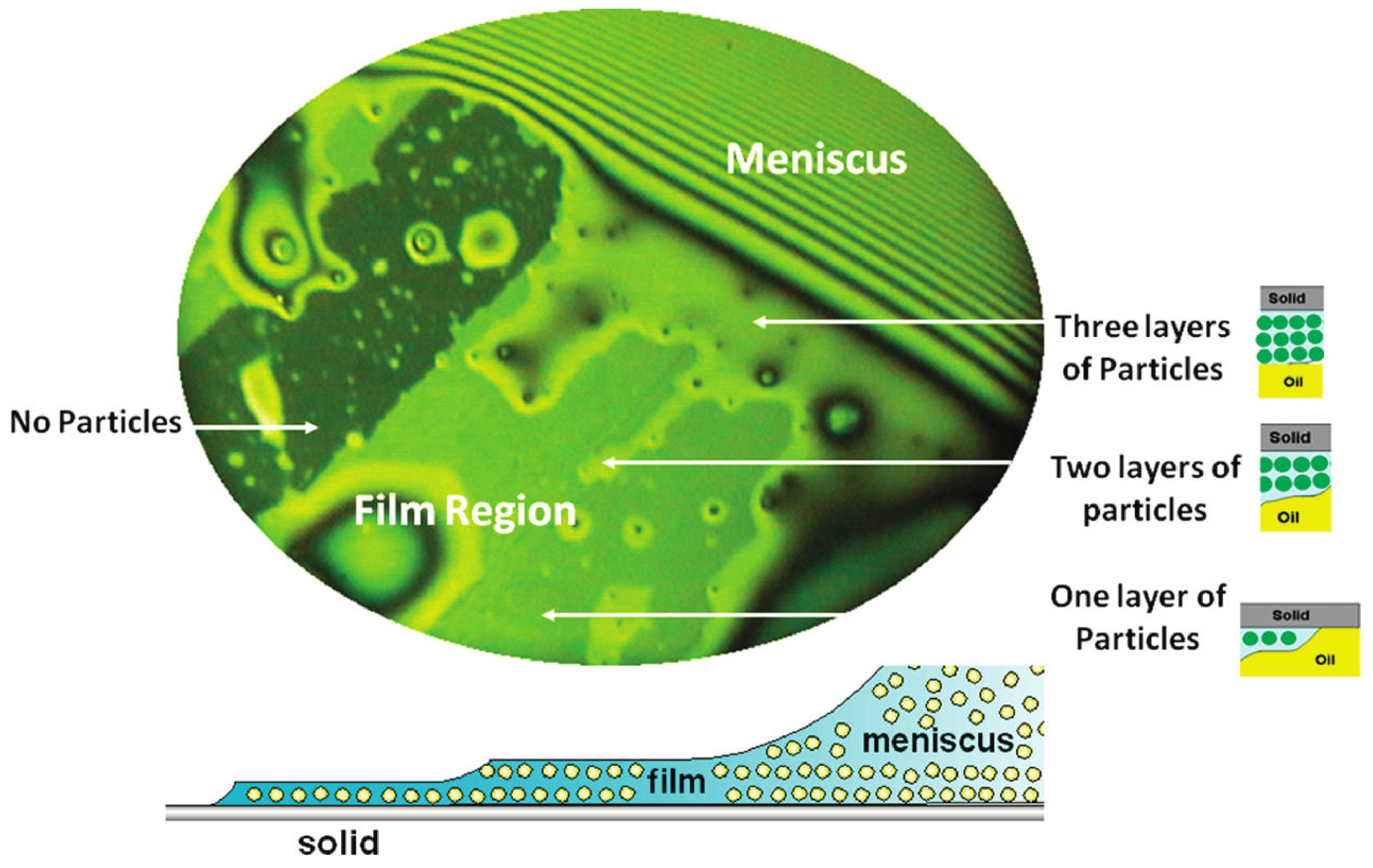
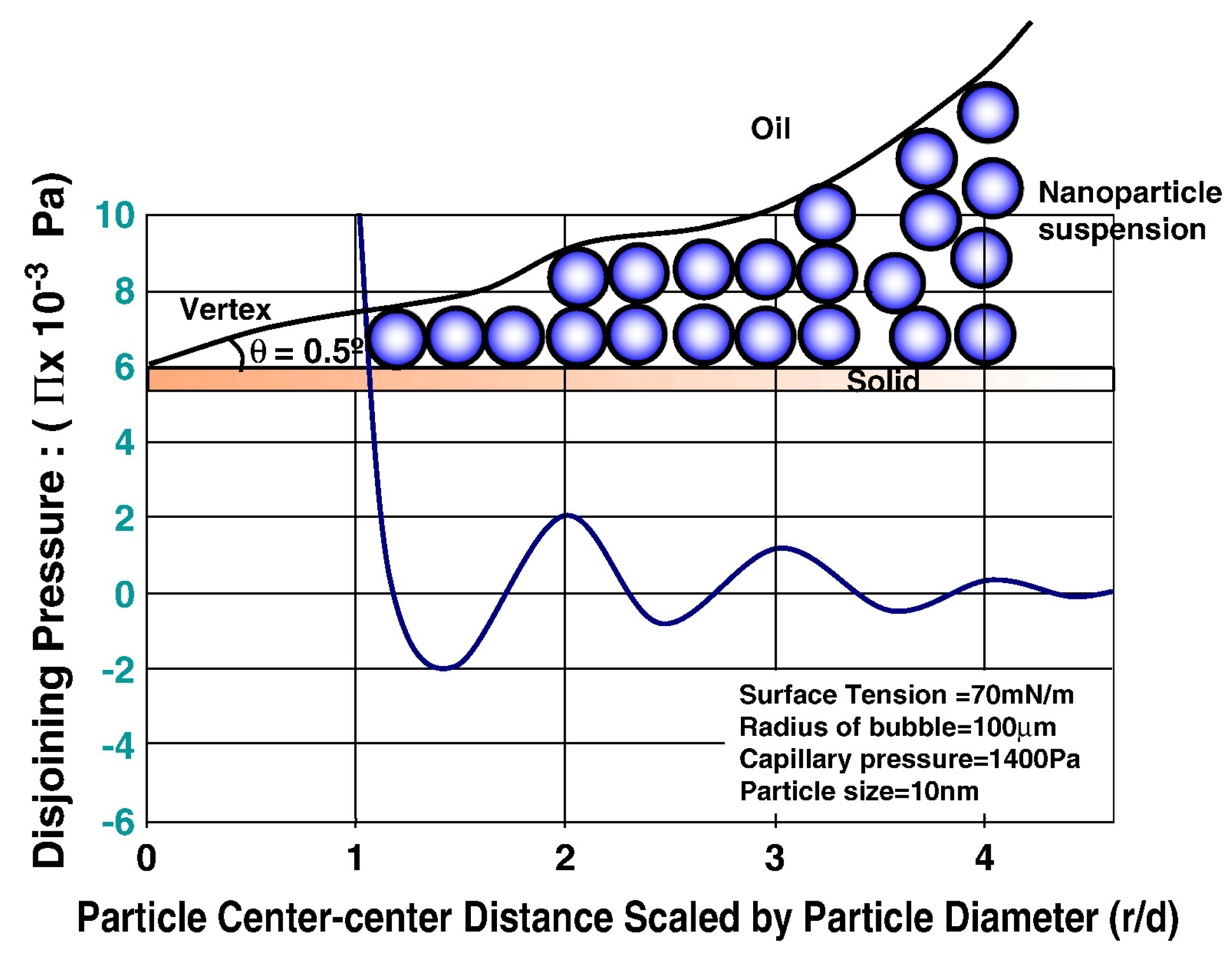





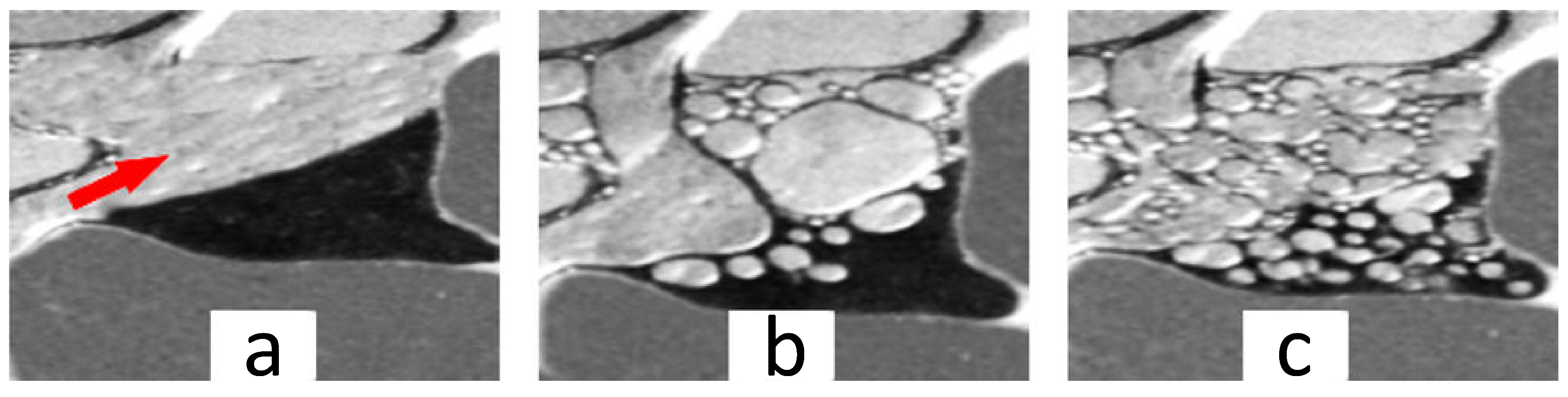
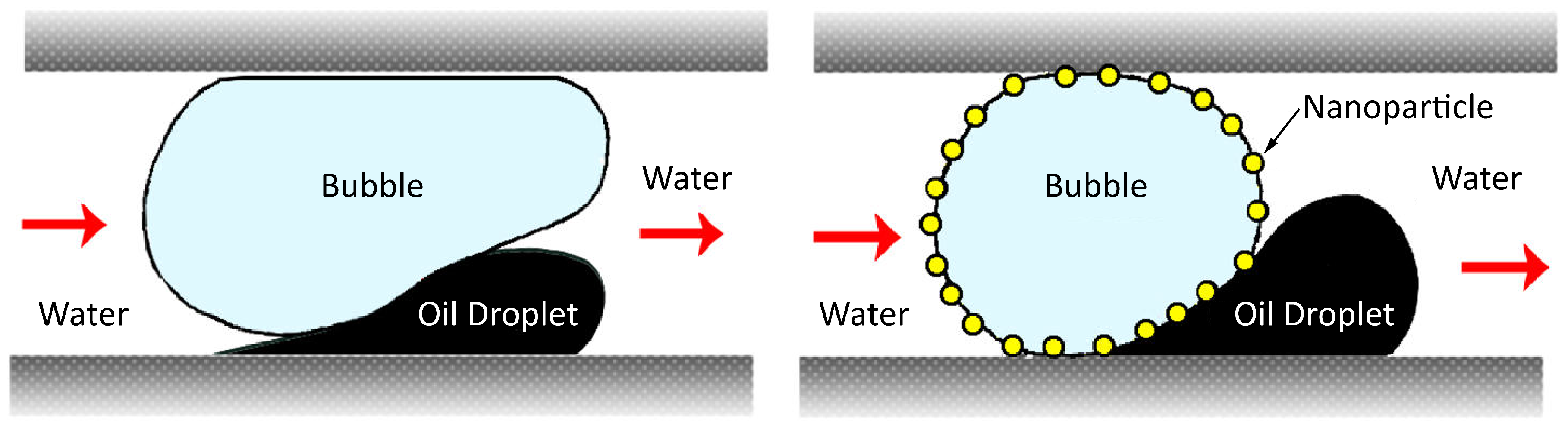
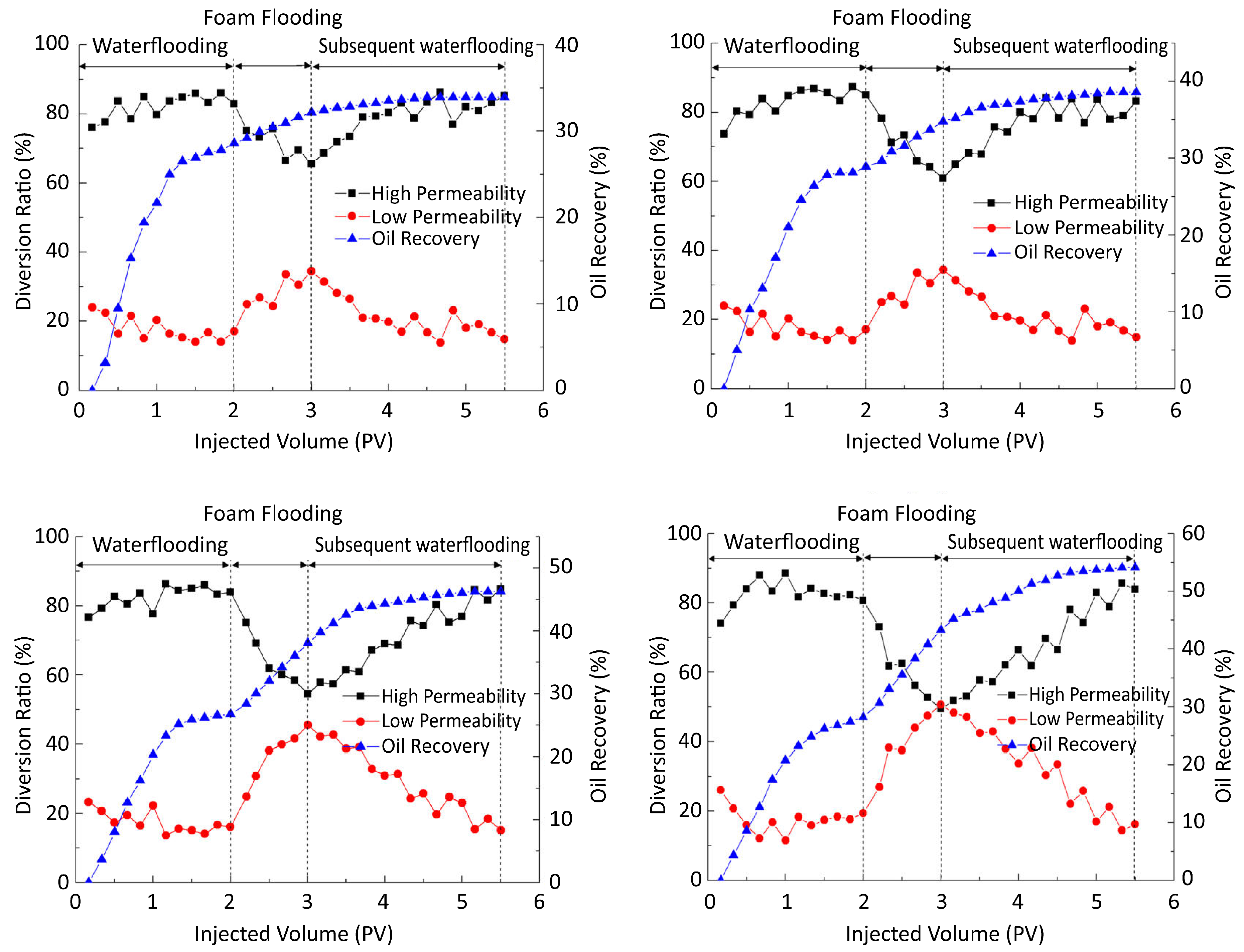
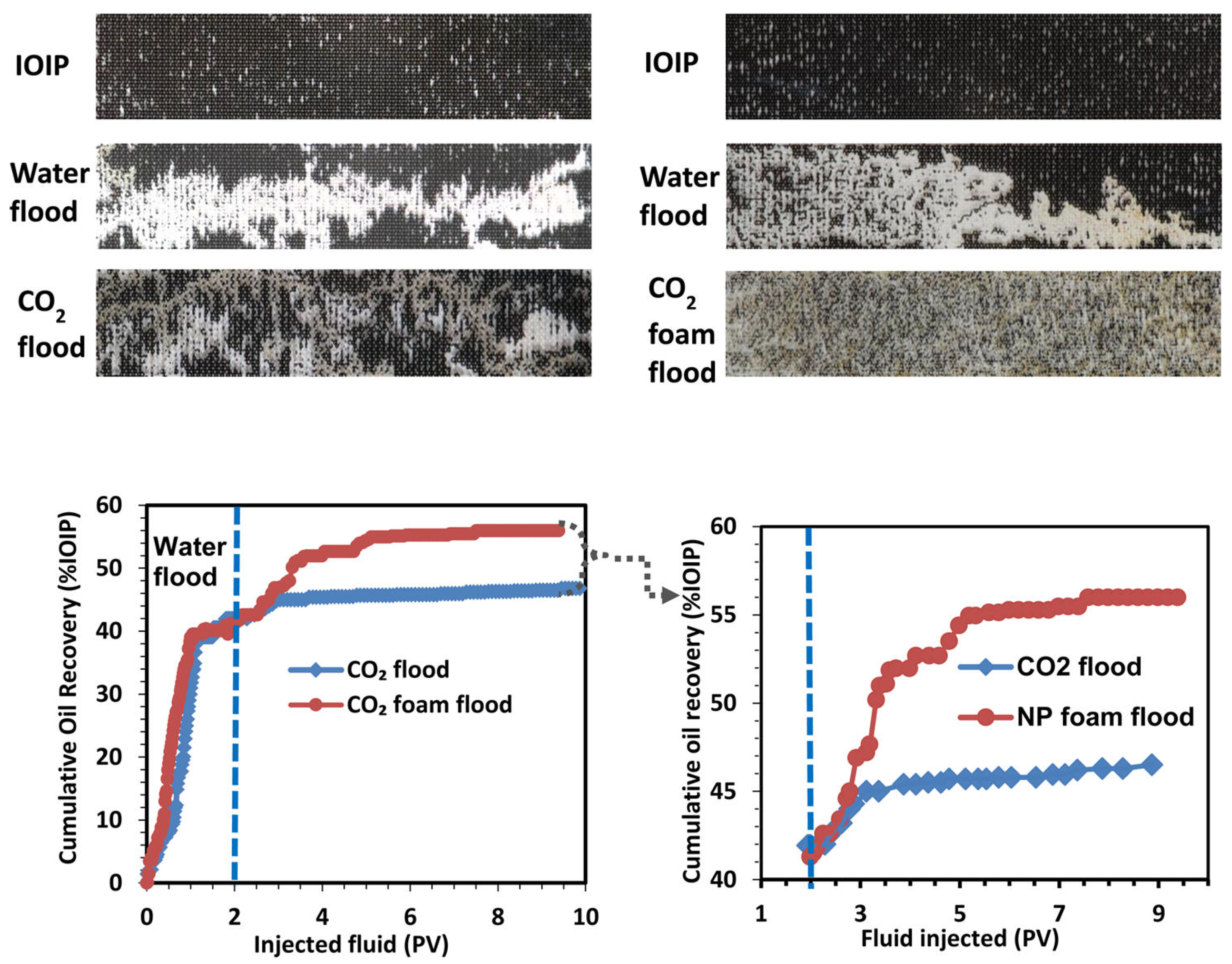
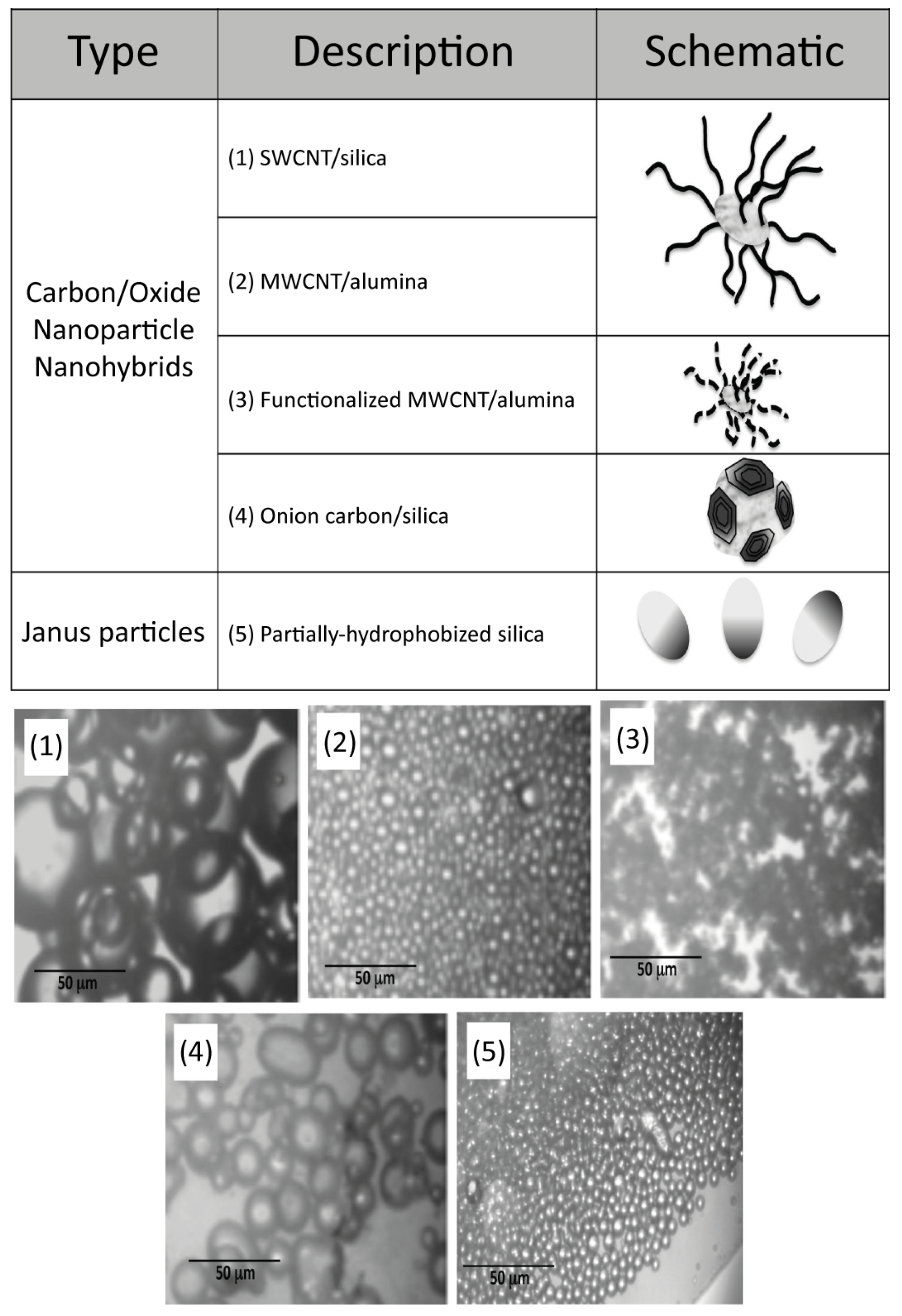
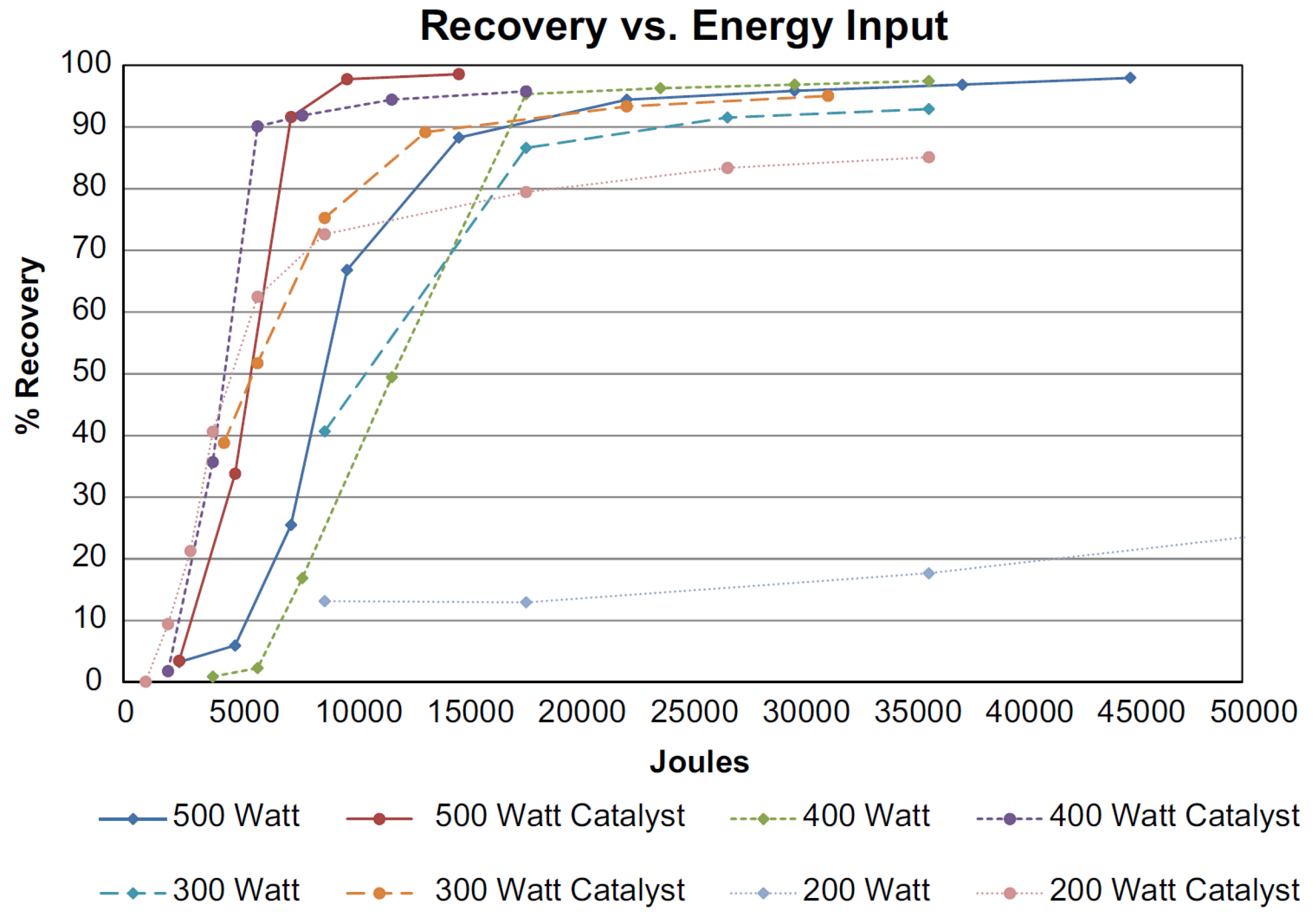
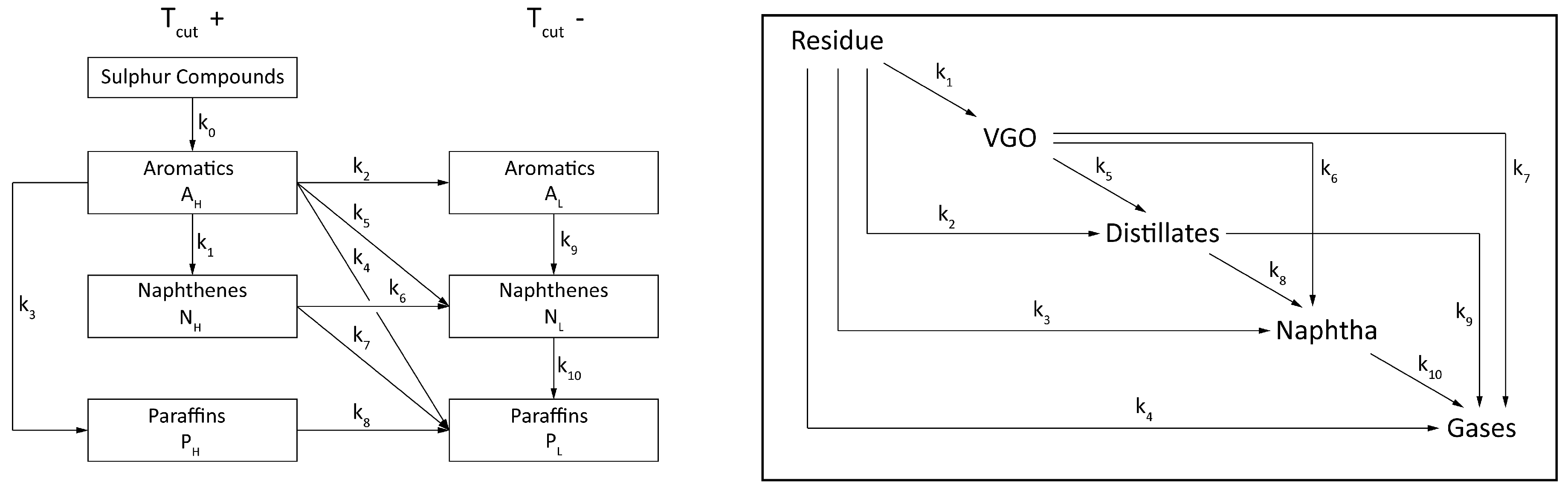
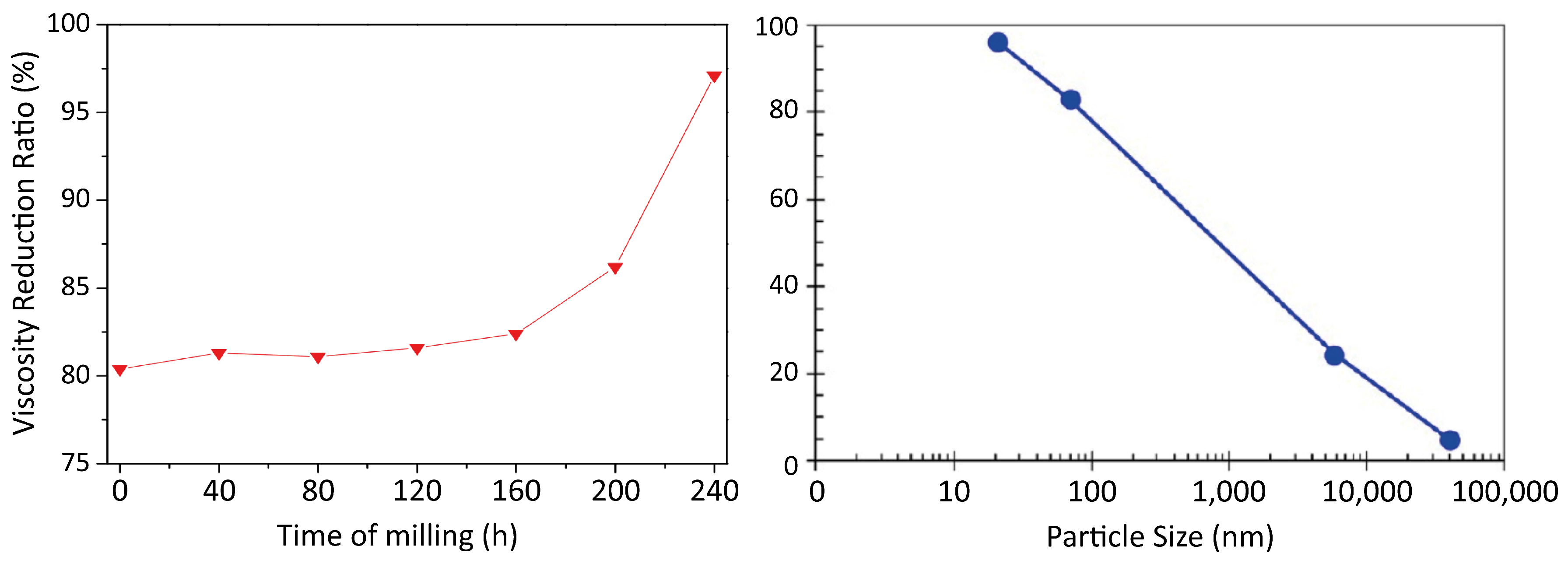

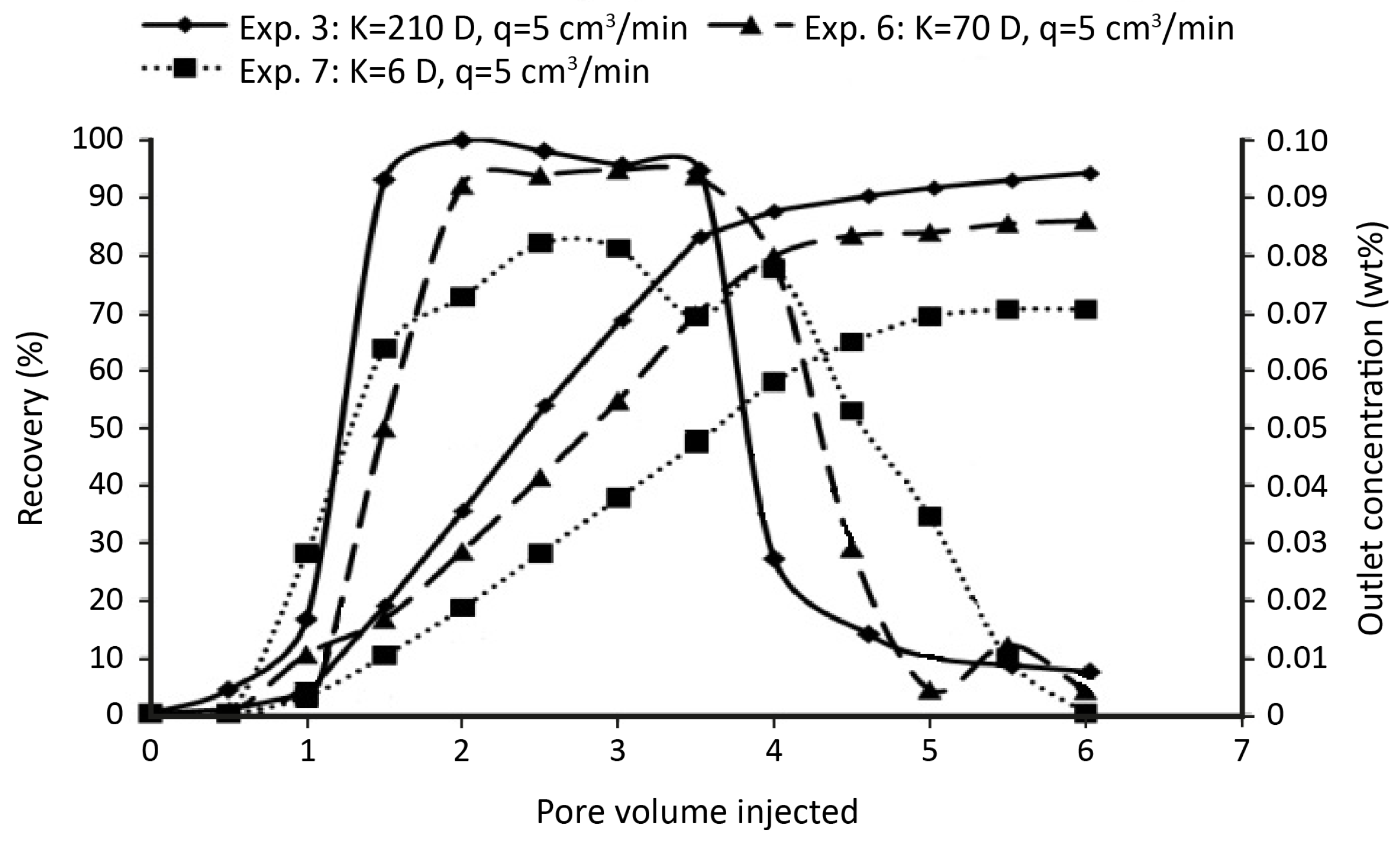
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Druetta, P.; Raffa, P.; Picchioni, F. Plenty of Room at the Bottom: Nanotechnology as Solution to an Old Issue in Enhanced Oil Recovery. Appl. Sci. 2018, 8, 2596. https://doi.org/10.3390/app8122596
Druetta P, Raffa P, Picchioni F. Plenty of Room at the Bottom: Nanotechnology as Solution to an Old Issue in Enhanced Oil Recovery. Applied Sciences. 2018; 8(12):2596. https://doi.org/10.3390/app8122596
Chicago/Turabian StyleDruetta, Pablo, Patrizio Raffa, and Francesco Picchioni. 2018. "Plenty of Room at the Bottom: Nanotechnology as Solution to an Old Issue in Enhanced Oil Recovery" Applied Sciences 8, no. 12: 2596. https://doi.org/10.3390/app8122596
APA StyleDruetta, P., Raffa, P., & Picchioni, F. (2018). Plenty of Room at the Bottom: Nanotechnology as Solution to an Old Issue in Enhanced Oil Recovery. Applied Sciences, 8(12), 2596. https://doi.org/10.3390/app8122596







