Selective Catalytic Reduction of NO with NH3 over Ce-W-Ti Oxide Catalysts Prepared by Solvent Combustion Method
Abstract
:1. Introduction
2. Materials and Methods
2.1. Catalyst Preparation
2.2. Catalyst Characterizations
2.3. Catalytic Activity Measurements
3. Results and Discussions
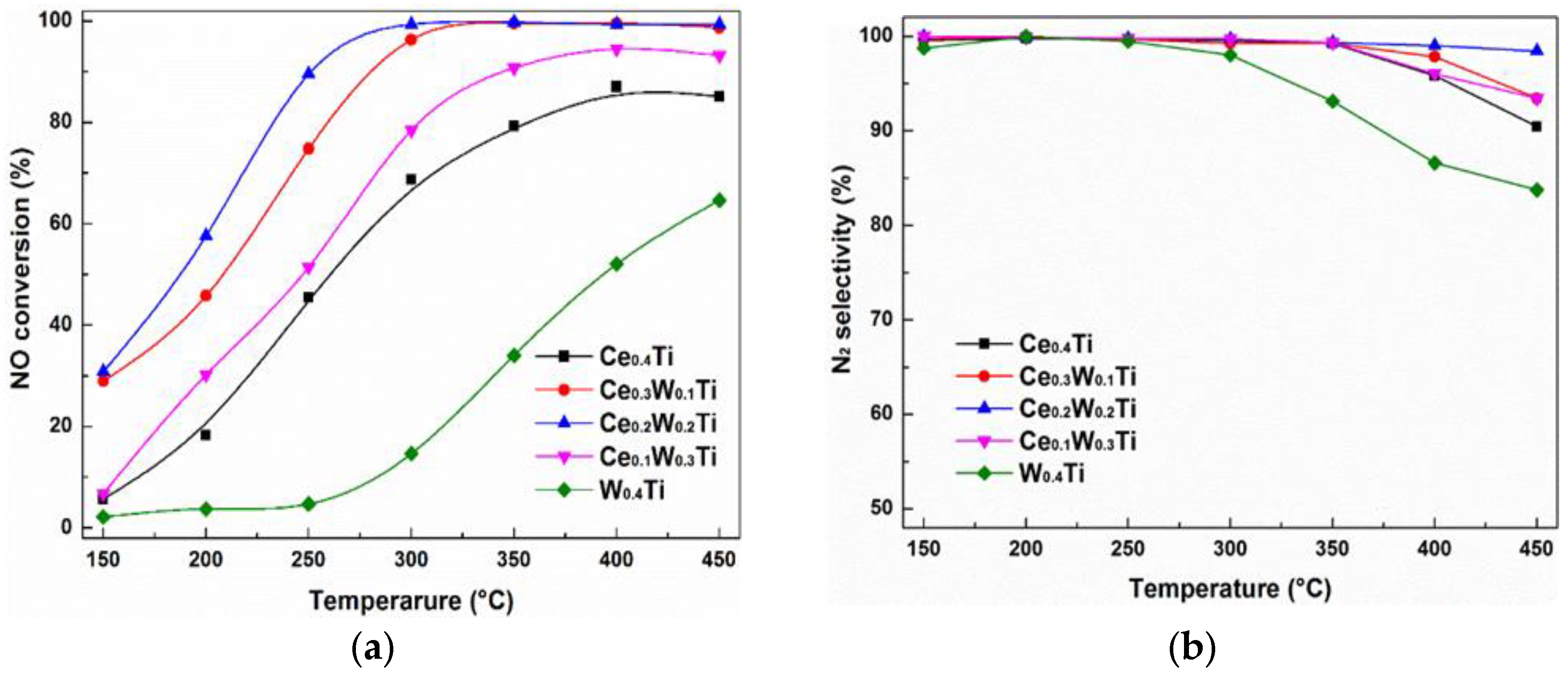
3.1. Catalytic Performance Test
3.2. N2 Adsorption and Desorption
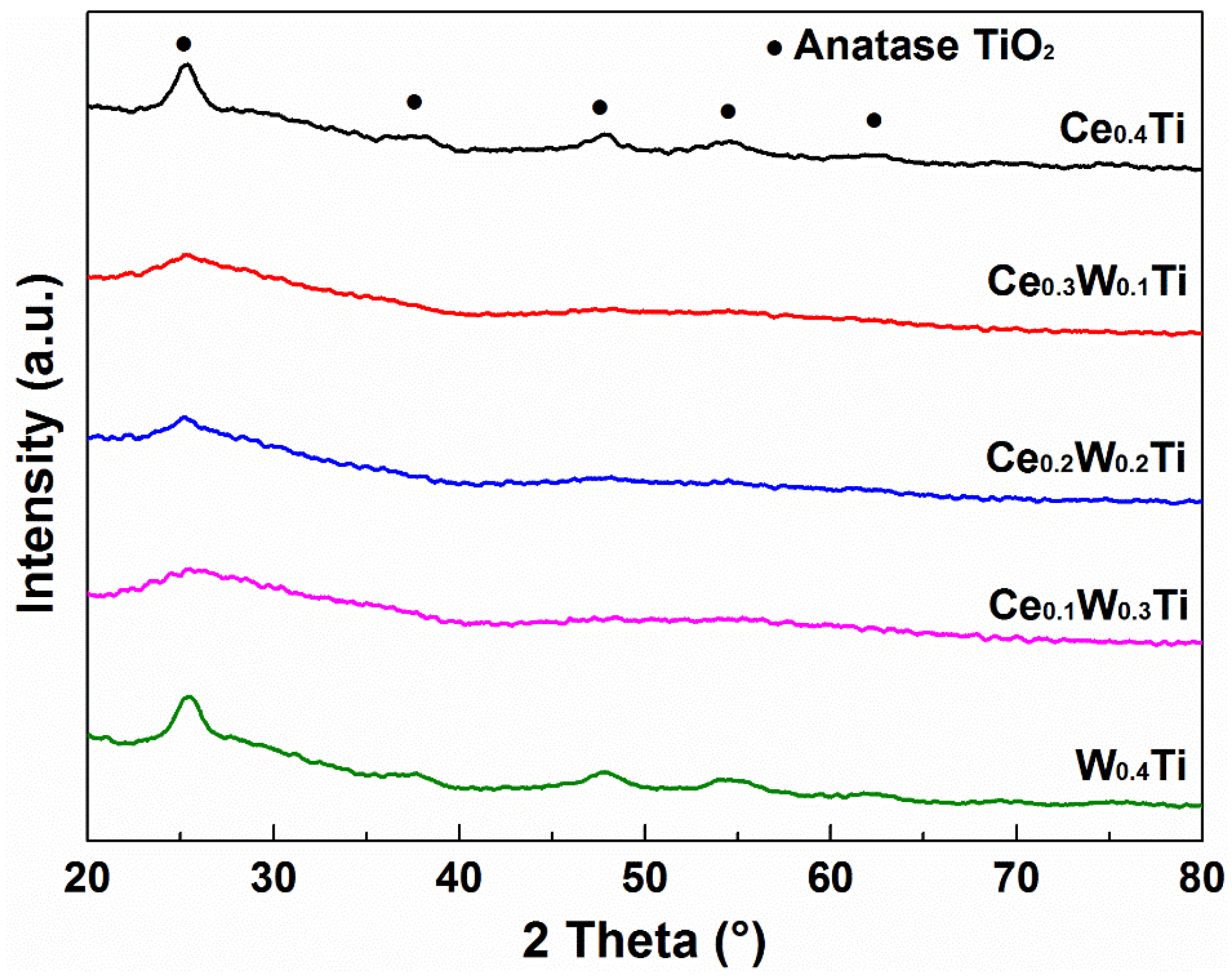
3.3. XRD Analysis
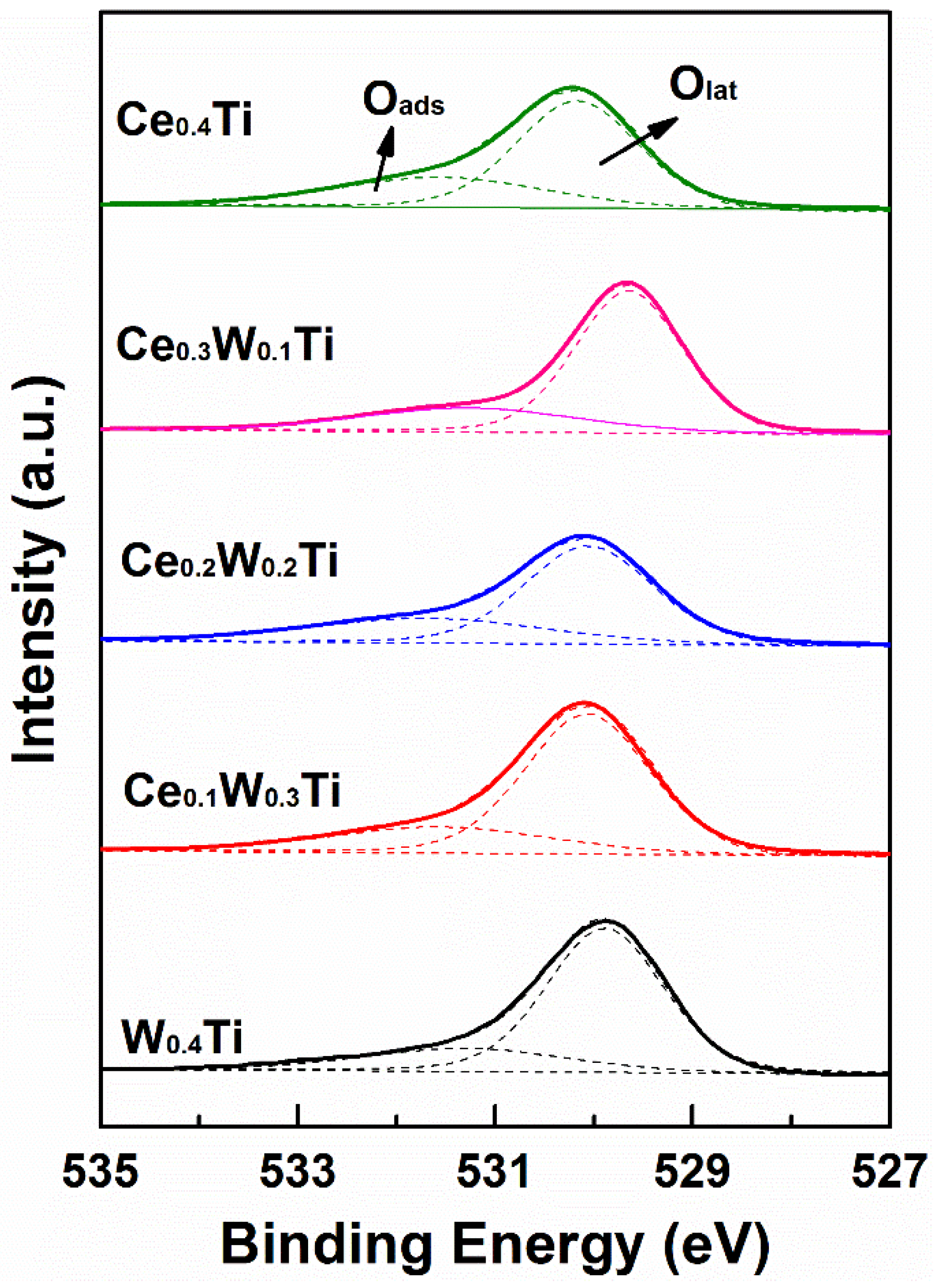
3.4. XPS Analysis
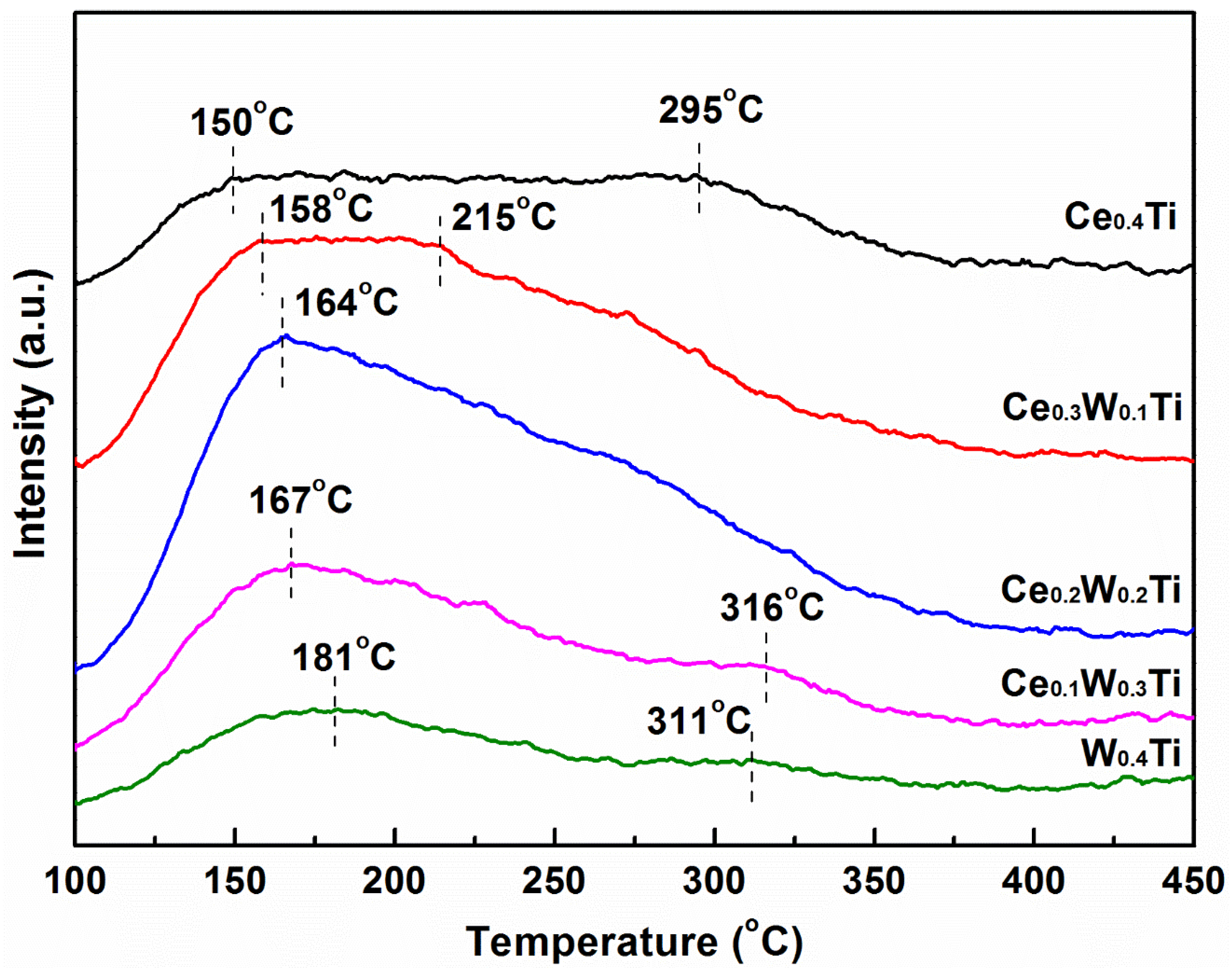
3.5. NH3-TPD Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shelef, M. Selective Catalytic Reduction of NOx with N-Free Reductants. Chem. Rev. 1995, 95, 209–225. [Google Scholar] [CrossRef]
- Pârvulescu, V.I.; Grange, P.; Delmon, B. Catalytic removal of NO. Catal. Today 1998, 46, 233–316. [Google Scholar] [CrossRef]
- Anstrom, M.; Dumesic, J.A.; Topsoe, N.Y. Theoretical insight into the nature of ammonia adsorption on vanadia-based catalysts for SCR reaction. Catal. Lett. 2002, 78, 281–289. [Google Scholar] [CrossRef]
- He, Y.; Ford, M.E.; Zhu, M.; Liu, Q.; Tumuluri, U.; Wu, Z.; Wachs, I.E. Influence of catalyst synthesis method on selective catalytic reduction (SCR) of NO by NH3 with V2O5-WO3/TiO2 catalysts. Appl. Catal. B Environ. 2016, 193, 141–150. [Google Scholar] [CrossRef]
- Zheng, Y.; Jensen, A.D.; Johnsson, J.E.; Thøgersen, J.R. Deactivation of V2O5-WO3-TiO2 SCR catalyst at biomass fired power plants: Elucidation of mechanisms by lab- and pilot-scale experiments. Appl. Catal. B Environ. 2008, 83, 186–194. [Google Scholar] [CrossRef]
- Zhu, L.; Zhong, Z.; Yang, H.; Wang, C. A comparative study of metal oxide and sulfate catalysts for selective catalytic reduction of NO with NH3. Environ. Technol. 2017, 38, 1285–1294. [Google Scholar] [CrossRef] [PubMed]
- Skalska, K.; Miller, J.S.; Ledakowicz, S. Trends in NOx abatement: A review. Sci. Total Environ. 2010, 408, 3976–3989. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Lai, J.-K.; Tumuluri, U.; Ford, M.E.; Wu, Z.; Wachs, I.E. Reaction Pathways and Kinetics for Selective Catalytic Reduction (SCR) of Acidic NOx Emissions from Power Plants with NH3. ACS Catal. 2017, 7, 8358–8361. [Google Scholar] [CrossRef]
- Devaiah, D.; Reddy, L.H.; Park, S.-E.; Reddy, B.M. Ceria–zirconia mixed oxides: Synthetic methods and applications. Catal. Rev. 2018, 60, 177–277. [Google Scholar] [CrossRef]
- Shan, W.; Liu, F.; Yu, Y.; He, H. The use of ceria for the selective catalytic reduction of NOx with NH3. Chin. J. Catal. 2014, 35, 1251–1259. [Google Scholar] [CrossRef]
- Zhang, R.D.; Yang, W.; Luo, N.; Li, P.X.; Lei, Z.G.; Chen, B.H. Low-temperature NH3-SCR of NO by lanthanum manganite perovskites: Effect of A-/B-site substitution and TiO2/CeO2 support. Appl. Catal. B Environ. 2014, 146, 94–104. [Google Scholar] [CrossRef]
- Boningari, T.; Somogyvari, A.; Smirniotis, P.G. Ce-based catalysts for the selective catalytic reduction of NOx in the presence of excess oxygen and simulated diesel engine exhaust conditions. Ind. Eng. Chem. Res. 2017, 56, 5483–5494. [Google Scholar] [CrossRef]
- Tang, C.; Zhang, H.; Dong, L. Ceria-based catalysts for low-temperature selective catalytic reduction of NO with NH3. Catal. Sci. Technol. 2016, 6, 1248–1264. [Google Scholar] [CrossRef]
- Shan, W.; Liu, F.; He, H.; Shi, X.; Zhang, C. An environmentally-benign CeO2-TiO2 catalyst for the selective catalytic reduction of NOx with NH3 in simulated diesel exhaust. Catal. Today 2012, 184, 160–165. [Google Scholar] [CrossRef]
- Qi, G.; Yang, R.T. Performance and kinetics study for low-temperature SCR of NO with NH3 over MnOx-CeO2 catalyst. J. Catal. 2003, 217, 434–441. [Google Scholar] [CrossRef]
- Zhang, G.; Han, W.; Zhao, H.; Zong, L.; Tang, Z. Solvothermal synthesis of well-designed ceria-tin-titanium catalysts with enhanced catalytic performance for wide temperature NH3-SCR reaction. Appl. Catal. B Environ. 2018, 226, 117–126. [Google Scholar] [CrossRef]
- Devaiah, D.; Thrimurthulu, G.; Smirniotis, P.G.; Reddy, B.M. Nanocrystalline alumina-supported ceria–praseodymia solid solutions: Structural characteristics and catalytic CO oxidation. RSC Adv. 2016, 6, 44826–44837. [Google Scholar] [CrossRef]
- Chen, L.; Li, J.; Ge, M. DRIFT study on cerium-tungsten/titania catalyst for selective catalytic reduction of NOx with NH3. Environ. Sci. Technol. 2010, 44, 9590–9596. [Google Scholar] [CrossRef] [PubMed]
- Michalow-Mauke, K.A.; Lu, Y.; Kowalski, K.; Graule, T.; Nachtegaal, M.; Kröcher, O.; Ferri, D. Flame-made WO3/CeOx-TiO2 catalysts for selective catalytic reduction of NOx by NH3. ACS Catal. 2015, 5, 5657–5672. [Google Scholar] [CrossRef]
- Geng, Y.; Huang, H.; Chen, X.; Ding, H.; Yang, S.; Liu, F.; Shan, W. The effect of Ce on a high-efficiency CeO2/WO3-TiO2 catalyst for the selective catalytic reduction of NOx with NH3. RSC Adv. 2016, 6, 64803–64810. [Google Scholar] [CrossRef]
- Chen, L.; Li, J.; Ge, M.; Zhu, R. Enhanced activity of tungsten modified CeO2/TiO2 for selective catalytic reduction of NOx with ammonia. Catal. Today 2010, 153, 77–83. [Google Scholar] [CrossRef]
- Jiang, Y.; Xing, Z.; Wang, X.; Huang, S.; Wang, X.; Liu, Q. Activity and characterization of a Ce-W-Ti oxide catalyst prepared by a single step sol-gel method for selective catalytic reduction of NO with NH3. Fuel 2015, 151, 124–129. [Google Scholar] [CrossRef]
- Jiang, Y.; Bao, C.; Liu, Q.; Liang, G.; Lu, M.; Ma, S. A novel CeO2-MoO3-WO3/TiO2 catalyst for selective catalytic reduction of NO with NH3. Catal. Commun. 2018, 103, 96–100. [Google Scholar] [CrossRef]
- Shan, W.; Liu, F.; He, H.; Shi, X.; Zhang, C. A superior Ce-W-Ti mixed oxide catalyst for the selective catalytic reduction of NOx with NH3. Appl. Catal. B Environ. 2012, 115, 100–106. [Google Scholar] [CrossRef]
- Shan, W.; Geng, Y.; Chen, X.; Huang, N.; Liu, F.; Yang, S. A highly efficient CeWOx catalyst for the selective catalytic reduction of NOx with NH3. Catal. Sci. Technol. 2016, 6, 1195–1200. [Google Scholar] [CrossRef]
- Xu, L.; Wang, C.; Chang, H.; Wu, Q.; Zhang, T.; Li, J. New insight into SO2 poisoning and regeneration of CeO2-WO3/TiO2 and V2O5-WO3/TiO2 catalysts for low-temperature NH3-SCR. Environ. Sci. Technol. 2018, 52, 7064–7071. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Xiong, S.; Shi, Y.; Shan, W.; Yang, S. Effect of H2O and SO2 on the selective catalytic reduction of NO with NH3 Over Ce/TiO2 Catalyst: Mechanism and Kinetic Study. J. Phys. Chem. C 2016, 120, 1066–1076. [Google Scholar] [CrossRef]
- Gao, X.; Jiang, Y.; Fu, Y.; Zhong, Y.; Luo, Z.; Cen, K. Preparation and characterization of CeO2/TiO2 catalysts for selective catalytic reduction of NO with NH3. Catal. Commun. 2010, 11, 465–469. [Google Scholar] [CrossRef]
- Xu, W.; He, H.; Yu, Y. Deactivation of a Ce/TiO2 catalyst by SO2 in the selective catalytic reduction of NO by NH3. J. Phys. Chem. C 2009, 113, 4426–4432. [Google Scholar] [CrossRef]
- Lippens, B.C.; De Boer, J. Studies on pore systems in catalysts: V. The t method. J. Catal. 1965, 4, 319–323. [Google Scholar] [CrossRef]
- Yu, J.C.; Zhang, L.; Yu, J. Rapid synthesis of mesoporous TiO2 with high photocatalytic activity by ultrasound-induced agglomeration. New J. Chem. 2002, 26, 416–420. [Google Scholar] [CrossRef]
- Zhao, K.; Han, W.; Lu, G.; Lu, J.; Tang, Z.; Zhen, X. Promotion of redox and stability features of doped Ce-W-Ti for NH3-SCR reaction over a wide temperature range. Appl. Surf. Sci. 2016, 379, 316–322. [Google Scholar] [CrossRef]
- Guo, R.T.; Chen, Q.L.; Ding, H.L.; Wang, Q.S.; Pan, W.G.; Yang, N.Z.; Lu, C.Z. Preparation and characterization of CeOx@MnOx core-shell structure catalyst for catalytic oxidation of NO. Catal. Commun. 2015, 69, 165–169. [Google Scholar] [CrossRef]
- Chen, H.; Xia, Y.; Huang, H.; Gan, Y.; Tao, X.; Liang, C.; Luo, J.; Fang, R.; Zhang, J.; Zhang, W.; et al. Highly dispersed surface active species of Mn/Ce/TiW catalysts for high performance at low temperature NH3-SCR. Chem. Eng. J. 2017, 330, 1195–1202. [Google Scholar] [CrossRef]
- Zhan, S.; Zhang, H.; Zhang, Y.; Shi, Q.; Li, Y.; Li, X. Efficient NH3-SCR removal of NOx with highly ordered mesoporous WO3(χ)-CeO2 at low temperatures. Appl. Catal. B Environ. 2017, 203, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Dupin, J.-C.; Gonbeau, D.; Vinatier, P.; Levasseur, A. Systematic XPS studies of metal oxides, hydroxides and peroxides. Phys. Chem. Chem. Phys. 2000, 2, 1319–1324. [Google Scholar] [CrossRef]
- Busca, G.; Lietti, L.; Ramis, G.; Berti, F. Chemical and mechanistic aspects of the selective catalytic reduction of NOx by ammonia over oxide catalysts: A review. Appl. Catal. B Environ. 1998, 18, 1–36. [Google Scholar] [CrossRef]
- Sanchez, C.; Livage, J.; Lucazeau, G. Infrared and Raman study of amorphous V2O5. J. Raman Spectrosc. 1982, 12, 68–72. [Google Scholar] [CrossRef]

| Samples | SBET (m2·g−1) | Pore Volume (cm3·g−1) | Average Pore Diameter (nm) | Oads/(Oads + Olat) (%) | NH3 Uptake (μmol·g−1) |
|---|---|---|---|---|---|
| Ce0.4Ti | 26.6 | 0.076 | 9.9 | 17.4 | 7.8 |
| Ce0.3W0.1Ti | 46.8 | 0.086 | 7.7 | 27.6 | 64.7 |
| Ce0.2W0.2Ti | 108.1 | 0.121 | 3.9 | 33.8 | 193.9 |
| Ce0.1W0.3Ti | 68.4 | 0.085 | 4.6 | 21.0 | 62.3 |
| W0.4Ti | 16.8 | 0.069 | 16.2 | 16.0 | 20.0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, X.; Wang, Y.; Huang, Y.; Cai, Y. Selective Catalytic Reduction of NO with NH3 over Ce-W-Ti Oxide Catalysts Prepared by Solvent Combustion Method. Appl. Sci. 2018, 8, 2430. https://doi.org/10.3390/app8122430
Zhu X, Wang Y, Huang Y, Cai Y. Selective Catalytic Reduction of NO with NH3 over Ce-W-Ti Oxide Catalysts Prepared by Solvent Combustion Method. Applied Sciences. 2018; 8(12):2430. https://doi.org/10.3390/app8122430
Chicago/Turabian StyleZhu, Xinbo, Yaolin Wang, Yu Huang, and Yuxiang Cai. 2018. "Selective Catalytic Reduction of NO with NH3 over Ce-W-Ti Oxide Catalysts Prepared by Solvent Combustion Method" Applied Sciences 8, no. 12: 2430. https://doi.org/10.3390/app8122430
APA StyleZhu, X., Wang, Y., Huang, Y., & Cai, Y. (2018). Selective Catalytic Reduction of NO with NH3 over Ce-W-Ti Oxide Catalysts Prepared by Solvent Combustion Method. Applied Sciences, 8(12), 2430. https://doi.org/10.3390/app8122430




