Purification of Lithium Carbonate from Sulphate Solutions through Hydrogenation Using the Dowex G26 Resin
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Equipment
2.3. Experimental Procedures
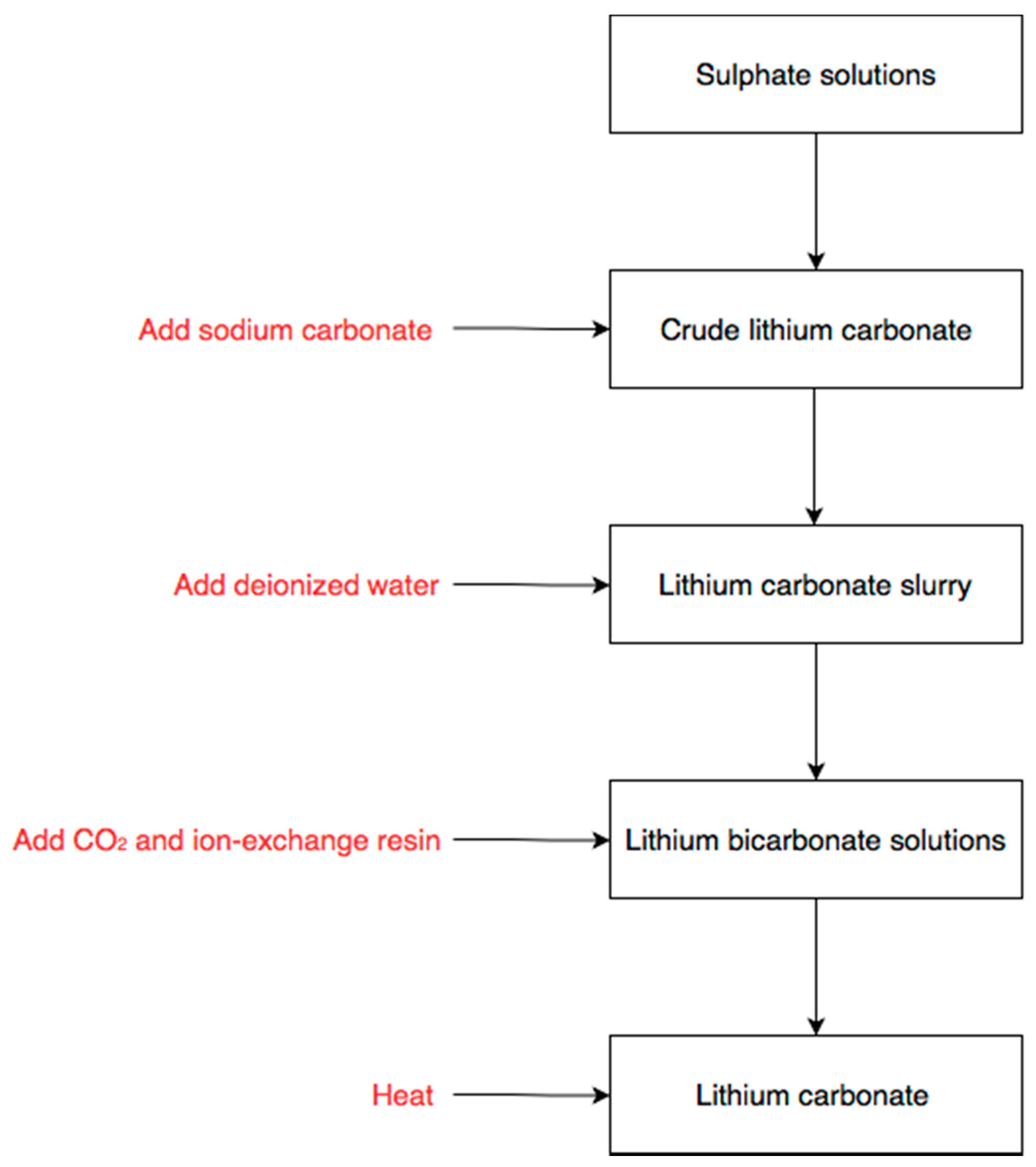
2.3.1. Chemical Precipitation
2.3.2. Lithium Carbonate Slurry
2.3.3. Hydrogenation Processing
2.3.4. Ion-Exchange
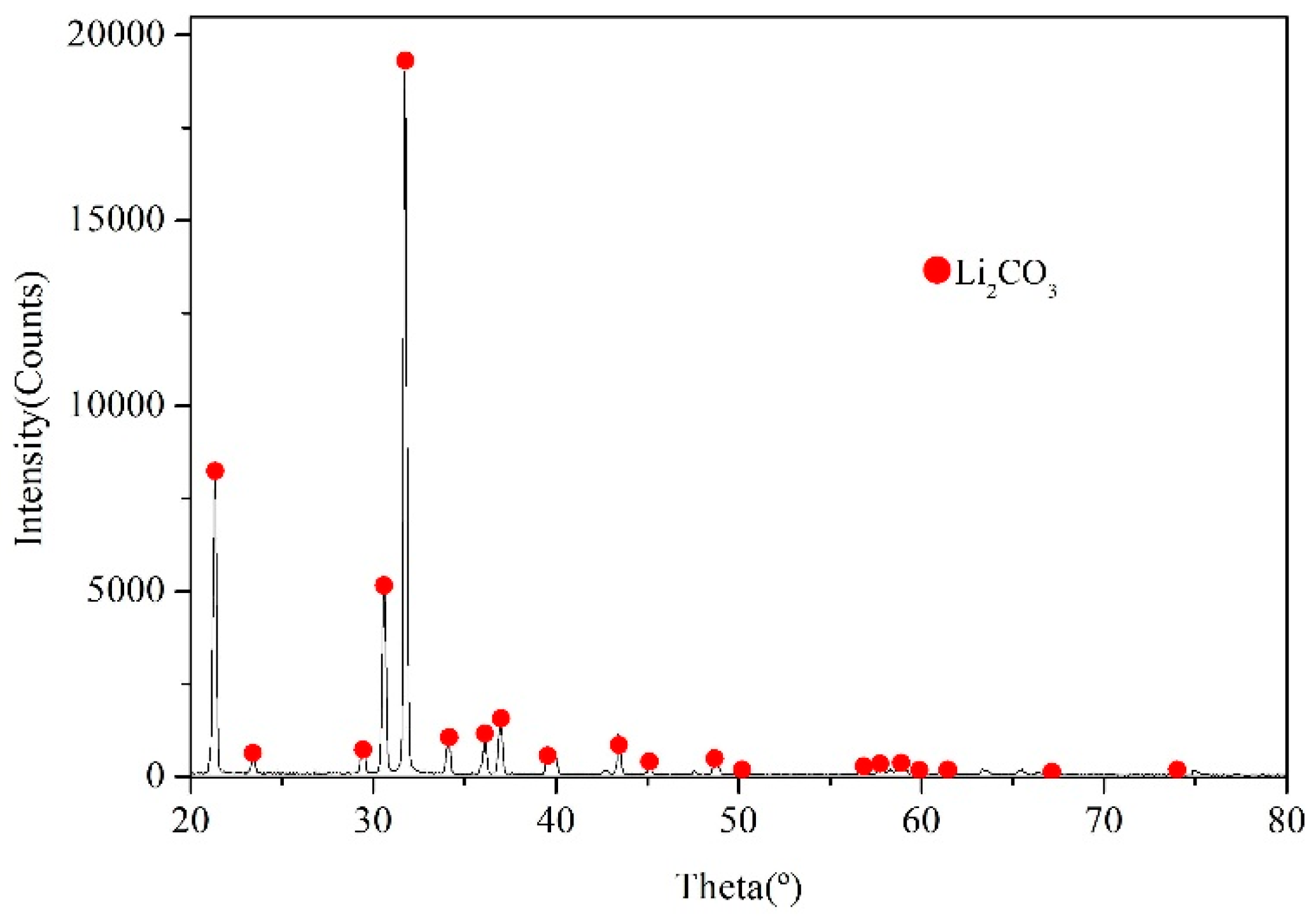
2.3.5. Decomposition Processing
3. Results and Discussion
3.1. Hydrogenation Processing
3.1.1. Effect of CO2 Aeration
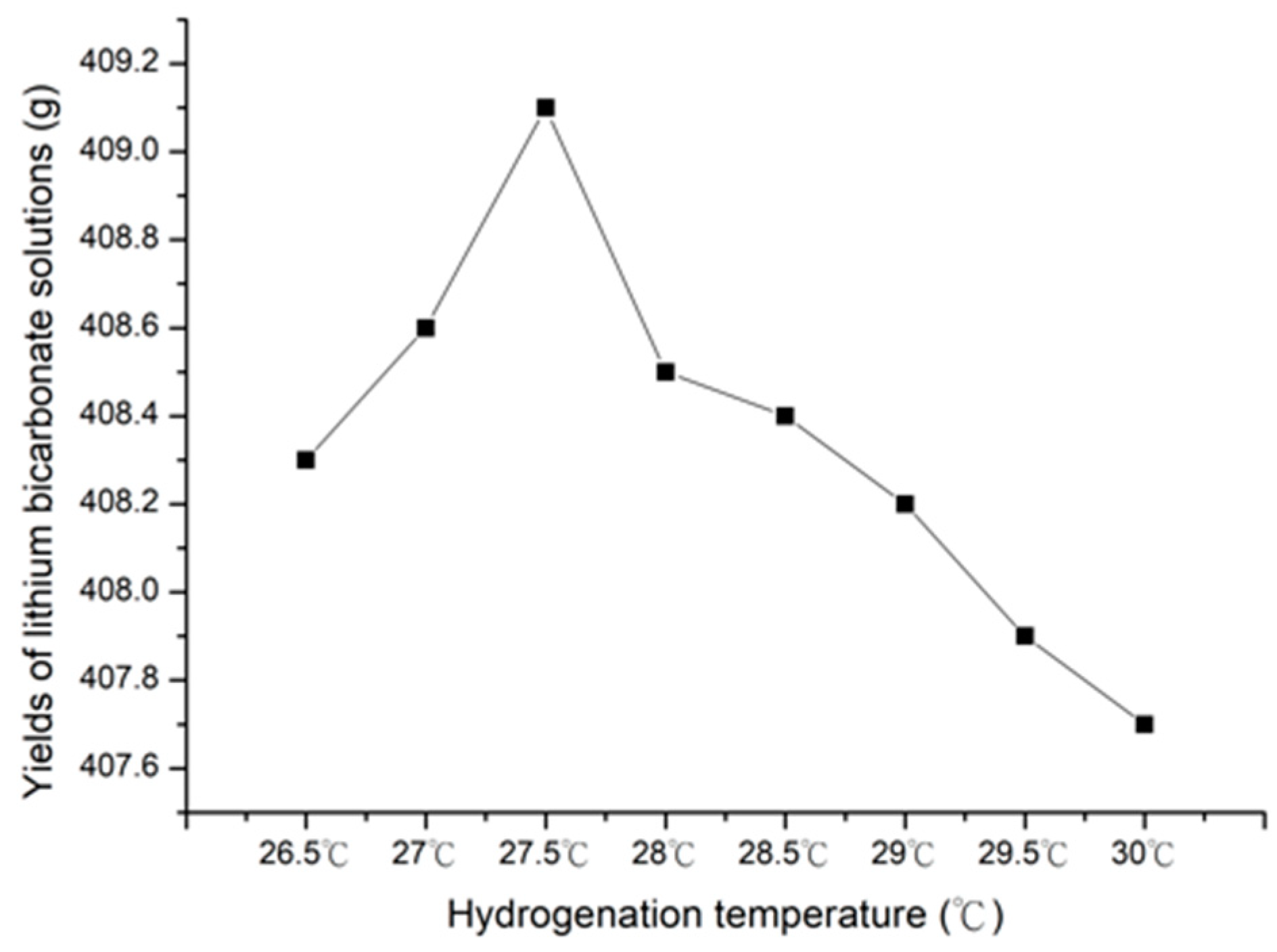
3.1.2. Effect of Hydrogenation Temperature
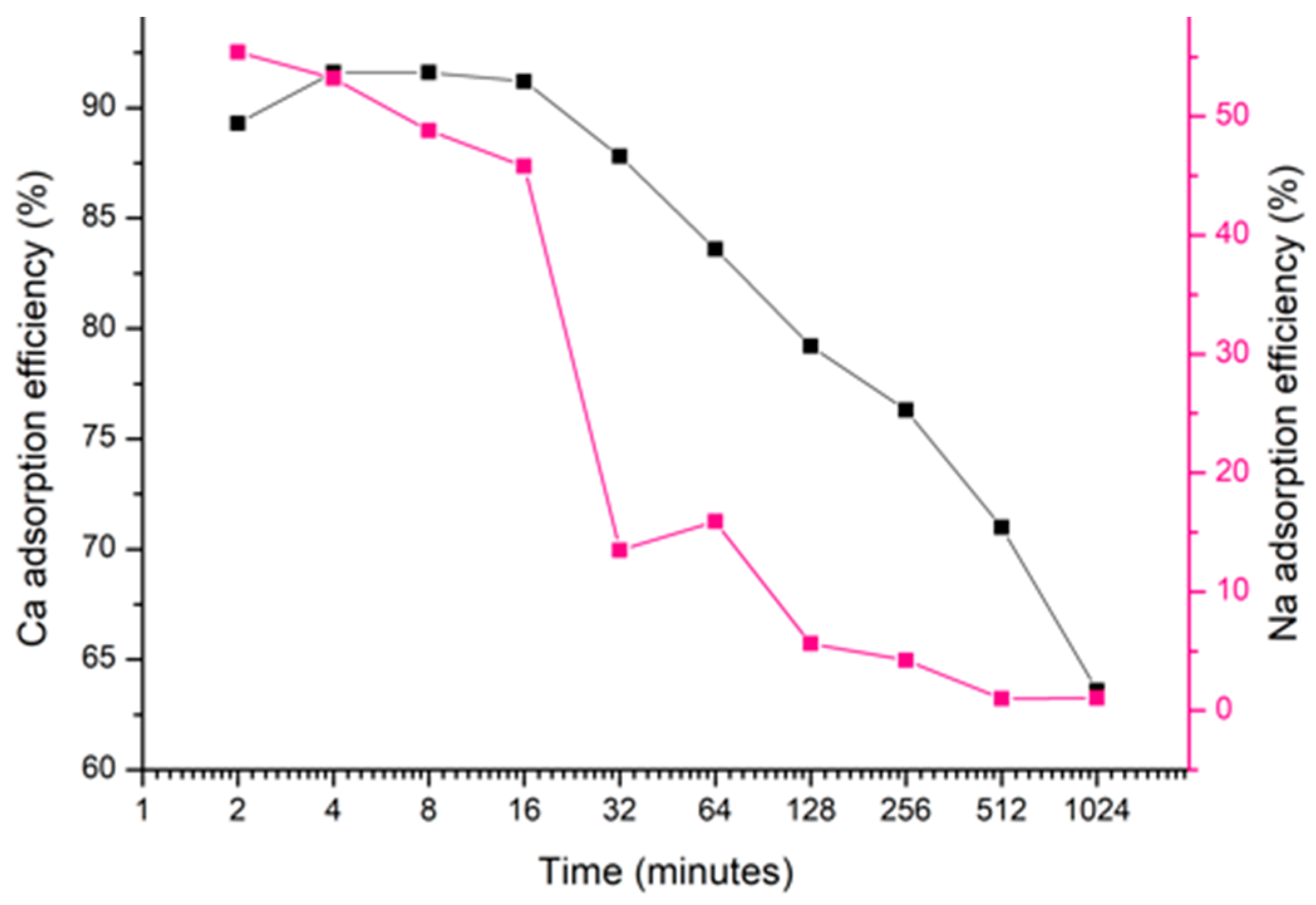
3.2. Ion-Exchange
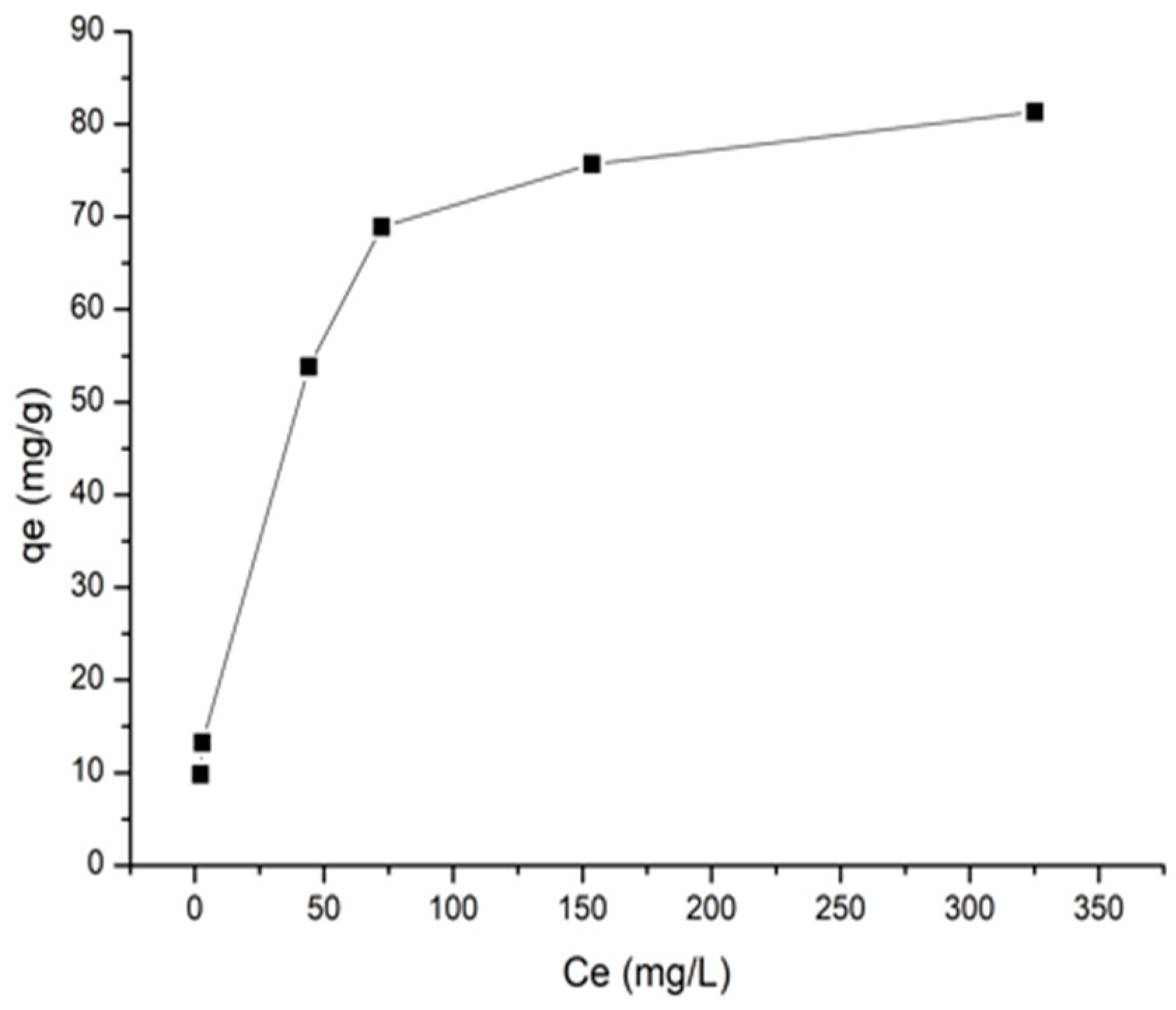
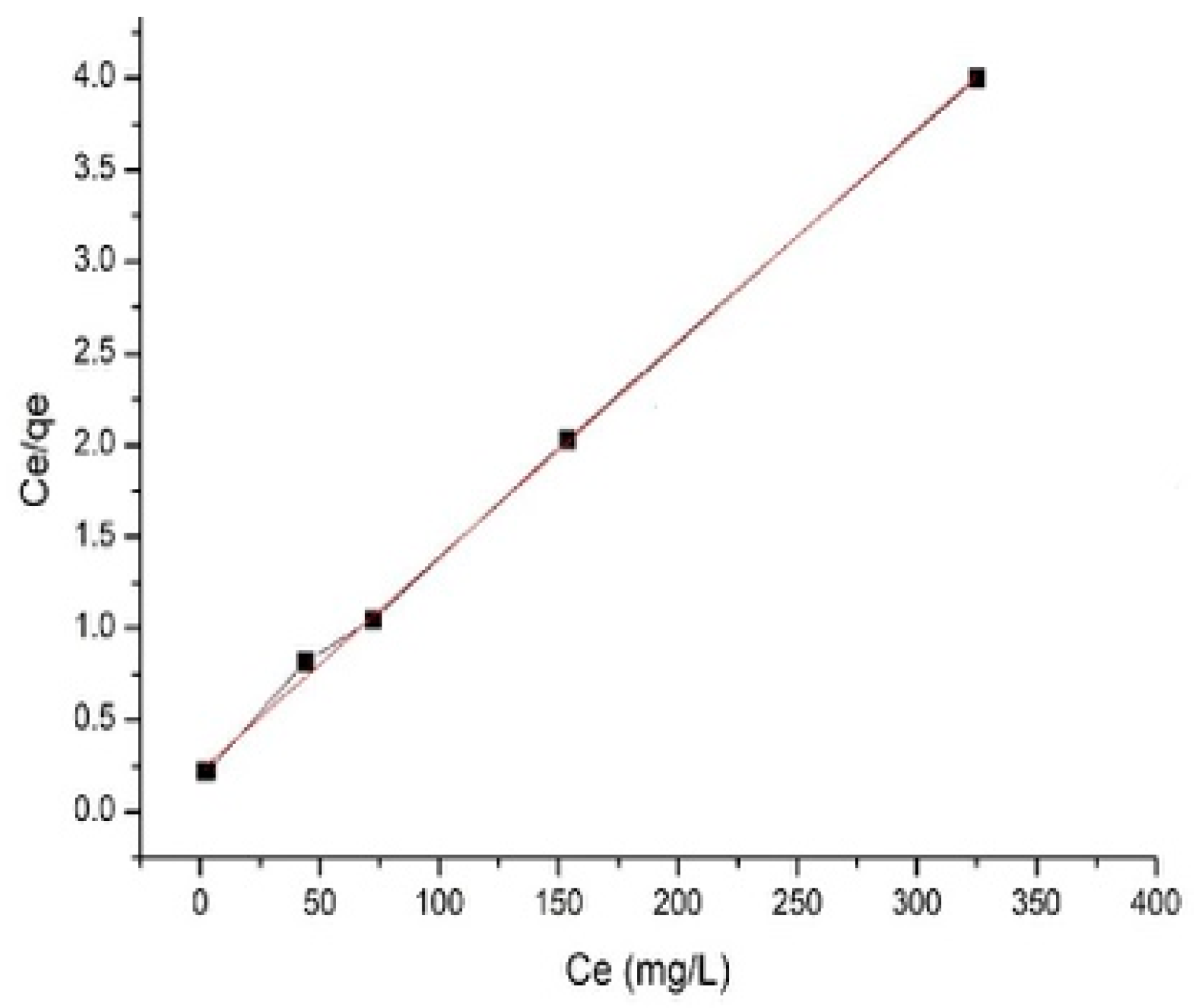
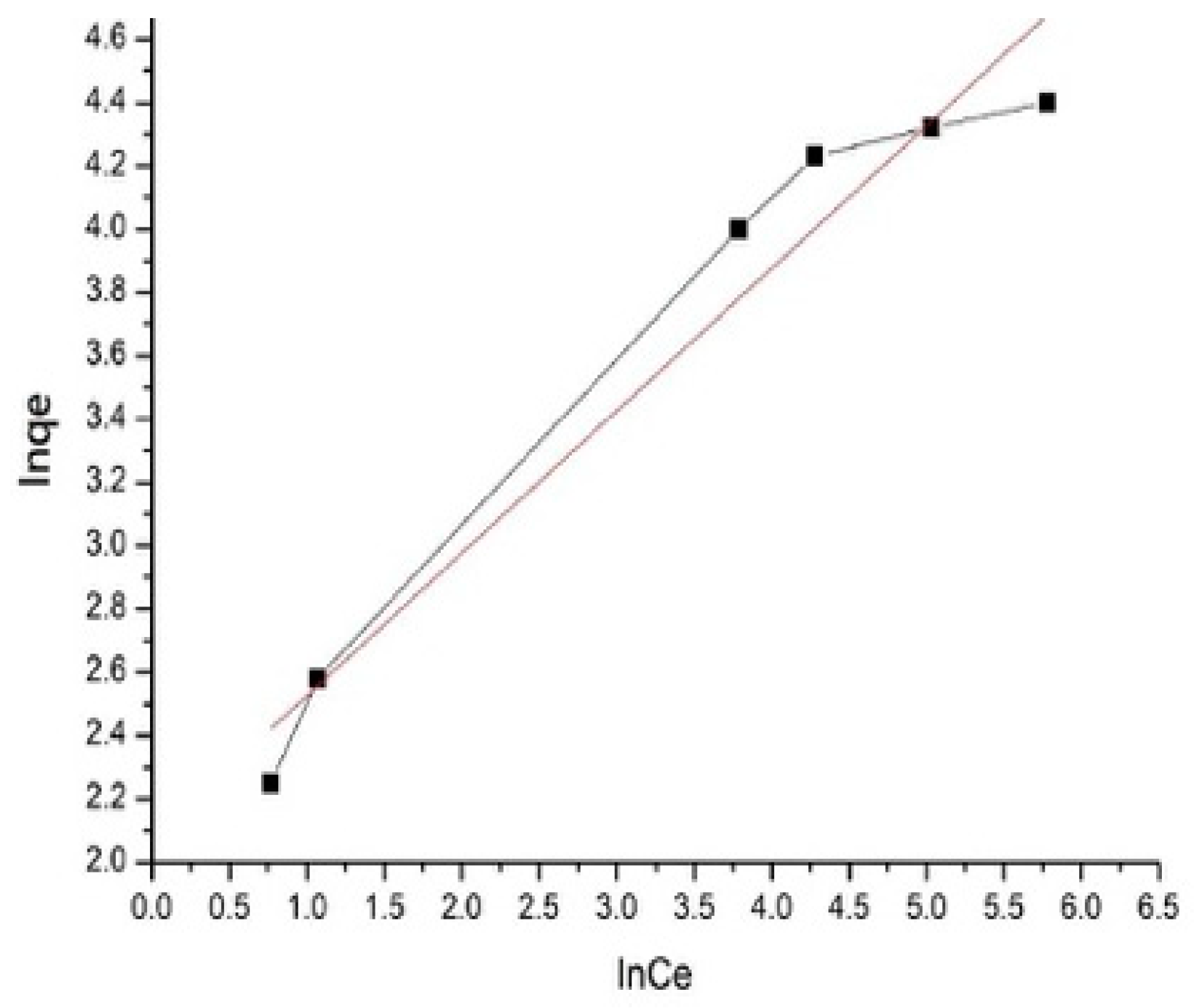
3.2.1. Isothermal Adsorption Models
3.2.2. Effect of the pH Value
3.2.3. Effect of Reaction Time
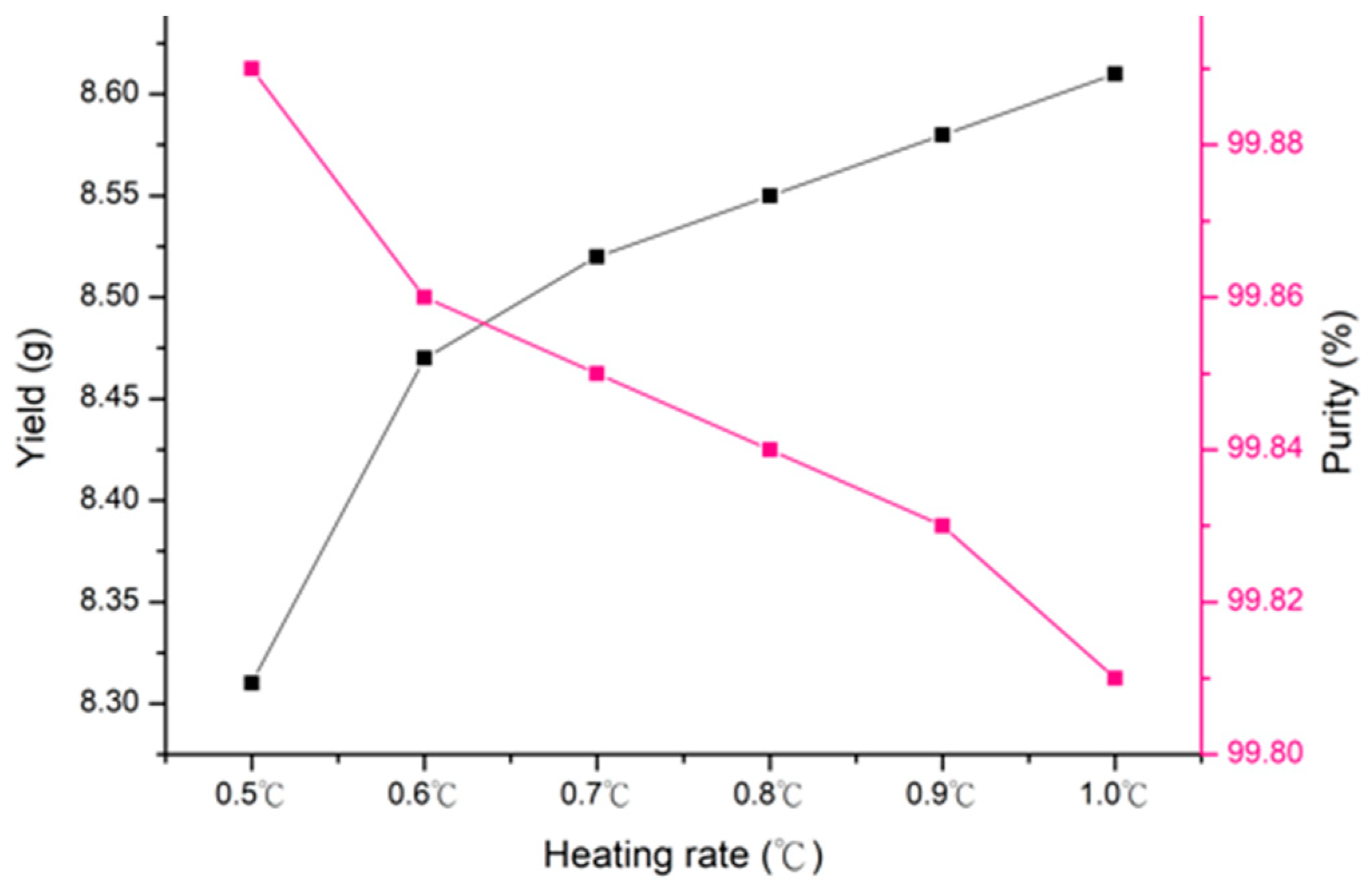
3.3. Decomposition Processing
The Effect of Heating Rate
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alias, N.; Mohamad, A. Advances of aqueous rechargeable lithium-ion battery: A review. J. Power Sources 2015, 274, 237–251. [Google Scholar] [CrossRef]
- Armand, M.; Tarascon, J. Building better batteries. Nature 2008, 451, 652–657. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Li, J.; Singh, N. Recycling of Spent Lithium-Ion Battery: A Critical Review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 1129–1165. [Google Scholar] [CrossRef]
- Chu, S.; Cui, Y.; Liu, N. The path towards sustainable energy. Nat. Mater. 2017, 16, 16–22. [Google Scholar] [CrossRef] [PubMed]
- U.S. Geological Survey. Mineral Commodity Summaries 2013; U.S. Geological Survey: Reston, VA, USA, 2013.
- U.S. Geological Survey. Mineral Commodity Summaries 2014; U.S. Geological Survey: Reston, VA, USA, 2014.
- U.S. Geological Survey. Mineral Commodity Summaries 2015; U.S. Geological Survey: Reston, VA, USA, 2015.
- U.S. Geological Survey. Mineral Commodity Summaries 2016; U.S. Geological Survey: Reston, VA, USA, 2016.
- U.S. Geological Survey. Mineral Commodity Summaries 2017; U.S. Geological Survey: Reston, VA, USA, 2017.
- Gao, D.; Yu, X.; Guo, Y.; Wang, S.; Liu, M.; Deng, T.; Chen, Y.; Belzile, N. Extraction of lithium from salt lake brine with triisobutyl phosphate in ionic liquid and kerosene. Chem. Res. Chin. Univ. 2015, 31, 621–626. [Google Scholar] [CrossRef]
- Liu, X.; Chen, X.; He, L.; Zhao, Z. Study on extraction of lithium from salt lake brine by membrane electrolysis. Desalination 2015, 376, 35–40. [Google Scholar] [CrossRef]
- Somrani, A.; Hamzaoui, A.; Pontie, M. Study on lithium separation from salt lake brines by nanofiltration (NF) and low pressure reverse osmosis (LPRO). Desalination 2013, 317, 184–192. [Google Scholar] [CrossRef]
- Medina, L.; El-Naggar, M. An alternative method for the recovery of lithium from spodumene. Metall. Trans. B 1984, 15, 725–726. [Google Scholar] [CrossRef]
- Fröhlich, P.; Lorenz, T.; Martin, G.; Brett, B.; Bertau, M. Valuable Metals-Recovery Processes, Current Trends, and Recycling Strategies. Angew. Chem. Int. Ed. 2017, 56, 2544–2580. [Google Scholar] [CrossRef] [PubMed]
- Geddes, J.; Burgess, S.; Hawton, K.; Jamison, K.; Goodwin, G. Long-Term Lithium Therapy for Bipolar Disorder: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Am. J. Psychiatry 2004, 161, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Severus, E.; Taylor, M.; Sauer, C.; Pfennig, A.; Ritter, P.; Bauer, M.; Geddes, J.R. Lithium for prevention of mood episodes in bipolar disorders: Systematic review and meta-analysis. Int. J. Bipolar Disord. 2014, 2, 15. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Xiao, X.; Li, F.; Ma, P. Discussion on Methods for the Preparation for High-purity Lithium Carbonate. J. Salt Lake Res. 2005, 2, 53–60. [Google Scholar]
- Chen, W.; Ho, H. Recovery of Valuable Metals from Lithium-Ion Batteries NMC Cathode Waste Materials by Hydrometallurgical Methods. Metals 2018, 8, 321. [Google Scholar] [CrossRef]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. Part II.—Liquids. J. Am. Chem. Soc. 1917, 184, 721. [Google Scholar] [CrossRef]
- Freundlich, H. Über die Adsorption in Lösungen. Z. Phys. Chem. 1907, 57, 385–470. [Google Scholar] [CrossRef]



| Element | Ni | Li |
|---|---|---|
| Concentration (ppm) | 130 ppm | 8000 ppm |
| Element | Lithium Carbonate | Ca2+ | Na+ | SO42− |
|---|---|---|---|---|
| Content (%) | 98% | 0.15% | 1.5% | 0.35% |
| Element | Li+ | Ca2+ | Na+ | SO42− |
|---|---|---|---|---|
| Content (%) | 99.475% | 0.015% | 0.5% | 0.01% |
| Element | Li+ | Ca2+ | Na+ | SO42− |
|---|---|---|---|---|
| Content (%) | 99.9% | 0.005% | 0.095% | N.D. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, W.-S.; Lee, C.-H.; Ho, H.-J. Purification of Lithium Carbonate from Sulphate Solutions through Hydrogenation Using the Dowex G26 Resin. Appl. Sci. 2018, 8, 2252. https://doi.org/10.3390/app8112252
Chen W-S, Lee C-H, Ho H-J. Purification of Lithium Carbonate from Sulphate Solutions through Hydrogenation Using the Dowex G26 Resin. Applied Sciences. 2018; 8(11):2252. https://doi.org/10.3390/app8112252
Chicago/Turabian StyleChen, Wei-Sheng, Cheng-Han Lee, and Hsing-Jung Ho. 2018. "Purification of Lithium Carbonate from Sulphate Solutions through Hydrogenation Using the Dowex G26 Resin" Applied Sciences 8, no. 11: 2252. https://doi.org/10.3390/app8112252
APA StyleChen, W.-S., Lee, C.-H., & Ho, H.-J. (2018). Purification of Lithium Carbonate from Sulphate Solutions through Hydrogenation Using the Dowex G26 Resin. Applied Sciences, 8(11), 2252. https://doi.org/10.3390/app8112252







