Formulation of Laccase Nanobiocatalysts Based on Ionic and Covalent Interactions for the Enhanced Oxidation of Phenolic Compounds
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Nanoparticles for Enzyme Immobilization
2.2. Laccase Activity
2.3. Functionalization of Laccase onto Silica and Silica-Coated Magnetic Nanoparticles
2.4. Immobilization of Laccase onto Silica and Polyethylenimine Nanoparticles
2.5. Immobilization of Laccase onto Polyacrylic Acid-Coated Magnetic Nanoparticles
2.6. Immobilization of Laccase by Ionic Exchange on Magnetite Nanoparticles
2.7. Biotransformation of Phenol by Laccase Immobilized onto fsNPs and Single-Core Silica-Coated Magnetic Nanoparticles in Batch Operation
2.8. Consecutive Cycles of Batch Biotransformation of Phenol by Laccase Immobilized onto Single-Core Silica-Coated Magnetic Nanoparticles
2.9. Phenol Analysis
2.10. Life Cycle Assessment Methodology
3. Results and Discussion
3.1. Immobilization of Laccase onto Different Types of Nanoparticles
3.2. Biotransformation of Phenol by Laccase Immobilized onto fsNPs and Silica-Coated Magnetic Nanoparticles in Batch Operation
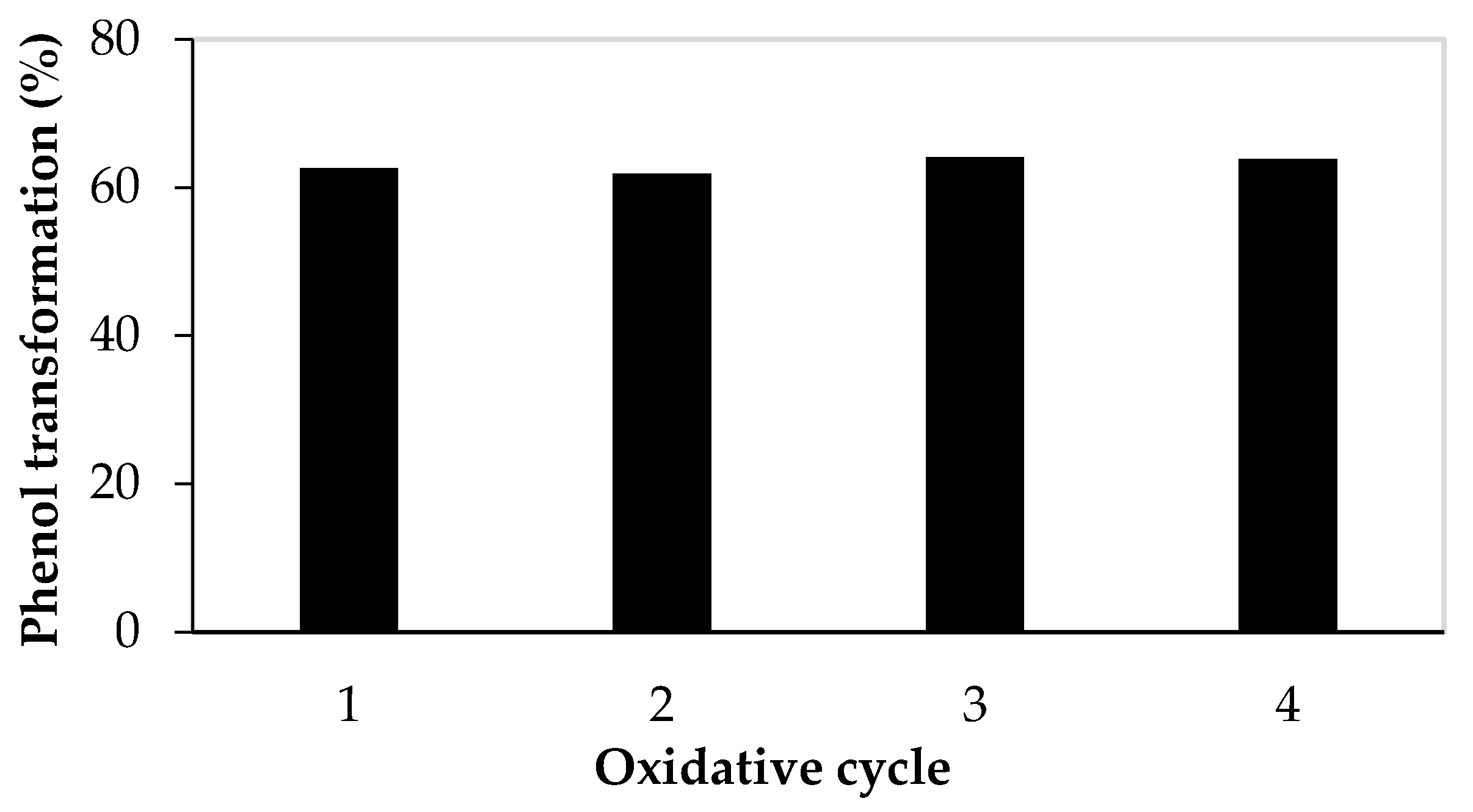
3.3. Sequential Batch Biotransformation of Phenol by Laccase Immobilized onto Silica-Coated Magnetic Nanoparticles
3.4. Environmental Indicators of the Single-Core Magnetic Nanoparticle
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kunamneni, A.; Ballesteros, A.; Plou, F.J.; Alcalde, M. Fungal laccase—A versatile enzyme for biotechnological applications. In Communicating Current Research and Educational Topics and Trends in Applied Microbiology; Mendez-Vilas, A., Ed.; Formex: Badajoz, Spain, 2007; Volume 1, pp. 233–245. ISBN 978-84-611-9422-3. [Google Scholar]
- Thurston, C.F. The structure and function of fungal laccases. Microbiology 1994, 140, 19–26. [Google Scholar] [CrossRef]
- Wesenberg, D.; Kyriakides, I.; Agathos, S.N. White-rot fungi and their enzymes for the treatment of industrial dye effluents. Biotechnol. Adv. 2003, 22, 161–187. [Google Scholar] [CrossRef] [PubMed]
- Misson, M.; Zhang, H.; Jin, B. Nanobiocatalyst advancements and bioprocessing applications. J. R. Soc. Interface 2015, 12, 20140891. [Google Scholar] [CrossRef] [PubMed]
- Illanes, R. Enzyme Biocatalysis : Principles and Applications; Springer: Berlin, Germany, 2008. [Google Scholar]
- Hernandez, R.; Fernandez-Lafuente, J.M. Control of protein immobilization: Coupling immobilization and site-directed mutagenesis to improve biocatalyst or biosensor performance. Enzyme Microb. Technol. 2011, 48, 107–122. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, R.C.; Ortiz, C.; Berenguer-Murcia, A.; Torres, R.; Fernández-Lafuente, R. Modifying enzyme activity and selectivity by immobilization. Chem. Soc. Rev. 2013, 42, 6290–6307. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A. Enzyme Immobilization: The Quest for Optimum Performance. Adv. Synth. Catal. 2007, 349, 1289–1307. [Google Scholar] [CrossRef]
- Duran, N.; Rosa, M.A.; D´Annibale, A.; Gianfreda, L. Applications of laccases and tyrosinases(phenoloxidases) immobilized on different supports: A review. Enzyme Microb. Technol. 2002, 31, 907–931. [Google Scholar] [CrossRef]
- Cui, J.; Jia, S.; Liang, L.; Zhao, Y.; Feng, Y. Mesoporous CLEAs-silica composite microparticles with high activity and enhanced stability. Sci. Rep. 2015, 5, 14203. [Google Scholar] [CrossRef] [PubMed]
- Matijosyte, I.; Arends, I.W.C.E.; Vries, S.; Sheldon, R.A. Preparation and use of cross-linked enzyme aggregates (CLEAs) of laccases. J. Mol. Catal. B Enzyme 2010, 62, 142–148. [Google Scholar] [CrossRef]
- Qu, X.; Alvarez, P.J.J.; Li, Q. Applications of nanotechnology in water and wastewater treatment. Water Res. 2013, 47, 3931–3946. [Google Scholar] [CrossRef] [PubMed]
- Ahn, M.-Y.; Zimmerman, A.R.; Martinez, C.E.; Archibald, D.D.; Bollag, J.-M.; Dec, J. Characteristics of Trametes villosa laccase adsorbed on aluminium hydroxide. Enzyme Microb. Technol. 2007, 41, 141–148. [Google Scholar] [CrossRef]
- Qiu, H.; Xu, C.; Huang, X.; Ding, Y.; Qu, Y.; Gao, P. Adsorption of laccase on the surface of nanoporous gold and the electron transfer between them. J. Phys. Chem. C 2008, 112, 14781–14785. [Google Scholar] [CrossRef]
- Fernandez-Fernandez, M.; Sanroman, M.A.; Moldes, D. Recent developments and applications of immobilized laccase. Biotechnol. Adv. 2013, 31, 1808–1825. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, M. Immobilized enzymes: Theory, methods of study and applications. ARS Pharm. 1998, 39, 23–39. [Google Scholar]
- Brady, D.; Jordaan, J. Advances in enzyme immobilisation. Biotechnol. Lett. 2009, 31, 1639–1650. [Google Scholar] [CrossRef] [PubMed]
- García-Galán, C.; Berenguer-Murcia, A.; Fernandez-Lafuente, R.; Rodrigues, R.C. Potential of Different Enzyme Immobilization Strategies to Improve Enzyme Performance. Adv. Synth. Catal. 2011, 353, 2885–2904. [Google Scholar] [CrossRef]
- Mazur, M.; Krysiński, P.; Michota-Kamińska, A.; Bukowska, J.; Rogalski, J.; Blanchard, G.J. Immobilization of laccase on gold, silver and indium tin oxide by zirconium–phosphonate–carboxylate (ZPC) coordination chemistry. Bioelectrochemistry 2007, 71, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Kalkan, N.; Aksoy, S.; Aksoy, E.; Hasirci, N. Preparation of chitosan-coated magnetite nanoparticles and application for immobilization of laccase. J. Appl. Polym. Sci. 2011, 123, 707–716. [Google Scholar] [CrossRef]
- Feng, W.; Ji, P. Enzymes immobilized on carbon nanotubes. Biotechnol. Adv. 2011, 29, 889–895. [Google Scholar] [CrossRef] [PubMed]
- DiCosimo, R.; McAuliffe, J.; Polouse, A.J.; Bohlmann, G. Industrial use of immobilized enzymes. Chem. Soc. Rev. 2013, 42, 6437–6474. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, Y.S.; Shahgaldian, P.; Corvini, P.F.X.; Hommes, G. Sorption-assisted surface conjugation: A way to stabilize laccase enzyme. Appl. Microbiol. Biotechnol. 2011, 92, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Nobs, L.; Buchegger, F.; Gurny, R.; Allemann, E. Surface modification of poly(lactic acid) nanoparticles by covalent attachment of thiol groups by means of three methods. Int. J. Pharm. 2003, 29, 327–337. [Google Scholar] [CrossRef]
- Barbosa, O.; Ortiz, C.; Berenguer-Murcia, A. Glutaraldehyde in bio-catalysts design: A useful crosslinker and a versatile tool in enzyme immobilization. RSC Adv. 2014, 4, 1583–1600. [Google Scholar] [CrossRef]
- Zucca, P.; Sanjust, E. Inorganic materials as supports for covalent enzyme immobilization: Methods and mechanisms. Molecules 2014, 19, 14139–14194. [Google Scholar] [CrossRef] [PubMed]
- Baumann, H.; Tillman, A.M. The Hitch Hilker’s Guide to LCA: An Orientation in Life Cycle Assessment Methodology and Application; Studentlitteratur: Lund, Sweden, 2004; ISBN 9144023642. [Google Scholar]
- Environmental Management—Life Cycle Assessment—Principles and Framework; ISO 14040:2006; International Organization for Standardization: Geneva, Switzerland, 2006.
- Feijoo, S.; Gonzalez-García, S.; Moldes-Diz, Y.; Vazquez-Vazquez, C.; Feijoo, G.; Moreira, M.T. Comparative life cycle assessment of different synthesis routes of magnetic nanoparticles. J. Clean. Prod. 2017, 43, 528–538. [Google Scholar] [CrossRef]
- Dones, R.; Bauer, C.; Bolliger, R.; Burger, B.; Faist, E.M.; Frischknecht, R.; Heck, T.; Jungbluth, N.; Röder, A.; Tuchschmid, M. Life Cycle Inventories of Energy Systems: Results for Current Systems in Switzerland and Other UCTE Countries; Ecoinvent Report No. 5; Swiss Centre for Life Cycle Inventories: Düberdorf, Switzerland, 2007; Available online: https://www.researchgate.net/profile/Deborah_Andrews2/publication/271710820_The_life_cycle_assessment_of_a_UK_data_centre/links/5620b9ec08aed8dd194054ea.pdf (accessed on 7 June 2016).
- Althaus, H.J.; Chudacoff, M.; Hischier, R.; Jungbluth, N.; Osses, M.; Primas, A. Life Cycle Inventories of Chemicals; Ecoinvent Report No.8 v2.0 EMPA; Swiss Centre for Life Cycle Inventories: Düberdorf, Switzerland, 2007. [Google Scholar]
- Doka, G. Life Cycle Inventories of Waste Treatment Services; Ecoinvent Report No. 13; Swiss Centre for Life Cycle Inventories: Dübendorf, Switzerland, 2003. [Google Scholar]
- Red Eléctrica de España. Avance del Informe del Sistema Eléctrico Español 2014. Available online: http://www.ree.es/sites/default/files/downloadable/avance_informe_sistema_electrico_2014.pdf (accessed on 10 June 2016).
- Goedkoop, M.J.; Heijungs, R.; Huijbregts, M.; Schryver, A.D.; Strujis, J.; Zelm, R. A Life Cycle Impact Assessment Method Which Comprises Harmonised Category Indicators at the Midpoint and the Endpoint Level. Available online: http://www.leidenuniv.nl/cml/ssp/publications/recipe_characterisation.pdf (accessed on 7 June 2016).
- PRé Consultants. 2017. Available online: http://www.pre.nl (accessed on 1 July 2017).
- Sheldon, R.A. Cross-linked enzyme aggregates (CLEAs): Stable and recyclable biocatalysts. Biochem. Soc. Trans. 2007, 35, 1583–1587. [Google Scholar] [CrossRef] [PubMed]
- Ovsejevi, K.; Manta, C.; Batista-Viera, F. Reversible covalent immobilization of enzymes via disulfide bonds. Methods Mol. Biol. 2013, 1051, 89–116. [Google Scholar] [CrossRef] [PubMed]
- Guisán, J.M. Immobilization of Enzymes and Cells, 2nd ed.; Humana Press Inc.: Clifton, NJ, USA, 2006; ISBN 978-1-58829-290-2. [Google Scholar]
- Flickinger, M.; Drew, S. Encyclopedia of Bioprocess Technology: Fermentation, Biocatalysis and Bioseparation Fermentation, Biocatalysis and Bioseparation; Wiley: New York, NY, USA, 1999; ISBN 0-471-13822-3. [Google Scholar]
- Majumder, A.B.; Mondal, K.; Singh, T.P.; Gupta, M.N. Designing cross-linked lipase aggregates for optimum performance as biocatalysts. Biocatal. Biotransform. 2008, 26, 235–242. [Google Scholar] [CrossRef]
- Kumar, S.; Jana, A.K.; Maiti, M.; Dhamija, I. Carbodiimide-mediated immobilization of serratiopeptidase on amino-, carboxyl-functionalized magnetic nanoparticles and characterization for target delivery. J. Nanopart. Res. 2014, 16, 2233. [Google Scholar] [CrossRef]
- Arca-Ramos, A.; Ammann, E.M.; Gasser, C.A.; Nastold, P.; Eibes, G.; Feijoo, G.; Lema, J.M.; Moreira, M.T.; Corvini, P.F.X. Assessing the use of nanoimmobilized laccases to remove micropollutants from wastewater. Environ. Sci. Pollut. Res. 2016, 23, 3217–3228. [Google Scholar] [CrossRef] [PubMed]
- Corgié, S.; Kahawong, P.; Duan, X.; Bowser, D.; Edward, J.B.; Walker, L.P.; Giannelis, E.P. Self-assembled complexes of horseradish peroxidase with magnetic nanoparticles showing enhanced peroxidase activity. Adv. Funct. Mater. 2012, 22, 1940–1951. [Google Scholar] [CrossRef]
- Claus, H.; Faber, G.; König, H. Redox-mediated decolorization of synthetic dyes by fungal laccases. Appl. Microbiol. Biotechnol. 2002, 59, 672–678. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Hu, Y.; Guo, C.; Huang, W.; Liu, C.-Z. Enhanced phenol degradation in cooking wastewater by immobilized laccase on magnetic mesoporous silica nanoparticles in a magnetically stabilized fluidized bed. Bioresour. Technol. 2012, 110, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Kurniawati, S.; Nicell, J.A. A Comprehensive Kinetic Model of Laccase-Catalyzed Oxidation of Aqueous Phenol. Biotechnol. Prog. 2009, 25, 763–773. [Google Scholar] [CrossRef] [PubMed]
- Bayramoglu, G.; Arica, M.Y. Enzymatic removal of phenol and p-chlorophenol in enzyme reactor: Horseradish peroxidase immobilized on magnetic beads. Hazard. Mater. 2008, 156, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Grieger, K.D.; Laurent, A.; Miseljic, M.; Christensen, F.; Baun, A.; Olsen, S.I. Analysis of current research addressing complementary use of life-cycle assessment and risk assessment for engineered nanomaterials: Have lessons been learned from previous experience with chemicals? J. Nanopart. Res. 2012, 14, 958. [Google Scholar] [CrossRef]

| Type of Nanoparticles | Size (nm) | Concentration (mg NPs mL−1) |
|---|---|---|
| Fumed silica nanoparticles (fsNP) | 7 | 59 |
| Silica-coated magnetic nanoparticles | ||
| FeO-2206W (single-core) | 21.5 ± 2.1 | 5 |
| S-57 (multi-core) | 11.8 ± 2.4 | 10.9 |
| Polyacrylic acid (PAA) magnetic nanoparticles | ||
| FeO-2204W | 10.1 ± 2.4 | 20.5 |
| FeO-36 | 23.1 ± 4.9 | 16.2 |
| Polyethylenimine (PEI) magnetic nanoparticles | ||
| VOZ-19 | 10 ± 1.2 | 56 |
| Non-coated magnetite nanoparticles | 9.9 ± 1.4 | 17.4 |
| Different Types of Nanoparticles | Washing Loss (%) | Activity Yield (%) | Enzyme Loading (U mg−1 NPs) |
|---|---|---|---|
| Covalent immobilization | |||
| Fumed silica nanoparticles (fsNP) | 5.6 ± 1.3 | 100 ± 6.1 | 1.78 ± 0.07 |
| Silica-coated magnetic nanoparticles | |||
| FeO-2206W (single-core) | 16.4 ± 2.81 | 99.7 ± 0.35 | 2.66 ± 0.65 |
| S-57 (multi-core) | 66.63 ± 1.67 | 31.3 ± 0.76 | 0.42 ± 0.05 |
| Polyacrylic acid (PAA) magnetic nanoparticles | |||
| FeO-2204W | 96.83 ± 1.4 | 2.55 ± 6.5 | 0.11 ± 0.34 |
| FeO-36 | 99.7 ± 2.34 | 0.12 ± 2.3 | 0.01 ± 0.24 |
| Polyethylenimine (PEI) magnetic nanoparticles | |||
| VOZ-19 | 27.18 ± 0.08 | 80.5 ± 0.21 | 1.54 ± 0.03 |
| Ionic exchange immobilization | |||
| Non-coated magnetite nanoparticles | 45.3 ± 0.9 | 58.5 ± 1.5 | 0.69 ± 0.05 |
| pH | Phenol Biotransformation (%) | Biotransformation Rate (mg L−1 h−1) |
|---|---|---|
| 5 | 67.9 | 0.383 |
| 6 | 63.9 | 0.326 |
| 7 | 48.1 | 0.189 |
| Scenarios | ||
|---|---|---|
| Impact Category | Silica-Coated mNPs | Silica Thin Shell |
| Climate change | 1.43 × 10−4 | 1.03 × 10−4 |
| Ozone depletion | 3.69 × 10−6 | 2.42 × 10−6 |
| Terrestrial acidification | 1.83 × 10−4 | 1.30 × 10−4 |
| Freshwater eutrophication | 9.11 × 10−4 | 5.42 × 10−4 |
| Marine eutrophication | 2.12 × 10−5 | 1.45 × 10−5 |
| Human toxicity | 5.93 × 10−4 | 3.98 × 10−4 |
| Photochemical oxidant formation | 1.33 × 10−4 | 9.05 × 10−5 |
| Terrestrial ecotoxicity | 6.18 × 10−6 | 4.37 × 10−6 |
| Freshwater ecotoxicity | 1.22 × 10−3 | 8.36 × 10−4 |
| Marine ecotoxicity | 1.45 × 10−3 | 9.84 × 10−4 |
| Fossil depletion | 6.20 × 10−4 | 5.18 × 10−4 |
| Normalized index | 5.28 × 10−3 | 3.62 × 10−3 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreira, M.T.; Moldes-Diz, Y.; Feijoo, S.; Eibes, G.; Lema, J.M.; Feijoo, G. Formulation of Laccase Nanobiocatalysts Based on Ionic and Covalent Interactions for the Enhanced Oxidation of Phenolic Compounds. Appl. Sci. 2017, 7, 851. https://doi.org/10.3390/app7080851
Moreira MT, Moldes-Diz Y, Feijoo S, Eibes G, Lema JM, Feijoo G. Formulation of Laccase Nanobiocatalysts Based on Ionic and Covalent Interactions for the Enhanced Oxidation of Phenolic Compounds. Applied Sciences. 2017; 7(8):851. https://doi.org/10.3390/app7080851
Chicago/Turabian StyleMoreira, Maria Teresa, Yolanda Moldes-Diz, Sara Feijoo, Gemma Eibes, Juan M. Lema, and Gumersindo Feijoo. 2017. "Formulation of Laccase Nanobiocatalysts Based on Ionic and Covalent Interactions for the Enhanced Oxidation of Phenolic Compounds" Applied Sciences 7, no. 8: 851. https://doi.org/10.3390/app7080851
APA StyleMoreira, M. T., Moldes-Diz, Y., Feijoo, S., Eibes, G., Lema, J. M., & Feijoo, G. (2017). Formulation of Laccase Nanobiocatalysts Based on Ionic and Covalent Interactions for the Enhanced Oxidation of Phenolic Compounds. Applied Sciences, 7(8), 851. https://doi.org/10.3390/app7080851






