Mechanical Behavior and Fracture Properties of NiAl Intermetallic Alloy with Different Copper Contents
Abstract
:1. Introduction
2. Material Preparation and Experimental Procedure
3. Results and Discussion
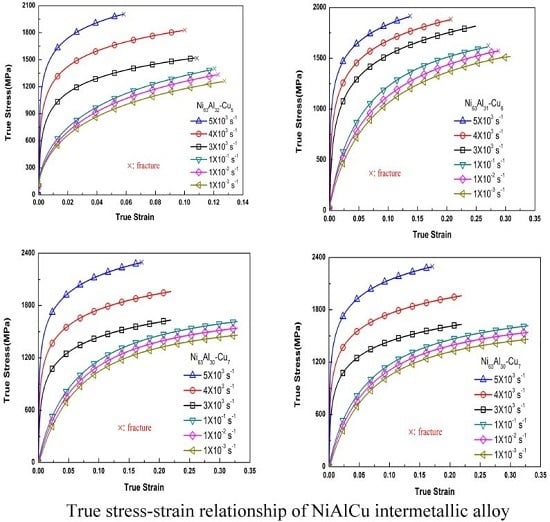
3.1. Stress–Strain Response
3.2. Strain Rate Effect
3.3. Microstructural Observations and Fracture Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Miracle, D.B. Overview No. 104. The physical and mechanical properties of NiAl. Acta Metall. Mater. 1993, 41, 649–684. [Google Scholar] [CrossRef]
- Zhao, H.L.; Qiu, F.; Jin, S.B.; Jiang, Q.C. High work-hardening effect of the pure NiAl intermetallic compound fabricated by the combustion synthesis and hot pressing technique. Mater. Lett. 2011, 65, 2604–2606. [Google Scholar] [CrossRef]
- Ozdemir, O.; Zeytin, S.; Bindal, C. Characterization of NiAl with cobalt produced by combustion synthesis. J. Alloys Compd. 2010, 508, 216–221. [Google Scholar] [CrossRef]
- Noebe, R.D.; Bowman, R.R.; Nathal, M.V. Physical and mechanical properties of the B2 compound NiAl. Int. Mater. Rev. 1993, 38, 193–232. [Google Scholar] [CrossRef]
- Liu, E.; Jia, J.; Bai, Y.; Wang, W.; Gao, Y. Study on preparation and mechanical property of nanocrystalline NiAl intermetallic. Mater. Des. 2014, 53, 596–601. [Google Scholar] [CrossRef]
- Zhou, Y.J.; Zhang, Y.; Wang, Y.L.; Chen, G.L. Solid solution alloys of AlCoCrFeNiTix with excellent room-temperature mechanical properties. Appl. Phys. Lett. 2007, 90, 181904-1–181904-3. [Google Scholar] [CrossRef]
- Zhu, J.M.; Fu, H.M.; Zhang, H.F.; Wang, A.M.; Li, H.; Hu, Z.Q. Microstructures and compressive properties of multicomponent AlCoCrFeNiMox alloys. Mater. Sci. Eng. A 2010, 527, 6975–6979. [Google Scholar] [CrossRef]
- Chuang, M.H.; Tsai, M.H.; Wang, W.R.; Lin, S.J.; Yeh, J.W. Microstructure and wear behavior of AlxCo1.5CrFeNi1.5Tiy high-entropy alloys. Acta Mater. 2011, 59, 6308–6317. [Google Scholar] [CrossRef]
- Lee, J.W.; Hyun, S.K.; Kim, M.S.; Kim, M.G.; Ide, T.; Nakajima, H. Ductility improvement of intermetallic compound NiAl by unidirectional pores. Mater. Lett. 2012, 74, 213–216. [Google Scholar] [CrossRef]
- Stephens, J.R. High Temperature Ordered Intermetallic Alloys. In Proceedings of the MRS Symp., Boston, MA, USA, 26 November 1985; Koch, C.C., Liu, C.T., Stoloff, N.S., Eds.; Materials Research Society: Warrendale, PA, USA, 1985; Volume 39, p. 381. [Google Scholar]
- Sheng, L.Y.; Guo, J.T.; Ye, H.Q. Microstructure and mechanical properties of NiAl–Cr(Mo)/Nb eutectic alloy prepared by injection-casting. Mater. Des. 2009, 30, 964–969. [Google Scholar] [CrossRef]
- Ferrandini, P.; Batista, W.W.; Caram, R. Influence of growth rate on the microstructure and mechanical behaviour of a NiAl–Mo eutectic alloy. J. Alloy. Compd. 2004, 381, 91–98. [Google Scholar] [CrossRef]
- Ishida, K.; Kainuma, R.; Ueno, N.; Nishizawa, T. Ductility enhancement in NiAl (B2)-base alloys by microstructural control. Metall. Trans. A 1991, 22A, 441–446. [Google Scholar] [CrossRef]
- Colín, J.; Serna, S.; Campillo, B.; Flores, O.; Juárez-Islas, J. Microstructural and lattice parameter study of as-cast and rapidly solidified NiAl intermetallic alloys with Cu additions. Intermetallics 2008, 16, 847–853. [Google Scholar] [CrossRef]
- Zhou, W.L.; Guo, J.T.; Chen, R.S.; Li, G.S.; Zhou, J.Y. Superplasticity in NiAl intermetallics macroalloyed with iron. Mater. Lett. 2001, 47, 30–34. [Google Scholar] [CrossRef]
- Duman, N.; Mekhrabov, A.O.; Akdeniz, M.V. Microalloying effects on the microstructure and kinetics of nanoscale precipitation in NiAlFe alloy. Intermetallics 2012, 23, 217–227. [Google Scholar] [CrossRef]
- Sheng, L.; Zhang, W.; Guo, J.; Yang, F.; Liang, Y.; Ye, H. Effect of Au addition on the microstructure and mechanical properties of NiAl intermetallic compound. Intermetallics 2010, 18, 740–744. [Google Scholar] [CrossRef]
- Jeon, S.J.; Lee, H.C. Effect of copper alloying on the deformation behavior of B2 NiA1 intermetallic compounds. Mater. Sci. Eng. A 1992, 153, 392–397. [Google Scholar] [CrossRef]
- Colin, J.; Gonzalez, C.; Herrera, R.; Juarez-Islas, J.A. Analysis of chill-cast NiAl intermetallic compound with copper additions. J. Mater. Eng. Perform. 2002, 11, 487–491. [Google Scholar] [CrossRef]
- Kainuma, R.; Imano, S.; Otani, H.; Ishida, K. Microstructural evolution in ductile Β(B2) + γ’(L12) Ni–Al–Fe alloys. Intermetallics 1996, 4, 37–45. [Google Scholar] [CrossRef]
- Noebe, R.D.; Cullers, C.L.; Bowman, R.R. The effect of strain rate and temperature on the tensile properties of NiAl. J. Mater. Res. 1992, 7, 605–612. [Google Scholar] [CrossRef]
- Moskovic, R. Mechanical properties of precipitation-strengthened Ni–Al–Cr alloy based on an NiAl intermetallic compound. J. Mater. Sci. 1978, 13, 1901–1906. [Google Scholar] [CrossRef]
- Busso, E.P.; McClintock, F.A. A dislocation mechanics-based crystallographic model of a B2-type intermetallic alloy. Inter. J. Plast. 1996, 12, 1–28. [Google Scholar] [CrossRef]
- Wright, T.W.; Batra, R.C. The initiation and growth of adiabatic shear bands. Inter. J. Plast. 1985, 1, 205–212. [Google Scholar] [CrossRef]
- Marchand, A.; Duffy, J. An experimental study of the formation process of adiabatic shear bands in a structural steel. J. Mech. Phys. Solids 1988, 36, 251–283. [Google Scholar] [CrossRef]
- Lee, W.S.; Liu, C.Y.; Chen, T.H. Adiabatic shearing behavior of different steels under extreme high shear loading. J. Nucl. Mater. 2008, 374, 313–319. [Google Scholar] [CrossRef]
- Philpot, K.A.; Munir, Z.A.; Holt, J.B. An investigation of the synthesis of nickel aluminides through gasless combustion. J. Mater. Sci. 1987, 22, 159–169. [Google Scholar] [CrossRef]
- Liu, C.T.; Stiegler, J.Q. ASM Handbook; ASM International: Novelty, OH, USA, 1990; Volume 11, p. 919. [Google Scholar]
- Egg, S.S.; Sue, Y.Q.; Hirsch, P.B. Strain rate dependence of the flow stress and work hardening of γ’. Mater. Sci. Eng. A 1995, 192–193, 45–52. [Google Scholar]
- Lee, W.S.; Chen, C.W. Dynamic mechanical properties and microstructure of Ti–6Al–7Nb biomedical alloy as function of strain rate. Mater. Sci. Technol. 2013, 9, 1055–1064. [Google Scholar] [CrossRef]
- Chen, T.H.; Hsu, Y.K.; Fang, T.E. The deformational behaviour and microstructural evolution of Al–Sc–Zr alloys with different copper content under high strain rate impact loading. J. Chin. Soc. Mech. Eng. 2014, 35, 499–506. [Google Scholar]
- Lee, W.S.; Lin, C.F. High-temperature deformation behaviour of Ti6Al4V alloy evaluated by high strain-rate compression tests. J. Mater. Process. Technol. 1998, 75, 127–136. [Google Scholar] [CrossRef]
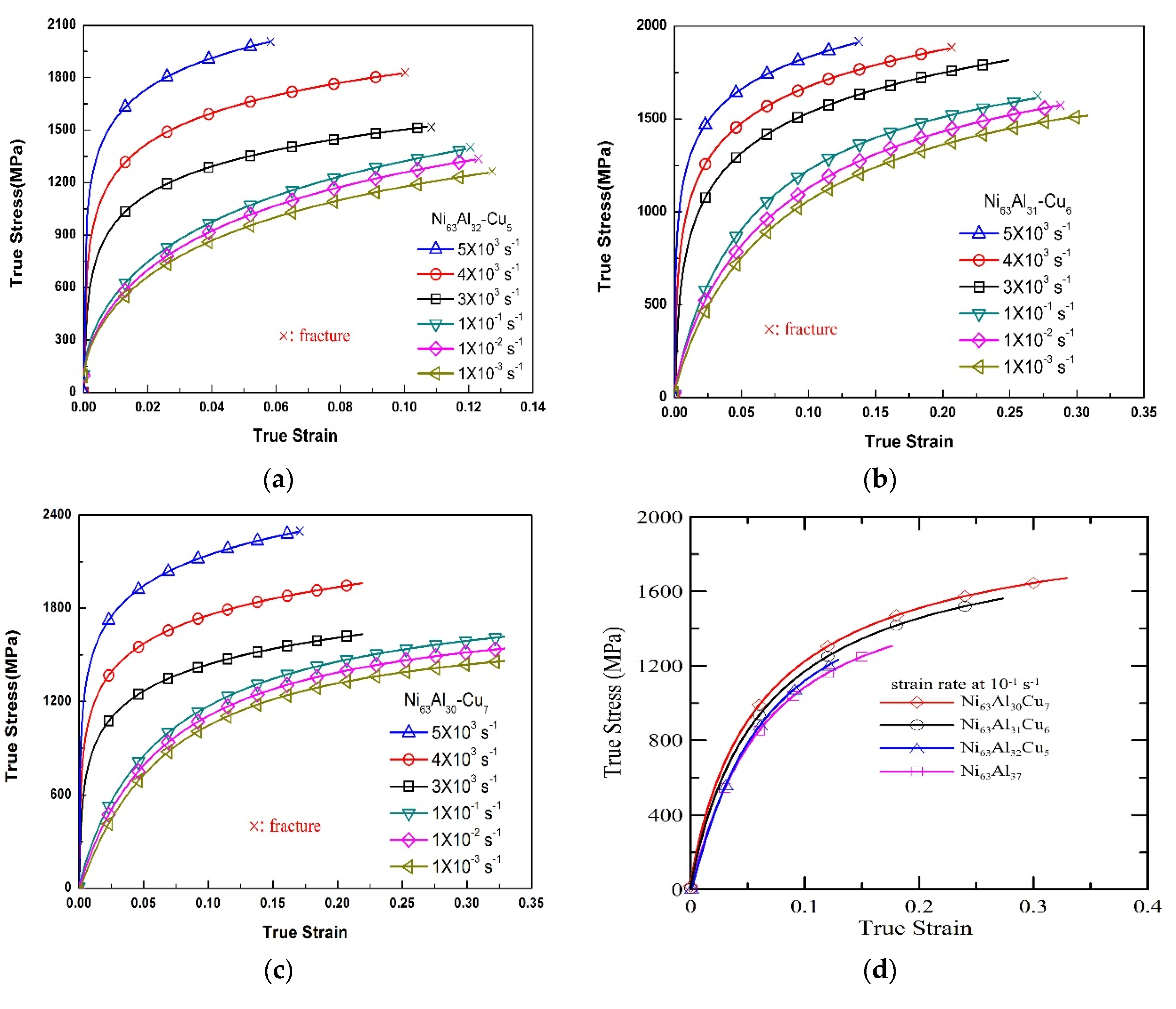
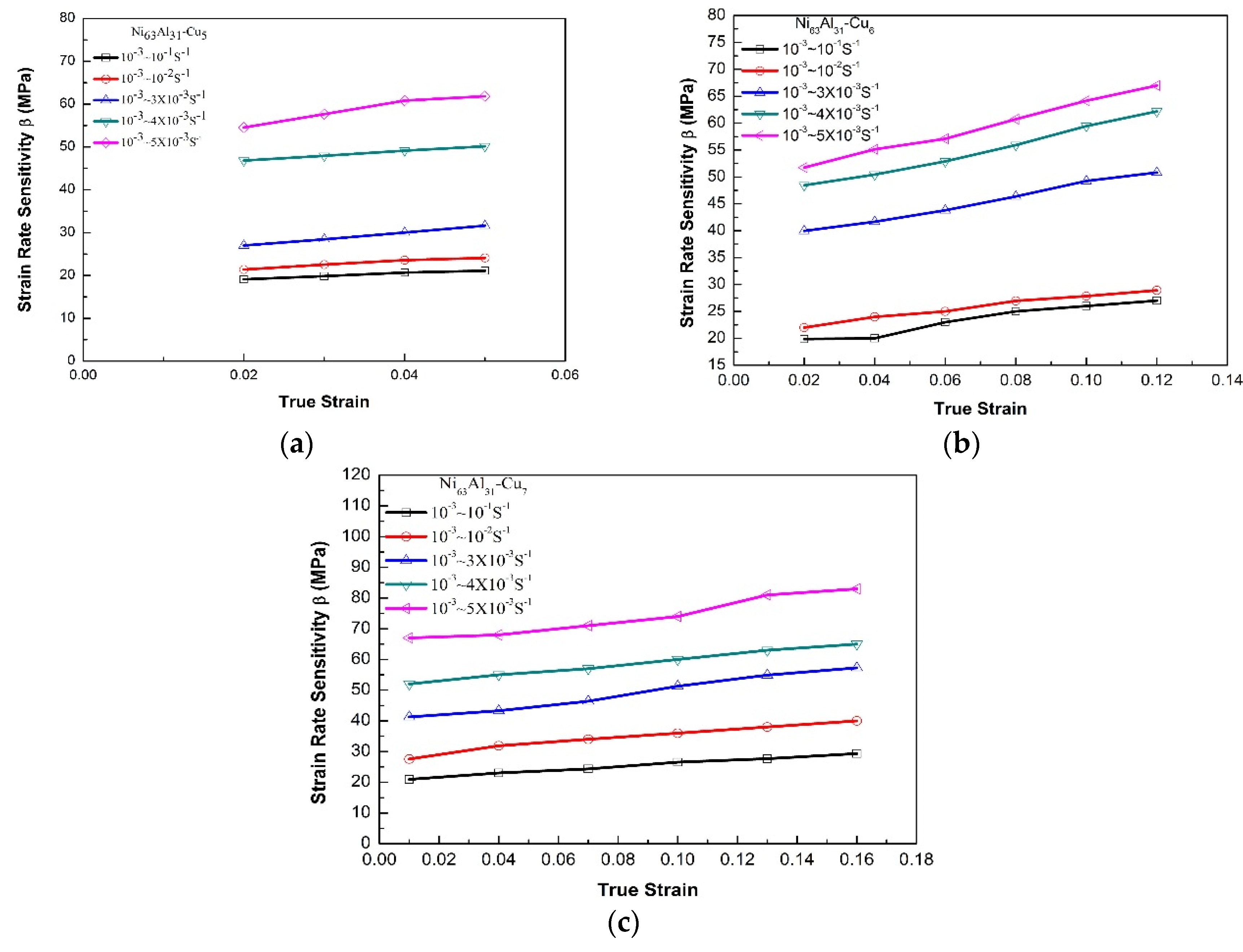

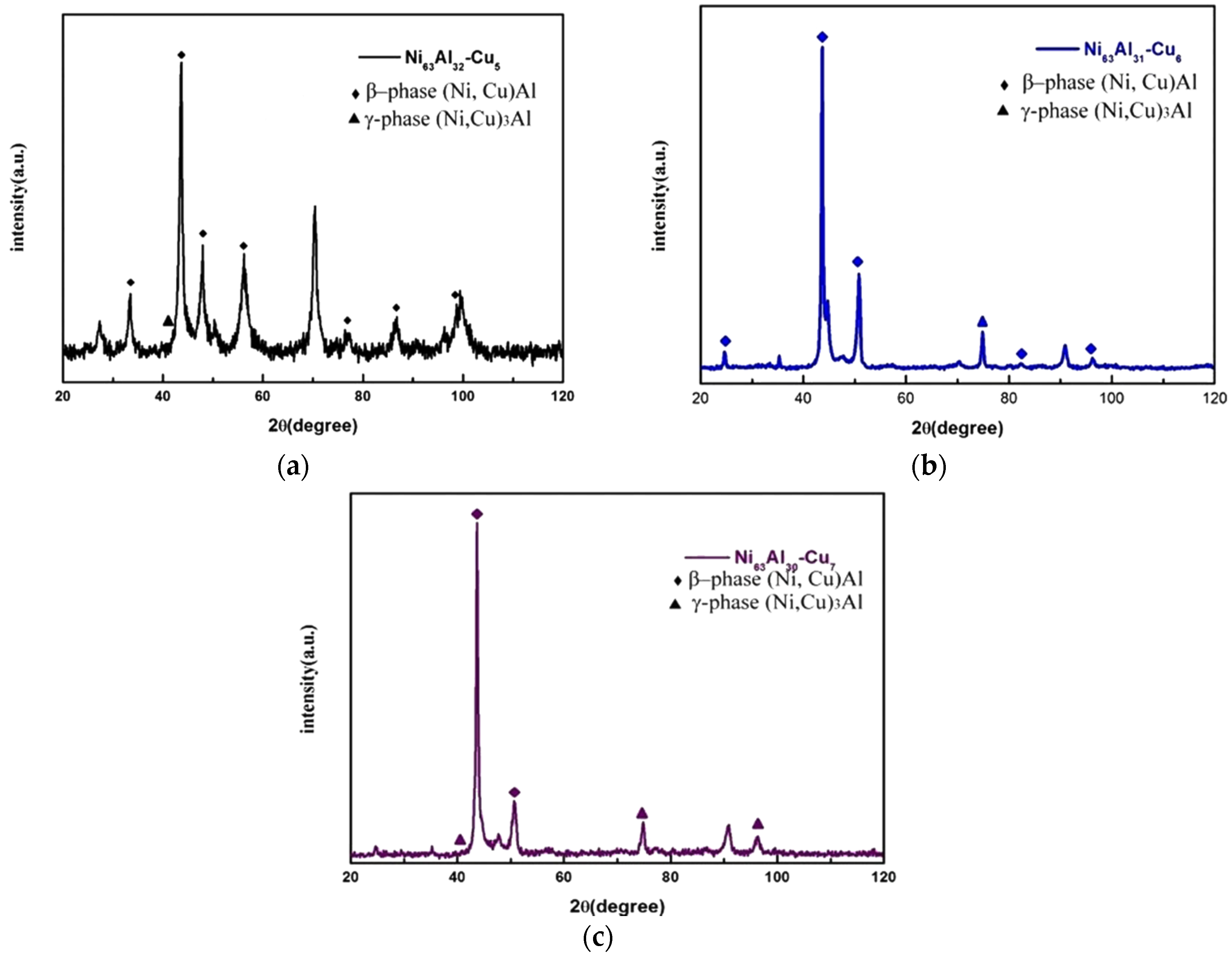

| Alloy | Strain Rate (s−1) | Yielding Strength, A (MPa) | Material Constant, B (MPa) | Work Hardening Coefficient, n |
|---|---|---|---|---|
| Ni63Al32Cu5 | 10−3 | 449.3 | 3056.6 | 0.62 |
| 10−2 | 475.9 | 3410.9 | 0.64 | |
| 10−1 | 520.67 | 3647.8 | 0.65 | |
| 3 × 103 | 925.8 | 2257.9 | 0.68 | |
| 4 × 103 | 1205.86 | 2626.8 | 0.72 | |
| 5 × 103 | 1562.66 | 2918.2 | 0.77 | |
| Ni63Al32Cu6 | 10−3 | 571.3 | 2072.8 | 0.63 |
| 10−2 | 656.57 | 2203.3 | 0.66 | |
| 10−1 | 771.74 | 2162.3 | 0.67 | |
| 3 × 103 | 1073.07 | 1802.4 | 0.7 | |
| 4 × 103 | 1236.94 | 1884.8 | 0.73 | |
| 5 × 103 | 1331.2 | 2171.2 | 0.78 | |
| Ni63Al32Cu7 | 10−3 | 695.5 | 1628.0 | 0.65 |
| 10−2 | 750.7 | 1697.4 | 0.68 | |
| 10−1 | 809.0 | 1744.0 | 0.70 | |
| 3 × 103 | 1048.6 | 1563.0 | 0.71 | |
| 4 × 103 | 1289.6 | 1661.6 | 0.75 | |
| 5 × 103 | 1676.5 | 2190.0 | 0.80 |
| Alloy | Average Hardness Value |
|---|---|
| Ni63Al32-Cu5 | 301.16(Hv) |
| Ni63Al31-Cu6 | 325.63(Hv) |
| Ni63Al30-Cu7 | 353.9(Hv) |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, T.-H.; Wu, J.-H. Mechanical Behavior and Fracture Properties of NiAl Intermetallic Alloy with Different Copper Contents. Appl. Sci. 2016, 6, 70. https://doi.org/10.3390/app6030070
Chen T-H, Wu J-H. Mechanical Behavior and Fracture Properties of NiAl Intermetallic Alloy with Different Copper Contents. Applied Sciences. 2016; 6(3):70. https://doi.org/10.3390/app6030070
Chicago/Turabian StyleChen, Tao-Hsing, and Jian-Hong Wu. 2016. "Mechanical Behavior and Fracture Properties of NiAl Intermetallic Alloy with Different Copper Contents" Applied Sciences 6, no. 3: 70. https://doi.org/10.3390/app6030070
APA StyleChen, T.-H., & Wu, J.-H. (2016). Mechanical Behavior and Fracture Properties of NiAl Intermetallic Alloy with Different Copper Contents. Applied Sciences, 6(3), 70. https://doi.org/10.3390/app6030070