1. Introduction
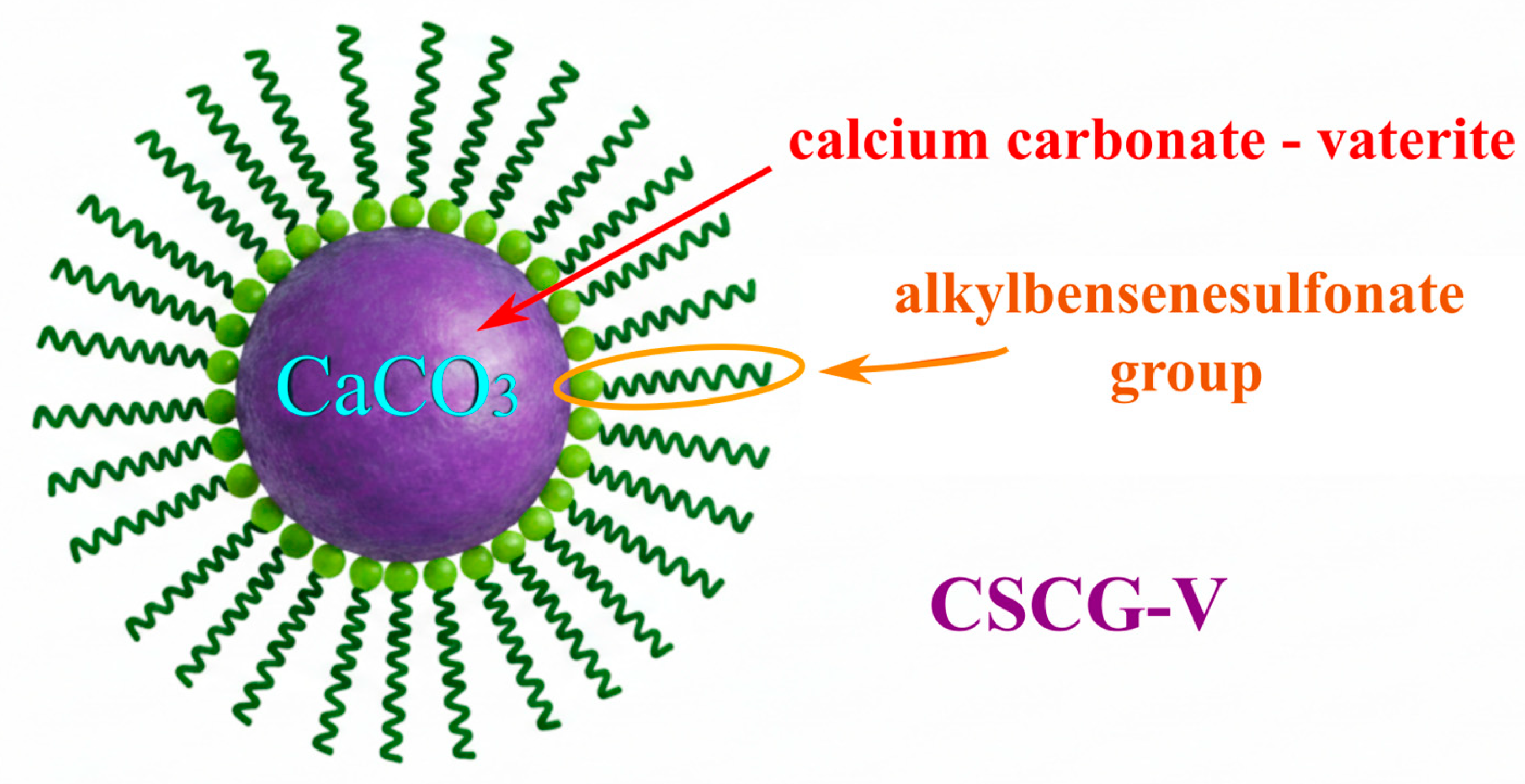
A grease based on the overbased calcium sulfonate is a colloidal system in which the dispersion medium is oil and the dispersed phase is calcium carbonate, stabilized by the surfactant calcium sulfonate. The core of the overbased calcium sulfonate molecule is amorphous calcium carbonate, which is surrounded by polar surfactant molecules. The hydrophobic surfactant chains, in turn, stabilize the colloid molecule in the oil [
1].
Figure 1 shows the micellar structure of calcium carbonate.
Hydrophilic groups within the calcium sulfonate capture and retain water molecules, resulting in excellent resistance of the grease to water leaching. Thanks to this property, sulfonate lubricating greases are widely used in various industries [
2]. This property of the grease is due to the structure of the thickener, overbased calcium sulfonate, which is made up of micelles. The core consists of a calcium carbonate molecule surrounded by molecules of overbased calcium sulfonate, which stabilizes the micelle structure and is responsible for the desired properties. Overbased calcium sulfonates act as surfactants (detergents) that stabilize the micelle system via polar sulfonate groups and hydrophobic alkyl tails. The micellar structure itself, in turn, is formed by the interaction between the sulfonate group and the base oil, forming a stable dispersion of calcium carbonate particles.
Lubricating greases operate in a variety of environments and are exposed to a range of factors that negatively affect their performance and, therefore, their service life. One such factor is contact with water. In the case of greases used in bearings, water can cause a number of problems: bearing corrosion, reduction in the consistency (hardening) of the grease, grease leakage from the bearing (when the consistency is increased, i.e., when the grease softens), or excessive grease aging due to the lack of channels (microspaces in the grease structure) [
3,
4]. The presence of water in greases almost always has a detrimental effect, deteriorating their structure and their lubricating ability. Furthermore, water in the grease can affect the thickness of the lubricating film and cause leakage [
5]. The presence of water can have a significant effect on the rheological properties of the grease: the grease can harden, and, in addition, water can reduce the effectiveness of the functional additives (anti-wear, anti-seizure and anti-oxidation additives) due to hydrolysis of their chemical compounds [
6].
There is no comprehensive information on the performance of water-contaminated grease. However, studies have been conducted on the effect of water on lubricating oil [
7,
8,
9,
10,
11]. Many esters hydrolyze when exposed to water, and ester-based lubricating oils, due to their hygroscopic nature, are more susceptible to water absorption. These oils are capable of forming association groups with water through hydrogen bonds, in contrast to mineral and polyalphaolefin (PAO) oils, which are hydrophobic, exhibiting very low affinity for water [
7].
Similarly to oils, some greases work well in contact with water because they do not absorb it. In general, a grease shows high water resistance if its individual components do not react with water and are not water soluble [
12].
When machinery and equipment operate in humid environments or under variable temperature conditions, it is recommended to use greases based on sodium and lithium soap. Sodium grease absorbs large amounts of water, i.e., it emulsifies with water, but can soften to the point where it flows out of the bearing. The grease based on lithium soap, in turn, does not emulsify with water, which means that, with the right additives, it provides good corrosion protection [
13].
When higher amounts of water come into contact with lithium grease, it has been found to soften, losing its structural stability or consistency [
14].
Complex greases thickened with overbased calcium sulfonate can absorb relatively large amounts of water, up to 80% by weight, while retaining their structure [
15]. This is due to the very strong polar nature of the calcium sulfonate that surrounds the calcite core. The presence of functional additives, which are usually polar compounds, may further facilitate the absorption of large amounts of water in these greases.
In another study [
5], four types of greases were analyzed—lithium greases, lithium complex greases, polyurethane greases, and calcium complex greases based on sulfonates—to assess the effect of water on their rheological properties. Viscosity measurements over a wide range of shear rates were described. The flow point (i.e., the smallest shear force that needs to be applied for the grease to start moving) and measured the elastic and viscous moduli (G′ and G″), along with the loss tangent (tan δ), were examined to understand the contribution of the elastic and viscous components in the material. From these studies, it was found that the structure of lithium and polyurethane greases weakened in the presence of water, whereas complex greases based on calcium sulfonates showed increased resistance—their viscosity at zero shear rate, flow point, and elastic moduli increased with increasing water content, and the loss tangent decreased, indicating a more elastic nature of the material. It has been observed that water in greases based on calcium sulfonates stabilized the structure of the thickener, strengthening the micromolecular network and making them extremely resistant to wet environments compared to other types of greases [
5].
The next work [
16] describes typical properties of sulfonate grease contaminated with 0%, 5%, and 10% water. The influence of water on the tribological properties of complex greases based on calcium sulfonates (CSCG) containing different polymorphic structures of calcium carbonate—vaterite (CSCG-V) and calcite (CSCG-C)—was examined. The research included rheological measurements, including viscosity at zero shear rate and flow point, to assess the effect of water on the structure of the grease and its resistance to deformation. Tribological tests were then performed at 30 °C and 80 °C, measuring the coefficient of friction and surface wear to determine the behavior of the grease under dynamic shear and loading conditions. Based on the obtained results, it was found that the addition of water to CSCG-V increased the viscosity at zero shear rate and the flow point and strengthened the grease structure, whereas in CSCG-C, the presence of water improved the tribological properties, providing better wear protection and a lower coefficient of friction. Additional analysis by XPS revealed the presence of CaCO
3 and CaO in the protective films on the wear surfaces regardless of the presence of water. The results indicate that the type of the polymorphic structure of CaCO
3 determines whether water will have a beneficial or negative effect on the grease based on calcium sulfonate. In practice, this means that in applications exposed to water, such as the maritime industry or machinery operating in humid conditions, CSCG-C greases should provide higher durability and performance than CSCG-V.
The microstructure of complex greases based on calcium sulfonates and the effect of water on the rheological and structural properties of the greases were investigated [
17] using atomic force microscopy (AFM). Microscopic observations allowed identification of the basic structural elements of the grease—the fibrous and spheroidal aggregates that form a three-dimensional network, responsible for maintaining the cohesion of the grease and its resistance to mechanical deformation.
The water in the sulfonate grease structure comes in two main forms: one is restricted water adsorbed around the calcium carbonate, and the other is unrestricted free water between the soap molecules. The main mechanism of grease-water interaction is the formation of inverted micelles that absorb water within the grease matrix. As the water content increases, the micelles become larger, leading to an increase in viscosity and strengthening the internal structure of the grease. Until the amount of absorbed water starts to exceed approximately 50–60% by weight, the grease maintains cohesion and shows increased shear resistance. Once this threshold is exceeded, free water appears and is no longer absorbed by the micelles, leading to a destabilization of the structure, a weakening of cohesion, and a partial “loosening” of the fibrospheroid network. The authors also showed that the presence of water affected the rheological properties of the grease depending on the amount of water. In the low (5–10%) and moderate (20–30%) ranges of water concentrations, an increase in flow point and shear strength is observed, indicating a strengthening of the internal structure. At the same time, under conditions of high (50–60% of grease weight) water content, the structure of the grease becomes unstable, and the rheological properties deteriorate (flow point, elastic modulus (G’), steady-state shear viscosity and susceptibility to oil discharge from the grease increase). The study also highlighted the phenomenon of shear banding—the formation of shear bands within the grease during mechanical deformation. This means that the material does not deform uniformly throughout the volume, but local zones are produced where the deformations are concentrated. The appearance of these bands is strongly correlated with structural changes caused by water content and is related to the uneven distribution of stresses in the grease micronetwork [
16] (see
Figure 2).
Furthermore, research conducted by Paszkowski et al. showed that sulfonate greases exhibited significant deterioration in functional properties after mining operations. The increase in frictional resistance of the grease after mining was attributed mainly to contamination with mine water. Water absorbed by the grease can not only increase its stiffness but also significantly reduce the surface activity of calcium carbonate, which compromises the lubricating properties [
18].
3. Results and Discussion
The results obtained from the grease specimens are summarized in
Table 1.
Adding water to the grease resulted in a change in its consistency. The base grease (which did not contain any water) was characterised by a consistency grade of NLGI 1. The addition of 1% water did not change the consistency of the grease (NLGI 1, like the base grease), but the incorporation of a higher water content caused the grease to harden by 1 consistency grade (NLGI 2) for 5–10% water and by 1.5 grades (NLGI 2/3) for 20–50% water.
The incorporation of water altered the grease’s dropping point. The dropping point decreased along with an increase in the amount of water. With the addition of 1% water, no change was observed; at 5%, there was a small change (within the uncertainty limit of the test method); and with the addition of higher water contents, a decrease of 10–16% was observed.
Mechanical stress via the prolonged worked penetration of grease causes it to soften slightly, increasing its consistency by a few units (up to half an NLGI grade); grease containing 50% water is just as resistant as grease that does not contain any water.
During the thermal oxidation stability test, the RSSOT (according to ASTM D8206, at 160 °C), the level of resistance to oxidation also decreased as the water content in the grease increased. For the sample containing 1% water, a slight decrease in oxidation resistance was found in comparison with the grease that did not contain any water. For samples containing 5–40% water, the decrease in resistance was similar but greater than that for a specimen that contained 1% water. For grease that contained 50% water, a significant decrease in oxidation resistance was observed.
In the thermal oxidation stability test, the RSSOT-PWP (according to ASTM D8206, at 160 °C, for grease samples subjected to mechanical stress by 100,000 double strokes), the level of resistance to oxidation also decreased as the water content in the grease increased. For the sample that contained 1% water, there was a decrease in oxidation resistance, expressed as a drop in oxygen pressure, which was one-half higher than that for the grease that did not contain any water, but the drop was two times higher for the grease that contained 50% water.
During the thermal oxidation stability test, the RSSOT-M (according to ASTM D8206, at 100 °C/100 h), the level of resistance to oxidation also decreased as the water content in the grease increased. For the sample that contained 1% water, a slight decrease in oxidation resistance was found in comparison with the grease that did not contain any water but was subjected to loading.
As the water content in the grease increased, there was a decrease in oxidation resistance, according to the ASTM D942 (at 100 °C for 100 h). For the sample containing 1–50% water, there was a decrease in oxygen pressure that was 3 to 4 times higher than that for the grease that did not contain any water.
The behaviour of the greases under similar conditions was compared. Thermal oxidation stability at 100 °C for 100 h was tested using two methods: conventional (acc. to ASTM D942) and modified RSSOT-M (acc. to ASTM D8206).
Figure 4 shows the percentage of oxygen pressure drop for both of these tests; the difference in the oxygen pressure drop percentage between the methods was the greatest for the grease sample that did not contain any water.
In order to assess the chemical changes that occurred during the thermal oxidation stability tests of lubricating grease with different water contents using different methods, infrared spectral analysis (FTIR) was performed; the spectra of the fresh grease and grease after testing were recorded. Due to the presence of strong bands in the spectrum being associated with the presence of calcium sulfonates and water, the spectra of the base grease and samples with added water were recorded using the ATR technique in the full range of 4000–550 cm
−1.
Figure 5 shows the spectra of the base grease and greases with 1%, 5%, 10%, 20%, 30%, 40%, and 50% water added. The main bands associated with sulfonate structures, apart from hydrocarbon bands in the range of 3000–2850 cm
−1, are bands at 1405, 1189, 1140, 1061, and 1012 cm
−1. The presence of polymorphic carbonate structures is indicated by the band at approx. 1793 cm
−1, while the sharp band at 879 cm
−1 originates from the vaterite structure. In addition to the bands described above, the tested samples contain bands characteristic of the introduced water at ~3360, 1640 cm
−1, and an increase in the background below 1000 cm
−1 is also observed. The intensity of the bands and background increases associated with the presence of water is proportional to its amount in the grease. No significant changes were found in the structure of the aforementioned bands originating from the components of sulfonate grease.
Infrared transmission spectra were recorded for lubricant samples that were degraded as a result of the thermal oxidation stability test. Due to the fact that water bands hinder the analysis of changes in the grease structure due to their intensity, the analysis of chemical changes in greases after testing was limited to the assessment of the oxidation level parameter determined by analysing the differences in the recorded spectra (
Table 2). In the case of grease containing 50% water, a very strong band in the range of 1700–1500 cm
−1 with a maximum of approx. 1640 cm
−1 made it impossible to determine this parameter.
Figure 6 shows a comparison of the FTIR spectra of greases to which water was not added: fresh, after 100,000 double strokes, and samples after undergoing thermal oxidative stability tests. The spectra show subtle differences in the absorbance of the bands at a wavenumber of approx. 1710 cm
−1 associated with the formation of carboxylic acids in typical oxidation processes. This process was most intense during the ASTM D942, where the degree of oxidation was 0.22 abs/0.1 cm
−1, while in the case of the other methods, the band of oxidation products was barely noticeable, and the degree of oxidation was approx. 0.15 abs/0.1 cm
−1. It was found that the 1793 cm
−1 band originating from polycarbonate structures remained unchanged. In addition, after the tests, changes in the spectrum structure in the range of 1590–1560 cm
−1 and approx. 1625 cm
−1 were also observed. It is not currently possible to attribute these changes to specific structural changes in the grease; thus, further research is required. Similarly, the degree of oxidation was determined for grease samples to which water was added, and the results are presented in
Table 2. It should be noted that they correlate with the previously described changes in the properties of grease with a water content of 0–40%. Even the addition of 1% water caused a significant deterioration in the stability of the greases, with the degree of oxidation increasing from approx. 30 for the grease that underwent a conventional method to over 70% for the lubricant that underwent the RSSOT-PWP test compared with the lubricant to which water was not added. After undergoing the RSSOT, RSSOT-PWP, and RSSOT-M tests, greases with a water content of 5–30% were characterised by an increase in the degree of oxidation by approx. 70–100%, which indicates their similar degradation, while in the case of greases that underwent a conventional test, a similar increase in the degree of oxidation by approx. 115–130% was observed for greases with a water content of 10–30%.
Greases with a water content of 40% have the lowest resistance to oxidation and, as a result, show the highest increases in oxidation levels, up to 150%, compared with grease that does not contain any water. The exception is the grease with a 40% water content after undergoing the RSSOT-M test, which showed an increase in oxidation of just over 70% compared with grease to which water was not added.
When assessing the differences in the degree of degradation of fresh grease and grease subjected to mechanical stress (RSSOT and RSSOT-PWP), it can be concluded that the degree of oxidation for the grease that underwent mechanical stress is higher after the test, within the range of 7–20% for all the tested samples, indicating that mechanical stress reduces the oxidation resistance of the grease.
Analysis of the spectral results obtained confirms that introducing water to lubricants impairs their thermal oxidation stability. It should be noted, however, that over a fairly wide range of water concentrations in the grease (10–30%), their susceptibility to degradation is similar. Considering all the tests used, it was found that the test acc. to ASTM D942 is the most degrading test for greases.
4. Conclusions
The incorporation of 1–50% water into calcium complex sulfonate grease causes the consistency of the grease to harden, which increases with the water content, but it does not significantly affect the working stability of the grease. The hardening of the grease’s consistency under the influence of water may result from the penetration of water molecules into the network structure of the hydrated calcium complex, where they can form hydrogen bonds with sulfonate groups, stabilising the micellar network and limiting the mobility of the base oil.
Due to the high resistance of the calcium sulfonate micellar structure to mechanical deformation, only a slight change in the consistency of greases subjected to mechanical stress is noticeable; in this case, by 100,000 double strokes.
The thermal oxidation stability of the tested calcium complex sulfonate grease decreases as the water content in the grease increases. For greases undergoing the conventional oxidation test according to ASTM D942, oxidation resistance decreases rapidly, even in the presence of a small amount of water in the grease (1%), while in those undergoing the RSSOT according to ASTM D8206, it decreases gradually. The difference in the observed rate of the decrease in oxidation stability may be mainly due to the conditions during the test. In both tests, the difference is the amount and distribution of the tested material, which means that the grease comes into contact differently with oxidising agents in such an environment. In addition, during the conventional method, according to ASTM D942, water is used, which is not the case during the RSSOT method. Water and oxygen promote the initiation of rapid oxidation, whereas in ASTM D8206, with five times less sample, uniform heating, and contact with oxygen, the oxidation process is gradual.
Studies have shown that mechanical stress reduces the thermal oxidation stability of calcium complex sulfonate greases by up to 40%. In the case of grease to which water is not added, mechanical stress causes a 20% decrease compared with unworking grease. In the case of grease containing water, even at a content of 1%, a decrease of about 10% is caused, which is within the margin of error of the method, compared with unworking grease. For grease with a water content of 5–40%, the decrease is already over 40% compared with grease that has not been subjected to mechanical stress. In the case of grease that contains 50% water, the decrease is approximately 25% compared with unworking grease.