1. Introduction
Red mud (RM) is a highly alkaline solid waste associated with the production of alumina by the Bayer or sintering process, and its output is 1–2 times that of alumina production [
1,
2]. Traditional disposal methods are limited by their strong alkalinity (pH10–13), complex composition, and high cost of neutralization, while the application in the field of building materials also faces technical bottlenecks. In recent years, the resource utilization of red mud has gradually become a research hotspot in which the mine-filling field shows significant advantages: From the geographical distribution point of view [
3], alumina plants are adjacent to mines, which can realize the short-distance transportation of red mud; from the analysis of material properties, red mud is rich in silicate, aluminate, and other active ingredients, which can play the role of cementing in the filling system, partially replacing the cement to enhance the strength of the filling body [
4]. At the same time, the iron and aluminum oxides contained in red mud can stimulate geopolymerization under alkaline conditions, forming a structure with self-hardening characteristics; from the engineering economic point of view, the annual average demand of hundreds of thousands of tons of materials for mine filling and the characteristics of the huge amount of red mud stockpile are highly compatible [
5], providing a feasible path for large-scale consumption. The research in this direction not only reduces the cost of mine cementation and filling by 15–30%, but also opens up a new way to utilize red mud in large quantities, and the relevant results have been verified in a number of bauxite mine synergistic bases [
6].
Although the application of RM in mine filling shows the potential for resource utilization, its highly alkaline characteristics still bring multiple technical challenges: On the one hand, the alkaline environment will inhibit the hydration reaction of the cementing system, leading to the lag in the early development of the strength of the filling body [
7]; on the other hand, the highly alkaline slurry is prone to corrosion of the metal pipeline and equipment, and the fine-grained red mud is prone to flocculation due to its high surface charge, leading to a slurry viscosity that is too large, and pumping strength increases significantly. In order to solve the above problems, scholars proposed to introduce an acidic solid waste synergistic regulation strategy, such as phosphogypsum (PG) [
8]. As a by-product of the phosphoric acid industry, PG is not only rich in free acid and sulfate, which can effectively neutralize the alkalinity of red mud [
9], but also its coarser particle characteristics (typical particle size of 0.1–0.5 mm) can form a complementary gradation with the red mud to optimize the microstructure of the filling body. Red mud with PG at a 1:1 mass ratio reduced the pH value of a mixed system from 12.3 to 8.5, and the synergistic effect of free Ca
2+ and SO
42− activated the dissolution of the silica–alumina phase of the red mud, which led to an increase of 40% in the generation of the hydration products of calichealumite (AFt) and C-S-H gel [
10], and the 28-day compressive strength reached 9.8 MPa, which was 2.3 times higher than that of the pure red mud system [
11].
However, PG also has the problem of solid waste utilization, and PG’s global annual production is about 280 million tons (China accounted for 75 million tons); at the same time, fluoride-containing compounds (e.g., CaF
2) [
6] are easily soluble in water, and seepage into the groundwater leads to fluorine exceeding the standard, which triggers the fluorine bone disease of humans and animals and the wilting of plants; PG contains sulfate SO
42−, with a pH of about 3–5, which pollutes the surrounding water bodies and soils. Therefore, the high-value utilization of PG is the key direction to solve its environmental problems. It is mainly applied to the fields of building materials, agriculture, and the chemical industry, such as using PG as a cement retarder; to improve acidic soil [
12], with the potential to increase crop yield and safety; and to prepare wall materials and road base materials. Applied to the filling field, Na(OH)
2, Na
2SiO
3, or Ca(OH)
2 of PG adjusts the pH value of the system and promotes the reaction between Ca
2+ in PG and silica–aluminum material to form C-(A)-S-H gel, and the acidity of PG will slow down the hydration reaction of cement [
13]. However, the “disadvantages” of PG are transformed into advantages in red mud filling, and the acidity of PG can neutralize the high alkalinity of red mud; the slow-setting property of PG can avoid premature solidification of RM slurry in the process of pumping and reduce the risk of pipeline clogging; at the same time, the coarser particles of PG are mixed with the fine particles of red mud, and the gradation is complementary so that the densification of the filling body is improved [
14].
At the same time, the slurry concentration is one of the core parameters of the filling process, which directly affects the delivery efficiency of the filling body, the strength of the filling body, and cost control. Low concentration (<65%): slurry flow state close to Newtonian fluid, pipeline strength is small but the risk of solid-phase settling is high, and it is easy to lead to pipe plugging. High concentration (>75%): viscosity rises sharply, pumping pressure needs to be increased, coarse particles (such as PG) may exacerbate pipeline wear, and has a concentration of 70% when the wear rate is higher than 60%; with a concentration increase of 5%, the pumping energy consumption increases by 10–15% but can reduce the cost of dewatering and the amount of cement, and the overall benefits need to be weighed; traditional tailings filling recommended a concentration of 65–75%, except for the fine particles of red mud, etc. The concentration is recommended to be 65–75% for traditional tailing sand filling, but fine particles, such as red mud and other solid wastes, need a lower concentration to ensure mobility due to high viscosity; the concentration should be optimized in synergy with aggregate gradation and cementitious material type to improve the red mud-PG compound system due to gradation improvement. A systematic summary of the mechanisms linking slurry consistency, additives, and filling performance is given in [
15], focusing on the latest research advances in red mud-based filling materials, including strategies for consistency optimization [
6].
Based on this, we propose using PG to alleviate the role of RM-based cemented paste backfill due to RM’s high alkalinity and high viscosity, resulting in construction difficulties and environmental risks, and PG’s acidic, slow-setting, and coarse aggregate properties that can be targeted to alleviate the above problems to determine the optimal mixing concentration interval of PG and RM, balancing the environmental protection, construction, and strength requirements, so that the mechanical properties of RCPB meet the requirements of mining [
16]; the high alkalinity of RM can be mitigated; the hydration process of RCPB can be investigated from a microscopic point of view, and the mechanism of F
− fixation can be elucidated; the fluidity of mixed slurry can be moderately high, the coagulation time can be controlled, and the pipeline conveying strength is low; the leaching of hazardous ions (F
−) can reach the national standard (<1.0 mg/L); and the optimal concentration of the PG-RM composite filling can be proposed to satisfy the practical requirements of the production. The optimal concentration ratio of the PG-RM compound filling body was proposed to meet the actual production demand. This study lays a scientific foundation for optimizing the use of PG resources and promoting the development of eco-friendly building materials while increasing the value of industrial solid waste and providing ideas for achieving the goals of a circular economy and sustainable development.
2. Materials
2.1. Raw Materials
The primary raw materials used to synthesize the CPB in this study were RM, PG, and OPC. PG was sourced from a chemical facility in Guizhou Province, China, and transported to the laboratory in airtight containers to prevent moisture exchange. The PG exhibited a pH of 3.74, confirming its strongly acidic nature. Moisture content was quantified two hours prior to experimentation to calibrate the water-to-solid ratio in the mixtures, ensuring consistency in slurry formulation.
RM was sourced from an alumina refinery in Guizhou Province, China. To eliminate residual moisture and enhance reactivity, the RM was oven-dried at 60 °C for 48 h prior to use. OPC (grade P.O 42.5) was supplied by Xiangping Co., Ltd. (Changsha, China). Toxicity Characteristic Leaching Procedure (TCLP) tests on RM confirmed that heavy metal concentrations in the leachate (e.g., Cr: 0.8 mg/L, Pb: 0.2 mg/L) were well below Chinese regulatory thresholds [
17]. The RM exhibited a pH of 11.47, establishing an alkaline environment that promotes hydration reactions critical to cementitious material formation.
X-ray fluorescence (XRF) analysis revealed that PG primarily consisted of CaO (38.72 ± 0.15 wt%) and SO3 (52.14 ± 0.20 wt%), aligning with the chemical formula of gypsum (CaSO4·2H2O). Trace components included P2O5 (1.85 ± 0.08 wt%) and F (1.34 ± 0.05 wt%), indicative of residual phosphate impurities and fluoride contamination. Cadmium (Cd) was detected at (0.012 ± 0.001 wt%), a critical parameter for environmental risk assessment.
RM was predominantly composed of Al2O3 (22.35 ± 0.15 wt%) and Fe2O3 (38.12 ± 0.20 wt%), characteristic of bauxite residue from alumina production. Elevated CaO (8.74 ± 0.10 wt%) and Na2O (6.85 ± 0.05 wt%) content corroborated RM’s strong alkalinity (pH > 10), which is central to its role in neutralizing acidic components and driving hydration reactions.
The analysis of the particle size of raw materials shows that RM has a wider particle size distribution but a more concentrated PG distribution (smaller D30/D10 ratio); RM has a finer particle size, probably due to a finer comminution process, and the PG particles are more homogeneous (D60/D10 = 11.2/2.88 ≈ 3.88 vs. RM = 5.33/0.56 ≈ 9.52). Also, the high specific surface area of RM may enhance the reactivity.
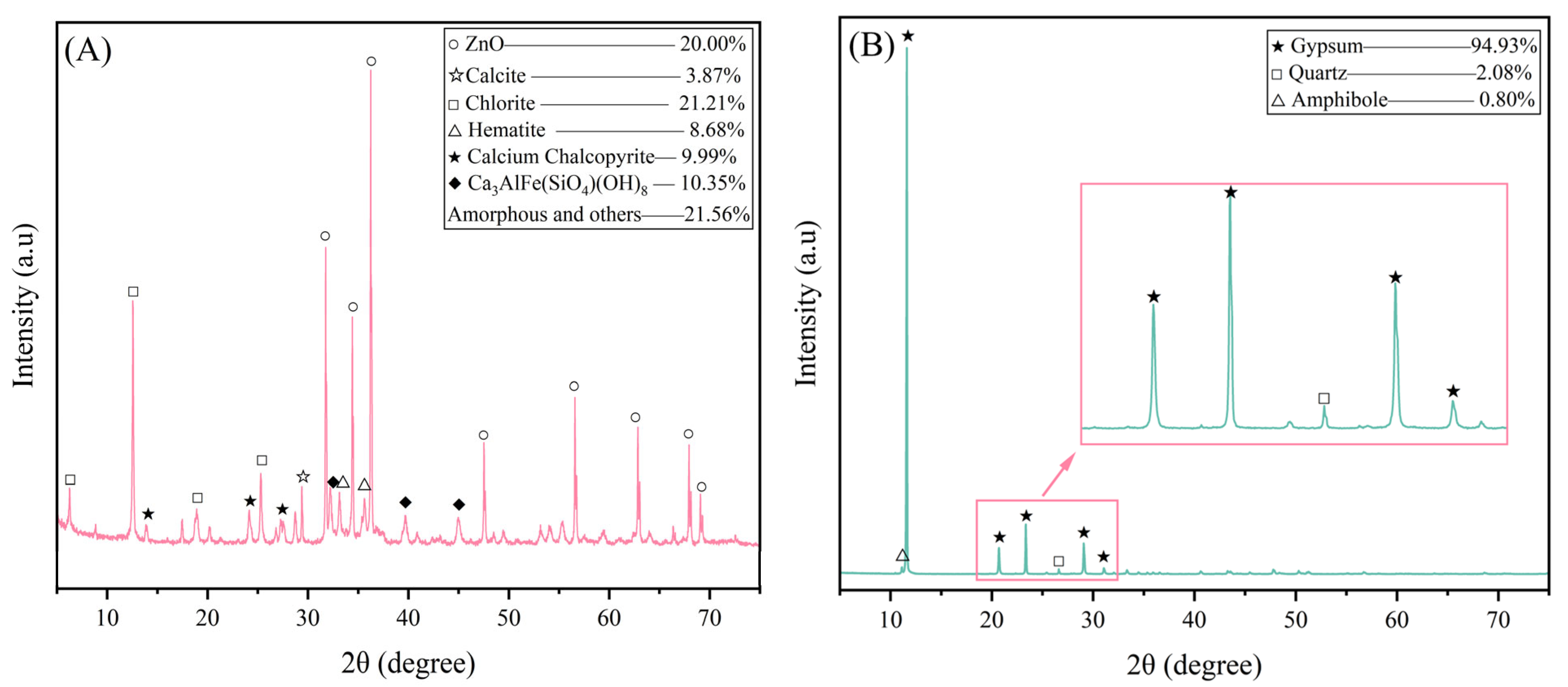
According to XRD, RM is dominated by amorphous and Chlorite, with nearly 10% Ca3AlFe(SiO4)(OH)8, Calcium Chalcopyrite, and hematite, while PG is mainly composed of gypsum.
Table 1 lists the results of the XRF analysis;
Figure 1 and
Table 2 present the particle size distribution of the raw materials, while the XRD spectrum is shown in
Figure 2:
2.2. PG-Based Cemented Paste Backfill (RCPB) Preparation
Figure 3 depicts the RCPB preparation procedure and analytical method adopted in this study. Based on the previous exploratory tests and the practical application of RCPB, and based on the preliminary ratio optimization study, the mass ratio of PG:RM = 1:3 was adopted in this experiment [
18], which was designed to take into account the following key factors: (1) ensuring that the system had sufficient initial alkalinity to maintain hydration activity; (2) maximizing the consumption of acidic components in PG through acid-base neutralization reaction; and (3) balancing the amount of solid waste admixture with the demand of mechanical properties. In order to better understand the role of slurry concentration in the RCPB system, the gravel-to-sand ratio was 1:8; the ratio of PG to RM was 1:3, which made the mixed slurry an alkaline environment and facilitated the hydration reaction. In this study, the slurry concentration ratios were controlled at 63%, 65%, 67%, and 69%. Nine specimens were prepared for each concentration level for subsequent testing and analysis, each group of samples was tested in triplicate, and the average was taken as the representative value for each specimen, with the specific mixing ratios detailed in
Table 3. The OPC, PG, and RM were dry-mixed in a mixer for 5 min in the specified ratios [
19]. Deionized water was then added to the mixture and stirred continuously for 5 min with an electric mixer [
20]. After the mixture was homogenized, the slurry was poured into a plastic cylinder (Φ 50 mm × H 100 mm), and the specimens were compacted on a vibration table at a speed of 1.3 mm/s for 5 min to remove residual air bubbles [
21]. After 3 d of hardening, the RCPB specimens were demolded and cured under constant temperature and humidity using a curing chamber (HWS-70B, Tianjin Teda Instrument Co., Ltd., Tianjin, China). The curing temperature and relative humidity were maintained at 20 ± 1 °C and 90%, respectively.
4. Results and Discussion
4.1. Mechanical Performance of RCPB
The compressive strength of RCPB generally increases with rising concentrations of cementitious materials, though further strength gains plateau and workability declines at excessively high concentrations [
31]. This study aims to identify the optimal concentration ratio.
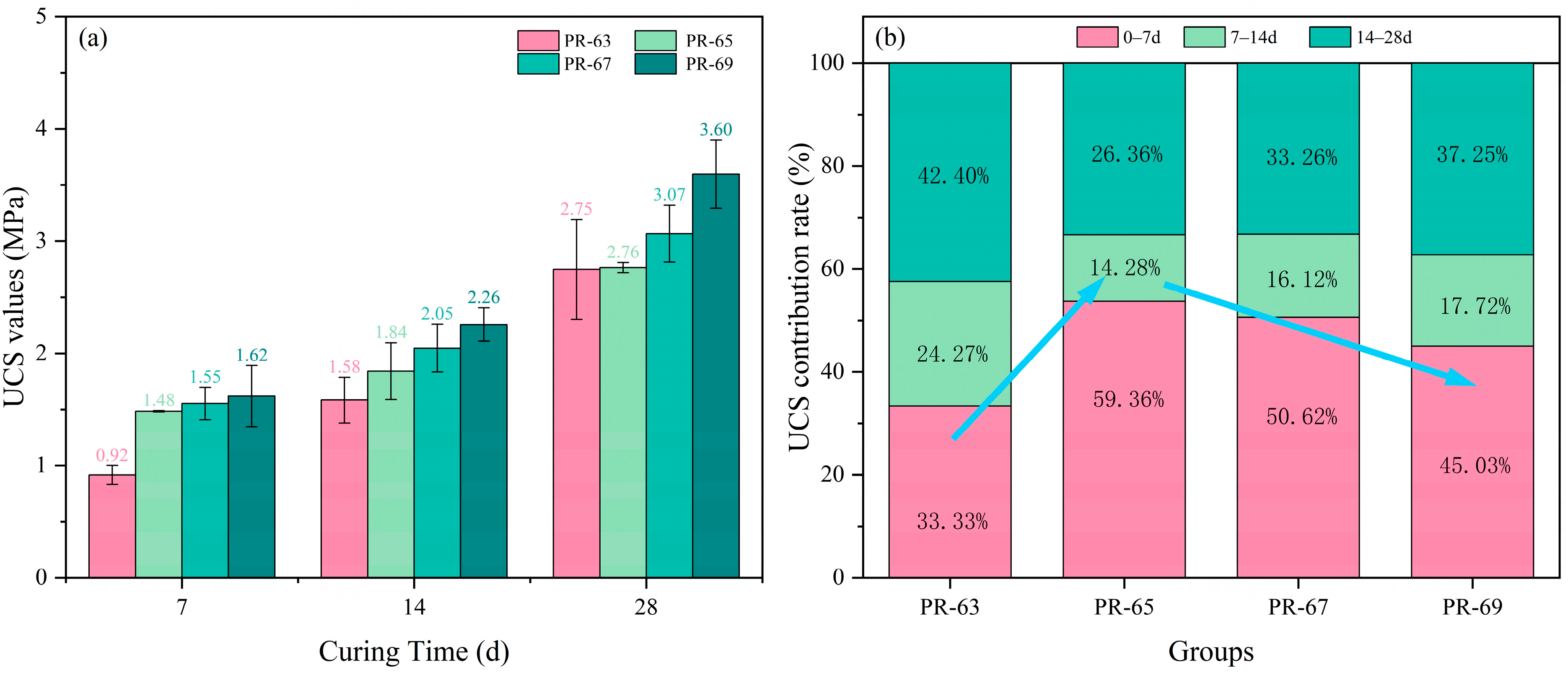
Figure 4 illustrates the evolution of UCS over curing periods (7, 14, and 28 days) and the percentage contribution to ultimate strength at each period, enabling evaluation of concentration effects on RCPB UCS.
All samples exhibited an overall increase in UCS with curing time. A clear positive correlation between concentration and strength was observed: Higher concentrations yielded proportionally greater UCS values. Notably, in the initial curing phase (7 days), specimens with concentrations ≥65% demonstrated comparable UCS ≥ 1.5 MPa, whereas the 63% concentration group showed significantly lower strength. This discrepancy is hypothesized to arise from insufficient cement and gypsum content in the 63% formulation (due to lower polymer-to-cement (PG) ratios), leading to delayed hydration kinetics and suboptimal early-stage reaction conditions.
At the lowest concentration (63%), early-age hydration contributed minimally to strength (only ≈30%), while later-stage reactions (14–28 days) accounted for a significantly higher proportion of strength development. In contrast, the 65% concentration specimens exhibited the highest early-stage strength contribution (59.36%) among the tested groups. This is hypothesized to arise from the formation of AFt in higher-concentration samples, which enhances early strength but leads to late-stage deterioration due to expansive AFt crystallization. At concentrations above 65%, further strength gains plateau, as the initial strength advantage from AFt formation is offset by its deleterious long-term effects.
It is hypothesized that the reduced rate of strength gain in higher-concentration samples stems from the increased presence of Ettringite, which limited mechanical property improvements despite a general trend of strength enhancement with concentration. Notably, samples with 63% Ettringite during later curing stages exhibited less pronounced strength increases than anticipated. Excessively high concentrations were also associated with practical challenges. For instance, at 69% concentration, the slurry’s high viscosity—attributable to RM—impaired fluidity, causing molding defects (e.g., bubble formation) and processing difficulties. Low fluidity further introduces operational risks in real-world applications, including transportation inefficiencies, inadequate densification, construction complexities, and pipeline blockages. Consequently, concentrations exceeding 69% were not evaluated in this study. After 28 days of curing, all samples demonstrated favorable long-term mechanical performance, achieving compressive strengths of 2.50–3.60 MPa. These results suggest the potential for partial substitution of conventional concrete, provided further toxicity leaching tests confirm environmental safety.
As the core active component of OPC, constituting 50–70% of its mass, tricalcium silicate (C
3S) governs the early-stage hydration kinetics of cementitious systems [
32]. The inherent lattice defects in C
3S crystals create preferential pathways for water infiltration, facilitating interfacial contact between the mineral surface and the aqueous phase. This initiates hydrolysis, characterized by continuous Ca
2+ dissolution and subsequent formation of silica-enriched regions on the particle surface. The resulting Ca-depleted silicate layer exhibits distinct compositional heterogeneity compared to later-stage hydration products like calcium silicate hydrate (C-S-H) gels. Its topochemical transformation follows the reaction pathway described in Equation (2), where surface restructuring drives the formation of a bilayer electrochemical interface:
Inner Stern layer: composed of specifically adsorbed Ca2+ and OH− ions, forming a rigid charge-dense region.
Outer diffuse layer: governed by electrostatic interactions, where counterion distribution follows the Gouy–Chapman model.
The negatively charged surface established during initial hydration critically modulates cation migration (e.g., Na+, K+, etc.), thereby regulating dissolution–reprecipitation equilibria and early-age reaction kinetics.
4.2. Analysis of the Hydrological Environment
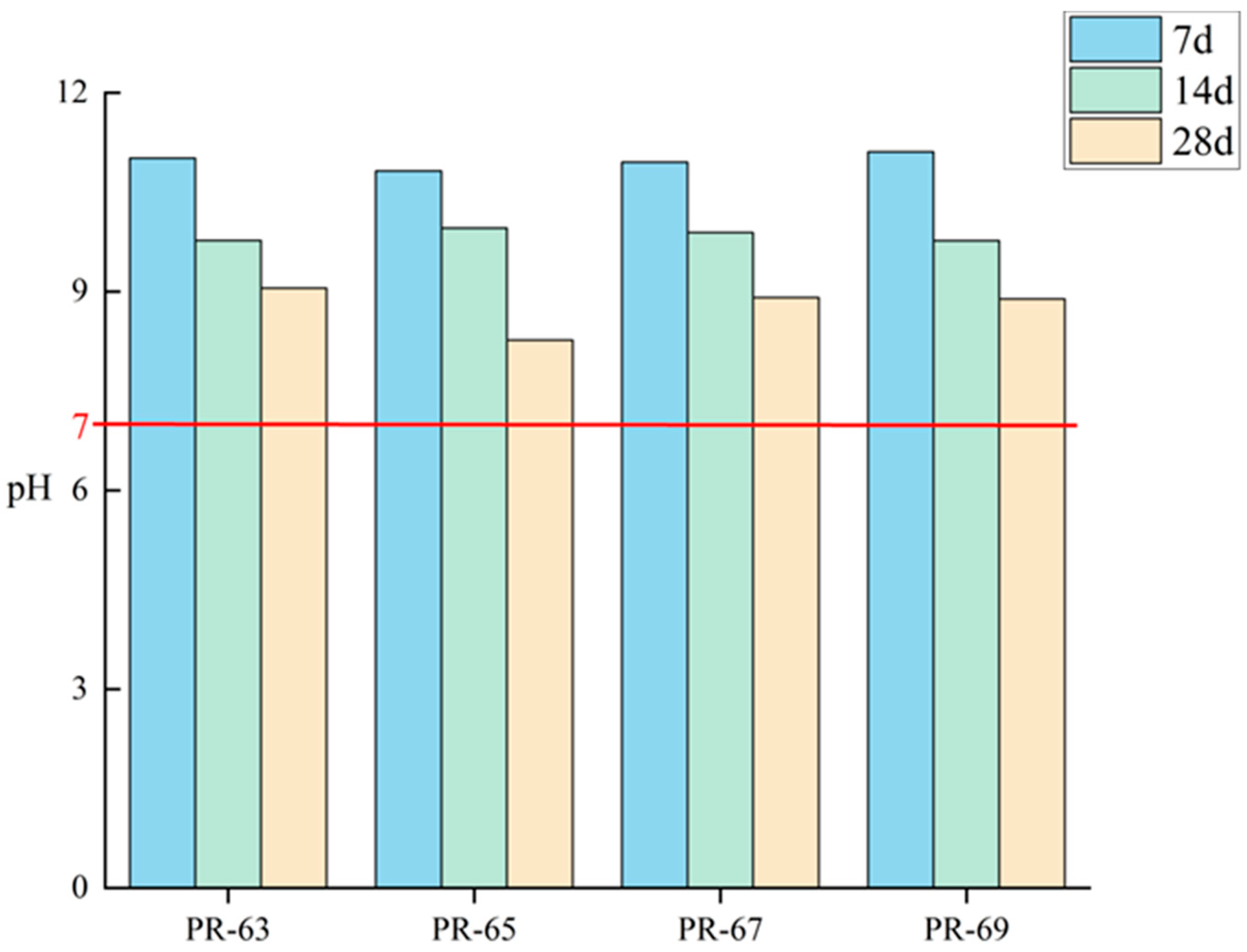
In order to evaluate the environmental compatibility of the composite cementitious material system, this study systematically monitored the pH evolution of the samples at different maintenance ages (as shown in
Figure 5), aiming to investigate the comprehensive influence of its acid–base equilibrium mechanism on the hydrological process, mechanical properties, and environmental safety so as to ensure that the pH of the samples is suitable for the hydrological reaction.
The test results showed that the pH value of the samples was maintained in the range of 9–12 at the early stage of conservation (0–7d), and this alkaline environment mainly originated from the dissolution of alkaline components such as Na
2O in RM, which effectively neutralized the soluble acids (e.g., residual phosphoric acid) contained in PG. The suitable alkaline conditions at this stage promote the rapid dissolution of cement clinker minerals, creating favorable conditions for the formation of hydration products such as C-S-H gel [
33]. By the 14d maintenance age, the pH value showed a significant decreasing trend (about 15–20%), which was closely related to the continuous consumption of OH
− ions by the hydration reaction: On the one hand, the continuous hydration of minerals such as C
3S produced Ca
2+ with OH
−, and on the other hand, the generated C-S-H gel fixed some of the OH
− by the chemical binding immobilizing some of the OH
−, leading to a decrease in the alkalinity of the solution phase [
34].
Notably, the pH of the system at 28 days stabilized at 9.0 ± 0.3, significantly lower than the original cement system (pH ≈ 12.5) but higher than the targeted neutral range (pH = 7). This deviation from neutrality likely arises from three interrelated mechanisms: (1) alkaline buffering in the RM-PG composite system: Hydroxyl ions (OH−) are continuously replenished by the gradual release of alkalis from RM; (2) kinetic limitations in later hydration stages: Slower reaction rates fail to fully deplete free alkalis in the system; (3) solubility constraints of Ca(OH)2: Reduced water-to-cement ratios limit calcium hydroxide dissolution. While the moderately alkaline pH (vs. the strongly alkaline leachate of traditional cement) may mitigate environmental risks, the long-term leaching behavior and its ecological implications require further assessment.
Three hydration stages were derived from the hydration process: initial passivation: The trapping of calcium ions by the bilayer leads to a limitation of the mineral dissolution rate and the formation of a short reaction lag. The presence of a surface passivation film at this stage is a key factor in inhibiting sustained hydration. Mid-phase acceleration: The accumulation of OH− concentration in the system triggers supersaturation of the solution, which leads to the formation of Ca(OH)2 nuclei. This phase transition process breaks the original solvation equilibrium, forming an autocatalytic reaction mechanism and entering the “accelerated period”; the hydration rate is obviously accelerated but also reflected in the pH value of the sample in the middle period, a pH value of 9–12, and the reaction rate is accelerated. Later decay: When the degree of hydration exceeds 60%, three synergistic factors lead to a decrease in the reaction rate: Firstly, the C-S-H gel densifies to form a diffusion barrier; secondly, the calcium ion concentration gradient of the pore solution decreases; and finally, the interfacial pH falls back to 9 from 11.
At the same time, RM and PG react synergistically: acid–base neutralization, which makes the final pH of the system alkaline, and the formation of the Na2SO4 electrolyte from RM and SO4 from PG, which leads to an intensification of the reaction in the accelerated phase.
XRD and SEM characterization (see
Section 4.3 for details) confirmed the formation of a dense C-S-H gel network structure at this ratio, which had a significant correlation with the phase change pattern of the system pH. In this study, the pH value of the system was stabilized at 9.0 ± 0.3, which could not only immobilize most of the heavy metals by alkaline precipitation but also realize the secondary curing by using the ion-exchange ability of the C-S-H gel.
4.3. Hydration Product Analysis of RCPB
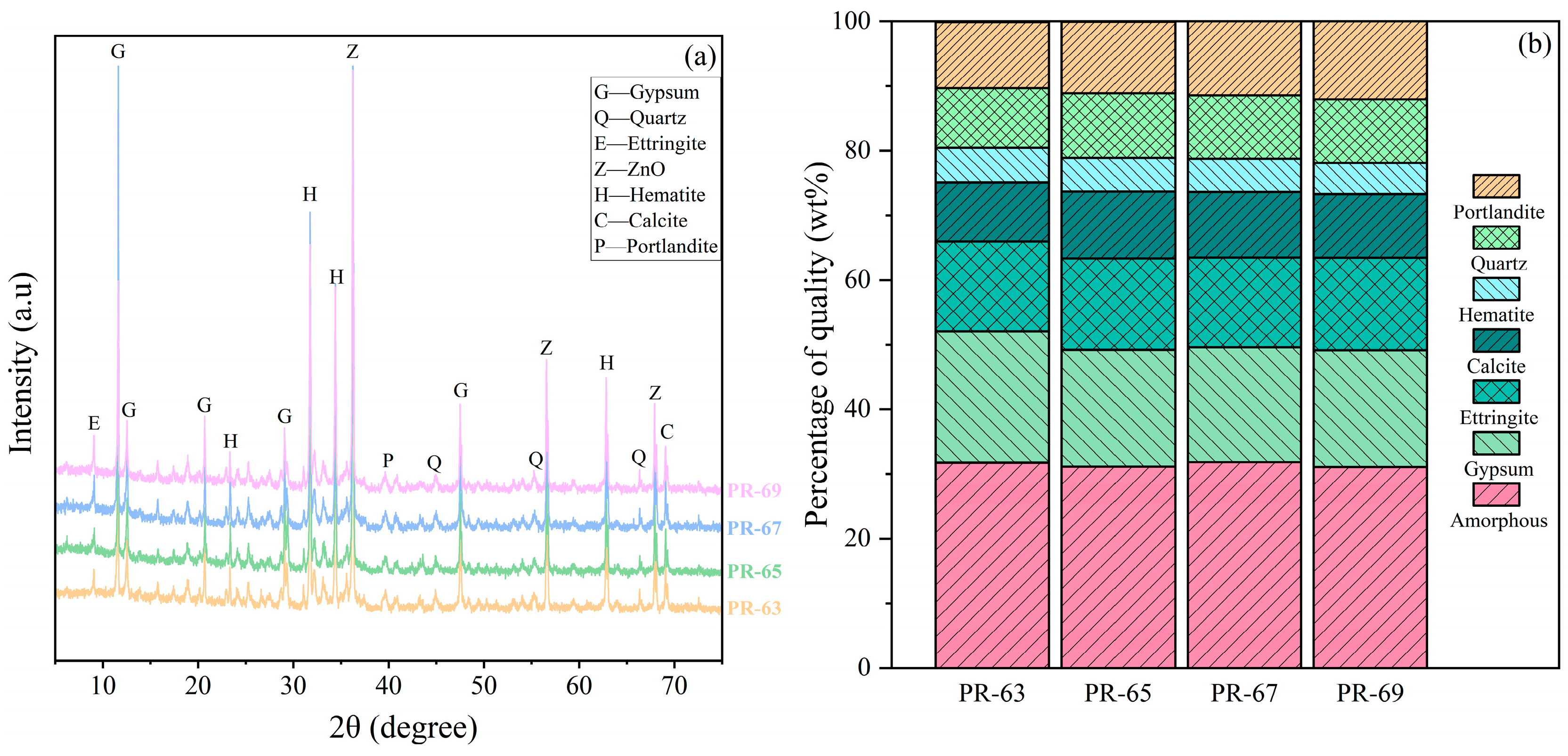
4.3.1. XRD
Figure 6 shows the X-ray diffraction (XRD) patterns of RCPB samples cured for 28 days and their quantitative analysis results. The positions and types of diffraction peaks are basically the same for samples of different concentrations, with only slight differences in peak intensities, and the percentage of amorphous is higher than 30%. The XRD results show that the samples mainly contain six typical mineral phases, which are Portlandite, Quartz, hematite, calcite, Ettringite, and gypsum. Among them, the diffraction peak intensity of gypsum is the highest except for amorphous, and gypsum is the main component of PG, indicating that it is more abundant in the sample. The second is Ettringite, but the high Ettringite is presumed to be probably due to the participation of PG in the reaction in the early stage, and the presence of Ettringite makes the RCPB have high strength in the early stage of maintenance, but the strength enhancement is not obvious in the late stage of maintenance due to the expansion of Ettringite, which is in agreement with the results of the UCS test. The presence of CaCO
3 is mainly attributed to Portlandite carbonation, while some of the mica undergoes a carbonation reaction with carbon dioxide during the curing process [
35].
The presence of hydroxide imparts not only base-activated properties to the RM, but the hematite also provides the skeletal structure for the formation of hydration products. The main components of the cementitious materials in the samples are silicate minerals (C
3S, C
3S) and aluminate minerals (C
3A, C
3AF). When the cement comes in contact with water, these minerals react chemically with water to produce gels and other hydration products [
36]. The reaction formula is given below:
The amorphous calcium silicate hydrate (C-S-H) gel, as the primary hydration product, dynamically evolves in calcium–silicate polymeric composition during cement hydration. It synergizes with calcium aluminate silicate hydrate (C-A-S-H) gel through covalent bonds, hydrogen bonds, and ionic interactions to form a three-dimensional network. This dual-phase system enhances concrete performance through complementary mechanisms: C-S-H gel primarily governs mechanical strength development while C-A-S-H gel improves structural stability, with their combined action reducing capillary porosity through progressive pore-filling.
In aluminate-rich cement systems, the coexistence of these gels creates an interconnected microstructure that enhances concrete’s mechanical properties (strength and toughness) and durability. Hydration advancement promotes gel cross-linking, which densifies the matrix by occupying interparticle voids and minimizing capillary pores. XRD patterns between 30 and 45° reveal diffraction peaks from both residual amorphous components in raw materials and newly formed amorphous hydration products. Increased peak intensity correlates with elevated gel production, directly contributing to improved mechanical strength and material performance.
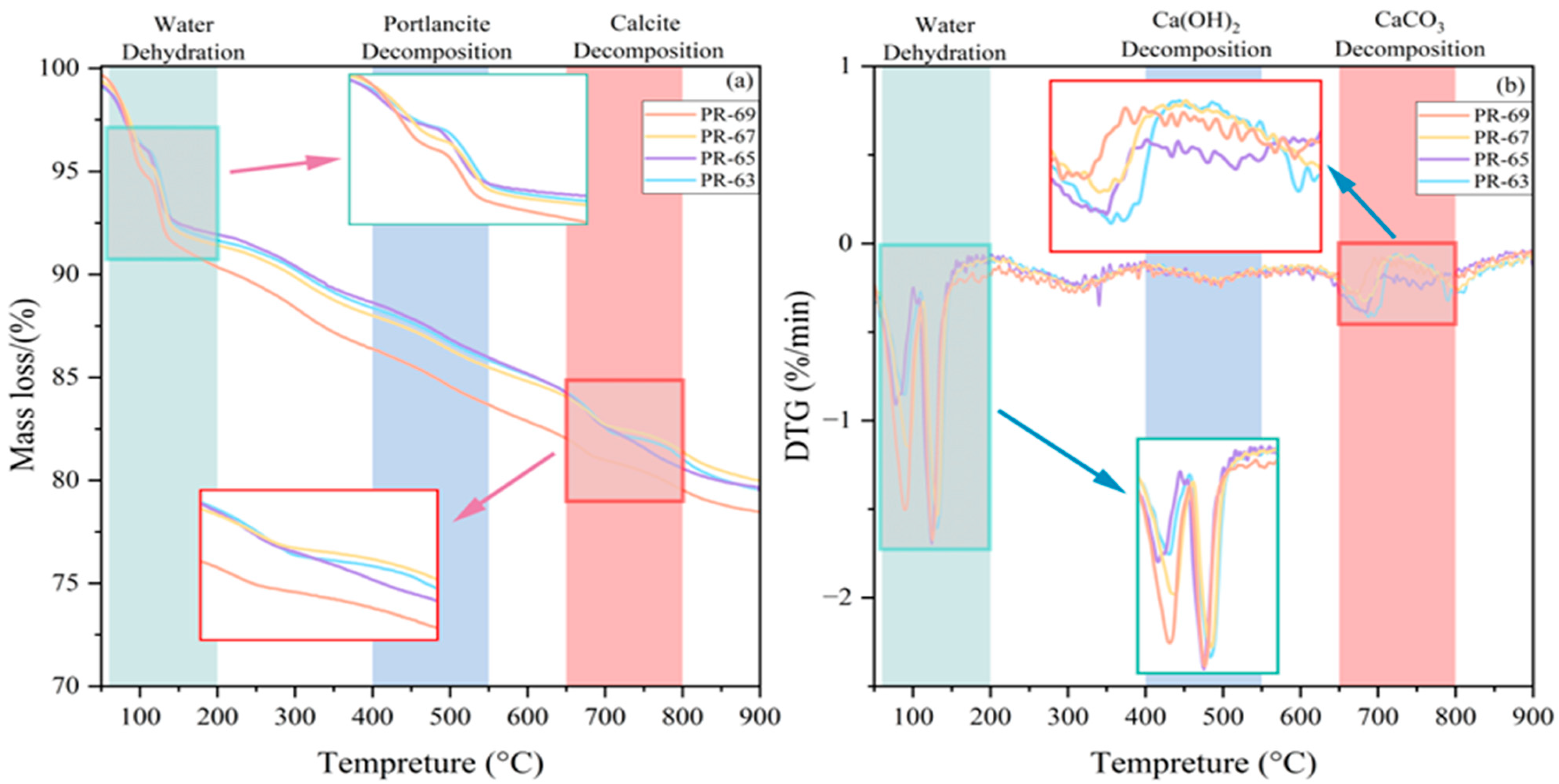
4.3.2. TG and DTG
Since C-S-H and C-A-S-H gels have low crystallinity and amorphous structures, it is challenging to detect these gels by XRD, so TG analysis was added to further detect the amount of C-S-H and C-A-S-H generated, and TG and DTG analysis was used to further determine the relative amount of amorphous phases in the filler. The TG and DTG analysis was performed to determine the differences in the substances formed in different concentrations. Two different weight loss scenarios can be most easily recognized as negative peaks in the DTG curves, with the intervals of highest weight loss being 60–200 °C and 650–800 °C [
37].
From the TG and DTG analysis of the tested samples (
Figure 7), the weight loss curves of all the samples showed similar profiles, which were mainly categorized into three weight loss temperature intervals:
(1) Within the temperature interval of 60 °C to 200 °C the material exhibited significant mass loss peaks, with calculated weight losses of 7.97 wt%, 7.37 wt%, 8.24 wt%, and 9.20 wt% for the respective samples, primarily attributed to the evaporation of free and bound water in hydration products [
38]. Specifically, gypsum dihydrate (CaSO
4·2H
2O) undergoes weight loss between 70 and 130 °C as it transforms into hemihydrate gypsum (CaSO
4·0.5H
2O), while the latter loses structural moisture at 190–230 °C. The weight loss of C-S-H and C-A-S-H gels occurs in the range of 105–200 °C, slightly higher than that of gypsum dihydrate but lower than hemihydrate gypsum, corresponding to the release of bound and free water in these gels. Concurrently, AFt demonstrates weight loss between 110 and 150 °C due to the escape of crystalline water. Notably, higher concentrations correlate with greater mass loss, exemplified by the 69% concentration sample showing a 9.3% mass loss. This is hypothesized to result from increased AFt content in high-concentration hydration products, aligning with prior observations from UCS tests where reduced late-stage strength enhancement in high-concentration specimens implied a dominant role of hydration product composition in overall mechanical performance, thereby validating the interplay between hydration chemistry and material strength.
(2) CH decomposes to CaO between 400 °C and 550 °C, and the following reactions usually occur:
In
Figure 7, it can be seen that the DTG graphs of the four groups of materials almost overlap, and all four groups lost about 3% of their mass, which proves that the concentration is less for CH content and proves that the degree of hydration is high.
(3) In the range of 650 °C to 800 °C, which is the region of carbonate decarbonization, there is a significant fluctuation in the DTG images of the materials [
39], with the TG curves showing a mass loss of about 3.27 wt%, 3.69 wt%, 2.67 wt%, and 2.52 wt%. This is attributed to the high-temperature decomposition of calcite:
4.3.3. SEM and EDS
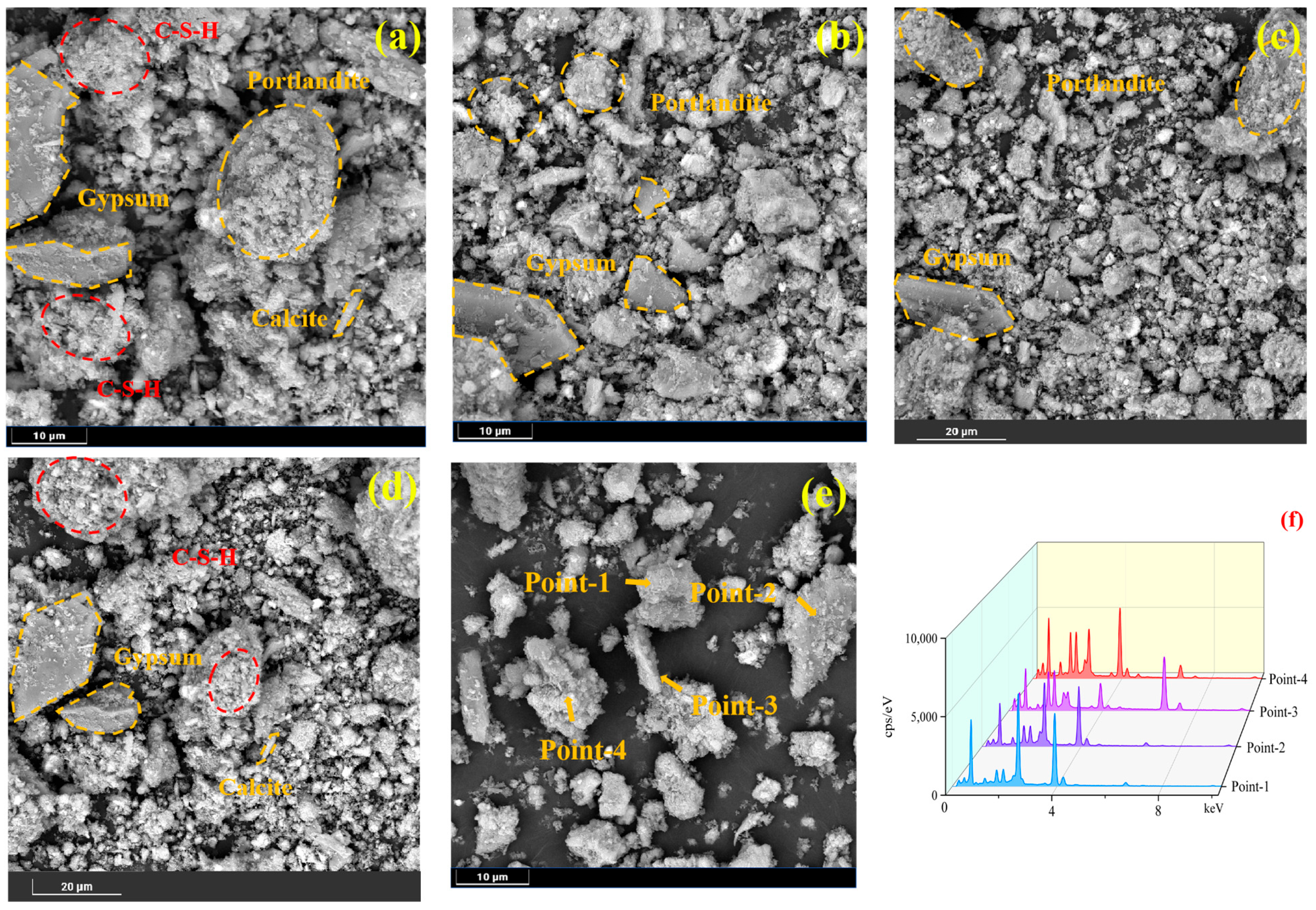
In order to further explore the composition in the samples, as well as the endowment morphology, characterize the typical morphology and quantity of hydration products, as well as the pore structure and distribution of the four groups of samples maintained for 28d [
40], SEM detection BES imaging, as well as the elemental composition of the specific points, were analyzed with energy spectroscopy analysis EDS detection; this study systematically revealed the solid waste doping on the formation of the microstructure of the RCPB system and the efficacy of the pollutant sequestration M = nechanism of action. SEM images magnified to 10 μm (
Figure 8) are shown for four samples, showing a large number of flocculent (C-S-H) gels or (C-A-S-H); the residual strong alkaline components in the RM elevate the initial alkalinity of the specimens, leading to a shortened induction period; Fe ions in the RM intensify the reaction in the accelerated period of the C-S-H gel; meanwhile, Al ions displace C-S-H in the bridging silicon sites, enhancing the macroscopic compressive strength. Lumpy (calcium hydroxide and gypsum): the reaction between soluble SO
4 in PG and the hydration product of C
3S, which reduces the concentration of Ca ions and prolongs the induction period; the formation of insoluble Ca
5(PO
4)
3OH by PO
4 impurities and Ca affects the hydration; gypsum crystallization leads to prolongation of the accelerated period and postponement of the onset of the decay period [
41]. Stick (AFt) as well as flocculent structures (complexes as well as unreacted material), with specific elemental compositions for each structure, were detected by EDS.
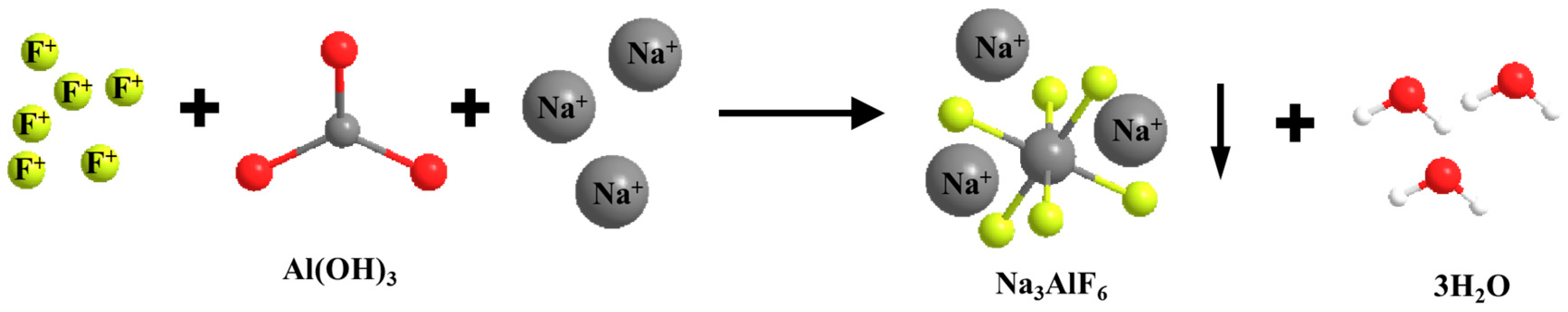
The flocculent gels (C-S-H or C-A-S-H), with EDS face scans showing Si/Ca molar ratios ranging from 0.45 to 0.62, are consistent with the structural features of low Ca-Si ratio C-S-H; the rod-like AFt crystals, with lengths of 1–3 μm, with EDS showing S/Al molar ratios of ≈3.0, corroborate the stoichiometric relationships of the sulfoaluminate phases. Residual gypsum phase: Flaky gypsum dihydrate (S/Ca = 1.02) showed disordered accumulation in the low-doped specimens, and its relative content was positively correlated with the PG/RM ratio, indicating that the RM basicity was not sufficient to completely neutralize the PG acidic component. Sodium hexafluoroaluminate precipitation: Na/Al/F≈3:1:6 was detected, confirming the formation of Na3AlF6 crystals (cryolite structure) with a specific surface area of 152 m2/g, which immobilized F by physisorption.
The SEM images clearly depicted the different microstructural features of each group of samples. The SEM of each group of samples had unreacted gypsum, indicating that the addition of RM was not sufficient to neutralize the impurities such as the phosphoric acid remaining in the original PG, and thus, there were some flocculated layers of gypsum mis-stacked. According to the EDS analysis, part of the flocculent and powdery structure should be sodium hexafluoroaluminate precipitation, and the precipitation is physically adsorbed with F ions at the same time.
Combined with XRD and thermogravimetric analysis results, it can be seen that increasing the concentration of RCPB makes the reaction intensify, resulting in more gypsum reaction consumption, and, at the same time, makes the F ions adsorbed and removed, and combined with the results of the pH test, the higher concentration is more friendly to environmental protection.
4.4. Fluoride Leaching Characteristic
As shown in
Table 4, the fluoride concentrations in RCPB sample leachates exhibited significant reductions compared to raw phosphogypsum (PG) containing 167.5 mg/L F
−. Experimental results revealed that all four tested groups achieved 69–96% fluoride removal efficiency, with the highest residual concentration measuring only 0.6686 mg/L. Notably, all treated samples complied with the Class I groundwater quality standard [
40] requiring fluoride levels ≤ 1 mg/L [
42].
There were quite a few Ca ions present in all of the samples in this study, so it was inferred that the decrease in the F ion concentration could be caused by the CaF
2 solid produced by the reaction between Ca and F. The CaF
2 solid was produced by the reaction between Ca and F, and the CaF
2 solid was produced by the reaction between Ca and F [
43]. However, although Ca ions can immobilize some F ions, the chemical precipitation reaction between Ca ions and F is not sufficient, and it is difficult to make the F ion concentration to such a low degree (the F content of all groups is under 1 mg/L) [
44], and the generation of CaF
2 can be inferred to a certain extent through the determination of the Ca ion fugacity; the specific amount of Ca ions retained needs to be further detected by microscopic means, such as XRD, SEM, etc., so presumably, there are other factors; there may be other ions involved in the reaction and precipitation of F ions, and F may react with Al, Fe, Mg, and other cations to form complex precipitation, thus fixing the F ions, for example, a F and Na and Al(OH)
3 reaction to generate sodium hexafluoroaluminate flocculent precipitation and physical adsorption of F ions so as to achieve the effect of removal of F; the chemical reaction equation is given by. The change in the molecule in the chemical reaction formula is shown in
Figure 9.
A similar reaction is found in Fe ions, FeF3 precipitation generated by the iron salt precipitation method or phosphorus salt precipitation method (the raw materials selected for this study contain P element), where F reacts with Ca(H2PO4)2 to form CaF2 precipitation.
Therefore, the F− removal mechanism is two parts: (1) chemical precipitation mechanism: The free Ca2+ in the system combines with F− through ionic bonding to form insoluble CaF2 precipitation, realizing the chemical immobilization of fluoride ions; (2) physical adsorption mechanism: Amorphous alumino–silicate gels generated by the hydration reaction and secondary minerals such as chalcocite, whose surfaces are rich in hydroxyl functional groups, can be combined with F− through the role of ligand exchange, and at the same time, the developed porous structure can produce a parcel immobilization effect on F− in solution.
5. Conclusions
In this study, PG was synergistically cured by RM and OPC to form a class of RCPB with excellent mechanical properties and environmental safety, and the slurry concentration was optimized to balance the mechanical properties, fluorine fixation efficacy, and environmental safety of the materials. The experiments showed the following:
(1) This study establishes that slurry concentration critically influences material performance, with optimal outcomes observed at elevated concentrations. The synergistic interaction between RM and PG underpins this enhancement, as RM’s alkaline components neutralize PG’s acidity and mitigate fluoride ion migration. This is achieved through accelerated hydration kinetics and the development of a robust microstructure anchored by C-S-H gel and AFt formation.
(2) Microstructural analysis reveals that higher slurry concentrations promote densification and reduced porosity, directly correlating with improved mechanical properties. However, excessive AFt generation introduces microstructural stresses, which may temper long-term strength progression. These findings underscore the importance of balancing hydration kinetics and crystalline phase formation in material design.
(3) The immobilization of fluoride ions is governed by a dual mechanism: physical encapsulation within the stabilized matrix and chemical interactions via precipitation and ion exchange processes. This combined approach ensures effective containment of fluoride, aligning with sustainable strategies for managing industrial by-products.
Chemical precipitation: Free Ca2+ reacts with F− to produce insoluble CaF2 precipitate, which reduces fluoride ion mobility.
Physically encapsulated hydration products (C-S-H gel and AFt) immobilize fluoride ions through surface hydroxyl adsorption and pore encapsulation.
In addition, the alkaline component (Na2O) of RM not only effectively neutralizes the acidic environment of PG but also promotes the formation of sodium Na3AlF6 precipitation, and Na+ reacts with Al3+ and F− in PG to produce sodium Na3AlF6 precipitation, and its high specific surface area (152 m2/g) strengthens the long-term stability of fluorine through physical adsorption, which significantly improves fluorine’s long-term stability.
This study not only provides theoretical support for the efficient resource utilization of PG but also provides innovative ideas for the co-disposal of industrial solid waste and the development of environmentally friendly building materials. Future research needs to further verify the long-term durability of the material and its adaptability in complex environments (e.g., freeze–thaw cycles, acidic precipitation, etc.) in order to promote its practical engineering applications. By optimizing the proportion and process parameters, this technology is expected to be popularized on a large scale in the fields of mine filling and foundation reinforcement, which will help realize the goals of circular economy and sustainable development.